Terminal Mannose Residues in Seminal Plasma Glycoproteins of Infertile Men Compared to Fertile Donors
Abstract
:1. Introduction
2. Results
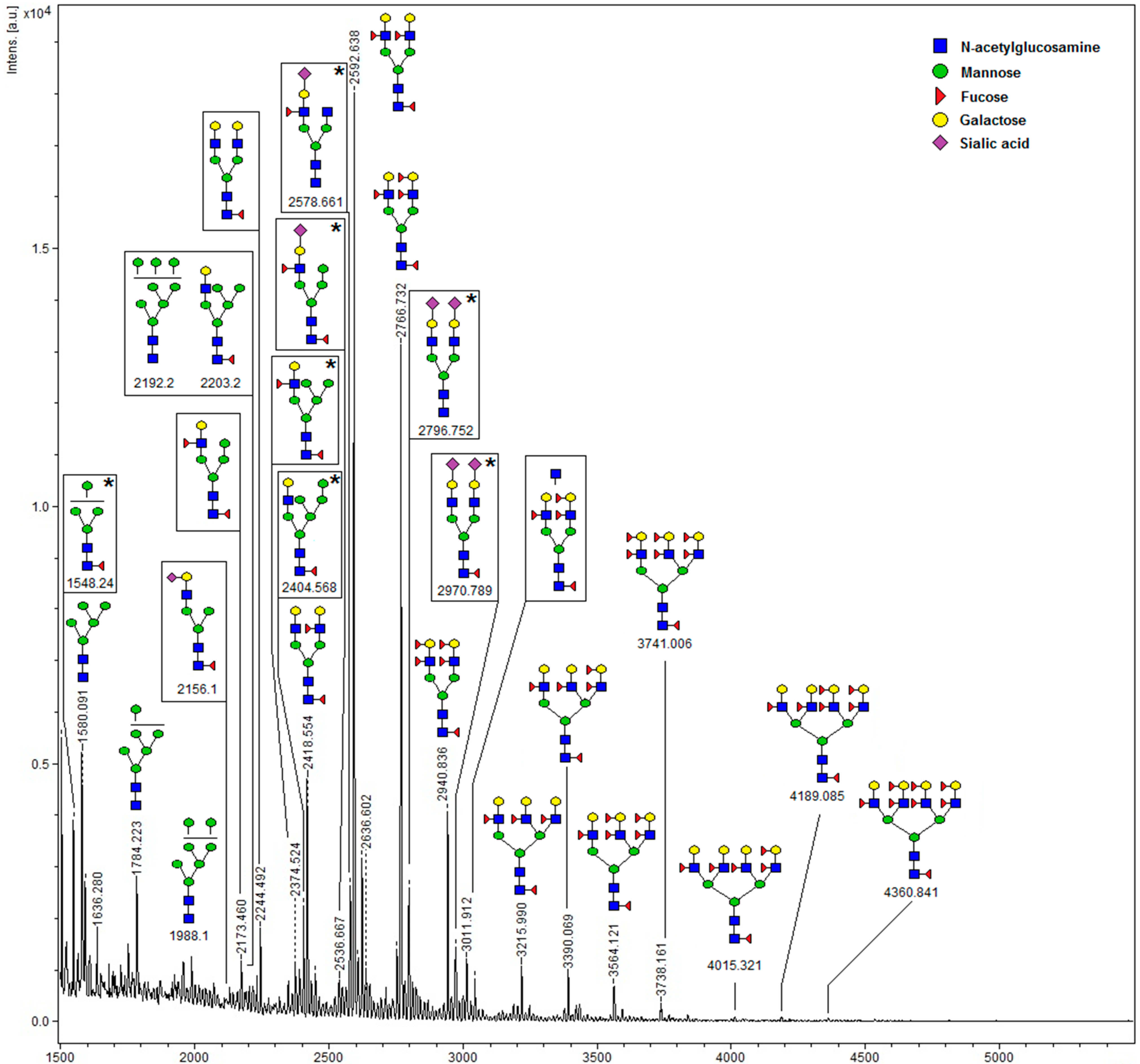
2.1. Detection of High-Mannose Type Oligosaccharides
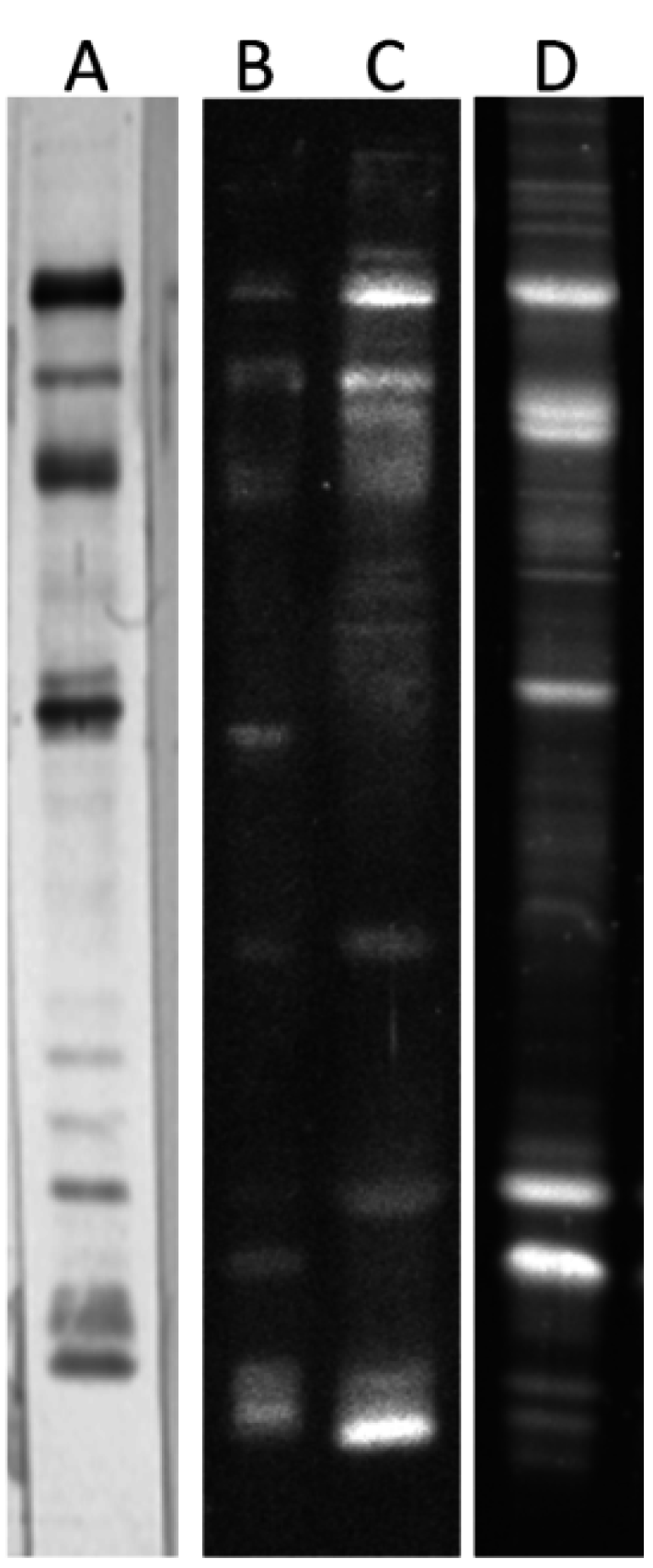
2.2. Altered Electrophoretic Patterns of GNL Reactivity
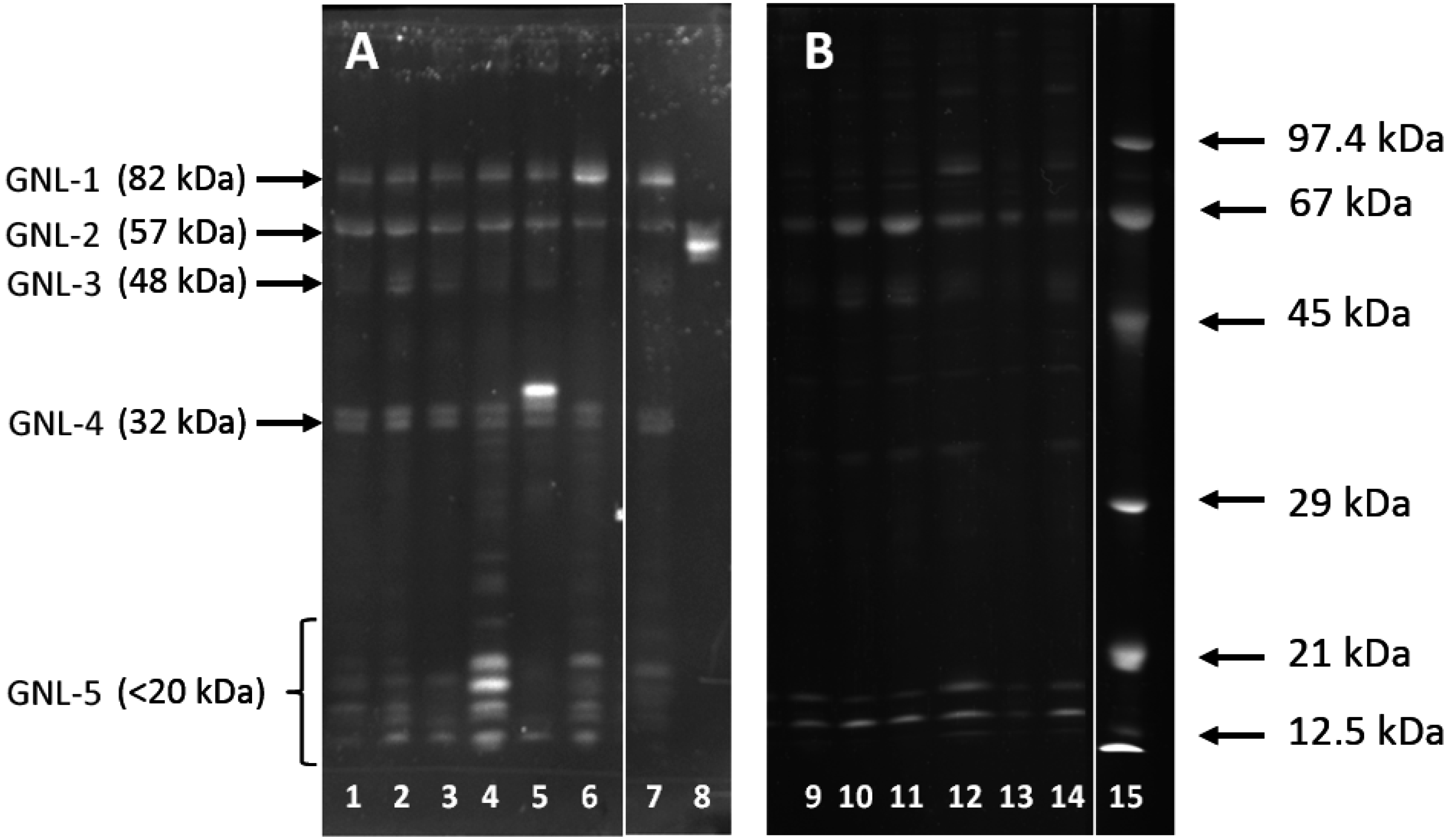
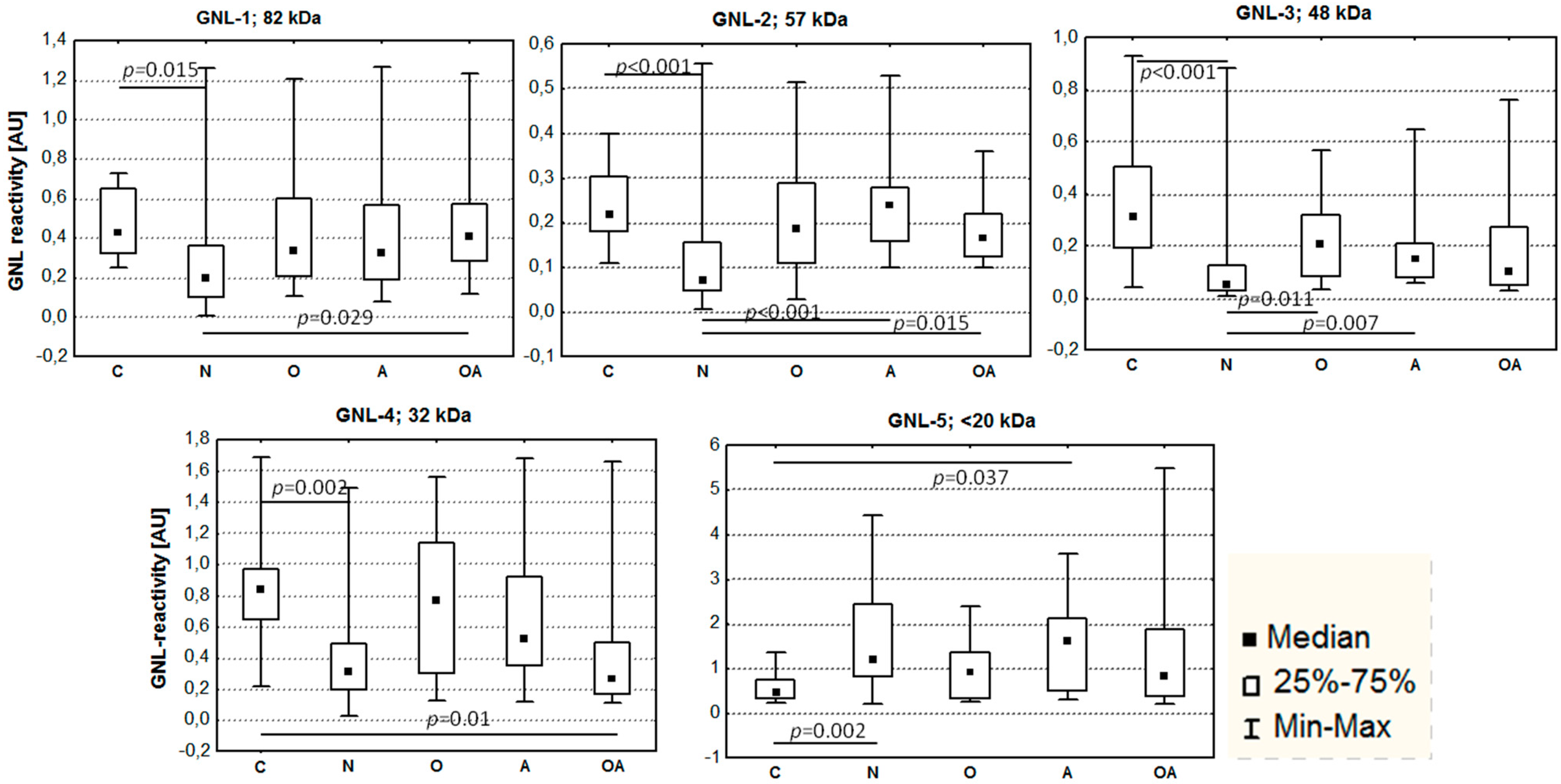
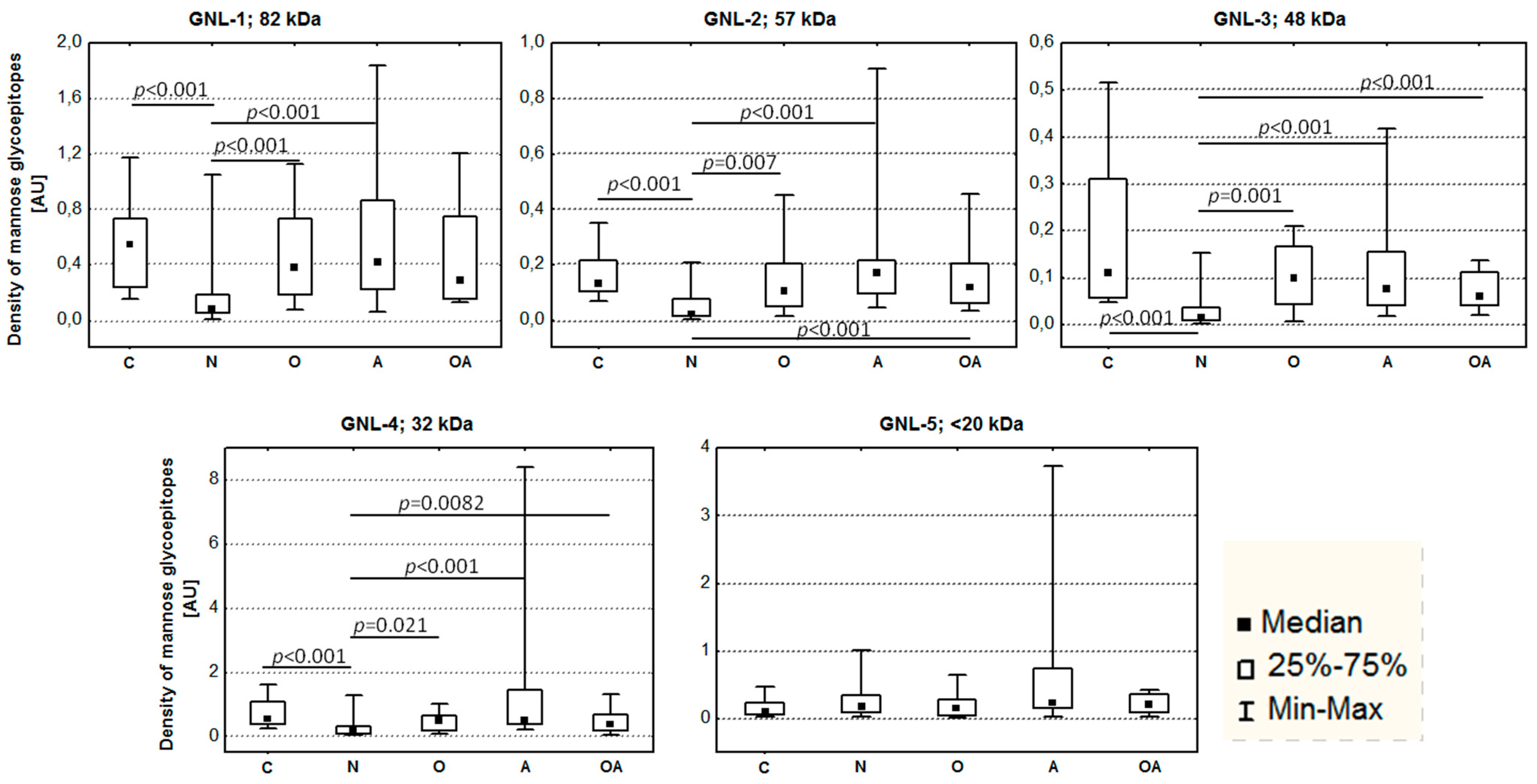
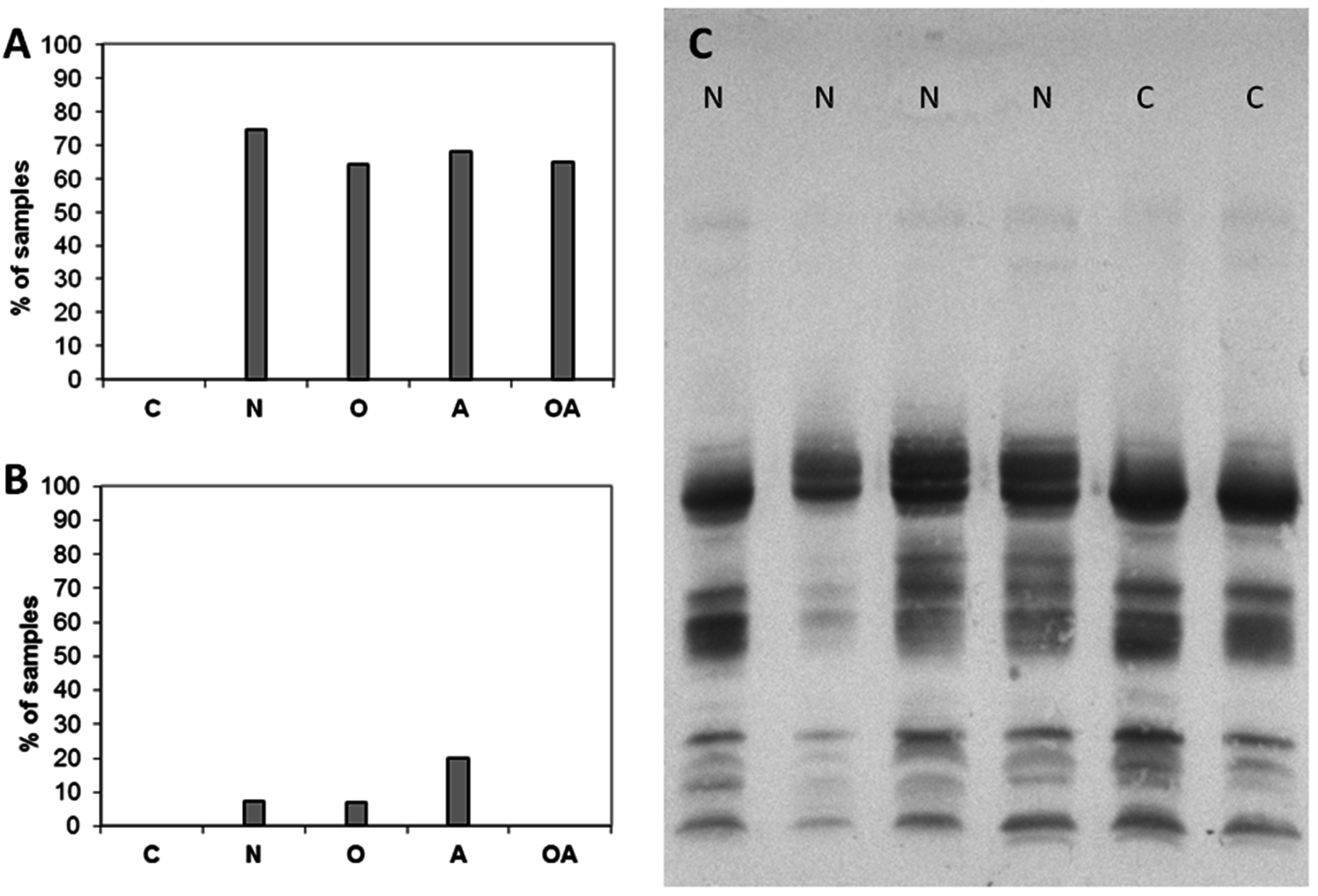
2.3. Identification of GNL-Reactive Bands in Mass Spectrometry
| Band No. | Protein Results of Mascot Database Search | Score | Matches | Antibody Reactivity [23] |
|---|---|---|---|---|
| GNL-1 | Chain X, structure of recombinant human lactoferrin | 6473 | 148 | Fn, PSA, Tf |
| lactoferrin precursor | 6405 | 148 | ||
| lactoferrin | 6379 | 146 | ||
| fibronectin precursor | 6334 | 145 | ||
| lactoferrin | 2004 | 41 | ||
| fibronectin 1, isoform CRA_f | 1517 | 29 | ||
| serotransferrin precursor | 872 | 25 | ||
| PREDICTED: mucin-6 isoform | 716 | 15 | ||
| extracellular matrix protein 1 | 508 | 12 | ||
| GNL-2 | PRO2044 | 3056 | 78 | Fn, hCG, ATIII |
| galectin-3-binding protein precursor | 1716 | 32 | ||
| fibronectin isoform 3 preproprotein | 1705 | 35 | ||
| lactoferrin | 1666 | 37 | ||
| prosaposin isoform a preproprotein | 1071 | 25 | ||
| serotransferrin precursor | 975 | 24 | ||
| l-plastin variant | 929 | 22 | ||
| heat shock protein | 656 | 12 | ||
| GNL-3 | prostatic acid phosphatase isoform PAP precursor | 3516 | 98 | PAP, IgG |
| fibronectin precursor | 2420 | 51 | ||
| Chain X, Structure Of Recombinant Human Lactoferrin | 1982 | 40 | ||
| plasma serine protease inhibitor precursor | 1723 | 46 | ||
| Chain A, 2.0 Angstrom Structure Of Intact Alpha-1-Antitrypsin: A Canonical Template For Active Serpins | 1341 | 35 | ||
| Chain A, The S Variant Of Human Alpha1-antitrypsin | 1323 | 36 | ||
| Chain A, Crystal Structure Of Cleaved Antitrypsin Polymer | 1285 | 35 | ||
