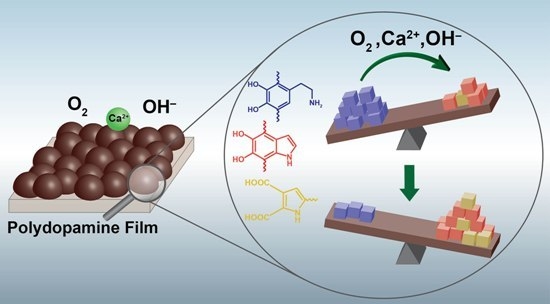
Calcium-Mediated Control of Polydopamine Film Oxidation and Iron Chelation
Abstract
:1. Introduction
2. Results and Discussion
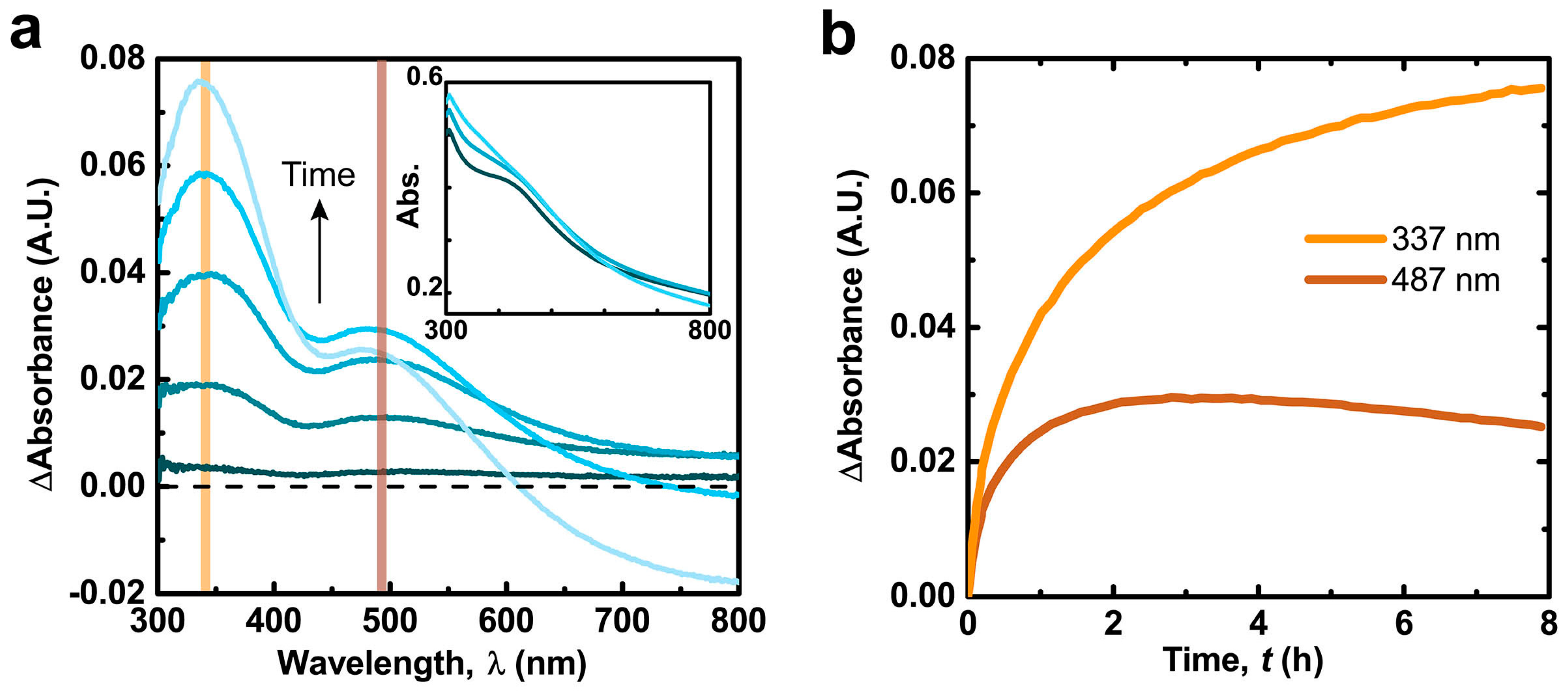
2.1. Alkaline Calcium Solutions Promote Generation of Semiquinone Radicals and Oxidative Evolution of Polydopamine (PDA) Films
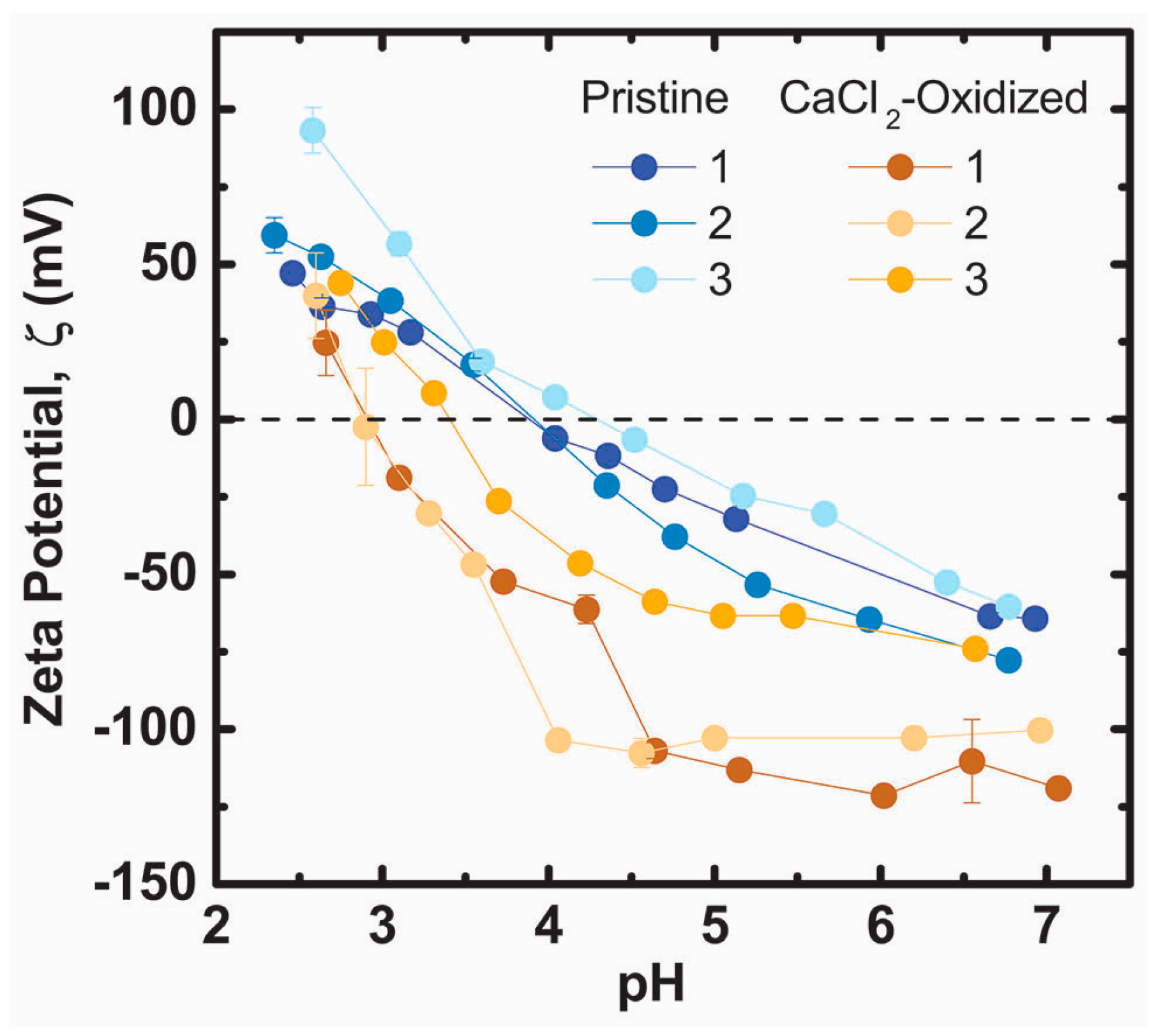
2.2. Alkaline Calcium Solutions Decrease Dopamine Content in PDA and Generate Carboxylic Acid Groups
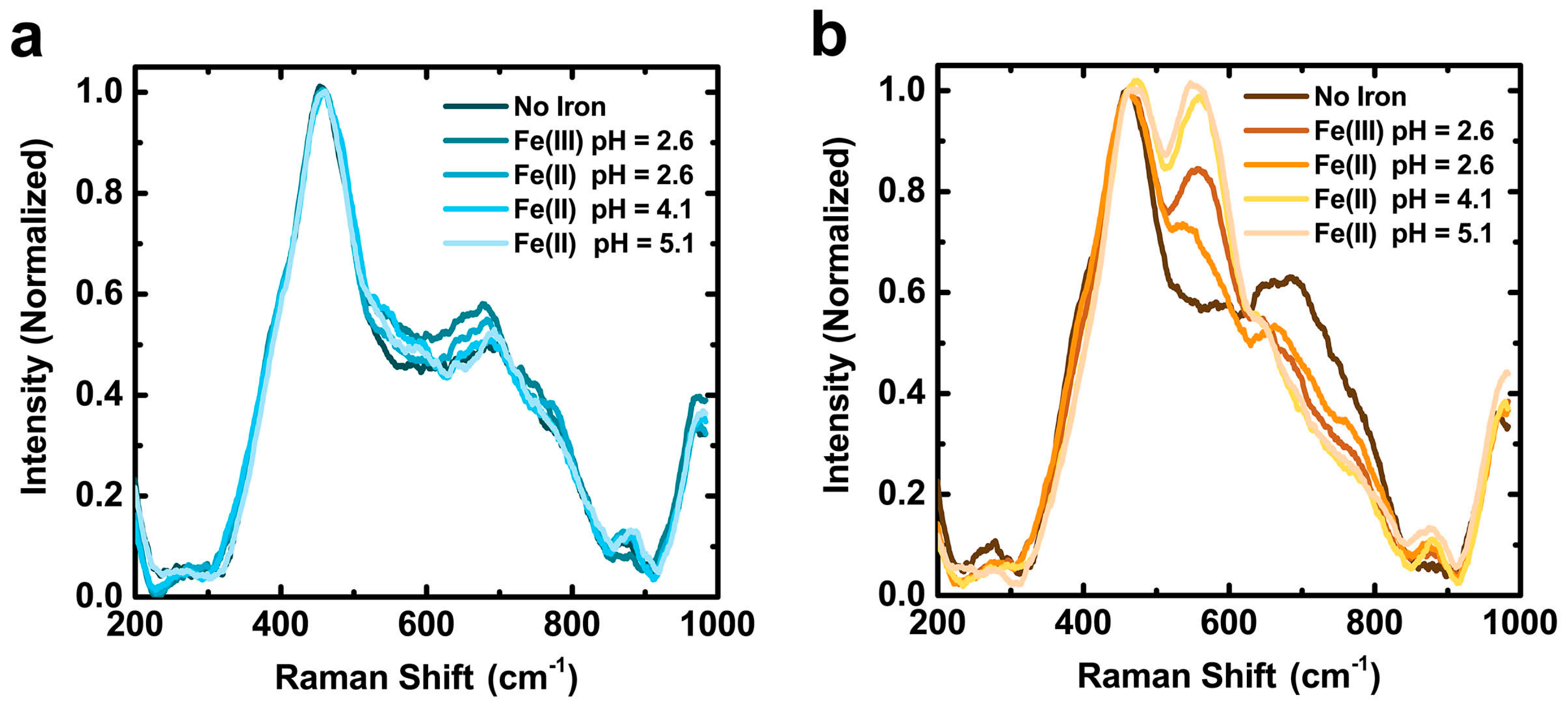
2.3. Films Incubated in Alkaline CaCl2 Solutions Exhibit Enhanced Iron Chelation
3. Materials and Methods
3.1. Materials
3.2. PDA Film Preparation and Iron Binding
3.3. Spectroscopic and Electrochemical Characterization of PDA Films
3.4. Morphological Characterization of PDA Films
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PDA | Polydopamine |
| DHI | 5,6-Dihydroxyindole |
| PDCA | Pyrrole-2,3,dicarboxylic acid |
| UV-Vis | Ultraviolet-visible |
| ATR-IR | Attenuated total reflectance-infrared |
| AFM | Atomic force microscopy |
References
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-Inspired Surface Chemistry for Multifunctional Coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Guardingo, M.; Bellido, E.; Miralles-Lluma, R.; Faraudo, J.; Sedo, J.; Tatay, S.; Verdaguer, A.; Busque, F.; Ruiz-Molina, D. Bioinspired Catechol-Terminated Self-Assembled Monolayers with Enhanced Adhesion Properties. Small 2014, 10, 1594–1602. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Scherer, N.F.; Messersmith, P.B. Single-molecule mechanics of mussel adhesion. Proc. Natl. Acad. Sci. USA 2006, 103, 12999–13003. [Google Scholar] [CrossRef] [PubMed]
- Pierpont, C.G.; Lange, C.W. The chemistry of transition metal complexes containing catechol and semiquinone ligands. In Progress in Inorganic Chemistry; Wiley: Hoboken, NJ, USA, 1993; Volume 41, pp. 331–442. [Google Scholar]
- Kim, E.; Liu, Y.; Leverage, W.T.; Yin, J.-J.; White, I.M.; Bentley, W.E.; Payne, G.F. Context-Dependent Redox Properties of Natural Phenolic Materials. Biomacromolecules 2014, 15, 1653–1662. [Google Scholar] [CrossRef] [PubMed]
- Lynge, M.E.; van der Westen, R.; Postma, A.; Städler, B. Polydopamine—A nature-inspired polymer coating for biomedical science. Nanoscale 2011, 3, 4916–4928. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ai, K.; Lu, L. Polydopamine and Its Derivative Materials: Synthesis and Promising Applications in Energy, Environmental, and Biomedical Fields. Chem. Rev. 2014, 114, 5057–5115. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Zhu, L.; Zhu, L.; Zhang, H.; Zhu, B.; Xu, Y. Antifouling and antimicrobial polymer membranes based on bioinspired polydopamine and strong hydrogen-bonded poly(N-vinyl pyrrolidone). ACS Appl. Mater. Interfaces 2013, 5, 12895–12904. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Su, Y.; Zhao, X.; He, X.; Zhang, R.; Zhao, J.; Fan, X.; Jiang, Z. Antifouling, High-Flux Nanofiltration Membranes Enabled by Dual Functional Polydopamine. ACS Appl. Mater. Interfaces 2014, 6, 5548–5557. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Rho, J.; Messersmith, P.B. Facile Conjugation of Biomolecules onto Surfaces via Mussel Adhesive Protein Inspired Coatings. Adv. Mater. 2009, 21, 431–434. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.; Ku, S.H.; Lee, H.; Park, C.B. Mussel-Inspired Polydopamine Coating as a Universal Route to Hydroxyapatite Crystallization. Adv. Funct. Mater. 2010, 20, 2132–2139. [Google Scholar] [CrossRef]
- Lee, M.; Rho, J.; Lee, D.E.; Hong, S.; Choi, S.J.; Messersmith, P.B.; Lee, H. Water detoxification by a substrate-bound catecholamine adsorbent. Chempluschem 2012, 77, 987–990. [Google Scholar] [CrossRef] [PubMed]
- Farnad, N.; Farhadi, K.; Voelcker, N.H. Polydopamine Nanoparticles as a New and Highly Selective Biosorbent for the Removal of Copper(II) Ions from Aqueous Solutions. Water Air Soil Pollut. 2012, 223, 3535–3544. [Google Scholar] [CrossRef]
- Park, H.-A.; Kim, Y.J.; Kwon, I.S.; Klosterman, L.; Bettinger, C.J. Lithium purification from aqueous solutions using bioinspired redox active melanin membranes. Polym. Int. 2016, 65, 1331–1338. [Google Scholar] [CrossRef]
- Herlinger, E.; Jameson, R.F.; Linert, W. Spontaneous Autoxidation of Dopamine. J. Chem. Soc. Perkin Trans. 1995, 2, 259–263. [Google Scholar]
- Bernsmann, F.; Ball, V.; Ponche, A.; Michel, M.; Gracio, D.A.; Ruch, D. Dopamine—Melanin Film Deposition Depends on the Used Oxidant and Buffer Solution. Langmuir 2011, 27, 2819–2825. [Google Scholar] [CrossRef] [PubMed]
- Klosterman, L.; Riley, J.K.; Bettinger, C.J. Control of Heterogeneous Nucleation and Growth Kinetics of Dopamine-Melanin by Altering Substrate Chemistry. Langmuir 2015, 31, 3451–3458. [Google Scholar] [CrossRef] [PubMed]
- Ball, V.; Frari, D.D.; Toniazzo, V.; Ruch, D. Kinetics of polydopamine film deposition as a function of pH and dopamine concentration: Insights in the polydopamine deposition mechanism. J. Colloid Interface Sci. 2012, 386, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Ponzio, F.; Barthes, J.; Bour, J.; Michel, M.; Bertani, P.; Hemmerlé, J.; D’Ischia, M.; Ball, V. Oxidant Control of Polydopamine Surface Chemistry in Acids: A Mechanism-Based Entry to Superhydrophilic-Superoleophobic Coatings. Chem. Mater. 2016, 28, 4697–4705. [Google Scholar] [CrossRef]
