Unsaturated Lipids Change in Olive Tree Drupe and Seed during Fruit Development and in Response to Cold-Stress and Acclimation
Abstract
:1. Introduction
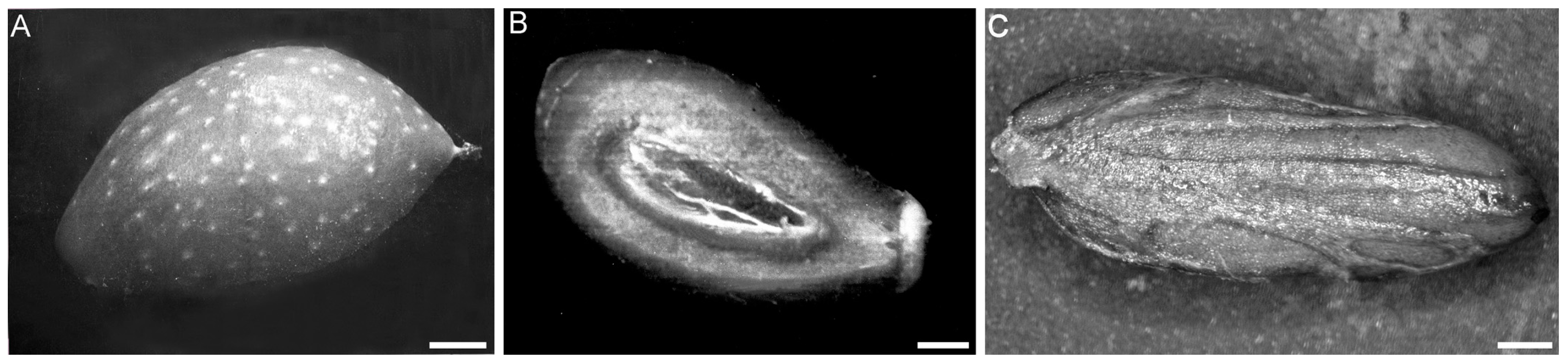
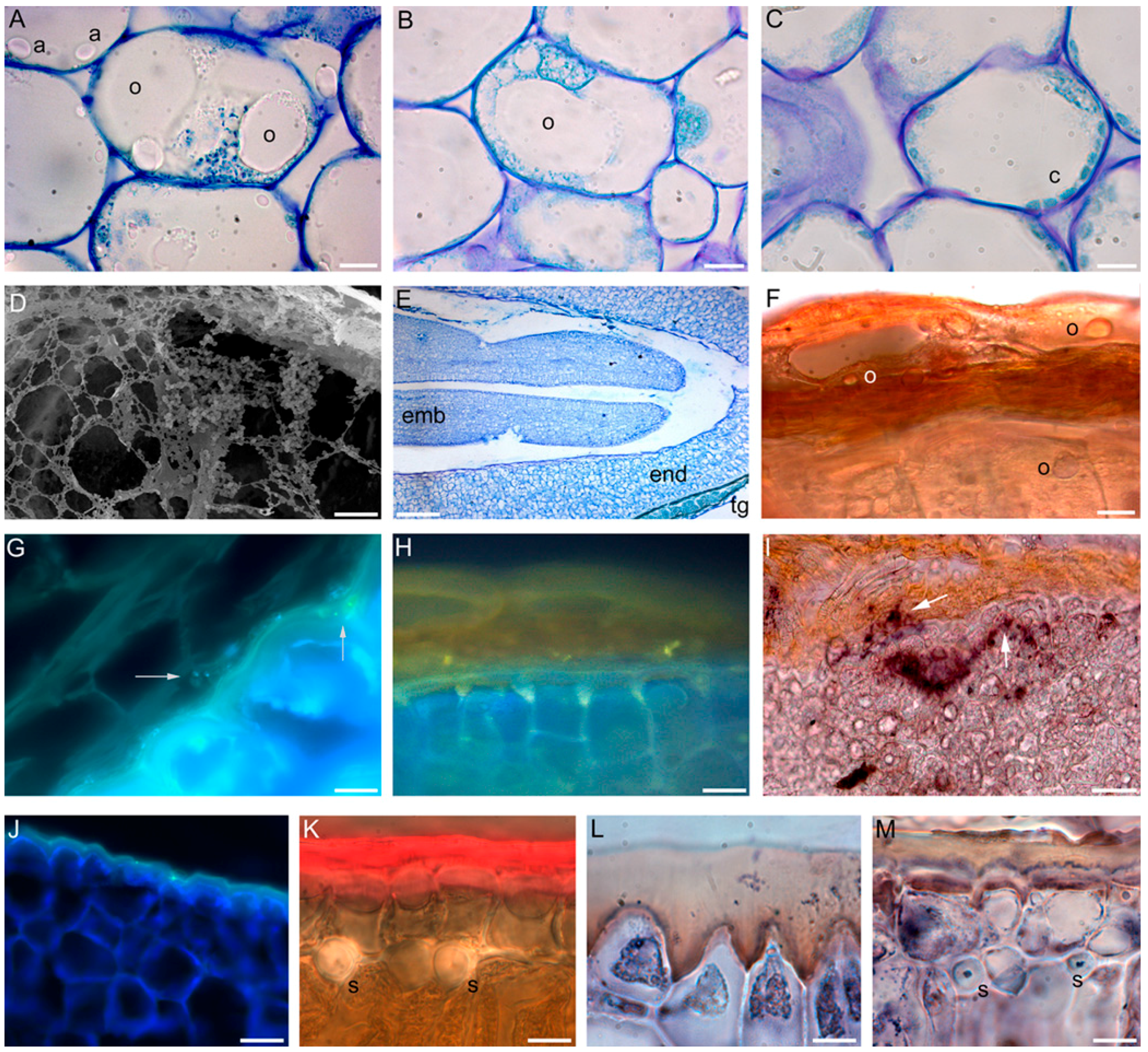
2. Oil Biogenesis in Olive Tree Drupe and Seed
3. Olive Oil and Fatty Acid Desaturation
4. FAD2.1, FAD2.2 and FAD7 Transcription Characterizes Oil Biogenesis during Drupe Development
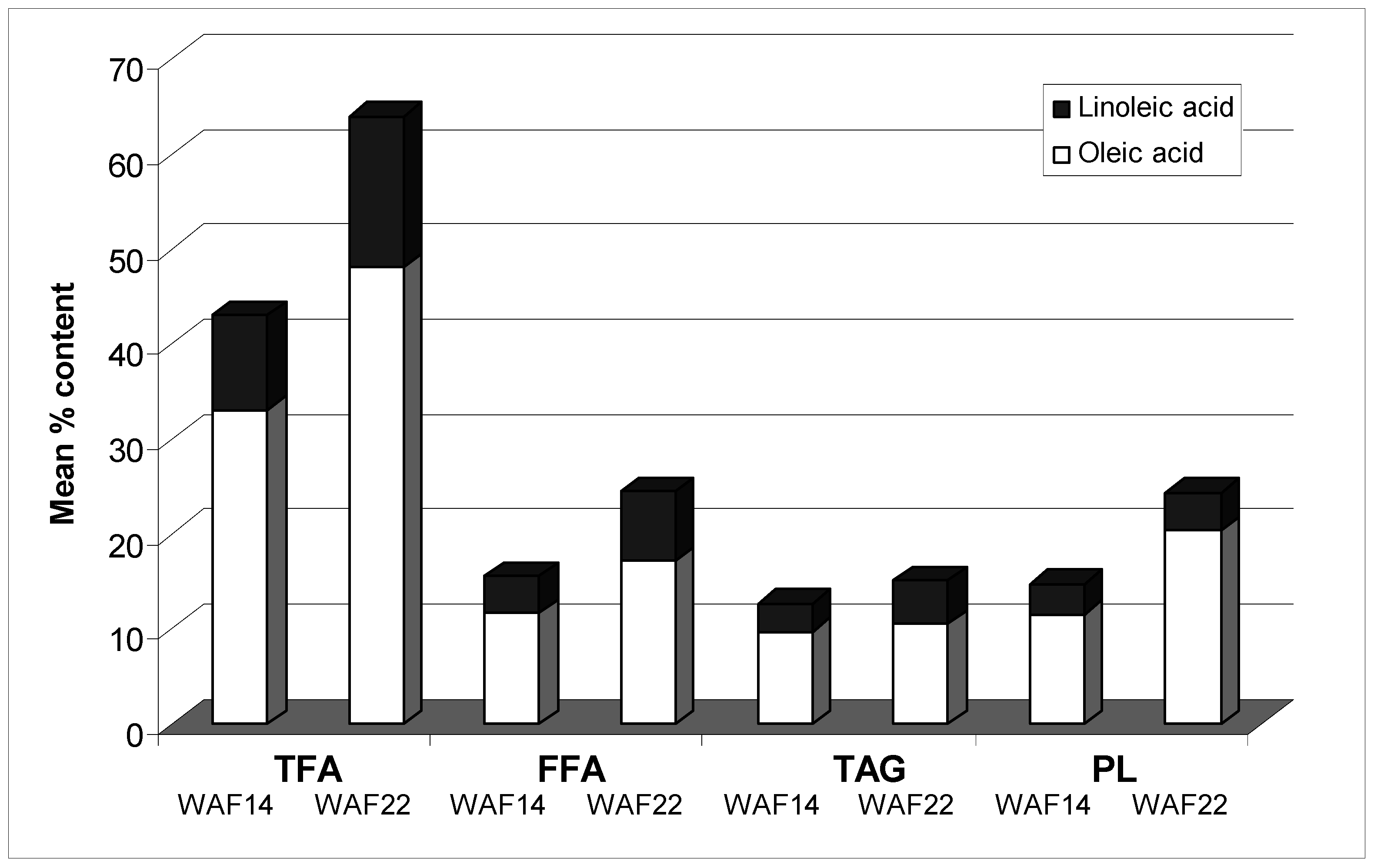
5. Cold Regulation of FAD2.2 and FAD7 Expression in the Drupe
6. OeFAD2.2 Transcription and C18:2-Content Increase in the Seed Coat of the Cold-Acclimated Drupe, and the Endosperm Cutinisation Increases Accordingly
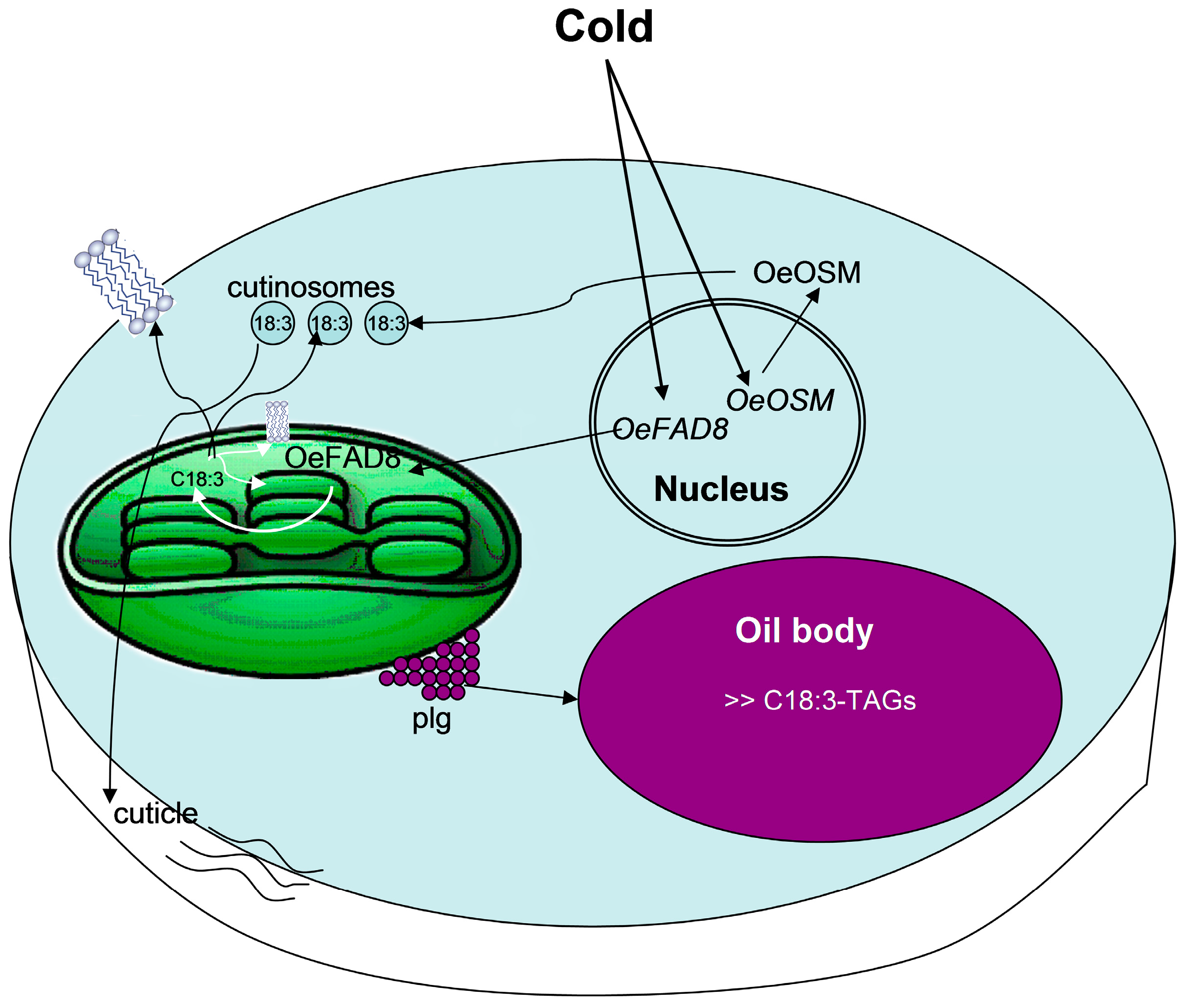
7. OeFAD8 Expression Is a Determinant for Cold-Acclimation in Drupes and Leaves, and Is Associated with Parallel Changes in C18:3-Lipids and in Cutinisation
8. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| OBs | Oil bodies |
| FA | Fatty acid |
| FAD | FA-desaturase |
| WAFs | Weeks after flowering |
| TAGs | Triacylglycerols |
| C18:1 | Oleic acid |
| C18:2 | ω6 linoleic acid |
| C18:3 | ω3-Linolenic acid |
| ER | Endoplasmic reticulum |
| FFA | Free fatty acid |
| PL | Polar lipid |
References
- Alagna, F.; D’Agostino, N.; Torchia, L.; Servili, M.; Rao, R.; Pietrella, M.; Giuliano, G.; Chiusano, M.L.; Baldoni, L.; Perrotta, G. Comparative 454 pyrosequencing of transcripts from two olive genotypes during fruit development. BMC Genom. 2009, 10, 399. [Google Scholar] [CrossRef] [PubMed]
- Hatzopoulos, P.; Banilas, G.; Giannoulia, K.; Gazis, F.; Nikoloudakis, N.; Milioni, D.; Haralampidis, K. Breeding, molecular markers and molecular biology of the olive tree. Eur. J. Lipid Sci. Technol. 2002, 104, 547–586. [Google Scholar] [CrossRef]
- Rugini, E.; Biasi, R.; Muleo, R. Olive (Olea europaea L. var. sativa) trasformation. In Molecular Biology of Woody Plants; Moahn, J.S., Minocha, S.C., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2000; pp. 245–279. [Google Scholar]
