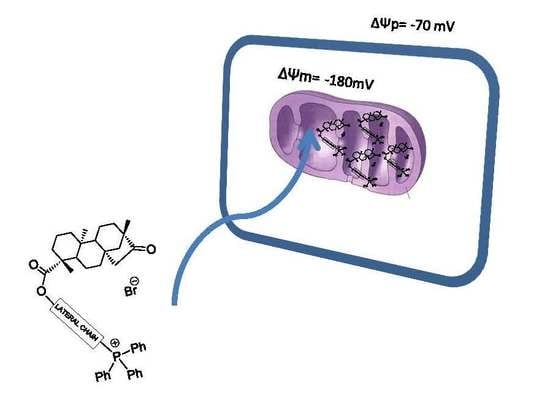
Mitochondriotropic and Cardioprotective Effects of Triphenylphosphonium-Conjugated Derivatives of the Diterpenoid Isosteviol
Abstract
:1. Introduction
2. Results
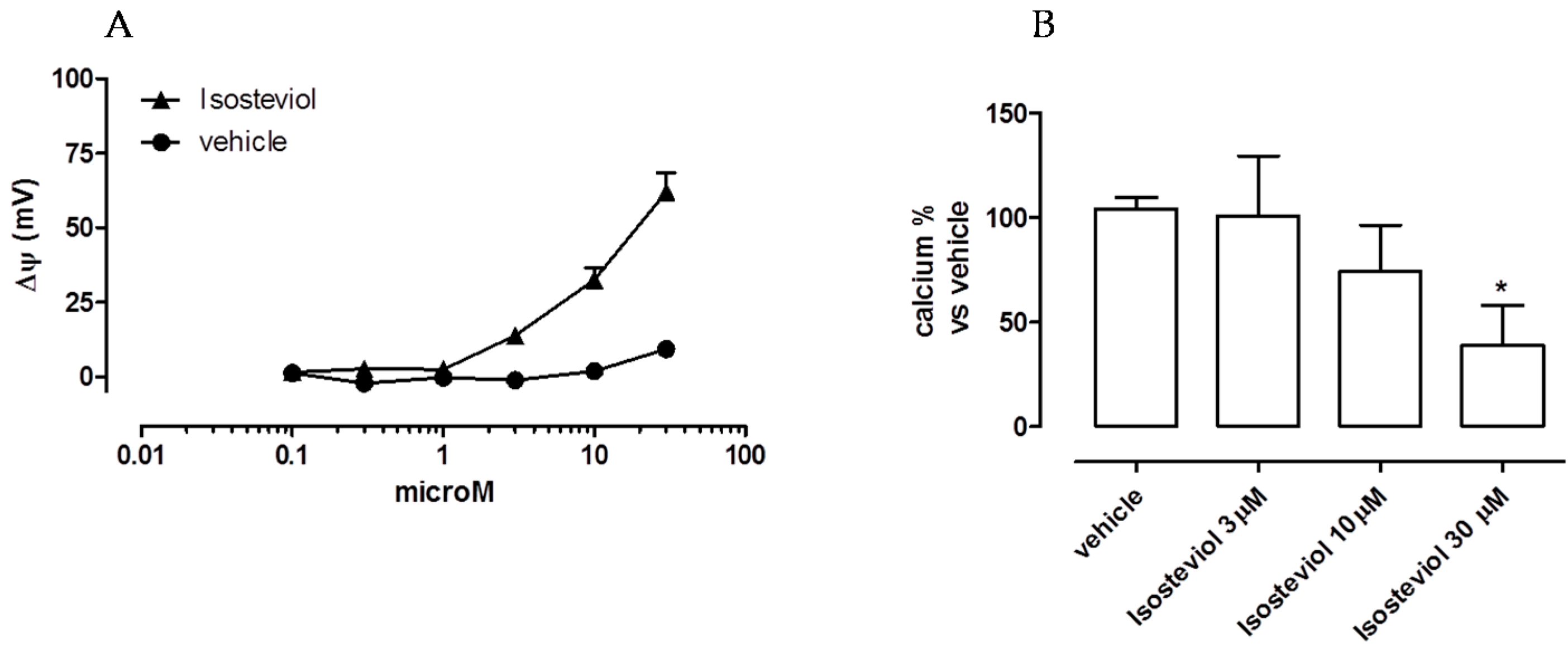
2.1. Mitochondriotropic Effects of Isosteviol
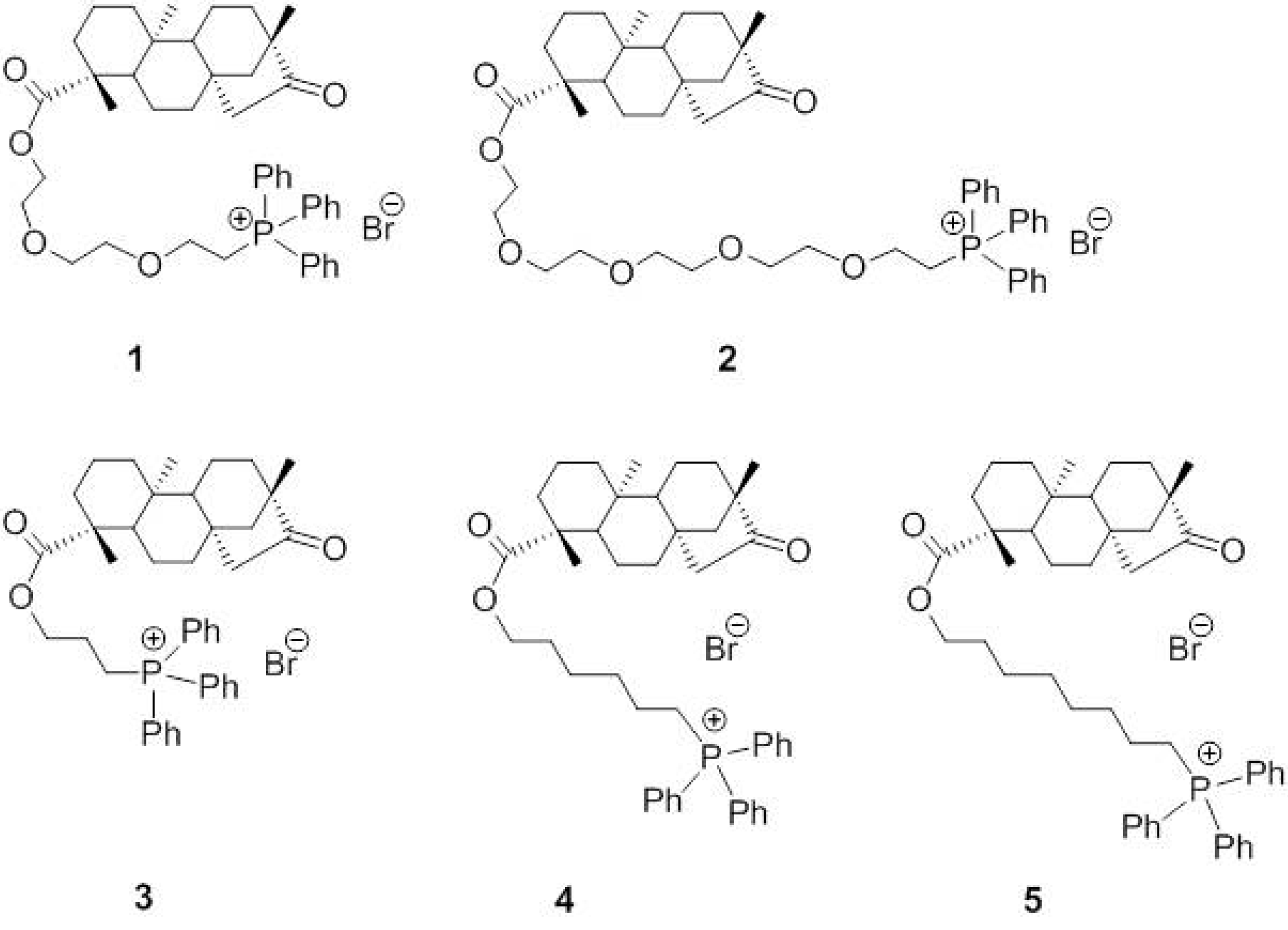
2.2. Mitochondriotropic Effects of Tripp-Conjugated Isosteviol Derivatives
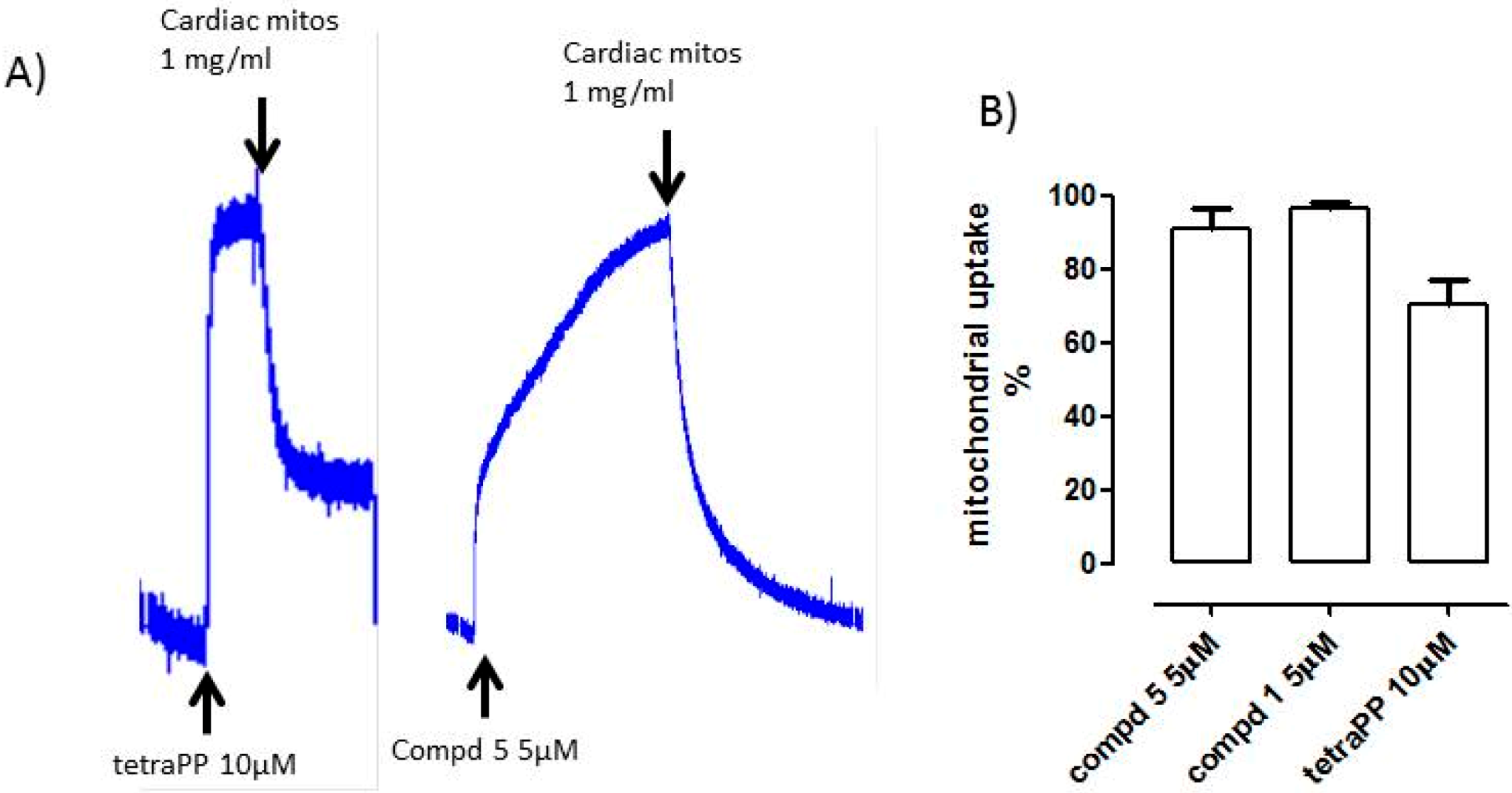
2.3. Uptake of Compounds 1 and 5 into the Mitochondrial Matrix
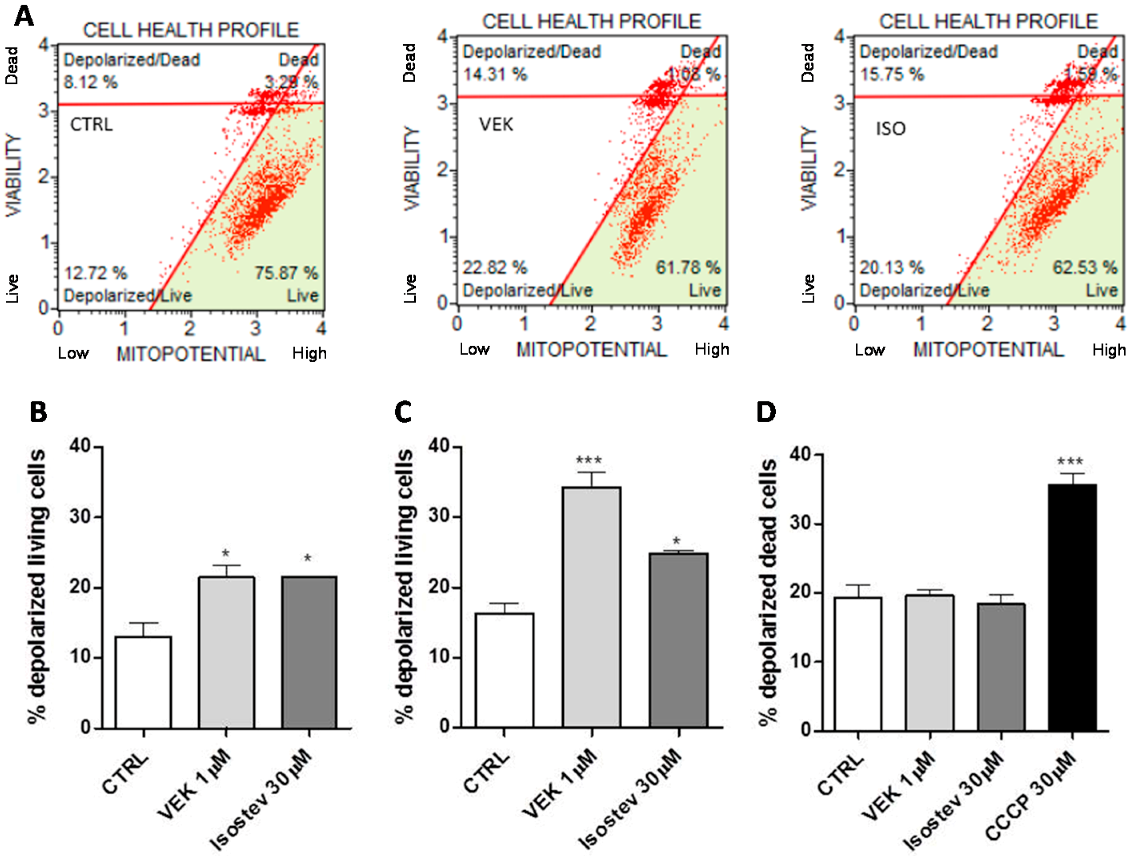
2.4. Effects of Isosteviol and Compound 5 Treatment on the Δψ of H9c2 Cells
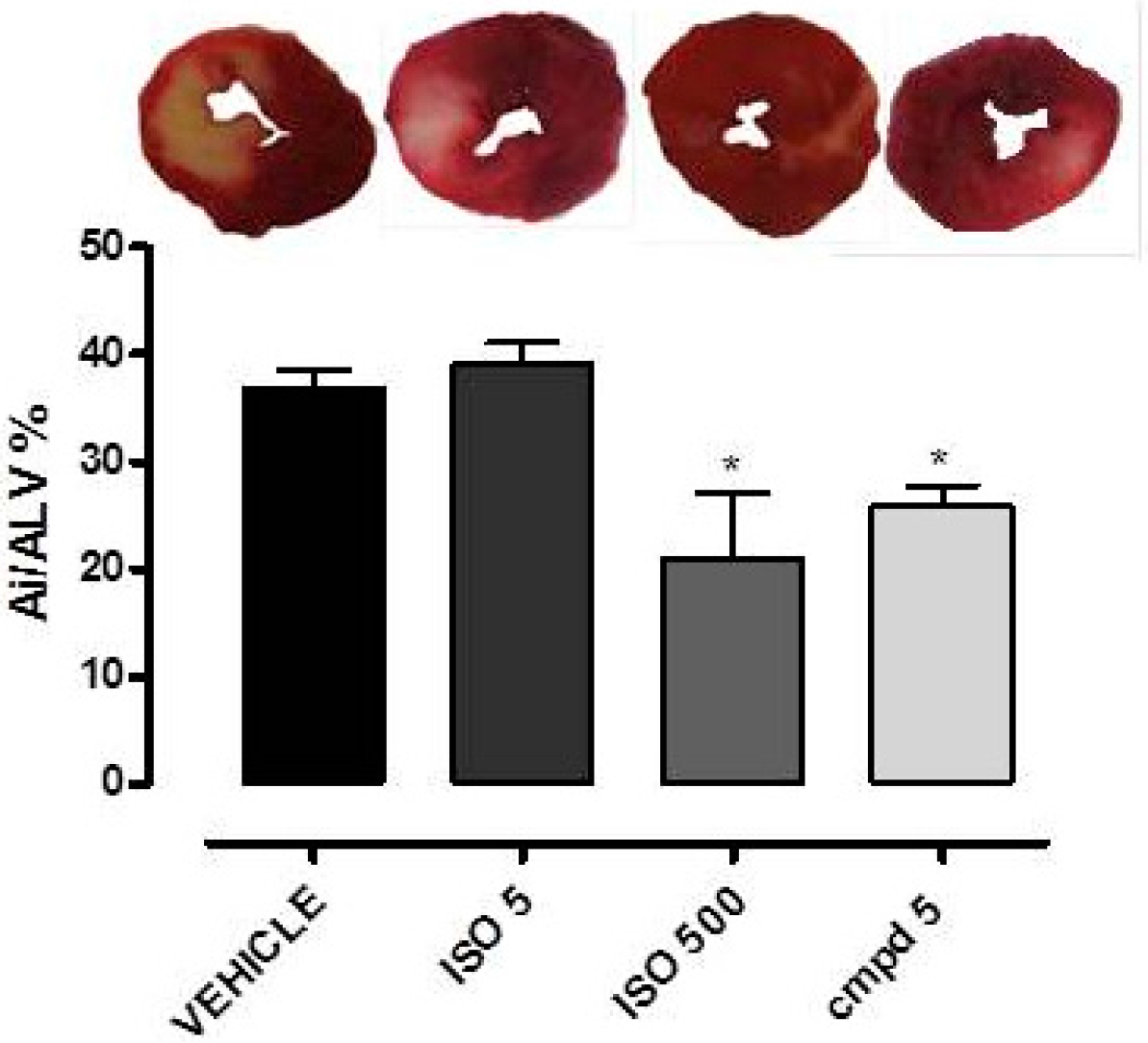
2.5. Cardioprotective Effects Observed with Isosteviol and Compound 5
3. Discussion
4. Materials and Methods
4.1. Substances
4.2. Experimental Procedures
4.2.1. Isolation Procedure of Mitochondria
Measurements of the Effects of Isosteviol on Mitochondrial Ca++ Uptake
Measurement of Isosteviol Effects on Mitochondrial Membrane Potential
Measurement of triPP-Conjugated Isosteviol Derivative Entry into Mitochondrial Matrix
4.2.2. H9c2 Cell Culture
Analysis of ΔΨ Changes on Intact H9c2 Cells
