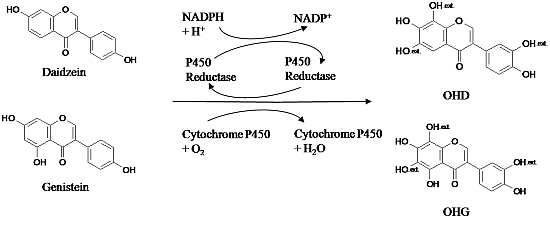
Isolation, Bioactivity, and Production of ortho-Hydroxydaidzein and ortho-Hydroxygenistein
Abstract
:1. Introduction
2. Isolation of OHD and OHG
3. Bioactivity of OHD and OHG
3.1. Anticancer-Related Bioactivity
3.2. Antimelanogenesis-Related Bioactivity
3.3. Other Bioactivity
4. Production of OHD and OHG
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Franke, A.A.; Custer, L.J.; Cerna, C.M.; Narala, K.K. Quantitation of phytoestrogens in legumes by HPLC. J. Agric. Food Chem 1994, 42, 1905–1913. [Google Scholar]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar]
- Vitale, D.C.; Piazza, C.; Melilli, B.; Drago, F.; Salomone, S. Isoflavones: Estrogenic activity, biological effect and bioavailability. Eur. J. Drug Metab. Pharmacokinet 2013, 38, 15–25. [Google Scholar]
- Scotti, L.; Bezerra Mendonça, F.J., Jr.; Magalhaes Moreira, D.R.; da Silva, M.S.; Pitta, I.R.; Scotti, M.T. SAR, QSAR and docking of anticancer flavonoids and variants: A review. Curr. Top. Med. Chem 2012, 12, 2785–2809. [Google Scholar]
- Roberts-Kirchhoff, E.S.; Crowley, J.R.; Hollenberg, P.F.; Kim, H. Metabolism of genistein by rat and human cytochrome P450s. Chem. Res. Toxicol 1999, 12, 610–616. [Google Scholar]
- Kulling, S.E.; Honig, D.M.; Simat, T.J.; Metzler, M. Oxidative in vitro metabolism of the soy phytoestrogens daidzein and genistein. J. Agric. Food Chem 2000, 48, 4963–4972. [Google Scholar]
- Kulling, S.E.; Honig, D.M.; Metzler, M. Oxidative metabolism of the soy isoflavones daidzein and genistein in humans in vitro and in vivo. J. Agric. Food Chem. 2001, 49, 3024–3033. [Google Scholar]
- Xiao, J.; Högger, P. Metabolism of dietary flavonoids in liver microsomes. Curr. Drug Metab 2013, 14, 381–391. [Google Scholar]
- Barnes, S.; Prasain, J.; D’Alessandro, T.; Arabshahi, A.; Botting, N.; Lila, M.A.; Jackson, G.; Janle, E.M.; Weaver, C.M. The metabolism and analysis of isoflavones and other dietary polyphenols in foods and biological systems. Food Funct 2011, 2, 235–244. [Google Scholar]
- Chen, K.I.; Erh, M.H.; Su, N.W.; Liu, W.H.; Chou, C.C.; Cheng, K.C. Soyfoods and soybean products: From traditional use to modern applications. Appl. Microbiol. Biotechnol 2012, 96, 9–22. [Google Scholar]
- Naim, M.; Gestetner, B.; Zilkah, S.; Birk, Y.; Bondi, A. Soybean isoflavones, characterization, and antifungal activity. J. Agric. Food Chem 1974, 22, 806–810. [Google Scholar]
- Kudou, S.; Fleury, Y.; Welti, D.; Magnolato, D.; Uchida, T.; Kitamura, K.; Okubo, K. Malonyl isoflavone glycosides in soybean seeds (Glycine max Merril). Agric. Biol. Chem 1991, 55, 2227–2233. [Google Scholar]
- Esaki, H.; Watanabe, R.; Onozaki, H.; Kawakishi, S.; Osawa, T. Formation mechanism for potent antioxidative o-dihydroxyisoflavones in soybeans fermented withAspergillus saitoi. Biosci. Biotechnol. Biochem 1999, 63, 851–858. [Google Scholar]
- Chang, T.S.; Ding, H.Y.; Tai, S.S.K.; Wu, C.Y. Metabolism of the soy isoflavone daidzein and genistein by the fungi used for the preparation of various fermented soybean foods. Biosci. Biotechnol. Biochem 2007, 71, 1330–1333. [Google Scholar]
