Programmed Effects in Neurobehavior and Antioxidative Physiology in Zebrafish Embryonically Exposed to Cadmium: Observations and Hypothesized Adverse Outcome Pathway Framework
Abstract
:1. Introduction
2. Results
2.1. Embryotoxicity
2.2. DNA Methylation
2.3. DNA Oxidation
2.4. Neurobehavioral Studies
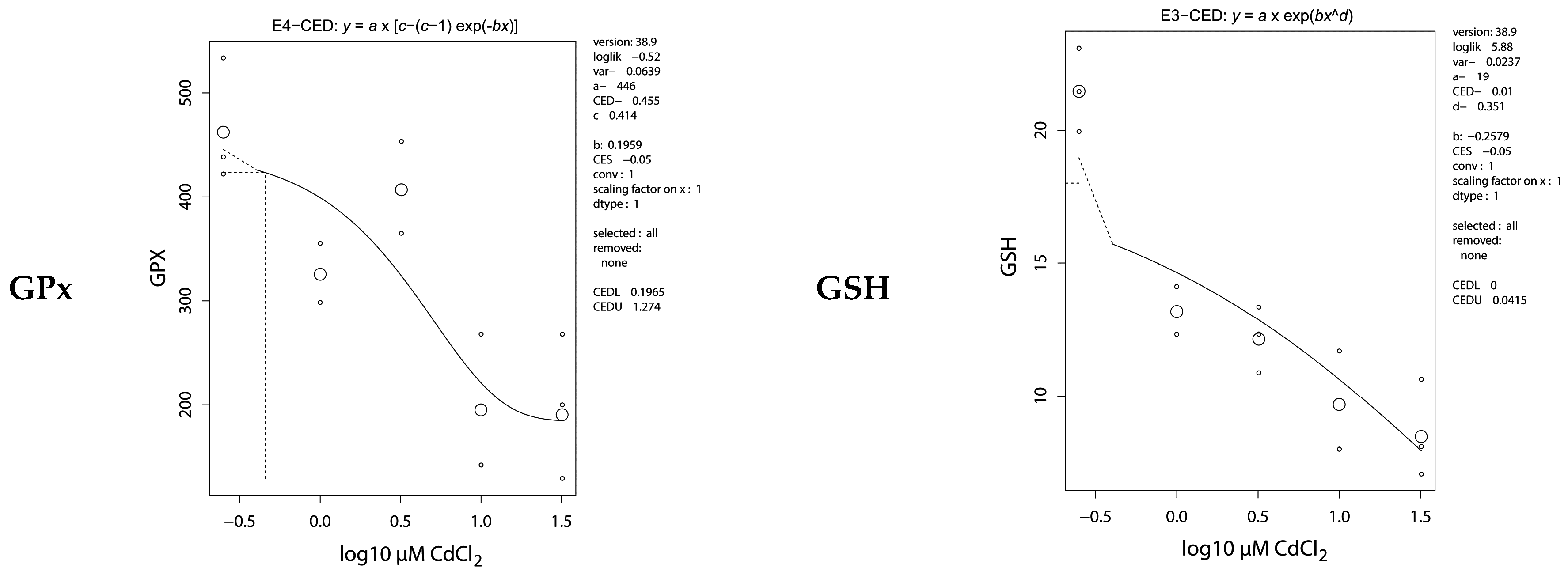
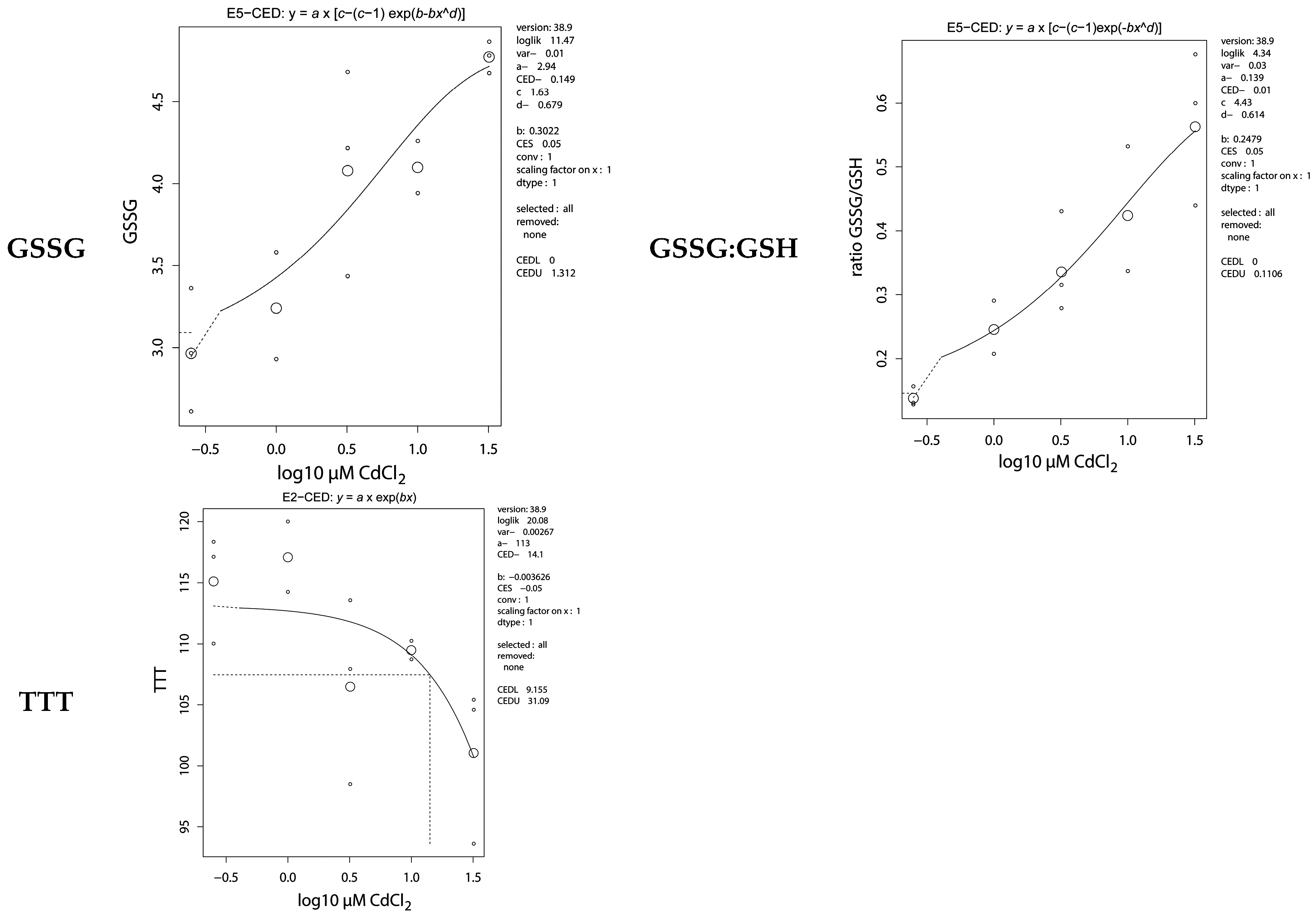
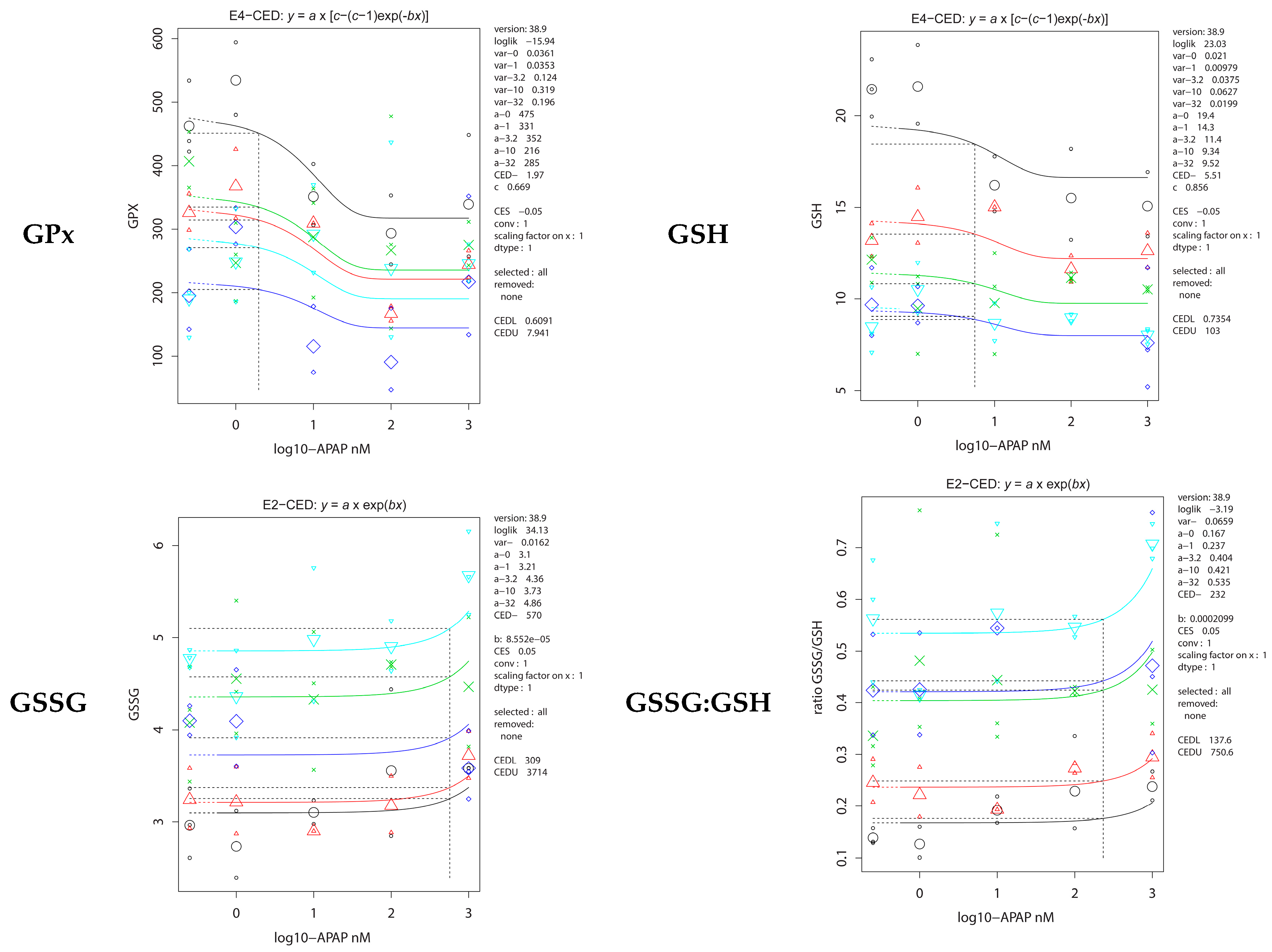
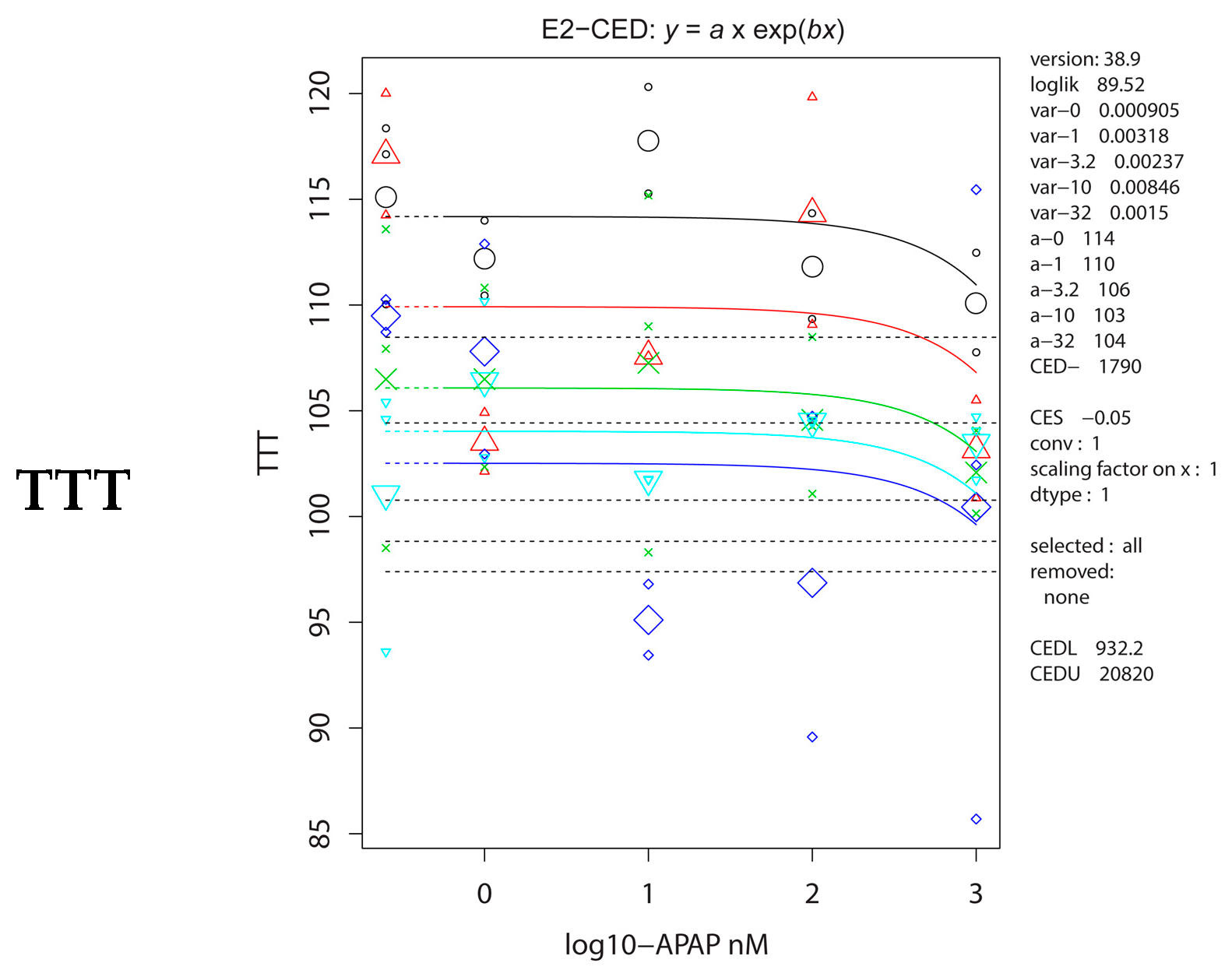
2.5. Antioxidative Parameters
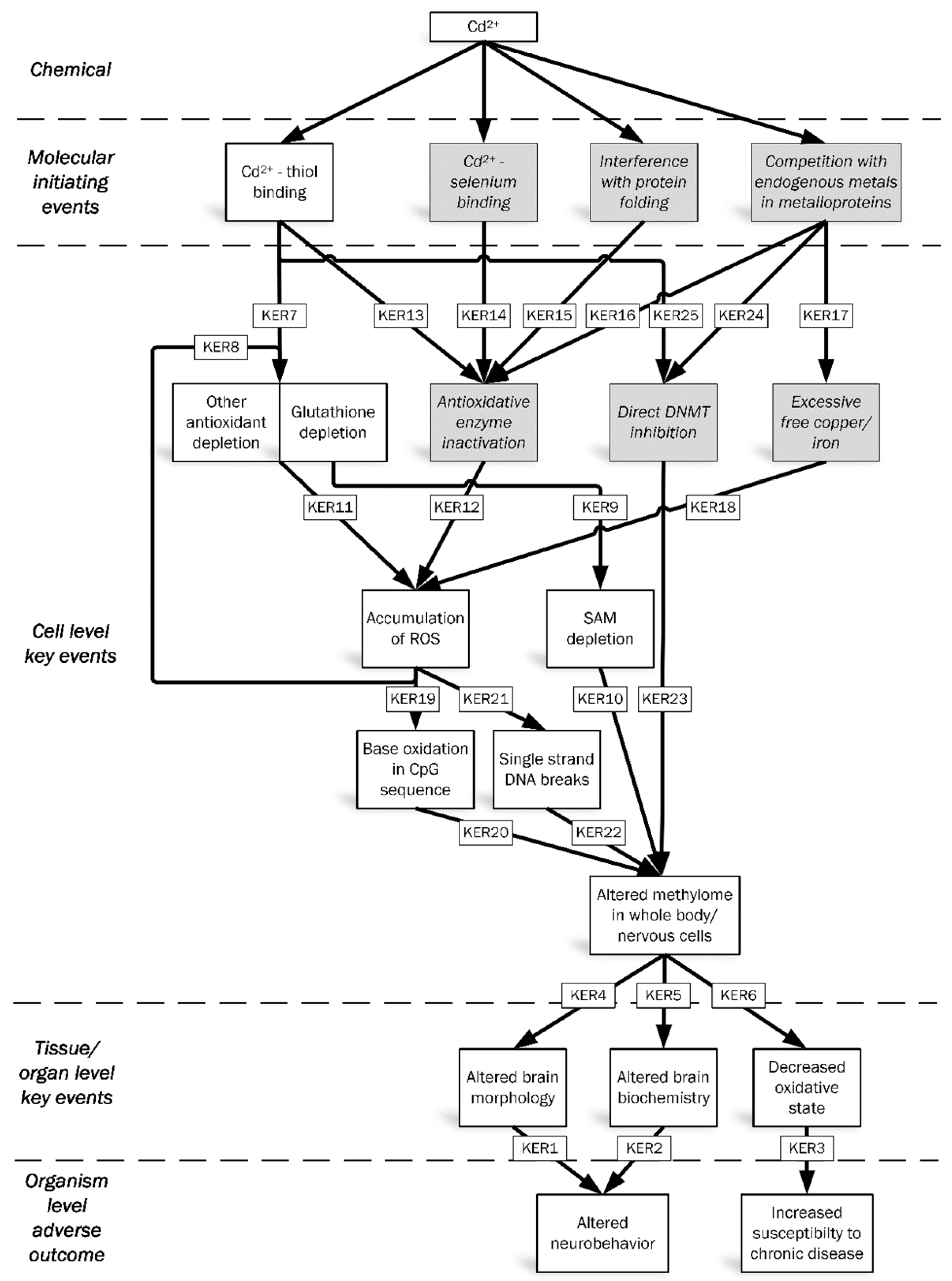
3. Discussion and Hypothesized AOP Framework
3.1. Neurobehavior
3.2. Oxidative Stress
3.3. Epigenetics
3.4. Mechanisms Leading to Methylome Alteration
3.5. Methylome Alteration via Oxidative DNA Damage
3.6. Informative Key Events (KEs)
4. Materials and Methods
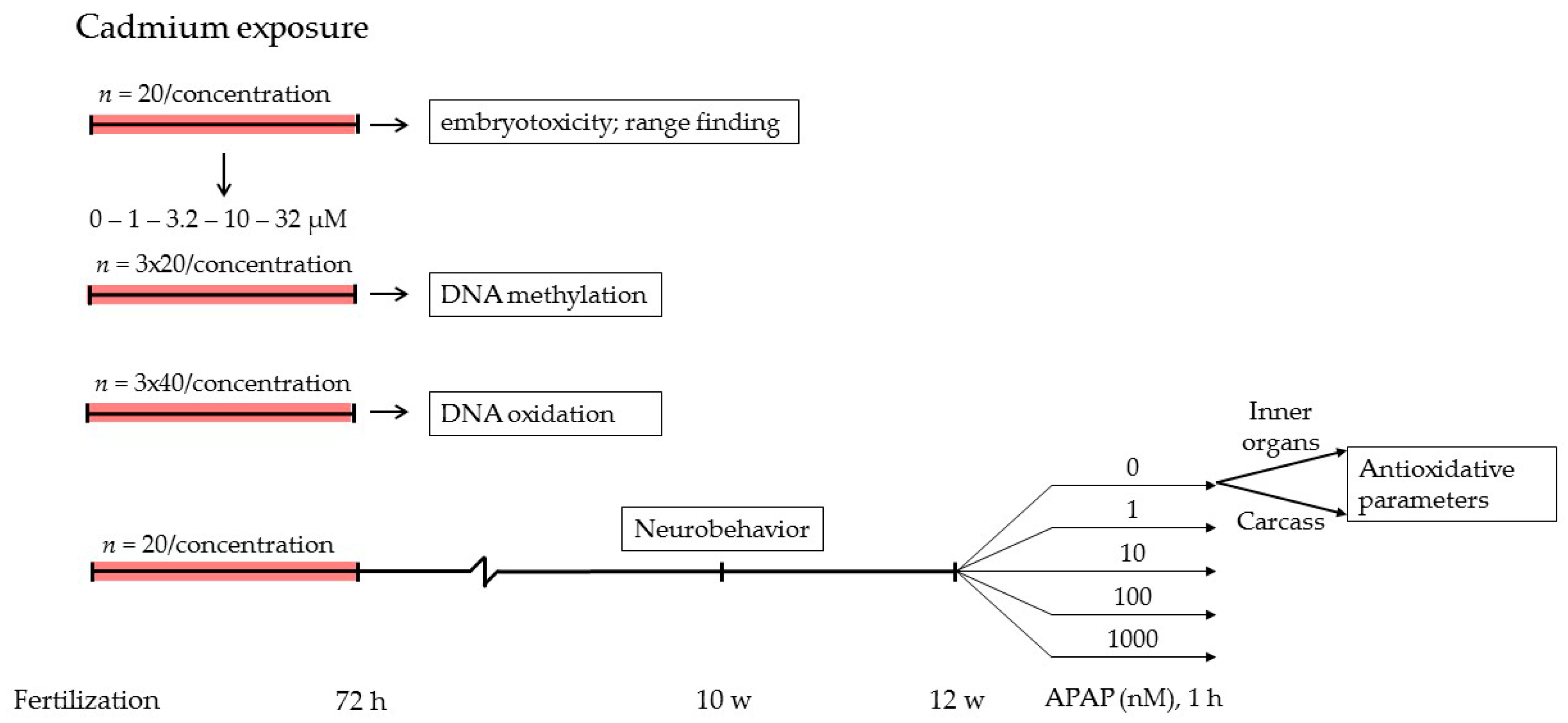
4.1. General Zebrafish Maintenance and Embryo Exposures
4.2. DNA Methylation
4.3. DNA Oxidation
4.4. Neurobehavior
4.5. Oxidative Stress Defense
4.6. Statistical Analysis
4.7. Hypothesized Adverse Outcome Pathway (AOP) Framework Development
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| NCD | Non-communicable disease |
| DOHaD | Developmental origins of health and disease |
| SAM | S-adenosyl-l-methionine |
| DNMT | DNA methyl transferase |
| AOP | Adverse outcome pathway |
| MIE | Molecular initiating event |
| KE | Key event |
| KER | Key event relationship |
| dpf | Days post fertilization |
| GSH | Reduced glutathione |
| GSSG | Oxidized glutathione |
| GPx | Glutathione peroxidase |
| GR | Glutathione reductase |
| SOD | Superoxide dismutase |
| TTT | Total thiols |
| BAP | Biological antioxidant potential |
| ROS | Reactive oxygen species |
| APAP | Acetaminophen, paracetamol |
| CED | Critical effect dose |
| 8-OHdG | 8-Hydroxyl-2′-deoxyguanosine |
References
- Bloom, D.E.; Cafiero, E.T.; Jané-Llopis, E.; Abrahams-Gessel, S.; Bloom, L.R.; Fathima, S.; Feigl, A.B.; Gaziano, T.; Mowafi, M.; Pandya, A.; et al. The Global Economic Burden of Noncommunicable Diseases; World Economic Forum: Cologny, Switzerland, 2011. [Google Scholar]
- Balbus, J.M.; Barouki, R.; Birnbaum, L.S.; Etzel, R.A.; Gluckman, P.D.; Grandjean, P.; Hancock, C.; Hanson, M.A.; Heindel, J.J.; Hoffman, K.; et al. Early-life prevention of non-communicable diseases. Lancet 2013, 381, 3–4. [Google Scholar] [CrossRef]
- Godfrey, K.M.; Barker, D.J. Fetal programming and adult health. Public Health Nutr. 2001, 4, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Gruenbaum, Y.; Stein, R.; Cedar, H.; Razin, A. Methylation of cpg sequences in eukaryotic DNA. FEBS Lett. 1981, 124, 67–71. [Google Scholar] [CrossRef]