| Serpin peptidase inhibitor, clade A (alpha-1 antiproteinase, antitrypsin) | 1269 | 34 | ||
| nucleobindin-2 precursor | 1066 | 23 | ||
| brain acid soluble protein 1 | 977 | 12 | ||
| 2-phosphopyruvate-hydratase alpha-enolase | 697 | 15 | ||
| monocyte differentiation antigen CD14 precursor | 642 | 10 | ||
| extracellular matrix protein 1 | 621 | 15 | ||
| serotransferrin precursor | 542 | 14 | ||
| Ig G1 H | 517 | 11 | ||
| Zn-alpha2-glycoprotein | 509 | 13 | ||
| GNL-4 | prostate specific antigen precursor, partial [Homo sapiens] | 3204 | 64 | PSA, PAP |
| apolipoprotein J precursor [Homo sapiens] | 1662 | 40 | ||
| fibronectin isoform 3 preproprotein [Homo sapiens] | 1198 | 28 | ||
| Chain A, Human Annexin V With Proline Substitution By Thioproline | 893 | 20 | ||
| annexin A1 [Homo sapiens] | 875 | 18 | ||
| cerebroside sulfate activator protein [Homo sapiens] | 659 | 13 | ||
| acidic epididymal glycoprotein homolog [Homo sapiens] | 521 | 8 | ||
| semenogelin-2 precursor [Homo sapiens] | 520 | 11 | ||
| annexin A3 [Homo sapiens] | 488 | 9 | ||
| semenogelin-1 preproprotein [Homo sapiens] | 418 | 11 | ||
| Zn-alpha2-glycoprotein [Homo sapiens] | 391 | 9 | ||
| GNL-6 | fibronectin isoform 3 preproprotein [Homo sapiens] | 1213 | 28 | Fn, IgG, PSA, GdS |
| placental protein 14 [Homo sapiens] * | 1207 | 23 | ||
| semenogelin-1 preproprotein [Homo sapiens] | 752 | 17 | ||
| apolipoprotein J precursor [Homo sapiens] | 683 | 15 | ||
| prostate specific antigen precursor, partial [Homo sapiens] | 662 | 13 | ||
| mucin-6 precursor [Homo sapiens] | 616 | 12 | ||
| protein DJ-1 [Homo sapiens] | 581 | 12 |
2.4. Isolation of GNL-Reactive Glycoproteins
| Protein Results of Mascot Database Search | Score | Matches |
|---|---|---|
| fibronectin | 72,742 | 1563 |
| prolactin-inducible protein | 65,038 | 1179 |
| prostatic acid phosphatase | 26,608 | 682 |
| semenogelin-1 | 15,725 | 442 |
| semenogelin-2 | 14,489 | 442 |