- Mannina, L.; Fontanazza, G.; Patumi, M.; Ansanelli, G.; Segre, A. Italian and Argentine olive oils: A NMR and gas chromatographic study. Grasas Y Aceites 2001, 52, 380–388. [Google Scholar] [CrossRef]
- Matteucci, M.; D’Angeli, S.; Errico, S.; Lamanna, R.; Perrotta, G.; Altamura, M.M. Cold affects the transcription of fatty acid desaturases and oil quality in the fruit of Olea europaea L. genotypes with different cold hardiness. J. Exp. Bot. 2011, 62, 3403–3420. [Google Scholar] [CrossRef] [PubMed]
- D’Angeli, S.; Malhò, R.; Altamura, M.M. Low-temperature sensing in olive tree: Calcium signalling and cold acclimation. Plant Sci. 2003, 165, 1303–1313. [Google Scholar] [CrossRef]
- D’Angeli, S.; Matteucci, M.; Fattorini, L.; Gismondi, A.; Ludovici, M.; Canini, A.; Altamura, M.M. OeFAD8, OeLIP and OeOSM expression and activity in cold-acclimation of Olea europaea, a perennial dicot without winter-dormancy. Planta 2016, 243, 1279–1296. [Google Scholar] [CrossRef] [PubMed]
- Green, P.S. A revision of Olea L. Kew Bull. 2002, 57, 91–140. [Google Scholar] [CrossRef]
- Morellò, J.; Motilva, M.; Ramo, T.; Romero, M. Effect of freeze injuries in olive fruit on virgin olive oil composition. Food Chem. 2003, 81, 547–553. [Google Scholar] [CrossRef]
- Bernardi, R.; Adamo, S.; Fontana, M.; Manzo, M.; Salvini, M.; Durante, M. Differential gene expression induced by cold stress in Olea europaea L. Acta Hortic. 2008, 791, 55–59. [Google Scholar] [CrossRef]
- Browse, J.; Xin, Z. Temperature sensing and cold acclimation. Curr. Opin. Plant Biol. 2001, 4, 241–246. [Google Scholar] [CrossRef]
- D’Angeli, S.; Altamura, M.M. Osmotin induces cold protection in olive tree by causing programmed cell death and affecting cytoskeleton organization. Planta 2007, 225, 1147–1163. [Google Scholar] [CrossRef] [PubMed]
