First Total Synthesis of Pestasulfamides A and B Through Iminoketene Dimerization of Anthranilic Acid in One-Pot Manner
Abstract
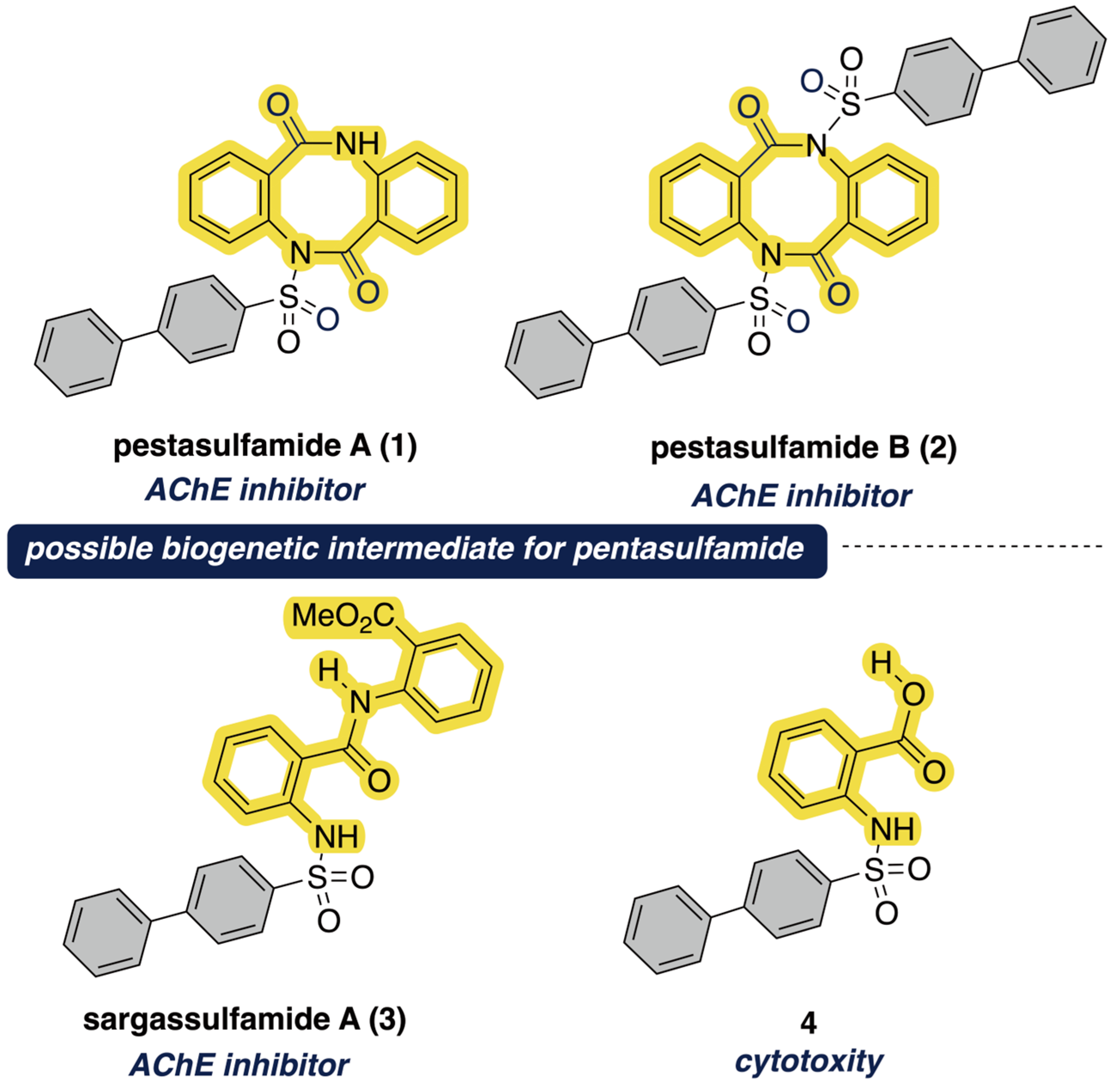
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Experimental Procedures and Characterization Data for the Synthetic Products
3.2.1. Synthesis of Pestasulfamide A (1) and Pestasulfamide B (2)
- [Typical procedure for run 3, Table 1]
- 5-([1,1′-Biphenyl]-4-ylsulfonyl)dibenzo[b,f][1,5]diazocine-6,12(5H,11H)-dione (pestasulfamide A, 1): Mp 273–275 °C dec.; IR (KBr) ν: 3539, 3093, 2979, 1711, 1653, 1564, 1498, 1282, 1093, 814 cm−1; 1H NMR (600 MHz, CDCl3) δ 12.38 (s, 1H), 8.24 (dd, J = 7.8, 1.2 Hz, 1H), 8.19 (dd, J = 7.8, 1.8 Hz, 1H), 7.89–7.93 (m, 3H), 7.78 (t, J = 9.0 Hz, 2H), 7.60 (td, J = 8.4, 1.2 Hz, 1H), 7.57 (d, J = 8.4 Hz, 2H), 7.49–7.51 (m, 3H), 7.42 (t, J = 7.8 Hz, 2H), 7.38 (tt, J = 9.9, 1.1 Hz, 1H), 7.16 (td, J = 8.7, 1.0 Hz, 1H); 13C{1H} NMR (151 MHz, CDCl3) δ 158.1, 157.2, 146.1, 145.0, 139.8, 139.1, 138.1, 137.3, 134.3, 129.9, 129.3, 129.1, 129.0, 128.7, 127.8, 127.3, 126.8, 123.5, 119.5, 116.7, 115.5; HRMS (ESI) m/z: [M+Na]+ calcd for C26H18N2O4SNa 477.0880; found 477.0881.
- 5,11-Bis([1,1′-biphenyl]-4-ylsulfonyl)dibenzo[b,f][1,5]diazocine-6,12(5H,11H)-dione (pestasulfamide B, 2): Mp 214–216 °C, dec.; IR (KBr) v: 3032, 2970, 1732, 1564, 1481, 1450, 1367, 1244, 1173, 1080, 768 cm−1; 1H NMR (600 MHz, CDCl3) δ 8.11 (d, J = 8.4 Hz, 4H), 7.75 (d, J = 8.4 Hz, 4H), 7.61 (d, J = 7.2 Hz, 4H), 7.40–7.47 (m, 8H), 7.33–7.35 (m, 4H), 7.15–7.18 (m, 2H); 13C{1H} NMR (151 MHz, CDCl3) δ 165.6, 147.4, 139.2, 136.1, 134.3, 133.5, 132.1, 130.7, 130.3, 129.3, 129.1, 129.0, 128.8, 127.8, 127.6; HRMS (ESI) m/z: [M+Na]+ calcd for C38H26N2O6S2Na 693.1125; found 693.1129.
3.2.2. Typical Procedure for Run 8, Table 1
3.2.3. Typical Procedure for Run 13, Table 1
3.2.4. Typical Procedure for Run 14, Table 1
3.2.5. Typical Procedure for Run 15, Table 1
3.3. Preparation of Dibenzo[b,f][1,5]Diazocine-6,12 (5H,11H)-Dione (7) [41]
3.4. Synthesis of 2-[([1,1′-Biphenyl]-4-Ylsulfonyl)Amino]Benzoic Acid (8) [42,43]
- 2-([1,1′-biphenyl]-4-sulfonamide)benzoic acid (8): Mp 210–212 °C, dec.; IR (KBr) v: 3392, 3234, 3043, 1680, 1493, 1342, 1159, 761 cm−1; 1H NMR (600 MHz, DMSO-d6) δ 7.90 (d, J = 7.2 Hz, 1H), 7.84 (d, J = 8.4 Hz, 2H), 7.74 (d, J = 7.8 Hz, 2H), 7.63 (d, J = 7.2 Hz, 2H), 7.44 (q, J = 8.2 Hz, 3H), 7.39 (t, J = 7.2 Hz, 1H), 7.26 (t, J = 7.2 Hz, 1H), 6.85 (t, J = 7.5 Hz, 1H); 13C{1H} NMR (151 MHz, DMSO-d6) δ 170.9, 144.0, 143.3, 140.6, 138.9, 132.1, 131.7, 129.5, 128.8, 127.64, 127.56, 127.4, 122.3, 121.0, 117.4; HRMS (ESI) m/z: [M+Na]+ calcd for C19H15NO4SNa 376.0614; found 376.0616.