| clusterin | 10,417 | 336 |
| glycodelin | 7447 | 163 |
| carboxypeptidase E | 3190 | 102 |
| cystatin-S | 2136 | 50 |
| mucin-6 | 2056 | 93 |
| lactotransferrin | 1923 | 42 |
| IgGFc-binding protein | 1114 | 32 |
| aminopeptidase N | 1015 | 26 |
| zinc-α2-glycoprotein | 991 | 23 |
| calcium-binding protein | 499 | 12 |
| α1-acid glycoprotein | 444 | 9 |
| prostate specific antigen | 439 | 11 |
3. Discussion
4. Experimental Section
4.1. Clinical Material
4.2. SDS-PAGE and Lectin Blotting
4.3. Lectin Reactivity
4.4. Densitometry
4.5. Reactivity with Antibodies
4.6. Affinity Chromatography
4.7. Mass Spectrometry Identification of Proteins
4.8. Glycan Analysis by MALDI-TOF-MS
4.9. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wassarman, P.M. Towards molecular mechanisms for gamete adhesion and fusion during mammalian fertilization. Curr. Opin. Cell Biol. 1995, 7, 658–664. [Google Scholar] [CrossRef]
- Seppälä, M.; Koistinen, H.; Koistinen, R.; Chiu, P.C.; Yeung, W.S. Glycosylation related actions of glycodelin: Gamete, cumulus cell, immune cell and clinical associations. Hum. Reprod. Update 2007, 13, 275–287. [Google Scholar] [CrossRef]
- Pang, P.C.; Chiu, P.C.; Lee, C.L.; Chang, L.Y.; Panico, M.; Morris, H.R.; Haslam, S.M.; Khoo, K.H.; Clark, G.F.; Yeung, W.S.; et al. Human sperm binding is mediated by the sialyl-Lewis(x) oligosaccharide on the zona pellucida. Science 2011, 333, 1761–1764. [Google Scholar] [CrossRef]
- Alavi, A.; Axford, J.S. Glyco-biomarkers: Potential determinants of cellular physiology and pathology. Dis. Markers 2008, 25, 193–205. [Google Scholar] [CrossRef]
- Alavi, A.; Axford, J.S. Sweet and sour: The impact of sugars on disease. Rheumatology 2008, 47, 760–770. [Google Scholar] [CrossRef]
- Pang, P.C.; Tissot, B.; Drobnis, E.Z.; Morris, H.R.; Dell, A.; Clark, G.F. Analysis of the human seminal plasma glycome reveals the presence of immunomodulatory carbohydrate functional groups. J. Proteome Res. 2009, 8, 4906–4915. [Google Scholar] [CrossRef]
