The Effects of Marine Carbohydrates and Glycosylated Compounds on Human Health
Abstract
:1. Introduction
2. Type of Sugar-Bound Compounds from Marine Organisms
2.1. Polysaccharides and Oligosaccharides
2.2. Glycosides
3. Biological Activities of Marine Carbohydrate and Glycosides
3.1. Antioxidant Activity
3.1.1. Polysaccharides from Hyriopsis cumingii (HCPS-3)
3.1.2. Crude Endopolysaccharides from Cerrena unicolor (c-EPL)
3.1.3. Polysaccharides from Mytilus coruscus (MP-I)
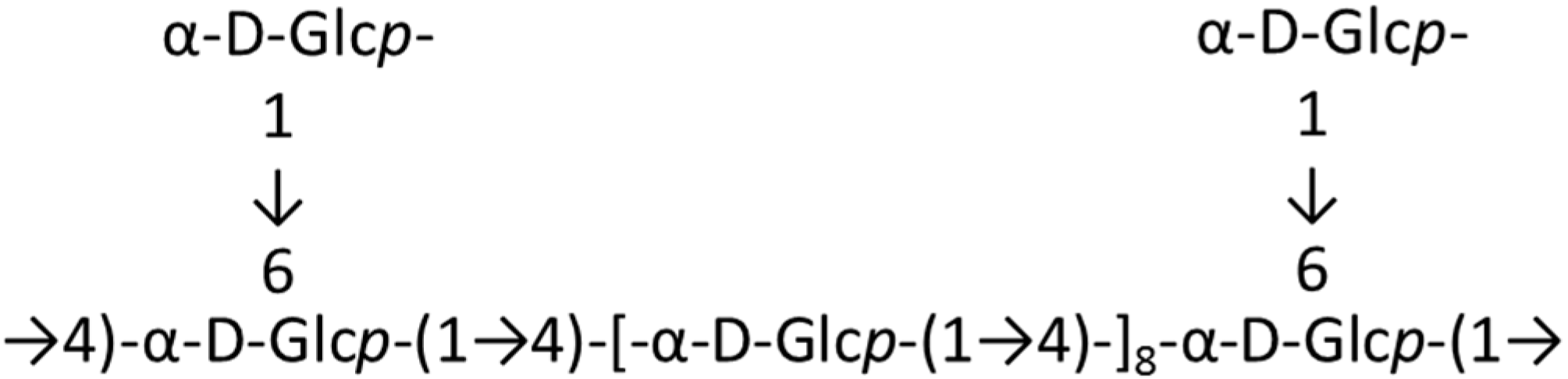
3.1.4. Polysaccharides from Mytilus edulis (HWS)
3.1.5. Polysaccharides from Penicillium sp. F23-2 (PS1-1, PS1-2 and PS2-1)
3.1.6. Sulfated Polysaccharide from Sargassum swartzii
3.1.7. Chitin Oligosaccharides from Crab Chitin Obtained by Acid Hydrolysis (NA-COS)
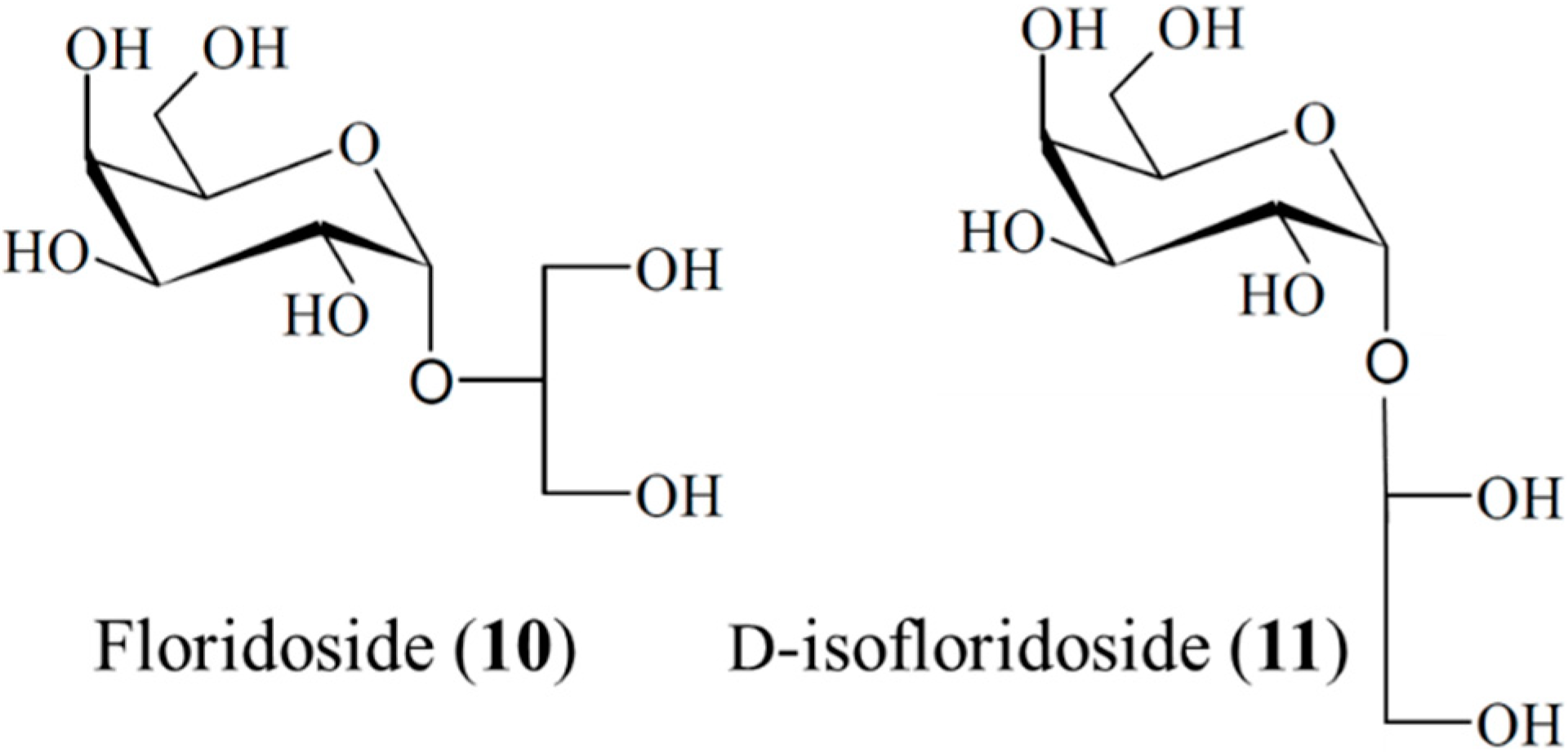
3.1.8. Floridoside and d-Isofloridoside
3.2. Antidiabetic Activity
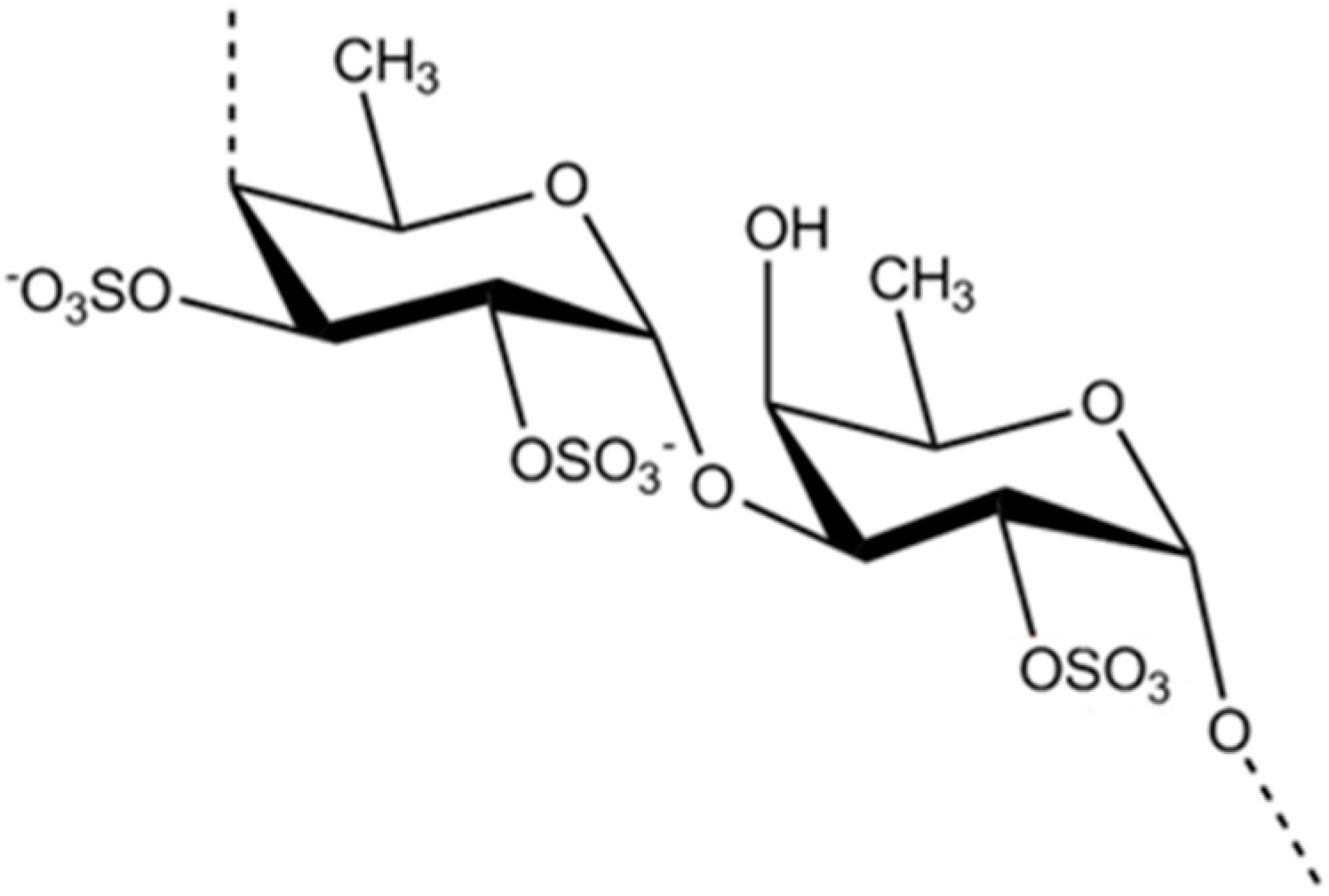
3.2.1. Fucoidan
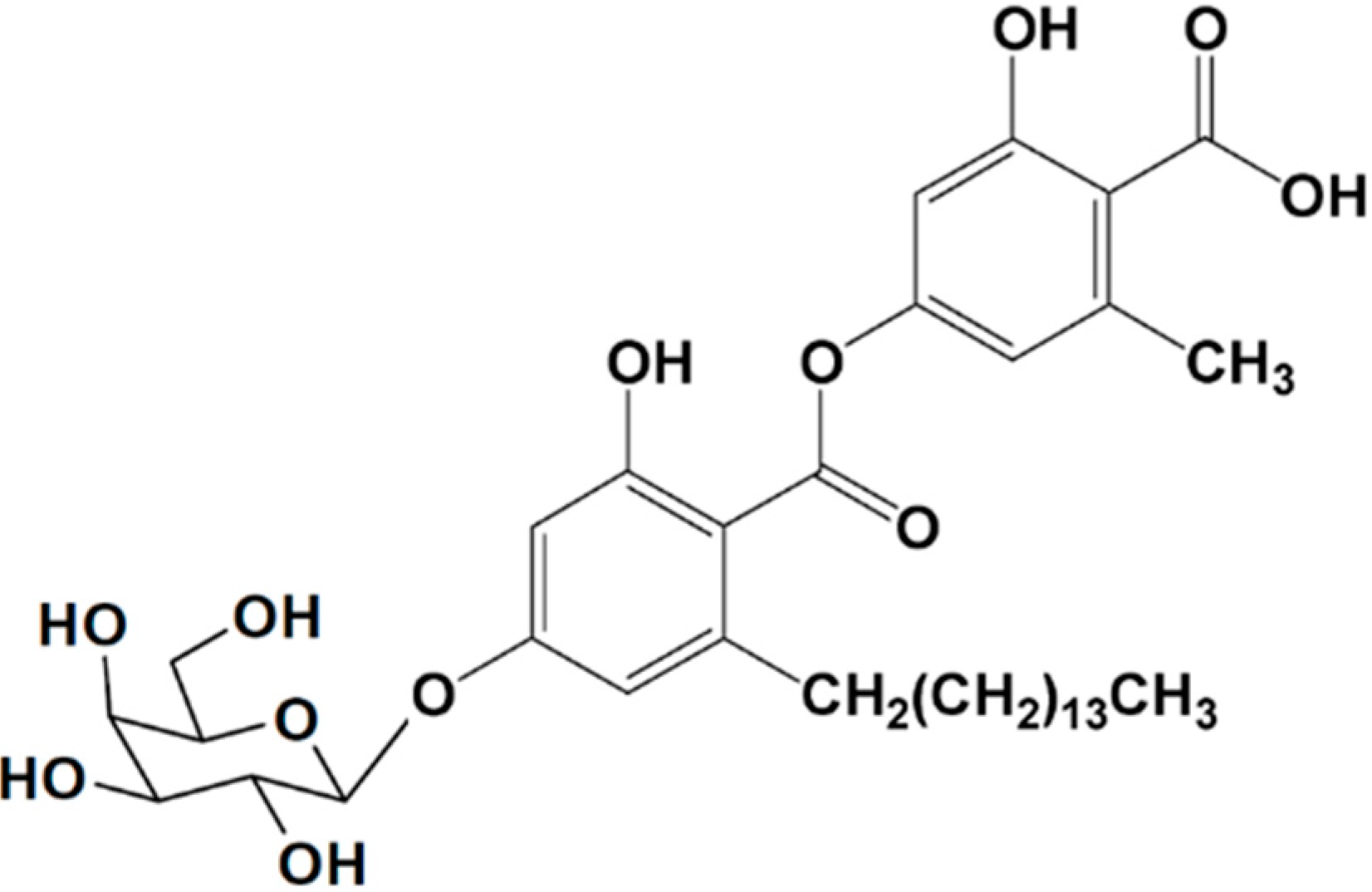
3.2.2. Aquastatin A
3.3. Antibacterial Activity
3.3.1. Polysaccharides from Sepia pharaonis
3.3.2. Gladius Polysaccharide
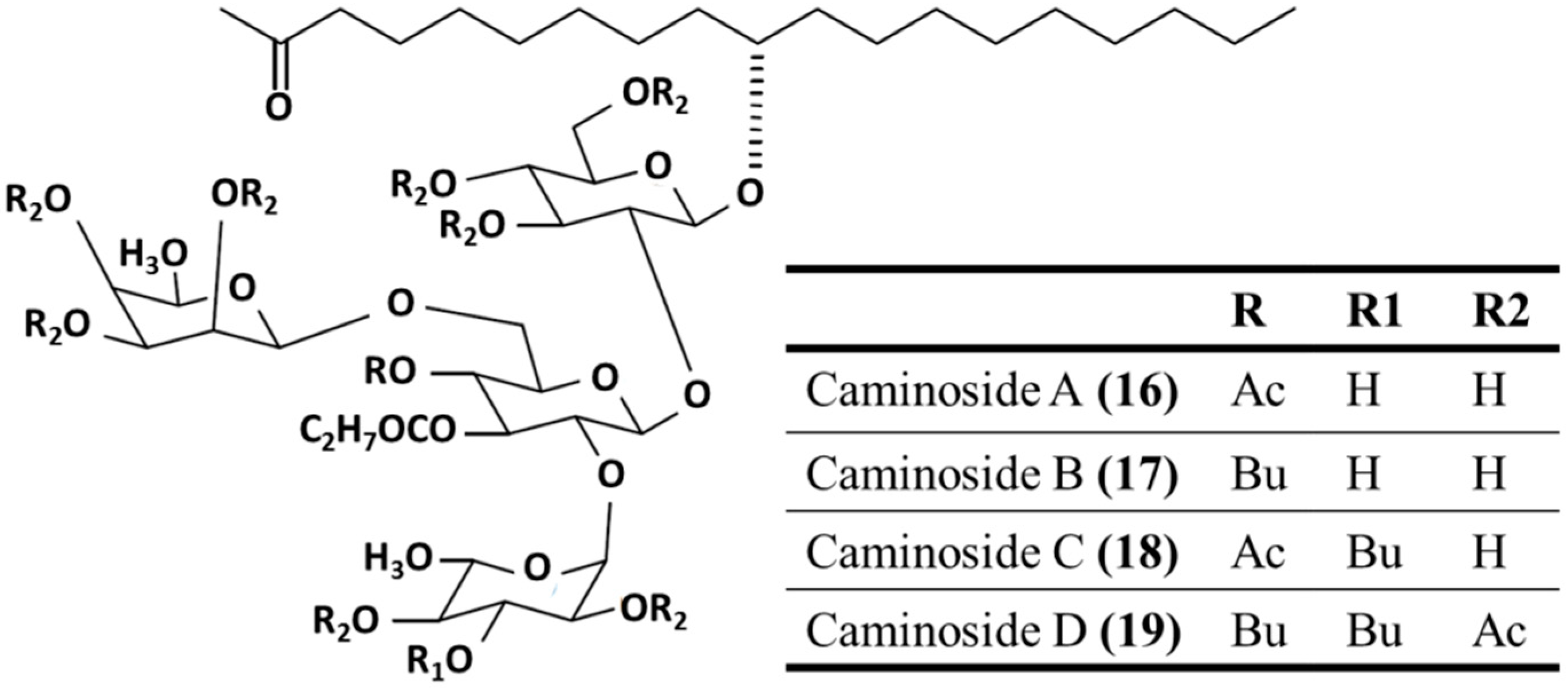
3.3.3. Caminosides A–D
3.3.4. Sulfate Galactan from Caminus aerea
3.4. Antifungal Activity
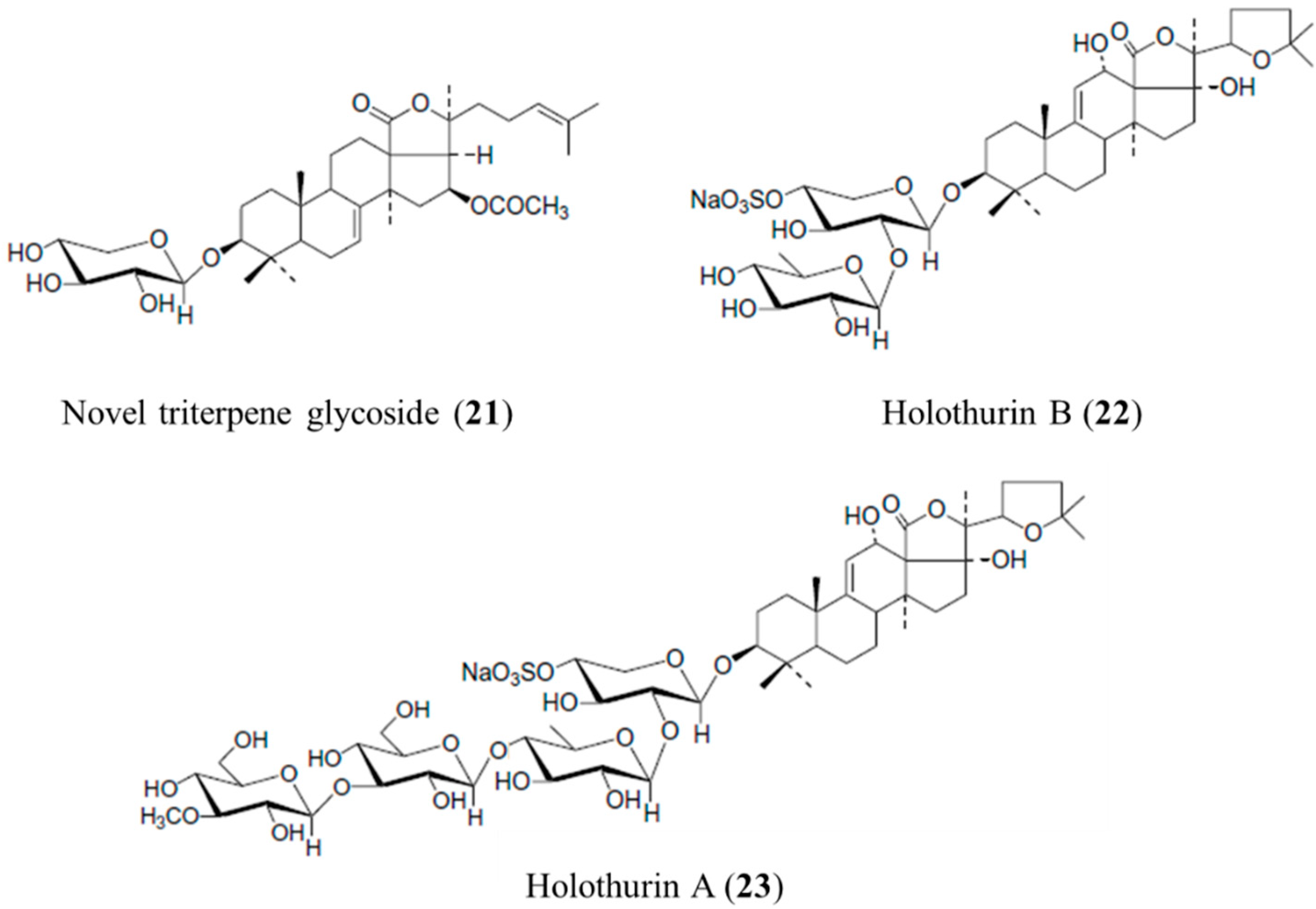
3.4.1. Novel Triterpene Glycoside, Holothurin B and Holothurin A
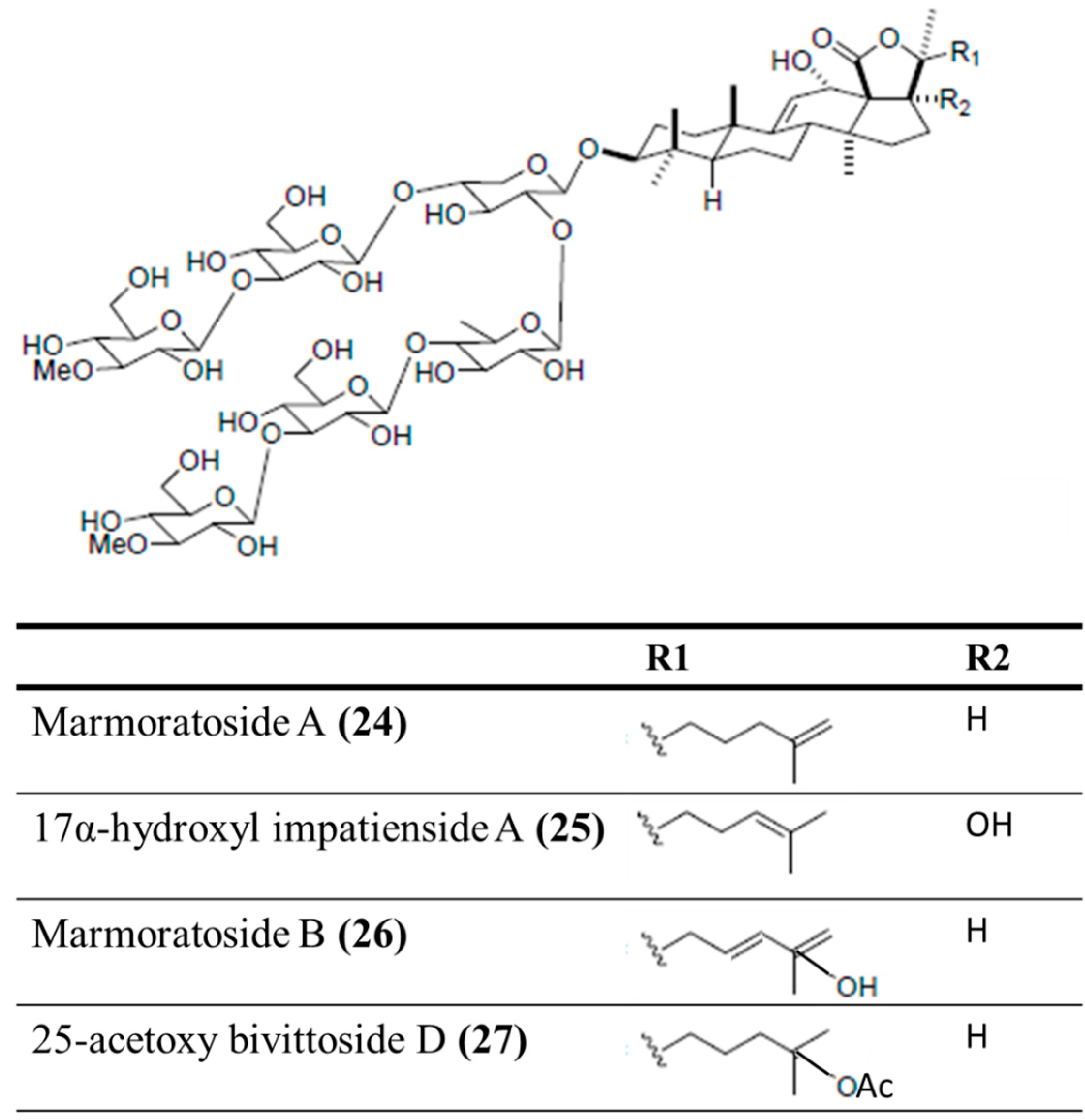
3.4.2. Marmoratoside A, 17α-Hydroxy Impatienside A, Marmoratoside B and 25-Acetoxy Bivittoside D
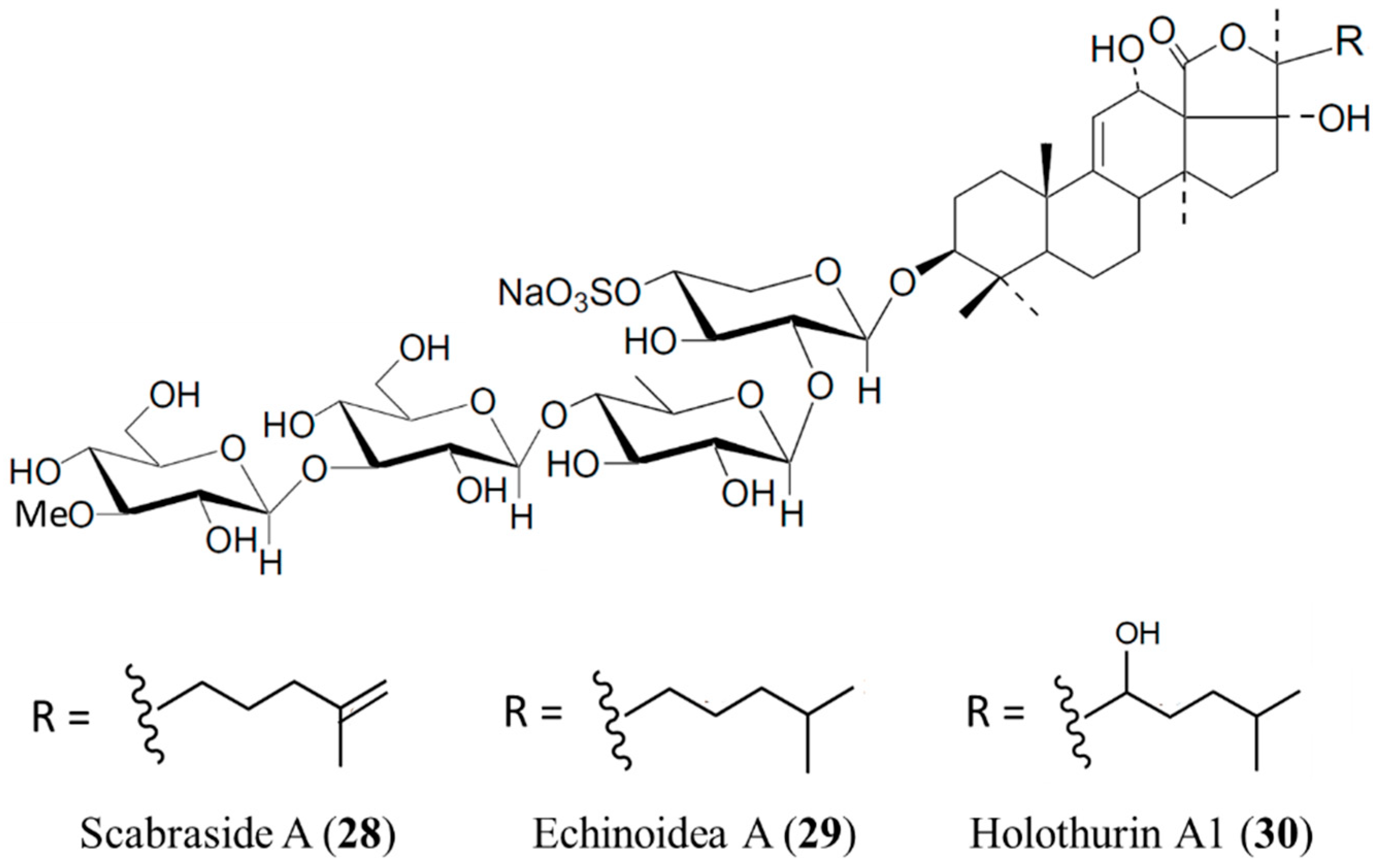
3.4.3. Scabraside A, Echinoidea A, and Holothurin A1
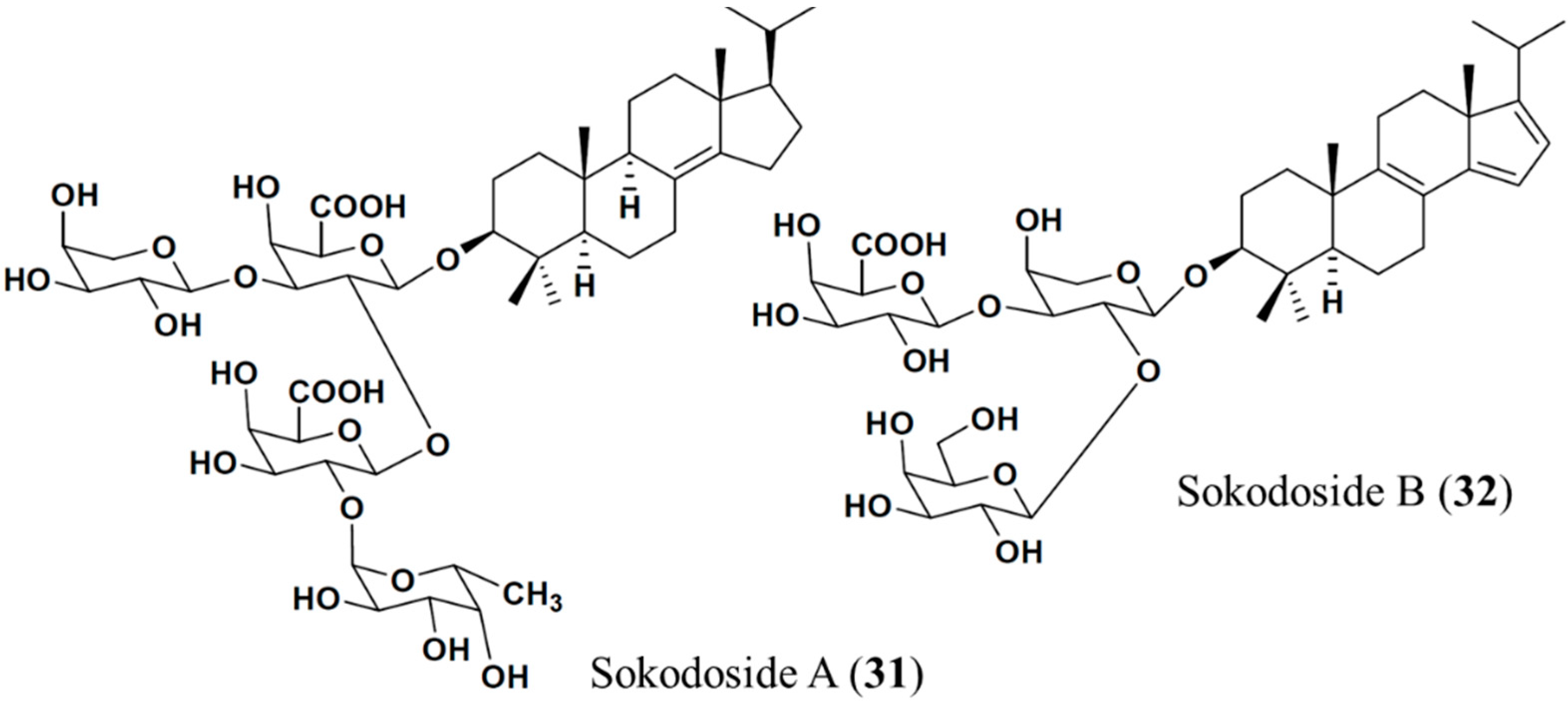
3.4.4. Sokodoside A and B
3.4.5. Variegatusides C–F
3.5. Anticoagulant Activity
3.5.1. Fucoidans
3.5.2. Acidic Polysaccharide from Laminaria cichorioides
3.5.3. Sulfated Polysaccharides from Brown Algae
3.5.4. Fucosylated Polysaccharide Sulfate (AMP-2)
3.5.5. Anticoagulant Polysaccharide from Lomentaria catenata
3.5.6. Halymenia floresia Sulfated Polysaccharides (Hf-SP)
3.5.7. Asparagopsis taxiformis Sulfated Polysaccharide (AtSP)
3.5.8. Caulerpa cupressoides var. lycopodium Sulfated Polysaccharides (CuSP)
3.6. Antiprotozoal Activity
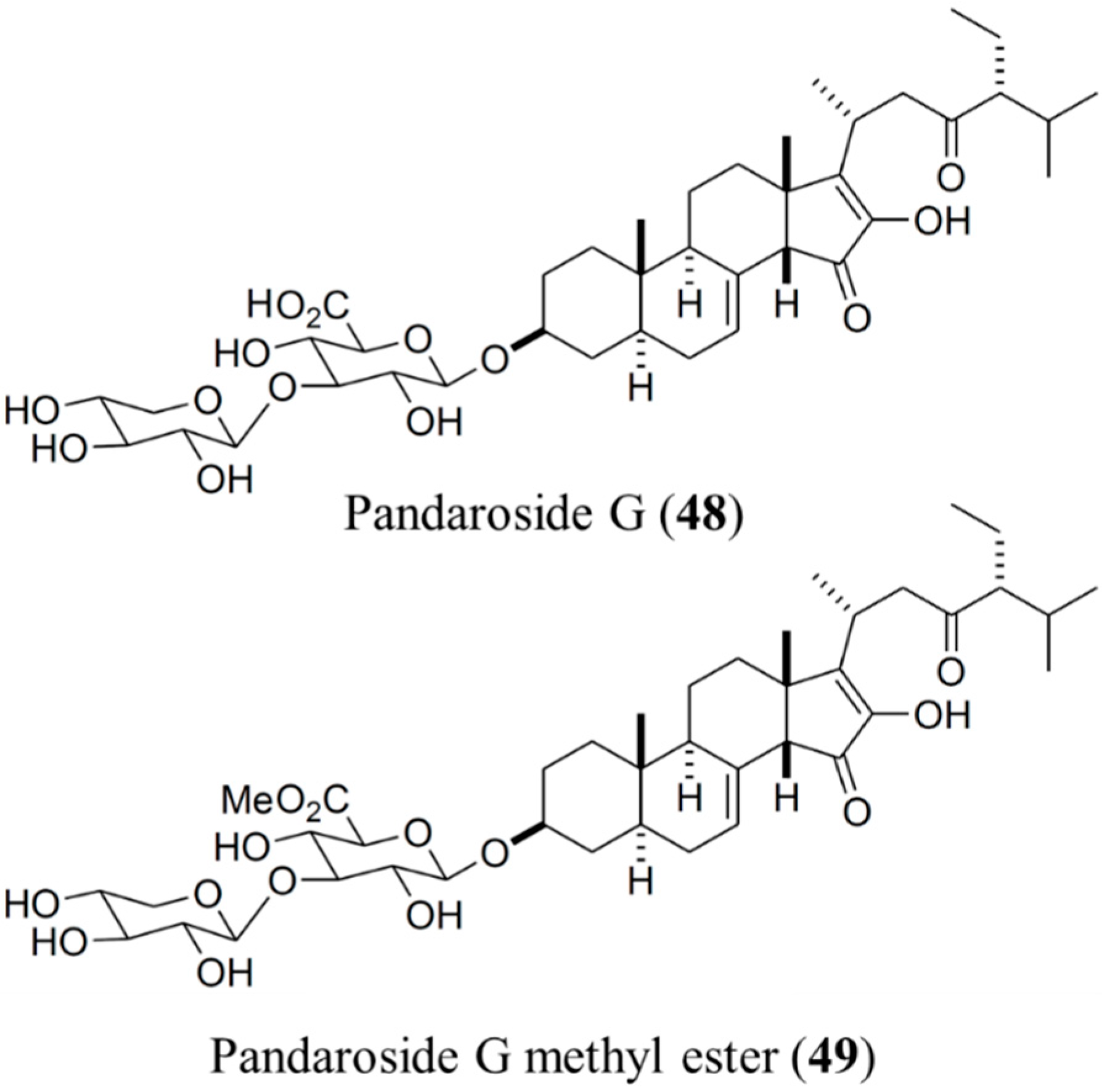
3.6.1. Pandaroside G and Pandaroside G Methyl Ester
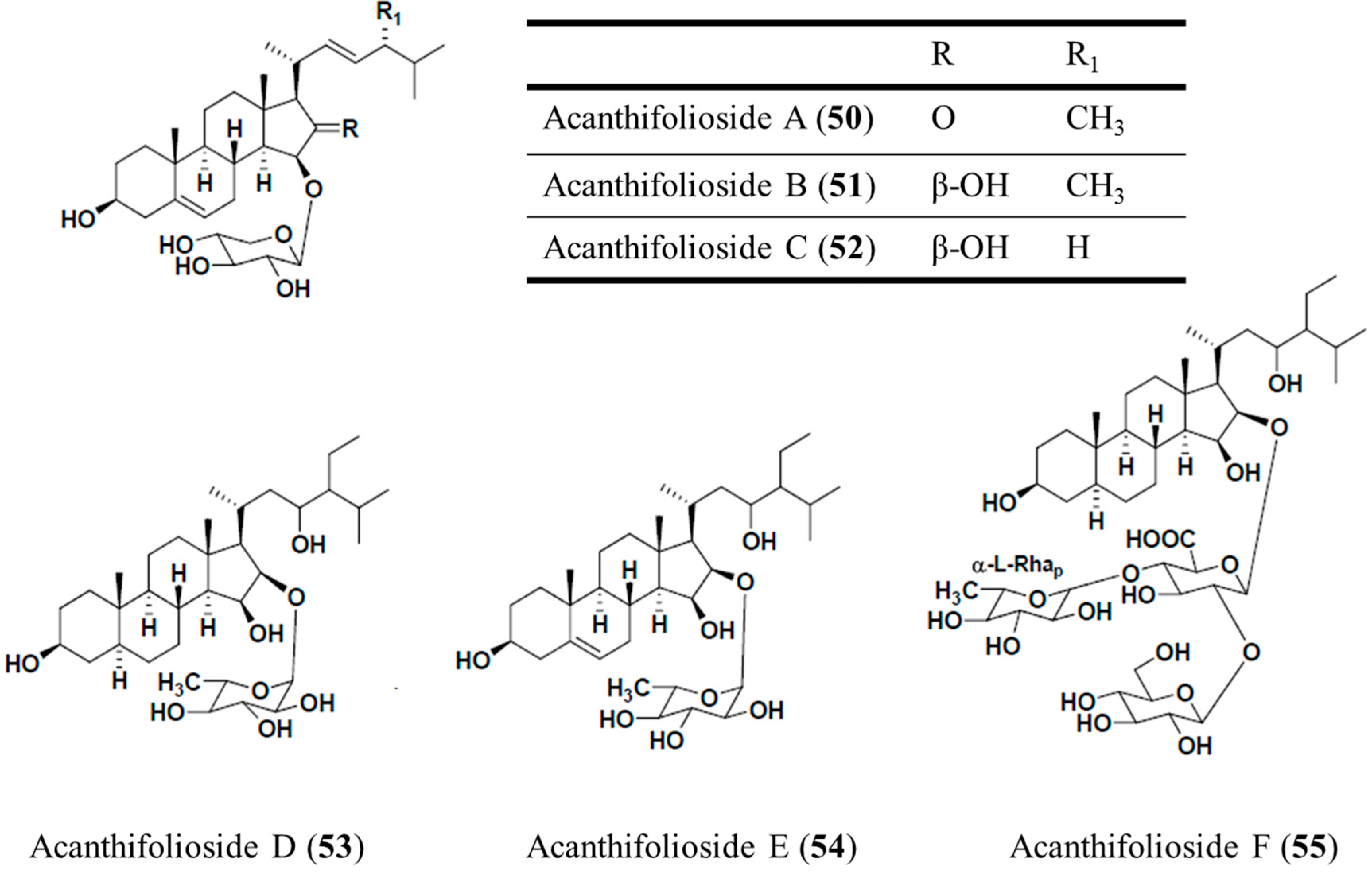
3.6.2. Acanthifoliosides A–F
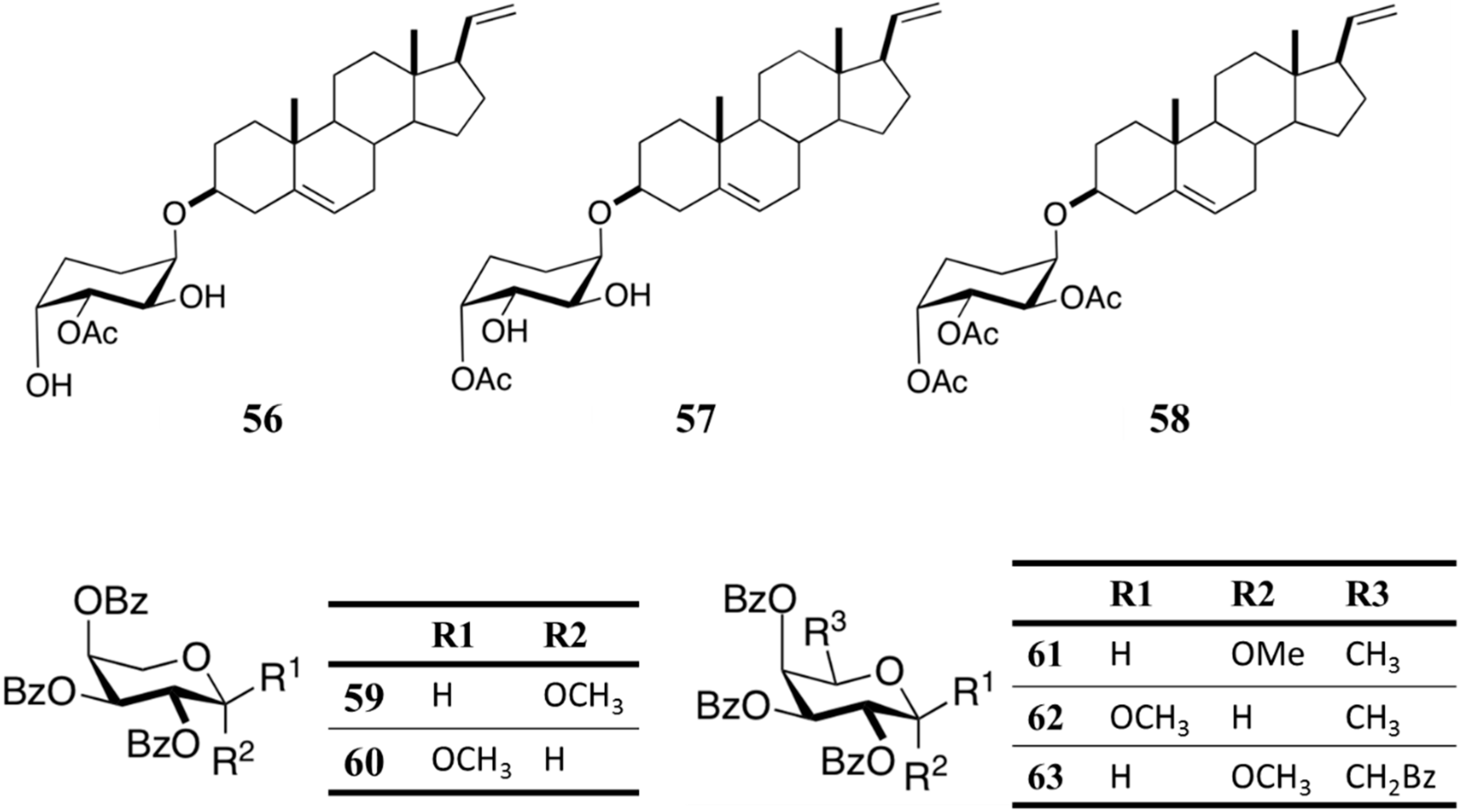
3.7. Anti-Malarial Activity
Glycosides From Muricea austera
3.8. Anti-Viral Activity
3.8.2. PSC and PBT
3.8.3. GFP and GLPE
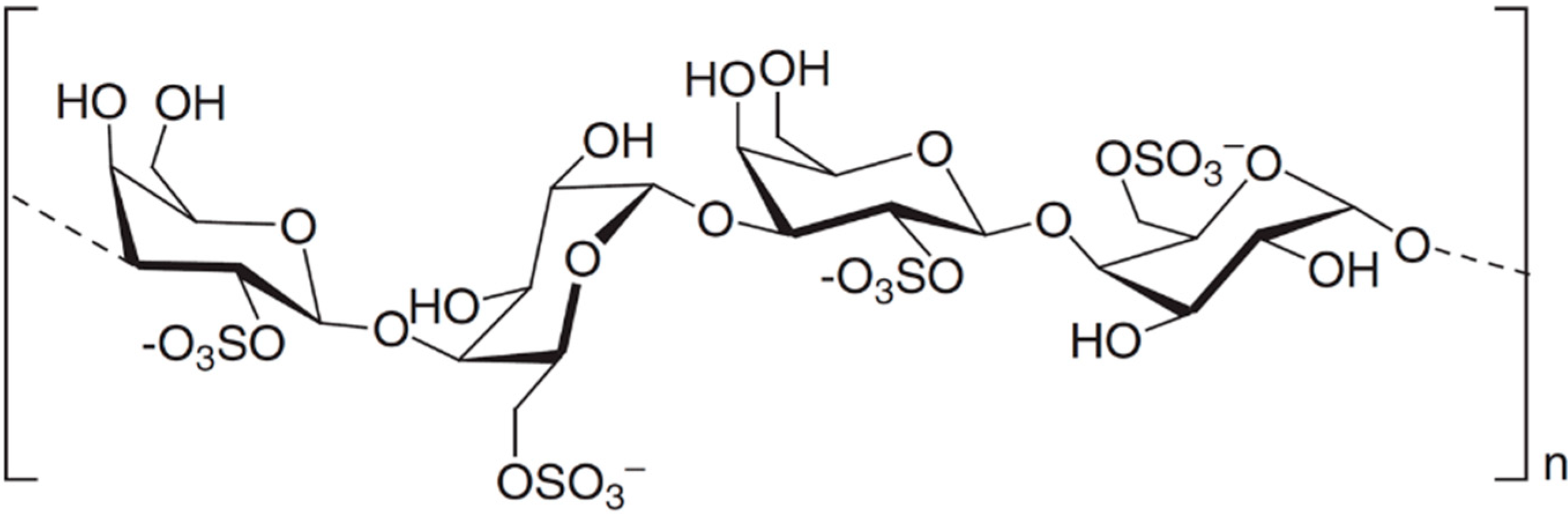
3.8.4. d,l-Galactan Hybrid C2S-3
3.8.5. Sulfated Xylomannan from Scinaia hatei
3.8.6. Galactofucan Fraction EA1-20
3.8.7. Sulfated Fucans
3.8.8. Sulfated Polymannuroguluronate
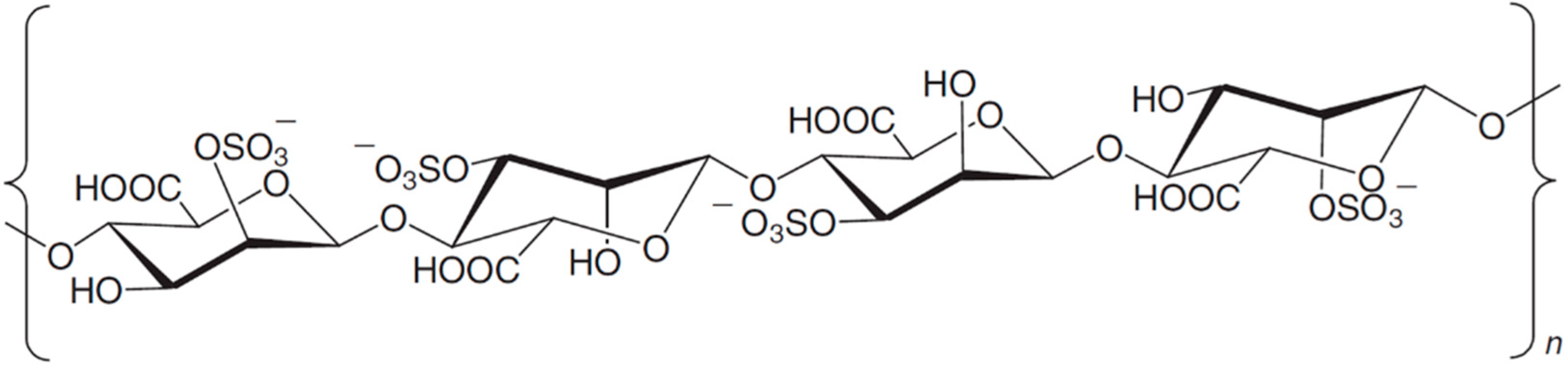
3.8.9. Carraguard
3.8.10. Aminoethyl-Chitosan, Sulfated Chitin and Sulfated Chitooligosaccharide
3.9. Anti-Imflammatory Activity
3.9.1. Styela plicata Dermatan Sulfate
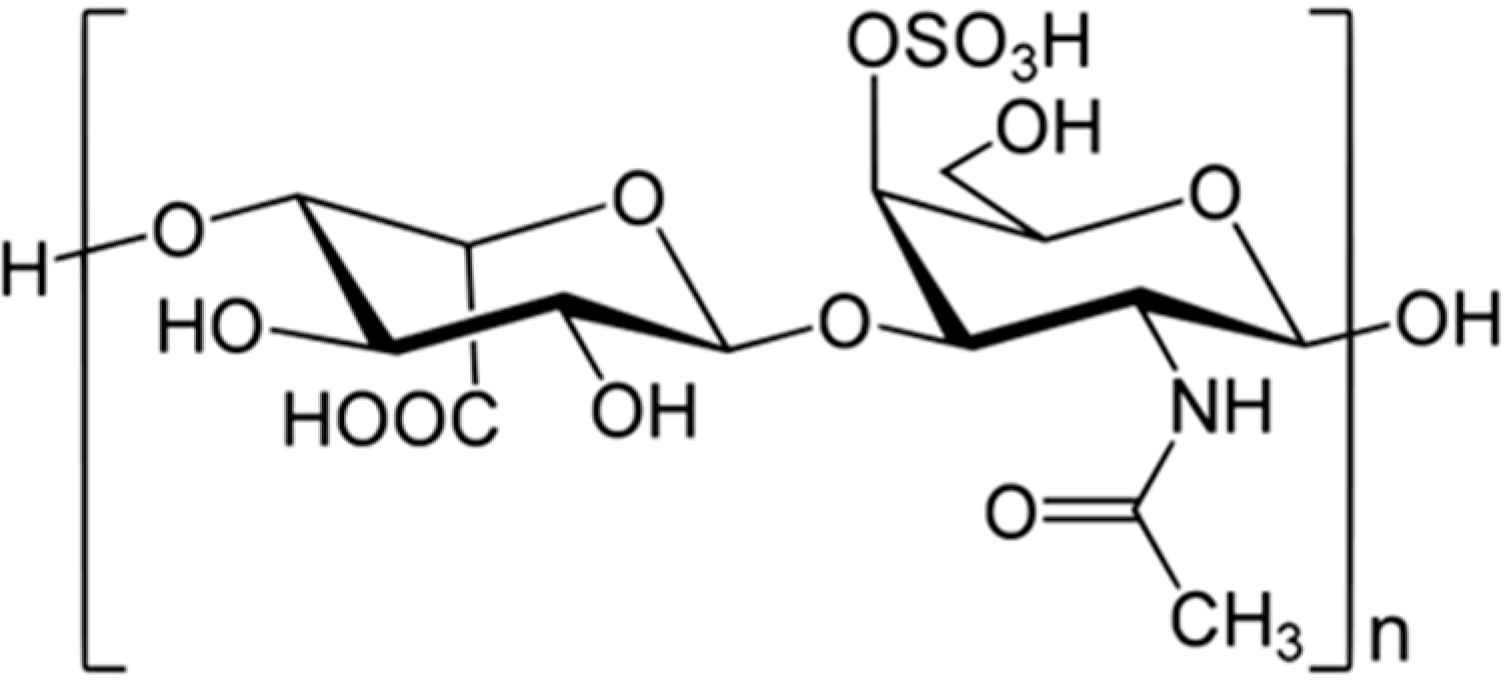
3.9.2. Carijoside A
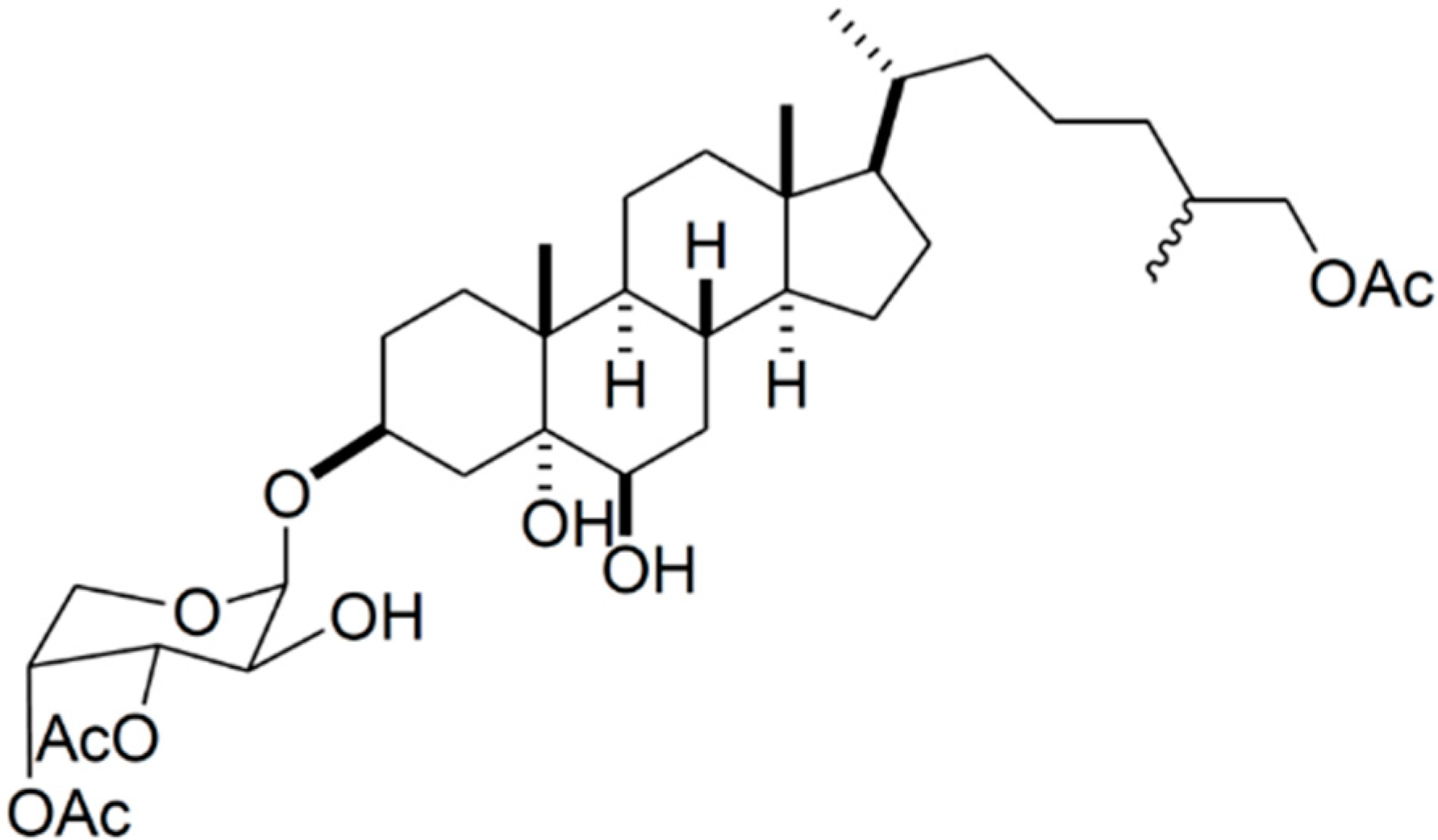
3.10. Immunomodulating Activity
3.10.1. Laminarin Polysaccharides and Oligosaccharides
3.10.2. ASLP
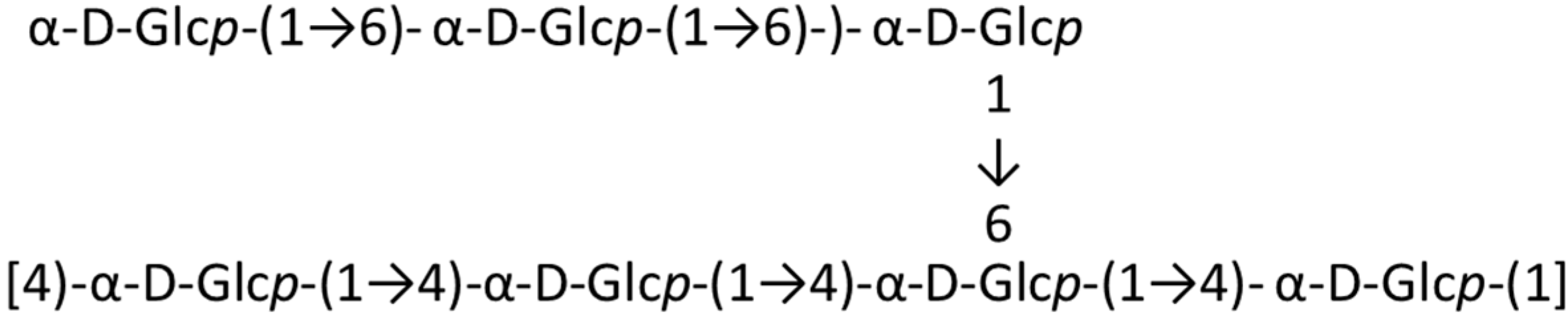
3.10.3. HCLPS-1
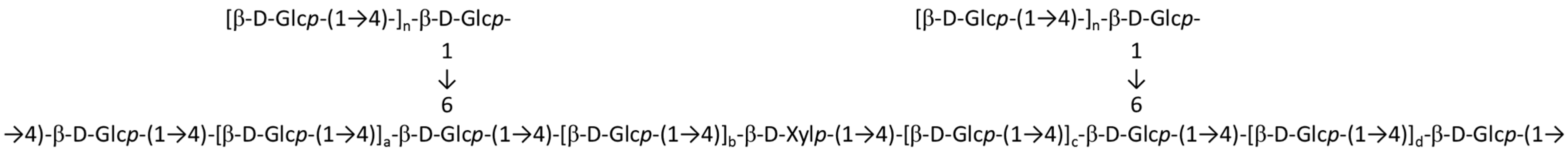
3.10.4. Floridoside
3.10.5. Frondoside A
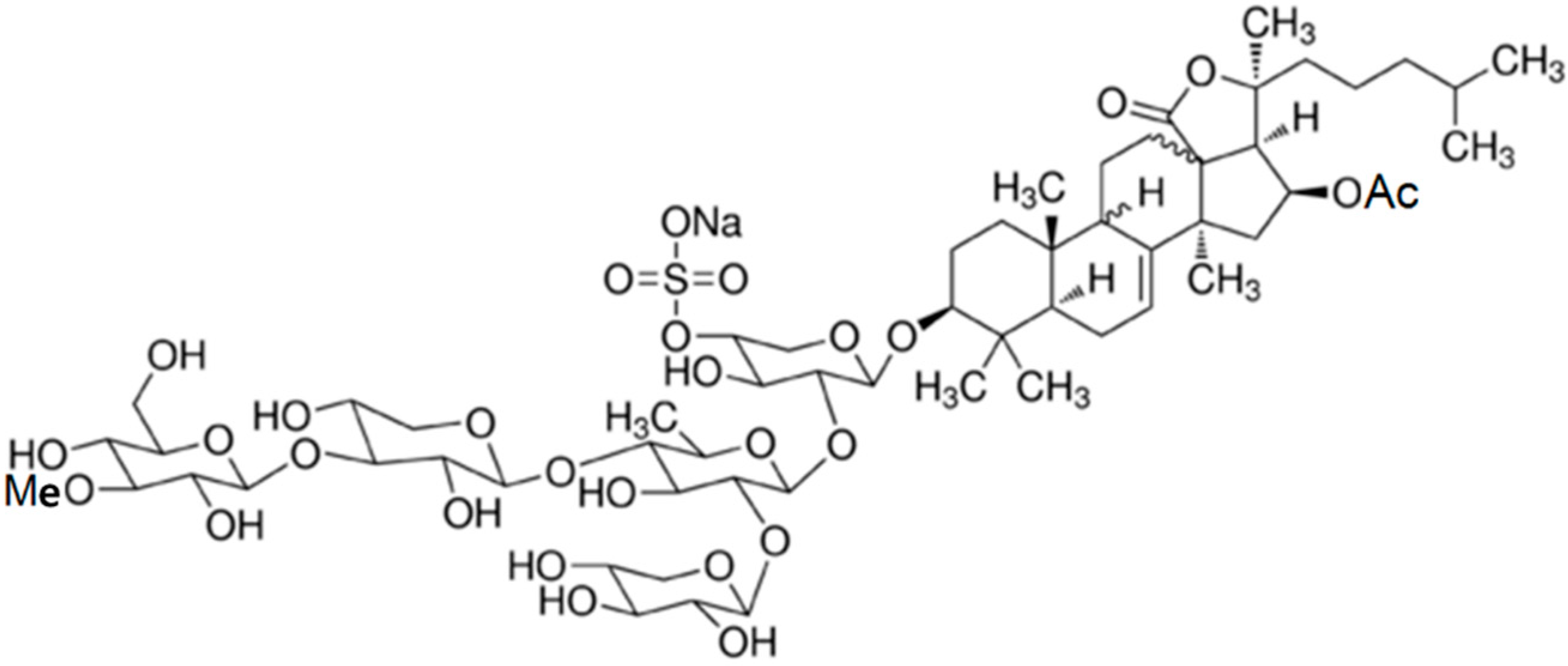
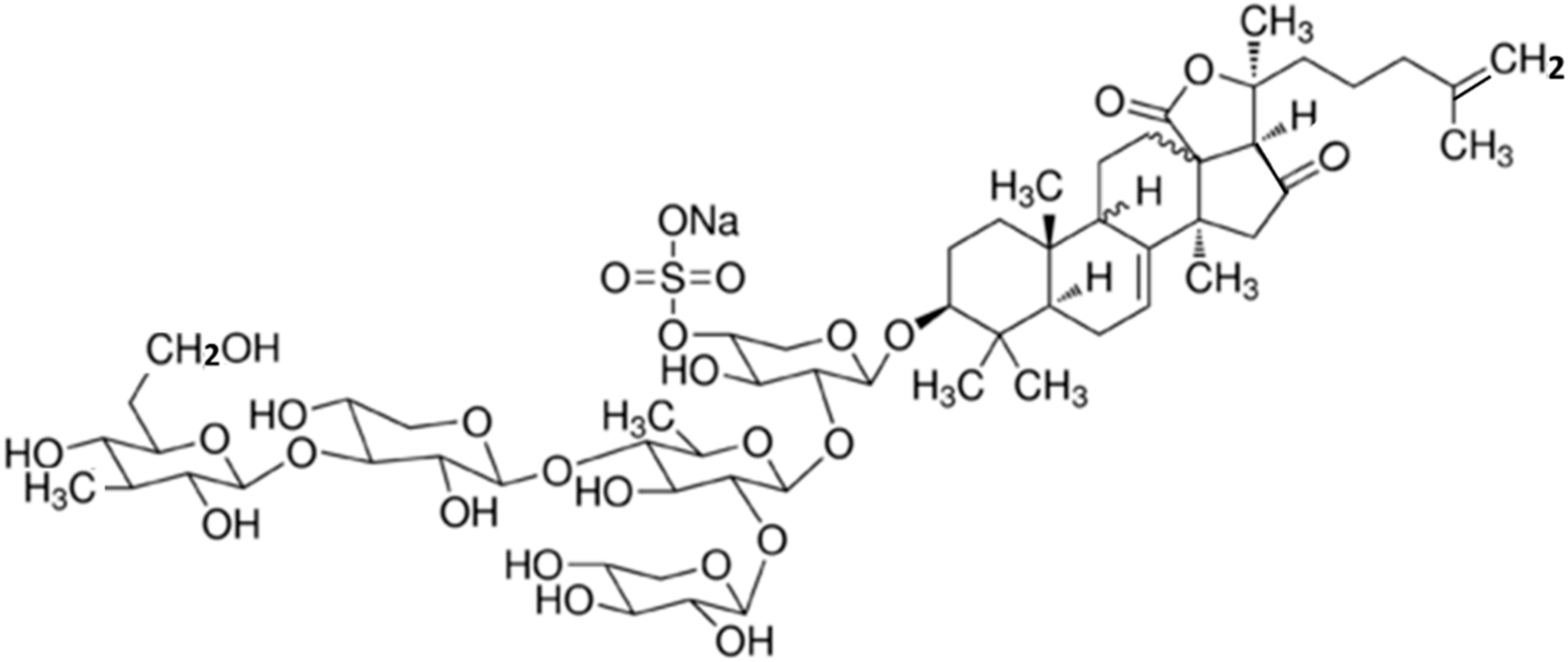
3.10.6. Cucumariosides
4. Conclusions
| Activity | Compound/Chemistry | Source of Original Carbohydrate | Pharmacologic Activity | Inhibitory Concentrations | Refs. |
|---|---|---|---|---|---|
| Antioxidant | HCPS-3 (1)/polysaccharides | Pearl mussel: Hyriopsis cumingii | Inhibition of MDA formation in vivo | ND | [34] |
| Antioxidant | c-EPL (2)/polysaccharide | Fungus: Cerrena unicolor | Scavenging activity against DPPH− and ABTS | ND | [35] |
| Antioxidant | MP-I (3)/polysaccharide | Mussel: Mytilus coruscus | ALT, AST, and MDA inhibition in vivo | ND | [36] |
| Antioxidant | HWS (4)/polysaccharide | Mussel: Mytilus coruscus | Scavenging activity against O−, OH−, and NO2− | ND | [37] |
| Antioxidant | PS1-1 (5), PS1-2 (6), PS2-1 (7)/polysaccharide | Fungus: Penicillium sp. F23-2 | Scavenging activity against DPPH−, O−, and OH− | 2.53–6.81 mg/mL (EC50) | [38] |
| Antioxidant | S. swartzii sulfate polysaccharide (8) | Algae: Sargassum swartzii | Scavenging activity against ABTS, H2O2 and DPPH− | ND | [39] |
