3D Visualization of Developmental Toxicity of 2,4,6-Trinitrotoluene in Zebrafish Embryogenesis Using Light-Sheet Microscopy
Abstract
:1. Introduction
2. Results
2.1. Pink Water Inhibits Melanin Synthesis and Affects Melanocyte Development
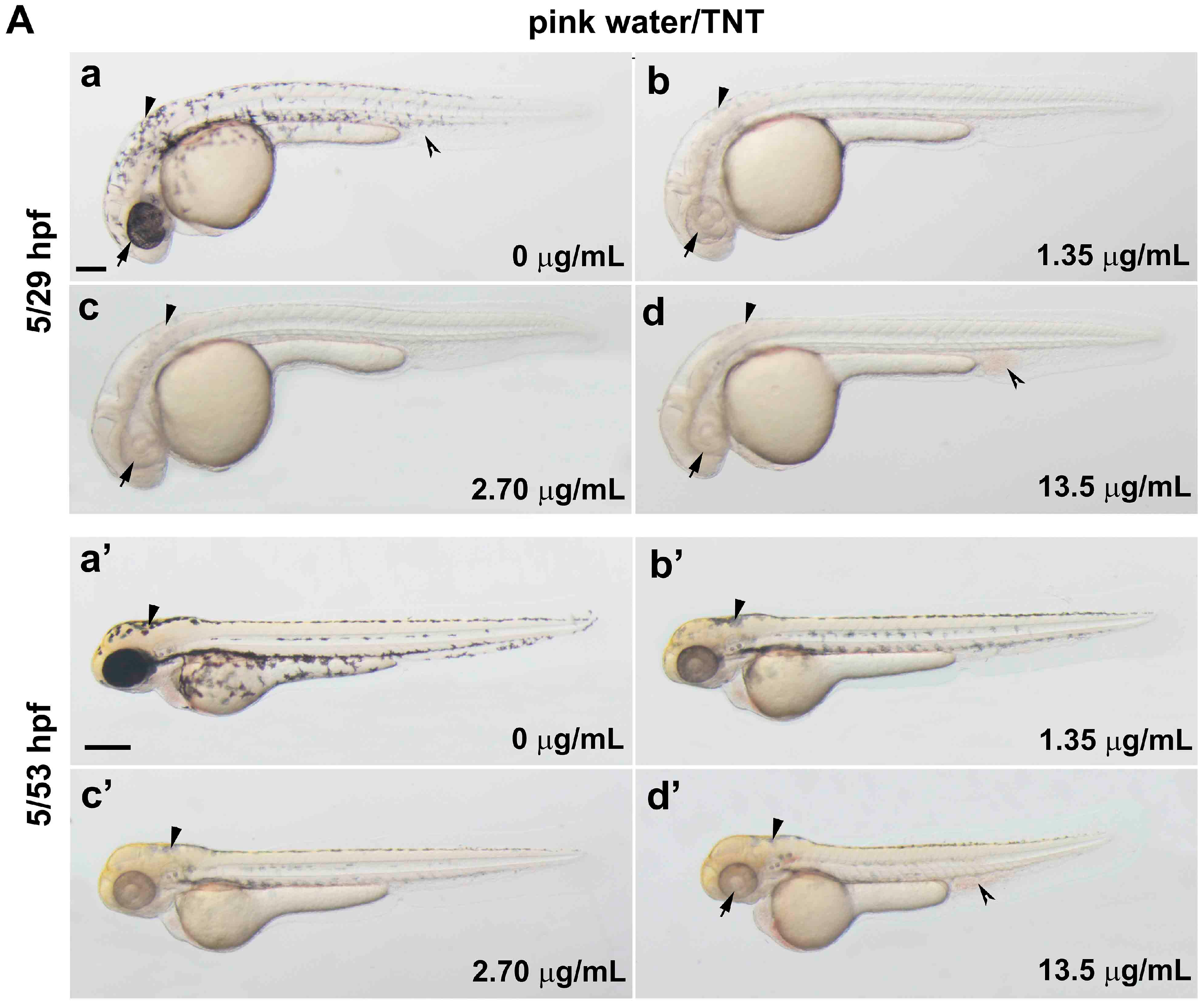
2.2. TNT in Pink Water Causes Early Developmental Defects in Zebrafish Embryogenesis
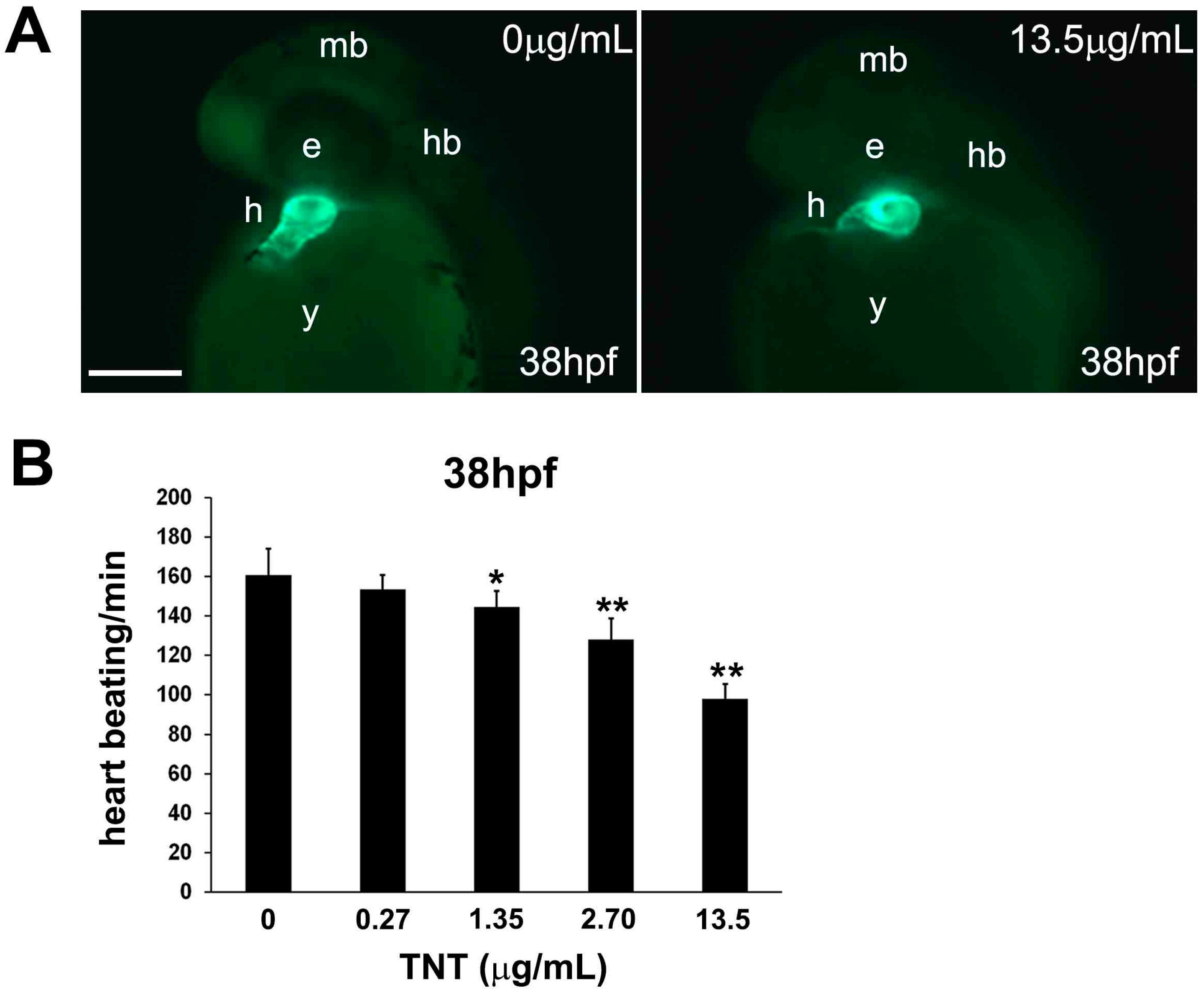
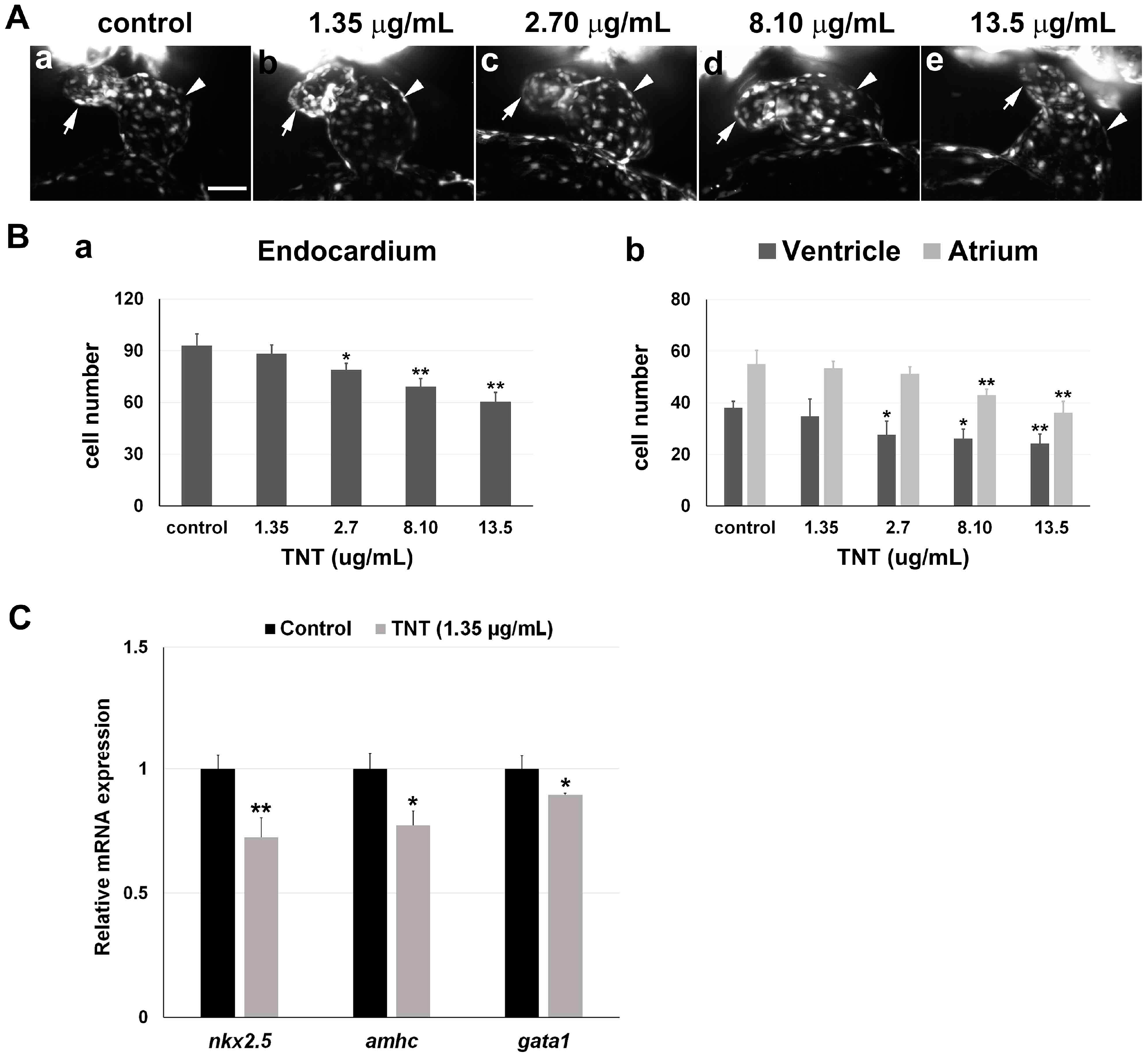
2.3. Pink Water Affects Heart Development and Function
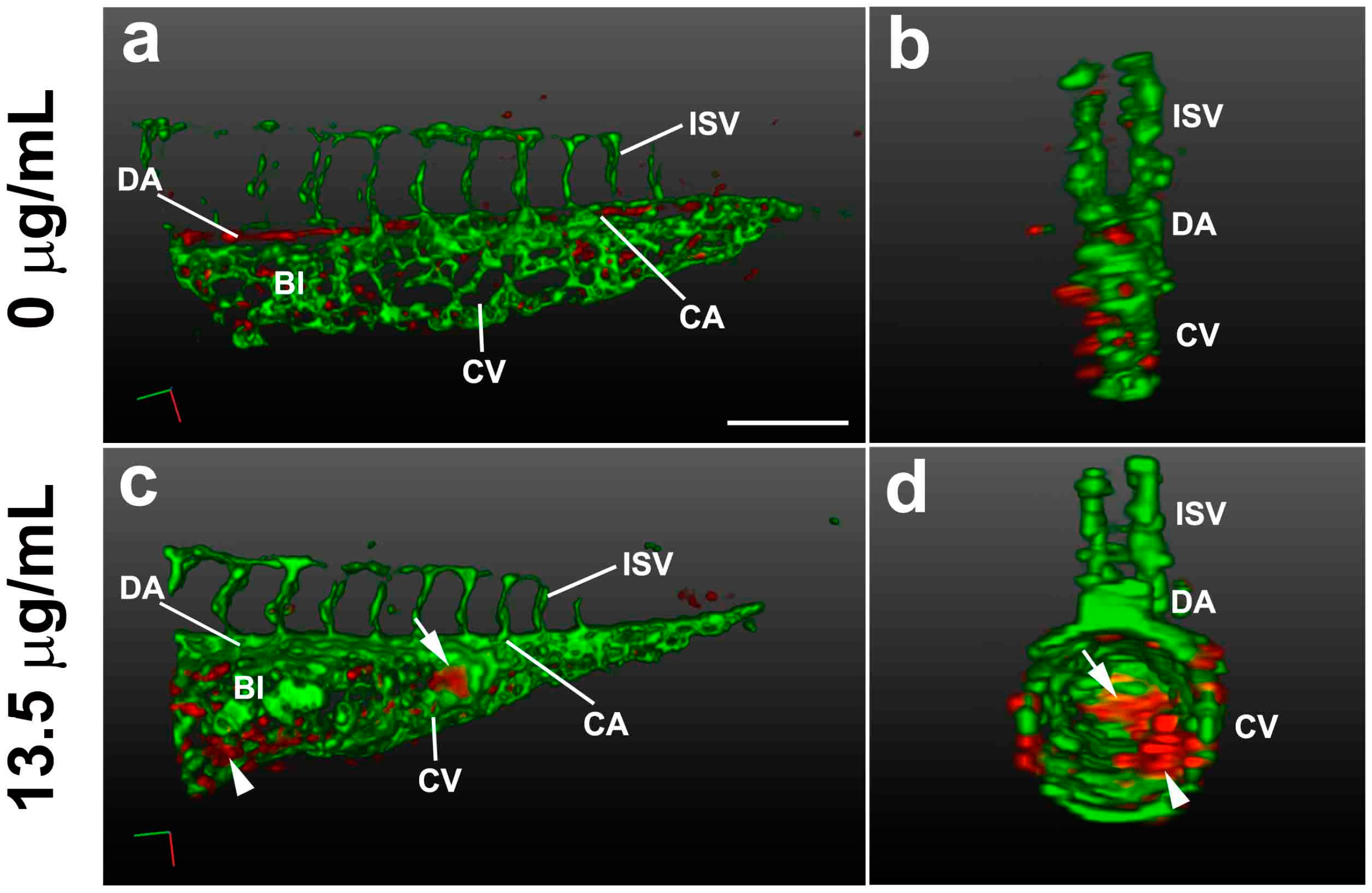
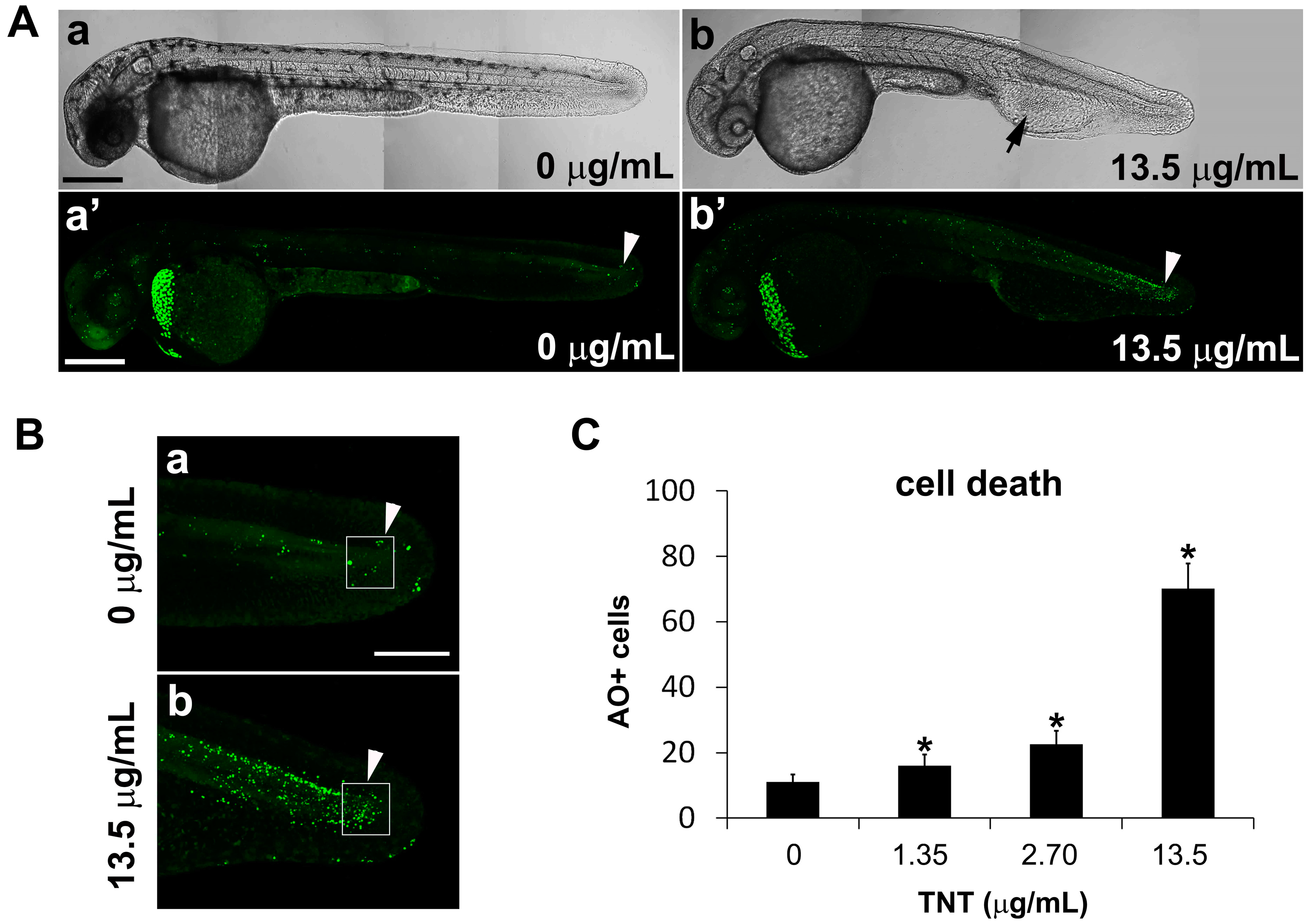
2.4. Live Imaging of TNT-Associated Cell Death
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Chemicals and High Performance Liquid Chromatography (HPLC) Analysis
4.3. Treatments
4.4. Live Imaging of Zebrafish Embryos
4.5. Acridine Orange Staining
4.6. Quantitative Real-Time PCR (qPCR)
4.7. Data and Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Talmage, S.S.; Opresko, D.M.; Maxwell, C.J.; Welsh, C.J.E.; Cretella, M.; Reno, P.H.; Daniel, F.B. Nitroaromatic Munition Compounds: Environmental Effects and Screening Values. In Reviews of Environmental Contamination and Toxicology; Ware, G.W., Ed.; Springer: New York, NY, USA, 1999; Volume 161, pp. 1–156. [Google Scholar]
- Harkonen, H.; Karki, M.; Lahti, A.; Savolainen, H. Early equatorial cataracts in workers exposed to trinitrotoluene. Am. J. Ophthalmol. 1983, 95, 807–810. [Google Scholar] [CrossRef]
- Hathaway, J.A. Trinitrotoluene: A review of reported dose-related effects providing documentation for a workplace standard. J. Occup. Med. 1977, 19, 341–345. [Google Scholar] [PubMed]
