Effective and Selective Recovery of Precious Metals by Thiourea Modified Magnetic Nanoparticles
Abstract
:1. Introduction
2. Results and Discussion
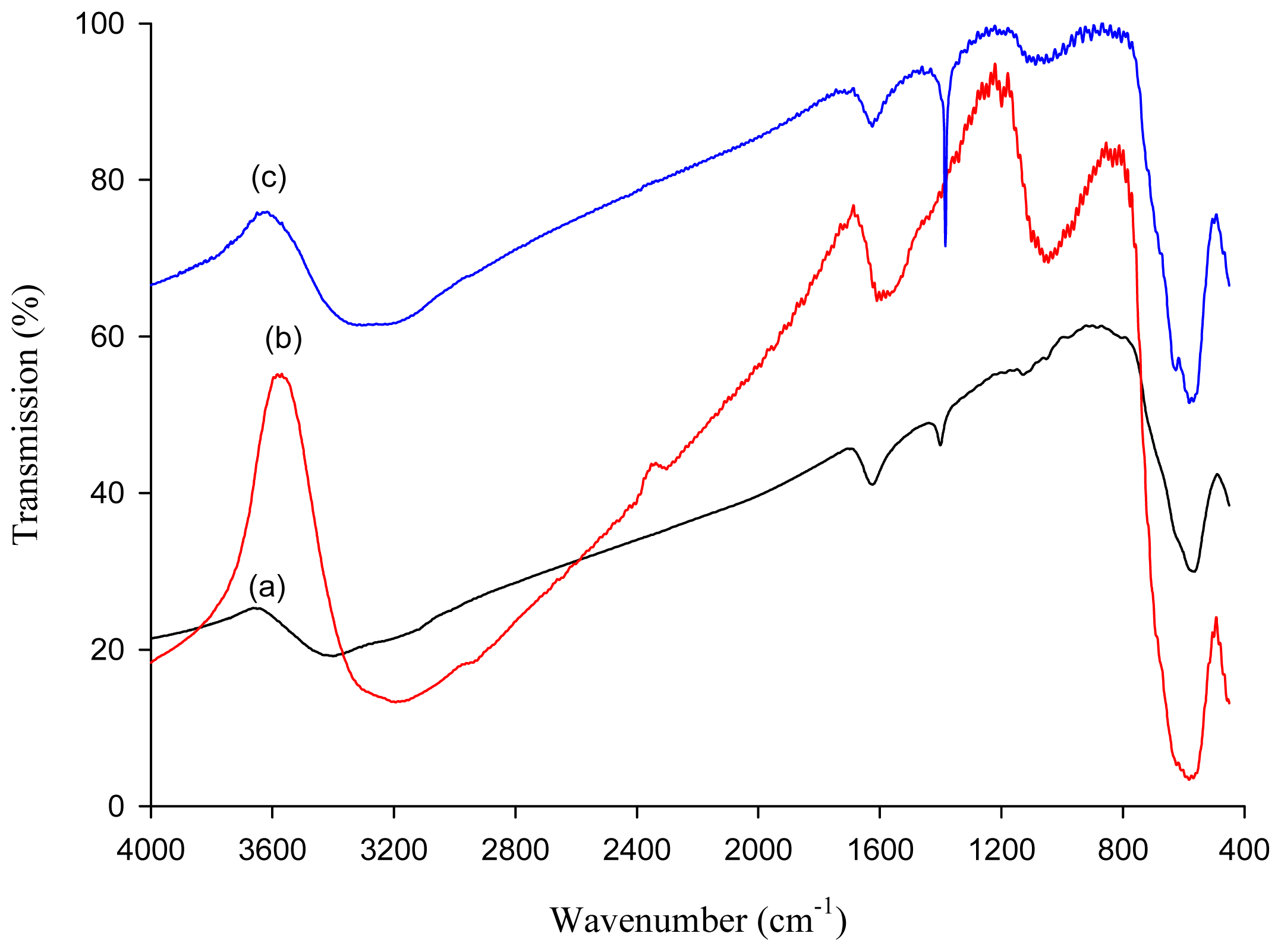
2.1. Characterization of Thiourea Modified Magnetic Nanoparticles (MNP-Tu)
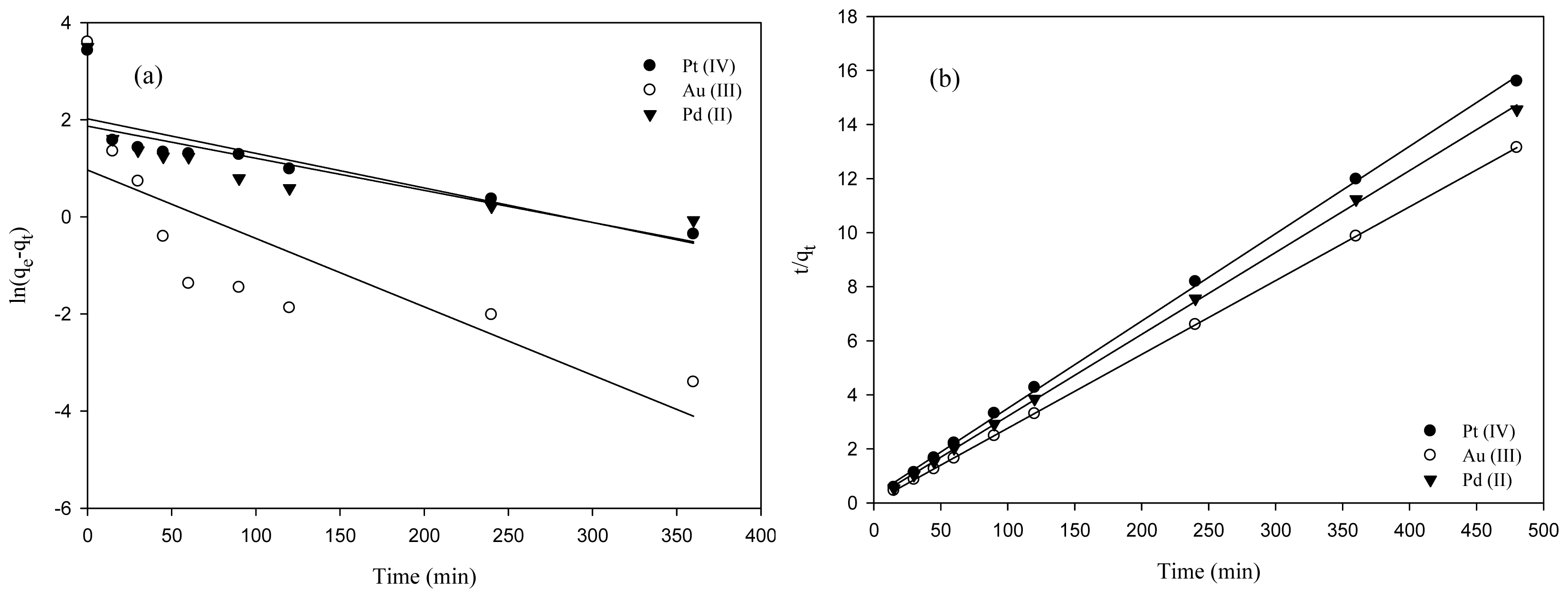
2.2. Adsorption Kinetics
2.3. Adsorption Isotherms
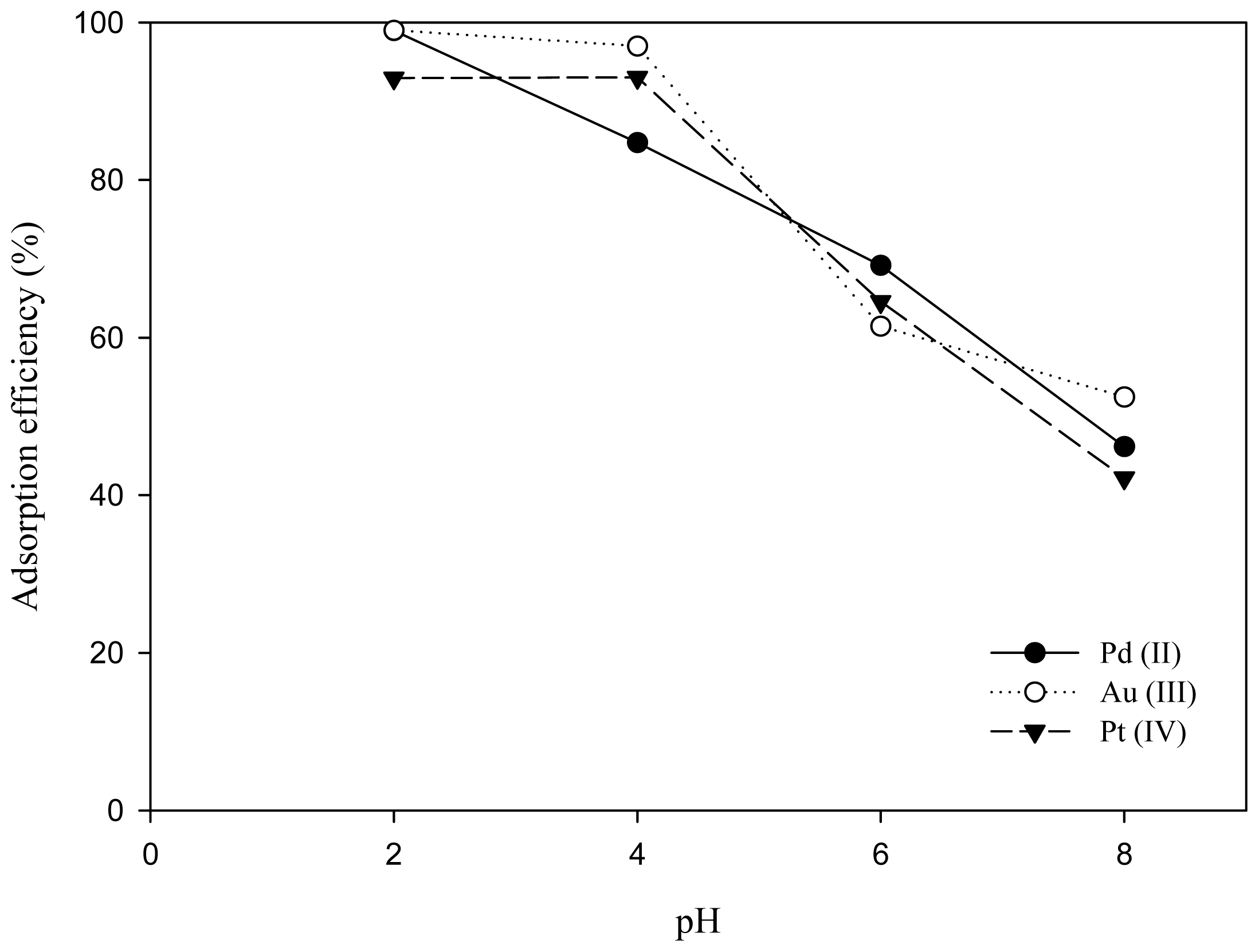
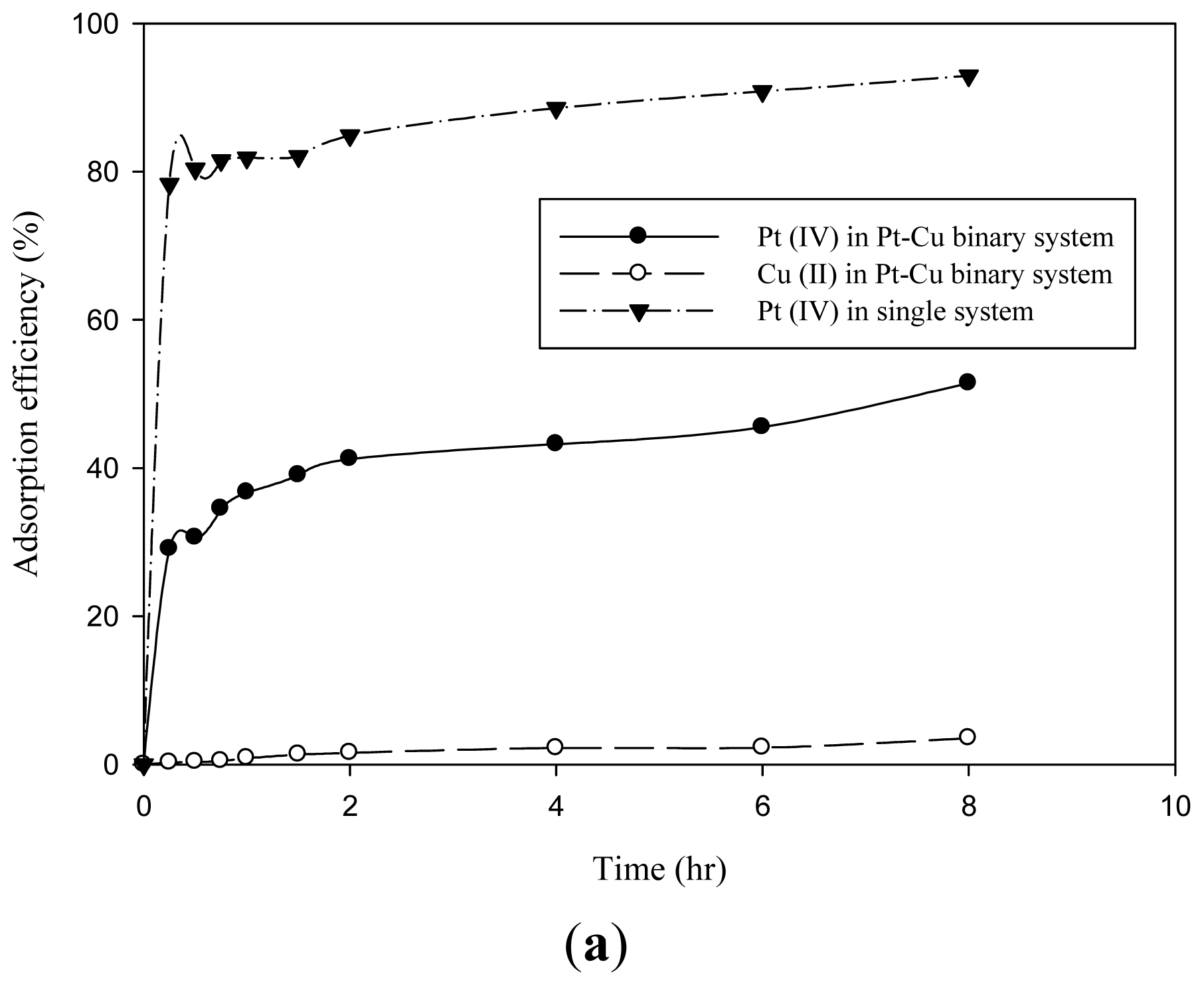
2.4. Competitive Adsorption
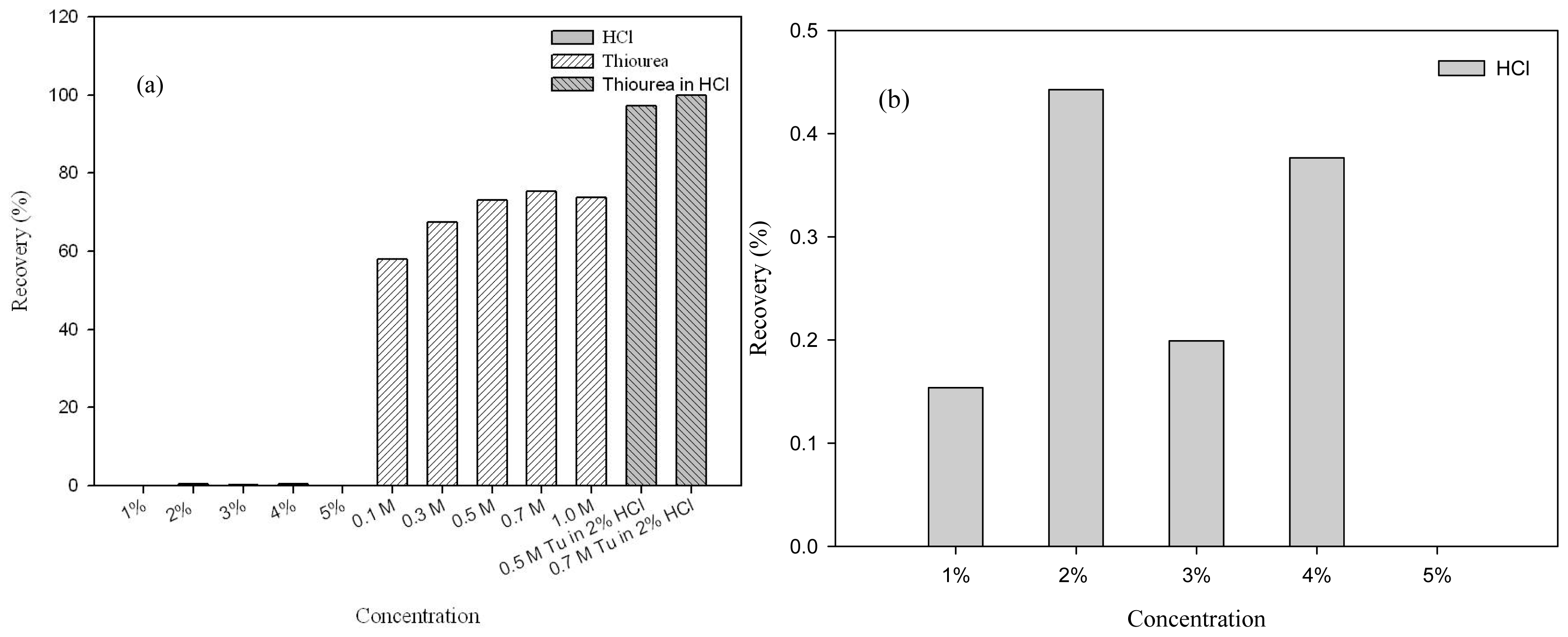
2.5. Desorption and Reusability
3. Experimental Section
3.1. Materials
3.2. Preparation of Magnetic Nanoparticles (MNPs)
3.3. Synthesis of Thiourea Modified Magnetic Nanoparticles (MNP-Tu)
3.4. Batch Tests
3.5. Metallic Ion Analysis
3.6. Characterization
4. Conclusions
Acknowledgements
References
- Widmer, R.; Oswald-Krapf, H.; Sinha-Khetriwal, D.; Schnellmann, M.; Böni, H. Global perspectives on e-waste. Environ. Impact Asses. Rev 2005, 25, 436–458. [Google Scholar]