| Antioxidant | NA-COS (9)/oligosaccharide | Crab: Chionoecetes opilio | Scavenging activity against DPPH−, hydroxyl radical, and alkyl radical | 0.8–1.75 mg/mL (IC50) | [41] |
| Antioxidant | Floridoside (10), d-isofloridoside (11)/glycoside | Algae: Laurencia undulate | Inhibition of free-radical oxidative stress | 22–43 μM (IC50) | [42] |
| Antidiabetic | Fucoidan (12)/polysaccharide | Algae: Fucus vesiculosus, Ecklonia kurome, Undaria pinnatifida | Decrease blood glucose, total cholesterol, and fat adiponectin levels in vivo | ND | [45,46,47,48,49] |
| Antidiabetic | Aquastatin A (13)/glycoside | Fungus: Cosmospora sp. SF-5060 | Tyrosine phosphataseinhibition | 0.19 μM (IC50) | [50] |
| Antibacterial | S. pharaobis polysaccharide (14) | Cuttlebone: Sepia pharaonis | Staphylococcus aureus, Escherichia coli, Salmonella typhii, Vibrio cholerae, Klebsiella oxytoca, Salmonella paratyphi, Proteus mirabilis, and Staphylococcus pyogenes inhibition | 40–100 mg/mL (MIC) | [52] |
| Antibacterial | Gladius polysaccharide (15) | Cuddalore: Loligo duvauceli, Mudasalodai: Doryteuthis sibogae | Bacillus subtilis, Shigella sp., S. typhii, Vibrio parahaemolyticus, Klebsiella pneumonia, and E. coli inhibition | 80–100 mg/mL (MIC) | [53] |
| Antibacterial | Caminoside A–D (16–19)/glycoside | Sponge: Chaetomorpha sphaeoroconia | Methicillin-resistant S. aureus, vancomycin-resistant Enterococcus, and E. coli inhibition | 6.3–12.5 μg/mL (MIC) | [54,55,56] |
| Antibacterial | C. sphaeoroconia sulfate galactan (20)/polysaccharide | Algae : Caminus sphaeoroconia | B. subtilis, Micrococus lutens and S.aureus inhibition | 40 mg/mL (MIC, MBC: S. aureus) | [57] |
| Antifungal | Triterpene glycoside (21), holothurin B (22), holothurin A (23)/glycoside | Sea cucumber: Actinopyga lecanora | Candida albicans, Cryptococcus neoformans, Sporothrix schenckii, Trychophyton mentagrophytes, and Aspergillus fumigatus inhibition | 1.56–50 μg/mL (MIC) | [60] |
| Antifungal | Marmoratoside A (24), 17α-hydroxy impatienside (25), marmoratoside B (26), 25-acetoxy bivittoside (27)/glycoside | Sea cucumber: Bohadschia marmorata | C. albicans, C. neoformans, A. fumigatus, Trichophyton rubrum, Candida tropicalis, and Candida krusei inhibition | 1–16 μg/mL (MIC80) | [61] |
| Antifungal | Scabraside A (28), ethinodea A (29), holothurin A1 (30)/glycoside | Sea cucumber: Holothuria scabra | C. albicans, C. neoformans, Candida pseudotropicalis, T. rubrum, Fonsecaea compacta, A. fumigatus, and Microsporum gypseum inhibition | 1–16 μg/mL (MIC80) | [62] |
| Antifungal | Sokodoside A (31), B (32)/glycoside | Sponge: Erylus placenta | Mortierella ramanniana, Saccharomyces cerevisiae inhibition | ND | [64] |
| Antifungal | Variegatuside C–F (33–36)/glycoside | Cucumber: Stichopus variegates | C. albicans, C. neoformans, C. pseudotropicalis, C. parapsilosis, C. tropicalis and Microsporum gypseum inhibition | 3.4–12.5 μg/mL (MIC80) | [65] |
| Anticoagulant | Fucoidan (12)/polysaccharide | Algae: Fucus vesiculosus, Ecklonia kurome, Undaria pinnatifida | Thrombin and factor Xa inhibition in vitro and in vivo | ND | [69,70] |
| Anticoagulant | L. catenata acid polysaccharide (37) | Algae: Lomentaria catenata | Thrombin and factor Xa inhibition in vitro | 0.045–25.47 USP units/mg | [71] |
| Anticoagulant | Brown algae Sulfate polysaccharide (38–42) | Algae : Sargassum tenerrimum (38), S. wightii (39), Turbinaria conoides (40), T. ornata (41), Padina tetrastromatica (42) | Thrombin inhibition and heparinoid activity in vitro | 14.5–25.41 haparin USP units/mg | [72] |
| Anticoagulant | AMP-2 (43)/polysaccharide | Cucumber: Acaudina molpadioidea | Thrombin, prothrombin and thrombin inhibition in vitro | 11.8–34.3 at 0.5 mg/mL | [73] |
| Anticoagulant | L. catenata polysaccharide (44) | Algae: Lomentaria catenata | Thrombin and factor Xa inhibition in vitro | 183 IU/mg | [74] |
| Anticoagulant | Hf-SP/polysaccharide (45) | Seaweed: Halymenia floresia | Thrombin inhibition in vitro | 10.72 IU/mg | [75] |
| Anticoagulant | A. taxiformis sulfated polysaccharide (46) | Algae: Asparagopsis taxiformis | Thrombin inhibition in vitro | 259.8 μg/mL | [76] |
| Anticoagulant | C. cupressoides polysaccharide 2 (47) | Algae: Caulerpa cupressoides var. lycopodium | Thrombin and factor Xa inhibition in vitro | ND | [77] |
