The Photodynamic Antibacterial Effects of Silicon Phthalocyanine (Pc) 4
Abstract
:1. Introduction
2. Results
2.1. Pc 4 Uptake by S. Aureus
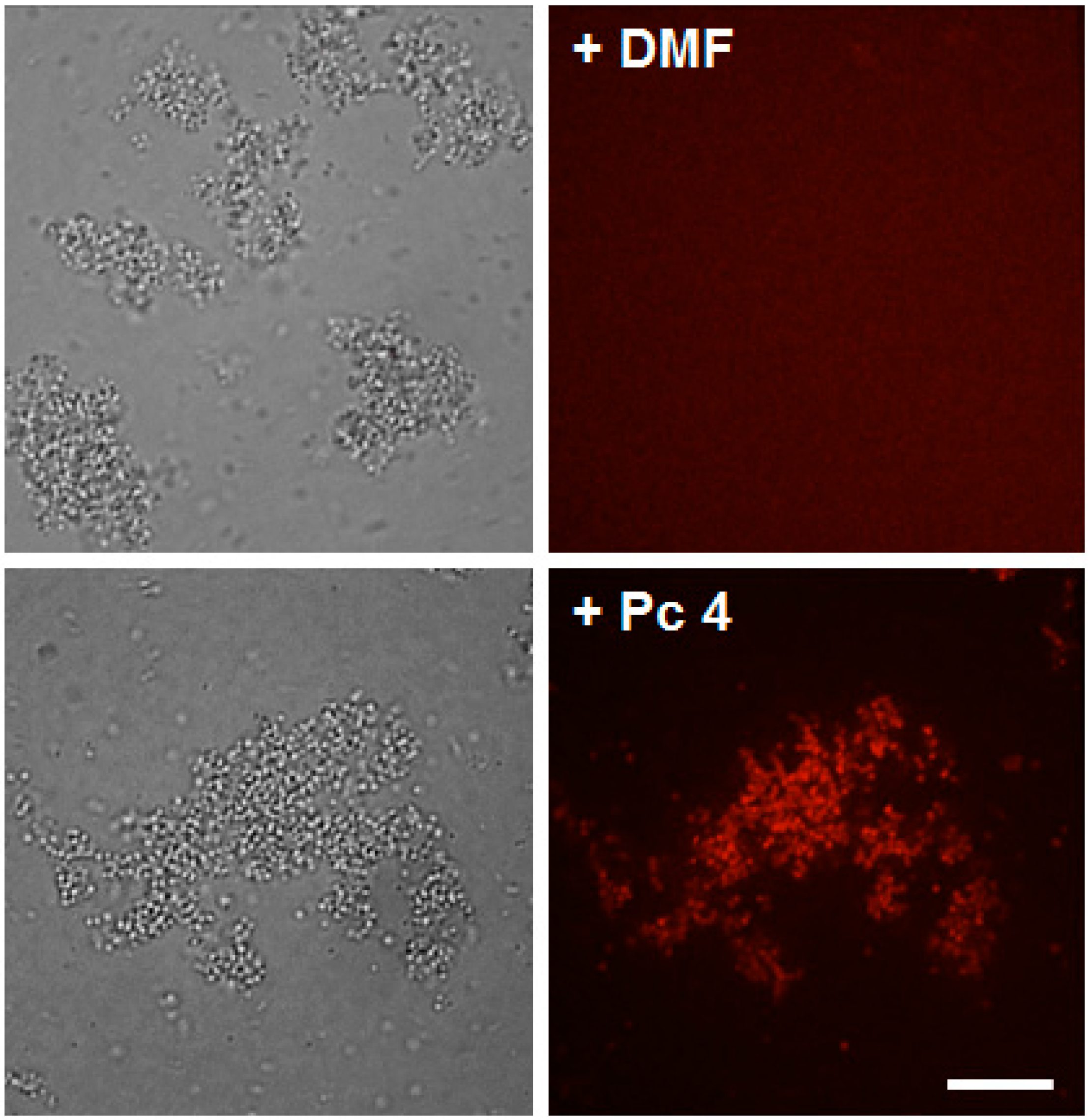
2.2. Toxicity Studies
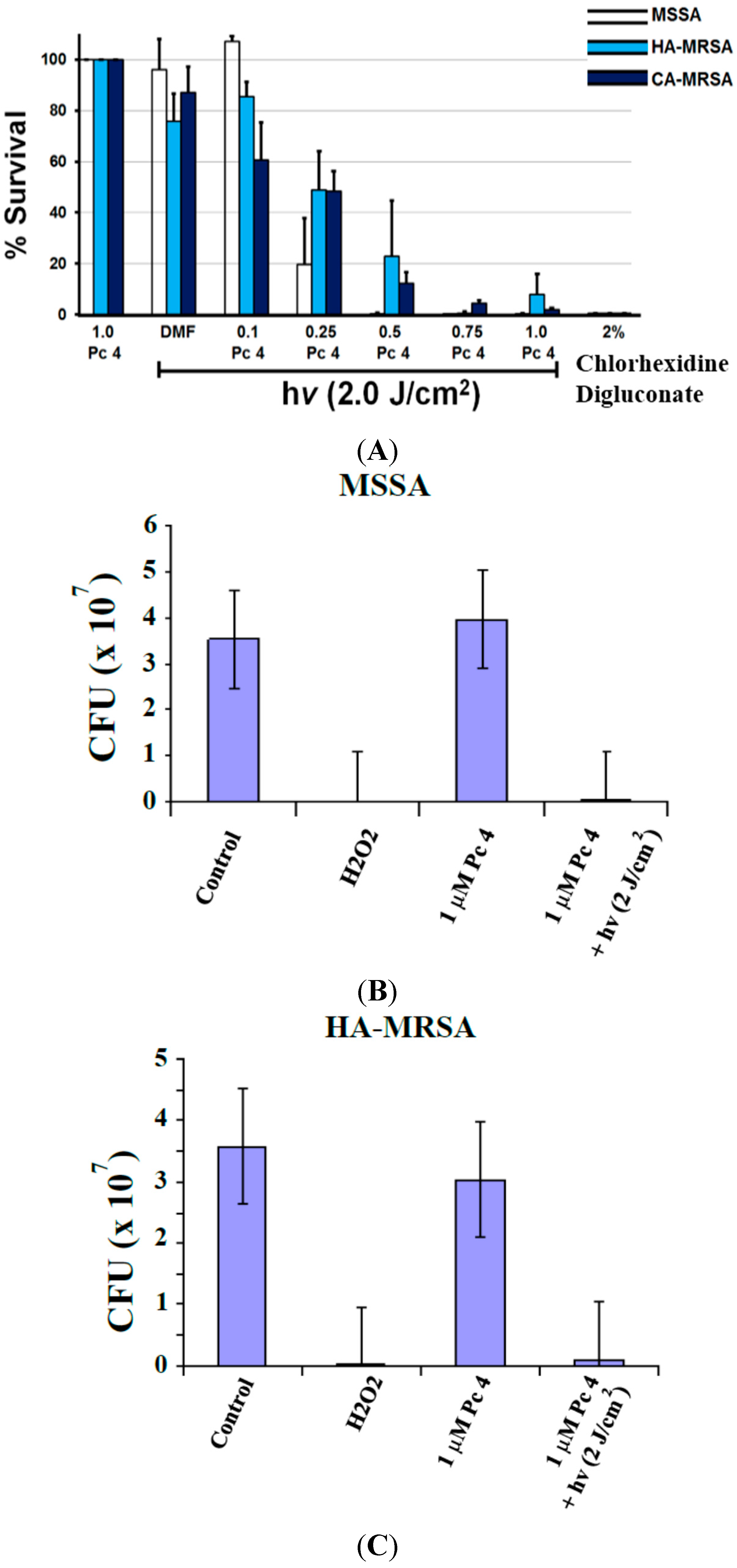
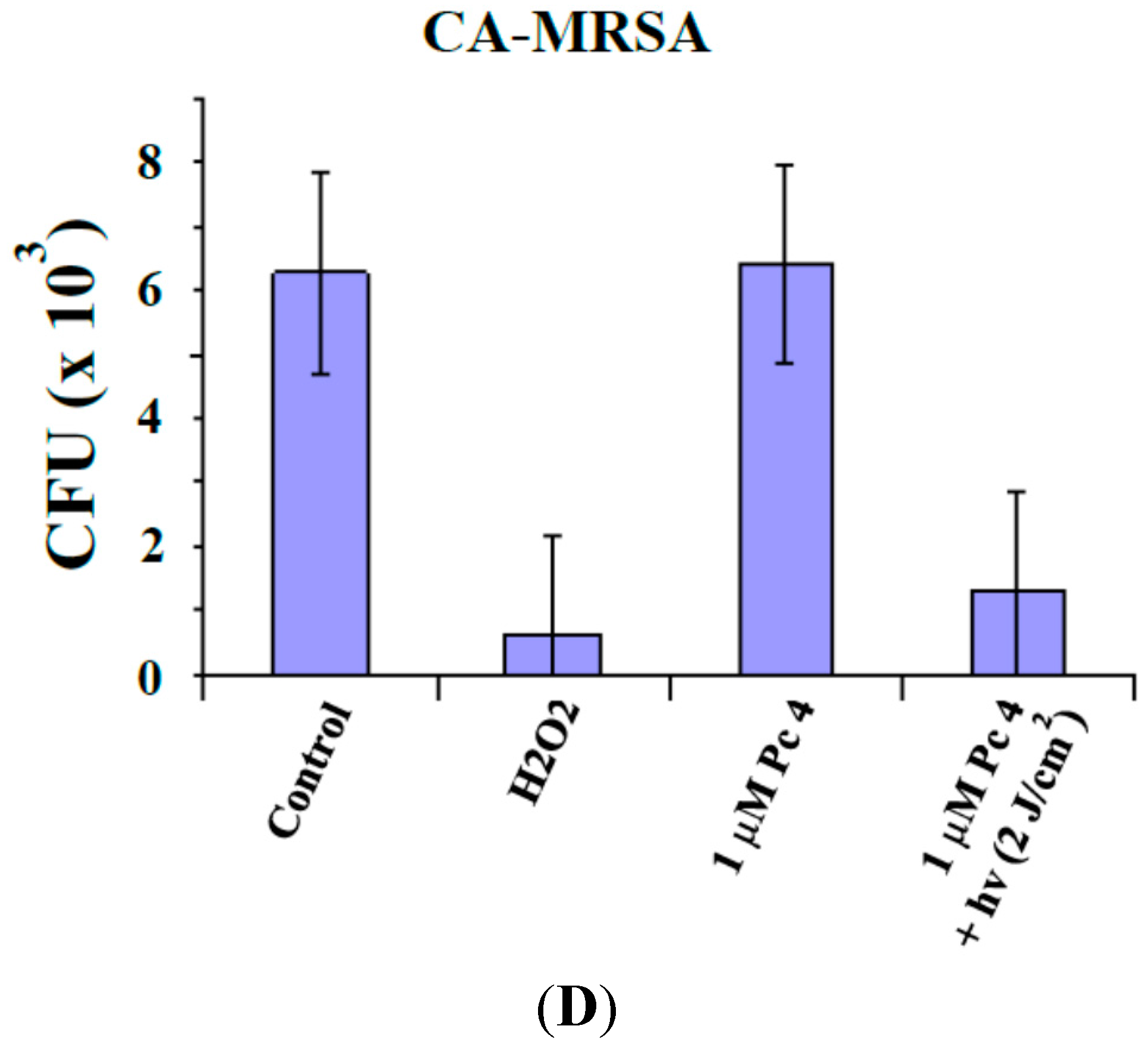
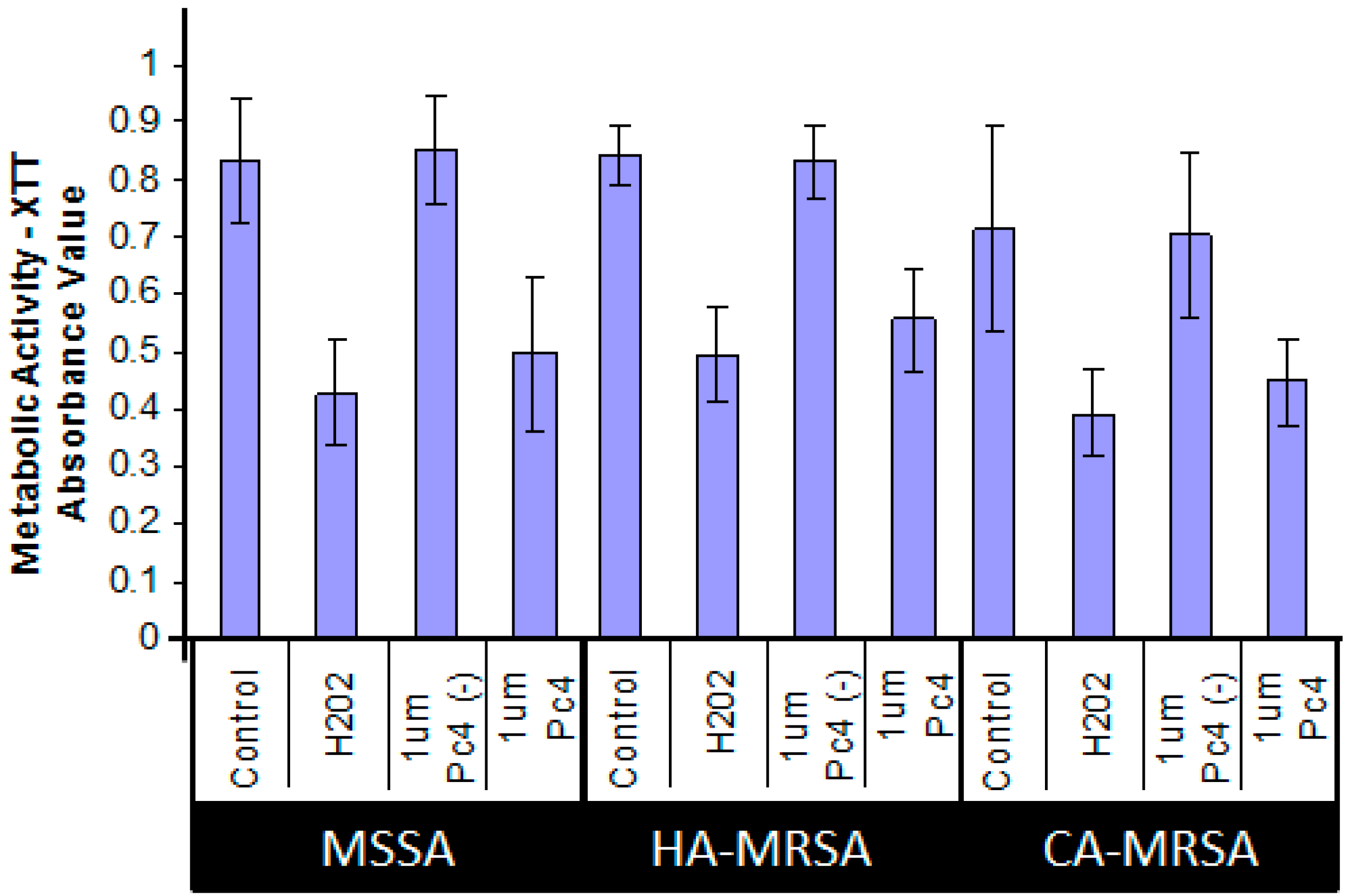
2.3. ROS Generation Immediately Following Pc 4-PDT
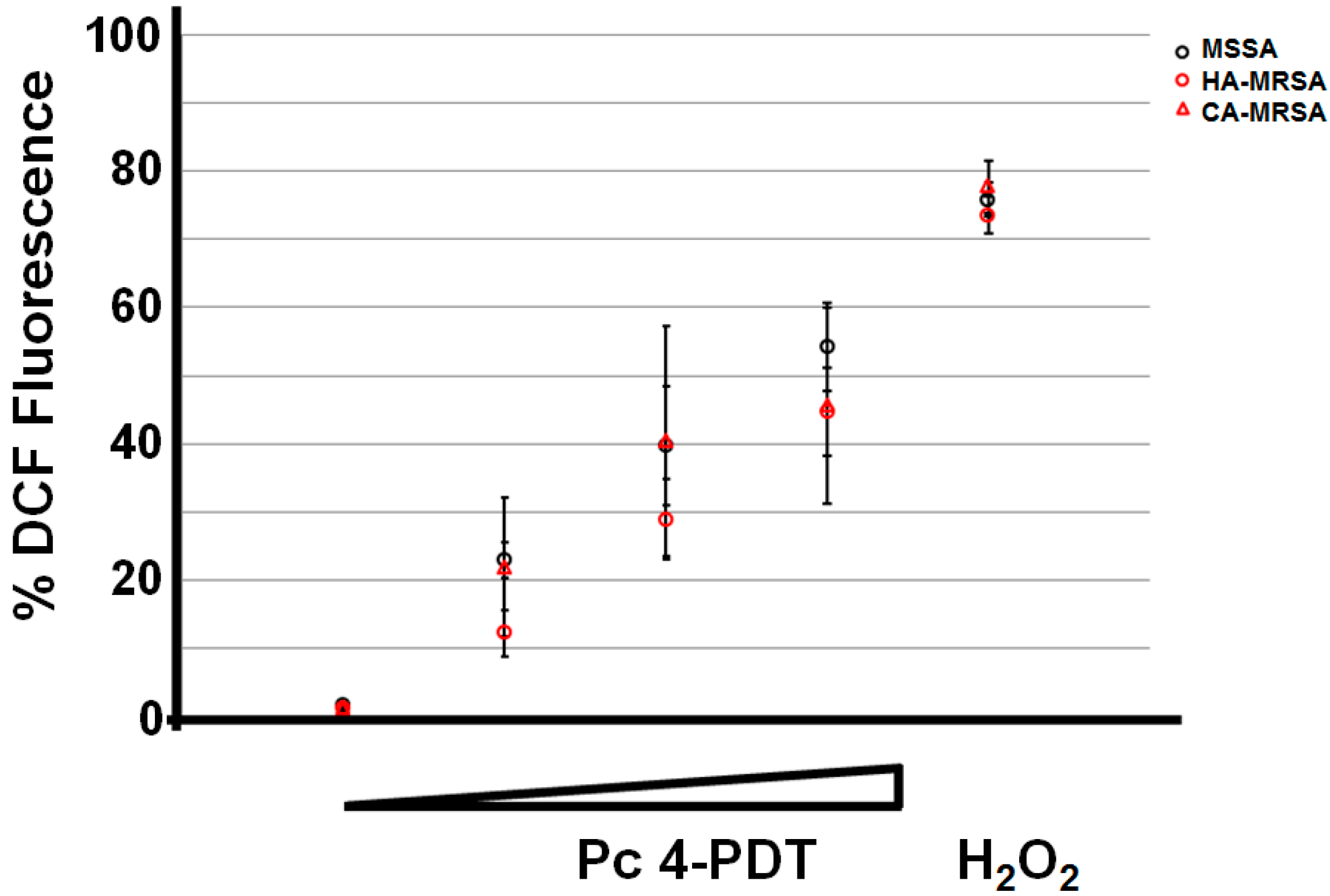
3. Discussions
4. Experimental Section
4.1. Bacteria
4.2. Pc 4-Photodynamic Treatment Conditions
4.3. Confocal Microscopy
4.4. Viable Cell Recovery Assay
4.5. Metabolic XTT Assay
4.6. Flow Cytometry
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Styers, D.; Sheehan, D.J.; Hogan, P.; Sahm, D.F. Laboratory-based surveillance of current antimicrobial resistance patterns and trends among Staphylococcus aureus: 2005 Status in the United States. Ann. Clin. Microbiol. Antimicrob. 2006, 5, 2. [Google Scholar]