- Cui, J.; Zhang, L. Metallurgical recovery of metals from electronic waste: A review. J. Hazard. Mater 2008, 158, 228–256. [Google Scholar]
- Chen, X.; Lam, K.F.; Mak, S.F.; Yeung, K.L. Precious metal recovery by selective adsorption using biosorbents. J. Hazard. Mater 2011, 186, 902–910. [Google Scholar]
- Xiong, Y.; Adhikari, C.R.; Kawakita, H.; Ohto, K.; Inoue, K.; Harada, H. Selective recovery of precious metals by persimmon waste chemically modified with dimethylamine. Bioresour. Technol 2009, 100, 4083–4089. [Google Scholar]
- Sun, C.; Zhang, G.; Wang, C.; Qu, Q.; Zhang, Y.; Gu, Q. A resin with high adsorption selectivity for Au(III): Preparation, characterization and adsorption properties. Chem. Eng. J 2011, 172, 713–720. [Google Scholar]
- Ertan, E.; Gulfen, M. Separation of gold(III) ions from copper(II) and zinc(II) ions using thiourea-formaldehyde or urea-formaldehyde chelating resins. J. Appl. Polym. Sci 2009, 111, 2798–2805. [Google Scholar]
- Parajuli, D.; Kawakita, H.; Inoue, K.; Funaoka, M. Recovery of gold(III), palladium(II), and platinum(IV) by aminated lignin derivatives. Ind. Eng. Chem. Res 2006, 45, 6405–6412. [Google Scholar]