- Bernsmann, F.; Voegel, J.C.; Ball, V. Different synthesis methods allow to tune the permeability and permselectivity of dopamine-melanin films to electrochemical probes. Electrochim. Acta 2011, 56, 3914–3919. [Google Scholar] [CrossRef]
- Del Frari, D.; Bour, J.; Ball, V.; Toniazzo, V.; Ruch, D. Degradation of polydopamine coatings by sodium hypochlorite: A process depending on the substrate and the film synthesis method. Polym. Degrad. Stab. 2012, 97, 1844–1849. [Google Scholar] [CrossRef]
- Della Vecchia, N.F.; Avolio, R.; Alfè, M.; Errico, M.E.; Napolitano, A.; D’Ischia, M. Building-block diversity in polydopamine underpins a multifunctional eumelanin-type platform tunable through a quinone control point. Adv. Funct. Mater. 2013, 23, 1331–1340. [Google Scholar] [CrossRef]
- Della Vecchia, N.F.; Luchini, A.; Napolitano, A.; D’Errico, G.; Vitiello, G.; Szekely, N.; d’Ishcia, M.; Paduano, L. Tris Buffer Modulates Polydopamine Growth, Aggregation, and Paramagnetic Properties. Langmuir 2014, 30, 9811–9818. [Google Scholar] [CrossRef] [PubMed]
- Proks, V.; Brus, J.; Pop-Georgievski, O.; Večerníková, E.; Wisniewski, W.; Kotek, J.; Urbanová, M.; Rypáček, F. Thermal-induced transformation of polydopamine structures: An efficient route for the stabilization of the polydopamine surfaces. Macromol. Chem. Phys. 2013, 214, 499–507. [Google Scholar] [CrossRef]
- Kim, S.; Gim, T.; Kang, S.M. Stability-enhanced polydopamine coatings on solid substrates by iron(III) coordination. Prog. Org. Coat. 2014, 77, 1336–1339. [Google Scholar] [CrossRef]
- Lim, C.; Huang, J.; Kim, S.; Lee, H.; Zeng, H.; Hwang, D.S. Nanomechanics of Poly(catecholamine) Coatings in Aqueous Solutions. Angew. Chem. Int. Ed. 2016, 55, 3342–3346. [Google Scholar] [CrossRef] [PubMed]
- Lebedev, A.V.; Ivanova, M.V.; Ruuge, E.K. Calcium-dioxolene complexes: Rate constants of pyrocatechol oxidation in the presence of Ca2+. Biophysics 2011, 56, 188–193. [Google Scholar] [CrossRef]
- Liebscher, J.; Mrówczyński, R.; Scheidt, H.A.; Filip, C.; Hădade, N.D.; Turcu, R.; Bende, A.; Beck, S. Structure of Polydopamine: A Never Ending Story? Langmuir 2013, 29, 10539–10548. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Na, Y.S.; Choi, S.; Song, I.T.; Kim, W.Y.; Lee, H. Non-Covalent Self-Assembly and Covalent Polymerization Co-Contribute to Polydopamine Formation. Adv. Funct. Mater. 2012, 22, 4711–4717. [Google Scholar] [CrossRef]
