Multifunctional Tacrine–Quinoline Hybrids as Cholinesterase Inhibitors, Aβ Aggregation Blockers, and Metal Chelators for Alzheimer’s Therapy
Abstract
1. Introduction
2. Result and Discussion
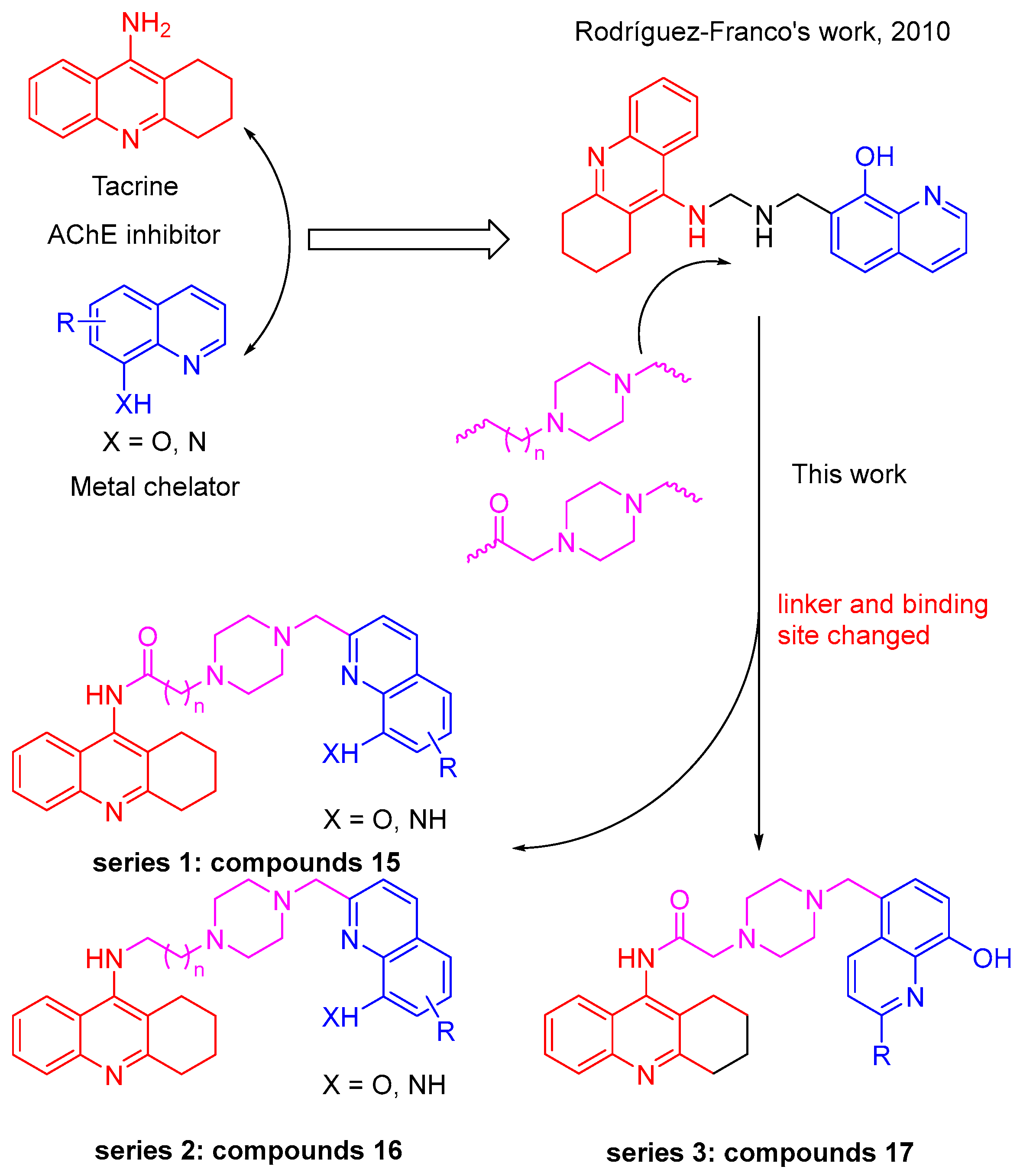
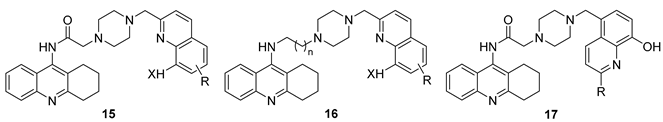
2.1. Design
2.2. Synthetic Methodology
2.3. Cholinesterase Inhibitory Activity
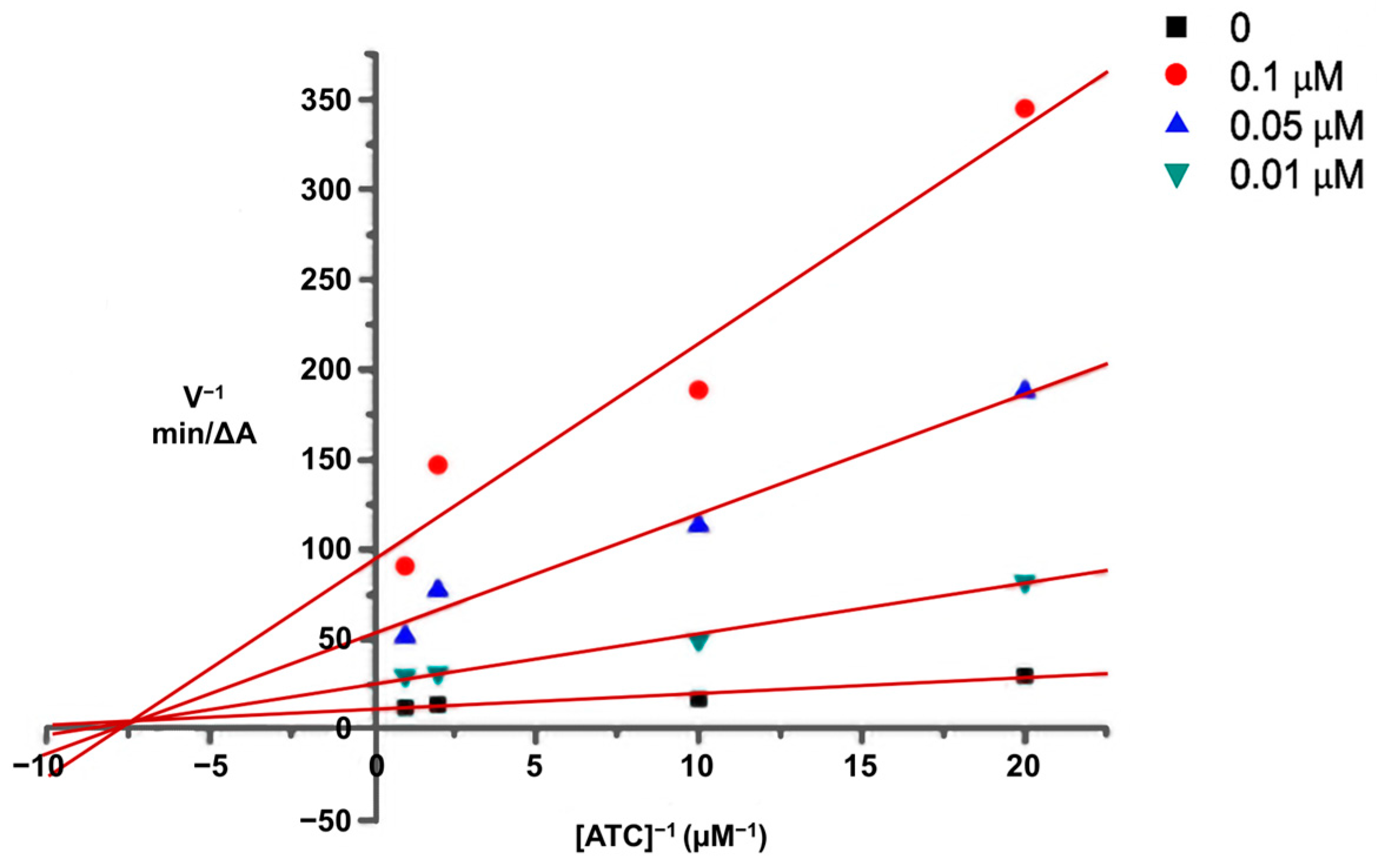
2.4. Kinetic Study of AChE Inhibition
2.5. Self-Induced Aβ1–42 Aggregation
2.6. Metal-Chelating Properties
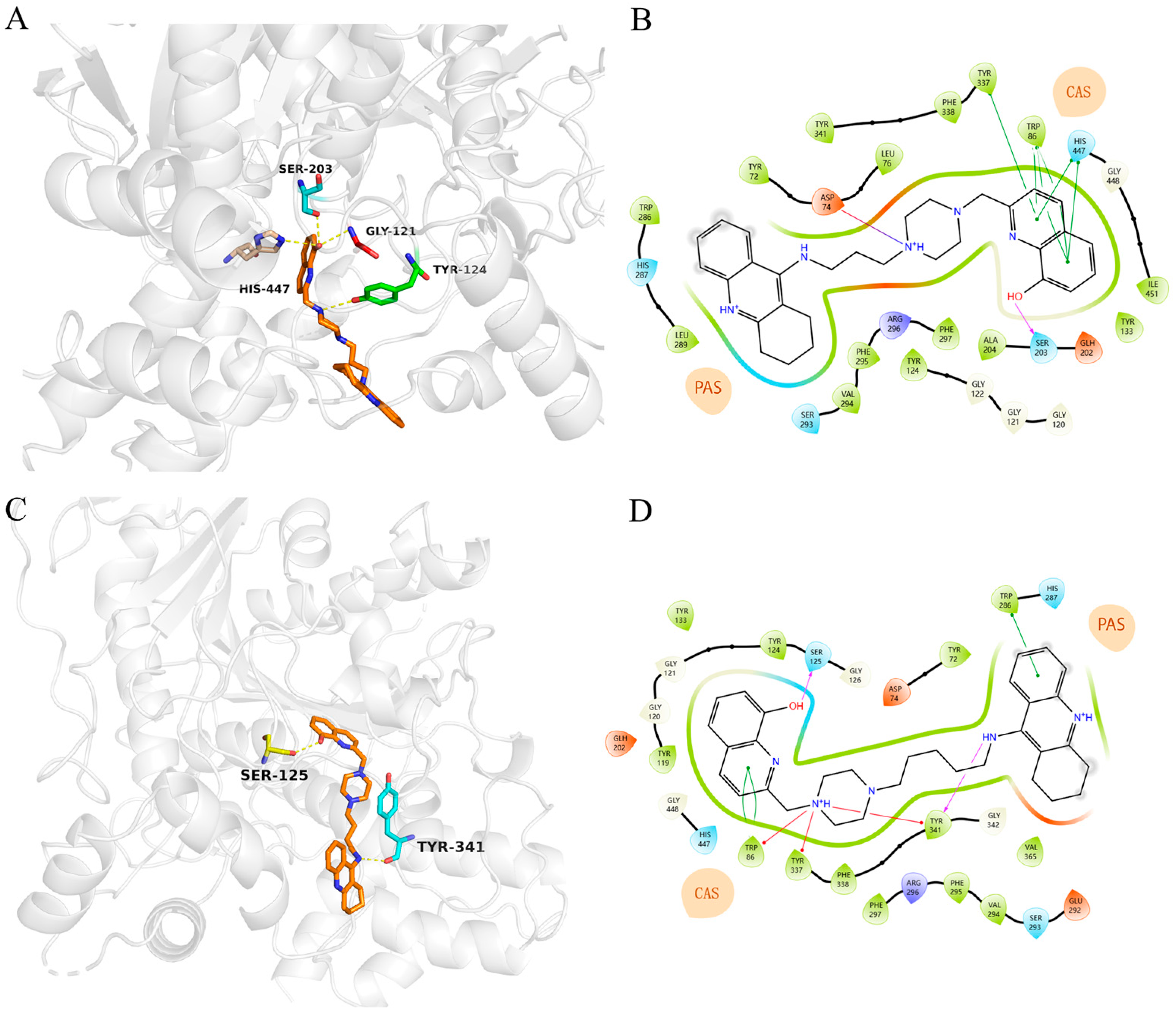
2.7. Molecular Docking into Cholinesterases
3. Materials and Methods
3.1. Chemistry
3.1.1. General Procedures for the Preparation of Compounds 15–16
- 2-(4-((8-Hydroxyquinolin-2-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (15a). Yield 66%, White solid; 1H NMR (400 MHz, CDCl3) δ 9.22 (s, 1H), 8.14 (d, J = 10.0 Hz, 1H), 8.00 (d, J = 8.0 Hz, 1H), 7.71 (d, J = 8.4 Hz, 1H), 7.66–7.61 (m, 2H), 7.48–7.41 (m, 2H), 7.32 (d, J = 8.4 Hz, 1H), 7.16 (d, J = 7.6 Hz, 1H), 3.88 (s, 2H), 3.33 (s, 2H), 