- Thomashow, M. So what’s new in the field of plant cold acclimation? Lots! Plant Physiol. 2001, 125, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Knight, M.R.; Knight, H. Low temperature perception leading to gene expression and cold tolerance in higher plants. New Phytol. 2012, 195, 737–751. [Google Scholar] [CrossRef] [PubMed]
- Welling, A.; Palva, T. Molecular control of cold acclimation in trees. Physiol Plant. 2006, 127, 167–181. [Google Scholar] [CrossRef]
- Arora, R.; Wisniewski, M.E.; Scorza, R. Cold acclimation in genetically related (sibling) deciduous and evergreen peach (Prunus persica L. Batsch). I. Seasonal changes in cold hardiness and polypeptides of bark and xylem tissues. Plant Physiol. 1992, 99, 1562–1568. [Google Scholar] [CrossRef] [PubMed]
- D’Angeli, S.; Falasca, G.; Matteucci, M.; Altamura, M.M. Cold perception and gene expression differ in Olea europaea seed coat and embryo during drupe cold acclimation. New Phytol. 2013, 197, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, J. Lipid photosynthesis in olive fruit. Prog. Lipid Res. 1994, 33, 97–104. [Google Scholar] [CrossRef]
- Hernandez, M.L.; Padilla, M.N.; Mancha, M.; Martìnez-Rivas, J.M. Expression analysis identifies FAD2–2 as olive oleate desaturase gene mainly responsible for the linoleic acid content in virgin olive oil. J. Agric. Food. Chem. 2009, 57, 6199–6206. [Google Scholar] [CrossRef] [PubMed]
- Lavee, S.; Wodner, M. Factors affecting the nature of oil accumulation in fruit of olive (Olea europaea L.) cultivars. J. Hortic. Sci. 1991, 66, 583–591. [Google Scholar] [CrossRef]
- Conde, C.; Delrot, S.; Gero’s, H. Physiological, biochemical and molecular changes occurring during olive development and ripening. J. Plant Physiol. 2008, 165, 1545–1562. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, H.T. Growth of the olive fruit. Proc. Am. Soc. Hortic. Sci. 1949, 54, 86–94. [Google Scholar]
- Roca, M.; Mínguez-Mosquera, M.I. Changes in chloroplast pigments of olive varieties during fruit ripening. J. Agric. Food Chem. 2001, 49, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Salas, J.J.; Sànchez, J.; Ramli, U.S.; Manaf, A.M. Biochemistry of lipid metabolism in olive and other oil fruits. Prog. Lipid Res. 2000, 39, 151–180. [Google Scholar] [CrossRef]