- White, K.Y.; Rodemich, L.; Nyalwidhe, J.O.; Comunale, M.A.; Clements, M.A.; Lance, R.S.; Schellhammer, P.F.; Mehta, A.S.; Semmes, O.J.; Drake, R.R. Glycomic characterization of prostate-specific antigen and prostatic acid phosphatase in prostate cancer and benign disease seminal plasma fluids. J. Proteome Res. 2009, 8, 620–630. [Google Scholar] [CrossRef]
- Stanley, P.; Schachter, H.; Taniguchi, N. N-Glycans. In Essentials of Glycobiology, 2nd ed.; Varki, A., Cummings, R.D., Esko, J.D., Freeze, H.H., Stanley, P., Bertozzi, C.R., Hart, G.W., Etzler, M.E., Eds.; Cold Spring Harbor Press: Woodbury, NY, USA, 2009. [Google Scholar]
- Morris, H.R.; Dell, A.; Easton, R.L.; Panico, M.; Koistinen, H.; Koistinen, R.; Oehninger, S.; Patankar, M.S.; Seppala, M.; Clark, G.F. Gender-specific glycosylation of human glycodelin affects its contraceptive activity. J. Biol. Chem. 1996, 271, 32159–32167. [Google Scholar] [CrossRef]
- Yeung, W.S.; Lee, K.F.; Koistinen, R.; Koistinen, H.; Seppala, M.; Ho, P.C.; Chiu, P.C. Roles of glycodelin in modulating sperm function. Mol. Cell. Endocrinol. 2006, 250, 149–156. [Google Scholar] [CrossRef]
- Yeung, W.S.; Lee, K.F.; Koistinen, R.; Koistinen, H.; Seppälä, M.; Chiu, P.C. Effects of glycodelins on functional competence of spermatozoa. J. Reprod. Immunol. 2009, 83, 26–30. [Google Scholar] [CrossRef]
- Mattsson, J.M.; Valmu, L.; Laakkonen, P.; Stenman, U.H.; Koistinen, H. Structural characterization and anti-angiogenic properties of prostate-specific antigen isoforms in seminal fluid. Prostate 2008, 68, 945–954. [Google Scholar] [CrossRef]
- Fukushima, K.; Satoh, T.; Baba, S.; Yamashita, K. Alpha1,2-Fucosylated and beta-N-acetyl-galactosaminylated prostate-specific antigen as an efficient marker of prostatic cancer. Glycobiology 2010, 20, 452–460. [Google Scholar] [CrossRef]