3.15 (t, J = 6.4 Hz, 2H), 2.85–2.80 (m, 6H), 2.70 (m, 4H), 1.99–1.95 (m, 2H), 1.90–1.85 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 168.6, 159.9, 156.8, 152.0, 147.0, 138.1, 137.5, 136.6, 129.0, 128.8, 127.6, 127.4, 127.1, 126.0, 123.7, 121.9, 121.8, 117.7, 110.1, 64.6, 61.8, 54.0, 53.5, 34.0, 25.8, 22.7, 22.5 ppm; ESI-MS m/z: 504.2 [M + Na]+.
- 2-(4-((5-Chloro-8-hydroxyquinolin-2-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (15b). Yield 63%, White solid; 1H NMR (400 MHz, CDCl3) δ = 9.21 (s, 1H), 8.49 (d, J = 8.8 Hz, 1H), 8.00 (d, J = 8.4 Hz, 1H), 7.76–7.70 (m, 2H), 7.63 (t, J = 7.2 Hz, 1H), 7.49–7.45 (m, 2H), 7.09 (d, J = 8.8 Hz, 1H), 3.89 (s, 2H), 3.31 (s, 2H), 3.15 (t, J = 6.4 Hz, 2H), 2.85–2.80 (m, 6H), 2.70 (m, 4H), 2.01–1.97 (m, 2H), 1.88–1.86 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ = 168.6, 159.9, 157.6, 151.2, 146.9, 138.1, 137.9, 133.9, 128.9, 128.8, 127.1, 126.0, 125.5, 123.7, 122.5, 121.8, 120.4, 110.2, 64.3, 61.8, 53.9, 53.5, 34.0, 25.8, 22.7, 22.5 ppm; ESI-MS m/z: 538.3 [M + Na]+.