4.2.3. In Vivo Model of Acute Infarct of Myocardium
4.3. Data Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Testai, L.; Rapposelli, S.; Martelli, A.; Breschi, M.C.; Calderone, V. Mitochondrial potassium channels as pharmacological target for cardioprotective drugs. Med. Res. Rev. 2015, 35, 520–553. [Google Scholar] [CrossRef] [PubMed]
- Testai, L.; Barrese, V.; Soldovieri, M.V.; Ambrosino, P.; Martelli, A.; Vinciguerra, I.; Miceli, F.; Greenwood, I.A.; Curtis, M.J.; Breschi, M.C.; et al. Expression and function of Kv7.4 channels in rat cardiac mitochondria: Possible targets for cardioprotection. Cardiovasc. Res. 2016, 110, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Kinghorn, A. Stevia: The genus Stevia. In Medicinal and Aromatic Plants—Industrial Profiles; Hardman, R., Ed.; Taylor and Francis: London, UK, 2002; Volume 19, p. 195. [Google Scholar]
- Hsu, Y.-H.; Liu, J.-C.; Kao, P.-F.; Lee, C.N.; Chen, Y.J.; Hsieh, M.H.; Chan, P. Antihypertensive effect of stevioside in different strains of hypertensive rats. Chin. Med. J. 2002, 65, 1–6. [Google Scholar]
