Active Silver Nanoparticles for Wound Healing
Abstract
:1. Introduction
2. Results and Discussion
2.1. In Vitro Study
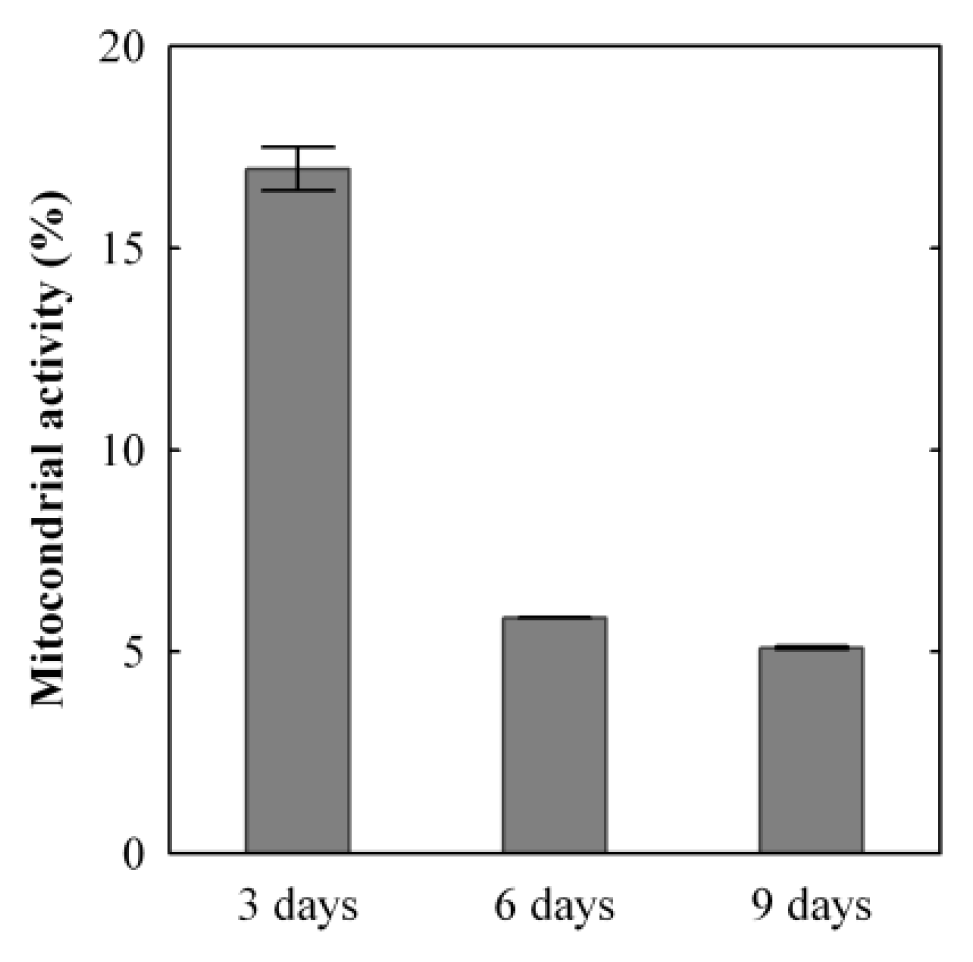
2.1.1. MTT Assay
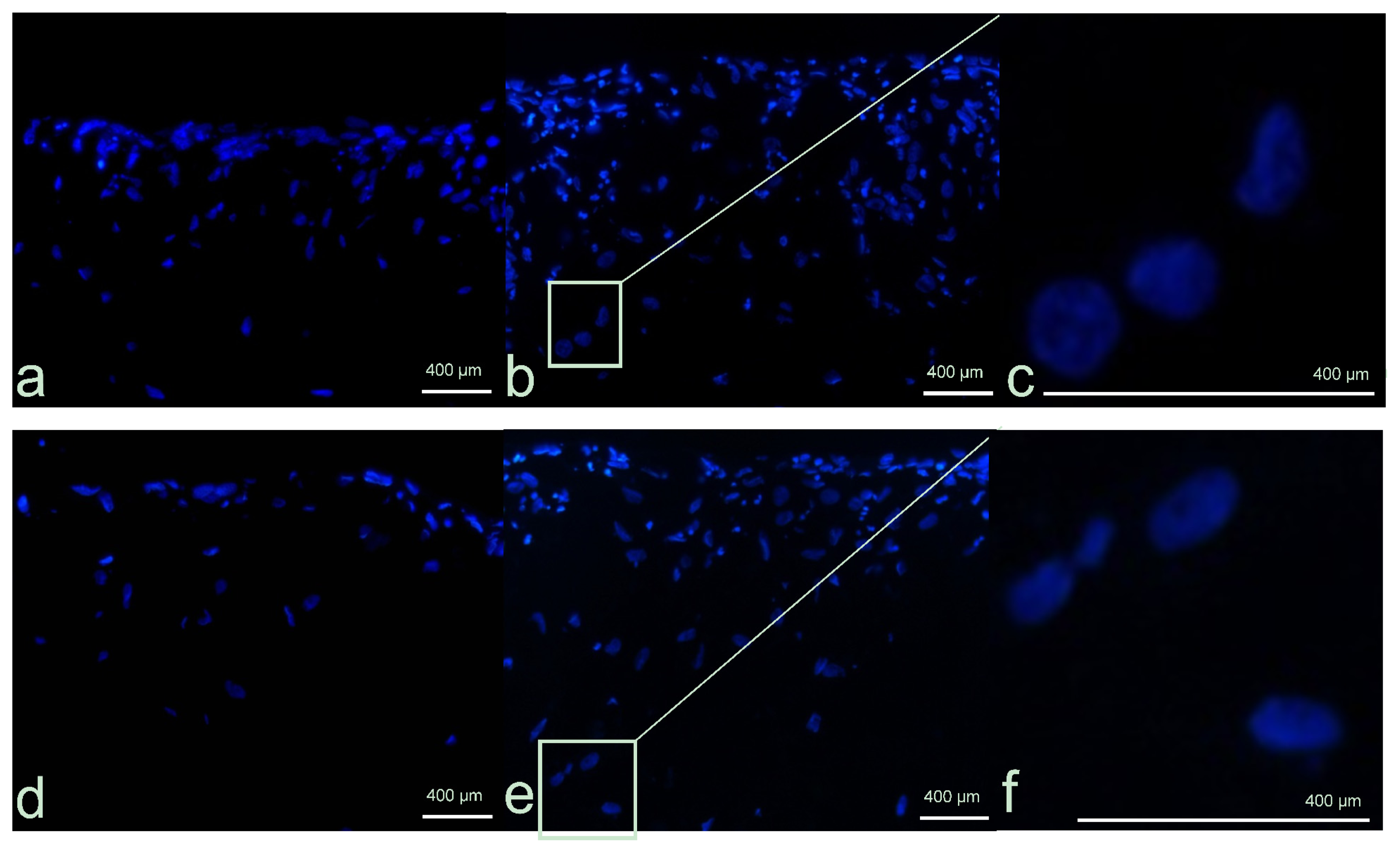
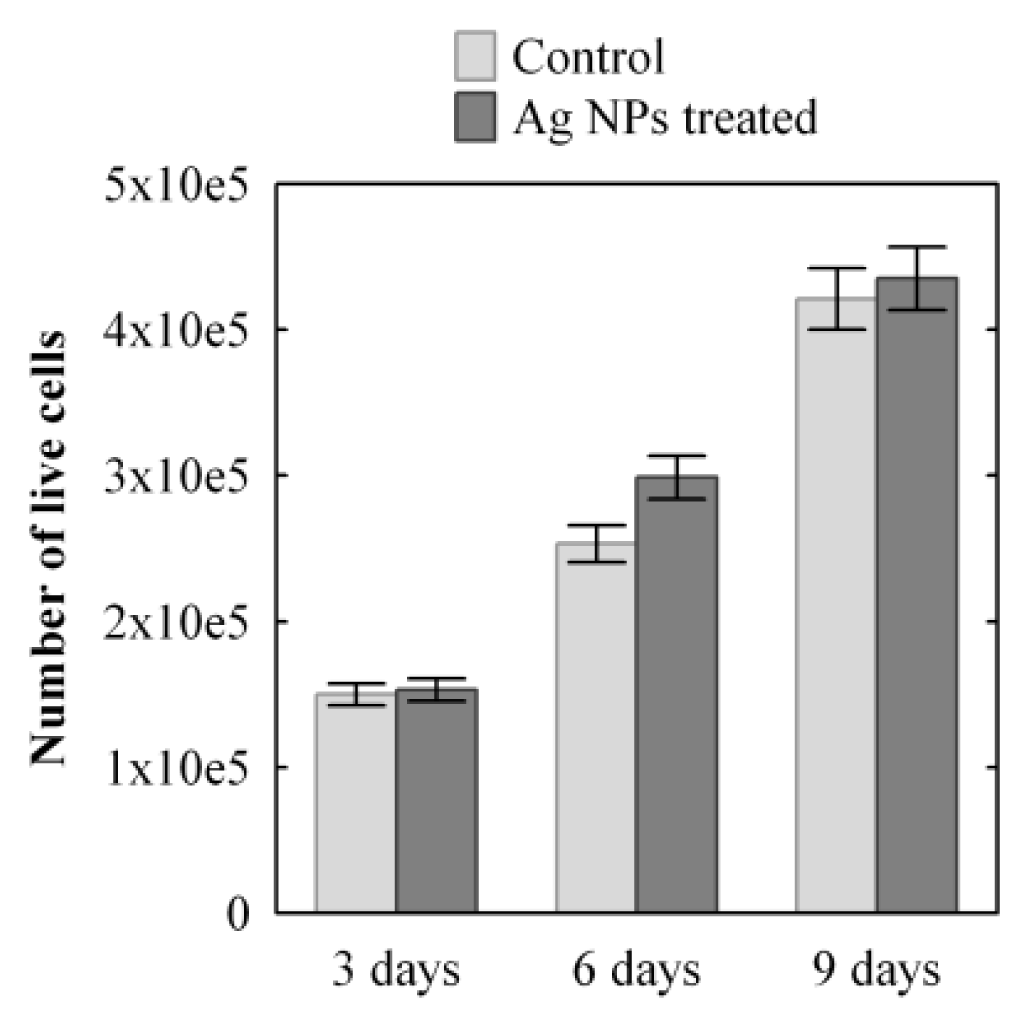
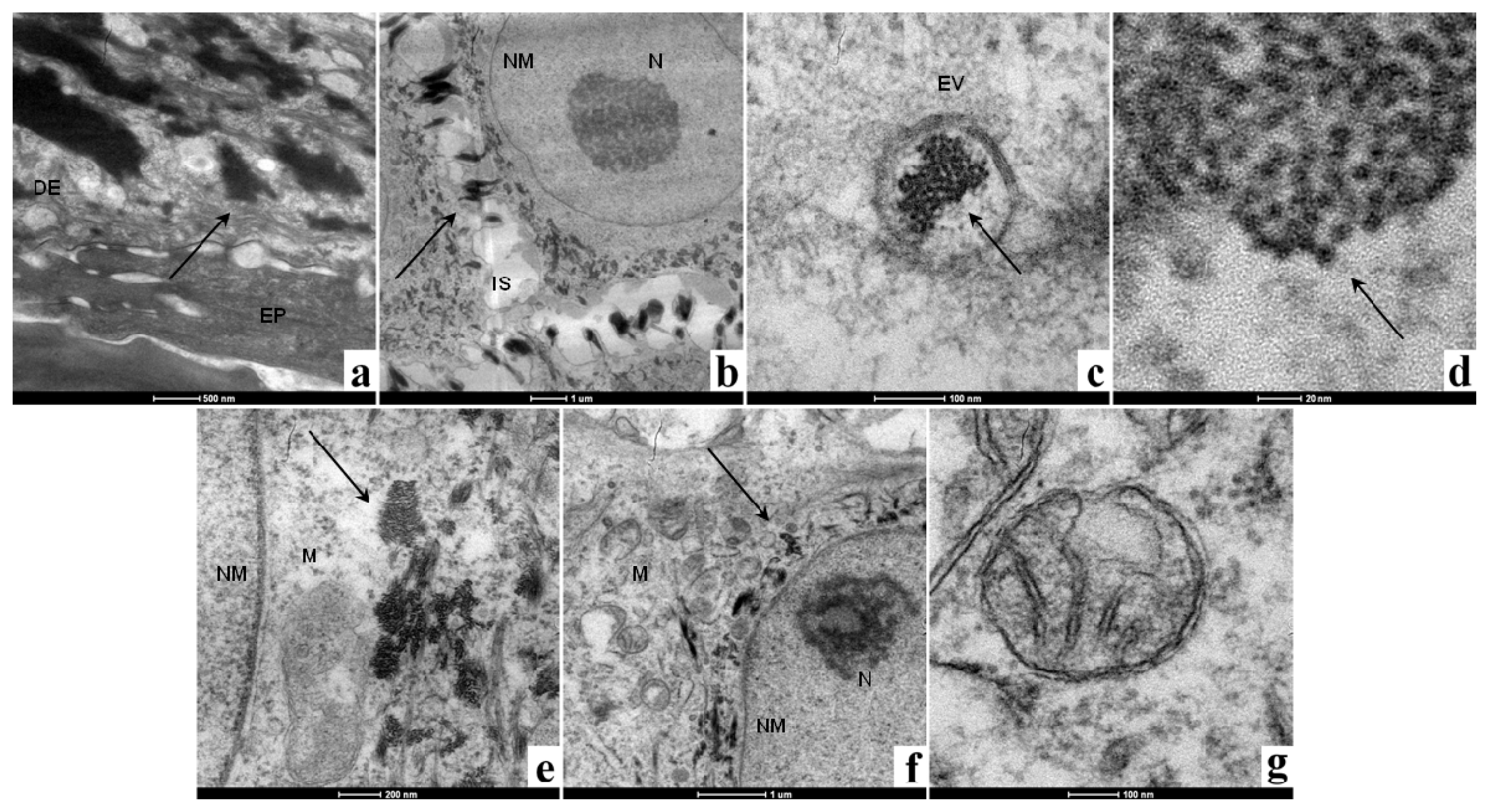
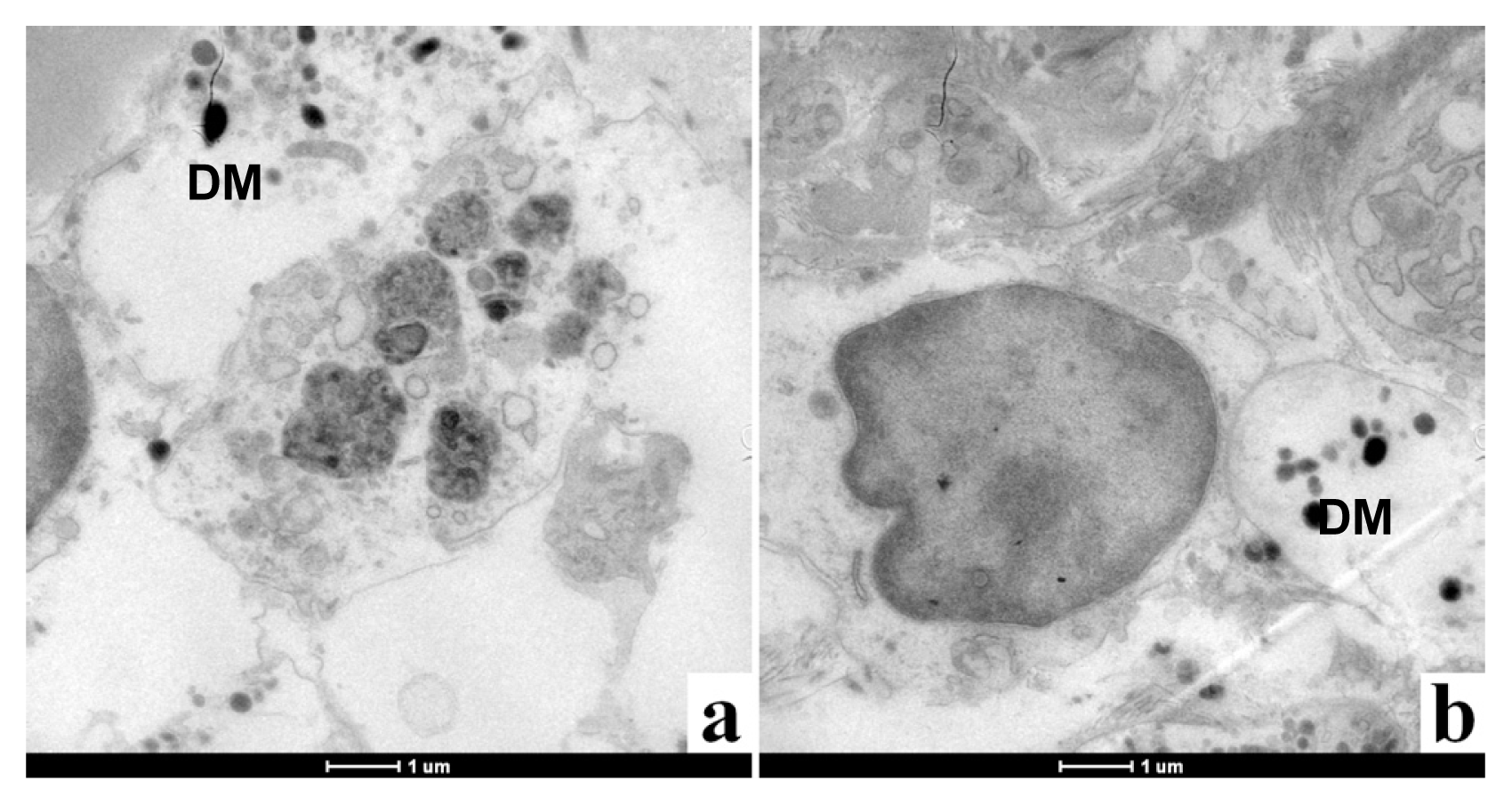
2.1.2. Morphological Analysis
2.1.3. Ag Release and Distribution
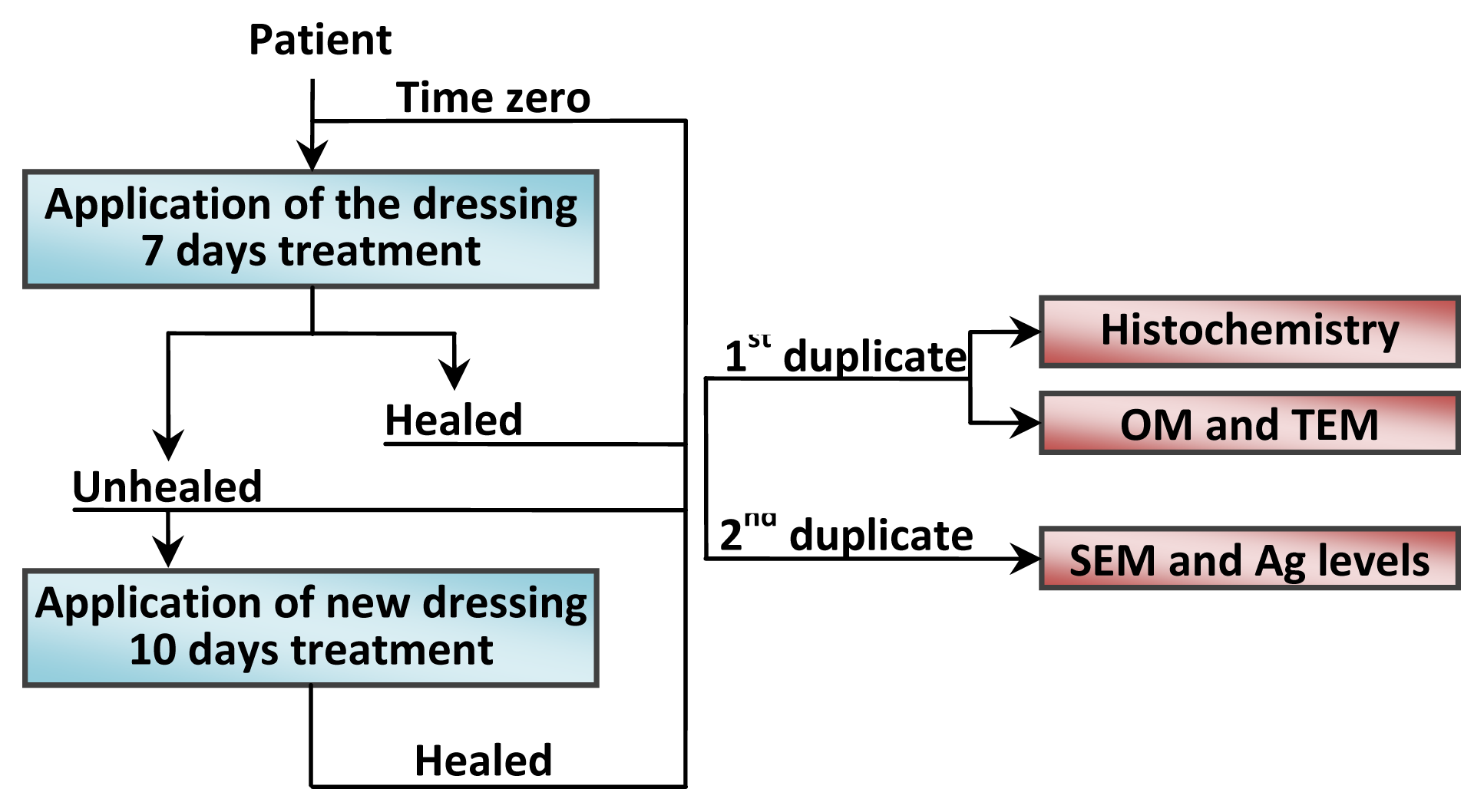
2.2. In Vivo Study
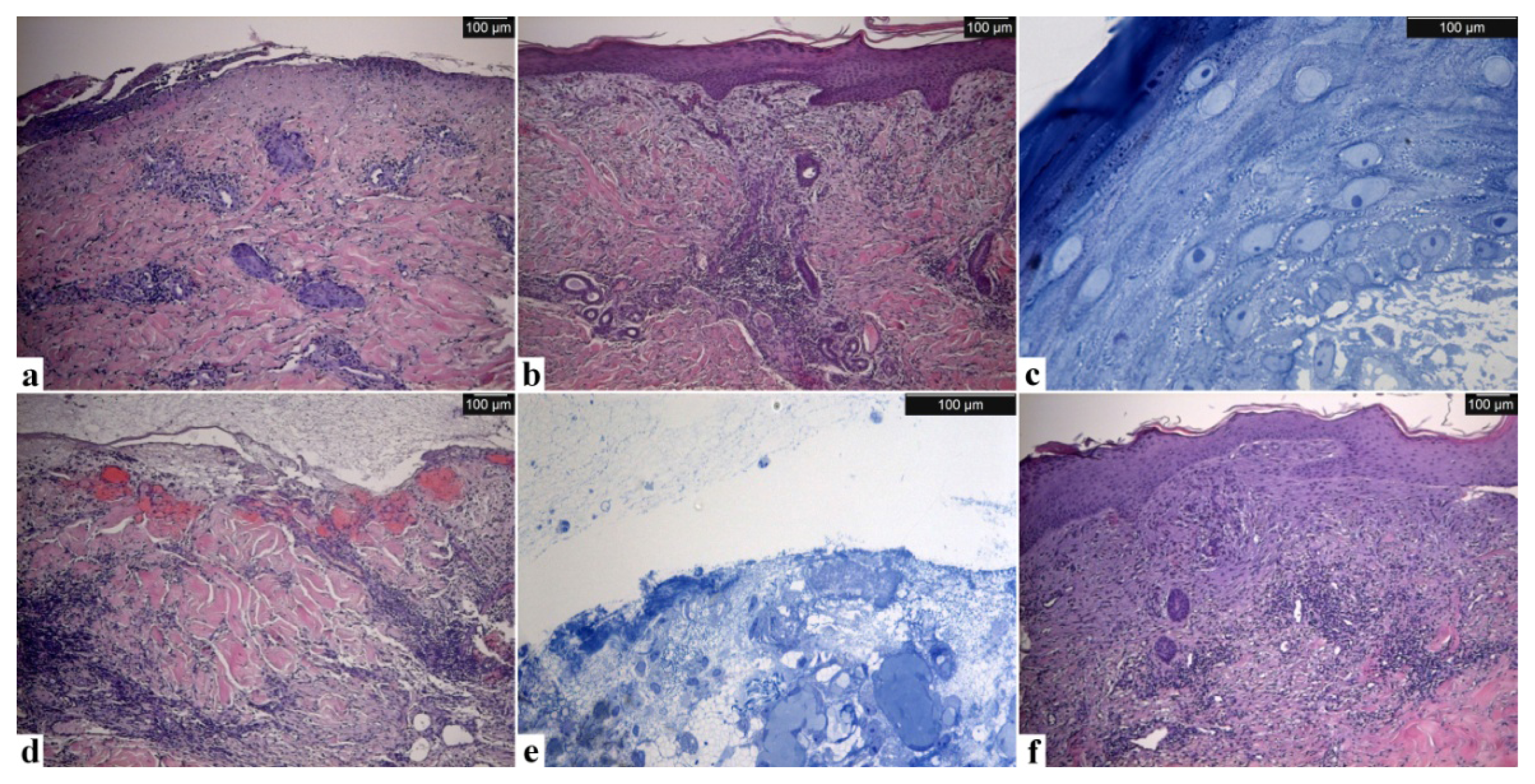
2.2.1. Microscopy
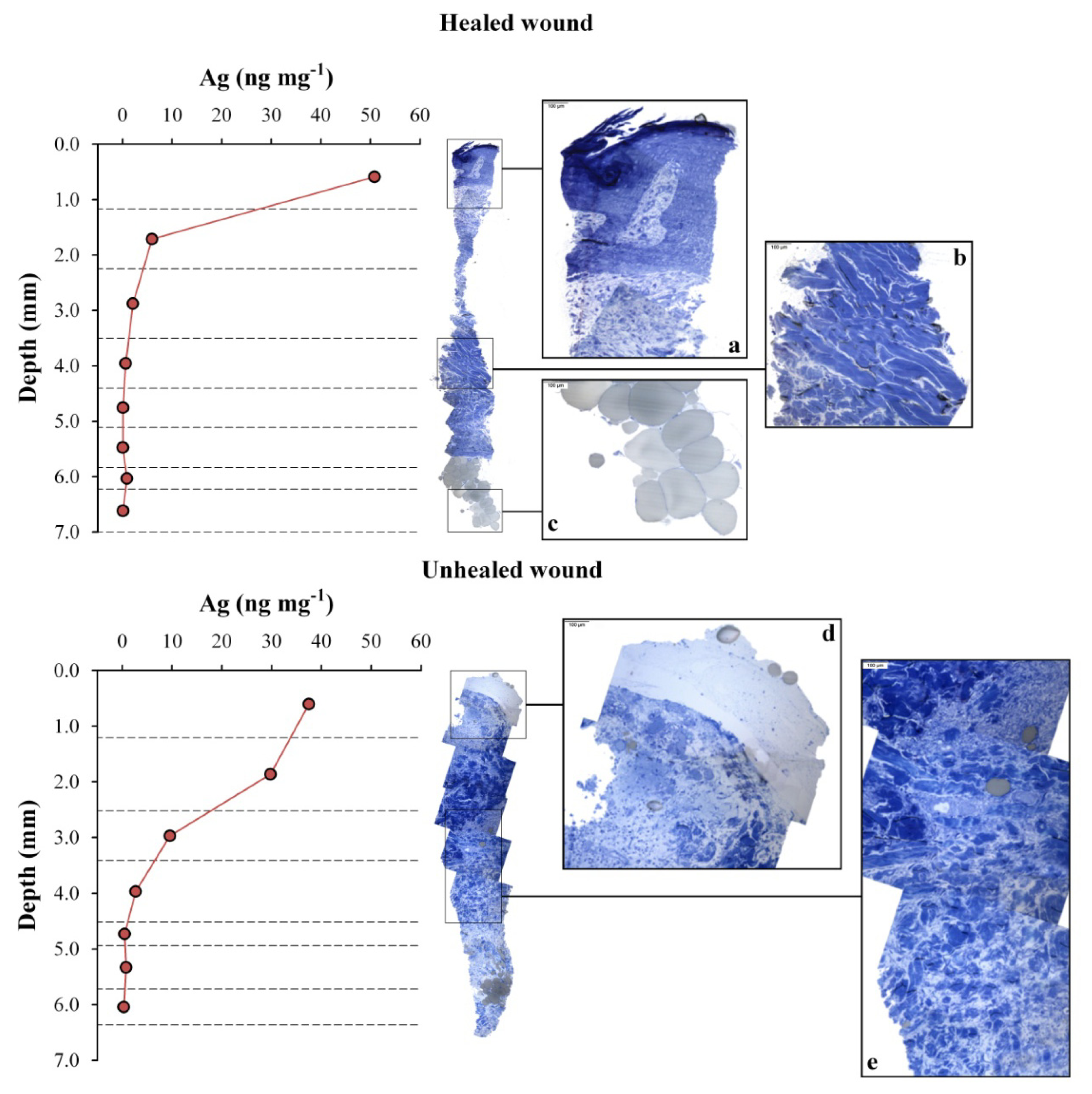
2.2.2. Ag Release and Depth Profiles
2.2.3. Clinical Observations
2.2.4. Limits of This Study and Future Perspectives
3 Material and Methods
3.1. Dressing
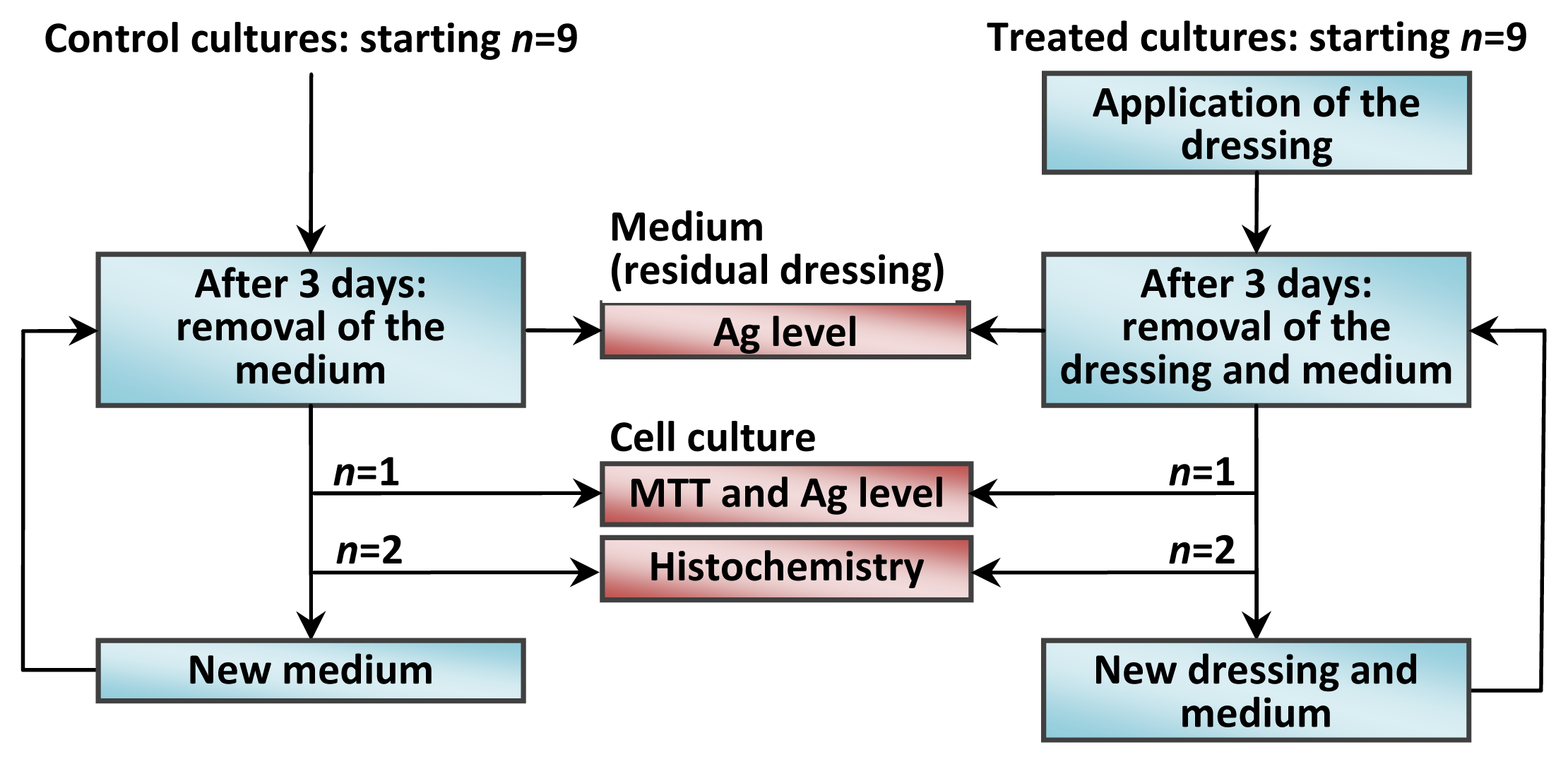
3.2. In Vitro Study
3.2.1. Cell Cultures
3.2.2. MTT Assay
3.2.3. Morphological Analysis
3.3. In Vivo Study
3.3.1. Human Skin Samples
3.3.2. Optical Microscopy, TEM and ESEM
3.4. Determination of Ag Levels by ICP-MS
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Klasen, H.J. Historical review of the use of silver in the treatment of burns. I. Early uses. Burns 2000, 26, 117–130. [Google Scholar]
- Klasen, H.J. A historical review of the use of silver in the treatment of burns. II. Renewed interest for silver. Burns 2000, 26, 131–138. [Google Scholar]
- Hussain, S.; Ferguson, C. Best evidence topic report. Silver sulphadiazine cream in burns. Emerg. Med. J 2006, 23, 929–932. [Google Scholar]
- Castellano, J.J.; Shafii, S.M.; Ko, F.; Donate, G.; Wright, T.E.; Mannari, R.J.; Payne, W.G.; Smith, D.J.; Robson, M.C. Comparative evaluation of silver-containing antimicrobial dressings and drugs. Int. Wound. J 2007, 4, 114–122. [Google Scholar]
- Ip, M.; Lui, S.L.; Poon, V.K.M.; Lung, I.; Burd, A. Antimicrobial activities of silver dressings: An in vitro comparison. J. Med. Microbiol 2006, 55, 59–63. [Google Scholar]
- Melayie, A.; Youngs, J.W. Silver and its application on antimicrobial agents. Expert. Opin. Ther. Pat 2005, 15, 125–130. [Google Scholar]