- Sabbioni, G.; Liu, Y.Y.; Yan, H.; Sepai, O. Hemoglobin adducts, urinary metabolites and health effects in 2,4,6-trinitrotoluene exposed workers. Carcinogenesis 2005, 26, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Shinkai, Y.; Li, S.; Kikuchi, T.; Kumagai, Y. Participation of metabolic activation of 2,4,6-trinitrotoluene to 4-hydroxylamino-2,6-dinitrotoluene in hematotoxicity. J. Toxicol. Sci. 2015, 40, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.S.; Furedi, E.M.; Gordon, D.E.; Lish, P.M.; Barkley, J.J. Subchronic toxicity of trinitrotoluene in fischer 344 rats. Toxicology 1984, 32, 253–265. [Google Scholar] [CrossRef]
- Crawford, M.A. Aplastic anaemia due to trinitrotoluene intoxication. Br. Med. J. 1954, 2, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Djerassi, L.S.; Vitany, L. Haemolytic episode in G6 PD deficient workers exposed to TNT. Br. J. Ind. Med. 1975, 32, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Robidoux, P.Y.; Svendsen, C.; Sarrazin, M.; Thiboutot, S.; Ampleman, G.; Hawari, J.; Weeks, J.M.; Sunahara, G.I. Assessment of a 2,4,6-trinitrotoluene-contaminated site using aporrectodea rosea and eisenia andrei in mesocosms. Arch. Environ. Contam. Toxicol. 2005, 48, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Leffler, P.; Brannas, E.; Ragnvaldsson, D.; Wingfors, H.; Berglind, R. Toxicity and accumulation of trinitrotoluene (TNT) and its metabolites in atlantic salmon alevins exposed to an industrially polluted water. J. Toxicol. Environ. Health A 2014, 77, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Talmage, S.S.; Opresko, D.M.; Maxwell, C.J.; Welsh, C.J.; Cretella, F.M.; Reno, P.H.; Daniel, F.B. Nitroaromatic munition compounds: Environmental effects and screening values. Rev. Environ. Contam. Toxicol. 1999, 161, 1–156. [Google Scholar] [PubMed]
- Robidoux, P.Y.; Hawari, J.; Thiboutot, S.; Ampleman, G.; Sunahara, G.I. Acute toxicity of 2,4,6-trinitrotoluene in earthworm (Eisenia andrei). Ecotoxicol. Environ. Saf. 1999, 44, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Dodard, S.G.; Renoux, A.Y.; Powlowski, J.; Sunahara, G.I. Lethal and subchronic effects of 2,4,6-trinitrotoluene (TNT) on enchytraeus albidus in spiked artificial soil. Ecotoxicol. Environ. Saf. 2003, 54, 131–138. [Google Scholar] [CrossRef]
- Kanekar, P.; Dautpure, P.; Sarnaik, S. Biodegradation of nitro-explosives. Indian J. Exp. Biol. 2003, 41, 991–1001. [Google Scholar] [PubMed]
- Corrales, J.; Kristofco, L.A.; Steele, W.B.; Saari, G.N.; Kostal, J.; Williams, E.S.; Mills, M.; Gallagher, E.P.; Kavanagh, T.J.; Simcox, N.; et al. Towards the design of less hazardous chemicals: Exploring comparative oxidative stress in two common animal models. Chem. Res. Toxicol. 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Olivares, C.I.; Sierra-Alvarez, R.; Abrell, L.; Chorover, J.; Simonich, M.; Tanguay, R.L.; Field, J.A. Zebrafish embryo toxicity of anaerobic biotransformation products from the insensitive munitions compound 2,4-dinitroanisole. Environ. Toxicol. Chem. 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Chardes, C.; Melenec, P.; Bertrand, V.; Lenne, P.F. Setting up a simple light sheet microscope for in toto imaging of C. elegans development. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