- Greally, J.M.; Jacobs, M.N. In vitro and in vivo testing methods of epigenomic endpoints for evaluating endocrine disruptors. Altex 2013, 30, 445–471. [Google Scholar] [CrossRef] [PubMed]
- Alyea, R.A.; Moore, N.P.; LeBaron, M.J.; Gollapudi, B.B.; Rasoulpour, R.J. Is the current product safety assessment paradigm protective for epigenetic mechanisms? J. Pharmacol. Toxicol. Methods 2012, 66, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Bouwmeester, M.C.; Ruiter, S.; Lommelaars, T.; Sippel, J.; Hodemaekers, H.M.; van den Brandhof, E.J.; Pennings, J.L.; Kamstra, J.H.; Jelinek, J.; Issa, J.J.; et al. Zebrafish embryos as a screen for DNA methylation modifications after compound exposure. Toxicol. Appl. Pharmacol. 2016, 291, 84–86. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, Y.; Shao, C.; Tan, Y.; Cai, L. Cadmium and its epigenetic effects. Curr. Med. Chem. 2012, 19, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Jomova, K.; Rhodes, C.J.; Kuca, K.; Musilek, K. Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch. Toxicol. 2016, 90, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Garrido, M.; Terron, M.P.; Rodriguez, A.B. Chrononutrition against oxidative stress in aging. Oxid. Med. Cell. Longev. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Stepniewska, J.; Golembiewska, E.; Dolegowska, B.; Domanski, M.; Ciechanowski, K. Oxidative stress and antioxidative enzyme activities in chronic kidney disease and different types of renal replacement therapy. Curr. Protein Pept. Sci. 2015, 16, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Fox, D.A.; Grandjean, P.; de Groot, D.; Paule, M.G. Developmental origins of adult diseases and neurotoxicity: Epidemiological and experimental studies. Neurotoxicology 2012, 33, 810–816. [Google Scholar] [CrossRef] [PubMed]
- Organisation for Economic Co-operation and Development (OECD). User’s Handbook Supplement to the Guidance Document for Developing and Assessing Aops; OECD: Paris, France, 2014. [Google Scholar]
- Groh, K.J.; Carvalho, R.N.; Chipman, J.K.; Denslow, N.D.; Halder, M.; Murphy, C.A.; Roelofs, D.; Rolaki, A.; Schirmer, K.; Watanabe, K.H. Development and application of the adverse outcome pathway framework for understanding and predicting chronic toxicity: II. A focus on growth impairment in fish. Chemosphere 2015, 120, 778–792. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Landrigan, P.J. Neurobehavioural effects of developmental toxicity. Lancet Neurol. 2014, 13, 330–338. [Google Scholar] [CrossRef]