- Silvestry-Rodriguez, N.; Sicairos-Ruelas, E.E.; Gerba, C.P.; Bright, K.R. Silver as disinfectant. Rev. Environ. Contam. T 2007, 191, 23–45. [Google Scholar]
- Atiyeh, B.S.; Costagliola, M.; Hayek, S.N.; Dibo, S.A. Effect of silver on burn wound infection control and healing: Review of the literature. Burns 2007, 33, 139–148. [Google Scholar]
- Neal, A.L. What can be inferred from bacterium-nanoparticle interactions about the potential consequences of environmental exposure to nanoparticles? Ecotoxicology 2008, 17, 362–371. [Google Scholar]
- Okan, D.; Woo, K.; Sibbald, R.G. So what if you are blue? Oral colloidal silver and argyria are out: safe dressings are in. Adv. Skin. Wound. Care 2007, 20, 326–330. [Google Scholar]
- Cutting, K.; White, R.; Edmonds, M. The safety and efficacy of dressings with silver—Addressing clinical concerns. Int. Wound. J 2007, 4, 177–184. [Google Scholar]
- Burd, A.; Kwok, C.H.; Hung, S.C.; Chan, H.S.; Gu, H.; Lam, W.K.; Huang, L. A comparative study of the cytotoxicity of silver-based dressings in monolayer cell, tissue explant, and animal models. Wound Repair. Regen 2007, 15, 94–104. [Google Scholar]
- le Duc, Q.; Breetveld, M.; Middelkoop, E.; Scheper, R.J.; Ulrich, M.M.W.; Gibbs, S. A cytotoxic analysis of antiseptic medication on skin substitutes and autograft. Brit. J. Dermatol 2007, 157, 33–40. [Google Scholar]
- Fong, J.; Wood, F. Nanocrystalline silver dressings in wound management: A review. Int. J. Nanomed 2006, 1, 441–449. [Google Scholar]
- Nair, L.S.; Laurencin, C.T. Silver nanoparticles: Synthesis and therapeutic applications. J. Biomed. Nanotechnol 2007, 3, 301–316. [Google Scholar]
- Galdiero, S.; Falanga, A.; Vitiello, M.; Cantisani, M.; Marra, V.; Galdiero, M. Silver nanoparticles as potential antiviral agents. Molecules 2011, 16, 8894–8918. [Google Scholar]
- Rai, M.K.; Deshmukh, S.D.; Ingle, A.P.; Gade, A.K. Silver nanoparticles: The powerful nanoweapon against multidrug-resistant bacteria. J. Appl. Microbiol 2012, 112, 841–852. [Google Scholar]