- 2-(4-((5-Fluoro-8-hydroxyquinolin-2-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (15c). Yield 60%, White solid; 1H NMR (400 MHz, CDCl3) δ 9.21 (s, 1H), 8.38 (d, J = 8.8 Hz, 1H), 8.00 (d, J = 8.4 Hz, 2H), 7.74–7.70 (m, 2H), 7.63 (t, J = 7.6 Hz, 1H), 7.47 (t, J = 7.6 Hz, 1H), 7.13–7.03 (m, 2H), 3.88 (s, 2H), 3.33 (s, 2H), 3.15 (t, J = 6.4 Hz, 2H), 2.86–2.80 (m, 6H), 2.71 (m, 4H), 2.01–1.97 (m, 2H), 1.90–1.86 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 168.6, 159.9, 158.0, 150.7 (JC-F = 244.1 Hz), 148.2 (JC-F = 2.7 Hz), 147.0, 138.0, 137.1, 137.0, 130.3 (JC-F = 3.1 Hz), 129.0, 128.9, 127.1, 125.9, 123.7, 121.8, 121.8 (JC-F = 1.9 Hz), 118.0 (JC-F = 18.7 Hz), 110.4 (JC-F = 20.5 Hz), 108.8 (JC-F = 7.1 Hz), 64.5, 61.8, 54.0, 53.5, 34.0, 25.8, 22.7, 22.5 ppm; ESI-MS m/z: 522.2 [M + Na]+.
- 2-(4-((6-Chloro-8-hydroxyquinolin-2-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (15d). Yield 59%, pale yellow solid; 1H NMR (400 MHz, CDCl3) δ 9.21 (s, 1H), 8.04 (d, J = 8.4 Hz, 1H), 8.00 (d, J = 8.4 Hz, 1H), 7.71–7.761 (m, 3H), 7.49–7.46 (m, 1H), 7.31 (d, J = 2.0 Hz, 1H), 7.14 (d, J = 2.4 Hz, 1H), 3.85 (s, 2H), 3.33 (s, 2H), 3.15 (d, J = 6.4 Hz, 2H), 2.85–2.81 (m, 6H), 2.77–2.69 (m, 4H), 1.99–1.98 (m, 2H), 1.89–1.86 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 168.6, 159.9, 157.1, 152.8, 147.0, 138.1, 136.2, 135.8, 133.1, 129.0, 128.9, 127.9, 127.1, 126.0, 123.7, 122.9, 121.8, 116.5, 111.5, 64.5, 61.8, 54.0, 53.5, 34.0, 25.8, 22.7, 22.5 ppm; ESI-MS m/z: 538.3 [M + Na]+.
- 2-(4-((5,7-Dichloro-8-hydroxyquinolin-2-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (15e). Yield 65%, pale yellow solid; 1H NMR (400 MHz, CDCl3) δ 9.19 (s, 1H), 8.46 (d, J = 8.8 Hz, 1H), 8.00 (d, J = 8.4 Hz, 1H), 7.76 (d, J = 8.8 Hz, 1H), 7.70 (d, J = 8.4 Hz, 1H), 7.63 (t, J = 8.0 Hz, 1H), 7.57 (s, 1H), 7.46 (t, J = 7.6 Hz, 1H), 3.88 (s, 2H), 3.31 (s, 2H), 3.15 (t, J = 6.4 Hz, 2H), 2.83–2.80 (m, 6H), 2.68 (m, 4H), 2.01–1.95 (m, 2H), 1.90–1.86 ppm (m, 2H); 13C NMR (100 MHz, CDCl3,) δ 168.6, 159.9, 158.9, 147.5, 146.9, 138.1, 137.9, 134.1, 129.0, 128.8, 127.9, 127.1, 126.0, 124.2, 123.7, 122.4, 121.8, 120.7, 115.5, 64.2, 61.8, 53.9, 53.5, 34.0, 25.8, 22.7, 22.5 ppm; ESI-MS m/z: 572.2 [M + Na]+.
- 2-(4-((8-Aminoquinolin-2-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (15f). Yield 61%, pale yellow solid; 1H NMR (400 MHz, CDCl3) δ 9.24 (s, 1H), 8.04 (d, J = 8.4 Hz, 1H), 8.00 (d, J = 8.4 Hz, 1H), 7.71 (d, J = 8.0 Hz, 1H), 7.63 (t, J = 7.6 Hz, 1H), 7.57 (d, J = 8.4 Hz, 1H), 7.46 (t, J = 7.6 Hz, 1H), 7.30 (t, J = 7.6 Hz, 1H), 7.13 (d, J = 8.0 Hz, 1H), 6.91 (d, J = 7.2 Hz, 1H), 4.99 (m, 2H), 3.88 (s, 2H), 3.33 (s, 2H), 3.15 (t, J = 6.4 Hz, 2H), 2.85–2.80 (m, 6H), 2.80–2.72 (m, 4H), 2.00–1.94 (m, 2H), 1.89–1.85 ppm (m, 2H); 13C NMR (100 MHz, CDCl3,) δ 168.7, 159.9, 156.0, 147.0, 143.7, 138.1, 137.5, 136.5, 128.9, 128.8, 127.9, 127.1, 125.9, 123.7, 121.9, 121.2, 115.9, 110.2, 65.0, 61.9, 54.1, 53.4, 34.0, 25.8, 22.7, 22.5 ppm; ESI-MS m/z: 503.3 [M + Na]+.
- 2-((4-(2-((1,2,3,4-Tetrahydroacridin-9-yl)amino)ethyl)piperazin-1-yl)methyl)quinolin-8-ol (16a). Yield 55%, pale yellow oil; 1H NMR (400 MHz, CDCl3) δ 8.54 (d, J = 8.4 Hz, 1H), 8.18 (d, J = 8.8 Hz, 1H), 8.14 (d, J = 8.8 Hz, 1H), 7.68 (t, J = 7.2 Hz, 1H), 7.62 (d, J = 8.8 Hz, 1H), 7.46–7.40 (m, 2H), 7.34–7.31 (m, 1H), 7.18–7.16 (m, 1H), 7.00 (m, 1H), 4.05–3.94 (m, 2H), 3.90 (s, 2H), 3.38–3.26 (m, 2H), 2.81 (t, J = 5.2 Hz, 2H), 2.76–2.67 (m, 8H), 2.57 (t, J = 5.2 Hz, 2H), 1.95–1.89 ppm (m, 4H); 13C NMR (100 MHz, CDCl3) δ 155.6, 153.8, 151.0, 150.9, 138.5, 136.4, 135.6, 131.1, 126.5, 126.4, 123.9, 122.9, 120.9, 120.8, 116.6, 115.0, 109.9, 109.1, 63.6, 55.0, 52.5, 51.3, 42.5, 27.6, 22.5, 21.0, 19.9 ppm; ESI-MS m/z: 468.3 [M + H]+.