- Nazir, K.; Ichinose, H.; Wariishi, H. Molecular characterization and isolation of cytochrome P450 genes from the filamentous fungusAspergillus oryzae. Arch. Microbiol 2010, 192, 395–408. [Google Scholar]
- Nazir, N.H.; Ichinose, H.; Wariishi, H. Construction and application of a functional library of cytochrome P450 monooxygenases from the filamentous fungusAspergillus oryzae. Appl. Environ. Microbiol 2011, 77, 3147–3150. [Google Scholar]
- Bernhardt, R. Cytochromes P450 as versatile biocatalysts. J. Biotechnol 2006, 124, 128–145. [Google Scholar]
- Peng, W.X.; Wang, L.S.; Li, H.D.; Abd El-Aty, A.M.; Chen, G.L.; Zhou, H.H. Evidence for the involvement of human liver microsomes CYP1A2 in the mono-hydroxylation of daidzein. Clin. Chim. Acta 2003, 334, 77–85. [Google Scholar]
- Hu, M.; Krausz, K.; Chen, J.; Ge, X.; Li, J.; Gelboin, H.L.; Gonzalez, F.J. Identification of CYP1A2 as the main isoform for the phase I hydroxylated metabolism of genistein and a prodrug converting enzyme of methylated isoflavones. Drug Metab. Dispos 2003, 31, 924–931. [Google Scholar]
- Breinholt, V.M.; Rasmussen, S.E.; Brøsen, K.; Friedberg, T.H. In vitro metabolism of genistein and tangeretin by human and murine cytochrome P450s. Pharmacol. Toxicol 2003, 93, 14–22. [Google Scholar]
- Pandey, B.P.; Lee, N.; Choi, K.Y.; Jung, E.; Jeong, D.H.; Kim, B.G. Screening of bacterial cytochrome P450s responsible for regiospecific hydroxylation of (iso)flavonoids. Enzym. Microbial. Technol 2011, 48, 386–392. [Google Scholar]
- Charaux, C.; Rabate, J. Constitution chimique de I’orobol. Bull. Soc. Chim. Biol 1939, 21, 1330–1333. [Google Scholar]
- Ma, C.H.; Chen, B.; Qi, H.Y.; Li, B.G.; Zhang, G.L. Two pyranocoumarins from the seeds ofCalophyllum polyanthum. J. Nat. Prod 2004, 67, 1598–1600. [Google Scholar]
- Matsuda, H.; Morikawa, T.; Xu, F.; Ninomiya, K.; Yoshikawa, M. New isoflavones and pterocarpane with hepatoprotective activity from the stems ofErycibe expansa. Planta Med 2004, 70, 1201–1209. [Google Scholar]
- Tewtrakul, S.; Subhadhirasakul, S.; Cheenpracha, S.; Karalai, C. HIV-1 protease and HIV-1 integrase inhibitory substances fromEclipta prostrata. Phytother. Res 2007, 21, 1092–1095. [Google Scholar]
- Chen, Q.C.; Zhang, W.Y.; Jin, W.; Lee, I.S.; Min, B.S.; Jung, H.J.; Na, M.; Lee, S.; Bae, K. Flavonoids and isoflavonoids from Sophorae Flos improve glucose uptake in vitro. Planta Med. 2010, 76, 79–81. [Google Scholar]
- Kiriakidis, S.; Högemeier, O.; Starcke, S.; Dombrowski, F.; Hahne, J.C.; Pepper, M.; Jha, H.C.; Wernert, N. Novel tempeh (fermented soyabean) isoflavones inhibit in vivo angiogenesis in the chicken chorioallantoic membrane assay. Br. J. Nutr 2005, 93, 317–323. [Google Scholar]
- Umezawa, H.; Tobe, H.; Shibamoto, N.; Nakamura, F.; Nakamura, K.; Matsuzaki, M.; Takeuchi, T. Isolation of isoflavones inhibiting dopa decarboxylase from fungi andStreptomyces. J. Antibiot 1975, 28, 947–952. [Google Scholar]