- Jin, Y.; Liu, Z.; Liu, F.; Ye, Y.; Peng, T.; Fu, Z. Embryonic exposure to cadmium (ii) and chromium (vi) induce behavioral alterations, oxidative stress and immunotoxicity in zebrafish (Danio rerio). Neurotoxicol. Teratol. 2015, 48, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Kalueff, A.V.; Gebhardt, M.; Stewart, A.M.; Cachat, J.M.; Brimmer, M.; Chawla, J.S.; Craddock, C.; Kyzar, E.J.; Roth, A.; Landsman, S.; et al. Towards a comprehensive catalog of zebrafish behavior 1.0 and beyond. Zebrafish 2013, 10, 70–86. [Google Scholar] [CrossRef] [PubMed]
- Mathur, P.; Guo, S. Differences of acute versus chronic ethanol exposure on anxiety-like behavioral responses in zebrafish. Behav. Brain Res. 2011, 219, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Kusch, R.C.; Krone, P.H.; Chivers, D.P. Chronic exposure to low concentrations of waterborne cadmium during embryonic and larval development results in the long-term hindrance of antipredator behavior in zebrafish. Environ. Toxicol. Chem. 2008, 27, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Antonio, M.T.; Benito, M.J.; Leret, M.L.; Corpas, I. Gestational administration of cadmium alters the neurotransmitter levels in newborn rat brains. J. Appl. Toxicol. 1998, 18, 83–88. [Google Scholar] [CrossRef]
- Glazer, L.; Hahn, M.E.; Aluru, N. Delayed effects of developmental exposure to low levels of the aryl hydrocarbon receptor agonist 3,3′,4,4′,5-pentachlorobiphenyl (PCB126) on adult zebrafish behavior. Neurotoxicology 2016, 52, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, Y.; Rampersad, M.; Gerlai, R. Impairment of social behaviour persists two years after embryonic alcohol exposure in zebrafish: A model of fetal alcohol spectrum disorders. Behav. Brain Res. 2015, 292, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, Y.; Rampersad, M.; Gerlai, R. Embryonic alcohol exposure impairs the dopaminergic system and social behavioral responses in adult zebrafish. Int. J. Neuropsychopharmacol. 2015, 18. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Redox potential of gsh/gssg couple: Assay and biological significance. Methods Enzymol. 2002, 348, 93–112. [Google Scholar] [PubMed]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Du, Y. Cadmium and its neurotoxic effects. Oxid. Med. Cell. Longev. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Stangenberg, S.; Nguyen, L.T.; Chen, H.; Al-Odat, I.; Killingsworth, M.C.; Gosnell, M.E.; Anwer, A.G.; Goldys, E.M.; Pollock, C.A.; Saad, S. Oxidative stress, mitochondrial perturbations and fetal programming of renal disease induced by maternal smoking. Int. J. Biochem. Cell Biol. 2015, 64, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Baxi, D.B.; Singh, P.K.; Vachhrajani, K.D.; Ramachandran, A.V. Plasticity changes in adult metabolic homeostasis and tissue oxidative stress: Neonatal programming by corticosterone and melatonin as deprogrammer. J. Matern. Fetal Neonatal Med. 2012, 25, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, N.B.; Hennington, B.S.; Williamson, D.T.; Hill, M.L.; Betson, N.E.; Sartori-Valinotti, J.C.; Reckelhoff, J.F.; Royals, T.P.; Alexander, B.T. Oxidative stress contributes to sex differences in blood pressure in adult growth-restricted offspring. Hypertension 2012, 60, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.A.; Gluckman, P.D. Developmental origins of health and disease—global public health implications. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.D.; Yosim, A.; Fry, R.C. Incorporating epigenetic data into the risk assessment process for the toxic metals arsenic, cadmium, chromium, lead, and mercury: Strategies and challenges. Front. Genet. 2014. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.H.; Lei, Y.X.; Wang, C.X. Analysis of aberrant methylation in DNA repair genes during malignant transformation of human bronchial epithelial cells induced by cadmium. Toxicol. Sci. 2012, 125, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Ronco, A.M.; Llaguno, E.; Epunan, M.J.; Llanos, M.N. Effect of cadmium on cortisol production and 11beta-hydroxysteroid dehydrogenase 2 expression by cultured human choriocarcinoma cells (JEG-3). Toxicol. In Vitro 2010, 24, 1532–1537. [Google Scholar] [CrossRef] [PubMed]
- Benbrahim-Tallaa, L.; Waterland, R.A.; Dill, A.L.; Webber, M.M.; Waalkes, M.P. Tumor suppressor gene inactivation during cadmium-induced malignant transformation of human prostate cells correlates with overexpression of de novo DNA methyltransferase. Environ. Health Perspect. 2007, 115, 1454–1459. [Google Scholar] [PubMed]
- Yara, S.; Lavoie, J.C.; Beaulieu, J.F.; Delvin, E.; Amre, D.; Marcil, V.; Seidman, E.; Levy, E. Iron-ascorbate-mediated lipid peroxidation causes epigenetic changes in the antioxidant defense in intestinal epithelial cells: Impact on inflammation. PLoS ONE 2013, 8, e63456. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Chen, F.; Chang, F.; Bai, Y.; Chen, L. Persistent overexpression of DNA methyltransferase 1 attenuating gabaergic inhibition in basolateral amygdala accounts for anxiety in rat offspring exposed perinatally to low-dose bisphenol a. J. Psychiatr. Res. 2013, 47, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Somji, S.; Garrett, S.H.; Toni, C.; Zhou, X.D.; Zheng, Y.; Ajjimaporn, A.; Sens, M.A.; Sens, D.A. Differences in the epigenetic regulation of mt-3 gene expression between parental and Cd2+ or As3+ transformed human urothelial cells. Cancer Cell Int. 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Liu, Y.; Xie, C.; Tu, W.; Xia, Y.; Costa, M.; Zhou, X. Cadmium induces histone H3 lysine methylation by inhibiting histone demethylase activity. Toxicol. Sci. 2015, 145, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, M.; Urani, C.; Sacco, M.G.; Procaccianti, C.; Gribaldo, L. Whole genome analysis and micrornas regulation in hepg2 cells exposed to cadmium. Altex 2012, 29, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Kloypan, C.; Srisa-Art, M.; Mutirangura, A.; Boonla, C. Line-1 hypomethylation induced by reactive oxygen species is mediated via depletion of S-adenosylmethionine. Cell Biochem. Funct. 2015, 33, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Yamaguchi, Y.; Kasai, H. Inhibition of 8-hydroxyguanine repair in testes after administration of cadmium chloride to gsh-depleted rats. Toxicol. Appl. Pharmacol. 1997, 147, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Timme-Laragy, A.R.; Goldstone, J.V.; Imhoff, B.R.; Stegeman, J.J.; Hahn, M.E.; Hansen, J.M. Glutathione redox dynamics and expression of glutathione-related genes in the developing embryo. Free Radic. Biol. Med. 2013, 65, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Ercal, N.; Gurer-Orhan, H.; Aykin-Burns, N. Toxic metals and oxidative stress part I: Mechanisms involved in metal-induced oxidative damage. Curr. Top. Med. Chem. 2001, 1, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Yang, J.M.; Zhang, F.; Miao, P.; Lin, Y.; Chen, M.L. Individual and joint toxic effects of cadmium sulfate and α-naphthoflavone on the development of zebrafish embryo. J. Zhejiang Univ. Sci. B 2014, 15, 766–775. [Google Scholar] [CrossRef] [PubMed]
