Eruca sativa Might Influence the Growth, Survival under Simulated Gastrointestinal Conditions and Some Biological Features of Lactobacillus acidophilus, Lactobacillus plantarum and Lactobacillus rhamnosus Strains
Abstract
:1. Introduction
2. Results and Discussion
2.1. Growth of Bacteria and Resistance to the Simulated Digestion
| T0 cfu/mL (SD) | T 24 h BGPJ treatment cfu/mL (SD) | T 24 h AGPJ treatment cfu/mL (SD) | |
|---|---|---|---|
| LA | 1.23 × 102 (0.01 × 102) | 1.25 × 108 (0.02 × 102) | 1.12 × 105 (0.03 × 102) |
| LA + E | 1.24 × 102 (0.02 × 102) | 8.15 × 107 (0.04 × 103) | 5.29 × 106 (0.02 × 103) |
| LP | 1.34 × 102 (0.02 × 102) | 1.55 × 108 (0.05 × 103) | 2.49 × 108 (0.03 × 103) |
| LP + E | 1.36 × 102 (0.01 × 102) | 2.46 ×108 (0.03 × 103) | 3.34 × 108 (0.05 × 103) |
| LR | 1.27 × 102 (0.02 × 102) | 1.14 × 108 (0.02 × 103) | 1.91 × 107 (0.03 × 103) |
| LR + E | 1.25 × 102 (0.02 × 102) | 4.43 × 107 (0.05 × 103) | 4.16 × 106 (0.03 × 103) |
2.2. Total Polyphenols and Antioxidant Activity
| Total polyphenols | ||
|---|---|---|
| T = 0 μg GAE/mL (SD) | T = 24 h μg GAE/mL (SD) | |
| MRS | 130.36 (1.07) | - |
| MRS + E.sativa | 230.48 (2.21) | - |
| LA | - | 139.25 (2.55) |
| LA + E.sativa | - | 218.14 (5.14) |
| LP | - | 138.04(2.12) |
| LP + E. sativa | - | 143.62 (3.01) |
| LR | - | 136.06 (0.55) |
| LR + E. sativa | - | 204.03 (1.43) |
| DDPPH test on growth medium | DDPPH test on microbial pellet | ||
|---|---|---|---|
| T = 0 | T = 24 h | T = 24 h | |
| EC50 (μL/mL ± SD) | EC50 (μL/mL ± SD) | Antioxidant power (%) | |
| MRS | 74.73 (2.64) | ||
| MRS + E.sativa | 49.29 (2.37) | ||
| LA | 92.13 (5.99) | 17.90 | |
| LA + E.sativa | 48.64 (1.15) | 22.95 | |