- De Oliveira, A.B.; Gottlieb, O.R.; Ollis, W.D. A quimica de leguminosas Brasileiras. XVII. Constituents do Machaerium villosum. Anais Acad. Brasil. Cienc 1968, 40, 147–150. [Google Scholar]
- Chan, S.C.; Chang, Y.S.; Wang, J.P.; Chen, S.C.; Kuo, S.C. Three new flavonoids and antiallergic, anti-inflammatory constituents from the heartwood ofDalbergia odorifera. Planta Med 1998, 64, 153–158. [Google Scholar]
- Kite, G.K.; Veitch, N.C.; Boalch, M.E.; Lewis, G.P.; Leon, C.J.; Simmonds, M.S.J. Flavonol tetraglycosides from fruits of Styphnolobium japonicum (Leguminosae) and the authentication of Fructus Sophorae and Flos Sophorae. Phytochemistry 2009, 70, 785–794. [Google Scholar]
- Gyoergy, P.; Murata, K.; Ikehata, H. Antioxidants isolated from fermented soybeans (tempeh). Nature 1964, 203, 870–872. [Google Scholar]
- Esaki, H.; Onozaki, H.; Morimitsu, Y. Potent antioxidative isoflavones isolated from soybean fermented withAspergillus saitoi. Biosci. Biotechnol. Biochem 1998, 62, 740–746. [Google Scholar]
- Esaki, H.; Kawakishi, S.; Morimitsu, Y.; Osawa, T. New potent antioxidative o-dihydroxyisoflavones in fermented Japanese soybean products. Biosci. Biotechnol. Biochem 1999, 63, 1637–1639. [Google Scholar]
- Hirota, A.; Taki, S.; Kawaii, S.; Yano, M.; Abe, N. 1,1-Diphenyl-2-picrylhydrazyl radical-scavenging compounds from soybean miso and antiproliferative activity of isoflavones from soybean miso toward the cancer cell lines. Biosci. Biotechnol. Biochem 2000, 64, 1038–1040. [Google Scholar]
- Chen, Y.C.; Inaba, M.; Abe, N.; Hirota, A. Antimutagenic activity of 8-hydroxyisoflavones and 6-hydroxydaidzein from soybean miso. Biosci. Biotechnol. Biochem 2003, 67, 903–906. [Google Scholar]
- Hirota, A.; Inaba, M.; Chen, Y.C.; Abe, N.; Taki, S.; Yano, M.; Kawaii, S. Isolation of 8-hydroxyglycitein and 6-hydroxydaidzein from soybean miso. Biosci. Biotechnol. Biochem 2004, 68, 1372–1374. [Google Scholar]
- Sugiyama, Y.; Sakurai, Y.; Hirota, A. Isolation of 2,4,4′-trihydroxydeoxybenzoin and 3′-hydroxydaidzein from soybean miso. Biosci. Biotechnol. Biochem 2010, 74, 1293–1294. [Google Scholar]
- Chen, Y.C.; Sugiyama, Y.; Abe, N.; Kuruto-Niwa, R.; Nozawa, R.; Hirota, A. DPPH radical-scavenging compounds from dou-chi, a soybean fermented food. Biosci. Biotechnol. Biochem 2005, 69, 999–1006. [Google Scholar]
- Park, J.S.; Park, H.Y.; Kim, D.H.; Kim, D.H.; Kim, H.K. ortho-Dihydroxyisoflavone derivatives from aged Doenjang (Korean fermented soypaste) and its radical scavenging activity. Bioorg. Med. Chem. Lett 2008, 18, 5006–5009. [Google Scholar]
- Klus, K.; Borger-Papendorf, G.; Barz, W. Formation of 6,7,4′-trihydroxyisoflavone (factor 2) from soybean seed isoflavones by bacteria isolated from tempe. Phytochemistry 1993, 34, 979–981. [Google Scholar]
- Klus, K.; Barz, W. Formation of polyhydroxylated isoflavones from the soybean seed isoflavones daidzein and glycitein by bacteria isolated from tempe. Arch. Microbiol 1995, 164, 428–434. [Google Scholar]
