Cloning, Expression and Characterization of a Novel Thermophilic Polygalacturonase from Caldicellulosiruptor bescii DSM 6725
Abstract
:1. Introduction
2. Results and Discussion
2.1. Sequence Alignment and Phylogenetic Analysis of ACM61449 and ACM60667
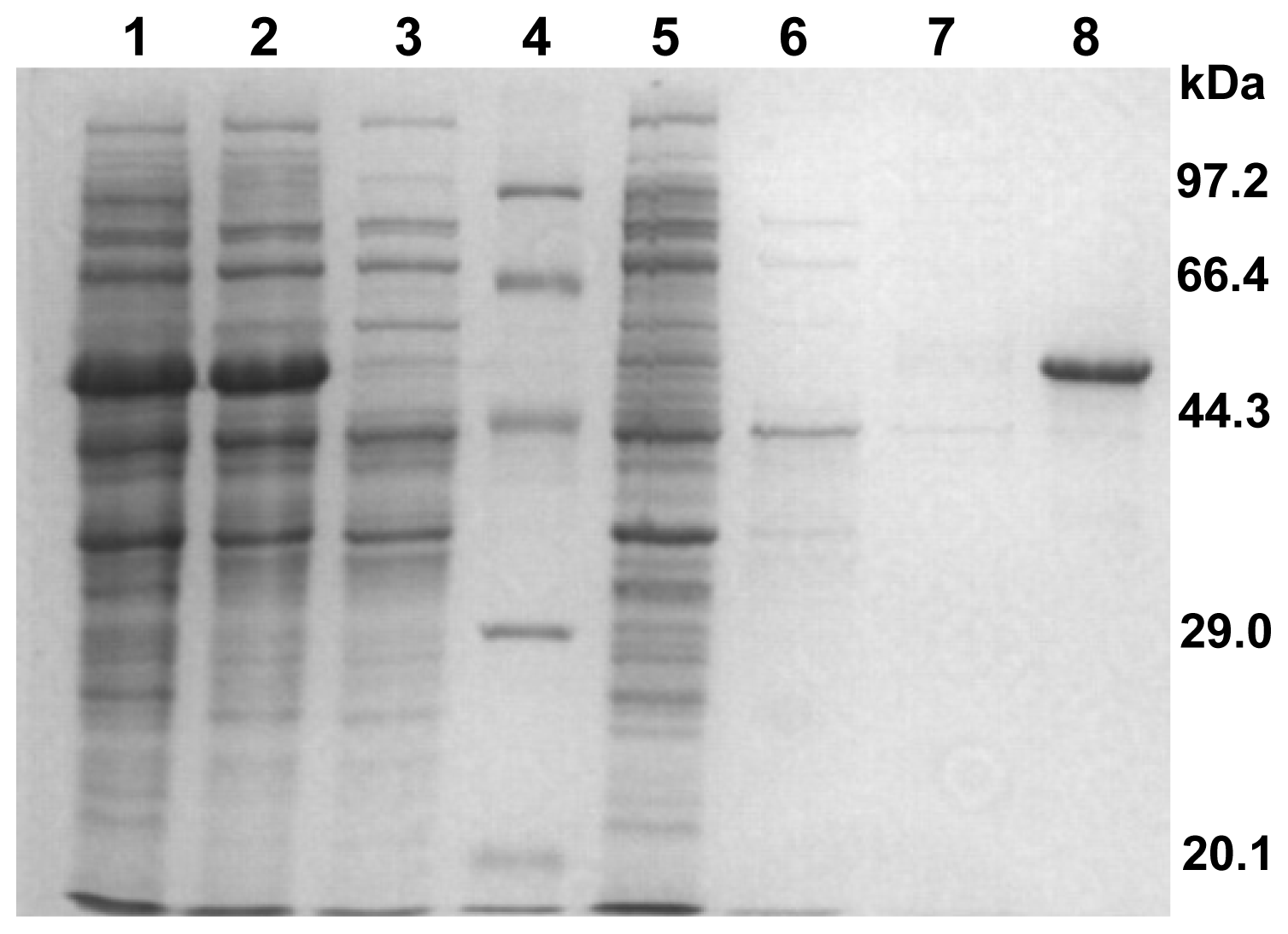