- Ramesh, A.; Hasegawa, H.; Sugimoto, W.; Maki, T.; Ueda, K. Adsorption of gold(III), platinum(IV) and palladium(II) onto glycine modified crosslinked chitosan resin. Bioresour. Technol 2008, 99, 3801–3809. [Google Scholar]
- Fujiwara, K.; Ramesh, A.; Maki, T.; Hasegawa, H.; Ueda, K. Adsorption of platinum (IV), palladium (II) and gold (III) from aqueous solutions onto l-lysine modified crosslinked chitosan resin. J. Hazard. Mater 2007, 146, 39–50. [Google Scholar]
- Uheida, A.; Iglesias, M.; Fontas, C.; Hidalgo, M.; Salvado, V.; Zhang, Y.; Muhammed, M. Sorption of palladium(II), rhodium(III), and platinum(IV) on Fe3O4 nanoparticles. J. Colloid Interf. Sci 2006, 301, 402–408. [Google Scholar]
- Chou, C.-M.; Lien, H.-L. Dendrimer-conjugated magnetic nanoparticles for removal of zinc(II) from aqueous solutions. J. Nanopart. Res 2011, 13, 2099–2107. [Google Scholar]
- Lo, I.M.C.; Hu, J.; Chen, G. Iron-Based Magnetic Nanoparticles for Removal of Heavy Metals from Electroplating and Metal-Finishing Wastewater. In Nanotechnologies for Water Environment Applications, 1st ed; Zhang, T.C., Surampalli, R.Y., Lai, K.C.K., Hu, Z., Tyagi, R.D., Lo, I.M.C., Eds.; American Society of Civil Engineers (ASCE): Reston, VA, USA, 2009; pp. 213–268. [Google Scholar]
- Hattori, S.; Watanabe, M.; Sasaki, K.; Yasuharu, H. Magnetization of activated sludge by an external magnetic field. Biotechnol. Lett 2002, 24, 65–69. [Google Scholar]
- Zhou, L.; Xu, J.; Liang, X.; Liu, Z. Adsorption of platinum(IV) and palladium(II) from aqueous solution by magnetic cross-linking chitosan nanoparticles modified with ethylenediamine. J. Hazard. Mater 2010, 182, 518–524. [Google Scholar]
- Ngomsik, A.; Bee, A.; Draye, M.; Cote, G.; Cabuil, V. Magnetic nano- and microparticles for metal removal and environmental aapplications: A review. C. R. Chim 2005, 8, 963–970. [Google Scholar]
- Pearson, R.G. Hard and soft acids, HSAB. PartI. Fundamental principles. J. Chem. Educ 1968, 45, 581–587. [Google Scholar]