| Antiprotozoal | Pandaroside G (48), pandaroside G methyl ester (49)/glycoside | Sponge: Pandaros acanthifolium | Trypanosoma brucei rhodesiense and Leishmania donovani inhibition | 0.038–1.3 μM (IC50) | [78] |
| Antiprotozoal | Acanthifoliosides A–F (50–55)/glycoside | Sponge: Pandaros acanthifolium | T. brucei rhodesiense and L. donovani inhibition | 5.7–94.8 μM (IC50) | [79] |
| Antimalarial | M. austere glycosides (56-63) | Octocoral: Muricea austere | Plasmodium falciparum inhibition | 21–80 μM (IC50) | [82] |
| Antiviral | Naviculan (64)/polysaccharide | Diatom: Navicula directa | HSV-1 and HSV-2 inhibition | 7–14 μM (IC50) | [85] |
| Antiviral | PSC (65), PBT (66)/polysaccharide | Algae: Sphaerococcus coronopifolius (PSC), Boergeseniella thuyoides (PBT) | HSV-1 inhibition | 4.1–17.2 μg/mL (EC50) | [86] |
| Antiviral | Sulfate GFP (67), GLPE (68)/polysaccharide | Algae: Grateloupia longifolia | HIV-1 inhibition | 0.003–0.010 μg/mL (EC50) | [87] |
| Antiviral | d,l-galatan hybrid C2S-3 (69)/polysaccharide | Seaweed: Cryptonemia crenulata | Dengue type 2 inhibition | 0.8–16 μg/mL (IC50) | [88] |
| Antiviral | Sulfate xylomannan (70)/polysaccharide | Seaweed: Scinaia hatei | HSV-1 and HSV-2 inhibition | 0.5–1.4 μg/mL (IC50) | [89] |
| Antiviral | Galactofucan EA-20 (71)/polysaccharide | Algae: Adenocystis utricularis | HIV-1 inhibition | 0.6 μg/mL (IC50) | [90] |
| Antiviral | Sulfate fucans (72)/polysaccharide | Seaweed: Dictyota mertensii, Lobophora variegata, Spatoglossum schroederi, Fucus vesiculosus | HIV-1 reverse transcriptase inhibition | ND | [91] |
| Antiviral | Sulfate SPMG (73)/polysaccharide | Algae: Laminaria japonica | Inhibition of HIV-1 infection | ND | [92,93] |
| Antiviral | Carraguard (74)/polysaccharide | Algae: Solieria chordalis | HIV-1 inhibition | ND | [94] |
| Antiviral | Aminoethyl-chitosan (75), sulfated chitin (76), chitosan (77)/polysaccharide | Fungus: Zygomycetes, Alage: Chlorella sp. Crab, crayfish, periwinkle and shrimp | HIV-1 inhibition | 17 μg/mL (IC50) | [95,98] |
| Antiviral | Sulfated SCOS (78)/oligosaccharide | Fungus: Zygomycetes, Alage: Chlorella sp. Crab, crayfish, periwinkle and shrimp | HIV-1 inhibition | 1.4–7.76 μg/mL (IC50) | [99] |
| Anti-inflammatory | Dermatan sulfate (79)/polysaccharide | Ascidian: Styela plicata | Colonic inflammation inhibition | 8 mg/kg (IC50) | [102] |
| Anti-inflammatory | Carijoside A (80)/glycoside | Coral: Carijoa sp. | Neutrophil superoxide and elastase inhibition | 1.8–6.8 μg/mL | [103] |
| Immune system | Laminarin polysaccharide LP1 (81), Laminarin oligosaccharide LO (82) | Alage: Laminaria japonica | Inhibition of lymphocyte apoptosis | 1–4 mg/mL | [106] |
| Immune system | ASLP (83)/polysaccharide | Lischke: Arca subcrenata | Increases splenocyte proliferation | <100 μg/mL (IC50) | [107] |
| Immune system | HCLPS-1 (84)/polysaccharide | Clam: Hyriopsis cumingii | In vivo & in vitro T and B cell activation | 20 mg/kg (IC50) | [108] |
| Immune system | Floridoside (10)/glycoside | Algae: Mastocarpus stellatus | Stimulation of IgM | 5.9–9.3 μg/mL (IC50) | [109] |
| Immune system | Frodoside A (85)/glycoside | Sea cucumber: Cucumaria frondosa | Lysosomal activity, phagocytosis and ROS activation | 0.1–0.001 μg/mL | [110] |
| Immune system | Cucumarioside A2-2 (86), I1 (87), I3 (88), I4 (89)/glycoside | Sea cucumber: Cucumaria japonica, Eupentacta fraudatrix | Simulation of lymphocytes and neutrophils | ND | [116,117,118,119] |
Acknowledgments
Conflicts of Interest
References
- Mayer, A.M.; Rodriguez, A.D.; Berlinck, R.G.; Fusetani, N. Marine pharmacology in 2009–2013: Marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 2013, 11, 2510–2573. [Google Scholar] [CrossRef] [PubMed]
- Masso-Silva, J.A.; Diamond, G. Antimicrobial peptides from fish. Pharmaceuticals 2014, 7, 265–310. [Google Scholar] [CrossRef] [PubMed]
- Winder, P.L.; Pomponi, S.A.; Wright, A.E. Natural products from the lithistida: A review of the literature since 2000. Mar. Drugs 2011, 9, 2643–2682. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.M.; Rodriguez, A.D.; Berlinck, R.G.; Fusetani, N. Marine pharmacology in 2007–8: Marine compounds with antibacterial, anticoagulant, antifungal, anti-inflammatory, antimalarial, antiprotozoal, antituberculosis and antiviral activities; affecting the immune and nervous system and other miscellaneous mechanism of action. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2011, 153, 191–222. [Google Scholar] [CrossRef] [PubMed]