- Chaloupka, K.; Malam, Y.; Seifalian, A.M. Nanosilver as a new generation of nanoproduct in biomedical applications. Trends Biotechnol 2010, 28, 580–588. [Google Scholar]
- Wilkinson, L.J.; White, R.J.; Chipman, J.K. Silver and nanoparticles of silver in wound dressings: A review of efficacy and safety. J. Wound Care 2011, 20, 543–549. [Google Scholar]
- Khundkar, R.; Malic, C.; Burge, T. Use of ActicoatTM dressing in burns: What is the evidence? Burns 2010, 36, 751–758. [Google Scholar]
- Wang, X.Q.; Kravchuk, O.; Kimble, R.M. A retrospective review of burn dressings on a porcine burn model. Burns 2010, 36, 680–687. [Google Scholar]
- Vlachou, E.; Chipp, E.; Shale, E.; Wilson, Y.T.; Papini, R.; Moiemen, N.S. The safety of nanocrystalline silver dressings on burns: A study of systemic silver absorption. Burns 2007, 33, 979–985. [Google Scholar]
- Moiemen, N.S.; Shale, E.; Drysdale, K.J.; Smith, G.; Wilson, Y.T.; Papini, R. Acticoat dressings and major burns: Systemic silver absorption. Burns 2011, 37, 27–35. [Google Scholar]
- Trop, M.; Novak, M.; Rodl, S.; Hellbom, B.; Kroell, W.; Goessler, W. Silver-coated dressing acticoat caused raised liver enzymes and argyria-like symptoms in burn patient. J. Trauma 2006, 60, 648–652. [Google Scholar]
- Poon, V.K.M.; Burd, A. In vitro cytotoxity of silver: Implication for clinical wound care. Burns 2004, 30, 140–147. [Google Scholar]
- Zanette, C.; Pelin, M.; Crosera, M.; Adami, G.; Bovenzi, M.; Larese, F.F.; Florio, C. Silver nanoparticles exert a long-lasting antiproliferative effect on human keratinocyte HaCaT cell line. Toxicol. In Vitro 2011, 25, 1053–1060. [Google Scholar]
- Foldbjerg, R.; Dang, D.A.; Autrup, H. Cytotoxicity and genotoxicity of silver nanoparticles in the human lung cancer cell line, A549. Arch. Toxicol 2011, 85, 743–750. [Google Scholar]
- AshaRani, P.V.; Low Kah Mun, G.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano 2009, 3, 279–290. [Google Scholar]