- Ross, J.H.E.; Sanchez, J.; Millan, F.; Murphy, D.J. Differential presence of oleosins in oleogenic seed and mesocarp tissues in olive (Olea europaea) and avocado (Persea americana). Plant Sci. 1993, 93, 203–210. [Google Scholar] [CrossRef]
- Hernandez, M.L.; Sicardo, M.D.; Martınez-Rivas, J.M. Differential contribution of endoplasmic reticulum and chloroplast o-3 fatty acid desaturase genes to the linolenic acid content of olive (Olea europaea) fruit. Plant Cell Physiol. 2016, 57, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Parvini, F.; Zeinanloo, A.A.; Ebrahimie, E.; Tahmasebi-Enferadi, S.; Hosseini-Mazinani, M. Differential expression of fatty acid desaturases in Mari and Shengeh olive cultivars during fruit development and ripening. Eur. J. Lipid Sci. Technol. 2015, 117, 523–531. [Google Scholar] [CrossRef]
- Borisjuk, N.; Hrmova, M.; Lopato, S. Transcriptional regulation of cuticle biosynthesis. Biotechnol. Adv. 2014, 32, 526–540. [Google Scholar] [CrossRef] [PubMed]
- Finch-Savage, W.E.; Leubner-Metzger, G. Seed dormancy and the control of germination. New Phytol. 2006, 171, 501–523. [Google Scholar] [CrossRef] [PubMed]
- Alagna, F.; Mariotti, R.; Panara, F.; Caporali, S.; Urbani, S.; Veneziani, G.; Esposto, S.; Taticchi, A.; Rosati, A.; Rao, R.; et al. Olive phenolic compounds: metabolic and transcriptional profiling during fruit development. BMC Plant Biol. 2012, 12, 162. [Google Scholar] [CrossRef] [PubMed]
- Ursin, V.M. Modification of plant lipids for human health: Development of functional land-based ω-3 fatty acids. J. Nutr. 2003, 133, 4271–4274. [Google Scholar] [PubMed]
- Upchurch, R.G. Fatty acid unsaturation, mobilization, and regulation in the response of plants to stress. Biotechnol. Lett. 2008, 30, 967–977. [Google Scholar] [CrossRef] [PubMed]