- Villa, F.A.; Gernwick, L. Marine natural product drug discovery: Leads for treatment of inflammation, cancer, infections and neurological disorders. Immunopharmacol. Immunotoxicol. 2010, 32, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Munso, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2011, 28, 196–268. [Google Scholar] [CrossRef] [PubMed]
- Luhn, S.; Grimm, J.C.; Alban, S. Simple and rapid quality control of sulfated glycans by a fluorescence sensor assay—Exemplarily developed for the sulfated polysaccharides from red algae Delesseria sanguine. Mar. Drugs 2014, 12, 2205–2227. [Google Scholar] [CrossRef] [PubMed]
- Laurienzo, P. Marine polysaccharides in pharmaceutical applications: An overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef] [PubMed]
- Abad, M.J.; Bedoya, L.M.; Bermejo, P. Marine compounds and their antimicrobial activities. In Science against Microbial Pathogens: Communicating Current Research and Technological Advances, 1st ed.; Mendez-Vilas, A., Ed.; FORMATEX: Badajoz, Spain, 2011; Volume 51, pp. 1293–1306. [Google Scholar]
- Gupta, S.; Abu-Ghannam, N. Bioactive potential and possible health effects of edible brown seaweeds. Trends Food Sci. Technol. 2011, 22, 315–326. [Google Scholar] [CrossRef]
- Vo, T.S.; Kim, S.K. Potential anti-HIV agents from marine resources: An overview. Mar. Drugs. 2010, 8, 2871–2892. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, S.X.; Guan, H.S. The antiviral activities and mechanisms of marine polysaccharides: An overview. Mar. Drugs 2012, 10, 2795–2816. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.V.; Kim, S.K. Beneficial effects of marine algal compounds in cosmeticals. Mar. Drugs 2013, 11, 146–164. [Google Scholar] [CrossRef] [PubMed]
- Duus, J.O.; Gotfredsen, C.H.; Bock, K. Carbohydrate structural determination by NMR spectroscopy: Modern methods and limitations. Chem. Rev. 2000, 100, 4589–4614. [Google Scholar] [CrossRef] [PubMed]
- Safari, D.; Dekker, H.A.T.; Joosten, J.A.F.; Michalik, D.; Carvalho de Souza, A.; Adamo, R.; Lahmann, M.; Sundgren, A.; Oscarson, S.; Johannis Kamerling, P.; et al. Identification of the smallest structure capable of evoking opsonophagocytic antibodies against Streptococcus pneumoniae Type 14. Infect. Immun. 2008, 76, 4615–4623. [Google Scholar] [CrossRef] [PubMed]
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H.S. Chemical structures and bioactivities of sulfated polysaccharides from marine algae. Mar. Drugs 2011, 9, 196–223. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, J.A.; Rodríguez-Amado, I.; Montemayor, M.I.; Fraguas, J.; González, M.D.P.; Murado, M.A. Chondroitin sulfate, hyaluronic acid and chitin/chitosan production using marine waste sources: Characteristics, applications and eco-friendly processes: A review. Mar. Drugs 2013, 11, 747–774. [Google Scholar] [CrossRef] [PubMed]
- Poli, A.; Anzelmo, G.; Nicolaus, B. Bacterial exopolysaccharides from extreme marine habitats: Production, characterization and biological activities. Mar. Drugs 2010, 8, 1779–1802. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Wang, L.; Wu, H.; Luan, H.M. Bio-function summary of marine oligosaccharides. Int. J. Biol. 2011, 3, 74–86. [Google Scholar]
- He, X.; Hwang, H.M.; Aker, W.G.; Wang, P.; Lin, Y.; Jiang, X.; Hed, X. Synergistic combination of marine oligosaccharides and azithromycin against Pseudomonas aeruginosa. Microbiol. Res. 2014, 169, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Jiang, X.; Jiang, Y.; Hu, X.; Mou, H.; Li, M.; Guan, H. In vitro antioxidative activities of three marine oligosaccharides. Nat. Prod. Res. 2007, 21, 646–654. [Google Scholar]
- Trincone, A. Molecular fishing: Marine oligosaccharies. Front. Mar. Sci. 2014, 1, 1–5. [Google Scholar]
- Kren, V.; Martínkova, L. Glycosides in medicine: “The role of glycosidic residue in biological activity”. Curr. Med. Chem. 2001, 8, 1303–1328. [Google Scholar] [CrossRef] [PubMed]
- Prassas, I.; Diamandis, E.P. Novel therapeutic applications of cardiac glycosides. Nat. Rev. Drug Discov. 2008, 7, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Francis, G.; Kerem, Z.; Makkar, H.P.S.; Becker, K. The biological action of saponins in animal systems: A review. Br. J. Nutr. 2002, 88, 587–605. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Ivanchina, N.V.; Krasokhin, V.B.; Makarieva, T.N.; Stonik, V.A. Glycosides from marine sponges (Porifera, Demospongiae): Structures, taxonomical distribution, biological activities and biological roles. Mar. Drugs 2012, 10, 1671–1710. [Google Scholar] [CrossRef] [PubMed]