- Lefièvre, L.; Bedu-Addo, K.; Conner, S.J.; Machado-Oliveira, G.S.; Chen, Y.; Kirkman-Brown, J.C.; Afnan, M.A.; Publicover, S.J.; Ford, W.C.; Barratt, C.L. Counting sperm does not add up any more: Time for a new equation? Reproduction 2007, 133, 675–684. [Google Scholar] [CrossRef]
- Barratt, C.L.; Mansell, S.; Beaton, C.; Tardif, S.; Oxenham, S.K. Diagnostic tools in male infertility—The question of sperm dysfunction. Asian J. Androl. 2011, 13, 53–58. [Google Scholar] [CrossRef]
- Esteves, S.C.; Miyaoka, R.; Agarwal, A. An update on the clinical assessment of the infertile male. Clinics 2011, 66, 691–700. [Google Scholar] [CrossRef]
- Hwang, K.; Lipshultz, L.I.; Lamb, D.J. Use of diagnostic testing to detect infertility. Curr. Urol. Rep. 2011, 12, 68–76. [Google Scholar] [CrossRef]
- Franken, D.R.; Oehninger, S. Semen analysis and sperm function testing. Asian J. Androl. 2012, 14, 6–13. [Google Scholar] [CrossRef]
- Nagler, H.M. Male factor infertility: A solitary semen analysis can never predict normal fertility. Nat. Rev. Urol. 2011, 8, 16–17. [Google Scholar] [CrossRef]
- Kratz, E.M.; Faundez, R.; Katnik-Prastowska, I. Fucose and sialic acid expressions in human seminal fibronectin and α1-acid glycoprotein associated with leukocytospermia of infertile men. Dis. Markers 2011, 31, 317–325. [Google Scholar] [CrossRef]
- Kratz, E.M.; Ferens-Sieczkowska, M.; Faundez, R.; Kątnik-Prastowska, I. Changes in fucosylation of human seminal IgG and secretory component of IgA in leukocytospermic patients. Glycoconj. J. 2014, 31, 51–60. [Google Scholar] [CrossRef]
- Kratz, E.M.; Ferens-Sieczkowska, M. Association of IgA secretory component sialylation with leucocytospermia of infertile men—A pilot study. Andrologia 2014, 46, 1200–1202. [Google Scholar] [CrossRef]
- Olejnik, B.; Kratz, E.M.; Zimmer, M.; Ferens-Sieczkowska, M. Glycoprotein fucosylation is increased in seminal plasma of subfertile men. Asian J. Androl. 2015, 17, 274–280. [Google Scholar]