- Miquel, M.; James, D., Jr.; Doonert, H.; Browse, J. Arabidopsis requires polyunsaturated lipids for low-temperature survival. Proc. Natl. Acad. Sci. USA 1993, 90, 6208–6212. [Google Scholar] [PubMed]
- Kodama, H.; Horiguchi, C.; Nishiuchi, T.; Nishimura, M.; Iba, K. Fatty acid desaturation during chilling acclimation is one of the factors involved in conferring low-temperature tolerance to young tobacco leaves. Plant Physiol. 1995, 107, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, O.; Sakamoto, H.; Hashimoto, T.; Iba, K. A temperature-sensitive mechanism that regulates post-translational stability of a plastidial ω-3 fatty acid desaturase (FAD8) in Arabidopsis leaf tissues. J. Biol. Chem. 2005, 280, 3597–3604. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ming, F.; Pittman, J.; Han, Y.; Hu, J.; Guo, B.; Shen, D. Characterization of a rice (Oryza sativa L.) gene encoding a temperature-dependent chloroplast ω-3 fatty acid desaturase. Biochem. Biophys. Res. Commun. 2006, 340, 1209–1216. [Google Scholar] [CrossRef] [PubMed]
- Wallis, J.G.; Browse, J. Mutants of Arabidopsis reveal many roles for membrane lipids. Prog. Lipid Res. 2002, 41, 254–278. [Google Scholar] [CrossRef]
- Hernandez, M.L.; Guschina, I.A.; Martìnez-Rivas, J.M.; Mancha, M.; Harwood, J.H. The utilization and desaturation of oleate and linoleate during glycerolipid biosinthesis in olive (Olea europaea L.) callus culture. J. Exp. Bot. 2008, 59, 2425–2435. [Google Scholar] [CrossRef] [PubMed]
- Banilas, G.; Moressis, A.; Nikiloudakis, N.; Hatzopoulos, P. Spatial and temporal expression of two distinc oleate desaturases from olive (Olea europaea L.). Plant Sci. 2005, 168, 547–555. [Google Scholar] [CrossRef]
- Hernandez, M.L.; Mancha, M.; Martìnez-Rivas, J.M. Molecular cloning and characterization of genes encoding two microsomal oleate desaturases (FAD2) from olive. Phytochemistry 2005, 66, 1417–1426. [Google Scholar] [CrossRef] [PubMed]
