Targeting Inflammation with Natural Products: A Mechanistic Review of Iridoids from Bulgarian Medicinal Plants
Abstract
1. Introduction
2. Bulgarian Species Rich in Iridoids: Phytochemistry and Therapeutic Potential
2.1. Veronica officinalis
2.2. Plantago spp.
2.3. Verbena officinalis
2.4. Scrophularia nodosa
2.5. Verbascum phlomoides
2.6. Galium spp.
2.7. Nepeta cataria L.
2.8. Lamium album L.
2.9. Euphrasia officinalis
3. Iridoids as Modulators of Inflammation: Evidence and Mechanistic Insights
3.1. Antioxidant Properties
3.2. NF-κB Inhibition
3.3. Modulation of the AMPK and Other Pathways Related to Metabolic Homeostasis
3.4. Antiapoptotic Activity
3.5. Immunomodulatory Activity
4. Current Limitations and Future Perspectives for Iridoid-Based Therapeutics
4.1. Safety Profile and Genotoxicity
4.2. Pharmacokinetics and Bioavailability
4.3. Chemical Modifications and Technological Approaches to Improve Iridoid Therapeutic Potential
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef]
- Phillips, C.M.; Chen, L.-W.; Heude, B.; Bernard, J.Y.; Harvey, N.C.; Duijts, L.; Mensink-Bout, S.M.; Polanska, K.; Mancano, G.; Suderman, M.; et al. Dietary Inflammatory Index and Non-Communicable Disease Risk: A Narrative Review. Nutrients 2019, 11, 1873. [Google Scholar] [CrossRef]
- Saikia, L.; Talukdar, N.C.; Dutta, P.P. Exploring the Therapeutic Role of Flavonoids Through AMPK Activation in Metabolic Syndrome: A Narrative Review. Phytother. Res. 2025, 39, 1403–1421. [Google Scholar] [CrossRef] [PubMed]
- Bhol, N.K.; Bhanjadeo, M.M.; Singh, A.K.; Dash, U.C.; Ojha, R.R.; Majhi, S.; Duttaroy, A.K.; Jena, A.B. The interplay between cytokines, inflammation, and antioxidants: Mechanistic insights and therapeutic potentials of various antioxidants and anti-cytokine compounds. Biomed. Pharmacother. 2024, 178, 117177. [Google Scholar] [CrossRef] [PubMed]
- Fürst, R.; Zündorf, I. Plant-Derived Anti-Inflammatory Compounds: Hopes and Disappointments regarding the Translation of Preclinical Knowledge into Clinical Progress. Mediators Inflamm. 2014, 2014, 146832. [Google Scholar] [CrossRef] [PubMed]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef]
- Huang, J.; Xie, M.; He, L.; Song, X.; Cao, T. Chlorogenic acid: A review on its mechanisms of anti-inflammation, disease treatment, and related delivery systems. Front. Pharmacol. 2023, 14, 1218015. [Google Scholar] [CrossRef]
- Choo, M.Z.Y.; Chua, J.A.T.; Lee, S.X.Y.; Ang, Y.; Wong, W.S.F.; Chai, C.L.L. Privileged natural product compound classes for anti-inflammatory drug development. Nat. Prod. Rep. 2025, 42, 856–875. [Google Scholar] [CrossRef]
- Al-Khayri, J.M.; Sahana, G.R.; Nagella, P.; Joseph, B.V.; Alessa, F.M.; Al-Mssallem, M.Q. Flavonoids as Potential Anti-Inflammatory Molecules: A Review. Molecules 2022, 27, 2901. [Google Scholar] [CrossRef] [PubMed]
- Danielewski, M.; Matuszewska, A.; Nowak, B.; Kucharska, A.Z.; Sozański, T. The Effects of Natural Iridoids and Anthocyanins on Selected Parameters of Liver and Cardiovascular System Functions. Oxid. Med. Cell. Longev. 2020, 2020, 2735790. [Google Scholar] [CrossRef]