- Funayama, S.; Anraku, Y.; Mita, A.; Komiyama, K.; Omura, S. Structure study of isoflavonoids possessing antioxidant activity from the fermentation broth of Streptomyces sp. J. Antibiot 1989, 42, 1350–1355. [Google Scholar]
- Komiyama, K.; Funayama, S.; Anraku, Y.; Mita, A.; Takahashi, Y.; Omura, S. Isolation of isoflavonoids possessing antioxidant activity from the fermentation broth of Streptomyces sp. J. Antibiot 1989, 42, 1344–1349. [Google Scholar]
- Klus, K.; Barz, W. Formation of polyhydroxylated isoflavones from the isoflavones genistein and biochanin A by bacteria isolated from tempe. Phytochemistry 1998, 47, 1045–1048. [Google Scholar]
- Tsuchihashi, R.; Kodera, M.; Sakamoto, S.; Nakajima, Y.; Yamazaki, T.; Niiho, Y.; Nohara, T.; Kinjo, J. Microbial transformation and bioconversion of isoflavones from Pueraria flowers by human intestinal bacterial strains. J. Nat. Med 2009, 63, 254–260. [Google Scholar]
- Goto, H.; Terao, Y.; Akai, S. Synthesis of various kinds of isoflavones, isoflavanes, and biphenyl-ketones and their 1,1-diphenyl-2-picrylhydrazyl radical-scavenging activities. Chem. Pharm. Bull 2009, 57, 346–360. [Google Scholar]
- Bennink, M.R. Dietary soy reduces colon carcinogenesis in human and rats. Soy and colon cancer. Adv. Exp. Med. Biol 2001, 492, 11–17. [Google Scholar]
- Farina, H.G.; Pomies, M.; Alonso, D.F.; Gomez, D.E. Antitumor and antiangiogenic activity of soy isoflavone genistein in mouse models of melanoma and breast cancer. Oncol. Rep 2006, 16, 885–891. [Google Scholar]
- Hewitt, A.L.; Singletary, K.W. Soy extract inhibits mammary adenocarcinoma growth in a syngeneic mouse model. Cancer Lett 2003, 192, 133–143. [Google Scholar]
- Jian, L. Soy, isoflavones, and prostate cancer. Mol. Nutr. Food Res 2009, 53, 217–226. [Google Scholar]
- Nguyen, D.T.; Hernandez-Montes, E.; Vauzour, D.; Schonthal, A.H.; Rice-Evans, C.; Cadenas, E.; Spencer, J.P.E. The intracellular genistein metabolite 5,7,3′,4′-tetrahydroxyisoflavone mediates G2-M cell cycle arrest in cancer cells via modulation of the p38 signaling pathway. Free Radic. Biol. Med 2006, 41, 1225–1239. [Google Scholar]
- Vauzour, D.; Vafeiadou, K.; Rice-Evans, C.; Cadenas, E.; Spencer, J.P.E. Inhibition of cellular proliferation by the genistein metabolite 5,7,30,40-tetrahydroxyisoflavone is mediated by DNA damage and activation of the ATR signalling pathway. Arch. Biochem. Biophy 2007, 468, 159–166. [Google Scholar]
- Lee, D.E.; Lee, K.W.; Jung, S.K.; Lee, E.J.; Hwang, J.A.; Lim, T.G.; Kim, B.Y.; Bode, A.M.; Lee, H.J.; Dong, Z. 6,7,4′-Trihydroxyisoflavone inhibits HCT-116 human colon cancer cell proliferation by targeting CDK1 and CDK2. Carcinogenesis 2011, 32, 629–635. [Google Scholar]
- Lee, D.E.; Lee, K.W.; Byun, S.; Jung, S.K.; Song, N.; Lim, S.H.; Heo, Y.S.; Kim, J.E.; Kang, N.J.; Kim, B.Y.; et al. 7,3′,4′-Trihydroxyisoflavone, a metabolite of the soy isoflavone daidzein, suppresses ultraviolet B-induced skin cancer by targeting Cot and MKK4. J. Biol. Chem 2011, 286, 14246–14256. [Google Scholar]