- Malanga, G.; Puntarulo, S. Oxidative stress and antioxidant content in Chlorella vulgaris after exposure to ultraviolet-B radiation. Physiol. Plant. 1995, 94, 672–679. [Google Scholar] [CrossRef]
- Dhalla, N.S.; Temsah, R.M.; Netticadan, T. Role of oxidative stress in cardiovascular diseases. J. Hypertens. 2000, 18, 655–673. [Google Scholar] [CrossRef]
- Cavas, L.; Yurdakoc, K. An investigation on the antioxidant status of the invasive alga Caulerpa racemosa var cylindracea (Sonder) Verlaque, Huisman, et Boudoresque (Caulerpales, Chlorophyta). J. Exp. Mar. Biol. Ecol. 2005, 325, 189–200. [Google Scholar] [CrossRef]
- Seven, A.; Guzel, S.; Aslan, M.; Hamuryudan, V. Lipid, protein, DNA oxidation and antioxidant status in rheumatoid arthritis. Clin. Biochem. 200 2008, 41, 538–543. [Google Scholar] [CrossRef]
- Schinella, G.R.; Tournier, H.A.; Priet, J.M.; Mordujovich de Buschiazzo, P.; Rios, J.L. Antioxidant activity of anti-inflammatory plant extracts. Life Sci. 2002, 70, 1023–1033. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, A.E.; Tirzite, D.; Tirzitis, G.; Roozen, J.P. Antioxidant activity of 1,4-dihydropyridine derivatives in-carotenemethyl linoleate, sunflower oil andemulsions. Food Chem. 1999, 66, 189–195. [Google Scholar] [CrossRef]
- Hayashi, Y.; Morimoto, K.; Miyata, N.; Sato, H. Quantitative cancer riskanalysis of BHA based on integration of pathological and biological/biochemicalinformation. Toxicol. Ind. Health 1993, 9, 243–249. [Google Scholar] [PubMed]
- Qiao, D.; Ke, C.; Hu, B.; Luo, J.; Ye, H.; Sun, Y.; Yan, X.; Zeng, X. Antioxidant activities of polysaccharides from Hyriopsis cumingii. Carbohydr. Polym. 2009, 78, 199–204. [Google Scholar] [CrossRef]
- Jaszek, M.; Osinska-Jaroszuk, M.; Janusz, G.; Matuszewska, A.; Stefaniuk, D.; Sulej, J.; Polak, J.; Ruminowicz, M.; Grzywnowicz, K.; Jarosz-Wilkołazka, A. New bioactive fungal molecules with high antioxidant and antimicrobial capacity isolated from Cerrena unicolor idiophasic cultures. Biomed. Res. Int. 2008, 89, 455–464. [Google Scholar]
- Xu, H.; Guo, T.; Guo, Y.F.; Zhang, J.; Li, Y.; Feng, W.; Jiao, B. Characterisation and protection on acute liver injury of a polysaccharide MP-I from Mytilus coruscus. Glycobiology 2008, 18, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Yu, X.; Zhang, Y. Extraction of polysaccharides from Mytilus edulis and their antioxidant activity in vitro. Shipin Gongye Keji 2010, 31, 132–134. [Google Scholar]
- Sun, H.H.; Mao, W.J.; Chen, Y.; Guo, S.D.; Li, H.Y.; Qi, X.H.; Chen, Y.L.; Xu, J. Isolation, chemical characteristics and antioxidant properties of the polysaccharides from marine fungus Penicillium sp. F23-2. Carbohydr. Polym. 2009, 78, 117–124. [Google Scholar] [CrossRef]
- Vijayabaskar, P.; Vaseela, N.; Thirumaran, G. Potential antibacterial and antioxidant properties of a sulfated polysaccharide from the brown marine algae Sargassum swartzii. Chin. J. Nat. Med. 2012, 10, 421–428. [Google Scholar]
- Shahidi, F.; Arachchi, J.K.V.; Jeon, Y.J. Food applications of chitin and chitosans. Trends Food Sci. Technol. 1999, 10, 37–51. [Google Scholar] [CrossRef]
- Ngo, D.N.; Kim, M.M.; Qian, Z.J.; Jung, W.K.; Lee, S.H.; Kim, S.K. Free radical-scavenging activity of low molecular weight chitin oligosaccharides lead to antioxidant effect in live cells. J. Food Biochem. 2010, 34, 161–177. [Google Scholar] [CrossRef]
- Li, Y.X.; Li, Y.; Lee, S.H.; Qian, Z.J.; Kim, S.K. Inhibitors of oxidation and matrix metalloproteinases, floridoside, and d-isofloridoside from marine red alga Laurencia undulata. J. Agric. Food Chem. 2010, 58, 578–586. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diabetes Mellitus: Report of a WHO Study Group; WHO Technical Report Series; World Health Organization: Geneva, Switzerland, 1985; p. 727. [Google Scholar]
- Rai, M.K. A review on some antidiabetic plants of India. Anc. Sci. Life 1995, 14, 42–54. [Google Scholar]
- Patankar, M.S.; Oehninger, S.; Barnett, T.; Williams, R.L.; Clark, G.F. A revised structure for fucoidan may explain some of its biological activities. J. Biol. Chem. 1993, 268, 21770–21776. [Google Scholar] [PubMed]