- Sun, H.; Hao, Y.; Liu, H.; Gao, F. The immunomodulatory effects of GLP-1 receptor agonists in neurogenerative diseases and ischemic stroke treatment. Front. Immunol. 2025, 16, 1525623. [Google Scholar] [CrossRef]
- Abiola, J.O.; Oluyemi, A.A.; Idowu, O.T.; Oyinloye, O.M.; Ubah, C.S.; Owolabi, O.V.; Somade, O.T.; Onikanni, S.A.; Ajiboye, B.O.; Osunsanmi, F.O.; et al. Potential Role of Phytochemicals as Glucagon-like Peptide 1 Receptor (GLP-1R) Agonists in the Treatment of Diabetes Mellitus. Pharmaceuticals 2024, 17, 736. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Qiao, J.; Huo, J.; Rupasinghe, H.P.V. Plant iridoids: Chemistry, dietary sources and potential health benefits. Food Chem. X 2025, 27, 102491. [Google Scholar] [CrossRef]
- Viljoen, A.; Mncwangi, N.; Vermaak, I. Anti-Inflammatory Iridoids of Botanical Origin. Curr. Med. Chem. 2012, 19, 2104–2127. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.; Menichini, F.; Statti, G.; Menichini, F. Biological and Pharmacological Activities of Iridoids: Recent Developments. Mini-Rev. Med. Chem. 2008, 8, 399–420. [Google Scholar] [CrossRef]
- Wang, C.; Gong, X.; Bo, A.; Zhang, L.; Zhang, M.; Zang, E.; Zhang, C.; Li, M. Iridoids: Research Advances in Their Phytochemistry, Biological Activities, and Pharmacokinetics. Molecules 2020, 25, 287. [Google Scholar] [CrossRef]
- Salehi, B.; Shivaprasad Shetty, M.; Anil Kumar, N.V.; Živković, J.; Calina, D.; Oana Docea, A.; Emamzadeh-Yazdi, S.; Sibel Kılıç, C.; Goloshvili, T.; Nicola, S.; et al. Veronica Plants—Drifting from Farm to Traditional Healing, Food Application, and Phytopharmacology. Molecules 2019, 24, 2454. [Google Scholar] [CrossRef]
- Rahamouz-Haghighi, S. Biological Activities and Analytical Methods for Detecting Aucubin and Catalpol Iridoid Glycosides in Plantago Species: A Review Study. Pharm. Biomed. Res. 2023, 9, 85–114. [Google Scholar] [CrossRef]
- Gvazava, L.N.; Kikoladze, V.S. Iridoids from Verbascum phlomoides and V. densiflorum. Chem. Nat. Compd. 2009, 45, 751–752. [Google Scholar] [CrossRef]
- Gründemann, C.; Garcia-Käufer, M.; Sauer, B.; Stangenberg, E.; Könczöl, M.; Merfort, I.; Zehl, M.; Huber, R. Traditionally used Veronica officinalis inhibits proinflammatory mediators via the NF-κB signalling pathway in a human lung cell line. J. Ethnopharmacol. 2013, 145, 118–126. [Google Scholar] [CrossRef]
- Scarlat, M.; Şandor, V.; Tǎmaş, M.; Cuparencu, B. Experimental anti-ulcer activity of Veronica officinalis L. extracts. J. Ethnopharmacol. 1985, 13, 157–163. [Google Scholar] [CrossRef]
- Živković, J.; Ćebović, T.; Maksimović, Z. In vivo and in vitro antioxidant effects of three Veronica species. Open Life Sci. 2012, 7, 559–568. [Google Scholar] [CrossRef]
- Kiss, B.; Popa, D.S.; Crişan, G.I.; Bojiţă, M.A.; Loghin, F.E. The evaluation of antioxidant potential of Veronica officinalis and Rosmarinus officinalis extracts by monitoring malondialdehide and glutathione levels in rats. Farmacia 2009, 57, 432–441. [Google Scholar]