- Hsieh, M.-H.; Chan, P.; Sue, Y.-M.; Liu, J.C.; Liang, T.H.; Huang, T.Y.; Tomlinson, B.; Chow, M.S.; Kao, P.F.; Chen, Y.J. Efficacy and tolerability of oral stevioside in patients with mild essential hypertension: A two-year, randomized, placebo-controlled study. Clin. Ther. 2003, 25, 2797–2808. [Google Scholar] [CrossRef]
- Chatsudthipong, V.; Muanprasat, C. Stevioside and related compounds: Therapeutic benefits beyond sweetness. Pharmacol. Ther. 2009, 121, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Ma, Z.; Wang, J.; Milne, R.W.; Xu, D.; Davey, A.K.; Evans, A.M. Isosteviol reduces plasma glucose levels in the intravenous glucose tolerance test in Zucker diabetic fatty rats. Diabetes Obes. Metab. 2007, 9, 597–599. [Google Scholar] [CrossRef] [PubMed]
- Ferrazzano, G.F.; Cantile, T.; Alcidi, B. Is Stevia rebaudiana Bertoni a Non Cariogenic Sweetener? A Review. Molecules 2016, 21, 38. [Google Scholar] [CrossRef] [PubMed]
- Mizushina, Y.; Akihisa, T.; Ukiya, M.; Hamasaki, Y.; Murakami-Nakai, C.; Kuriyama, I.; Takeuchi, T.; Sugawara, F.; Yoshida, H. Structural analysis of isosteviol and related compounds as DNA polymerase and DNA topoisomerase inhibitors. Life Sci. 2005, 77, 2127–2140. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Du, W.; Zhao, L.; Davey, A.K.; Wang, J. The neuroprotective effects of isosteviol against focal cerebral ischemia injury induced by middle cerebral artery occlusion in rats. Planta Med. 2008, 74, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Zhang, S.; Foster, D.J.; Wang, J. The effects of isosteviol against myocardium injury induced by ischaemia-reperfusion in the isolated guinea pig heart. Clin. Exp. Pharmacol. Physiol. 2007, 34, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Li, Y.; Wang, J.; Wang, J.; Davey, A.K.; Zhang, S.; Evans, A.M. The cardioprotective effect of isosteviol on rats with heart ischemia-reperfusion injury. Life Sci. 2007, 80, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Wen, T.; Chen, Y.; Huang, L.; Lin, W.; Yin, C.; Tan, W. Isosteviol Sensitizes sarcKATP Channels towards Pinacidil and Potentiates Mitochondrial Uncoupling of Diazoxide in Guinea Pig Ventricular Myocytes. Oxid. Med. Cell. Longev. 2016, 2016, 6362812. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sato, T.; O’Rourke, B.; Marban, E. Mitochondrial ATP-dependent potassium channels: Novel effectors of cardioprotection? Circulation 1998, 97, 2463–2469. [Google Scholar] [CrossRef] [PubMed]