- Lee, D.E.; Lee, K.W.; Song, N.R.; Seo, S.K.; Heo, Y.S.; Kang, N.J.; Bode, A.M.; Lee, H.J.; Dong, Z. 7,3′,4′-Trihydroxyisoflavone inhibits epidermal growth factor-induced proliferation and transformation of JB6 P+ mouse epidermal cells by suppressing cyclin-dependent kinases and phosphatidylinositol 3-kinase. J. Biol. Chem 2010, 285, 21458–21466. [Google Scholar]
- Lo, Y.L.; Wang, W.; Ho, C.T. 7,3′,4′-Trihydroxyisoflavone modulates multidrug resistance transporters and induces apoptosis via production of reactive oxygen species. Toxicology 2012, 302, 221–232. [Google Scholar]
- Lo, Y.L. A potential daidzein derivative enhances cytotoxicity of epirubicin on human colon adenocarcinoma caco-2 cells. Int. J. Mol. Sci 2012, 14, 158–176. [Google Scholar]
- Chang, T.S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci 2009, 10, 2440–2475. [Google Scholar]
- Chang, T.S.; Ding, H.Y.; Lin, H.C. Identifying 6,7,4′-trihydroxyisoflavone as a potent tyrosinase inhibitor. Biosci. Biotechnol. Biochem 2005, 69, 1999–2001. [Google Scholar]
- Chang, T.S.; Ding, H.Y.; Tai, S.S.K.; Wu, C.Y. Tyrosinase inhibitors isolated from soygerm koji fermented with Aspergillus oryzae BCRC 32288. Food Chem 2007, 105, 1430–1438. [Google Scholar]
- Chang, T.S. Two potent suicide substrates of mushroom tyrosinase: 7,8,4′-Trihydroxyisoflavone and 5,7,8,4′-tetrahydroxyisoflavone. J. Agric. Food Chem 2007, 55, 2010–2015. [Google Scholar]
- Tai, S.S.K.; Lin, C.G.; Wu, M.H.; Chang, T.S. Evaluation of depigmenting activity by 8-hydroxydaidzein in mouse B16 melanoma cells and human volunteers. Int. J. Mol. Sci 2009, 10, 4257–4266. [Google Scholar]
- Park, J.S.; Kim, D.H.; Lee, J.K.; Lee, J.Y.; Kim, D.H.; Kim, H.K.; Lee, H.J.; Kim, H.C. Natural ortho-dihydroxyisoflavone derivatives from aged Korean fermented soybean paste as potent tyrosinase and melanin formation inhibitors. Bioorg. Med. Chem. Lett 2010, 20, 1162–1164. [Google Scholar]
- Goh, M.J.; Park, J.S.; Bae, J.H.; Kim, D.H.; Kim, H.K.; Na, Y.J. Effects of ortho-dihydroxyisoflavone derivatives from Korean fermented soybean paste on melanogenesis in B16 melanoma cells and human skin equivalents. Phytother. Res 2012, 26, 1107–1112. [Google Scholar]
- Chang, T.S. Natural melanogenesis inhibitors acting through the down-regulation of tyrosinase activity. Materials 2012, 5, 1661–1685. [Google Scholar]
- Kim, E.S.; Shin, J.H.; Seok, S.H.; Kim, J.B.; Chang, H.; Park, S.J.; Jo, Y.K.; Choi, E.S.; Park, J.S.; Yeom, M.H.; et al. Autophagy mediates anti-melanogenic activity of 30-ODI in B16F1 melanoma cells. Biochem. Biophy. Res. Commun 2013, 442, 165–170. [Google Scholar]
- Tewtrakul, S.; Subhadhirasakul, S.; Tansakul, P.; Cheenpracha, S.; Karalai, C. Antiinflammatory constituents from Eclipta prostrata using RAW264.7 macrophage cells. Phytother. Res 2011, 25, 1313–1316. [Google Scholar]
- Fujita, T.; Funako, T.; Hayashi, H. 8-Hydroxydaidzein, an aldose reductase inhibitor from okara fermented with Aspergillus sp. HK-388. Biosci. Biotechnol. Biochem 2004, 68, 1588–1590. [Google Scholar]