- Lee, H.Y.; Ghimeray, A.K.; Yim, J.H.; Chang, M.S. Antioxidant, Collagen Synthesis Activity in Vitro and Clinical Test on Anti-Wrinkle Activity of Formulated Cream Containing Veronica officinalis Extract. J. Cosmet. Dermatol. Sci. Appl. 2015, 5, 45–51. [Google Scholar] [CrossRef]
- Beara, I.; Živković, J.; Lesjak, M.; Ristić, J.; Šavikin, K.; Maksimović, Z.; Janković, T. Phenolic profile and anti-inflammatory activity of three Veronica species. Ind. Crops Prod. 2015, 63, 276–280. [Google Scholar] [CrossRef]
- Harput, U.S.; Saracoglu, I.; Inoue, M.; Ogihara, Y. Anti-inflammatory and Cytotoxic Activities of Five Veronica Species. Biol. Pharm. Bull. 2002, 25, 483–486. [Google Scholar] [CrossRef]
- Zhakipbekov, K.; Turgumbayeva, A.; Issayeva, R.; Kipchakbayeva, A.; Kadyrbayeva, G.; Tleubayeva, M.; Akhayeva, T.; Tastambek, K.; Sainova, G.; Serikbayeva, E.; et al. Antimicrobial and Other Biomedical Properties of Extracts from Plantago major, Plantaginaceae. Pharmaceuticals 2023, 16, 1092. [Google Scholar] [CrossRef] [PubMed]
- Laanet, P.-R.; Bragina, O.; Jõul, P.; Vaher, M. Plantago major and Plantago lanceolata Exhibit Antioxidant and Borrelia burgdorferi Inhibiting Activities. Int. J. Mol. Sci. 2024, 25, 7112. [Google Scholar] [CrossRef] [PubMed]
- Alipieva, K.; Korkina, L.; Orhan, I.E.; Georgiev, M.I. Verbascoside—A review of its occurrence, (bio)synthesis and pharmacological significance. Biotechnol. Adv. 2014, 32, 1065–1076. [Google Scholar] [CrossRef]
- Zubair, M.; Widén, C.; Renvert, S.; Rumpunen, K. Water and ethanol extracts of Plantago major leaves show anti-inflammatory activity on oral epithelial cells. J. Tradit. Complement. Med. 2019, 9, 169–171. [Google Scholar] [CrossRef]
- Farid, A.; Sheibani, M.; Shojaii, A.; Noori, M.; Motevalian, M. Evaluation of anti-inflammatory effects of leaf and seed extracts of Plantago major on acetic acid-induced ulcerative colitis in rats. J. Ethnopharmacol. 2022, 298, 115595. [Google Scholar] [CrossRef]
- Triastuti, A.; Pradana, D.A.; Setiawan, I.D.; Fakhrudin, N.; Himmi, S.K.; Widyarini, S.; Rohman, A. In vivo anti-inflammatory activities of Plantago major extract and fractions and analysis of their phytochemical components using a high-resolution mass spectrometry. Res. Pharm. Sci. 2022, 17, 665–676. [Google Scholar] [CrossRef]
- Ghiasian, M.; Niroomandi, Z.; Dastan, D.; Poorolajal, J.; Zare, F.; Ataei, S. Clinical and phytochemical studies of Plantago major in pressure ulcer treatment: A randomized controlled trial. Complement. Ther. Clin. Pract. 2021, 43, 101325. [Google Scholar] [CrossRef] [PubMed]
- Ghanadian, M.; Soltani, R.; Homayouni, A.; Khorvash, F.; Jouabadi, S.M.; Abdollahzadeh, M. The Effect of Plantago major Hydroalcoholic Extract on the Healing of Diabetic Foot and Pressure Ulcers: A Randomized Open-Label Controlled Clinical Trial. Int. J. Low. Extrem. Wounds 2024, 23, 475–481. [Google Scholar] [CrossRef]
- Baghizadeh, A.; Davati, A.; Heidarloo, A.J.; Emadi, F.; Aliasl, J. Efficacy of Plantago major seed in management of ulcerative colitis symptoms: A randomized, placebo controlled, clinical trial. Complement. Ther. Clin. Pract. 2021, 44, 101444. [Google Scholar] [CrossRef] [PubMed]