- Zhou, L.; Liu, J.; Liu, Z. Adsorption of platinum(IV) and palladium(II) from aaqueous solution by thiourea-modified chitosan microspheres. J. Hazard. Mater 2009, 172, 439–446. [Google Scholar]
- Wang, L.; Xing, R.; Liu, S.; Yu, H.; Qin, Y.; Li, K.; Feng, J.; Li, R.; Li, P. Recovery of silver (I) using a thiourea-modified chitosan resin. J. Hazard. Mater 2010, 180, 577–582. [Google Scholar]
- Birinci, E.; Gülfen, M.; Aydın, A.O. Separation and recovery of palladium(II) from base metal ions by melamine-formaldehyde-thiourea (MFT) chelating resin. Hydrometallurgy 2009, 95, 15–21. [Google Scholar]
- Lam, K.F.; Fong, C.M.; Yeung, K.L. Separation of precious metals using selective mesoporous adsorbents. Gold Bull 2007, 40, 192–198. [Google Scholar]
- Lam, K.F.; Yeung, K.L.; McKay, G. An investigation of gold adsorption from a binary mixture with selective mesoporous silica adsorbents. J. Phys. Chem. B 2006, 110, 2187–2194. [Google Scholar]
- Lam, K.F.; Fong, C.M.; Yeung, K.L.; Mckay, G. Selective adsorption of gold from complex mixtures using mesoporous adsorbents. Chem. Eng. J 2008, 145, 185–195. [Google Scholar]