- Seo, S.G.; Yang, H.; Shin, S.H.; Min, S.; Kim, Y.A.; Yu, J.G.; Lee, D.E.; Chung, M.Y.; Heo, Y.S.; Kwon, J.Y.; et al. A metabolite of daidzein, 6,7,4′-trihydroxyisoflavone, suppresses adipogenesis in 3T3-L1 preadipocytes via ATP-competitive inhibition of PI3K. Mol. Nutr. Food Res 2013, 57, 1446–1455. [Google Scholar]
- Chen, L.; Li, Q.Y.; Shi, X.J.; Mao, S.L.; Du, Y.L. 6-Hydroxydaidzein enhances adipocyte differentiation and glucose uptake in 3T3-L1 cells. J. Agric. Food Chem 2013, 61, 10714–10719. [Google Scholar]
- Tasdemir, D.; Kaiser, M.; Brun, R.; Yardley, V.; Schmidt, T.J.; Tosun, F.; Ruedi, P. Antitrypanosomal and antileishmanial activities of flavonoids and their analogues: In vitro, in vivo, structure-activity relationship, and quantitative structure-activity relationship studies. Antimicrob. Agents Chemother 2006, 50, 1352–1364. [Google Scholar]
- Salem, M.M.; Werbovetz, K.A. Isoflavonoids and other compounds from Psorothamnus arborescens with antiprotozoal activities. J. Nat. Prod 2006, 69, 43–49. [Google Scholar]
- Kim, B.B.; Kim, J.R.; Kim, J.H.; Kim, Y.A.; Park, J.S.; Yeom, M.H.; Lee, H.; Lee, K.W.; Kang, N.J. 7,3′,4′-Trihydroxyisoflavone ameliorates the development of dermatophagoides farinae-induced atopic dermatitis in NC/Nga Mice. Evid. Based Complement. Alternat. Med 2013, 2013, 636597. [Google Scholar]
- Roh, C.; Choi, K.Y.; Pandey, B.P.; Kim, B.G. Hydroxylation of daidzein by CYP107H1 from Bacillus subtilis 168. J. Mol. Catal. B Enzym 2009, 59, 248–253. [Google Scholar]
- Choi, K.Y.; Kim, T.J.; Koh, S.K.; Roh, C.H.; Pandey, B.P.; Lee, N.; Kim, B.G. A-ring ortho-specific monohydroxylation of daidzein by cytochrome P450s of Nocardia farcinica IFM10152. Biotechnol. J 2009, 4, 1586–1595. [Google Scholar]
- Choi, K.Y.; Park, H.Y.; Kim, B.G. Characterization of bi-functional CYP154 from Nocardia farcinica IFM10152 in the O-deakylation and ortho-hydroxylation of formononetin. Enzym. Microb. Technol 2010, 47, 327–334. [Google Scholar]
- Choi, K.Y.; Jung, E.O.; Jung, D.H.; Pandey, B.P.; Yun, H.; Park, H.Y.; Kazlauskas, R.J.; Kim, B.G. Cloning, expression and characterization of CYP102D1, a self-sufficient P450 monooxygenase fromStreptomyces avermitilis. FEBS J 2012, 279, 1650–1662. [Google Scholar]
- Choi, K.Y.; Jung, E.O.; Jung, D.H.; An, B.R.; Pandey, B.P.; Yun, H.; Sung, C.; Park, H.Y.; Kim, B.G. Engineering of daidzein 3′-hydroxylase P450 enzyme into catalytically self-sufficient cytochrome P450. Microb. Cell Fact 2012, 11, 81. [Google Scholar]
- Pandey, B.P.; Roh, C.; Choi, K.Y. Regioselective hydroxylation of daidzein using P450 (CYP105D7) from Streptomyces avermitilis MA4680. Biotechnol. Bioeng 2010, 105, 697–704. [Google Scholar]
- Choi, K.Y.; Jung, E.O.; Kazlauskas, R.; Kim, B.G. Development of colorimetric HTS assay of cytochrome P450 for ortho-specific hydroxylation and engineering of CYP102D1 with enhanced catalytic activity and regioselectivity. Chembiochem 2013, 14, 1231–1238. [Google Scholar]