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chhablani, A.; Jain, V.; Makasana, J. Sulfonamides: Historical Perspectives, Therapeutic Insights, Applications, Challenges, and Synthetic Strategies. ChemistrySelect 2025, 10, e2122. [Google Scholar] [CrossRef]
- Ilardi, E.A.; Vitaku, E.; Njardarson, J.T. Datamining for Sulfur and Fluorine: An Evaluation of Pharmaceuticals to Reveal Opportunities for Drug Design and Discovery. J. Med. Chem. 2014, 57, 2832–2842. [Google Scholar] [CrossRef]
- Ovung, A.; Bhattacharyya, J. Sulfonamide drugs: Structure, Antibacterial Property, Toxicity, and Biophysical Interactions. Biophys. Rev. 2021, 13, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Delost, M.D.; Qureshi, M.H.; Smith, D.T.; Njardarson, J.T. A survey of the Structures of US FDA Approved Combination Drugs. J. Med. Chem. 2019, 62, 4265–4311. [Google Scholar] [CrossRef]
- Zuercher, W.J.; Buchholz, R.G.; Campobasso, N.; Collins, J.L.; Galardi, C.M.; Gampe, R.T.; Hyatt, S.M.; Merrihew, S.L.; Moore, J.T.; Oplinger, J.A.; et al. Discovery of Tertiary Sulfonamides as Potent Liver X Receptor Antagonists. J. Med. Chem. 2010, 53, 3412–3416. [Google Scholar] [CrossRef]
- Kosak, U.; Benedik, N.S.; Knez, D.; Zakelj, S.; Trontelj, J.; Pislar, A.; Horvant, S.; Bolje, A.; Znidarsic, N.; Grgurevic, N.; et al. Lead Optimization of a Butylcholinesterase Inhibitor for the Treatment of Alzheimer’s Disease. J. Med. Chem. 2025, 68, 11693–11723. [Google Scholar] [CrossRef]
- Jiang-Song; Gao, Q.-L.; Wu, B.-W.; Li, D.; Shi, L.; Zhu, T.; Lou, J.-F.; Jin, C.-Y.; Zhang, Y.-B.; Zhang, S.-Y.; et al. Novel Tertiary Sulfonamide Derivatives Containing Benzimidazole Moiety as Potent Anti-gastric Cancer Agents: Design, Synthesis, and SAR Studies. Eur. J. Med. Chem. 2019, 183, 111731. [Google Scholar] [CrossRef]
- Giampietro, L.; Marinacci, B.; Valle, A.D.; D’Agostino, I.; Lauro, A.; Mori, M.; Carradori, S.; Ammazzzalorso, A.; De Filippis, B.; Maccallini, C.; et al. Azobenzenesulfonamide Carbonic Anhydrase Inhibitors as New Weapons to Fight Helicobacter pylori: Synthesis, Bioactivity Evaluation, In Vivo Toxicity, and Computational Studies. Pharmaceuticals 2024, 17, 1027. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Yang, J.; Zhou, Y.; Li, X.; Zou, Y.; Zheng, Z.; Guo, M.; Chen, Z.; Cho, W.J.; Chaattipakorn, N.; et al. Design, Synthesis, and Bioactivity Evaluation of Novel Amide/Sulfonamide Derivatives as Potential Anti-inflammatory Agents Against Acute Lung Injury and Ulcerative Colitis. Eur. J. Med. Chem. 2023, 259, 115706. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, W.; Xie, B.; Chen, T.; Chen, S.; Liu, Z.; Sun, B.; She, Z. First discovery of sulfonamides derivatives with acetylcholinesterase inhibitory activity in fungus Pestalotiopsis sp. HNY36-1D. Tetrahedron 2023, 142, 133524. [Google Scholar] [CrossRef]
- Xu, Y.; Liang, X.; Li, Y.; Liang, Z.; Huang, W.; Zhang, H.; Jiang, Y.; Cui, J.; Song, X. Arylsulfonamides from the Roots and Rhizomes of Tupistra chinensis Baker. Nat. Prod. Commun. 2020, 15, 1934578X20921665. [Google Scholar] [CrossRef]
- Peng, Y.; Cao, L.; Liu, Y.; Huang, R. Sargassulfamide A, An Unprecedented Amide Derivatives from the Seaweed Sargassum naozhouense. Chem. Nat. Compd. 2020, 56, 98–100. [Google Scholar] [CrossRef]
- Mujumdar, P.; Polsen, S.-A. Natural Product Primary Sulfonamides and Primary Sulfamates. J. Nat. Prod. 2015, 78, 1470–1477. [Google Scholar] [CrossRef]
- Baunach, M.; Ding, L.; Willing, K.; Hertweck, C. Bacterial Synthesis of Unusual Sulfonamide and Sulfone Antibiotics by Flavoenzyme-Mediated Sulfur Dioxide Capture. Angew. Chem. Int. Ed. 2015, 54, 13279–13283. [Google Scholar] [CrossRef]
- Jain, P.; Saravanan, C.; Singh, S.K. Sulphonamides: Deserving Class of MMP Inhibitors? Eur. J. Med. Chem. 2013, 60, 89–100. [Google Scholar] [CrossRef]
- Kaludercic, N.; Lindsey, M.L.; Tavazzi, B.; Lazzarino, G.; Paolocci, N. Inhibiting Metalloproteases with PD 166793 in Heart Failure: Impact of Cardic Remodeling and Beyond. Cardiovasc. Ther. 2008, 26, 24–37. [Google Scholar] [CrossRef]
- Cooper, F.C.; Partridge, M.W. 564. Cyclic Amidines. Part V. 5:11-endo-Substituted 5:6:11:12-tetrahydro-2:8-dimethylphenhomazines. J. Chem. Soc. 1957, 2888–2893. [Google Scholar] [CrossRef]
- Olszewska, T.; Gdaniec, M.; Polonski, T. Planner Chiral Dianthranilide and Dithiodianthranilide Molecules: Optical Resolution, Chroptical Spectra, and Molecular Self-Assembly. J. Org. Chem. 2004, 69, 1248–1255. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Pan, Y.; Jin, L.L.; Yin, Y.; Yang, B.-Z.; Sun, X.-Q. Chiral Exploration of 6,12-Diphenyldibenzo[b.f][1,5]diazocine with Stable Conformation. Chirality 2017, 29, 134–139. [Google Scholar] [CrossRef]