- Andrzejewska, A.; Krysztafkiewicz, A.; Jesionowski, T. Adsorption of organic ddyes on the aminosilane modified TiO2 surface. Dye. Pigment 2004, 62, 121–130. [Google Scholar]
- Ye, L.; Pelton, R.; Brook, M.A. Biotinylation of TiO2 nanoparticles and their conjugation with sstreptavidin. Langmuir 2007, 23, 5630–5637. [Google Scholar]
- Pan, B.F.; Gao, F.; Gu, H.C. Dendrimer modified magnetic nanoparticles for protein immobilization. J. Colloid Interface Sci 2005, 284, 1–5. [Google Scholar]
- El-Bahy, G.M.S.; El-Sayed, B.A.; Shabana, A.A. Vibrational and electronic studies on some metal thiourea complexes. Vib. Spectrosc 2003, 31, 101–107. [Google Scholar]
- Groenewald, T. Potential application of thiourea in the processing of gold. J. S. Afr. Inst. Min. Metall 1977, 77, 217–223. [Google Scholar]
- Crane, R.A.; Dickinson, M.; Popescu, I.C.; Scott, T.B. Magnetite and zero-valent iron nanoparticles for the remediation of uranium contaminated environmental water. Water Res 2011, 45, 2931–1942. [Google Scholar]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem 1999, 34, 451–465. [Google Scholar]