- Kim, K.J.; Yoon, K.Y.; Lee, B.Y. Fucoidan regulate blood glucose homeostasis in C57BL/KSJ m+/+db and C57BL/KSJ db/db mice. Fitoterapia 2012, 83, 1105–1109. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.T.; Kim, Y.D.; Jung, Y.M.; Park, D.C.; Lee, D.S.; Ku, S.K.; Li, X.; Lu, Y.; Chao, G.H.; Kim, K.J.; et al. Low molecular weight fucoidan improves endoplasmic reticulum stress-reduced insulin sensitivity through AMP-activated protein kinase activation in L6 myotubes and restores lipid homeostasis in a mouse model of type 2 diabetes. Mol. Pharmacol. 2013, 84, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Lee, O.H.; Lee, B.Y. Fucoidan, a sulfated polysaccharide, inhibits adipogenesis through the mitogen-activated protein kinase pathway in 3T3-L1 preadipocytes. Sci. Life 2010, 86, 791–797. [Google Scholar] [CrossRef]
- Lutay, N.; Nilsson, I.; Wadstrom, T.; Ljungh, A. Effect of heparin, fucoidan and other polysaccharides on adhesion of enterohepatic helicobacter species to murine macrophages. Appl. Biochem. Biotechnol. 2011, 164, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Seo, C.; Sohn, J.H.; Oh, H.; Kim, B.Y.; Ahn, J.S. Isolation of the protein tyrosine phosphatase 1B inhibitory metabolite from the marine-derived fungus Cosmospora sp. SF-5060. Bioorg. Med. Chem. Lett. 2009, 19, 6095–6097. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.; Hu, Y.; Bax, R.; Page, C. The future challenges facing the development of new antimicrobial drugs. Nat. Rev. Drug Discov. 2002, 1, 895–910. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthi, J.; Sudharsan, S.; Shanmugam, V.; Shanmugam, A. In vitro antibacterial screening of methanolic extract of whole body tissue and ethylene diamine tetra acetate (EDTA) extract of cuttlebone of Sepia pharaonis (Ehrenberg, 1831) against selected clinical isolates. Afr. J. Microbiol. Res. 2014, 8, 3551–3557. [Google Scholar]
- Vino, A.B.; Shanmugam, V.; Shanmugam, A. Antimicrobial activity of methanolic extract and fractionated polysaccharide from Loligo duvauceli Orbingy 1848 and Doryteuthis sibogae Adam 1954 on human pathogenic microorganisms. Afr. J. Microbiol. Res. 2014, 8, 230–236. [Google Scholar] [CrossRef]
- Linington, R.G.; Robertson, M.; Gauthier, A.; Finlay, B.B.; MacMillan, J.B.; Molinski, T.F.; van Soest, R.; Andersen, R.J. Caminosides B–D, antimicrobial glycolipids isolated from the marine sponge Caminus sphaeroconia. J. Nat. Prod. 2006, 69, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Deibel, C.; Krämer, S.; Chakraborty, T.; Ebel, F. EspE, a novel secreted protein of attaching and effacing bacteria, is directly translocated into infected host cells, where it appears as a tyrosine-phosphorylated 90 kDa protein. Mol. Microbiol. 1998, 28, 463–474. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zong, C.; Song, G.; Lv, G.; Chun, Y.; Wang, P.; Ding, N.; Li, Y. Total synthesis of caminoside B, a novel antimicrobial glycolipid isolated from the marine sponge Caminus sphaeroconia. Carbohydr. Res. 2010, 345, 750–760. [Google Scholar] [CrossRef] [PubMed]
- Pierre, G.; Sopena, V.; Juin, C.; Mastouri, A.; Graber, M.; Maugard, T. Antibacterial activity of a sulfated galactan extracted from the marine alga Chaetomorpha aerea against Staphylococcus aureus. Biotechnol. Bioprocess Eng. 2011, 16, 937–945. [Google Scholar] [CrossRef]
- Asano, N. Glycosidase inhibitors: Update and perspectives on practical use. Glycobiology 2003, 13, 93R–104R. [Google Scholar] [CrossRef] [PubMed]
- Danac, R.; Ball, L.; Gurr, S.J.; Muller, T.; Fairbanks, A.J. Carbohydrate chain terminators: Rational design of novel carbohydrate-based antifungal agents. Chembiochem 2007, 8, 1241–1245. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Chaturvedi, A.K.; Shukla, P.K.; Lakshmi, V. Antifungal activity in triterpene glycosides from the sea cucumber Actinopyga lecanora. Bioorg. Med. Chem. Lett. 2007, 17, 4387–4391. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.H.; Yi, Y.H.; Tang, H.F.; Liu, B.S.; Wang, Z.L.; Sun, G.Q.; Zhang, W.; Li, L.; Sun, P. Antifungal triterpene glycosides from the sea cucumber Bohadschia marmorata. Planta Med. 2009, 75, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Yi, Y.H.; Li, L.; Liu, B.S.; La, M.P.; Zhang, H.W. Antifungal active triterpene glycosides from sea cucumber Holothuria scabra. Yao Xue Xue Bao 2009, 44, 620–624. [Google Scholar] [PubMed]