- Huisken, J.; Stainier, D.Y. Even fluorescence excitation by multidirectional selective plane illumination microscopy (mSPIM). Opt. Lett. 2007, 32, 2608–2610. [Google Scholar] [CrossRef] [PubMed]
- Keller, P.J.; Schmidt, A.D.; Wittbrodt, J.; Stelzer, E.H. Reconstruction of zebrafish early embryonic development by scanned light sheet microscopy. Science 2008, 322, 1065–1069. [Google Scholar] [CrossRef] [PubMed]
- Keller, P.J.; Schmidt, A.D.; Wittbrodt, J.; Stelzer, E.H. Digital scanned laser light-sheet fluorescence microscopy (DSLM) of zebrafish and Drosophila embryonic development. Cold Spring Harb. Protoc. 2011, 2011, 1235–1243. [Google Scholar] [CrossRef] [PubMed]
- Mertz, J.; Kim, J. Scanning light-sheet microscopy in the whole mouse brain with hilo background rejection. J. Biomed. Opt. 2010, 15, 016027. [Google Scholar] [CrossRef] [PubMed]
- Park, O.K.; Kwak, J.; Jung, Y.J.; Kim, Y.H.; Hong, H.S.; Hwang, B.J.; Kwon, S.H.; Kee, Y. 3D light-sheet fluorescence microscopy of cranial neurons and vasculature during zebrafish embryogenesis. Mol. Cells 2015, 38, 975–981. [Google Scholar] [PubMed]
- Santi, P.A.; Johnson, S.B.; Hillenbrand, M.; GrandPre, P.Z.; Glass, T.J.; Leger, J.R. Thin-sheet laser imaging microscopy for optical sectioning of thick tissues. Biotechniques 2009, 46, 287–294. [Google Scholar]