- Gregory, M.A.; Zhang, B.; Tilley, A.J.; Scheerlinck, T.; White, J.M. Amine-Substituted Diazocine Derivatives—Synthesis, Structure, and Photophysical Properties. Helv. Chim. Acta 2018, 101, e180146. [Google Scholar] [CrossRef]
- Nonnenmacher, E.; Hevertt, A.; Mahamoudt, A.; Aubert, C.; Molnartt, J.; Barbe, J. A Novel route to new dibenzo[b,f][1,5]diazocine Derivatives as Chemosensitizers. Org. Prep. Proced. Int. 1997, 29, 711–715. [Google Scholar] [CrossRef]
- Hoorfar, A.; Ollis, W.D.; Price, J.A.; Stephanatou, J.S.; Stoddart, J.F. Conformational Behaviour of Medium-sized Rings. Part 11. Dianthranilides and Trianthranilides. J. Chem. Soc. Perkin Trans. 1982, 1, 1649–1699. [Google Scholar] [CrossRef]
- Gordon-Wylie, S.W.; Teplin, E.; Morris, J.C.; Trombley, M.I.; McCarthy, S.M.; Cleaver, W.M.; Clark, G.R. Exploring Hydrogen-Bonded Structures: Synthesis and X-ray Crystallographic Screening of a Cisoid Cyclic Dipeptide Mini-Library. Cryst. Growth Des. 2004, 4, 789–797. [Google Scholar] [CrossRef]
- Qiu, L.; Wang, X.; Zhao, N.; Xu, S.; An, Z.; Shuang, X.; Lan, Z.; Wen, L.; Wan, X. Reductive Ring Closure Methodology toward Heteroacenes Bearing a Dihydropyrrolo[3,2-b]pyrrole Core: Scope and Limitation. J. Org. Chem. 2014, 79, 11339–11348. [Google Scholar] [CrossRef] [PubMed]
- Bieszczad, B.; Garbicz, D.; Trzybinski, D.; Mielecki, D.; Wozniak, K.; Grzesiuk, E.; Mieczkowski, A. Unsymmetrically Substituted Dibenzo[b,f][1,5]-diazocine-6,12(5H,11H)dione–A Convenient Scaffold for Bioactive Molecule Design. Molecules 2020, 25, 906. [Google Scholar] [CrossRef]
- Acs, P.; Müller, E.; Rangits, G.; Lóránd, T.; Kollár, L. Palladium-Catalyzed Carbonylation of 4-Substituted 2-Iodoaniline Derivatives: Carbonylative Cyclization and Aminocarbonylation. Tetrahedron 2006, 62, 12051–12056. [Google Scholar] [CrossRef]
- Kamal, A.; Ramana, K.V.; Ankati, H.B.; Ramana, A.V. Mild and Efficient Reduction of Azides to Amines: Synthesis of Fused [2,1]Quinazolines. Tetrahedron Lett. 2002, 43, 6861–6863. [Google Scholar] [CrossRef]
- Kametani, T.; Asagi, S.; Nakamura, S.; Satoh, M.; Wagatsuma, N.; Takano, S. Compounds containing Amino, Hydroxyl, or Carboxyl Groups with Mesyl Chloride. Yakugaku Zasshi 1966, 86, 107–109. [Google Scholar] [CrossRef]
- Wang, B.; Mi, M.; Li, S.-J.; Huang, J.-F.; Feng, P.; Lin, L.; Tang, Y. The Studies on the Synthesizing Dibenzodiazocinediimine via Constructing an Eight-Membered Ring. J. Org. Chem. 2025, 90, 6655–6661. [Google Scholar] [CrossRef]