- Wang, J.; Han, H.; Chen, X.; Yi, Y.; Sun, H. Cytotoxic and apoptosis-inducing activity of triterpene glycosides from Holothuria scabra and Cucumaria frondosa against HepG2 cells. Mar. Drugs 2014, 12, 4274–4290. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, S.; Pramanik, K.; Mikhopadhyay, B. Oiligosaccharides through reactivity tuning: Convergent synthesis of the trisaccharides of the steroid glycoside sokodoside B isolated from marine sponge Erylus placenta. Tetrahedron 2007, 63, 12310–12316. [Google Scholar] [CrossRef]
- Wang, X.H.; Zou, Z.R.; Yi, Y.H.; Han, H.; Li, L.; Pan, M.X. Variegatusides: New non-sulphated triterpene glycosides from the sea cucumber Stichopus variegates semper. Mar. Drugs 2014, 12, 2004–2018. [Google Scholar] [CrossRef] [PubMed]
- Rivas, G.G.; Gutierrez, C.M.G.; Arteaga, G.A.; Mercado, I.E.S.; Sanchez, N.E.A. Screening for anticoagulant activity in marine algae from the Northwest Mexican pacific coast. J. Appl. Phycol. 2011, 23, 495–503. [Google Scholar] [CrossRef]
- Desai, U.R. New antithrombin-based anticoagulants. Med. Res. Rev. 2004, 24, 151–181. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.S.; Melo, F.R.; Mourao, P.A.S. Is there a correlation between structure and anticoagulant action of sulfated galactans and sulfated fucans? Glycobiology 2002, 12, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Drozd, N.N.; Tolstenkov, A.S.; Makarov, V.A.; Kuznetsova, T.A.; Besednova, N.N.; Shevchenko, N.M.; Zvyagintseva, T.N. Pharmacodynamic parameters of anticoagulants based on sulfated polysaccharides from marine algae. Bull. Exp. Biol. Med. 2006, 142, 591–593. [Google Scholar] [CrossRef]
- Shanthi, N.; Eluvakkal, T.; Arunkumar, K. Characterization of galactose rich fucoidan with anticoagulation potential isolated from Turbinaria decurrens Bory de Saint-Vincent occurring along the coast of Gulf of Mannar (Pamban), India. J. Pharmacogn. Phytochem. 2014, 3, 132–137. [Google Scholar]
- Yoon, S.J.; Pyun, Y.R.; Hwang, J.K.; Mourao, P.A. A sulphated fucan from the brown alga Laminaria cichorioides has mainly heparin cofactor II-dependant anticoagulant activity. Carbohydr. Res. 2007, 342, 2326–2330. [Google Scholar] [CrossRef] [PubMed]
- Manoj Saravana, G.M.; Maheshkumar, P.S.; Vasanthi, M.; Achary, A. Anticoagulant property of sulphated polysaccharides extracted from marine brown algae collected from Mandapam Island, India. Afr. J. Biotechnol. 2013, 12, 1937–1945. [Google Scholar]
- Ye, L.; Xu, L.; Li, J. Preparation and anticoagulant activity of a fucosylated polysaccharide sulfate from a sea cucumber Acaudina molpadioidea. Carbohydr. Polym. 2012, 87, 2052–2057. [Google Scholar] [CrossRef]
- Pushpamali, W.A.; Nikapitiya, C.; Zoysa, M.D.; Whang, I.; Kim, S.J.; Lee, J. Isolation and purification of an anticoagulant from fermented red seaweed Lomentaria catenata. Carbohydr. Polym. 2008, 73, 274–279. [Google Scholar] [CrossRef]
- Rodrigues, J.A.G.; de Queiroz, I.N.L.; Bessa, E.F.; Coura, C.O.; Amorim, R.C.N.; Benevides, N.M.B. Anticoagulant activity of sulfated polysaccharides fractions from an aqueous extract obtained from the red seaweed Halymenia floresia (Clemente) C. Agardh. Acta Sci. Technol. Mar. 2011, 33, 371–378. [Google Scholar]
- Manilal, A.; Sujith, S.; Selvin, J.; Panikkar, M.V.N.; George, S. Anticoagulant potential of polysaccharide isolated from Indian red alga, Asparagopsis taxiformis (Delile) Trevisan. Thalass. Int. J. Mar. Sci. 2012, 28, 9–15. [Google Scholar]
- Rodrigues, J.A.G.; Neto, E.M.; Teixeira, L.A.C.; de Paula, R.C.M.; Mourao, P.A.S.; Benevides, N.M.B. Structural features and inactivation of coagulation proteases of a sulfated polysaccharidic fraction from Caulerpa cupressoides var. lycopodium (Caulerpaceae, Chlorophyta). Acta Sci. Technol. Mar. 2013, 35, 611–619. [Google Scholar]
- Regalado, E.L.; Tasdemir, D.; Kaiser, M.; Cachet, N.; Amade, P.; Thomas, O.P. Antiprotozoal steroidal saponins from the marine sponge Pandaros acanthifolium. J. Nat. Prod. 2010, 73, 1404–1410. [Google Scholar] [CrossRef] [PubMed]
- Regalado, E.; Jimenez-Romero, C.; Genta-Jouve, G.; Tasdemir, D.; Amade, P.; Nogueiras, C.; Thomas, O.P. Acanthifoliosides, minor steroidal saponins from the sponge Pandaros acanthifolium. Tetrahedron 2011, 67, 1011–1018. [Google Scholar] [CrossRef]
- Word Health Organization Home Page. Available online: http://www.who.int/neglected_diseases/en/ (accessed on 17 November 2014).