2.2. Cloning, Expression, and Purification of CbPelA
2.3. Enzyme Properties of CbPelA
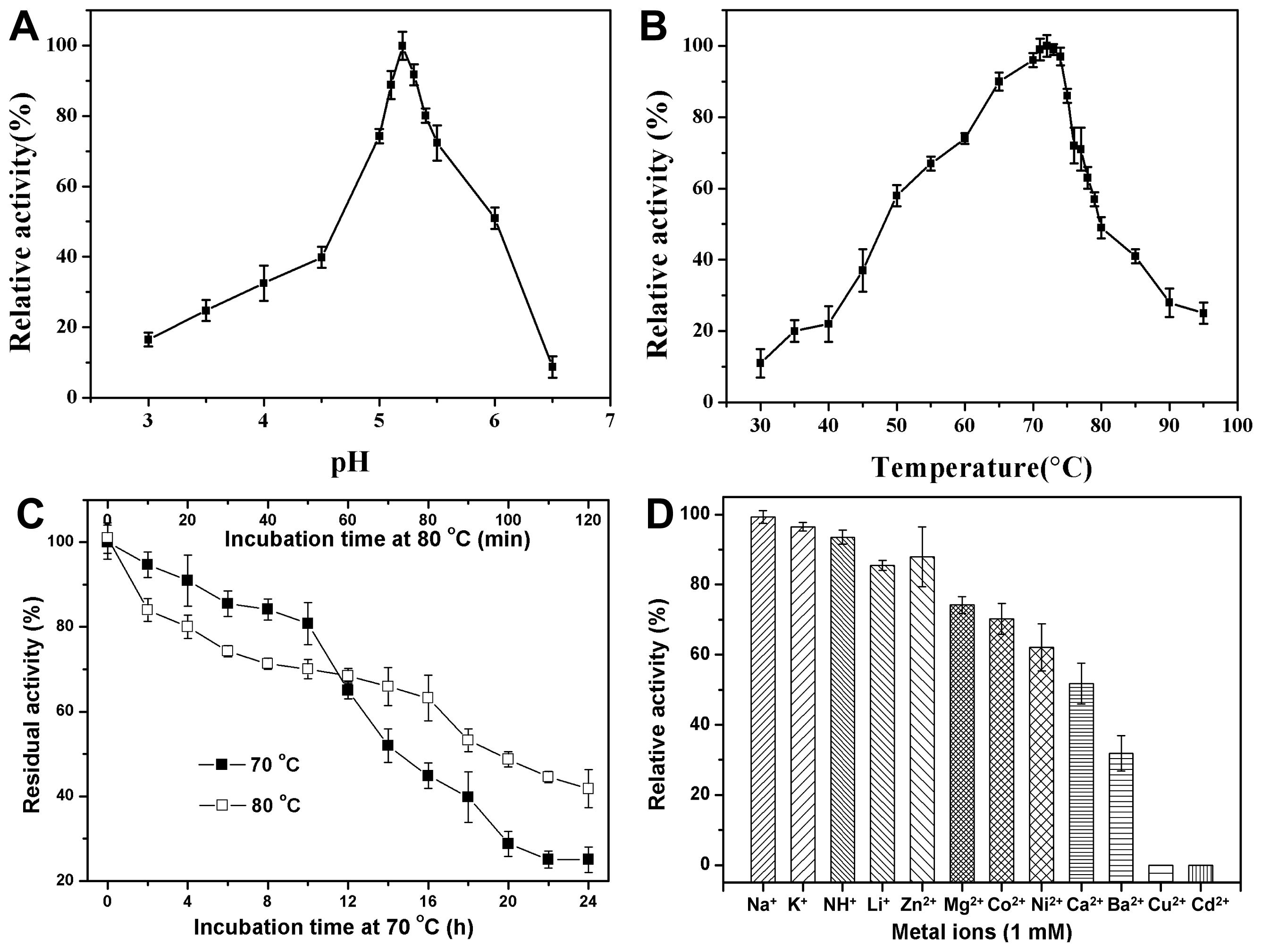
2.3.1. Effect of pH on CbPelA Activity
2.3.2. Effect of Temperature on CbPelA Activity
2.3.3. Thermo-Stability of CbPelA
2.3.4. The Effect of Metal Ions on CbPelA Activity
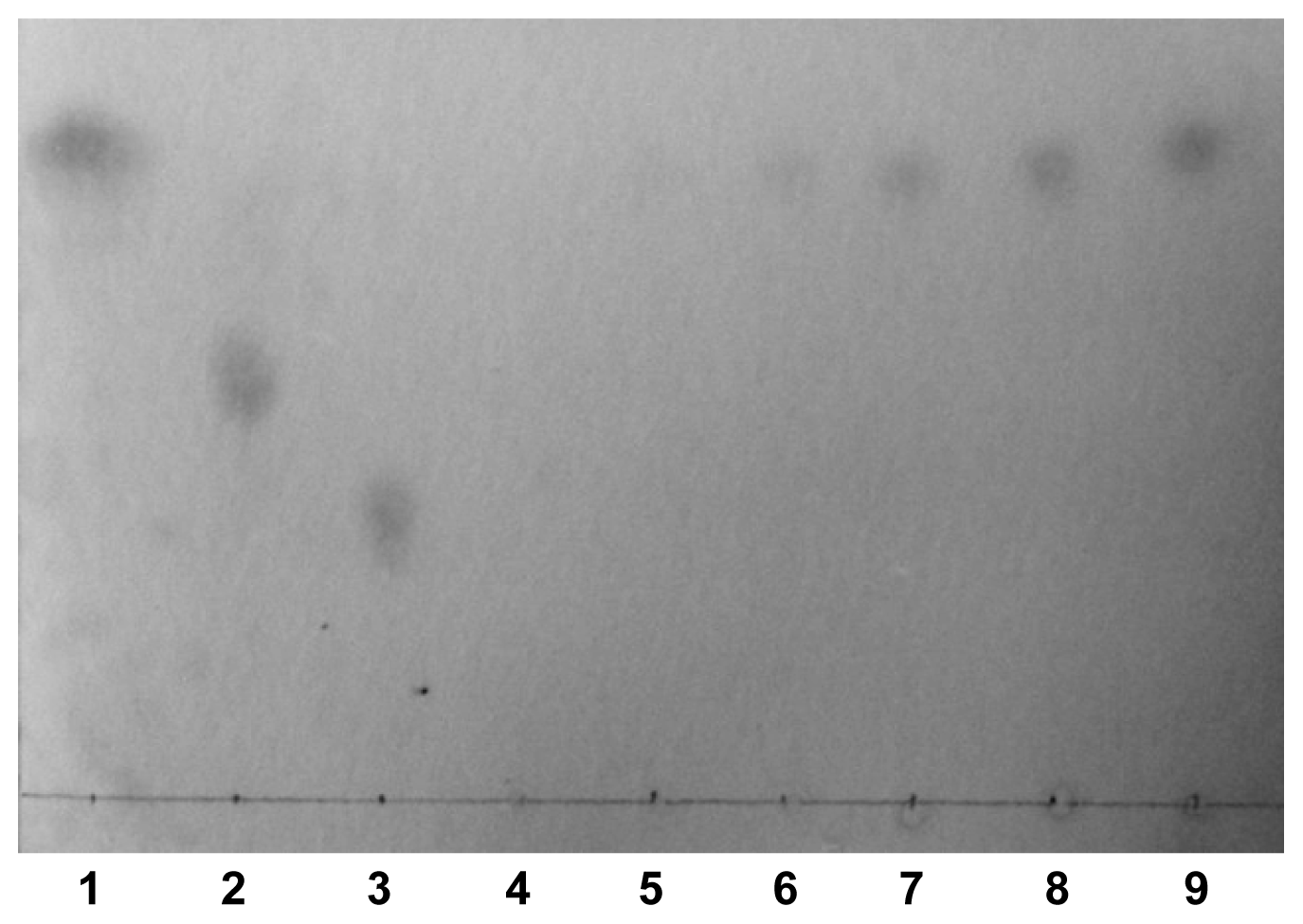