- Dantes, R.; Mu, Y.; Belflower, R.; Aragon, D.; Dumyati, G.; Harrison, L.H.; Lessa, F.C.; Lynfield, R.; Nadle, J.; Petit, S. National burden of invasive methicillin-resistant Staphylococcus aureus infections, United States, 2011. JAMA Intern. Med. 2011, 173, 1970–1978. [Google Scholar]
- Boucher, H.W.; Corey, G.R. Epidemiology of methicillin-resistant Staphylococcus aureus. Clin. Infect. Dis. 2008, 46, S344–S349. [Google Scholar] [CrossRef] [PubMed]
- Boucher, H.; Miller, L.G.; Razonable, R.R. Serious infections caused by methicillin-resistant Staphylococcus aureus. Clin. Infect. Dis. 2010, 51, S183–S197. [Google Scholar] [CrossRef] [PubMed]
- Archibald, L.; Phillips, L.; Monnet, D.; McGowan, J.E., Jr.; Tenover, F.; Gaynes, R. Antimicrobial resistance in isolates from inpatients and outpatients in the United States: Increasing importance of the intensive care unit. Clin. Infect. Dis. 1997, 24, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Steinkraus, G.; White, R.; Friedrich, L. Vancomycin MIC creep in non-vancomycin-intermediate Staphylococcus aureus (VISA), vancomycin-susceptible clinical methicillin-resistant S. aureus (MRSA) blood isolates from 2001–05. J. Antimicrob. Chemother. 