- 5-Fluoro-2-((4-(2-((1,2,3,4-tetrahydroacridin-9-yl)amino)ethyl)piperazin-1-yl)methyl)quinolin-8-ol (16b). Yield 51%, pale yellow oil; 1H NMR (400 MHz, CDCl3) δ 8.36 (d, J = 8.4 Hz, 1H), 8.04 (d, J = 8.0 Hz, 1H), 7.99 (d, J = 8.4 Hz, 1H), 7.70 (d, J = 8.4 Hz, 1H), 7.58–7.54 (m, 1H), 7.36–7.32 (m, 1H), 7.13–7.04 (m, 2H), 5.39 (m, 1H), 3.88 (s, 2H), 3.61 (t, J = 4.8 Hz, 2H), 3.15–3.05 (m, 2H), 2.80–2.75 (m, 2H), 2.64–2.59 (m, 10H), 1.93–1.91 ppm (m, 4H); 13C NMR (100 MHz, CDCl3) δ 158.1, 157.7, 151.5, 150.7 (JC-F = 244.3 Hz), 149.5, 148.3 (JC-F = 3.2 Hz), 146.4, 137.1 (JC-F = 2.6 Hz), 130.3 (JC-F = 3.5 Hz), 128.8, 123.7, 122.9, 121.8 (JC-F = 2.2 Hz), 119.8, 117.9 (JC-F = 15.8 Hz), 115.4, 110.4 (JC-F = 20.1 Hz), 108.8 (JC-F = 7.7 Hz), 64.7, 57.4, 53.6, 52.6, 45.0, 33.3, 24.8, 23.0, 22.6 ppm; ESI-MS m/z: 486.3 [M + H]+.
- 5,7-Dichloro-2-((4-(2-((1,2,3,4-tetrahydroacridin-9-yl)amino)ethyl)piperazin-1-yl)methyl)quinolin-8-ol (16c). Yield 56%, pale yellow oil; 1H NMR (400 MHz, CDCl3) δ 8.45–8.41 (m, 1H), 8.04–7.99 (m, 2H), 7.74–7.70 (m, 1H), 7.58–7.55 (m, 2H), 7.36–7.32 (m, 1H), 5.47 (m, 1H), 3.87–3.85 (m, 2H), 3.70–3.55 (m, 2H), 3.17–3.03 (m, 2H), 2.80–2.73 (m, 2H), 2.64–2.63 (m, 10H), 1.98–1.82 ppm (m, 4H); 13C NMR (100 MHz, CDCl3) δ 158.9, 157.2, 151.8, 147.8, 145.9, 138.1, 133.9, 129.0, 127.8, 127.4, 124.1, 123.8, 123.0, 122.5, 120.4, 119.5, 115.5, 115.1, 64.3, 57.3, 53.6, 52.5, 44.9, 32.9, 24.7, 23.0, 22.5 ppm; ESI-MS m/z: 536.2 [M + H]+.
- N-(2-(4-((8-aminoquinolin-2-yl)methyl)piperazin-1-yl)ethyl)-1,2,3,4-tetrahydroacridin-9-amine (16d). Yield 49%, colorless oil; 1H NMR (400 MHz, CDCl3) δ 8.48 (d, J = 8.4 Hz, 1H), 8.10 (d, J = 8.8 Hz, 1H), 7.97 (d, J = 8.4 Hz, 1H), 7.62–7.58 (m, 1H), 7.48 (d, J = 8.4 Hz, 1H), 7.36–7.32 (m, 1H), 7.23 (t, J = 8.0 Hz, 1H), 7.08–7.05 (m, 1H), 7.02 (m, 1H), 6.87–6.85 (m, 1H), 3.92 (t, J = 5.2 Hz, 2H), 3.83 (s, 2H), 3.24 (t, J = 5.2 Hz, 2H), 2.74 (t, J = 5.6 Hz, 2H), 2.70–2.52 (m, 8H), 2.48 (t, J = 6.0 Hz, 2H), 1.85–1.78 ppm (m, 4H); 13C NMR (100 MHz, CDCl3) δ 155.5, 155.0, 151.6, 143.7, 139.3, 137.5, 136.4, 132.2, 127.8, 127.2, 125.0, 123.9, 121.5, 121.3, 115.9, 115.8, 110.8, 110.2, 64.9, 56.0, 53.4, 52.3, 43.5, 28.4, 23.5, 22.0, 20.8 ppm; ESI-MS m/z: 467.3 [M + H]+.
- 2-((4-(3-((1,2,3,4-Tetrahydroacridin-9-yl)amino)propyl)piperazin-1-yl)methyl)quinolin-8-ol (16e). Yield 53%, colorless oil; 1H NMR (400 MHz, CDCl3) δ 8.39 (d, J = 8.4 Hz, 1H), 8.15 (d, J = 8.4 Hz, 1H), 8.06 (d, J = 8.4 Hz, 1H), 7.69 (m, 1H), 7.57–7.52 (m, 2H), 7.35 (t, J = 8.0 Hz, 1H), 7.30–7.23 (m, 2H), 7.10–7.08 (m, 1H), 4.00 (t, J = 5.2 Hz, 2H), 3.81 (s, 2H), 3.26–3.12 (m, 2H), 2.65–2.60 (m, 10H), 2.51–2.50 (m, 2H), 1.90 (t, J = 4.8 Hz, 2H), 1.77–1.76 ppm (m, 4H); 13C NMR (100 MHz, CDCl3) δ 156.4, 155.4, 152.0, 151.4, 139.6, 137.5, 136.6, 131.8, 127.6, 127.5, 124.6, 124.4, 121.9, 121.5, 117.7, 116.1, 110.9, 110.2, 64.7, 58.3, 53.9, 52.7, 50.0, 28.7, 25.6, 25.4, 21.8, 20.9 ppm; ESI-MS m/z: 482.3 [M + H]+.
- 5-Fluoro-2-((4-(3-((1,2,3,4-tetrahydroacridin-9-yl)amino)propyl)piperazin-1-yl)methyl)quinolin-8-ol (16f). Yield 59%, pale yellow oil; 1H NMR (400 MHz, CDCl3) δ 8.42 (d, J = 8.4 Hz, 1H), 8.30 (d, J = 8.4 Hz, 1H), 8.16 (d, J = 8.8 Hz, 1H), 7.74 (s, 1H), 7.62–7.56 (m, 2H), 7.31 (t, J = 8.0 Hz, 1H), 7.06–6.97 (m, 2H), 4.03 (t, J = 5.6 Hz, 2H), 3.83 (s, 2H), 3.22 (t, J = 5.6 Hz, 2H), 2.68–2.62 (m, 10H), 2.53 (t, J = 5.6 Hz, 2H), 1.92 (t, J = 4.8 Hz, 2H), 1.81–1.78 ppm (m, 4H); 13C NMR (100 MHz, CDCl3) δ 157.6, 155.5, 151.4, 150.7 (JC-F = 244.7 Hz), 148.2 (JC-F = 3.3 Hz), 139.5, 137.1 (JC-F = 3.3 Hz), 132.0, 130.4 (JC-F = 1.8 Hz), 124.7, 124.4, 121.8 (JC-F = 3.0 Hz), 121.5, 117.9 (JC-F = 18.8 Hz), 116.0, 110.8, 110.5 (JC-F = 21.4 Hz), 108.9 (JC-F = 7.4 Hz), 64.7, 58.4, 53.9, 52.7, 50.2, 29.7, 25.6, 25.3, 21.8, 20.8 ppm; ESI-MS m/z: 500.3 [M + H]+.