- Munoz-Munoz, J.L.; García-Molina, F.; Varón, R.; Tudela, J.; García-Cánovas, F.; Rodríguez-López, J.N. Generation of hydrogen peroxide in the melanin biosynthesis pathway. Biochim. Biophys. Acta 2009, 1794, 1017–1029. [Google Scholar] [CrossRef] [PubMed]
- Reale, S.; Crucianelli, M.; Pezzella, A.; D’Ischia, M.; De Angelis, F. Exploring the frontiers of synthetic eumelanin polymers by high-resolution matrix-assisted laser/desorption ionization mass spectrometry. J. Mass Spectrom. 2012, 47, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Gracio, J.D.A.; Singh, Â.M.K.; Ruch, D.; Buehler, M.J.; Al, C.E.T.; Ball, V. Self-Assembly of Tetramers of 5,6-Dihydroxyindole Explains the Primary Physical Properties of Eumelanin: Experiment, Simulation, and Design. ACS Nano 2013, 7, 1524–1532. [Google Scholar]
- Watt, A.A.R.; Bothma, J.P.; Meredith, P. The supramolecular structure of melanin. Soft Matter 2009, 5, 3754. [Google Scholar] [CrossRef]
- d’Ischia, M.; Napolitano, A.; Ball, V.; Chen, C.; Buehler, M.J. Polydopamine and Eumelanin: From Structure—Property Relationships to a Unified Tailoring Strategy. Acc. Chem. Res. 2014, 47, 3541–3550. [Google Scholar] [CrossRef] [PubMed]
- Mrówczyński, R.; Coy, L.E.; Scheibe, B.; Czechowski, T.; Augustyniak-Jabłokow, M.; Jurga, S.; Tadyszak, K. Electron Paramagnetic Resonance Imaging and Spectroscopy of Polydopamine Radicals. J. Phys. Chem. B 2015, 119, 10341–10347. [Google Scholar] [CrossRef] [PubMed]
- Steenken, S.; Neta, P. One-electron redox potentials of phenols. Hydroxy- and aminophenols and related compounds of biological interest. J. Phys. Chem. 1982, 86, 3661–3667. [Google Scholar] [CrossRef]
- Lambert, C.; Chacon, J.N.; Chedekel, M.R.; Land, E.J.; Riley, P.A.; Thompson, A.; Truscott, T.G. A pulse radiolysis investigation of the oxidation of indolic melanin precursors: Evidence for indolequinones and subsequent intermediates. Biochim. Biophys. Acta 1989, 993, 12–20. [Google Scholar] [CrossRef]
- Thompson, A.; Land, E.J.; Chedekel, M.R.; Subbarao, K.V.; Truscott, T.G. A pulse radiolysis investigation of the oxidation of the melanin precursors 3,4-dihydroxyphenylalanine (dopa) and the cysteinyldopas. Biochim. Biophys. Acta 1985, 843, 49–57. [Google Scholar] [CrossRef]
- Craw, M.; Chedekel, M.R.; Truscott, T.G.; Land, E.J. The Photochemical Interaction Between the Triplet State of 8-Methoxypsoralen and the Melanin Precursor l-3,4 Dihydroxyphenylalanine. Photochem. Photobiol. 1984, 39, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Dryhurst, G. Oxidation Chemistry of Dopamine: Possible Insights into the Age-Dependent Loss of Dopaminergic Nigrostriatal Neurons. Bioorg. Chem. 1993, 21, 392–410. [Google Scholar] [CrossRef]