- Lopez, E.; Arce, C.; Oset-Gasque, M.J.; Canadas, S.; Gonzalez, M.P. Cadmium induces reactive oxygen species generation and lipid peroxidation in cortical neurons in culture. Free Radic. Biol. Med. 2006, 40, 940–951. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Sui, Y.; Wang, X.; Luo, Y.; Ji, L. Hydroxyl radical production and oxidative damage induced by cadmium and naphthalene in liver of carassius auratus. Comp. Biochem. Physiol. Toxicol. Pharmacol. 2005, 140, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, H.; Zhang, T.; Zhang, R.; Liu, R.; Chen, Y. Molecular mechanism on cadmium-induced activity changes of catalase and superoxide dismutase. Int. J. Biol. Macromol. 2015, 77, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Quig, D. Cysteine metabolism and metal toxicity. Altern. Med. Rev. 1998, 3, 262–270. [Google Scholar] [PubMed]
- Rani, A.; Kumar, A.; Lal, A.; Pant, M. Cellular mechanisms of cadmium-induced toxicity: A review. Int. J. Environ. Health Res. 2014, 24, 378–399. [Google Scholar] [CrossRef] [PubMed]
- Zachara, B.A. Selenium and selenium-dependent antioxidants in chronic kidney disease. Adv. Clin. Chem. 2015, 68, 131–151. [Google Scholar] [PubMed]
- Franco, R.; Schoneveld, O.; Georgakilas, A.G.; Panayiotidis, M.I. Oxidative stress, DNA methylation and carcinogenesis. Cancer Lett. 2008, 266, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Turk, P.W.; Laayoun, A.; Smith, S.S.; Weitzman, S.A. DNA adduct 8-hydroxyl-2′-deoxyguanosine (8-hydroxyguanine) affects function of human DNA methyltransferase. Carcinogenesis 1995, 16, 1253–1255. [Google Scholar] [CrossRef] [PubMed]
- Tan, N.W.; Li, B.F. Interaction of oligonucleotides containing 6-O-methylguanine with human DNA (cytosine-5-)-methyltransferase. Biochemistry 1990, 29, 9234–9240. [Google Scholar] [CrossRef] [PubMed]
- Watjen, W.; Beyersmann, D. Cadmium-induced apoptosis in c6 glioma cells: Influence of oxidative stress. Biometals 2004, 17, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, N.; Nakashima, H.; Hosoda, K.; Eitaki, Y.; Shimada, N.; Omae, K. Urinary excretion of an oxidative stress marker, 8-hydroxyguanine (8-OH-Gua), among nickel-cadmium battery workers. J. Occup. Health 2008, 50, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Christman, J.K.; Sheikhnejad, G.; Marasco, C.J.; Sufrin, J.R. 5-methyl-2′-deoxycytidine in single-stranded DNA can act in cis to signal de novo DNA methylation. Proc. Natl. Acad. Sci. USA 1995, 92, 7347–7351. [Google Scholar] [CrossRef] [PubMed]
- Filipic, M. Mechanisms of cadmium induced genomic instability. Mutat. Res. 2012, 733, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Takiguchi, M.; Achanzar, W.E.; Qu, W.; Li, G.; Waalkes, M.P. Effects of cadmium on DNA-(cytosine-5) methyltransferase activity and DNA methylation status during cadmium-induced cellular transformation. Exp. Cell Res. 2003, 286, 355–365. [Google Scholar] [CrossRef]