- Foldbjerg, R.; Olesen, P.; Hougaard, M.; Dang, D.A.; Hoffmann, H.J.; Autrup, H. PVP-coated silver nanoparticles and silver ions induce reactive oxygen species, apoptosis and necrosis in THP-1 monocytes. Toxicol. Lett 2009, 190, 156–162. [Google Scholar]
- Hsin, Y.H.; Chena, C.F.; Huang, S.; Shih, T.S.; Lai, P.S.; Chueh, P.J. The apoptotic effect of nanosilver is mediated by a ROS- and JNK-dependent mechanism involving the mitochondrial pathway in NIH3T3 cells. Toxicol. Lett 2008, 179, 130–139. [Google Scholar]
- Carlson, C.; Hussain, S.M.; Schrand, A.M.; Braydich-Stolle, L.K.; Hess, K.L.; Jones, R.L.; Schlager, J.J. Unique cellular interaction of silver nanoparticles: Size-dependent generation of reactive oxygen species. J. Phys. Chem. B 2008, 112, 13608–13619. [Google Scholar]
- Hackenberg, S.; Scherzed, A.; Kessler, M.; Hummel, S.; Technau, A.; Froelich, K.; Ginzkey, C.; Koehler, C.; Hagen, R.; Kleinsasser, N. Silver nanoparticles: Evaluation of DNA damage, toxicity and functional impairment in human mesenchymal stem cells. Toxicol. Lett 2011, 201, 27–33. [Google Scholar]
- Labouta, H.I.; Schneider, M. Interaction of inorganic nanoparticles with the skin barrier: Current status and critical review. Nanomed. Nanotechnol 2013, 9, 39–54. [Google Scholar]
- Dunn, K.; Edwards-Jones, V. The role of Acticoat™ with nanocrystalline silver in the management of burns. Burns 2004, 30, S1–S9. [Google Scholar]
- Nadworny, P.L.; Landry, B.K.; Wang, J.; Tredget, E.E.; Burrell, R.E. Does nanocrystalline silver have a transferable effect? Wound Repair Regen 2010, 18, 254–265. [Google Scholar]
- Burrell, R.E.; Morris, L.R. Antimicrobial Coating for Medical Device. U.S. Patent 5,753,251, 19 May 1998. [Google Scholar]
- Kempf, M.; Kimble, R.M.; Cuttle, L. Cytotoxicity testing of burn wound dressings, ointments and creams: A method using polycarbonate cell culture inserts on a cell culture system. Burns 2011, 37, 994–1000. [Google Scholar]