- Strobykina, I.Y.; Belenok, M.G.; Semenova, M.N.; Semenov, V.V.; Babaev, V.M.; Rizvano, I.K.; Mironov, V.F.; Kataev, V.E. Triphenylphosphonium Cations of the Diterpenoid Isosteviol: Synthesis and Antimitotic Activity in a Sea Urchin Embryo Model. J. Nat. Prod. 2015, 78, 1300–1308. [Google Scholar] [CrossRef] [PubMed]
- Ross, M.F.; Filipovska, A.; Smith, R.A.; Gait, M.J.; Murphy, M.P. Cell-penetrating peptides do not cross mitochondrial membranes even when conjugated to a lipophilic cation: Evidence against direct passage through phospholipid bilayers. Biochem. J. 2004, 383, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Testai, L. Flavonoids and mitochondrial pharmacology: A new paradigm for cardioprotection. Life Sci. 2015, 135, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Testai, L.; Martelli, A.; Marino, A.; D’Antongiovanni, V.; Ciregia, F.; Giusti, L.; Lucacchini, A.; Chericoni, S.; Breschi, M.C.; Calderone, V. The activation of mitochondrial BK potassium channels contributes to the protective effects of naringenin against myocardial ischemia/reperfusion injury. Biochem. Pharmacol. 2013, 85, 1634–1643. [Google Scholar] [CrossRef] [PubMed]