- Jainae, K.; Sanuwong, K.; Nuangjamnong, J.; Sukpirom, N.; Unob, F. Extraction and recovery of precious metal ions in wastewater by polystyrene-coated magnetic particles functionalized with 2-(3-(2-aminoethylthio)propylthio)ethanamine. Chem. Eng. J 2010, 160, 586–593. [Google Scholar]
- Qu, R.; Sun, C.; Wang, M.; Ji, C.; Xu, Q.; Zhang, Y.; Wang, C.; Chen, H.; Yin, P. Adsorption of Au(III) from aqueous solution using cotton fiber/chitosan composite adsorbents. Hydrometallurgy 2009, 100, 65–71. [Google Scholar]
- Mehta, R.V.; Upadhyay, R.V.; Charles, S.W.; Ramchand, C.N. Direct binding of protein to magnetic particles. Biotechnol. Tech 1997, 77, 493–496. [Google Scholar]

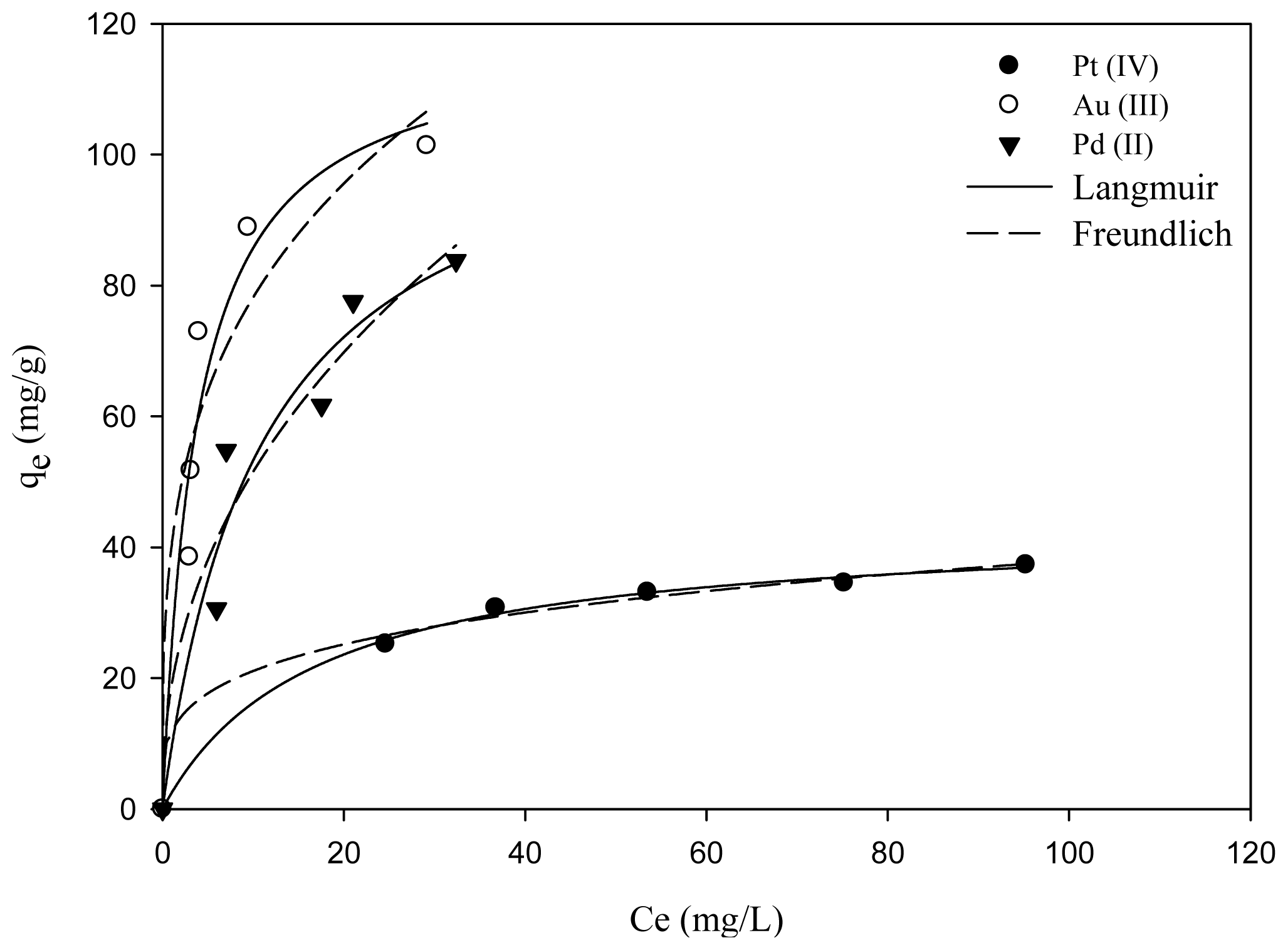



| Langmuir model | Freundlich model | |||||
|---|---|---|---|---|---|---|
| qmax (mg/g) | KL (L/mg) | R2 | Kf | n | R2 | |
| Pt(IV) | 43.34 | 0.06 | 0.9975 | 11.73 | 3.92 | 0.995 |
| Au(III) | 118.46 | 0.26 | 0.946 | 40.13 | 3.45 | 0.9076 |
| Pd(II) | 111.58 | 0.09 | 0.9457 | 18.83 | 2.29 | 0.9422 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, T.-L.; Lien, H.-L. Effective and Selective Recovery of Precious Metals by Thiourea Modified Magnetic Nanoparticles. Int. J. Mol. Sci. 2013, 14, 9834-9847. https://doi.org/10.3390/ijms14059834
Lin T-L, Lien H-L. Effective and Selective Recovery of Precious Metals by Thiourea Modified Magnetic Nanoparticles. International Journal of Molecular Sciences. 2013; 14(5):9834-9847. https://doi.org/10.3390/ijms14059834
Chicago/Turabian StyleLin, Tai-Lin, and Hsing-Lung Lien. 2013. "Effective and Selective Recovery of Precious Metals by Thiourea Modified Magnetic Nanoparticles" International Journal of Molecular Sciences 14, no. 5: 9834-9847. https://doi.org/10.3390/ijms14059834
APA StyleLin, T.-L., & Lien, H.-L. (2013). Effective and Selective Recovery of Precious Metals by Thiourea Modified Magnetic Nanoparticles. International Journal of Molecular Sciences, 14(5), 9834-9847. https://doi.org/10.3390/ijms14059834