- Ascione, L.; Pezzella, A.; Ambrogi, V.; Carfagna, C.; D’Ischia, M. Intermolecular π-electron perturbations generate extrinsic visible contributions to eumelanin black chromophore in model polymers with interrupted interring conjugation. Photochem. Photobiol. 2013, 89, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Tran, M.L.; Powell, B.J.; Meredith, P. Chemical and structural disorder in eumelanins: A possible explanation for broadband absorbance. Biophys. J. 2006, 90, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Meredith, P.; Powell, B.J.; Riesz, J.; Nighswander-Rempel, S.; Pederson, M.R.; Moore, E. Towards Structure-Property-Function Relationships for Eumelanin. Soft Matter 2006, 2, 37–44. [Google Scholar] [CrossRef]
- Korytowski, W.; Sarna, T. Bleaching of melanin pigments. Role of copper ions and hydrogen peroxide in autooxidation and photooxidation of synthetic dopa-melanin. J. Biol. Chem. 1990, 265, 12410–12416. [Google Scholar] [PubMed]
- Littrell, K.C.; Gallas, J.M.; Zajac, G.W.; Thiyagarajan, P. Structural studies of bleached melanin by synchrotron small-angle X-ray scattering. Photochem. Photobiol. 2003, 77, 115–120. [Google Scholar] [CrossRef]
- Lebedev, A.V.; Ivanova, M.V.; Timoshin, A.A.; Ruuge, E.K. Effect of group II metal cations on catecholate oxidation. Chemphyschem 2007, 8, 1863–1869. [Google Scholar] [CrossRef] [PubMed]
- Lebedev, A.V.; Ivanova, M.V.; Timoshin, A.A.; Ruuge, E.K. Effect of calcium cations on acid-base properties and free radical oxidation of dopamine and pyrocatechol. Biomed. Khim. 2008, 54, 687–695. [Google Scholar] [PubMed]
- Felix, C.C.; Hyde, J.S.; Sarna, T.; Sealy, R.C. Interactions of melanin with metal ions. Electron spin resonance evidence for chelate complexes of metal ions with free radicals. J. Am. Chem. Soc. 1978, 100, 3922–3926. [Google Scholar] [CrossRef]
- Ishimitsu, T.; Hirose, S.; Sakurai, H. Microscopic Acid Dissociation Constants of 3,4-Dihydroxyphenethylamine (Dopamine). Chem. Pharm. Bull. 1978, 26, 74–78. [Google Scholar] [CrossRef]
- Mostert, A.B.; Powell, B.J.; Pratt, F.L.; Hanson, G.R.; Sarna, T.; Gentle, I.R.; Meredith, P. Role of semiconductivity and ion transport in the electrical conduction of melanin. Proc. Natl. Acad. Sci. USA 2012, 109, 8943–8947. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S.; Hayon, E. Ionization Constants and Spectral Characteristics of Some Semiquinone Radicals in Aqueous Solution. J. Phys. Chem. 1973, 77, 2274–2276. [Google Scholar] [CrossRef]
- Mostert, A.B.; Hanson, G.R.; Sarna, T.; Gentle, I.R.; Powell, B.J.; Meredith, P. Hydration-controlled X-band EPR spectroscopy: A tool for unravelling the complexities of the solid-state free radical in eumelanin. J. Phys. Chem. B 2013, 117, 4965–4972. [Google Scholar] [CrossRef] [PubMed]