- Du Toit, D.F.; Page, B.J. An in vitro evaluation of the cell toxicity of honey and silver dressings. J. Wound Care 2009, 18, 383–389. [Google Scholar]
- Uygur, F.; Oncül, O.; Evinç, R.; Diktas, H.; Acar, A.; Ulkür, E. Effects of three different topical antibacterial dressings on Acinetobacter baumannii-contaminated full-thickness burns in rats. Burns 2009, 35, 270–273. [Google Scholar]
- Wright, J.B.; Lam, K.; Buret, A.G.; Olson, M.E.; Burrell, R.E. Early healing events in a porcine model of contaminated wounds: Effects of nanocrystalline silver on matrix metalloproteinases, cell apoptosis, and healing. Wound Repair Regen 2002, 10, 141–151. [Google Scholar]
- Huang, Y.; Li, X.; Liao, Z.; Zhang, G.; Liu, Q.; Tang, J.; Peng, Y.; Liu, X.; Luo, Q. A randomized comparative trial between Acticoat and SD-Ag in the treatment of residual burn wounds, including safety analysis. Burns 2007, 33, 161–166. [Google Scholar]
- Tonello, C.; Zavan, B.; Cortivo, R.; Brun, P.; Panfilo, S.; Abatangelo, G. In vitro reconstruction of human dermal equivalent enriched with endothelial cells. Biomaterials 2003, 24, 1205–1211. [Google Scholar]
- Tonello, C.; Vindigni, V.; Zavan, B.; Abatangelo, S.; Abatangelo, G.; Brun, P.; Cortivo, R. In vitro reconstruction of an endothelialized skin substitute provided with a microcapillary network using biopolymer scaffolds. FASEB J 2005, 19, 1546–1548. [Google Scholar]
- Rigo, C.; Roman, M.; Munivrana, I.; Vindigni, V.; Azzena, B.; Barbante, C.; Cairns, W.R.L. Characterization and evaluation of silver release from four different dressings used in burns care. Burns 2012, 38, 1131–1142. [Google Scholar]
- Thaler, A.; Heidari, N.; Cox, B.G.; Schneider, H. Stability constants of copper(I) and silver(I) complexes with open-chain, macrocyclic and -bicyclic aza-ligands in acetonitrile and comparison with results in dimethylsulfoxide. Inorg. Chim. Acta 1999, 286, 160–168. [Google Scholar]