- Banilas, G.; Nikiforiadis, A.; Makariti, I.; Moressis, A.; Hatzopoulos, P. Discrete roles of a microsomal linoleate desaturase gene in olive identified by spatiotemporal transcriptional analysis. Tree Physiol. 2007, 27, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Poghosyan, Z.; Haralampidis, K.; Martsinkovskaya, A.; Murphy, D.; Hatzopoulos, P. Developmental regulation and spatial expression of a plastidial fatty acid desaturase from Olea europaea. Plant Physiol Biochem. 1999, 37, 109–119. [Google Scholar] [CrossRef]
- Sabetta, W.; Blanco, A.; Zelasco, S.; Lombardo, L.; Perri, E.; Mangini, G.; Montemurro, C. FAD7 gene identification and fatty acids phenotypic variation in an olive collection by EcoTILLING and sequencing approaches. Plant Physiol. Biochem. 2013, 69, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cruz, F.; Julca, I.; Gómez-Garrido, J.; Loska, D.; Marcet-Houben, M.; Cano, E.; Galán, B.; Frias, L.; Ribeca, P.; Derdak, S.; et al. Genome sequence of the olive tree, Olea europaea. GigaScience 2016, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, M.L.; Padilla, M.N.; Sicardo, M.D.; Mancha, M.; Martinez-Rivas, J.M. Effect of different environmental stresses on the expression of oleate desaturase genes and fatty acid composition in olive fruit. Phytochemistry 2011, 72, 178–187. [Google Scholar] [CrossRef] [PubMed]
- De la O Leyva-Pérez, M.; Valverde-Corredor, A.; Valderrama, R.; Jiménez-Ruiz, J.; Muñioz-Merida, A.; Trelles, O.; Barroso, J.B.; Mercado-Blanco, J.; Luque, F. Early and delayed long-term transcriptional changes and short-term transient responses during cold acclimation in olive leaves. DNA Res. 2015, 22, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, R.; Kaimainen, M.; Kallio, H. Cutin composition of selected northern berries and seeds. Food Chem. 2010, 122, 137–144. [Google Scholar] [CrossRef]
- Newton, S.; Duman, J.G. An osmotin-like cryoprotective protein from bittersweet nightshade Solanum dulcamara. Plant Mol. Biol. 2000, 44, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Yeats, T.; Howe, K.; Matas, A.; Buda, G.; Thannhauser, T.; Rose, J. Mining the surface proteome of tomato (Solanum lycopersicum) fruit for proteins associated with cuticle biogenesis. J. Exp. Bot. 2010, 61, 3759–3771. [Google Scholar] [CrossRef] [PubMed]