- Soltani, G.M.; Hemati, S.; Sarvizadeh, M.; Kamalinejad, M.; Tafazoli, V.; Latifi, S.A. Efficacy of the Plantago major L. syrup on radiation induced oral mucositis in head and neck cancer patients: A randomized, double blind, placebo-controlled clinical trial. Complement. Ther. Med. 2020, 51, 102397. [Google Scholar] [CrossRef]
- Matev, M.; Angelova, I.; Koĭchev, A.; Leseva, M.; Stefanov, G. Clinical trial of a Plantago major preparation in the treatment of chronic bronchitis. Vutr. Boles. 1982, 21, 133–137. [Google Scholar]
- Sosa, S.; Faudale, M.; Zacchigna, M.; Cateni, F.; Del Favero, G.; Tubaro, A.; Della Loggia, R. Topical anti-inflammatory activity of Plantago lanceolata L. leaves: The relevance of triterpenic acids. Planta Med. 2011, 77, PM132. [Google Scholar] [CrossRef]
- Vigo, E.; Cepeda, A.; Gualillo, O.; Perez-Fernandez, R. In-vitro anti-inflammatory activity of Pinus sylvestris and Plantago lanceolata extracts: Effect on inducible NOS, COX-1, COX-2 and their products in J774A.1 murine macrophages. J. Pharm. Pharmacol. 2005, 57, 383–391. [Google Scholar] [CrossRef]
- Pol, M.; Schmidtke, K.; Lewandowska, S. Plantago lanceolata—An overview of its agronomically and healing valuable features. Open Agric. 2021, 6, 479–488. [Google Scholar] [CrossRef]
- Abate, L.; Bachheti, R.K.; Tadesse, M.G.; Bachheti, A. Ethnobotanical Uses, Chemical Constituents, and Application of Plantago lanceolata L. J. Chem. 2022, 2022, 1532031. [Google Scholar] [CrossRef]
- Kubica, P.; Szopa, A.; Dominiak, J.; Luczkiewicz, M.; Ekiert, H. Verbena officinalis (Common Vervain)—A Review on the Investigations of This Medicinally Important Plant Species. Planta Med. 2020, 86, 1241–1257. [Google Scholar] [CrossRef]
- Dai, X.; Zhou, X.; Shao, R.; Zhao, R.; Yanamandra, A.K.; Xing, Z.; Ding, M.; Wang, J.; Liu, T.; Zheng, Q.; et al. Bioactive Constituents of Verbena officinalis Alleviate Inflammation and Enhance Killing Efficiency of Natural Killer Cells. Int. J. Mol. Sci. 2023, 24, 7144. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, E. Short-term effects of Verbena officinalis Linn decoction on patients suffering from chronic generalized gingivitis: Double-blind randomized controlled multicenter clinical trial. Quintessence Int. 2016, 47, 491–498. [Google Scholar] [CrossRef]
- Nisar, R.; Ahmad, S.; Khan, K.-R.; Sherif, A.E.; Alasmari, F.; Almuqati, A.F.; Ovatlarnporn, C.; Khan, M.A.; Umair, M.; Rao, H.; et al. Metabolic Profiling by GC-MS, In Vitro Biological Potential, and In Silico Molecular Docking Studies of Verbena officinalis. Molecules 2022, 27, 6685. [Google Scholar] [CrossRef]
- Stojković, D.; Đorđevski, N.; Rajaković, M.; Filipović, B.; Božunović, J.; Bolevich, S.; Zengin, G.; Bolevich, S.; Gašić, U.; Soković, M. Investigation of Bioactive Compounds Extracted from Verbena officinalis and Their Biological Effects in the Extraction by Four Butanol/Ethanol Solvent Combinations. Pharmaceuticals 2025, 18, 1012. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.I. Anti-inflammatory and analgesic activity of the topical preparation of Verbena officinalis L. J. Ethnopharmacol. 2006, 107, 380–382. [Google Scholar] [CrossRef]