- Szpoganicz, B.; Gidanian, S.; Kong, P.; Farmer, P. Metal binding by melanins: Studies of colloidal dihydroxyindole-melanin, and its complexation by Cu(II) and Zn(II) ions. J. Inorg. Biochem. 2002, 89, 45–53. [Google Scholar] [CrossRef]
- D’Ischia, M.; Napolitano, A.; Pezzella, A.; Land, E.J.; Ramsden, C.A.; Riley, P.A. 5,6-Dihydroxyindoles and Indole-5,6-diones. Adv. Heterocycl. Chem. 2005, 89, 1–63. [Google Scholar]
- Liu, Y.; Hong, L.; Kempf, V.R.; Wakamatsu, K.; Ito, S.; Simon, J.D. Ion-exchange and adsorption of Fe(III) by Sepia melanin. Pigment Cell Res. 2004, 17, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Liu, J.; Liu, S.; Zhou, F. Pdop layer exhibiting zwitterionicity: A simple electrochemical interface for governing ion permeability. Chem. Commun. 2010, 46, 5900–5902. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Su, L.; Tong, Y.; Guan, M.; Zhang, X. Ion permeability of polydopamine films revealed using a prussian blue-based electrochemical method. J. Phys. Chem. B 2014, 118, 12781–12787. [Google Scholar] [CrossRef] [PubMed]
- Gunasekaran, S.; Kumar, R.T.; Ponnusamy, S. Vibrational spectra and normal coordinate analysis of adrenaline and dopamine. Indian J. Pure Appl. Phys. 2007, 45, 884–892. [Google Scholar]
- Ball, V. Impedance spectroscopy and ζ potential titration of dopa-melanin films produced by oxidation of dopamine. Colloids Surfaces A Physicochem. Eng. Asp. 2010, 363, 92–97. [Google Scholar] [CrossRef]
- Richter, H.W.; Waddell, W.H. Mechanism of the Oxidation of Dopamine by the Hydroxyl Radical in Aqueous Solution. J. Am. Chem. Soc. 1983, 105, 5434–5440. [Google Scholar] [CrossRef]
- Samokhvalov, A.; Liu, Y.; Simon, J.D. Characterization of the Fe(III)-binding site in Sepia eumelanin by resonance Raman confocal microspectroscopy. Photochem. Photobiol. 2004, 80, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Harrington, M.J.; Masic, A.; Holten-andersen, N.; Waite, J.H.; Fratzl, P. Iron-Clad Fibers: A Metal-Based Biological Strategy for Hard Flexible Coatings. Science 2010, 328, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- Capozzi, V.; Perna, G.; Gallone, A.; Biagi, P.F.; Carmone, P.; Fratello, A.; Guida, G.; Zanna, P.; Cicero, R. Raman and optical spectroscopy of eumelanin films. J. Mol. Struct. 2005, 744–747, 717–721. [Google Scholar] [CrossRef]