- Gravante, G.; Montone, A. A retrospective analysis of ambulatory burn patients: focus on wound dressings and healing times. Ann. R. Coll. Surg. Engl 2010, 92, 118–123. [Google Scholar]
- Xu, R.X. Physiological Healing Procedure and Histological Observation on Deep Second-Degree Burns Treated with MEBO. In Burns Regenerative Medicine and Therapy; Sun, X., Ed.; Karger: Basel, Switzerland, 2004; pp. 106–111. [Google Scholar]
- Kim, S.; Choi, J.E.; Choi, J.; Chung, K.H.; Park, K.; Yi, J.; Ryu, D.Y. Oxidative stress-dependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol. In Vitro 2009, 23, 1076–1084. [Google Scholar]
- Greulich, C.; Diendorf, J.; Simon, T.; Eggeler, G.; Epple, M.; Koeller, M. Uptake and intracellular distribution of silver nanoparticles in human mesenchymal stem cells. Acta Biomater 2011, 7, 347–354. [Google Scholar]
- Hirokawa, N.; Bloom, G.S.; Vallee, R.B. Cytoskeletal architecture and immunocytochemical localization of microtubule-associated proteins in regions of axons associated with rapid axonal transport: The β,β′-iminodipropionitrile-intoxicated axon as a model system. J. Cell. Biol 1985, 101, 227–239. [Google Scholar]
- Ball, E.H.; Singer, S. Mitochondria are associated with microtubules and not with intermediate filaments in cultured fibroblasts. Prod. Natl. Acad. Sci. USA 1982, 79, 123–126. [Google Scholar]
- Lee, H.C.; Yin, P.H.; Lu, C.Y.; Chi, C.W.; Wei, Y.H. Increase of mitochondria and mitochondrial DNA in response to oxidative stress in human cells. Biochem. J 2000, 348, 425–432. [Google Scholar]
- Marchi, S.; Giorgi, C.; Suski, J.M.; Agnoletto, C.; Bononi, A.; Bonora, M.; de Marchi, E.; Missiroli, S.; Patergnani, S.; Poletti, F.; et al. Mitochondria-ros crosstalk in the control of cell death and aging. J. Signal. Transduct. 2012, 2012. [Google Scholar] [CrossRef]
- Kim, H.R.; Kim, M.J.; Lee, S.Y.; Oh, S.M.; Chung, K.H. Genotoxic effects of silver nanoparticles stimulated by oxidative stress in human normal bronchial epithelial (BEAS-2B) cells. Mutat. Res. Gen. Tox. En 2011, 726, 129–135. [Google Scholar]