- Madan, S.; Ahmad, S.; Singh, G.N. Stevia rebaudiana (Bert.) Bertoni—A Review. Indian J. Nat. Prod. Resour. 2010, 1, 267–286. [Google Scholar]
- Jeppesen, P.B.; Gregersen, S.; Alstrup, K.K.; Hermansen, K. Stevioside induces antihyperglycaemic, insulinotropic and glucagonostatic effects in vivo: Studies in the diabetic Goto-Kakizaki (GK) rats. Phytomedicine 2002, 9, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Gregersen, S.; Jeppesen, P.B.; Holst, J.J.; Hermansen, K. Antihyperglycemic Effects of Stevioside in Type 2 Diabetic Subjects. Metabolism 2004, 53, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Chappell, J.B.; Hansford, R.G. Preparation of mitochondria from animal tissues and yeasts. In Subcellular Components: Preparation and Fractionation; Birnie, G.D., Ed.; Butterworths: London, UK, 1972; pp. 77–91. [Google Scholar]
- Calderone, V.; Testai, L.; Martelli, A.; Rapposelli, S.; Digiacomo, M.; Balsamo, A.; Breschi, M.C. Anti-ischemic properties of a new spiro-cyclic benzopyran activator of the cardiac mito-KATP channel. Biochem. Pharmacol. 2010, 79, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Drew, B.; Leeuwenburgh, C. Method for measuring ATP production in isolated mitochondria: ATP production in brain and liver mitochondria of Fischer-344 rats with age and caloric restriction. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003, 285, R1259–R1267. [Google Scholar] [CrossRef] [PubMed]
- Labajova, A.; Vojtiskova, A.; Krivakova, P.; Kofranek, J.; Drahota, Z.; Houstek, J. Evaluation of mitochondrial membrane potential using a computerized device with a tetraphenylphosphonium-selective electrode. Anal. Biochem. 2006, 353, 37–42. [Google Scholar] [CrossRef] [PubMed]
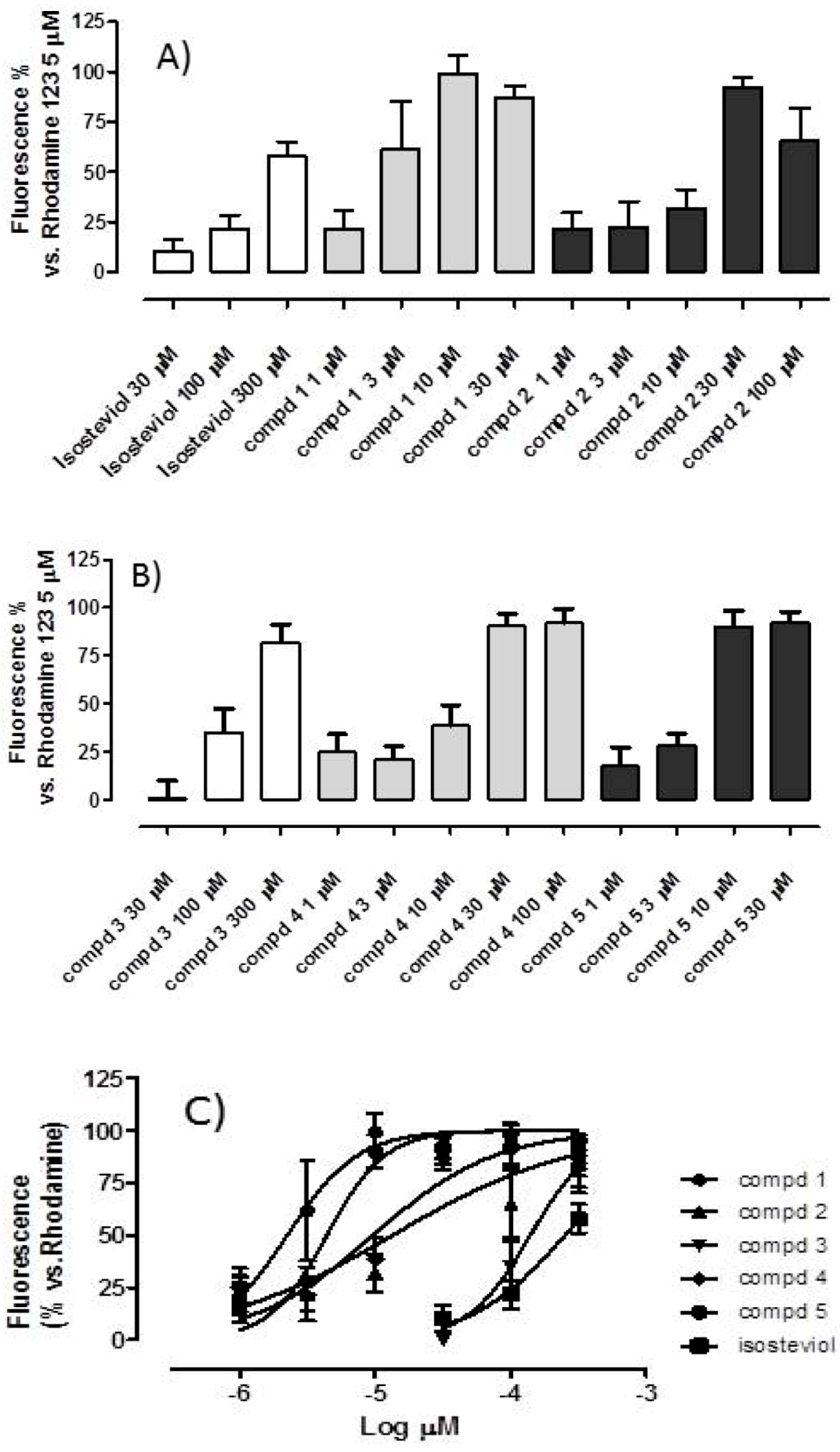
| Compounds | pEC50 | Emax (%) |
|---|---|---|
| Isosteviol | 3.59 ± 0.074 | 58 ± 7 |
| Cmpd 1 | 5.65 ± 1.67 | Full |
| Cmpd 2 | 4.87 ± 0.65 | Full |
| Cmpd 3 | 3.85 ± 0.090 | 82 ± 9 |
| Cmpd 4 | 5.01 ± 0.095 | Full |
| Cmpd 5 | 5.36 ± 0.060 | Full |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Testai, L.; Strobykina, I.; Semenov, V.V.; Semenova, M.; Pozzo, E.D.; Martelli, A.; Citi, V.; Martini, C.; Breschi, M.C.; Kataev, V.E.; et al. Mitochondriotropic and Cardioprotective Effects of Triphenylphosphonium-Conjugated Derivatives of the Diterpenoid Isosteviol. Int. J. Mol. Sci. 2017, 18, 2060. https://doi.org/10.3390/ijms18102060