- Wang, X.; Li, J.; Zhao, N.; Wan, X. A Rapid and Efficient Access to Diaryldibenzo[b,f][1,5]diazocines. Org. Lett. 2011, 13, 709–711. [Google Scholar] [CrossRef] [PubMed]
- Mao, D.; Tang, J.; Wang, W.; Wu, S.; Liu, X.; Yu, J.; Wang, L. Scandium Pentafluorobenzoate-Catalyzed Unexpected Cascade Reaction of 2-Aminobenzaldehydes with Primary Amines: A Process for the Preparation of Ring-Fused Aminals. J. Org. Chem. 2013, 78, 12848–12854. [Google Scholar] [CrossRef]
- Pettersson, B.; Bergman, J.; Svensson, P.H. Synthetic Studies towards 1,5-Benzodiazocines. Tetrahedron 2013, 69, 2647–2654. [Google Scholar] [CrossRef]
- Rode, N.D.; Arcadi, A.; Chiarini, M.; Marinelli, F.; Portalone, G. Gold-Catalyzed Synthesis of Dibenzo[1,5]diazocines from β-(2-Aminophenyl)-α,β-ynones. Adv. Synth. Catal. 2017, 359, 3371–3377. [Google Scholar] [CrossRef]
- Ramle, A.Q.; Tiekink, E.R.T. Synthetic Strategies and diversification of Dibenzo[1,5]diazocines. Org. Biomol. Chem. 2023, 21, 2870–2888. [Google Scholar] [CrossRef]
- Abe, T.; Itoh, T.; Choshi, T.; Hibino, S.; Ishikura, M. One-pot Synthesis of Tryptanthrin by the Dakin Oxidation of Indole-3-Carbaldehyde. Tetrahedron Lett. 2014, 55, 5268–5270. [Google Scholar] [CrossRef]
- Abe, T.; Tarasaki, M. Synthesis of Phaitanthrin E and Tryptanthrin through Amination/Cyclization Cascade. Helv. Chim. Acta 2018, 101, e1700284. [Google Scholar] [CrossRef]
- Itoh, T.; Abe, T.; Choshi, T.; Nishiyama, T.; Ishikura, M. A One-pot Synthesis of Phaitanthrin E through Intermolecular Condensation/Intramolecular Aryl C–H Amination Cascade. Heterocycles 2016, 92, 1132–1136. [Google Scholar]
- Abe, T.; Takeda, H.; Miwa, Y.; Yamada, K.; Yanada, R.; Ishikura, M. Copper-Catalyzed Ritter-type Reaction of Unactivated Alkenes with Dichloramine-T. Helv. Chim. Acta 2010, 93, 233–241. [Google Scholar] [CrossRef]
- Abe, T.; Kida, K.; Yamada, K. A Copper-Catalyzed Ritter-type Cascade via Iminoketene for the Synthesis of Quinazolin-4(3H)-ones and Diazocines. Chem. Commun. 2017, 53, 4362–4365. [Google Scholar] [CrossRef]
- Kametani, T.; Higa, T.; Loc, C.V.; Ihara, M.; Koizumi, M.; Fukumoto, K. Iminoketene Cycloaddition. 1. A Facile Synthesis of Quinazolone System by Condensation of Iminoketene with Imines–A Total Synthesis of Evodiamine and Rutecarpine by Retro Mass-Spectral Synthesis. J. Am. Chem. Soc. 1976, 98, 6186–6188. [Google Scholar] [CrossRef]