2.4. Analysis of the Degradation Products of CbPelA
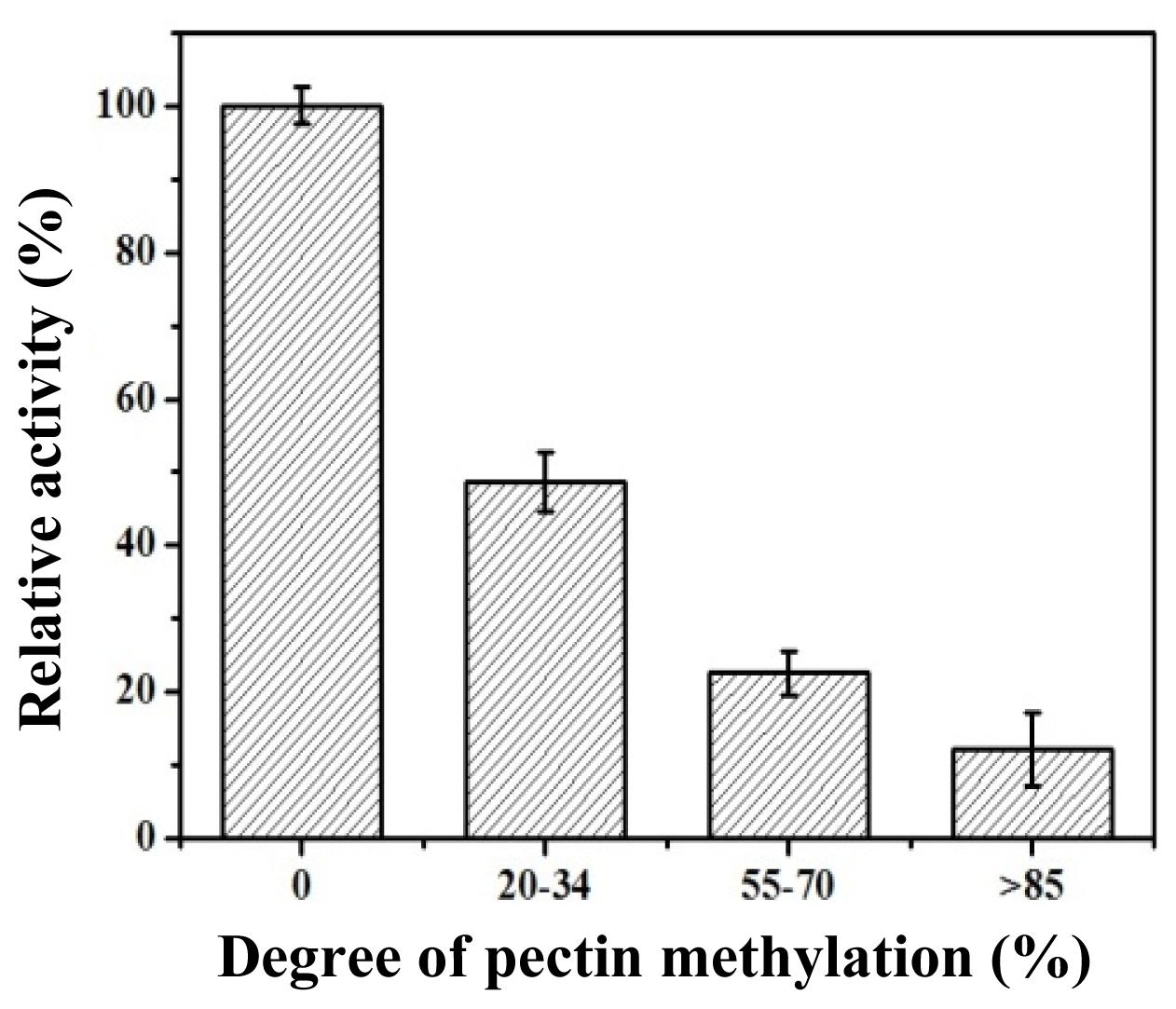
2.5. Substrate Specificity
3. Methods
3.1. Recombinant DNA Techniques
3.2. Expression and Purification of pCbPelA
3.3. Enzyme Activity Assay
3.4. Enzyme Characterization of CbPelA
3.5. Thin Layer Chromatography
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsConceived and designed the experiments: Y.C., D.S., Y.Z., Z.Z. Performed the experiments: Y.C., L.L., W.H., B.Z. Analyzed the data: Y.C., D.S., Y.Z., W.H., B.Z., Z.W. Contributed reagents/materials/analysis tools: W.H., B.Z., Z.W., Z.Z. Wrote the paper: Y.C., Z.W., Z.Z.