- Heredia-Guerrero, J.A.; Benítez, J.J.; Heredia, A. Self-assembled polyhydroxy fatty acids vesicles: A mechanism for plant cutin synthesis. BioEssays 2008, 30, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.; Rose, J. There’s more than one way to skin a fruit: formation and functions of fruit cuticles. J. Exp. Bot. 2014, 65, 4639–4651. [Google Scholar] [CrossRef] [PubMed]
- Bacelar, E.; Correia, C.; Moutinho-Pereira, J.; Gonçalves, B.; Lopes, J.; Torres-Pereira, J. Sclerophylly and leaf anatomical traits of five field-grown olive cultivars growing under drought conditions. Tree Physiol. 2004, 24, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, S.M.; Jenks, M.A. Plant cuticle function as a barrier to water loss. In Plant Abiotic Stress; Jenks, M.A., Hasegawa, P.M., Eds.; Blackwell Publishing: Oxford, UK, 2005; pp. 14–36. [Google Scholar]
- Gomes, S.; Bacelar, E.; Martins-Lopes, P.; Carvalho, T.; Guedes-Pinto, H. Infection process of olive fruits by Colletotrichum acutatum and the protective role of the cuticle and epidermis. J. Agric. Sci. 2012, 4, 101–110. [Google Scholar] [CrossRef]
- Degenkolbe, T.; Giavalisco, P.; Zuther, E.; Seiwert, B.; Hincha, D.K.; Willmitzer, L. Differential remodeling of the lipidome during cold acclimation in natural accessions of Arabidopsis thaliana. Plant J. 2012, 72, 972–982. [Google Scholar] [CrossRef] [PubMed]
- Szymanski, J.; Brotman, Y.; Willmitzer, L.; Cuadros-Inostroza, Á. Linking gene expression and membrane lipid composition of Arabidopsis. Plant Cell 2014, 26, 915–928. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, T.; Hernández, M.; Pennycooke, J.; Jiménez, P.; Martínez-Rivas, J.; Sanz, C.; Stockinger, E.; Sánchez-Serrano, J.; Sanmartín, M. Increasing ω-3 desaturase expression in tomato results in altered aroma profile and enhanced resistance to cold stress. Plant Physiol. 2010, 153, 655–665. [Google Scholar] [CrossRef] [PubMed]