- Tomer, R.; Khairy, K.; Amat, F.; Keller, P.J. Quantitative high-speed imaging of entire developing embryos with simultaneous multiview light-sheet microscopy. Nat. Methods 2012, 9, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Truong, T.V.; Supatto, W.; Koos, D.S.; Choi, J.M.; Fraser, S.E. Deep and fast live imaging with two-photon scanned light-sheet microscopy. Nat. Methods 2011, 8, 757–760. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.; Huisken, J. Light sheet microscopy for real-time developmental biology. Curr. Opin. Genet. Dev. 2011, 21, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.J.; Tu, C.T.; Hsiao, C.D.; Hsieh, F.J.; Tsai, H.J. Germ-line transmission of a myocardium-specific gfp transgene reveals critical regulatory elements in the cardiac myosin light chain 2 promoter of zebrafish. Dev. Dyn. 2003, 228, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.H.; Li, F.P.; Zhang, H.P.; Jiang, Y.; Mao, L.C.; Wu, L.L.; Chen, L. Comparative analysis of water quality and toxicity assessment methods for urban highway runoff. Sci. Total Environ. 2016, 553, 519–523. [Google Scholar] [CrossRef] [PubMed]
- He, D.F.; Chen, R.R.; Zhu, E.H.; Chen, N.; Yang, B.; Shi, H.H.; Huang, M.S. Toxicity bioassays for water from black-odor rivers in Wenzhou, China. Environ. Sci. Pollut. Res. Int. 2015, 22, 1731–1741. [Google Scholar]
- Guarienti, M.; Gianoncelli, A.; Bontempi, E.; Moscoso Cardozo, S.; Borgese, L.; Zizioli, D.; Mitola, S.; Depero, L.E.; Presta, M. Biosafe inertization of municipal solid waste incinerator residues by cosmos technology. J. Hazard. Mater. 2014, 279, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Islam, A.M.T.; Kwak, J.; Jung, Y.J.; Kee, Y. Animal models of amyotrophic lateral sclerosis and Huntington′s disease. Genes Genom. 2014, 36, 399–413. [Google Scholar] [CrossRef]