- Lee, H.; Kim, B.G.; Ahn, J.H. Production of bioactive hydroxyflavones by using monooxygenase fromSaccharothrix espanaensis. J. Biotechnol 2014, 176, 11–17. [Google Scholar]
- Kim, H.J.; Choi, K.Y.; Jung, D.H.; Jung, J.Y.; Jung, E.O.; Yang, Y.H.; Kim, B.G.; Oh, M.K. Transcriptomic study for screening genes involved in the oxidative bioconversions ofStreptomyces avermitilis. Bioprocess Biosyst. Eng 2013, 36, 1621–1630. [Google Scholar]
- Pandey, B.P.; Lee, N.; Choi, K.Y.; Kim, J.N.; Kim, E.J.; Kim, B.G. Identification of the specific electron transfer proteins, ferredoxin, and ferredoxin reductase, for CYP105D7 in Streptomyces avermitilis MA4680. Appl. Microbiol. Biotechnol 2014. [Google Scholar] [CrossRef]
- Jung, S.T.; Lauchli, R.; Arnold, F.H. Cytochrome P450: Taming a wild type enzyme. Curr. Opin. Biotechnol 2011, 22, 1–9. [Google Scholar]
- Hlavica, P. Assembly of non-natural electron transfer conduits in the cytochrome P450 system: A critical assessment and update of artificial redox constructs amenable to exploitation in biotechnological areas. Biotechnol. Adv 2009, 27, 103–121. [Google Scholar]
- Chang, T.S.; Chao, S.Y.; Chen, Y.C. Production of ortho-hydroxydaidzein derivatives by a recombinant strain of Pichia pastoris harboring a cytochrome P450 fusion gene. Process Biochem 2013, 48, 426–429. [Google Scholar]
- Chang, T.S. Department of Biological Science and Technology; National University of Tainan; Unpublished observation; 2014. [Google Scholar]
- Chang, T.S. Department of Biological Science and Technology; National University of Tainan; Unpublished observation; 2014. [Google Scholar]
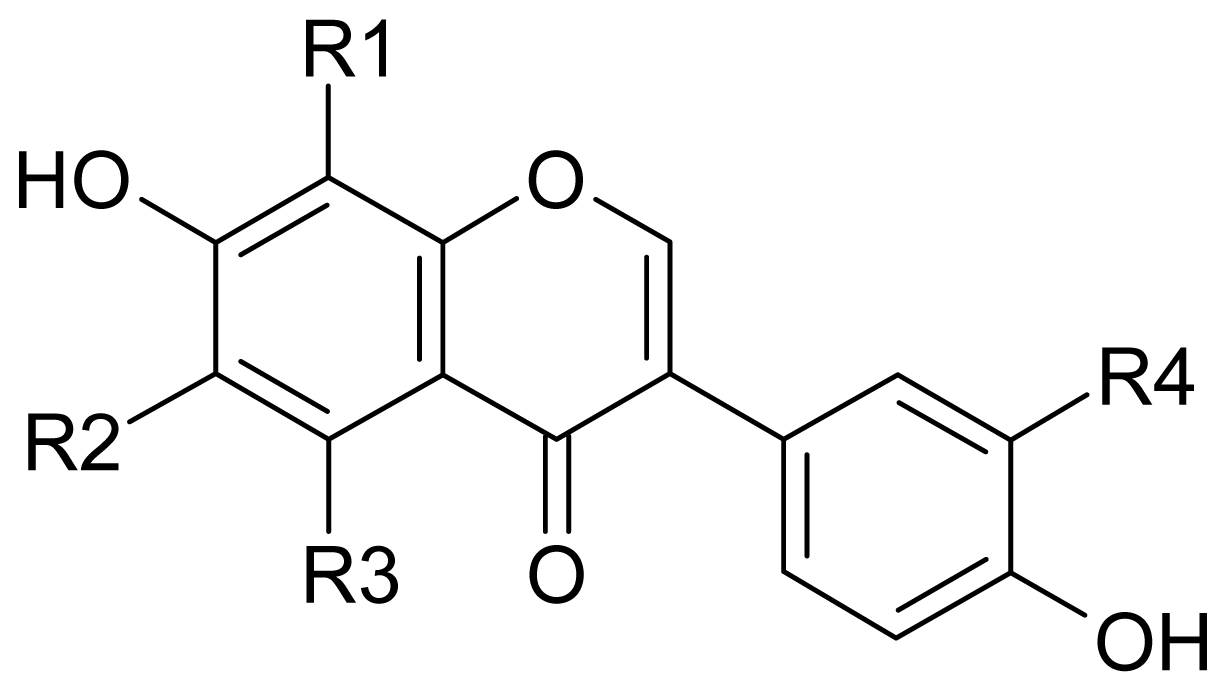

| Sources | Name | Microorganisms | Products | Ref. |
|---|---|---|---|---|
| Plants | Orobus tuberuosus | --- a | 3′-OHG | [22] |
| Calophyllum polyanthum | --- | 3′-OHG | [23] | |
| Erycibe expansa | --- | 3′-OHG | [24] | |
| Eclipta prostrata | --- | 3′-OHG | [25] | |
| Sophora japonica | --- | 3′-OHG | [26] | |
| Machaerium villosum | --- | 3′-OHD | [29] | |
| Dalbergia odorifera | --- | 3′-OHD | [30] | |
| Styphnolobium japponicum | --- | 3′-OHD | [31] | |
| Soybean Foods | Indonesian Tempeh | Rhizopus and other bacteria | 6-OHD, 8-OHD, 3′-OHG | [27,32] |
| Japanese Soybean koji | A. saitoi | 8-OHD, 8-OHG, 6-OHD | [33,34] | |
| Japanese Miso | A. oryzae | 8-OHD, 8-OHG, 6-OHD, 3′-OHD | [35–38] | |
| Chinese Douchi | A. oryzae | 6-OHD, 8-OHD, 3′-OHD, 8-OHG | [39] | |
| Korean Doenjang | diverse fungi and bacteria | 6-OHD, 8-OHD, 3′-OHD | [40] | |
| Microbial Fermentation Broth | --- | A. niger | 8-OHG, 3′-OHG | [28] |
| --- | Streptomyces sp. | 8-OHD, 3′-OHD | [43,44] | |
| --- | Micrococcus sp. or Arthrobacter sp. | 6-OHG | [45] | |
| --- | P. productus | 6-OHG | [46] | |
| Classification | Compound | Bioactivity | Ref. |
|---|---|---|---|
| Anticancer-Related Activity | 3′-OHG | Antiproliferative activity toward T47D tumorigenic breast epithelial cells; | [52,53] |
| 8-OHD | Increases life span against S180 bearing mice; | [43] | |
| Suppression of MDR in Caco-2 colon adenocarcinoma cells; | [58] | ||