- Mishra, S.K.; Satpathy, S.K.; Mohanty, S. Survey of malaria treatment and deaths. Bull. World Health Organ. 1999, 77, 1020. [Google Scholar] [PubMed]
- Gutiérrez, M.; Capson, T.L.; Guzman, H.M.; Gonzalez, J.; Ortega-Barría, E.; Quiñoa, E.; Riguera, R. Antiplasmodial metabolites isolated from the marine octocoral Muricea austera. J. Nat. Prod. 2006, 69, 1379–1383. [Google Scholar] [CrossRef]
- Borowitzka, M.A. Microalgae as sources of pharmaceuticals and other biologically active compounds. J. Appl. Phycol. 1995, 7, 65–68. [Google Scholar] [CrossRef]
- Smelcerovic, A.; Knezevic-Jugovic, Z.; Petronijevic, Z. Microbial polysaccharides and their derivatives as current and prospective pharmaceuticals. Curr. Pharm. Des. 2008, 14, 3168–3195. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.B.; Hayashi, K.; Hirata, M.; Kuroda, E.; Suzuki, E.; Kubo, Y.; Hayashi, T. Antiviral sulfated polysaccharide from Navicula directa, a diatom collected from deep-sea water in Toyama Bay. Biol. Pharm. Bull. 2006, 29, 2135–2139. [Google Scholar] [CrossRef] [PubMed]
- Bouhlal, R.; Haslin, C.; Chermann, J.C.; Colliec-Jouault, S.; Sinquin, C.; Simon, G.; Cerantola, S.; Riadi, H.; Bourgougnon, N. Antiviral activities of sulfated polysaccharides isolated from Sphaerococcus coronopifolius (Rhodophytha, Gigartinales) and Boergeseniella thuyoides (Rhodophyta, Ceramiales). Mar. Drugs 2011, 9, 1187–1209. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C.; Bligh, S.W.A.; Shi, S.S.; Wang, Z.; Hu, Z.B.; Crowder, J.; Branford-White, C.; Vella, C. Structural features and anti-HIV-1 activity of novel polysaccharides from red algae Grateloupia longifolia and Grateloupia filicina. Int. J. Biol. Macromol. 2007, 41, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Talarico, L.B.; Duarte, M.E.; Zibetti, R.G.; Noseda, M.D.; Damonte, E.B. An algalderived d,l-galactan hybrid is an efficient preventing agent for in vitro dengue virus infection. Planta Med. 2007, 73, 1464–1468. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.X.; Li, J.; Sun, Y.X.; Qi, X.; Wang, Q.J.; Xin, X.L.; Geng, M.Y. Sulfated polymannuroguluronate, a novel anti-AIDS drug candidate, inhibits HIV-1 Tat induced angiogenesis in Kaposi’s sarcoma cells. Biochem. Pharmacol. 2007, 74, 1330–1339. [Google Scholar] [CrossRef] [PubMed]
- Trinchero, J.; Ponce, N.M.A.; Cordoba, O.L.; Flores, M.L.; Pampuro, S.; Stortz, C.A.; Salomon, H.; Turk, G. Antiretroviral activity of fucoidans extracted from the brown seaweed Adenocystis utricularis. Phytother. Res. 2009, 23, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, K.C.S.; Medeiros, V.P.; Queiroz, L.S.; Abreu, L.R.D.; Rocha, H.A.O.; Ferreira, C.V.; Juca, M.B.; Aoyama, H.; Leite, E.L. Inhibition of reverse transcriptase activity of HIV by polysaccharides of brown algae. Biomed. Pharmacother. 2008, 62, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Hui, B.; Li, J.; Geng, M.Y. Sulfated polymannuroguluronate, a novel anti-acquired immune deficiency syndrome drug candidate, decreased vulnerability of PC12 cells to human immunodeficiency virus tat protein through attenuating calcium overload. J. Neurosci. Res. 2008, 86, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Stephanie, B.; Eric, D.; Sophie, F.M.; Christian, B.; Yu, G.L. Carrageenan from Solieria chordalis (Gigartinales): Structural analysis and immunological activities of the low molecular weight fractions. Carbohydr. Polym. 2010, 81, 448–460. [Google Scholar] [CrossRef]
- Skoler-Karpoff, S.; Ramjee, G.; Ahmed, K.; Altini, L.; Plagianos, M.G.; Friedland, B.; Govender, S.; de Kock, A.; Cassim, N.; Palanee, T.; et al. Efficacy of Carraguard for prevention of HIV infection in women in South Africa: A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1977–1987. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, R.; Nwe, N.; Tokura, S.; Tamura, H. Sulfated chitin and chitosan as novel biomaterials. Int. J. Biol. Macromol. 2007, 40, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Suwan, J.; Zhang, Z.; Li, B.; Vongchan, P.; Meepowpan, P.; Zhang, F.; Mousa, S.A.; Mousa, S.; Premanode, B.; Kongtawelert, P.; et al. Sulfonation of papain-treated chitosan and its mechanism for anticoagulant activity. Carbohydr. Res. 2009, 344, 1190–1196. [Google Scholar] [CrossRef] [PubMed]
- Prabaharan, M.; Reis, R.L.; Mano, J.F. Carboxymethyl chitosan-graft-phosphatidylethanolamine: Amphiphilic matrices for controlled drug delivery. React. Funct. Polym. 2007, 67, 43–52. [Google Scholar] [CrossRef]
- Artan, M.; Karadeniz, F.; Kim, M.M.; Kim, S.K. Chitosan derivatives as HIV-1 inhibitors. J. Biotechnol. 2008, 136S, S527–S540. [Google Scholar] [CrossRef]
- Artan, M.; Karadeniz, F.; Karagozlu, M.Z.; Kim, M.M.; Kim, S.K. Anti-HIV-1 activity of low molecular weight sulfated chitooligosaccharides. Carbohydr. Res. 2010, 345, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Kazlowska, K.; Hsu, T.; Hou, C.C.; Yang, W.C.; Tsai, G.J. Anti-inflammatory properties of phenolic compounds and crude extract from Porphyra dentata. J. Ethanopharmacol. 2010, 128, 123–130. [Google Scholar] [CrossRef]
- Nguemfo, E.L.; Dimo, T.; Azebaze, A.G.B.; Asongalem, E.A.; Alaoui, K.; Dongmo, A.B.; Cherrah, Y.; Kamtchouing, P. Anti-inflammatory and anti-nociceptive activities of the stem bark extracts from Allanblackia monticola STANER L.C. (Guttiferae). J. Ethanopharmacol. 2007, 114, 417–424. [Google Scholar]
- Belmiro, C.L.; Castelo-Branco, M.T.; Melim, L.M.; Schanaider, A.; Elia, C.; Madi, K.; Pavao, M.S.; de Souza, H.S. Unfractionated heparin and new heparin analogues from ascidians (chordate-tunicate) ameliorate colitis in rats. J. Biol. Chem. 2009, 284, 11267–11278. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.Y.; Hwang, T.L.; Lin, M.R.; Chen, Y.H.; Chang, Y.C.; Fang, L.S.; Wang, W.H.; Wu, Y.C.; Sung, P.J. Carijoside A, a bioactive sterol glycoside from an octocoral Carijoa sp. (Clavulariidae). Mar. Drugs 2010, 8, 2014–2020. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghea, W.A.J.P.; Jeon, Y.J. Biological activities and potential industrial applications of fucose rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Wijesekar, I.; Pangestuti, R.; Kim, S.K. Biological activities and potential health benefits of sulfated polysaccharides derived from marine algae. Carbohydr. Polym. 2011, 84, 14–21. [Google Scholar] [CrossRef]
- Kim, K.H.; Kim, Y.W.; Kim, H.B.; Lee, B.J.; Lee, D.S. Anti-apoptotic activity of laminarin polysaccharides and their enzymatically hydrolyzed oligosaccharides from Laminaria japonica. Biotechnol. Lett. 2006, 28, 439–446. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Liu, C.; Chen, Y.; Ji, A.; Shen, Z.; Xi, T.; Yao, Q. Isolation and structural characterization of a novel polysaccharide prepared from Arca subcrenata Lischke. J. Biosci. Bioeng. 2007, 104, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Zhang, H.; Zhang, Y.; Wang, H. Chemical properties and immunostimulatory activity of a water-soluble polysaccharide from the clam of Hyriopsis cumingii lea. Carbohydr. Polym. 2009, 77, 365–369. [Google Scholar] [CrossRef]
- Courtois, A.; Simon-Colin, C.; Boisset, C.; Berthou, C.; Deslandes, E.; Guezennec, J.; Bordron, A. Floridoside extracted from the red alga Mastocarpus stellatus is a potent activator of the classical complement pathway. Mar. Drugs 2008, 6, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Agafonova, I.G.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A.; Collin, P.D.; Woodward, C. Immunomodulatory properties of frondoside A, a major triterpene glycoside from the North Atlantic commercially harvested sea cucumber Cucumaria frondosa. J. Med. Food 2008, 11, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Roginsky, A.B.; Ding, X.Z.; Woodward, C.; Collin, P.; Newman, R.A. Review of the apoptosis pathways in pancreatic cancer and the anti-apoptotic effects of the novel sea cucumber compound, Frondoside A. Ann. N. Y. Acad. Sci. 2008, 1138, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Roginsky, A.B.; Ding, X.Z.; Woodward, C.; Ujiki, M.B.; Singh, B.; Bell, R.H., Jr.; Collin, P.; Adrian, T.E. Anti-pancreatic cancer effects of a polar extract from the edible sea cucumber, Cucumaria frondosa. Pancreas 2010, 39, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Al Marzouqi, N.; Iratni, R.; Nemmar, A.; Arafat, K.; Ahmed Al Sultan, M.; Yasin, J.; Collin, P.; Mester, J.; Adrian, T.E.; Attoub, S. Frondoside A inhibits human breast cancer cell survival, migration, invasion and the growth of breast tumor xenografts. Eur. J. Pharmacol. 2011, 668, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Attoub, S.; Arafat, K.; Gelaude, A.; Al Sultan, M.A.; Bracke, M.; Collin, P.; Takahashi, T.; Adrian, T.E.; de Wever, O. Frondoside A suppressive effects on lung cancer survival, tumor growth, angiogenesis, invasion, and metastasis. PLoS One 2013, 8, e53087. [Google Scholar] [CrossRef] [PubMed]
- Holt, D.M.; Ma, X.; Kundu, N.; Collin, P.D.; Fulton, A.M. Modulation of host natural killer cell functions in breast cancer via prostaglandin E2 receptors EP2 and EP4. J. Immunother. 2012, 35, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Pinegin, B.V.; Pichugina, L.V.; Zaporozhets, T.S.; Agafonova, I.G.; Boguslavski, V.M.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A. Immunomodulatory properties of cumaside. Int. Immunopharmacol. 2006, 6, 1070–1082. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Menchinskaya, E.S.; Aminin, D.L.; Kalinin, V.I. Structure of cucumarioside I2 from the sea cucumber Eupentacta fraudatrix (Djakonov et Baranova) and cytotoxic and immunostimulatory activities of this saponin and relative compounds. Nat. Prod. Res. 2013, 27, 1776–1783. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Gorpenchenko, T.Y.; Bulgakov, V.P.; Andryjashchenko, P.V.; Avilov, S.A.; Kalinin, V.I. Triterpene glycoside cucumarioside A2-2 from sea cucumber stimulates mouse immune cell adhesion, spreading, and motility. J. Med. Food 2011, 14, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological action of cucumariosides I1, I3, I4, three new minor disulfated pentaosides. Nat. Prod. Commun. 2013, 8, 1053–1058. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, H.-K.; Seo, C.H.; Park, Y. The Effects of Marine Carbohydrates and Glycosylated Compounds on Human Health. Int. J. Mol. Sci. 2015, 16, 6018-6056. https://doi.org/10.3390/ijms16036018
Kang H-K, Seo CH, Park Y. The Effects of Marine Carbohydrates and Glycosylated Compounds on Human Health. International Journal of Molecular Sciences. 2015; 16(3):6018-6056. https://doi.org/10.3390/ijms16036018
Chicago/Turabian StyleKang, Hee-Kyoung, Chang Ho Seo, and Yoonkyung Park. 2015. "The Effects of Marine Carbohydrates and Glycosylated Compounds on Human Health" International Journal of Molecular Sciences 16, no. 3: 6018-6056. https://doi.org/10.3390/ijms16036018
APA StyleKang, H.-K., Seo, C. H., & Park, Y. (2015). The Effects of Marine Carbohydrates and Glycosylated Compounds on Human Health. International Journal of Molecular Sciences, 16(3), 6018-6056. https://doi.org/10.3390/ijms16036018