- Raslan, A.A.; Kee, Y. Tackling neurodegenerative diseases: Animal models of alzheimer' disease and Parkinson’s disease. Genes Genom. 2013, 35, 425–440. [Google Scholar] [CrossRef]
- Wu, T.S.; Yang, J.J.; Yu, F.Y.; Liu, B.H. Cardiotoxicity of mycotoxin citrinin and involvement of microrna-138 in zebrafish embryos. Toxicol. Sci. 2013, 136, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, X.; Zhu, J.; Zhu, S.; Hu, X.; Zhu, C.; Guo, X.; Yu, Z.; Han, S. Toxic effects of polychlorinated biphenyls on cardiac development in zebrafish. Mol. Biol. Rep. 2014, 41, 7973–7983. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Yu, Y.; Li, Y.; Yu, Y.; Sun, Z. Cardiovascular toxicity evaluation of silica nanoparticles in endothelial cells and zebrafish model. Biomaterials 2013, 34, 5853–5862. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ball, J.; Panzica-Kelly, J.; Augustine-Rauch, K. In vitro developmental toxicology screens: A report on the progress of the methodology and future applications. Chem. Res. Toxicol. 2016, 29, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Stainier, D.Y.; Yelon, D. Screening mosaic F1 females for mutations affecting zebrafish heart induction and patterning. Dev. Genet. 1998, 22, 288–299. [Google Scholar] [CrossRef]
- Berdougo, E.; Coleman, H.; Lee, D.H.; Stainier, D.Y.; Yelon, D. Mutation of weak atrium/atrial myosin heavy chain disrupts atrial function and influences ventricular morphogenesis in zebrafish. Development 2003, 130, 6121–6129. [Google Scholar] [CrossRef] [PubMed]