- N-(3-(4-((8-aminoquinolin-2-yl)methyl)piperazin-1-yl)propyl)-1,2,3,4-tetrahydroacridin-9-amine (16g). Yield 61%, colorless oil; 1H NMR (400 MHz, CDCl3) δ 8.12 (d, J = 8.4 Hz, 1H), 8.04 (d, J = 8.4 Hz, 1H), 7.93 (d, J = 8.4 Hz, 1H), 7.48–7.43 (m, 2H), 7.24–7.17 (m, 2H), 7.02 (d, J = 8.0 Hz, 1H), 6.82 (d, J = 7.6 Hz, 1H), 6.72 (m, 1H), 4.97 (m, 2H), 3.77–3.72 (m, 4H), 3.13–3.07 (m, 2H), 2.68–2.35 (m, 12H), 1.82–1.79 (m, 2H), 1.77–1.73 ppm (m, 4H); 13C NMR (100 MHz, CDCl3) δ 155.7, 154.1, 153.7, 143.8, 142.7, 137.5, 136.3, 130.3, 127.8, 127.0, 124.2, 124.1, 124.0, 121.3, 117.7, 115.7, 112.8, 110.2, 65.1, 58.1, 53.8, 52.9, 49.6, 30.8, 26.1, 25.6, 22.2, 21.6 ppm; ESI-MS m/z: 481.3 [M + H]+.
- 2-((4-(5-((1,2,3,4-Tetrahydroacridin-9-yl)amino)pentyl)piperazin-1-yl)methyl)quinolin-8-ol (16h). Yield 55%, pale yellow oil; 1H NMR (400 MHz, CD3OD) δ 8.39 (d, J = 8.4 Hz, 1H), 8.22 (d, J = 8.8 Hz, 1H), 7.86–7.76 (m, 2H), 7.60–7.56 (m, 2H), 7.43–7.33 (m, 2H), 7.10–7.08 (m, 1H), 3.99–3.95 (m, 4H), 3.33–3.30 (m, 2H), 3.01–2.81 (m, 10H), 2.71–2.69 (m, 2H), 1.96–1.87 (m, 6H), 1.74–1.70 (m, 2H), 1.51–1.47 ppm (m, 2H); 13C NMR (100 MHz, CD3OD) δ 156.6, 155.3, 152.8, 150.5, 138.4, 137.9, 136.7, 132.7, 128.2, 127.2, 125.1, 125.0, 121.5, 118.8, 117.5, 115.8, 111.6, 110.8, 63.0, 57.0, 51.8, 50.9, 48.4, 29.7, 28.0, 24.3, 23.7, 23.6, 21.6, 20.5 ppm; ESI-MS m/z: 510.3 [M + H]+.
- 5-Fluoro-2-((4-(5-((1,2,3,4-tetrahydroacridin-9-yl)amino)pentyl)piperazin-1-yl)methyl)quinolin-8-ol (16i). Yield 57%, pale yellow oil; 1H NMR (400 MHz, CDCl3) δ 8.31 (d, J = 8.0 Hz, 1H), 8.26 (d, J = 8.8 Hz, 1H), 8.14 (d, J = 8.8 Hz, 1H), 7.58–7.52 (m, 2H), 7.34 (t, J = 8.0 Hz, 1H), 7.03–6.93 (m, 2H), 6.17 (m, 1H), 3.85 (t, J = 6.8 Hz, 2H), 3.81 (s, 2H), 3.23–3.10 (m, 2H), 2.72–2.57 (m, 12H), 1.82–1.77 (m, 6H), 1.66–1.61 (m, 2H), 1.46–1.42 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 156.3, 154.3, 150.8, 149.6 (JC-F = 244.1 Hz), 147.5 (JC-F = 3.1 Hz), 138.3, 136.2 (JC-F = 3.3 Hz), 131.0, 129.3 (JC-F = 2.7 Hz), 124.0, 123.2, 120.7 (JC-F = 2.2 Hz), 120.3, 117.0 (JC-F = 18.7 Hz), 115.1, 110.2, 109.5 (JC-F = 21.0 Hz), 108.0 (JC-F = 7.7 Hz), 63.1, 56.5, 51.6, 51.0, 47.0, 29.5, 27.8, 24.2, 23.2, 23.0, 21.0, 19.8 ppm; ESI-MS m/z: 528.3 [M + H]+.
- 5-Chloro-2-((4-(5-((1,2,3,4-tetrahydroacridin-9-yl)amino)pentyl)piperazin-1-yl)methyl)quinolin-8-ol (16j). Yield 58%, pale yellow oil; 1H NMR (400 MHz, CDCl3) δ 8.43 (d, J = 8.8 Hz, 1H), 8.38 (d, J = 8.8 Hz, 1H), 8.23 (d, J = 8.8 Hz, 1H), 7.68–7.60 (m, 2H), 7.45–7.38 (m, 2H), 7.05 (d, J = 8.4 Hz, 1H), 6.38 (m, 1H), 3.97–3.90 (m, 2H), 3.86 (s, 2H), 3.24–3.23 (m, 2H), 2.68–2.66 (m, 10H), 2.52 (t, J = 6.8 Hz, 2H), 1.89–1.86 (m, 6H), 1.64–1.62 (m, 2H), 1.50–1.47 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 157.4, 155.1, 151.9, 151.4, 139.7, 138.0, 133.8, 131.7, 127.0, 125.4, 124.9, 124.3, 122.5, 121.5, 120.2, 116.4, 111.5, 110.3, 64.1, 57.8, 52.8, 52.6, 48.1, 30.7, 29.1, 25.7, 24.4, 24.2, 22.1, 20.9 ppm; ESI-MS m/z: 544.3 [M + H]+.
- N-(5-(4-((8-aminoquinolin-2-yl)methyl)piperazin-1-yl)pentyl)-1,2,3,4-tetrahydroacridin-9-amine (16k). Yield 62%, colorless oil; 1H NMR (400 MHz, CDCl3) δ 8.30–8.25 (m, 1H), 8.11 (d, J = 8.8 Hz, 1H), 7.93–7.91 (m, 1H), 7.55–7.51 (m, 1H), 7.45–7.42 (m, 1H), 7.33–7.29 (m, 1H), 7.19–7.17 (m, 1H), 7.03–7.01 (m, 1H), 6.84–6.82 (m, 1H), 6.01 (m, 1H), 4.96 (m, 2H), 3.77–3.74 (m, 4H), 3.14–3.13 (m, 2H), 2.58–2.34 (m, 12H), 1.78–1.76 (m, 6H), 1.54–1.49 (m, 2H), 1.41–1.38 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 155.9, 154.6, 152.6, 143.8, 140.5, 137.5, 136.3, 131.4, 127.8, 127.0, 124.8, 124.1, 122.3, 121.3, 116.7, 115.7, 111.9, 110.1, 64.9, 58.0, 53.1, 52.8, 48.3, 30.9, 29.5, 26.0, 24.5, 24.2, 22.2, 21.1 ppm; ESI-MS m/z: 509.3 [M + H]+.
3.1.2. General Procedures for the Preparation of Compounds 17
- 2-(4-((8-Hydroxyquinolin-5-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (17a). Yield 92%, pale yellow solid; 1H NMR (400 MHz, CDCl3) δ 9.26 (s, 1H), 8.80 (d, J = 3.2 Hz, 1H), 8.67 (d, J = 8.0 Hz, 1H), 8.01 (d, J = 8.4 Hz, 1H), 7.72 (d, J = 8.0 Hz, 1H), 7.64 (t, J = 7.2 Hz, 1H), 7.49–7.46 (m, 2H), 7.33 (d, J = 7.6 Hz, 1H), 7.08 (d, J = 8.0 Hz, 1H), 3.85 (s, 2H), 3.30 (s, 2H), 3.15 (t, J = 6.4 Hz, 2H), 2.83–2.77 (m, 6H), 2.62 (m, 4H), 2.00–1.97 (m, 2H), 1.89–1.86 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 168.7, 160.0, 152.0, 147.7, 146.9, 138.8, 138.1, 134.0, 129.0, 128.95, 128.8, 127.9, 127.1, 125.9, 124.2, 123.7, 121.9, 121.5, 108.6, 61.8, 60.6, 54.1, 53.1, 34.0, 25.8, 22.7, 22.5 ppm; ESI-MS m/z: 504.3 [M + Na]+.