- Pollard, M.; Beisson, F.; Li, Y.; Ohlrogge, J.B. Building lipid barriers: biosynthesis of cutin and suberin. Trends Plant Sci. 2008, 13, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Rani, S.H.; Krishna, T.H.A.; Saha, S.; Negi, A.S.; Rajasekharan, R. Defective in cuticular ridges (DCR) of Arabidopsis thaliana, a gene associated with surface cutin formation, encodes a soluble diacylglycerol acyltransferase. J. Biol. Chem. 2010, 285, 38337–38347. [Google Scholar] [CrossRef] [PubMed]
- Burgos, A.; Szymanski, J.; Seiwert, B.; Degenkolbe, T.; Hannah, M.A.; Giavalisco, P.; Willmitzer, L. Analysis of short-term changes in the Arabidopsis thaliana glycerolipidome in response to temperature and light. Plant J. 2011, 66, 656–668. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Angeli, S.; Altamura, M.M. Unsaturated Lipids Change in Olive Tree Drupe and Seed during Fruit Development and in Response to Cold-Stress and Acclimation. Int. J. Mol. Sci. 2016, 17, 1889. https://doi.org/10.3390/ijms17111889
D’Angeli S, Altamura MM. Unsaturated Lipids Change in Olive Tree Drupe and Seed during Fruit Development and in Response to Cold-Stress and Acclimation. International Journal of Molecular Sciences. 2016; 17(11):1889. https://doi.org/10.3390/ijms17111889
Chicago/Turabian StyleD’Angeli, Simone, and Maria Maddalena Altamura. 2016. "Unsaturated Lipids Change in Olive Tree Drupe and Seed during Fruit Development and in Response to Cold-Stress and Acclimation" International Journal of Molecular Sciences 17, no. 11: 1889. https://doi.org/10.3390/ijms17111889
APA StyleD’Angeli, S., & Altamura, M. M. (2016). Unsaturated Lipids Change in Olive Tree Drupe and Seed during Fruit Development and in Response to Cold-Stress and Acclimation. International Journal of Molecular Sciences, 17(11), 1889. https://doi.org/10.3390/ijms17111889