| LP | 84.80 (6.69) | 19.41 | |
| LP + E.sativa | 63.98 (1.98) | 24.02 | |
| LR | 70.38 (2.53) | 15.23 | |
| LR + E.sativa | 51.14 (2.39) | 14.03 | |
2.3. Antimicrobial Activity
2.4. Protein Profile
| LP | LP + E | LA | LA + E | LR | LR + E | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BGPJ | AGPJ | BGPJ | AGPJ | BGPJ | AGPJ | BGPJ | AGPJ | BGPJ | AGPJ | BGPJ | AGPJ | |
| BC 4313 | ||||||||||||
| 5 µL | - | - | 5 (0.5) | 2 (0.3) | 3.4 (0.3) | - | 3 (0) | - | 5 (0.3) | 1.2 (0) | 2 (0) | 2 (0) |
| 10 µL | 4 (0.3) | - | 7 (0.3) | 4 (0.2) | 6.3 (0.3) | 3 (0.3) | 6 (0.1) | 4 (0) | 7 (0.5) | 3 (0) | 6 (0.1) | 3 (0) |
| 20 µL | 8 (0.8) | 10 (0.4) | 12 (0.2) | 6 (0.3) | 10.3 (0.3) | 2.5 (0.5) | 7 (0.6) | 10.5 (0.3) | 12 (0.7) | 5 (0.5) | 10 (0.4) | 6 (0.2) |
| BC 4384 | ||||||||||||
| 5 µL | 2.5 (0.1) | 3 (0.1) | 5 (0.5) | 4 (0.3) | 4 (0) | - | 5 (0.2) | 2 (0) | 4 (0.3) | 2 (0.2) | 3.2 (0.3) | 3 (0.3) |
| 10 µL | 7 (0.3) | 9 (0.3) | 6 (0.4) | 6 (0.2) | 7 (0.3) | 3 (0) | 6.5 (0.5) | 4 (0.1) | 7.3 (0.4) | 4 (0.4) | 7.1 (0.4) | 3 (0.3) |
| 20 µL | 12 (0.7) | 12 (0.4) | 11 (0.4) | 12 (0.4) | 9.4 (0.6) | - | 10 (0.3) | 10 (0.3) | 11 (0.5) | 6 (0.1) | 11.3 (0.3) | 5.9 (0.3) |
| SA | ||||||||||||
| 5 µL | - | - | 5 (0.5) | 2 (0.2) | 4.5 (0.5) | - | 3.5 (0.5) | 3 (0.3) | 5 (0) | 2 (0) | 4 (0.3) | 1.5 (0) |
| 10 µL | 2 (0.2) | 6 (0.2) | 6 (0.1) | 3 (0.3) | 6.5 (0.5) | 3 (3.1) | 6.5 (0.3) | 5 (0.4) | 7 (0.4) | 3 (0) | 6 (0.2) | 3 (0) |
| 20 µL | 7 (0.4) | 6 (0.3) | 10 (0.3) | 4 (0.2) | 8.5 (0.5) | 6 (0.2) | 7.8 (0.3) | 10 (0.3) | 9 (0.3) | 5 (0.5) | 9 (0.3) | 5 (0.5) |
| PA | ||||||||||||