- Albano, L.G.S.; Di Mauro, E.; Kumar, P.; Cicoira, F.; Graeff, C.F.O.; Santato, C. Novel insights on the physicochemical properties of eumelanins and their DMSO derivatives. Polym. Int. 2016, 65, 1315–1322. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A.B. Chemical and structural properties of carbonaceous products obtained by hydrothermal carbonization of saccharides. Chem. A Eur. J. 2009, 15, 4195–4203. [Google Scholar] [CrossRef] [PubMed]
- John, K.J.; Manoj, B. Raman Spectroscopy Investigation of Camphor Soot: Spectral Analysis and Structural Information. Int. J. Electrochem. Sci. 2013, 8, 9421–9428. [Google Scholar]
- Sánchez-Cortés, S.; Francioso, O.; Ciavatta, C.; García-Ramos, J.V.; Gessa, C. pH-Dependent Adsorption of Fractionated Peat Humic Substances on Different Silver Colloids Studied by Surface-Enhanced Raman Spectroscopy. J. Colloid Interface Sci. 1998, 198, 308–318. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A.B. The production of carbon materials by hydrothermal carbonization of cellulose. Carbon 2009, 47, 2281–2289. [Google Scholar] [CrossRef]
- Tylli, H.; Konschin, H. A raman spectroscopic study of the OH and OD torsion in 1,2-dihydroxybenzene. J. Mol. Struct. 1979, 57, 13–19. [Google Scholar] [CrossRef]
- Fujita, J.; Martell, A.E.; Nakamoto, K. Infrared Spectra of Metal Chelate Compounds. VI. A Normal Coordinate Treatment of Oxalato Metal Complexes. J. Chem. Phys. 1962, 36, 324. [Google Scholar] [CrossRef]
- Michaud-Soret, I.; Andersson, K.K.; Que, L.; Haavik, J. Resonance Raman studies of catecholate and phenolate complexes of recombinant human tyrosine hydroxylase. Biochemistry 1995, 34, 5504–5510. [Google Scholar] [CrossRef] [PubMed]
- Salama, S.; Stong, J.D.; Neilands, J.B.; Spiro, T.G. Electronic and resonance Raman spectra of iron(III) complexes of enterobactin, catechol, and N-methyl-2,3-dihydroxybenzamide. Biochemistry 1978, 17, 3781–3785. [Google Scholar] [CrossRef] [PubMed]
- Que, L., Jr.; Heistand, R.H., II. Resonance Raman Studies on Pyrocatechase. Am. Chem. Soc. 1979, 101, 2219–2221. [Google Scholar] [CrossRef]
- Hurst, S.J.; Fry, H.C.; Gosztola, D.J.; Rajh, T. Utilizing chemical Raman enhancement: A route for metal oxide support-based biodetection. J. Phys. Chem. C 2011, 115, 620–630. [Google Scholar] [CrossRef]
- Musumeci, A.; Gosztola, D.; Schiller, T.; Dimitrijevic, N.M.; Mujica, V.; Martin, D.; Rajh, T. SERS of semiconducting nanoparticles (TiO(2) hybrid composites). J. Am. Chem. Soc. 2009, 131, 6040–6041. [Google Scholar] [CrossRef] [PubMed]
- Charkoudian, L.K.; Franz, K.J. Fe(III)-Coordination Properties of Neuromelanin Components: 5,6-Dihydroxyindole and 5,6-Dihydroxyindole-2-Carboxylic Acid. Inorg. Chem. 2006, 45, 3657–3664. [Google Scholar] [CrossRef] [PubMed]