- Zhu, H.; Li, K.; Liang, J.; Zhang, J.; Wu, Q. Changes in the levels of DNA methylation in testis and liver of sd rats neonatally exposed to 5-aza-2′-deoxycytidine and cadmium. J. Appl. Toxicol. 2011, 31, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Poirier, L.A.; Vlasova, T.I. The prospective role of abnormal methyl metabolism in cadmium toxicity. Environ. Health Perspect. 2002, 110, 793–795. [Google Scholar] [CrossRef] [PubMed]
- Thiesen, H.J.; Bach, C. Transition metals modulate DNA-protein interactions of sp1 zinc finger domains with its cognate target site. Biochem. Biophys. Res. Commun. 1991, 176, 551–557. [Google Scholar] [CrossRef]
- Razmiafshari, M.; Zawia, N.H. Utilization of a synthetic peptide as a tool to study the interaction of heavy metals with the zinc finger domain of proteins critical for gene expression in the developing brain. Toxicol. Appl. Pharmacol. 2000, 166, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hermsen, S.A.; van den Brandhof, E.J.; van der Ven, L.T.; Piersma, A.H. Relative embryotoxicity of two classes of chemicals in a modified zebrafish embryotoxicity test and comparison with their in vivo potencies. Toxicol. In Vitro 2011, 25, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Blaser, R.E.; Rosemberg, D.B. Measures of anxiety in zebrafish (Danio rerio): Dissociation of black/white preference and novel tank test. PLoS ONE 2012, 7, e36931. [Google Scholar] [CrossRef] [PubMed]
- Pittman, J.T.; Lott, C.S. Startle response memory and hippocampal changes in adult zebrafish pharmacologically-induced to exhibit anxiety/depression-like behaviors. Physiol. Behav. 2014, 123, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Cianca, V.; Bartolini, T.; Porfiri, M.; Macri, S. A robotics-based behavioral paradigm to measure anxiety-related responses in zebrafish. PLoS ONE 2013, 8, e69661. [Google Scholar] [CrossRef] [PubMed]
- Mueller, K.P.; Neuhauss, S.C. Quantitative measurements of the optokinetic response in adult fish. J. Neurosci. Methods 2010, 186, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Slob, W. Dose-response modeling of continuous endpoints. Toxicol. Sci. 2002, 66, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Becker, R.A.; Ankley, G.T.; Edwards, S.W.; Kennedy, S.W.; Linkov, I.; Meek, B.; Sachana, M.; Segner, H.; van Der Burg, B.; Villeneuve, D.L.; et al. Increasing scientific confidence in adverse outcome pathways: Application of tailored bradford-hill considerations for evaluating weight of evidence. Regul. Toxicol. Pharmacol. 2015, 72, 514–537. [Google Scholar] [CrossRef] [PubMed]