- 2-(4-((8-Hydroxy-2-methylquinolin-5-yl)methyl)piperazin-1-yl)-N-(1,2,3,4-tetrahydroacridin-9-yl)acetamide (17b). Yield 93%, pale yellow solid; 1H NMR (400 MHz, CDCl3) δ 9.26 (s, 1H), 8.53 (d, J = 8.4 Hz, 1H), 8.00 (d, J = 8.0 Hz, 1H), 7.71 (d, J = 8.4 Hz, 1H), 7.65–7.61 (m, 1H), 7.49–7.45 (m, 1H), 7.34 (d, J = 8.8 Hz, 1H), 7.25 (d, J = 7.6 Hz, 1H), 7.03 (d, J = 7.6 Hz, 1H), 3.82 (s, 2H), 3.29 (s, 2H), 3.15 (t, J = 6.4 Hz, 2H), 2.82 (t, J = 6.4 Hz, 2H), 2.76 (m, 4H), 2.73 (s, 3H), 2.60 (m, 4H), 2.00–1.97 (m, 2H), 1.89–1.86 ppm (m, 2H); 13C NMR (100 MHz, CDCl3) δ 168.7, 160.0, 156.6, 151.5, 147.0, 138.13, 138.11, 134.1, 129.0, 128.8, 128.0, 127.1, 125.9, 123.9, 123.7, 122.4, 121.9, 108.5, 61.8, 60.6, 54.1, 53.1, 34.0, 25.8, 24.9, 22.7, 22.6 ppm; ESI-MS m/z: 518.3 [M + Na]+.
3.2. Biological Evaluation
3.2.1. Inhibition Experiments of ChEs
3.2.2. Kinetic Analysis of AChE Inhibition
3.2.3. Inhibition of Self-Induced Aβ1–42 Aggregation
3.2.4. Evaluation of Metal-Chelating Properties
3.2.5. Molecular Docking Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kumar, A.; Singh, A.; Ekavali. A review on Alzheimer’s disease pathophysiology and its management: An update. Pharmacol. Rep. 2015, 67, 195–203. [Google Scholar] [CrossRef]
- Dickson, D.W.; Crystal, H.A.; Mattiace, L.A.; Masur, D.M.; Blau, A.D.; Davies, P. Identification of normal and pathological aging in prospectively studied nondemented elderly humans. Neurobiol. Aging 1992, 13, 179–189. [Google Scholar] [CrossRef]
- Iqbal, K.; Flory, M.; Khatoon, S.; Soininen, H.; Pirttila, T.; Lehtovirta, M. Subgroups of Alzheimer’s disease based on cerebrospinal fluid molecular markers. Ann. Neurol. 2005, 58, 748–757. [Google Scholar] [CrossRef]
- Abeysinghe, A.A.D.T.; Deshapriya, R.D.U.S.; Udawatte, C. Alzheimer’s disease; a review of the pathophysiological basis and therapeutic interventions. Life Sci. 2020, 256, 117996. [Google Scholar] [CrossRef]
- Uddin, M.S.; Kabir, M.T.; Tewari, D.; Mamun, A.A.; Mathew, B.; Aleya, L.; Barreto, G.E.; Bin-Jumah, M.N.; Abdel-Daim, M.M.; Ashraf, G.M. Revisiting the role of brain and peripheral Aβ in the pathogenesis of Alzheimer’s disease. J. Neurol. Sci. 2020, 416, 116974. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Sharma, A.; Fayaz, F.; Wakode, S.; Pottoo, F.H. Biological signatures of Alzheimer’s disease. Curr. Top. Med. Chem. 2020, 20, 770–781. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, L.W.; Mohamed, K.O.; Sayed, H.S.; Mahmoud, Z. Recent modifications of anti-dementia agents focusing on tacrine and/or donepezil analogs. Med. Chem. 2023, 19, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Goedert, M.; Spillantini, M.G. A century of Alzheimer’s disease. Science 2006, 314, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Alzheimer’s Disease International. World Alzheimer Report 2021: Journey Through the Diagnosis of Dementia; Alzheimer’s Disease International: London, UK, 2021. [Google Scholar]
- Miguel, M.; Avila, J. New perspectives on the role of tau in Alzheimer’s disease: Implications for therapy. Biochem. Pharmacol. 2014, 88, 540–547. [Google Scholar]
- Ayton, S.; Lei, P.; Bush, A.I. Metallostasis in Alzheimer’s disease. Free Radic. Biol. Med. 2013, 62, 76–89. [Google Scholar] [CrossRef]
- Iqbal, K.; Liu, F.; Gong, C.X. Alzheimer disease therapeutics: Focus on the disease and not just plaques and tangles. Biochem. Pharmacol. 2014, 88, 631–639. [Google Scholar] [CrossRef]
- Sweeney, P.; Park, H.; Baumann, M.; Dunlop, J.; Frydman, J.; Kopito, R.; McCampbell, A.; Leblanc, G.; Venkateswaran, A.; Nurimi, A.; et al. Protein misfolding in neurodegenerative diseases: Implications and strategies. Transl. Neurodegener. 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Budimir, A. Metal ions, Alzheimer’s disease and chelation therapy. Acta Pharm. 2011, 61, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Terry, R.D.; Masliah, E.; Salmon, D.P.; Butters, N.; DeTeresa, R.; Hill, R.; Hansen, L.A.; Katzman, R. Physical basis of cognitive alterations in alzheimer’s disease: Synapse loss is the major correlate of cognitive impairment. Ann. Neurol. 1991, 30, 572–580. [Google Scholar] [CrossRef]
- Sharma, K. Cholinesterase inhibitors as Alzheimer’s therapeutics (review). Mol. Med. Rep. 2019, 20, 1479–1487. [Google Scholar] [CrossRef]
- Augustinsson, K.B.; Nachmansohn, D. Distinction between acetylcholine-esterase and other choline ester-splitting enzymes. Science 1949, 110, 98–99. [Google Scholar] [CrossRef]
- Xu, M.L.; Luk, W.K.W.; Bi, C.W.C.; Liu, E.Y.L.; Wu, K.Q.Y.; Yao, P.; Dong, T.T.X.; Tsim, K.W.K. Erythropoietin regulates the expression of dimeric form of acetylcholinesterase during differentiation of erythroblast. J. Neurochem. 2018, 146, 390–402. [Google Scholar] [CrossRef]
- Selim, M.S.; Azzam, A.M.; Shenashen, M.A.; Higazy, S.A.; Mostafa, B.B.; El-Safty, S.A. Comparative study between three carbonaceous nanoblades and nanodarts for antimicrobial applications. J. Environ. Sci. 2024, 136, 594–605. [Google Scholar] [CrossRef]
- Li, X.; Jia, Y.; Li, J.; Zhang, P.; Li, T.; Lu, L.; Yao, H.; Liu, J.; Zhu, Z.; Xu, J. Novel and Potent Acetylcholinesterase inhibitors for the treatment of Alzheimer’s disease from natural (±)-7,8-dihydroxy-3-methyl-isochroman-4-one. Molecules 2022, 27, 3090. [Google Scholar] [CrossRef] [PubMed]