Testai L, Strobykina I, Semenov VV, Semenova M, Pozzo ED, Martelli A, Citi V, Martini C, Breschi MC, Kataev VE, et al. Mitochondriotropic and Cardioprotective Effects of Triphenylphosphonium-Conjugated Derivatives of the Diterpenoid Isosteviol. International Journal of Molecular Sciences. 2017; 18(10):2060. https://doi.org/10.3390/ijms18102060
Chicago/Turabian StyleTestai, Lara, Irina Strobykina, Victor V. Semenov, Marina Semenova, Eleonora Da Pozzo, Alma Martelli, Valentina Citi, Claudia Martini, Maria C. Breschi, Vladimir E. Kataev, and et al. 2017. "Mitochondriotropic and Cardioprotective Effects of Triphenylphosphonium-Conjugated Derivatives of the Diterpenoid Isosteviol" International Journal of Molecular Sciences 18, no. 10: 2060. https://doi.org/10.3390/ijms18102060
APA StyleTestai, L., Strobykina, I., Semenov, V. V., Semenova, M., Pozzo, E. D., Martelli, A., Citi, V., Martini, C., Breschi, M. C., Kataev, V. E., & Calderone, V. (2017). Mitochondriotropic and Cardioprotective Effects of Triphenylphosphonium-Conjugated Derivatives of the Diterpenoid Isosteviol. International Journal of Molecular Sciences, 18(10), 2060. https://doi.org/10.3390/ijms18102060