2007, 60, 788–794. [Google Scholar]
- Wang, G.; Hindler, J.F.; Ward, K.W.; Bruckner, D.A. Increased vancomycin MICs for Staphylococcus aureus clinical isolates from a university hospital during a 5-year period. J. Clin. Microbiol. 2006, 44, 3883–3886. [Google Scholar] [CrossRef] [PubMed]
- Van Hal, S.J.; Fowler, V.G., Jr. Reply to parra-ruiz. Clin. Infect. Dis. 2013, 57, 1219–1220. [Google Scholar] [CrossRef] [PubMed]
- Fridkin, S.K. Vancomycin-intermediate and -resistant Staphylococcus aureus: What the infectious disease specialist needs to know. Clin. Infect. Dis. 2001, 32, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Sievert, D.M.; Rudrik, J.T.; Patel, J.B.; McDonald, L.C.; Wilkins, M.J.; Hageman, J.C. Vancomycin-resistant Staphylococcus aureus in the United States, 2002–2006. Clin. Infect. Dis. 2008, 46, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Tenover, F.C.; Weigel, L.M.; Appelbaum, P.C.; McDougal, L.K.; Chaitram, J.; McAllister, S.; Clark, N.; Killgore, G.; O'Hara, C.M.; Jevitt, L.; et al. Vancomycin-resistant Staphylococcus aureus isolate from a patient in Pennsylvania. Antimicrob Agents Chemother. 2004, 48, 275–280. [Google Scholar]
- Moellering, R.C. Linezolid: The first oxazolidinone antimicrobial. Ann. Intern. Med. 2003, 138, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.K.; Sakoulas, G.; Wennersten, C.; Eliopoulos, G.M.; Moellering, R.C., Jr.; Ferraro, M.J.; Gold, H.S. Linezolid resistance in Staphylococcus aureus: Characterization and stability of resistant phenotype. J. Infect. Dis. 2002, 186, 1603–1607. [Google Scholar] [CrossRef] [PubMed]
- Kurlenda, J.; Grinholc, M. Alternative therapies in Staphylococcus aureus diseases. Acta Biochim. Pol. 2012, 59, 171–184. [Google Scholar] [PubMed]