| 5 µL | 2 (0.2) | 2 (0.1) | 4 (0.4) | 2 (0.1) | 4 (0.3) | 1.5 (0.3) | 4.5 (0.5) | - | - | - | 2.2 (0.3) | - |
| 10 µL | 5 (0.1) | 3 (0.2) | 7 (0.4) | 3 (0.3) | 8 (0.4) | 4 (0.2) | 6 (0.6) | 2 (0.1) | 7 (0.5) | 2 (0.1) | 6 (0.4) | 2 (0.3) |
| 20 µL | 10 (0.3) | 5 (0.2) | 12 (0.8) | 3 (0.2) | 8.5 (0.5) | 4 (0.1) | 9 (0.3) | 2 (0.1) | 11 (0.4) | 3 (0.1) | 10.5 (0.3) | 3.2 (0.3) |
| EC | ||||||||||||
| 5 µL | - | - | 5 (0.2) | 2 (0.1) | 2.5 (0.1) | - | 4.8 (0.3) | 2 (0.2) | 3 (0.1) | 2 (0) | - | - |
| 10 µL | 2 (0.1) | - | 8 (0.3) | 1 (0) | 7 (0.4) | 3 (0.5) | 6.5 (0.5) | 4 (0.3) | 6 (0.2) | 1.3 (0) | 7.5 (0.3) | - |
| 20 µL | 9 (0.8) | 3 (0.3) | 12 (0.3) | 3 (0) | 10.4 (0.1) | 6 (0.1) | 10 (0.3) | 10 (0.6) | 11 (0.4) | 5 (0) | 11 (0.4) | 3 (0) |
| Gram-positive tester strains | Gram-negative tester strains | ||||
|---|---|---|---|---|---|
| P. aeruginosa | E. coli | S. aureus | B. cereus 4313 | B. cereus 4384 | |
| DMSO | 0 | 0 | 0 | 0 | 0 |
| Tetracycline (7 μg) | 9.8 (1.6) | 12 (1.2) | 11 (0.4) | 9 (0.6) | 8.4 (1.4) |
| MRS | 0 | 0 | 0 | 0 | 0 |
| MRS + E. sativa | 0 | 0 | 0 | 0 | 0 |
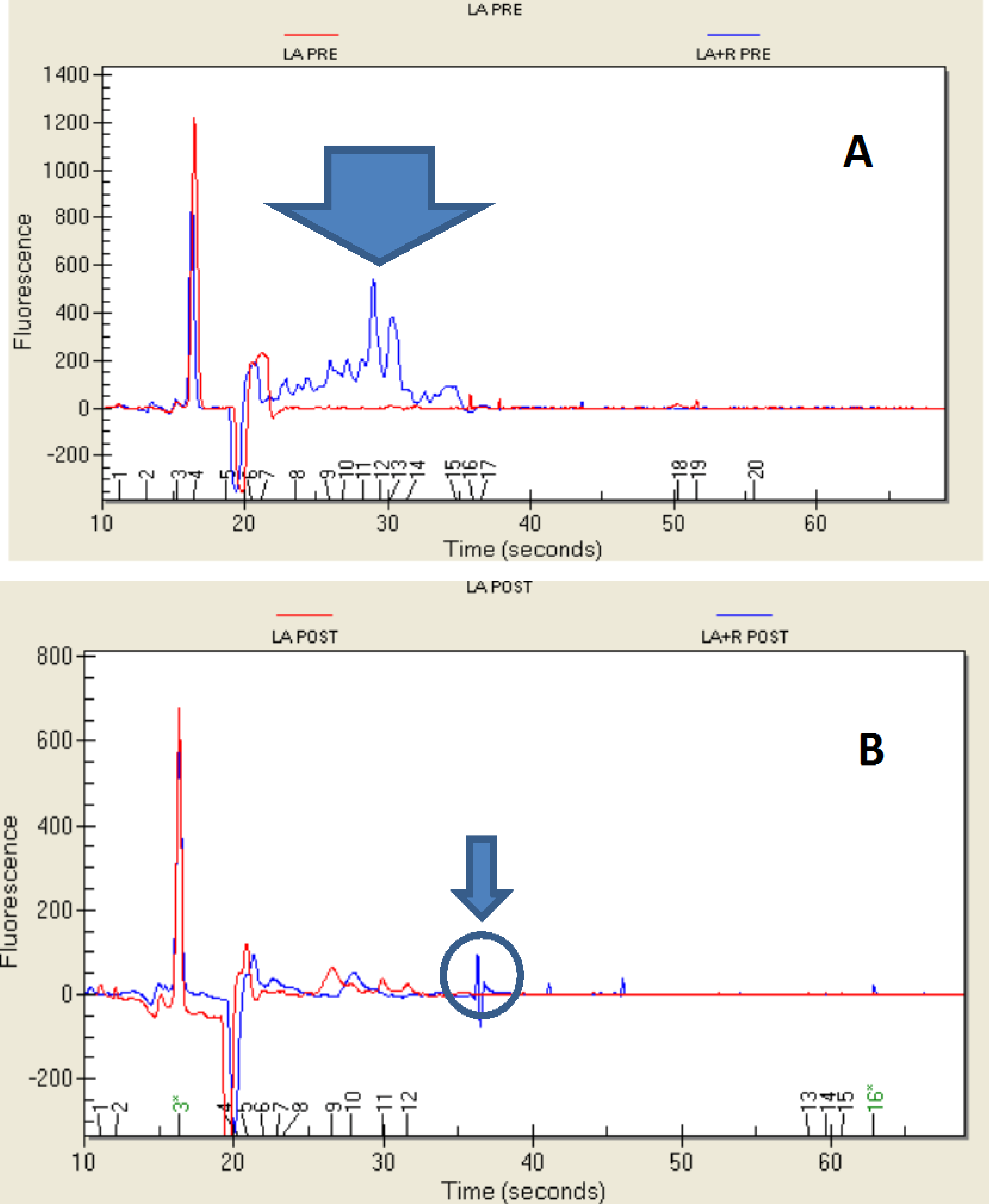
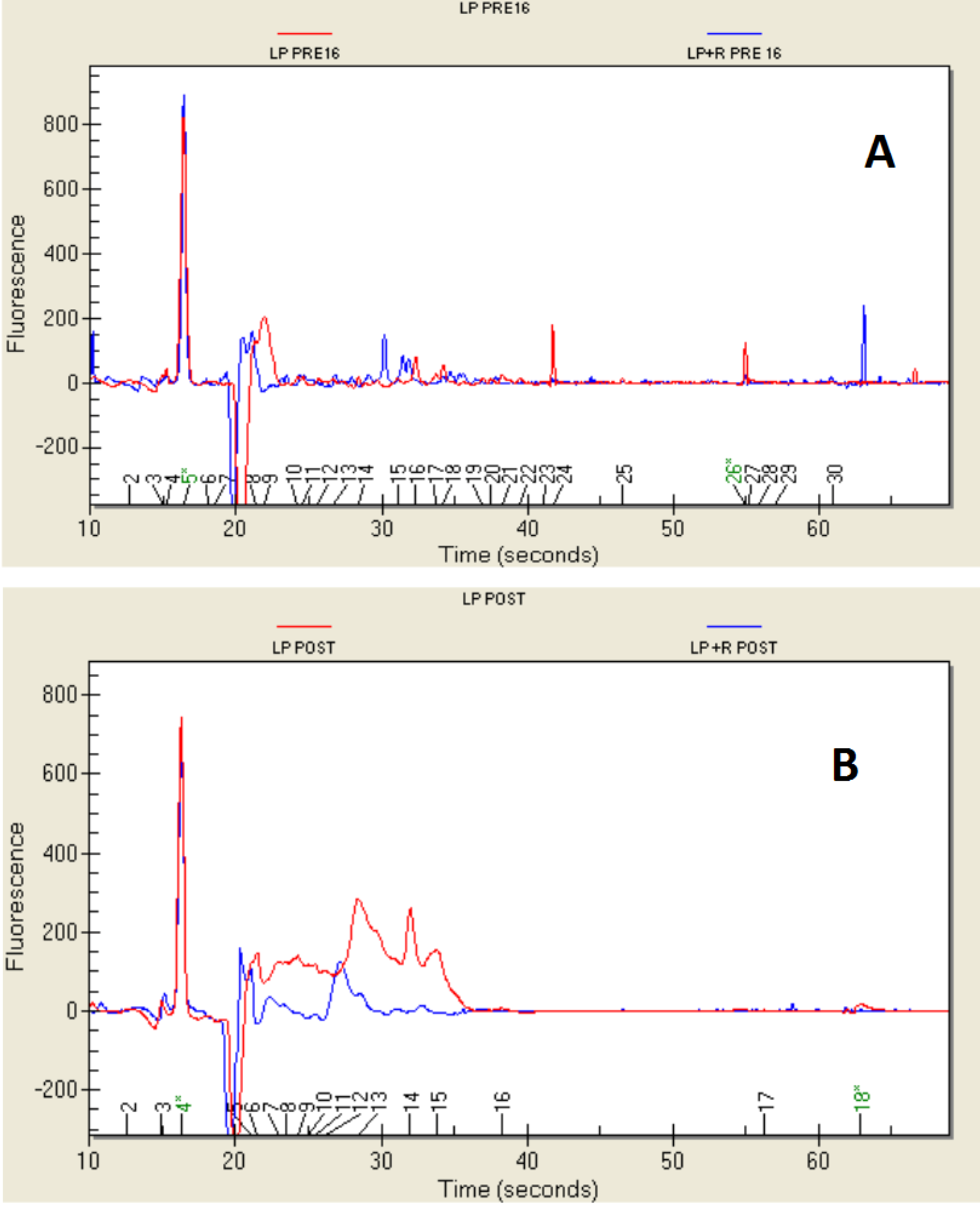
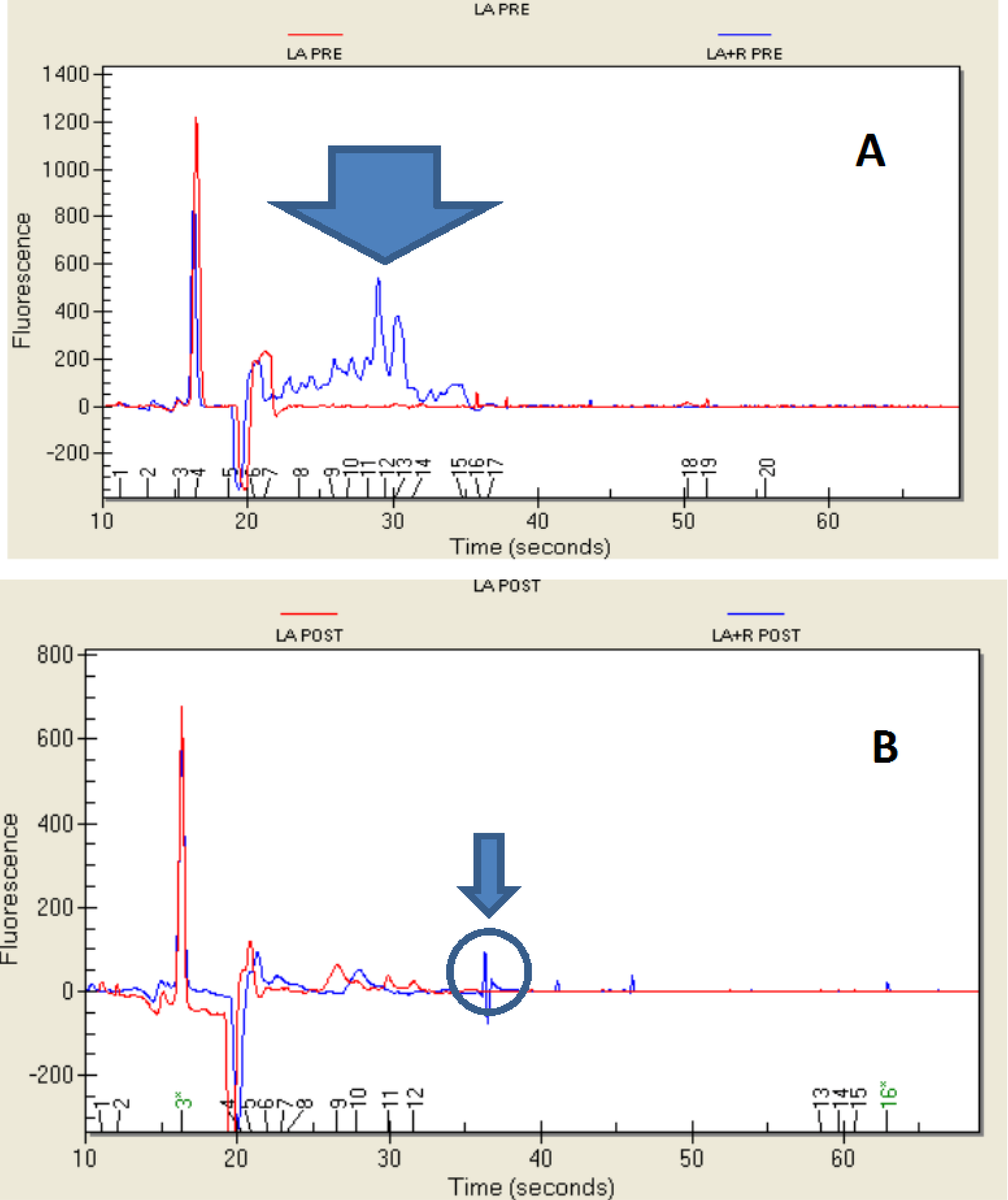
3. Experimental Section
3.1. Strain and Culture Conditions
3.2. Preparation of MRS Broth + Eruca sativa
3.3. Resistance to Simulated Gastric and Intestinal Juices
3.4. Colorimetric Analysis of Polyphenols
3.5. Preparation of Heat Killed Cells
3.6. Free Radical Scavenging Capacity
3.7. Protein Profile
3.8. Antimicrobial Assays