References
- Jayani, R.S.; Saxena, S.; Gupta, R. Microbial pectinolytic enzymes: A review. Proc. Biochem 2005, 40, 2931–2944. [Google Scholar]
- Benoit, I.; Coutinho, P.; Schols, H.; Gerlach, J.; Henrissat, B.; de Vries, R. Degradation of different pectins by fungi: Correlations and contrasts between the pectinolytic enzyme sets identified in genomes and the growth on pectins of different origin. BMC Genomics 2012, 13, 1–11. [Google Scholar]
- O’Neill, M.A.; Ishii, T.; Albersheim, P.; Darvill, A.G.; Rhamnogalacturonan, I.I. Structure and function of a borate cross-linked cell wall pectic polysaccharide. Ann. Rev. Plant Biol 2004, 55, 109–139. [Google Scholar]
- De Vries, R.P.; Visser, J. Aspergillus enzymes involved in degradation of plant cell wall polysaccharides. Microbiol. Mol. Biol. Rev 2001, 65, 497–522. [Google Scholar]
- Gummadi, S.N.; Kumar, D.S. Microbial pectic transeliminases. Biotechnol. Lett 2005, 27, 451–458. [Google Scholar]
- Markovič, O.; Janeček, Š. Pectin degrading glycoside hydrolases of family 28: Sequence-structural features, specificities and evolution. Protein Eng 2001, 14, 615–631. [Google Scholar]
- Kobayashi, T.; Higaki, N.; Yajima, N.; Suzumatsu, A.; Hagihara, H.; Kawai, S.; Ito, S. Purification and properties of a galacturonic acid-releasing exo-polygalacturonase from a strain ofBacillus. Biosci. Biotechnol. Biochem 2001, 65, 842–847. [Google Scholar]
- Yuan, P.; Meng, K.; Wang, Y.; Luo, H.; Shi, P.; Huang, H.; Bai, Y.; Yang, P.; Yao, B. A protease-resistant exo-polygalacturonase from Klebsiella sp. Y1 with good activity and stability over a wide pH range in the digestive tract. Bioresour. Technol 2012, 123, 171–176. [Google Scholar]
- He, S.Y.; Collmer, A. Molecular cloning, nucleotide sequence, and marker exchange mutagenesis of the exo-poly-alpha-d-galacturonosidase-encoding pehX gene of Erwinia chrysanthemi EC16. J. Bacteriol 1990, 172, 4988–4995. [Google Scholar]
- Nasser, W.; Shevchik, V.E.; Hugouvieux-Cotte-Pattat, N. Analysis of three clustered polygalacturonase genes in Erwinia chrysanthemi 3937 revealed an anti-repressor function for the PecS regulator. Mol. Microbiol 1999, 34, 641–650. [Google Scholar]
- Garcia-Maceira, F.; di Pietro, A.; Roncero, M.I.G. Cloning and disruption of pgx4 encoding an in planta expressed exo-polygalacturonase fromFusarium oxysporum. Mol. Plant Microbe Interac 2000, 13, 359–365. [Google Scholar]
- De las Heras, A.; Patiño, B.; Posada, M.L.; Martínez, M.J.; Vázquez, C.; González Jaén, M.T. Characterization and in vitro expression patterns of an exo-polygalacturonase encoding gene from Fusarium oxysporum f. sp. radicis lycopersici. J. Appl. Microbiol 2003, 94, 856–864. [Google Scholar]
- Pedrolli, D.B.; Carmona, E.C. Purification and characterization of the exo-polygalacturonase produced by Aspergillus giganteus in submerged cultures. J. Ind. Microbiol. Biotechnol 2010, 37, 567–573. [Google Scholar]
- Fahmy, A.S.; EI-Beih, F.M.; Mohamed, S.A.; Abdel-Gany, S.S.; Abd-Elbaky, E.A. Characterization of an exo-polygalacturonase fromAspergillus niger. Appl. Biochem. Biotechnol 2008, 149, 205–217. [Google Scholar]
- Sakai, T.; Sakamoto, T.; Hallaert, J.; Vandamme, E.J. Pectin, pectinase and protopectinase: Production, properties, and applications. Adv. Appl. Microbiol 1993, 39, 213–294. [Google Scholar]
- Eriksson, K.E.; Athens, G.A. Biotechnology in the pulp and paper industry. Wood Sci. Technol 1990, 24, 79–101. [Google Scholar]