- Ceroni, A.; Maass, K.; Geyer, H.; Geyer, R.; Dell, A.; Haslam, S.M. GlycoWorkbench: A tool for the computer-assisted annotation of mass spectra of glycans. J. Proteome Res. 2008, 7, 1650–1659. [Google Scholar] [CrossRef]
- Damerell, D.; Ceroni, A.; Maass, K.; Ranzinger, R.; Dell, A.; Haslam, S.M. The GlycanBuilder and GlycoWorkbench glycoinformatics tools: Updates and new developments. Biol. Chem. 2012, 393, 1357–1362. [Google Scholar] [CrossRef]
- Juyena, N.S.; Stelletta, C. Seminal plasma: An essential attribute to spermatozoa. J. Androl. 2012, 33, 536–551. [Google Scholar] [CrossRef]
- Bromfield, J.J. Seminal fluid and reproduction: Much more than previously thought. J. Assist. Reprod. Genet. 2014, 31, 627–636. [Google Scholar] [CrossRef]
- Bedford, J.M. The functions—Or not—Of seminal plasma? Biol. Reprod. 2015, 92, 18. [Google Scholar] [CrossRef]
- Rickard, J.P.; Pini, T.; Soleilhavoup, C.; Cognie, J.; Bathgate, R.; Lynch, G.W.; Evans, G.; Maxwell, W.M.; Druart, X.; de Graaf, S.P. Seminal plasma aids the survival and cervical transit of epididymal ram spermatozoa. Reproduction 2014, 148, 469–478. [Google Scholar] [CrossRef]
- Leahy, T.; de Graaf, S.P. Seminal plasma and its effect on ruminant spermatozoa during processing. Reprod. Domest. Anim. 2012, 47, 207–213. [Google Scholar] [CrossRef]
- Rickard, J.P.; Schmidt, R.E.; Maddison, J.W.; Bathgate, R.; Lynch, G.W.; Druart, X.; de Graaf, S.P. Variation in seminal plasma alters the ability of ram spermatozoa to survive cryopreservation. Reprod. Fertil. Dev. 2014. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, H.; Kvist, U.; Ernerudh, J.; Sanz, L.; Calvete, J.J. Seminal plasma proteins: What role do they play? Am. J. Reprod. Immunol. 2011, 66, 11–22. [Google Scholar] [CrossRef]
- Druart, X.; Rickard, J.P.; Mactier, S.; Kohnke, P.L.; Kershaw-Young, C.M.; Bathgate, R.; Gibb, Z.; Crossett, B.; Tsikis, G.; Labas, V.; et al. Proteomic characterization and cross species comparison of mammalian seminal plasma. J. Proteomics 2013, 91, 13–22. [Google Scholar] [CrossRef]