- Rydeen, A.B.; Waxman, J.S. Cyp26 enzymes are required to balance the cardiac and vascular lineages within the anterior lateral plate mesoderm. Development 2014, 141, 1638–1648. [Google Scholar] [CrossRef] [PubMed]
- Paik, E.J.; Zon, L.I. Hematopoietic development in the zebrafish. Int. J. Dev. Biol. 2010, 54, 1127–1137. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; He, Q.; Ma, D.; Xue, Y.; Liu, F. Irf4 regulates the choice between T lymphoid-primed progenitor and myeloid lineage fates during embryogenesis. Dev. Cell 2015, 34, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Paden, N.E.; Smith, E.E.; Kendall, R.J. Acute toxicity of 2,4,6-trinitrotoluene, 2,4-dinitrotoluene, and 2,6-dinitrotoluene in the adult bullfrog (Lithobates catesbeiana). Bull. Environ. Contam. Toxicol. 2008, 80, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
- Lawson, N.D.; Vogel, A.M.; Weinstein, B.M. Sonic hedgehog and vascular endothelial growth factor act upstream of the notch pathway during arterial endothelial differentiation. Dev. Cell 2002, 3, 127–136. [Google Scholar] [CrossRef]
- Traver, D.; Paw, B.H.; Poss, K.D.; Penberthy, W.T.; Lin, S.; Zon, L.I. Transplantation and in vivo imaging of multilineage engraftment in zebrafish bloodless mutants. Nat. Immunol. 2003, 4, 1238–1246. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.; Park, O.K.; Jung, Y.J.; Hwang, B.J.; Kwon, S.H.; Kee, Y. Live image profiling of neural crest lineages in zebrafish transgenic lines. Mol. Cells 2013, 35, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative pcr and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eum, J.; Kwak, J.; Kim, H.J.; Ki, S.; Lee, K.; Raslan, A.A.; Park, O.K.; Chowdhury, M.A.U.; Her, S.; Kee, Y.; et al. 3D Visualization of Developmental Toxicity of 2,4,6-Trinitrotoluene in Zebrafish Embryogenesis Using Light-Sheet Microscopy. Int. J. Mol. Sci. 2016, 17, 1925. https://doi.org/10.3390/ijms17111925
Eum J, Kwak J, Kim HJ, Ki S, Lee K, Raslan AA, Park OK, Chowdhury MAU, Her S, Kee Y, et al. 3D Visualization of Developmental Toxicity of 2,4,6-Trinitrotoluene in Zebrafish Embryogenesis Using Light-Sheet Microscopy. International Journal of Molecular Sciences. 2016; 17(11):1925. https://doi.org/10.3390/ijms17111925
Chicago/Turabian StyleEum, Juneyong, Jina Kwak, Hee Joung Kim, Seoyoung Ki, Kooyeon Lee, Ahmed A. Raslan, Ok Kyu Park, Md Ashraf Uddin Chowdhury, Song Her, Yun Kee, and et al. 2016. "3D Visualization of Developmental Toxicity of 2,4,6-Trinitrotoluene in Zebrafish Embryogenesis Using Light-Sheet Microscopy" International Journal of Molecular Sciences 17, no. 11: 1925. https://doi.org/10.3390/ijms17111925
APA StyleEum, J., Kwak, J., Kim, H. J., Ki, S., Lee, K., Raslan, A. A., Park, O. K., Chowdhury, M. A. U., Her, S., Kee, Y., Kwon, S.-H., & Hwang, B. J. (2016). 3D Visualization of Developmental Toxicity of 2,4,6-Trinitrotoluene in Zebrafish Embryogenesis Using Light-Sheet Microscopy. International Journal of Molecular Sciences, 17(11), 1925. https://doi.org/10.3390/ijms17111925