- Dougherty, T.J. An update on photodynamic therapy applications. J. Clin. Laser Med. Surg. 2002, 20, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Taub, A.F. Photodynamic therapy: Other uses. Dermatol. Clin. 2007, 25, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R.; Newman, E.L. On the mechanism of the tumour-localising effect in photodynamic therapy. J. Photochem. Photobiol. B 1994, 23, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Baldea, I.; Filip, A.G. Photodynamic therapy of cancer: An update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, F.S.; Bentley, M.V. Photodynamic therapy of skin cancers: Sensitizers, clinical studies and future directives. Pharm. Res. 2000, 17, 1447–1455. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R.; Hasan, T. Photodynamic therapy: A new antimicrobial approach to infectious disease. Photochem. Photobiol. Sci. 2004, 3, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Maisch, T. Anti-microbial photodynamic therapy: Useful in the future. Lasers Med. Sci. 2007, 22, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.; Dimaano, M.L.; Oyetakin-White, P.; Retuerto, M.A.; Chandra, J.; Mukherjee, P.K.; Ghannoum, M.A.; Cooper, K.D.; Baron, E.D. Silicon phthalocyanine 4 phototoxicity in Trichophyton rubrum. Antimicrob Agents Chemother. 2014, 58, 3029–3034. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.; Jou, P.C.; Lattif, A.A.; Lee, Y.; Malbasa, C.L.; Mukherjee, P.K.; Oleinick, N.L.; Ghannoum, M.A.; Cooper, K.D.; Baron, E.D. Photodynamic therapy with Pc 4 induces apoptosis of Candida albicans. Photochem. Photobiol. 2011, 87, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Kamp, H.; Tietz, H.J.; Lutz, M.; Piazena, H.; Sowyrda, P.; Lademann, J.; Blume-Peytavi, U. Antifungal effect of 5-aminolevulinic acid PDT in Trichophyton rubrum. Mycoses 2005, 48, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Eruslanov, E.; Kusmartsev, S. Identification of ROS using oxidized DCFDA and flow-cytometry. Methods Mol. Biol. 2012, 59, 57–72. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dimaano, M.L.; Rozario, C.; Nerandzic, M.M.; Donskey, C.J.; Lam, M.; Baron, E.D. The Photodynamic Antibacterial Effects of Silicon Phthalocyanine (Pc) 4. Int. J. Mol. Sci. 2015, 16, 7851-7860. https://doi.org/10.3390/ijms16047851
Dimaano ML, Rozario C, Nerandzic MM, Donskey CJ, Lam M, Baron ED. The Photodynamic Antibacterial Effects of Silicon Phthalocyanine (Pc) 4. International Journal of Molecular Sciences. 2015; 16(4):7851-7860. https://doi.org/10.3390/ijms16047851
Chicago/Turabian StyleDimaano, Matthew L., Chantal Rozario, Michelle M. Nerandzic, Curtis J. Donskey, Minh Lam, and Elma D. Baron. 2015. "The Photodynamic Antibacterial Effects of Silicon Phthalocyanine (Pc) 4" International Journal of Molecular Sciences 16, no. 4: 7851-7860. https://doi.org/10.3390/ijms16047851
APA StyleDimaano, M. L., Rozario, C., Nerandzic, M. M., Donskey, C. J., Lam, M., & Baron, E. D. (2015). The Photodynamic Antibacterial Effects of Silicon Phthalocyanine (Pc) 4. International Journal of Molecular Sciences, 16(4), 7851-7860. https://doi.org/10.3390/ijms16047851