- Qiu, L.; Yu, C.; Zhao, N.; Chen, W.; Guo, Y.; Wan, X.; Yang, R.; Liu, Y. An Expedient Synthesis of Fused Heteroacenes Bearing a Pyrrolo[3,2-b]pyrrole Core. Chem. Comm. 2012, 48, 12225–12227. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.M.; Turner, J.; Pina, I.C.; Garrabrant, K.A.; Geerts, D.; Bachmann, A.S.; Peterson, Y.K.; Woster, P.M. Synthesis and Evaluation of Small Molecule Inhibitors of LSD1 for Use Against MYCN-Expressing Neuroblastoma. Eur. J. Med. Chem. 2022, 244, 114818. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Shi, L.; Liu, M.; Zhang, Z.; Liu, F.; Long, T.; Wen, S.; Huang, D.; Wang, K.; Zhou, R.; et al. Design, Synthesis, and Cytotoxic Activity of Novel Natural Arylsulfonamide-Inspired Molecules. Molecules 2022, 27, 1479. [Google Scholar] [CrossRef] [PubMed]





 | ||||||
|---|---|---|---|---|---|---|
| Run a | PhBsCl (X Equiv.) | Solvent (0.2 M) | Temp. (°C) | Time (h) | Yield (%) of 1 b | Yield (%) of 2 b |
| 1 | 1.0 | pyridine | rt | 0.1 | 21 | 8 |
| 2 | 1.5 | pyridine | rt | 0.1 | 33 | 16 |
| 3 | 2.0 | pyridine | rt | 0.1 | 47 | 37 |
| 4 | 2.5 | pyridine | rt | 0.1 | 40 | 39 |
| 5 | 3.0 | pyridine | rt | 0.1 | 35 | 20 |
| 6 | 2.0 | pyridine | 0 | 0.1 | 34 | 41 |
| 7 | 2.0 | pyridine | 60 | 0.1 | 46 | 45 |
| 8 | 2.0 | pyridine | reflux | 0.1 | 50 | 40 |
| 9 | 2.0 | Et3N | rt | 24 | 0 | 0 |
| 10 | 2.0 | AcOEt | rt | 24 | 0 | 0 |
| 11 | 2.0 | THF | rt | 24 | 0 | 0 |
| 12 | 2.0 | DMSO | rt | 24 | 0 | 0 |
| 13 | 2.0 | pyridine/THF (1/1, v/v) | rt | 0.1 | 50 | 41 |
| 14 | 2.0 | pyridine/DMSO (1/1, v/v) | rt | 0.1 | 23 | 16 |
| 15 | 2.0 | pyridine/DMF (1/1, v/v) | rt | 0.1 | 0 | 15 |
 | ||||
|---|---|---|---|---|
| Position | Synthetic 1 δH Mult (J in Hz) 600 MHz, CDCl3 | Natural 1 δH Mult (J in Hz) 600 MHz, CDCl3 | Synthetic 1 δc 151 MHz, CDCl3 | Natural 1 δc 150 MHz, CDCl3 |
| 1 | 158.1 | 165.6 | ||
| 2 | 116.7 | 119.6 | ||
| 3 | 8.24, dd (1.2, 7.8) | 7.93, dd (1.5, 8.1) | 129.0 | 126.8 |
| 4 | 7.60, td (1.2, 8.4) | 7.48, overlap | 129.3 | 129.2 |
| 5 | 7.91, m | 7.50, overlap | 137.3 | 137.3 |
| 6 | 7.78, t (9.0) | 7.62, m | 126.8 | 116.8 |
| 7 | 145.0 | 139.9 | ||
| 8 | 138.1 | 143.7 | ||
| 9 | 7.91, m | 7.71, overlap | 127.8 | 128.7 |
| 10 | 7.57, d (8.7) | 7.90, overlap | 127.8 | 127.8 |
| 11 | 146.1 | 145.1 | ||
| 12 | 7.57, d (8.7) | 7.90, overlap | 127.8 | 127.8 |
| 13 | 7.91, m | 7.71, overlap | 127.8 | 128.7 |
| 14 | 139.1 | 146.2 | ||
| 15 | 7.50, m | 7.57, overlap | 127.3 | 128.7 |
| 16 | 7.42, t (7.8) | 7.44, overlap | 129.1 | 129.3 |