- Chen, Y.; Gan, Y.; Yu, J.; Ye, X.; Yu, W. Key ingredients in Verbena officinalis and determination of their anti-atherosclerotic effect using a computer-aided drug design approach. Front. Plant Sci. 2023, 14, 1154266. [Google Scholar] [CrossRef]
- Stepanova, S.I.; Shtrygol, S.Y.; Tovchiga, O.V.; Demeshko, O.V. Clinical and pharmacological aspects of vervain (Verbena officinalis L.) using in the treatment of respiratory system diseases (literature review). Farm. Zh. 2023, 5, 71–83. [Google Scholar] [CrossRef]
- Pasdaran, A.; Hamedi, A. The genus Scrophularia: A source of iridoids and terpenoids with a diverse biological activity. Pharm. Biol. 2017, 55, 2211–2233. [Google Scholar] [CrossRef]
- De Santos Galíndez, J.; Díaz Lanza, A.; Fernández Matellano, L. Biologically Active Substances from the Genus Scrophularia. Pharm. Biol. 2002, 40, 45–59. [Google Scholar] [CrossRef]
- Stevenson, P.C.; Simmonds, M.S.J.; Sampson, J.; Houghton, P.J.; Grice, P. Wound healing activity of acylated iridoid glycosides from Scrophularia nodosa. Phytother. Res. 2002, 16, 33–35. [Google Scholar] [CrossRef]
- Zhu, T.; Zhang, L.; Ling, S.; Duan, J.; Qian, F.; Li, Y.; Xu, J.-W. Scropolioside B Inhibits IL-1β and Cytokines Expression through NF-κB and Inflammasome NLRP3 Pathways. Mediators Inflamm. 2014, 2014, 819053. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Zhang, L.; Ling, S.; Qian, F.; Li, Y.; Xu, J.-W. Anti-Inflammatory Activity Comparison among Scropoliosides—Catalpol Derivatives with 6-O-Substituted Cinnamyl Moieties. Molecules 2015, 20, 19823–19836. [Google Scholar] [CrossRef]
- Kerdar, T.; Rabienejad, N.; Alikhani, Y.; Moradkhani, S.; Dastan, D. Clinical, in vitro and phytochemical, studies of Scrophularia striata mouthwash on chronic periodontitis disease. J. Ethnopharmacol. 2019, 239, 111872. [Google Scholar] [CrossRef] [PubMed]
- Grigore, A.; Colceru-Mihul, S.; Litescu, S.; Panteli, M.; Rasit, I. Correlation between polyphenol content and anti-inflammatory activity of Verbascum phlomoides (mullein). Pharm. Biol. 2013, 51, 925–929. [Google Scholar] [CrossRef]
- El Gizawy, H.A.E.H.; Hussein, M.A.; Abdel-Sattar, E. Biological activities, isolated compounds and HPLC profile of Verbascum nubicum. Pharm. Biol. 2019, 57, 485–497. [Google Scholar] [CrossRef]
- Paun, G.; Neagu, E.; Albu, C.; Radu, G.L. Verbascum phlomoides and Solidago virgaureae herbs as natural source for preventing neurodegenerative diseases. J. Herb. Med. 2016, 6, 180–186. [Google Scholar] [CrossRef]
- Pongkitwitoon, B.; Putalun, W.; Triwitayakorn, K.; Kitisripanya, T.; Kanchanapoom, T.; Boonsnongcheep, P. Anti-inflammatory activity of verbascoside- and isoverbascoside-rich Lamiales medicinal plants. Heliyon 2024, 10, e23644. [Google Scholar] [CrossRef] [PubMed]
- Saha, R.; Majie, A.; Baidya, R.; Sarkar, B. Verbascoside: Comprehensive review of a phenylethanoid macromolecule and its journey from nature to bench. Inflammopharmacology 2024, 32, 2729–2751. [Google Scholar] [CrossRef]