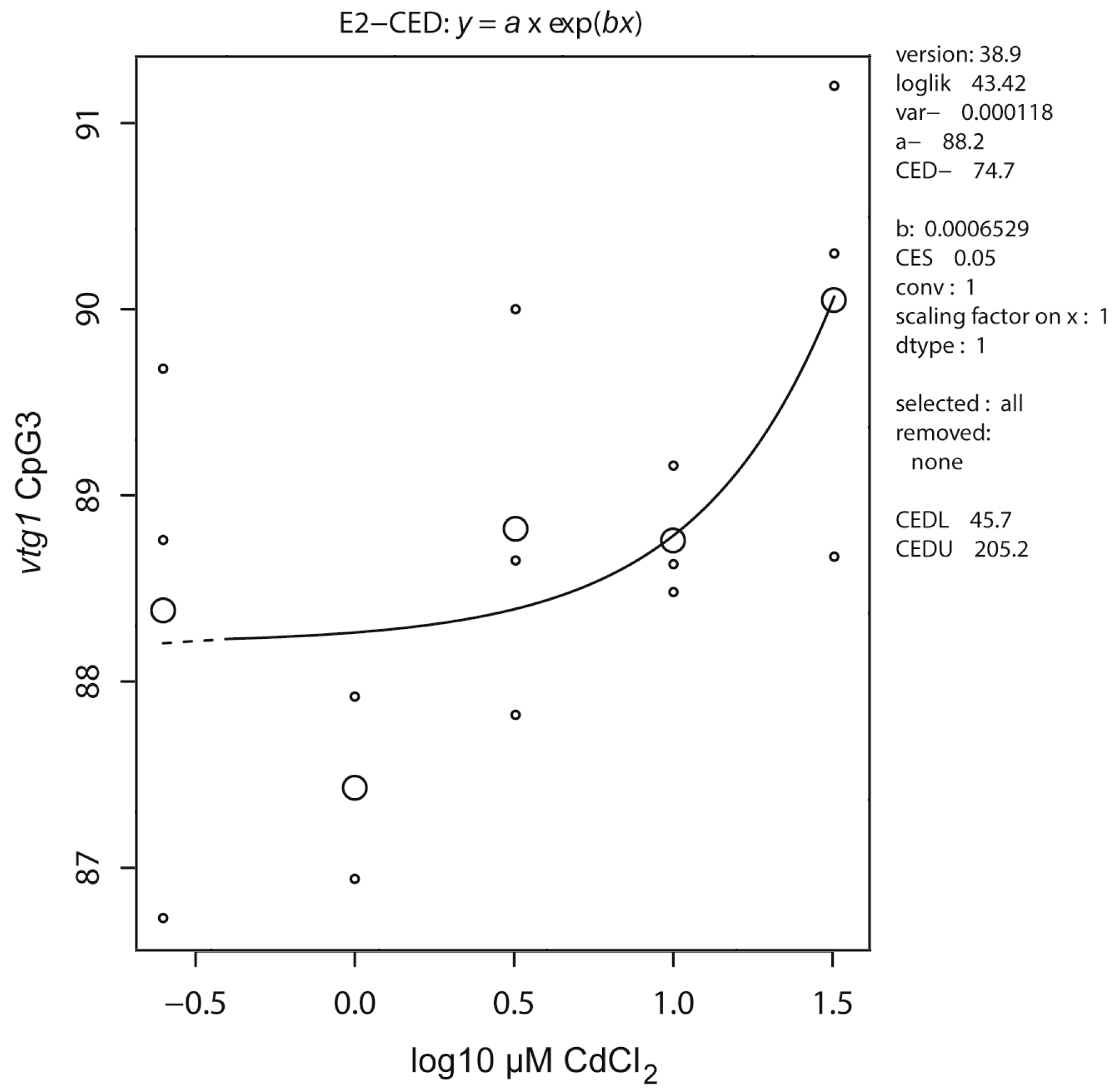
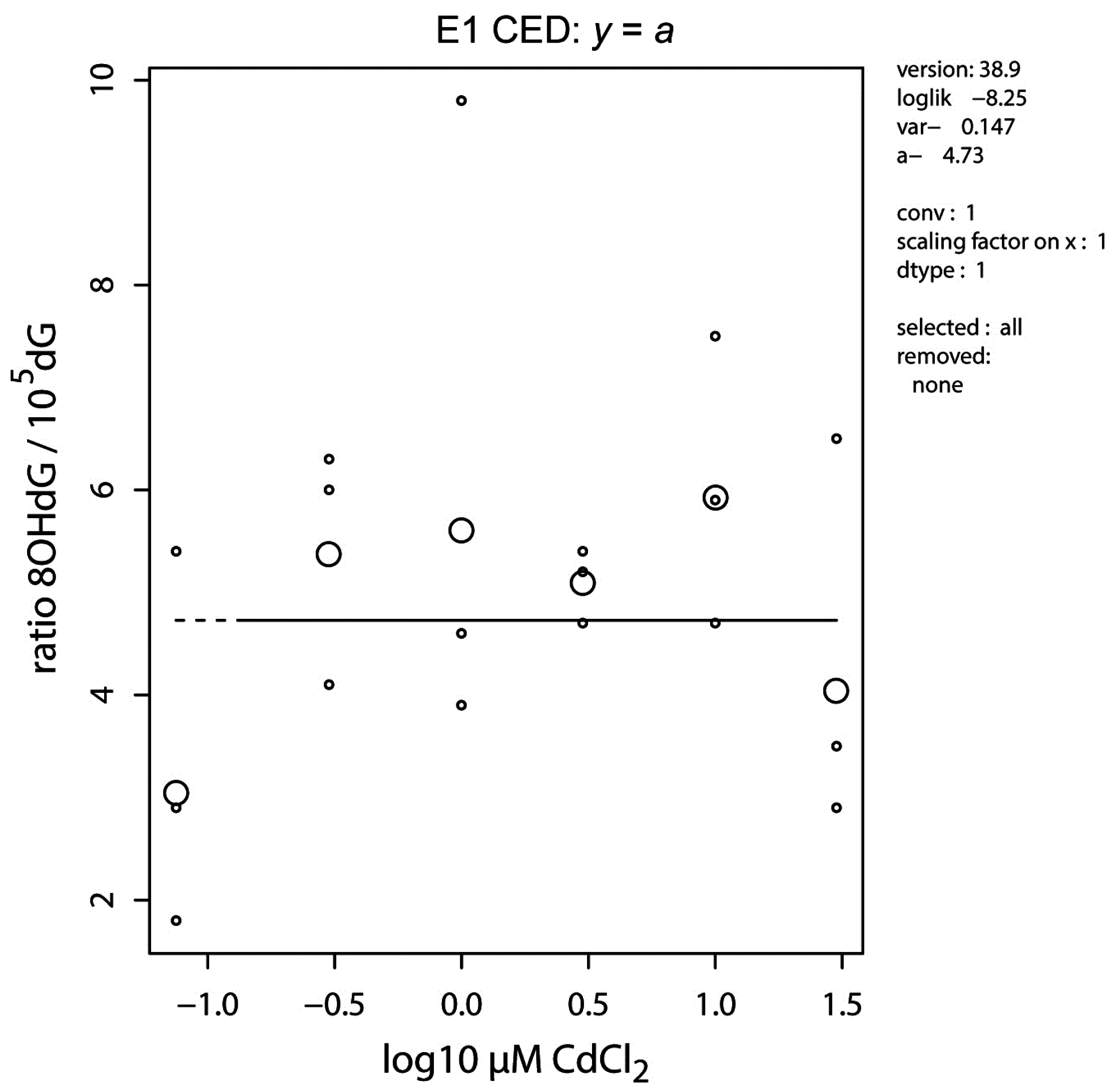
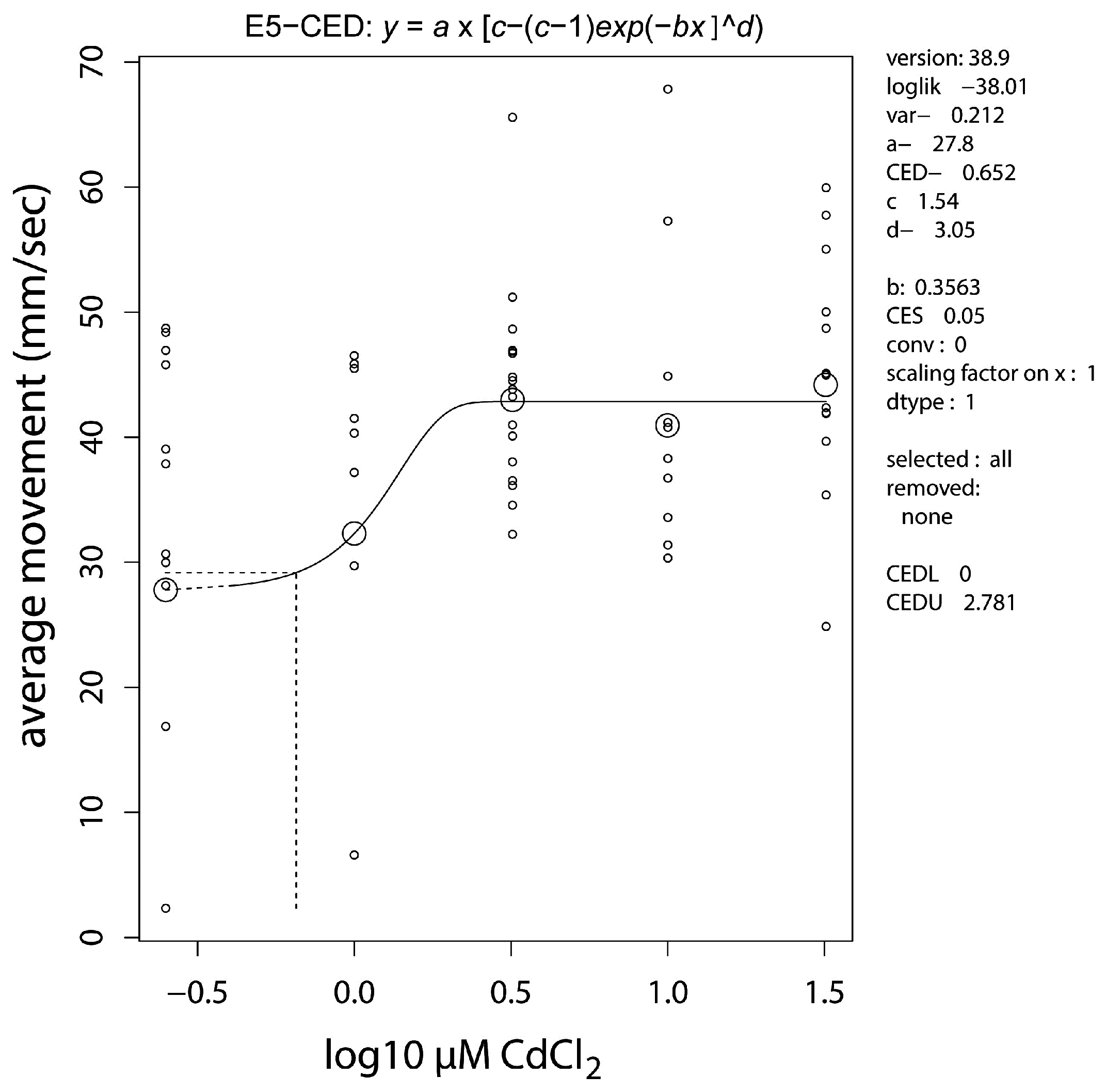
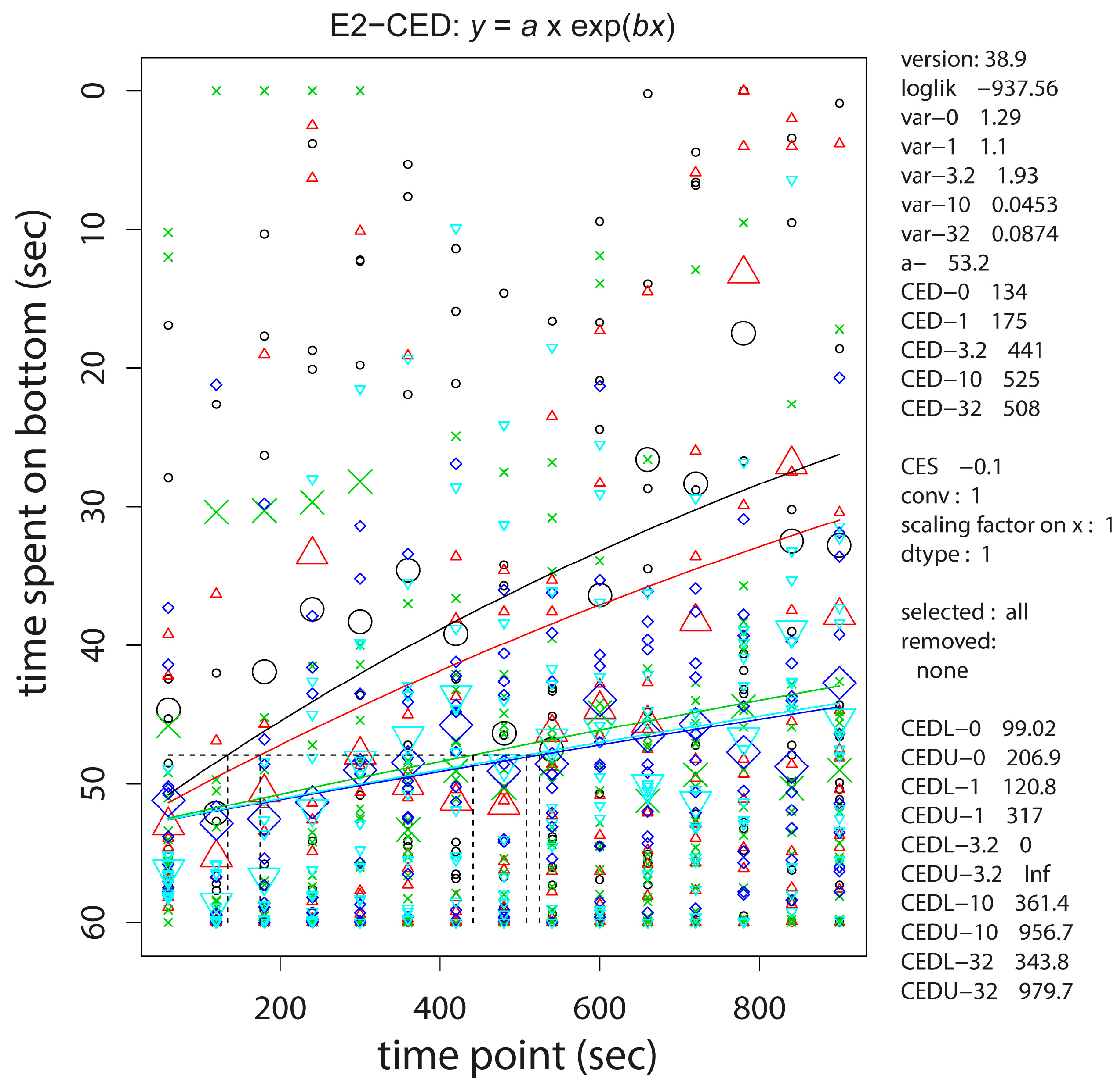
| Target | cyp19a2-1 | vasa-3 | vasa-5 | vtg1-3 |
|---|---|---|---|---|
| effect | -/↑ 164 | ↑ 61.6/- | ↑ 143/- | -/↑ 74.7 |
| Parameter | Carcass (μM) | Organ (μM) |
|---|---|---|
| GPx | 0.46 (0.20–1.3) | 0.30 (0.04–2.8) |
| GSH | 0.01 (0–0.04) | 0.04 (0–0.42) |
| GSSG | 0.15 (0–1.3) | 0.47 (0.17–0.92) |
| GSSG:GSH ratio | 0.01 (0–0.11) | 0.01 (0–0.54) |
| TTT | 14.1 (9.16–31.09) | 0.10 (0.04–0.27) |
| GR | ns | ns |
| SOD | ns | ns |
| BAP | ns | ns |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiter, S.; Sippel, J.; Bouwmeester, M.C.; Lommelaars, T.; Beekhof, P.; Hodemaekers, H.M.; Bakker, F.; Van den Brandhof, E.-J.; Pennings, J.L.A.; Van der Ven, L.T.M. Programmed Effects in Neurobehavior and Antioxidative Physiology in Zebrafish Embryonically Exposed to Cadmium: Observations and Hypothesized Adverse Outcome Pathway Framework. Int. J. Mol. Sci. 2016, 17, 1830. https://doi.org/10.3390/ijms17111830
Ruiter S, Sippel J, Bouwmeester MC, Lommelaars T, Beekhof P, Hodemaekers HM, Bakker F, Van den Brandhof E-J, Pennings JLA, Van der Ven LTM. Programmed Effects in Neurobehavior and Antioxidative Physiology in Zebrafish Embryonically Exposed to Cadmium: Observations and Hypothesized Adverse Outcome Pathway Framework. International Journal of Molecular Sciences. 2016; 17(11):1830. https://doi.org/10.3390/ijms17111830
Chicago/Turabian StyleRuiter, Sander, Josefine Sippel, Manon C. Bouwmeester, Tobias Lommelaars, Piet Beekhof, Hennie M. Hodemaekers, Frank Bakker, Evert-Jan Van den Brandhof, Jeroen L. A. Pennings, and Leo T. M. Van der Ven. 2016. "Programmed Effects in Neurobehavior and Antioxidative Physiology in Zebrafish Embryonically Exposed to Cadmium: Observations and Hypothesized Adverse Outcome Pathway Framework" International Journal of Molecular Sciences 17, no. 11: 1830. https://doi.org/10.3390/ijms17111830
APA StyleRuiter, S., Sippel, J., Bouwmeester, M. C., Lommelaars, T., Beekhof, P., Hodemaekers, H. M., Bakker, F., Van den Brandhof, E.-J., Pennings, J. L. A., & Van der Ven, L. T. M. (2016). Programmed Effects in Neurobehavior and Antioxidative Physiology in Zebrafish Embryonically Exposed to Cadmium: Observations and Hypothesized Adverse Outcome Pathway Framework. International Journal of Molecular Sciences, 17(11), 1830. https://doi.org/10.3390/ijms17111830