- Ghanei-Nasab, S.; Khoobi, M.; Hadizadeh, F.; Marjani, A.; Moradi, A.; Nadri, H.; Emami, S.; Foroumadi, A.; Shafiee, A. Synthesis and anticholinesterase activity of coumarin-3-carboxamides bearing tryptamine moiety. Eur. J. Med. Chem. 2016, 121, 40–46. [Google Scholar] [CrossRef]
- Mantoani, S.P.; Chierrito, T.P.C.; Vilela, A.F.L.; Cardoso, C.L.; Martínez, A.; Carvalho, I. Novel triazole-quinoline derivatives as selective dual binding site acetylcholinesterase inhibitors. Molecules 2016, 21, 193. [Google Scholar] [CrossRef]
- Kumar, S.; Mitra1, R.; Ayyannan, S.R. Design, synthesis and evaluation of benzothiazole-derived phenyl thioacetamides as dual inhibitors of monoamine oxidases and cholinesterases. Mol. Divers. 2024, 1–23. [Google Scholar] [CrossRef]
- Mesulam, M.M.; Geula, C. Acetylcholinesterase-rich neurons of the human cerebral cortex: Cytoarchitectonic and ontogenetic patterns of distribution. J. Comp. Neurol. 1991, 306, 193–220. [Google Scholar] [CrossRef]
- Zemek, F.; Drtinova, L.; Nepovimova, E.; Sepsova, V.; Korabecny, J.; Klimes, J.; Kuca, K. Outcomes of Alzheimer’s disease therapy with acetylcholinesterase inhibitors and memantine. Expert Opin. Drug Saf. 2014, 13, 759–774. [Google Scholar]
- Novak, M.; Vajrychova, M.; Koutsilieri, S.; Sismanoglou, D.C.; Kobrlova, T.; Prchal, L.; Svobodova, B.; Korabecny, J.; Zarybnicky, T.; Raisova-Stuchlikova, L.; et al. Tacrine first-phase biotransformation and associated hepatotoxicity: A possible way to avoid Quinone Methide formation. ACS Chem. Biol. 2023, 18, 1993–2002. [Google Scholar] [CrossRef]
- Fan, F.; Liu, H.; Shi, X.; Ai, Y.; Liu, Q.; Cheng, Y. The efficacy and safety of Alzheimer’s disease therapies: An updated umbrella review. J. Alzheimer’s Dis. 2022, 85, 1195–1204. [Google Scholar] [CrossRef]
- Pourtaher, H.; Mohammadi, Y.; Hasaninejad, A.; Iraji, A. Highly efficient, catalyst-free, one-pot sequential four-component synthesis of novel spiroindolinone-pyrazole scaffolds as anti-Alzheimer agents: In silico study and biological screening. RSC Med. Chem. 2024, 15, 207–222. [Google Scholar] [CrossRef]
- Darvesh, S.; Cash, M.K.; Reid, G.A.; Martin, E.; Mitnitski, A.; Geula, C. Butyrylcholinesterase is associated with β-amyloid plaques in the transgenic APPSWE/PSEN1dE9 mouse model of Alzheimer disease. J. Neuropathol. Exp. Neurol. 2012, 71, 2–14. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.; Martínez, A. Targeting Beta-Amyloid Pathogenesis Through Acetylcholinesterase Inhibitors. Curr. Pharm. Des. 2006, 12, 4377–4387. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.B.; Mu, Y.; Lei, H.; Wang, P.; Li, X.; Leng, Q.; Han, L.; Qu, X.D.; Wang, Z.Y.; Huang, X.S. Design, synthesis and evaluation of novel tacrine-ferulic acid hybrids as multifunctional drug candidates against Alzheimer’s disease. Molecules 2016, 21, 1338. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Y.; Wang, X.B.; Xie, S.S.; Jiang, N.; Wang, K.D.G.; Yao, H.Q.; Sun, H.B.; Kong, L.Y. Multifunctional tacrine–flavonoid hybrids with cholinergic, β-amyloid-reducing, and metal chelating properties for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2013, 69, 632–646. [Google Scholar] [CrossRef]
- Huang, W.H.; Wei, W.H.; Shen, Z. Drug-like chelating agents: A potential lead for Alzheimer’s disease. RSC Adv. 2014, 4, 52088–52099. [Google Scholar] [CrossRef]
- Guilloreau, L.; Combalbert, S.; Sournia-Saquet, A.; Mazarguil, H.; Faller, P. Redox chemistry of copper–amyloid-β: The generation of hydroxyl radical in the presence of ascorbate is linked to redox-potentials and aggregation state. ChemBioChem 2007, 8, 1317–1325. [Google Scholar] [CrossRef]
- Ha, C.; Ryu, J.; Park, C.B. Metal ions differentially influence the aggregation and deposition of Alzheimer’s β-amyloid on a solid template. Biochemistry 2007, 46, 6118–6125. [Google Scholar] [CrossRef]
- Amélia Santos, M.; Chand, K.; Chaves, S. Recent progress in multifunctional metal chelators as potential drugs for Alzheimer’s disease. Coordin. Chem. Rev. 2016, 327–328, 287–303. [Google Scholar] [CrossRef]
- Cuajungco, M.P.; Fagét, K.Y.; Huang, X.D.; Tanzi, R.E.; Bush, A.I. Metal chelation as a potential therapy for Alzheimer’s disease. Ann. N. Y. Acad. Sci. 2000, 920, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Sales, T.A.; Prandi, I.G.; de Castro, A.A.; Leal, D.H.S.; da Cunha, E.F.F.; Kuca, K.; Ramalho, T.C. Recent developments in metal-based drugs and chelating agents for neurodegenerative diseases treatments. Int. J. Mol. Sci. 2019, 20, 1829. [Google Scholar] [CrossRef]
- Zhang, P.F.; Xu, S.T.; Zhu, Z.Y.; Xu, J.Y. Multi-target design strategies for the improved treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2018, 158, 463–477. [Google Scholar]
- Savelieff, M.G.; Nam, G.; Kang, J.; Lee, H.J.; Lee, M.; Lim, M.H. Development of multifunctional molecules as potential therapeutic candidates for alzheimer’s disease, parkinson’s disease, and amyotrophic lateral sclerosis in the last decade. Chem. Rev. 2019, 119, 1221–1322. [Google Scholar] [CrossRef] [PubMed]
- Guzior, N.; Wieckowska, A.; Panek, D.; Malawska, B. Recent Development of multifunctional agents as potential drug candidates for the treatment of Alzheimer’s disease. Curr. Med. Chem. 2015, 22, 373–404. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.; Robert, A.; Sournia-Saquet, A.; Vendier, L.; Meunier, B. Characterization of new specific copper chelators as potential drugs for the treatment of Alzheimer’s disease. Chem. Eur. J. 2014, 20, 6771–6785. [Google Scholar] [CrossRef]