- Ciriolo, M.R.; Civitareale, P.; Carri, M.; De Martino, A.; Galiazzo, F.; Rotilio, G. Purification and characterization of Ag, Zn-superoxide dismutase from Saccharomyces cerevisiae exposed to silver. J. Biol. Chem 1994, 269, 25783–25787. [Google Scholar]
- Lansdown, A.B.G. Silver in health care: Antimicrobial effects and safety in use. Curr. Probl. Dermatol 2006, 33, 17–34. [Google Scholar]
- Cuttle, L.; Naidu, S.; Mill, J.; Hoskins, W.; Das, K.; Kimble, R.M. A retrospective cohort study of Acticoat™ versus Silvazine™ in a paediatric population. Burns 2007, 33, 701–707. [Google Scholar]
- Marazzi, M.; de Angelis, A.; Ravizza, A.; Ordanini, M.N.; Falcone, L.; Chiaratti, A.; Crovato, F.; Calò, D.; Veronese, S.; Rapisarda, V. Successful management of deep facial burns in a patient with extensive third-degree burns: The role of a nanocrystalline silver dressing in facilitating resurfacing. Int. Wound J 2007, 4, 8–14. [Google Scholar]
- Strand, O.; San Miguel, L.; Rowan, S.; Sahlqvist, A. Retrospective comparison of two years in a paediatric burns unit, with and without acticoat as a standard dressing. Ann. Burns Fire Disasters 2010, 23, 182–185. [Google Scholar]
- Rheinwald, J.G.; Green, H. Serial cultivation of strains of human epidermal keratinocytes: The formation of keratinizing colonies from single cells. Cell 1975, 6, 331–343. [Google Scholar]
- Brun, P.; Cortivo, R.; Zavan, B.; Vecchiato, N.; Abatangelo, G. In vitro reconstructed tissues on hyaluronan-based temporary scaffolding. J. Mater. Sci. Mater. M 1999, 10, 683–688. [Google Scholar]
- Denizot, F.; Lang, R. Rapid colorimetric assay for cell-growth and survival—Modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J. Immunol. Methods 1986, 89, 271–277. [Google Scholar]

| Ag (μg cm−2) | |||||
|---|---|---|---|---|---|
| Days | Sample size | Total applied | Dressing | Medium | Culture * |
| 1–3 | n = 9 | 821 | 767 ± 4 | 37 ± 5 | 18 ± 3 |