- Vieille, C.; Zeikus, G.J. Hyperthermophilic enzymes: Sources, uses, and molecular mechanisms for thermostability. Microbiol. Mol. Biol. Rev 2001, 65, 1–43. [Google Scholar]
- Pijning, T.; van Pouderoyen, G.; Kluskens, L.; van der Oost, J.; Dijkstra, B.W. The crystal structure of a hyperthermoactive exo-polygalacturonase from Thermotoga maritima reveals a unique tetramer. FEBS Lett 2009, 583, 3665–3670. [Google Scholar]
- Kumar, S.S.; Palanivelu, P. Purification and characterization of an extracellular polygalacturonase from the thermophilic fungusThermomyces lanuginosus. World J. Microbiol. Biotechnol 1999, 15, 643–646. [Google Scholar]
- Puchart, V.R.; Katapodis, P.; Biely, P.; Kremnický, L.R.; Christakopoulos, P.; Vršanská, M.; Kekos, D.; Macris, B.J.; Bhat, M.K. Production of xylanases, mannanases, and pectinases by the thermophilic fungusThermomyces lanuginosus. Enzym. Microb. Technol 1999, 24, 355–361. [Google Scholar]
- Martins, E.S.; Silva, D.; Da Silva, R.; Gomes, E. Solid state production of thermostable pectinases from thermophilicThermoascus aurantiacus. Proc. Biochem 2002, 37, 949–954. [Google Scholar]
- Kaur, G.; Kumar, S.; Satyanarayana, T. Production, characterization and application of a thermostable polygalacturonase of a thermophilic mould Sporotrichum thermophile Apinis. Bioresour. Technol 2004, 94, 239–243. [Google Scholar]
- Kapoor, M.; Khalil Beg, Q.; Bhushan, B.; Dadhich, K.; Hoondal, G. Production and partial purification and characterization of a thermo-alkali stable polygalacturonase from Bacillus sp. MG-cp-2. Proc. Biochem 2000, 36, 467–473. [Google Scholar]
- Kluskens, L.; van Alebeek, G.; Voragen, A.; de Vos, W.; van der Oost, J. Molecular and biochemical characterization of the thermoactive family 1 pectate lyase from the hyperthermophilic bacteriumThermotoga maritima. Biochem. J 2003, 370, 651–659. [Google Scholar]
- Kozianowski, G.; Canganella, F.; Rainey, F.; Hippe, H.; Antranikian, G. Purification and characterization of thermostable pectate-lyases from a newly isolated thermophilic bacterium, Thermoanaerobacter italicus sp. nov. Extremophiles 1997, 1, 171–182. [Google Scholar]
- Takao, M.; Nakaniwa, T.; Yoshikawa, K.; Terashita, T.; Sakai, T. Purification and characterization of thermostable pectate lyase with protopectinase activity from thermophilic Bacillus sp. TS47. Biosci. Biotechnol. Biochem 2000, 64, 2360–2367. [Google Scholar]
- Kluskens, L.D.; van Alebeek, G.J.W.; Walther, J.; Voragen, A.G.; de Vos, W.M.; van der Oost, J. Characterization and mode of action of an exo-polygalacturonase from the hyperthermophilic bacteriumThermotoga maritima. FEBS J 2005, 272, 5464–5473. [Google Scholar]
- Parisot, J.; Langlois, V.; Sakanyan, V.; Rabiller, C. Cloning expression and characterization of a thermostable exo-polygalacturonase fromThermotoga maritima. Carbohydr. Res 2003, 338, 1333–1337. [Google Scholar]
- Singh, R.; Dhawan, S.; Singh, K.; Kaur, J. Cloning, expression and characterization of a metagenome derived thermoactive/thermostable pectinase. Mol. Biol. Rep 2012, 39, 8353–8361. [Google Scholar]
- Kataeva, I.A.; Yang, S.J.; Dam, P.; Poole, F.L.; Yin, Y.; Zhou, F.; Chou, W.C.; Xu, Y.; Goodwin, L.; Sims, D.R. Genome sequence of the anaerobic, thermophilic, and cellulolytic bacterium “Anaerocellum thermophilum” DSM 6725. J. Bacteriol 2009, 191, 3760–3761. [Google Scholar]