- Varki, A.; Freeze, H.F.; Gagneux, P. Evolution of glycan diversity. In Essentials of Glycobiology, 2nd ed.; Varki, A., Cummings, R.D., Esko, J.D., Freeze, H.H., Stanley, P., Bertozzi, C.R., Hart, G.W., Etzler, M.E., Eds.; Cold Spring Harbor Press: Woodbury, NY, USA, 2009. [Google Scholar]
- Chiu, P.C.; Chung, M.K.; Tsang, H.Y.; Koistinen, R.; Koistinen., H.; Seppala, M.; Lee, K.F.; Yeung, W.S. Glycodelin-S in human seminal plasma reduces cholesterol efflux and inhibits capacitation of spermatozoa. J. Biol. Chem. 2005, 280, 25580–25589. [Google Scholar] [CrossRef]
- World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen, 5th ed.; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Sigma Aldrich Co. SYPRO®Ruby Protein Gel Stain. Product S 4942 Datasheet. Available online: http://www.sigmaaldrich.com/catalog/search?term=494275&interface=Product%20No.&N=0&mode=mode%20matchpartialmax&lang=pl®ion=PL&focus=product (accessed on 30 June 2015).
- Towbin, H.; Stachelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 1979, 76, 4350–4353. [Google Scholar] [CrossRef]
- Shibuya, N.; Goldstein, I.J.; van Damme, E.J.; Peumans, W.J. Binding properties of a mannose-specific lectin from the snowdrop (Galanthus nivalis) bulb. J. Biol. Chem. 1988, 263, 728–734. [Google Scholar]
- Barre, A.; Bourne, Y.; van Damme, E.J.; Peumans, W.J.; Rougé, P. Mannose-binding plant lectins: Different structural scaffolds for a common sugar-recognition process. Biochimie 2001, 83, 645–651. [Google Scholar] [CrossRef]
- Van Damme, E.J.; Nakamura-Tsuruta, S.; Smith, D.F.; Ongenaert, M.; Winter, H.C.; Rougé, P.; Goldstein, I.J.; Mo, H.; Kominami, J.; Culerrier, R.; et al. Phylogenetic and specificity studies of two-domain GNA-related lectins: Generation of multispecificity through domain duplication and divergent evolution. Biochem. J. 2007, 404, 51–61. [Google Scholar]