| 3–6 | n = 6 | 821 | 765 ± 3 | 31 ± 3 | 25 ± 2 |
| 6–9 | n = 3 | 821 | 765 ± 4 | 32 ± 2 | 25 ± 3 |
| Days | Estimated | MTT + iDMSO | MTT | iDMSO | TMAH |
|---|---|---|---|---|---|
| 3 | 16.5 | 16.3 | 5.8 | 10.5 | - |
| 6 | 40.9 | 30.9 | 8.0 | 22.9 | - |
| 9 | 62.9 | 46.5 | 10.3 | 36.2 | - |
| Control experiment in absence of cellsn= 3 replicates ± 1 SD | |||||
| 3 | - | 12.5 ± 5.1 | 8.9 ± 3.7 | 3.3 ± 1.8 | 0.7 ± 0.3 |
| RF power | 1500 W |
| Plasma gas flow rate | 15 L min−1 |
| Carrier gas flow rate | 1.2 mL min−1 |
| Nebulizer | V-groove |
| Ions lens setting | Optimized daily for best sensitivity using a 10 ng mL−1 Ag in NH4OH 2.8% w/w solution |
| Monitored masses | 107, 109 |
| Points per peak | 3 |
| Acquisition time per point | 1 s |
| Replicates | 5 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rigo, C.; Ferroni, L.; Tocco, I.; Roman, M.; Munivrana, I.; Gardin, C.; Cairns, W.R.L.; Vindigni, V.; Azzena, B.; Barbante, C.; et al. Active Silver Nanoparticles for Wound Healing. Int. J. Mol. Sci. 2013, 14, 4817-4840. https://doi.org/10.3390/ijms14034817
Rigo C, Ferroni L, Tocco I, Roman M, Munivrana I, Gardin C, Cairns WRL, Vindigni V, Azzena B, Barbante C, et al. Active Silver Nanoparticles for Wound Healing. International Journal of Molecular Sciences. 2013; 14(3):4817-4840. https://doi.org/10.3390/ijms14034817
Chicago/Turabian StyleRigo, Chiara, Letizia Ferroni, Ilaria Tocco, Marco Roman, Ivan Munivrana, Chiara Gardin, Warren R. L. Cairns, Vincenzo Vindigni, Bruno Azzena, Carlo Barbante, and et al. 2013. "Active Silver Nanoparticles for Wound Healing" International Journal of Molecular Sciences 14, no. 3: 4817-4840. https://doi.org/10.3390/ijms14034817
APA StyleRigo, C., Ferroni, L., Tocco, I., Roman, M., Munivrana, I., Gardin, C., Cairns, W. R. L., Vindigni, V., Azzena, B., Barbante, C., & Zavan, B. (2013). Active Silver Nanoparticles for Wound Healing. International Journal of Molecular Sciences, 14(3), 4817-4840. https://doi.org/10.3390/ijms14034817







