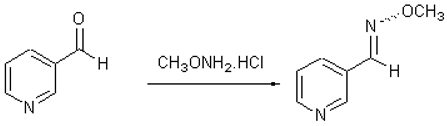
Pyridine-3-carboxaldehyde O-methyloxime
Supplementary Materials
Reference
Sample availability: available from the authors. |
© 2001 MDPI. All rights reserved. Molecules website www.mdpi.org/molecules/
Share and Cite
Caprio, V.E.; Jones, M.W.; Brimble, M.A. Pyridine-3-carboxaldehyde O-methyloxime. Molecules 2001, 6, M202. https://doi.org/10.3390/M202
Caprio VE, Jones MW, Brimble MA. Pyridine-3-carboxaldehyde O-methyloxime. Molecules. 2001; 6(3):M202. https://doi.org/10.3390/M202
Chicago/Turabian StyleCaprio, Vittorio E., Michael W. Jones, and Margaret A. Brimble. 2001. "Pyridine-3-carboxaldehyde O-methyloxime" Molecules 6, no. 3: M202. https://doi.org/10.3390/M202
APA StyleCaprio, V. E., Jones, M. W., & Brimble, M. A. (2001). Pyridine-3-carboxaldehyde O-methyloxime. Molecules, 6(3), M202. https://doi.org/10.3390/M202