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Block, G.; Patterson, B.; Subar, A. Fruit, vegetables and cancer prevention. A review of the epidemiological evidence. Nutr. Cancer 1992, 18, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Protection against tissue damage in vivo by desferrioxammine: What is its mechanism of action? Free Rad. Biol. Med. 1989, 7, 645–651. [Google Scholar]
- Hertog, M.G.L.; Freskens, E.J.M.; Hollman, P.C.H.; Katan, M.B.; Kromhout, D. Dietary antioxidant flavonoids and the risk of coronary heart disease: The zutphen elverly study. Lancet 1993, 342, 1007–1011. [Google Scholar] [CrossRef] [PubMed]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Higdon, J.V.; Delage, B.; Williams, D.E.; Dashwood, R.H. Cruciferous vegetables and human cancer risk: Epidemiologic evidence and mechanistic basis. Pharm. Res. 2007, 55, 224–236. [Google Scholar] [CrossRef]
- Jin, J.; Koroleva, O.A.; Gibson, T.; Swanston, J.; Magan, J.; Zhang, Y.; Rowland, I.A.; Wagstaff, C. Analysis of phytochemical composition and chemoprotective capacity of rocket (Eruca sativa and Diplotaxis tenuifolia) leafy salad following cultivation in different environments. J. Agric. Food Chem. 2009, 57, 5227–5234. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Espín, J.C.; Tomás-Barberán, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef] [PubMed]
- Wells, C.L.; Jechorek, R.P.; Kinneberg, K.M.; Debol, S.M.; Erlandsen, S.L. The isoflavone genistein inhibits internalization of enteric bacteria by cultured Caco-2 and HT-29 enterocytes. J. Nutr. 1999, 129, 634–640. [Google Scholar] [PubMed]
- Parkar, S.G.; Stevenson, D.E.; Skinner, M.A. The potential influence of fruit polyphenols on colonic microflora and human gut health. Int. J. Food Microbiol. 2008, 124, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Vattem, D.A.; Lin, Y.T.; Ghaedian, R.; Shetty, K. Cranberry synergies for dietary management of Helicobacter pylori infections. Process Biochem. 2005, 40, 1583–1592. [Google Scholar] [CrossRef]