- Cantarel, B.L.; Coutinho, P.M.; Rancurel, C.; Bernard, T.; Lombard, V.; Henrissat, B. The carbohydrate-active enzyme database (CAZy), an expert resource for glycogenomic. Nucleic Acids Res 2009, 37, D233–D238. [Google Scholar]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J.; Clustal, W. Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 1994, 22, 4673–4680. [Google Scholar]
- Kester, H.C.; Kusters-van Someren, M.A.; Müller, Y.; Visser, J. Primary structure and characterization of an exo-polygalacturonase fromAspergillus tubingensis. Eur. J. Biochem 1996, 240, 738–746. [Google Scholar]
- Dong, Z.; Wang, Z. Isolation and characterization of an exo-polygalacturonase from Fusarium oxysporum f. sp. cubense race 1 and race 4. BMC Biochem 2011, 12, 51. [Google Scholar]
- Warrilow, A.G.; Jones, M.G. Different forms of tomato pectinesterase have different kinetic properties. Phytochemistry 1995, 39, 277–282. [Google Scholar]
- Yang, S.J.; Kataeva, I.; Hamilton-Brehm, S.D.; Engle, N.L.; Tschaplinski, T.J.; Doeppke, C.; Davis, M.; Westpheling, J.; Adams, M.W. Efficient degradation of lignocellulosic plant biomass, without pretreatment, by the thermophilic anaerobe “Anaerocellum thermophilum” DSM 6725. Appl. Environ. Microbiol 2009, 75, 4762–4769. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Biochem 1959, 31, 426–428. [Google Scholar]
- Li, G.; Rao, L.; Xue, Y.; Zhou, C.; Zhang, Y.; Ma, Y. Cloning, expression, and characterization of a highly active alkaline pectate lyase from alkaliphilic Bacillus sp. N16–5. J. Microbiol. Biotechnol 2010, 20, 670–677. [Google Scholar]


| Organism | Name | Molecular mass (kDa) | pH optimum | Temperature optimum (°C) | Vmax (U/mg) | Reference |
|---|---|---|---|---|---|---|
| Caldiceelulosiruptor bescii | PelA | 50 | 5.2 | 72 | 384.6 | This study |
| Thermotoga maritime | PelB | 50 | 6.4 | 80 | 1170 | [27] |
| Metagenome | PecJKR01 | 47.9 | 7.0 | 70 | - | [29] |
| Klebsiella sp. Yl | PguB | 72 | 6.0 | 40–50 | 47.0 | [8] |
| Aspergillus tubingensis | PgaX | 78 | 4.2 | 30 | 602.8 | [33] |
| Aspergillus giganteus | PG | 69.7 | 6.0 | 55–60 | 255 | [13] |
| Fusarium oxysporum | Pgc2 | 63 | 5.0 | 50 | 95.24 | [34] |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, Y.; Sun, D.; Zhou, Y.; Liu, L.; Han, W.; Zheng, B.; Wang, Z.; Zhang, Z. Cloning, Expression and Characterization of a Novel Thermophilic Polygalacturonase from Caldicellulosiruptor bescii DSM 6725. Int. J. Mol. Sci. 2014, 15, 5717-5729. https://doi.org/10.3390/ijms15045717
Chen Y, Sun D, Zhou Y, Liu L, Han W, Zheng B, Wang Z, Zhang Z. Cloning, Expression and Characterization of a Novel Thermophilic Polygalacturonase from Caldicellulosiruptor bescii DSM 6725. International Journal of Molecular Sciences. 2014; 15(4):5717-5729. https://doi.org/10.3390/ijms15045717
Chicago/Turabian StyleChen, Yanyan, Dejun Sun, Yulai Zhou, Liping Liu, Weiwei Han, Baisong Zheng, Zhi Wang, and Zuoming Zhang. 2014. "Cloning, Expression and Characterization of a Novel Thermophilic Polygalacturonase from Caldicellulosiruptor bescii DSM 6725" International Journal of Molecular Sciences 15, no. 4: 5717-5729. https://doi.org/10.3390/ijms15045717
APA StyleChen, Y., Sun, D., Zhou, Y., Liu, L., Han, W., Zheng, B., Wang, Z., & Zhang, Z. (2014). Cloning, Expression and Characterization of a Novel Thermophilic Polygalacturonase from Caldicellulosiruptor bescii DSM 6725. International Journal of Molecular Sciences, 15(4), 5717-5729. https://doi.org/10.3390/ijms15045717