- Fernandez-Bachiller, M.I.; Perez, C.; Gonzalez-Munoz, G.C.; Conde, S.; Lopez, M.G.; Villarroya, M.; Garcia, A.G.; Rodriguez-Franco, M.I. Novel Tacrine−8-Hydroxyquinoline Hybrids as Multifunctional Agents for the Treatment of Alzheimer’s Disease, with Neuroprotective, Cholinergic, Antioxidant, and Copper-Complexing Properties. J. Med. Chem. 2010, 53, 4927–4937. [Google Scholar] [CrossRef]
- Xie, S.S.; Wang, X.B.; Jiang, N.; Yu, W.Y.; Wang, K.D.G.; Lan, J.S.; Li, Z.R.; Kong, L.Y. Multi-target tacrine-coumarin hybrids: Cholinesterase and monoamine oxidase B inhibition properties against Alzheimer’s disease. Eur. J. Med. Chem. 2015, 95, 153–165. [Google Scholar] [CrossRef]
- Xie, S.S.; Wang, X.B.; Li, J.Y.; Yang, L.; Kong, L.Y. Design, synthesis and evaluation of novel tacrine–coumarin hybrids as multifunctional cholinesterase inhibitors against Alzheimer’s disease. Eur. J. Med. Chem. 2013, 64, 540–553. [Google Scholar] [CrossRef]
- Blanc, G. Preparation of aromatic chloromethylenic derivatives. Bull. Soc. Chim. Fr. 1923, 33, 313. [Google Scholar]
- Biancalana, M.; Koide, S. Molecular mechanism of Thioflavin-T binding to amyloid fibrils. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2010, 18, 1405–1412. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–90. [Google Scholar] [CrossRef]
- Huang, C.Y. [27] Determination of binding stoichiometry by the continuous variation method: The job plot. Method Enzymol. 1982, 87, 509–525. [Google Scholar]
- Ferrada, E.; Arancibia, V.; Loeb, B.; Norambuena, E.; Olea-Azar, C.; Huidobro-Toro, J.P. Stoichiometry and conditional stability constants of Cu(II) or Zn(II) clioquinol complexes; implications for Alzheimer’s and Huntington’s disease therapy. Neurotoxicology 2007, 28, 445–449. [Google Scholar] [CrossRef] [PubMed]



 | |||||
| Compd. | X, R, n | IC50 (μM) a | Selectivity Index b | Aβ Aggregation Inhibition (%) c | |
|---|---|---|---|---|---|
| AChE Inhibition | BuChE Inhibition | ||||
| 15a | X = O, R = H | 2.02 ± 0.12 | 7.74 ± 0.89 | 0.26 | 46.8 ± 4.1 |
| 15b | X = O, R = 5-chloro | 2.18 ± 0.17 | 11.45 ± 1.56 | 0.19 | n.d |
| 15c | X = O, R = 6-chloro | 1.51 ± 0.31 | 11.98 ± 1.07 | 0.13 | n.d |
| 15d | X = O, R = 5-fluoro | 1.18 ± 0.10 | 8.41 ± 1.41 | 0.14 | 88.7 ± 5.2 |
| 15e | X = O, R = 5,7-dichloro | 5.73 ± 0.17 | 5.53 ± 0.27 | 1.04 | 55.9 ± 2.5 |
| 15f | X = NH, R = H | 1.24 ± 0.21 | 7.85 ± 0.71 | 0.16 | 22.0 ± 1.8 |
| 16a | X = O, R = H, n = 1 | 0.44 ± 0.006 | 0.27 ± 0.004 | 1.63 | 77.0 ± 4.3 |
| 16b | X = O, R = 5-fluoro, n = 1 | 0.19 ± 0.005 | 0.20 ± 0.003 | 0.95 | 22.9 ± 2.4 |
| 16c | X = O, R = 5,7-dichloro, n = 1 | 2.68 ± 0.30 | 3.41 ± 0.006 | 0.79 | 92.5 ± 3.3 |
| 16d | X = NH, R = H, n = 1 | 0.18 ± 0.004 | 0.21 ± 0.003 | 0.86 | 84.7 ± 3.6 |
| 16e | X = O, R = H, n = 2 | 0.10 ± 0.0032 | 0.043 ± 0.005 | 2.33 | 80.5 ± 4.4 |
| 16f | X = O, R = 5-fluoro, n = 2 | 0.087 ± 0.002 | 0.028 ± 0.002 | 3.11 | 54.4 ± 2.9 |
| 16g | X = NH, R = H, n = 2 | 0.052 ± 0.001 | 0.018 ± 0.003 | 2.89 | 94.6 ± 4.2 |
| 16h | X = O, R = H, n = 4 | 0.21 ± 0.003 | 0.10 ± 0.01 | 2.10 | 93.2 ± 3.9 |
| 16i | X = O, R = 5-fluoro, n = 4 | 0.40 ± 0.006 | 0.12 ± 0.02 | 3.33 | 55.6 ± 5.3 |
| 16j | X = O, R = 5-chloro, n = 4 | 0.13 ± 0.02 | 0.045 ± 0.011 | 2.89 | 74.8 ± 4.7 |
| 16k | X = NH, R = H, n = 4 | 0.21 ± 0.003 | 0.054 ± 0.004 | 3.89 | 86.7 ± 3.8 |
| 17a | R = H | 4.46 ± 0.33 | 4.13 ± 0.10 | 1.08 | 24.6 ± 3.7 |
| 17b | R = Me | 2.53 ± 0.20 | 4.39 ± 0.27 | 0.58 | 23.3 ± 3.2 |
| tacrine | 0.17 ± 0.04 | 0.029 ± 0.005 | 5.86 | – | |
| curcumin | – | – | 58.3 ± 2.5 | ||
| Comp. | LogK | |
|---|---|---|
| Cu | Zn | |
| 15a | 10.14 | 9.91 |
| 15b | 10.09 | 11.34 |
| 15c | 10.05 | 9.64 |
| 15d | 10.08 | 9.81 |
| 15e | 9.59 | 9.65 |
| 15f | - | - |
| 16a | 9.63 | 9.48 |
| 16b | 9.99 | 9.63 |
| 16c | 9.75 | 9.28 |
| 16d | - | - |
| 16e | 9.46 | 9.63 |
| 16f | 9.62 | 9.45 |
| 16g | - | - |
| 16h | 9.87 | 9.63 |
| 16i | 9.49 | 9.49 |
| 16j | 9.96 | 9.65 |
| 16k | - | - |
| 17a | 9.50 | 9.67 |
| 17b | 9.42 | 9.71 |
| clioquinol | 9.86 (10.08) a | 8.38 (8.84) a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Ma, M.; Feng, Y.; Liu, J.; Wang, G. Multifunctional Tacrine–Quinoline Hybrids as Cholinesterase Inhibitors, Aβ Aggregation Blockers, and Metal Chelators for Alzheimer’s Therapy. Molecules 2025, 30, 3489. https://doi.org/10.3390/molecules30173489
Wang X, Ma M, Feng Y, Liu J, Wang G. Multifunctional Tacrine–Quinoline Hybrids as Cholinesterase Inhibitors, Aβ Aggregation Blockers, and Metal Chelators for Alzheimer’s Therapy. Molecules. 2025; 30(17):3489. https://doi.org/10.3390/molecules30173489
Chicago/Turabian StyleWang, Xiaohua, Minglan Ma, Yalan Feng, Jian Liu, and Gang Wang. 2025. "Multifunctional Tacrine–Quinoline Hybrids as Cholinesterase Inhibitors, Aβ Aggregation Blockers, and Metal Chelators for Alzheimer’s Therapy" Molecules 30, no. 17: 3489. https://doi.org/10.3390/molecules30173489
APA StyleWang, X., Ma, M., Feng, Y., Liu, J., & Wang, G. (2025). Multifunctional Tacrine–Quinoline Hybrids as Cholinesterase Inhibitors, Aβ Aggregation Blockers, and Metal Chelators for Alzheimer’s Therapy. Molecules, 30(17), 3489. https://doi.org/10.3390/molecules30173489