- Rodríguez-Vaquero, M.J.; Alberto, M.R.; Manca de Nadra, M.C. Antibacterial effect of phenolic compounds from different wines. Food Control 2007, 18, 93–101. [Google Scholar] [CrossRef]
- Almajano, M.P.; Carbó, R.; López-Jiménez, J.A.; Gordon, M.H. Antioxidant and antimicrobial activities of tea infusions. Food Chem. 2008, 108, 55–63. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Coppola, R. Quorum sensing and phytochemicals. Int. J. Mol. Sci. 2013, 14, 12607–12619. [Google Scholar] [CrossRef]
- Fratianni, F.; de Martino, L.; Melone, A.; de Feo, V.; Coppola, R.; Nazzaro, F. Preservation of chicken breast meat treated with thyme and balm essential oils. J. Food Sci. 2010, 75, M528–M535. [Google Scholar]
- Rodríguez, H.; Curiel, J.A.; Landete, J.M.; de Las Rivas, B.; de Felipe, F.L.; Gómez-Cordovés, C.; Mancheño, J.M.; Muñoz, R. Food phenolics and lactic acid bacteria. Int. J. Food Microbiol. 2009, 132, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Hervert-Hernández, D.; Pintado, C.; Rotger, R.; Goñi, I. Stimulatory role of grape pomace polyphenols on Lactobacillus acidophilus growth. Int. J. Food Microbiol. 2009, 136, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Fratianni, F.; Coppola, R.; Sada, A.; Mendiola, J.A.; Ibanez, E.; Nazzaro, F. A novel functional probiotic product containing phenolics and anthocyanins. Int. J. Probiotics Prebiotics 2010, 5, 85–90. [Google Scholar]