| 8-OHG | Antimutagenesis activity; | [36] | |
| Antiproliferative activity toward HL-60 promyelocytic leukemia cells; | [35] | ||
| 6-OHD | Suppression of HCT-116 colon cancer cell proliferation in vitro and in vivo; | [54] | |
| 3′-OHD | Suppression of EGF receptor-positive skin cancer cell proliferation in vitro and in vivo; | [55,56] | |
| Suppression of MDR transporters; | [57] | ||
| Antimelanogenesis- Related Activity | 6-OHD | Competitive tyrosinase inhibitor; | [60] |
| 8-OHD | Irreversible tyrosinase inhibitor; | [61,62] | |
| Antimelanogenesis activity in human volunteers; | [63] | ||
| 8-OHG | Irreversible tyrosinase inhibitor; | [61,62] | |
| 3′-OHD | Antimelanogenesis activity in human skin equivalents; | [64,65] | |
| Potent autophage inducer; | [67] | ||
| Other Bioactivities | 3′-OHG | HIV-1 integrase inhibitor; | [25] |
| Antiinflammatory activity; | [68] | ||
| Hepatoprotective activity; | [24] | ||
| 6-OHG | Hepatoprotective activity; | [46] | |
| 8-OHD | Aldose reductase inhibitor; | [69] | |
| 6-OHD | Suppression on adipogenesis of 3T3-L1 preadipocytes; | [70] | |
| Promoting differentiation of 3T3-L1 preadipocytes; | [71] | ||
| Antitrypanosomal activity | [72] | ||
| 3′-OHD | Antitrypanosomal activity; | [72,73] | |
| Improving atopic dermatitis symptoms; | [74] | ||
| Monooxygenase name | Monooxygenase sources | Recombinant hosts | Products | Yield (mg/L) | Ref. |
|---|---|---|---|---|---|
| CYP107H1 a | B. subtilis | E. coli | 3′-OHD | <0.1 | [75] |
| Nfa33880 a | N. farcinica | E. coli | 8-OHD 6-OHD | 0.76 0.56 | [76] |
| CYP154 a | N. farcinica | E. coli | 3′-OHD | 0.99 | [77] |
| CYP102D1 b | S. avermitilis | E. coli | 8-OHD 6-OHD | 2.42 1.18 | [78] |
| CYP105D7 c | S. avermitilis | S. avermitilis | 3′-OHD | 9.3 | [79] |
| CYP105D7 a | S. avermitilis | S. avermitilis | 3′-OHD | 37.5 | [80] |
| CYP107Y1 a | S. avermitilis | S. avermitilis | 3′-OHG | 6.75 | [21] |
| Sam5 | S. espanaensis | E. coli | 3′-OHD | 75 | [82] |
| CYP57B3 a | A. oryzae | S. cerevisiae | 3′-OHG | N.D. d | [16] |
| CYP57B3 c | A. oryzae | P. pastoris | 6-OHD | 9.1 | [87] |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, T.-S. Isolation, Bioactivity, and Production of ortho-Hydroxydaidzein and ortho-Hydroxygenistein. Int. J. Mol. Sci. 2014, 15, 5699-5716. https://doi.org/10.3390/ijms15045699
Chang T-S. Isolation, Bioactivity, and Production of ortho-Hydroxydaidzein and ortho-Hydroxygenistein. International Journal of Molecular Sciences. 2014; 15(4):5699-5716. https://doi.org/10.3390/ijms15045699
Chicago/Turabian StyleChang, Te-Sheng. 2014. "Isolation, Bioactivity, and Production of ortho-Hydroxydaidzein and ortho-Hydroxygenistein" International Journal of Molecular Sciences 15, no. 4: 5699-5716. https://doi.org/10.3390/ijms15045699
APA StyleChang, T.-S. (2014). Isolation, Bioactivity, and Production of ortho-Hydroxydaidzein and ortho-Hydroxygenistein. International Journal of Molecular Sciences, 15(4), 5699-5716. https://doi.org/10.3390/ijms15045699