- Kossowska, B.; Ferens-Sieczkowska, M.; Gancarz, R.; Passowicz-Muszyńska, E.; Jankowska, R. Fucosylation of serum glycoproteins in lung cancer patients. Clin. Chem. Lab. Med. 2005, 43, 361–369. [Google Scholar] [CrossRef]
- Ferens-Sieczkowska, M.; Kossowska, B.; Gancarz, R.; Dudzik, D.; Knas, M.; Popko, J.; Zwierz, K. Fucosylation in synovial fluid as a novel clinical marker for differentiating joint diseases—A preliminary study. Clin. Exp. Rheumatol. 2007, 25, 92–95. [Google Scholar]
- Shevchenko, A.; Tomas, H.; Havlis, J.; Olsen, J.V.; Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 2006, 1, 2856–2860. [Google Scholar] [CrossRef]
- Morelle, W.; Michalski, J.C. Analysis of protein glycosylation by mass spectrometry. Nat. Protoc. 2007, 2, 1585–1602. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olejnik, B.; Jarząb, A.; Kratz, E.M.; Zimmer, M.; Gamian, A.; Ferens-Sieczkowska, M. Terminal Mannose Residues in Seminal Plasma Glycoproteins of Infertile Men Compared to Fertile Donors. Int. J. Mol. Sci. 2015, 16, 14933-14950. https://doi.org/10.3390/ijms160714933
Olejnik B, Jarząb A, Kratz EM, Zimmer M, Gamian A, Ferens-Sieczkowska M. Terminal Mannose Residues in Seminal Plasma Glycoproteins of Infertile Men Compared to Fertile Donors. International Journal of Molecular Sciences. 2015; 16(7):14933-14950. https://doi.org/10.3390/ijms160714933
Chicago/Turabian StyleOlejnik, Beata, Anna Jarząb, Ewa M. Kratz, Mariusz Zimmer, Andrzej Gamian, and Mirosława Ferens-Sieczkowska. 2015. "Terminal Mannose Residues in Seminal Plasma Glycoproteins of Infertile Men Compared to Fertile Donors" International Journal of Molecular Sciences 16, no. 7: 14933-14950. https://doi.org/10.3390/ijms160714933
APA StyleOlejnik, B., Jarząb, A., Kratz, E. M., Zimmer, M., Gamian, A., & Ferens-Sieczkowska, M. (2015). Terminal Mannose Residues in Seminal Plasma Glycoproteins of Infertile Men Compared to Fertile Donors. International Journal of Molecular Sciences, 16(7), 14933-14950. https://doi.org/10.3390/ijms160714933