| 17 | 7.38, tt (1.1, 9.9) | 7.38, overlap | 128.7 | 127.4 |
| 18 | 7.42, t (7.8) | 7.44, overlap | 129.1 | 129.3 |
| 19 | 7.50, m | 7.44, overlap | 127.3 | 128.7 |
| 1′ | 157.2 | 163.4 | ||
| 2′ | 115.5 | 123.5 | ||
| 3′ | 8.19, dd (1.8, 7.8) | 8.19, dd (1.5, 8.1) | 129.9 | 129.9 |
| 4′ | 7.16, td (1.0, 8.7) | 7.16, m | 123.5 | 123.5 |
| 5′ | 7.50, m | 7.52, overlap | 134.3 | 134.3 |
| 6′ | 7.78, t (9.0) | 8.25, dd (1.1, 7.9) | 119.5 | 115.6 |
| 7′ | 139.8 | 138.2 | ||
| 1-NH | 12.38, s | 12.39, brs | --- | --- |
 | ||||
|---|---|---|---|---|
| Position | Synthetic 2 δH Mult (J in Hz) 600 MHz, CDCl3 | Natural 2 δH Mult (J in Hz) 500 MHz, CDCl3 | Synthetic 2 δc 151 MHz, CDCl3 | Natural 2 δc 125 MHz, CDCl3 |
| 1 | 165.6 | 165.6 | ||
| 2 | 133.5 | 133.6 | ||
| 3 | 7.44, m | 7.44, overlap | 129.0 | 130.7 |
| 4 | 7.17, m | 7.18, m | 129.3 | 129.3 |
| 5 | 7.34, m | 7.35, overlap | 132.1 | 132.1 |
| 6 | 7.34, m | 7.35, overlap | 130.7 | 129.1 |
| 7 | 134.3 | 134.4 | ||
| 8 | 136.1 | 139.3 | ||
| 9 | 8.11, d (8.4) | 7.77, d (7.9) | 130.3 | 127.8 |
| 10 | 7.75, d (8.4) | 8.13, d (7.9) | 127.8 | 130.5 |
| 11 | 147.4 | 136.2 | ||
| 12 | 7.75, d (8.4) | 8.13, d (7.9) | 127.8 | 130.5 |
| 13 | 8.11, d (8.4) | 7.77, d (7.9) | 130.3 | 127.8 |
| 14 | 139.2 | 147.5 | ||
| 15 | 7.61, d (7.2) | 7.62, d (7.2) | 127.6 | 127.6 |
| 16 | 7.44, m | 7.45, overlap | 129.1 | 129.2 |
| 17 | 7.44, m | 7.44, overlap | 128.8 | 128.8 |
| 18 | 7.44, m | 7.45, overlap | 129.1 | 129.2 |
| 19 | 7.61, d (7.2) | 7.62, d (7.2) | 127.6 | 127.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kobori, Y.; Abe, T. First Total Synthesis of Pestasulfamides A and B Through Iminoketene Dimerization of Anthranilic Acid in One-Pot Manner. Molecules 2026, 31, 47. https://doi.org/10.3390/molecules31010047
Kobori Y, Abe T. First Total Synthesis of Pestasulfamides A and B Through Iminoketene Dimerization of Anthranilic Acid in One-Pot Manner. Molecules. 2026; 31(1):47. https://doi.org/10.3390/molecules31010047
Chicago/Turabian StyleKobori, Yuito, and Takumi Abe. 2026. "First Total Synthesis of Pestasulfamides A and B Through Iminoketene Dimerization of Anthranilic Acid in One-Pot Manner" Molecules 31, no. 1: 47. https://doi.org/10.3390/molecules31010047
APA StyleKobori, Y., & Abe, T. (2026). First Total Synthesis of Pestasulfamides A and B Through Iminoketene Dimerization of Anthranilic Acid in One-Pot Manner. Molecules, 31(1), 47. https://doi.org/10.3390/molecules31010047