- Fratianni, F.; Cardinale, F.; Russo, I.; Iuliano, C.; Tremonte, P.; Coppola, R.; Nazzaro, F. Ability of synbiotic encapsulated Saccharomyces cerevisiae boulardii to grow in berry juice and to survive under simulated gastrointestinal conditions. J. Microencapsul. 2013, 31, 299–305. [Google Scholar] [CrossRef]
- Fratianni, F.; Cardinale, F.; Russo, I.; Iuliano, C.; Cucciniello, A.C.; Maione, M.; d’Acierno, A.; Nazzaro, F. Fermentation of tomato juice with the probiotic yeast Saccharomyces cerevisiae boulardii. In Functional Foods: Sources, Biotechnology Applications, and Health Challenges; Robinson, A., Emerson, D., Eds.; Nova Science Publisher: New York, NY, USA, 2013; pp. 143–152. [Google Scholar]
- Nazzaro, F.; Fratianni, F.; Sada, A.; Orlando, P. Synbiotic potential of carrot juice supplemented with Lactobacillus spp. and inulin or fructooligosaccharides. J. Sci. Food Agric. 2008, 88, 2271–2276. [Google Scholar] [CrossRef]
- China, R.; Mukherjee, S.; Sen, S.; Bose, S.; Datta, S.; Koley, H.; Ghosh, S.; Dhar, P. Antimicrobial activity of Sesbania grandiflora flower polyphenol extracts on some pathogenic bacteria and growth stimulatory effect on the probiotic organism Lactobacillus acidophilus. Microbiol. Res. 2012, 167, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Granato, D.; Branco, G.F.; Nazzaro, F.; Cruz, A.; Faria, J.A.F. Functional foods and nondairy probiotic food development: Trends, concepts, and products. Compr. Rev. Food Sci. Food Saf. 2010, 9, 292–302. [Google Scholar] [CrossRef]
- FAO/WHO Experts’ Report, 2001 FAO/WHO Experts’ Report. Processing of Health and Nutritional Properties of Probiotics in Food Including Powder Milk With Live Lactic Acid Bacteria, Córdoba, Argentina, 1–4 October 2001.