- Ryou, M.-H.; Lee, Y.M.; Park, J.-K.; Choi, J.W. Mussel-inspired polydopamine-treated polyethylene separators for high-power li-ion batteries. Adv. Mater. 2011, 23, 3066–3070. [Google Scholar] [CrossRef] [PubMed]
- Nikanorov, A.M.; Brazhnikova, L.V. Water Chemical Composition of Rivers, Lakes, and Wetlands. In Types and Properties of Waters; EOLSS Publishers: Oxford, UK, 2009; Volume II, pp. 42–79. [Google Scholar]
- Poh, C.K.; Shi, Z.; Lim, T.Y.; Neoh, K.G.; Wang, W. The effect of VEGF functionalization of titanium on endothelial cells in vitro. Biomaterials 2010, 31, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.; Choi, I.S.; Nam, Y. A biofunctionalization scheme for neural interfaces using polydopamine polymer. Biomaterials 2011, 32, 6374–6380. [Google Scholar] [CrossRef] [PubMed]
- Oyane, A.; Kim, H.-M.; Furuya, T.; Kokubo, T.; Miyazaki, T.; Nakamura, T. Preparation and assessment of revised simulated body fluids. J. Biomed. Mater. Res. 2003, 65, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Misawa, T. The Thermodynamic Consideration for Fe-H2O System at 25 °C. Corros. Sci. 1973, 13, 659–676. [Google Scholar] [CrossRef]
- Morgan, B.; Lahav, O. The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution—Basic principles and a simple heuristic description. Chemosphere 2007, 68, 2080–2084. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Hoggard, J.D.; Sides, P.J.; Prieve, D.C. Measurement of the streaming potential and streaming current near a rotating disk to determine its ζ potential. Langmuir 2005, 21, 7433–7438. [Google Scholar] [CrossRef] [PubMed]
- Aigen, S.; Saiget, T.; Sides, P.J. ZetaSpin: A Revolution in ζ Potential Measurement. Available online: https://www.andrew.cmu.edu/course/39-801/index.html (accessed on 29 November 2016).




© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klosterman, L.; Bettinger, C.J. Calcium-Mediated Control of Polydopamine Film Oxidation and Iron Chelation. Int. J. Mol. Sci. 2017, 18, 14. https://doi.org/10.3390/ijms18010014
Klosterman L, Bettinger CJ. Calcium-Mediated Control of Polydopamine Film Oxidation and Iron Chelation. International Journal of Molecular Sciences. 2017; 18(1):14. https://doi.org/10.3390/ijms18010014
Chicago/Turabian StyleKlosterman, Luke, and Christopher J. Bettinger. 2017. "Calcium-Mediated Control of Polydopamine Film Oxidation and Iron Chelation" International Journal of Molecular Sciences 18, no. 1: 14. https://doi.org/10.3390/ijms18010014
APA StyleKlosterman, L., & Bettinger, C. J. (2017). Calcium-Mediated Control of Polydopamine Film Oxidation and Iron Chelation. International Journal of Molecular Sciences, 18(1), 14. https://doi.org/10.3390/ijms18010014