- Laparra, J.M.; Sanz, Y. Interactions of gut microbiota with functional food components and nutraceuticals. Pharm. Res. 2010, 61, 219–225. [Google Scholar] [CrossRef]
- Irshad, M.; Ahmad, I.; Goel, H.C.; Rizvi, M.M.A. Phytochemical screening and high performance TLC analysis of some cucirbits. Res. J. Phytochem. 2010, 4, 242–247. [Google Scholar] [CrossRef]
- Sharma, D.; Rawat, I.; Goel, H.C. Antioxidant and prebiotic potential of some Cucurbits. Res. J. Med. Plant 2012, 6, 500–510. [Google Scholar] [CrossRef]
- Lee, H.C.; Jenner, A.M.; Lowand, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar] [CrossRef]
- Chiva-Blancha, G.; Visioli, F. Polyphenols and health: Moving beyond antioxidants. J. Berry Res. 2012, 2, 63–71. [Google Scholar]
- De Martino, L.; de Feo, V.; Nazzaro, F. Chemical composition and in vitro antimicrobial and mutagenic activities of seven Lamiaceae essential oils. Molecules 2009, 14, 4213–4230. [Google Scholar] [CrossRef] [PubMed]
- De Man, J.D.; Rogosa, M.; Sharpe, M.E. A medium for the cultivation of Lactobacilli. J. Appl. Bacteriol. 1960, 23, 130–135. [Google Scholar] [CrossRef]
- De Giulio, B.; Orlando, P.; Barba, G.; Coppola, R.; de Rosa, M.; Sada, A.; de Prisco, P.P.; Nazzaro, F. Use of alginate and cryo-protective sugars to improve the viability of lactic acid bacteria after freezing and freeze-drying. World J. Microbiol. Biotechnol. 2005, 2, 739–748. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolic with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Liu, C.F.; Pan, T.M. In vitro effects of lactic acid bacteria on cancer cell viability and antioxidant activity. J. Food Drug Anal. 2010, 18, 77–86. [Google Scholar]
- Nazzaro, F.; Fratianni, F.; Orlando, P.; Coppola, R. Biochemical traits, survival and biological properties of the probiotic Lactobacillus plantarum grown in the presence of prebiotic inulin and pectin as energy source. Pharmaceuticals 2012, 5, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Nicolaus, B.; Poli, A.; Orlando, P. The prebiotic source influences the growth, biochemical features and survival under simulated gastrointestinal conditions of the probiotic Lactobacillus acidophilus. Anaerobe 2012, 18, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fratianni, F.; Pepe, S.; Cardinale, F.; Granese, T.; Cozzolino, A.; Coppola, R.; Nazzaro, F. Eruca sativa Might Influence the Growth, Survival under Simulated Gastrointestinal Conditions and Some Biological Features of Lactobacillus acidophilus, Lactobacillus plantarum and Lactobacillus rhamnosus Strains. Int. J. Mol. Sci. 2014, 15, 17790-17805. https://doi.org/10.3390/ijms151017790
Fratianni F, Pepe S, Cardinale F, Granese T, Cozzolino A, Coppola R, Nazzaro F. Eruca sativa Might Influence the Growth, Survival under Simulated Gastrointestinal Conditions and Some Biological Features of Lactobacillus acidophilus, Lactobacillus plantarum and Lactobacillus rhamnosus Strains. International Journal of Molecular Sciences. 2014; 15(10):17790-17805. https://doi.org/10.3390/ijms151017790
Chicago/Turabian StyleFratianni, Florinda, Selenia Pepe, Federica Cardinale, Tiziana Granese, Autilia Cozzolino, Raffaele Coppola, and Filomena Nazzaro. 2014. "Eruca sativa Might Influence the Growth, Survival under Simulated Gastrointestinal Conditions and Some Biological Features of Lactobacillus acidophilus, Lactobacillus plantarum and Lactobacillus rhamnosus Strains" International Journal of Molecular Sciences 15, no. 10: 17790-17805. https://doi.org/10.3390/ijms151017790
APA StyleFratianni, F., Pepe, S., Cardinale, F., Granese, T., Cozzolino, A., Coppola, R., & Nazzaro, F. (2014). Eruca sativa Might Influence the Growth, Survival under Simulated Gastrointestinal Conditions and Some Biological Features of Lactobacillus acidophilus, Lactobacillus plantarum and Lactobacillus rhamnosus Strains. International Journal of Molecular Sciences, 15(10), 17790-17805. https://doi.org/10.3390/ijms151017790