- Mitova, M.I.; Anchev, M.E.; Handjieva, N.V.; Popov, S.S. Iridoid Patterns in Galium L. and Some Phylogenetic Considerations. Z. Für Naturforschung C 2002, 57, 226–234. [Google Scholar] [CrossRef]
- Herre, I.; Stegemann, T.; Zidorn, C. Comprehensive analysis of natural products of Galium odoratum (Rubiaceae), focusing on the formation of coumarin during the drying process. Phytochemistry 2025, 236, 114485. [Google Scholar] [CrossRef]
- Petkova, M.K.; Grozeva, N.H.; Tzanova, M.T.; Todorova, M.H. A Review of Phytochemical and Pharmacological Studies on Galium verum L., Rubiaceae. Molecules 2025, 30, 1856. [Google Scholar] [CrossRef]
- Bradic, J.; Andjic, M.; Novakovic, J.; Kocovic, A.; Tomovic, M.; Petrovic, A.; Nikolic, M.; Mitrovic, S.; Jakovljevic, V.; Pecarski, D. Lady’s Bedstraw as a Powerful Antioxidant for Attenuation of Doxorubicin-Induced Cardiotoxicity. Antioxidants 2023, 12, 1277. [Google Scholar] [CrossRef]
- Bradic, J.; Zivkovic, V.; Srejovic, I.; Jakovljevic, V.; Petkovic, A.; Turnic, T.N.; Jeremic, J.; Jeremic, N.; Mitrovic, S.; Sobot, T.; et al. Protective Effects of Galium verum L. Extract against Cardiac Ischemia/Reperfusion Injury in Spontaneously Hypertensive Rats. Oxid. Med. Cell. Longev. 2019, 2019, 4235405. [Google Scholar] [CrossRef] [PubMed]
- Antoniak, K.; Studzińska-Sroka, E.; Szymański, M.; Dudek-Makuch, M.; Cielecka-Piontek, J.; Korybalska, K. Antiangiogenic, Anti-Inflammatory and Antioxidant Properties of Bidens tripartite Herb, Galium verum Herb and Rumex hydrolapathum Root. Molecules 2023, 28, 4966. [Google Scholar] [CrossRef]
- Lotfy, B.M.; Mousa, M.R.; El-Shehry, M.S.; Ahmed, S.H.; Ali, S.B.; Al Shawoush, A.M.; Mohamed, A.S. Therapeutic Potency of Gallium verum Extract on Ethanol-Induced Gastric Ulcer in Rats. Biointerface Res. Appl. Chem. 2021, 12, 6010–6020. [Google Scholar] [CrossRef]
- Lee, S.; Park, S.; Park, H. Immuno-Enhancing Effects of Galium aparine L. in Cyclophosphamide-Induced Immunosuppressed Animal Models. Nutrients 2024, 16, 597. [Google Scholar] [CrossRef]
- Razzivina, V.; Vasiljeva, A.; Kronberga, A.; Skudrins, G.; Nakurte, I.; Zogota, M.; Mezaka, I.; Pugovics, O.; Grinberga, S.; Dambrova, M.; et al. Phenolic Content and Anti-Inflammatory Activity of Cultivated and Wild-Type Galium odoratum Extracts in Murine Bone Marrow-Derived Macrophages. Antioxidants 2024, 13, 1447. [Google Scholar] [CrossRef]
- Patel, H.; Gomes, E.N.; Yuan, B.; Lyu, W.; Wu, Q.; Simon, J.E. Investigation of Volatile Iridoid Terpenes in Nepeta cataria L. (Catnip) Genotypes. Molecules 2022, 27, 7057. [Google Scholar] [CrossRef]
- Reichert, W.; Villani, T.; Pan, M.; Ho, C.; Simon, J.E.; Wu, Q. Phytochemical Analysis and Anti-Inflammatory Activity of Nepeta cataria Accessions. J. Med. Act. Plants 2018, 7, 19–27. [Google Scholar] [CrossRef]
- Ricci, E.L.; Toyama, D.O.; Lago, J.H.; Romoff, P.; Kirsten, T.B.; Reis-Silva, T.M.; Bernardi, M.M. Antinociceptive and anti-inflammatory actions of Nepeta cataria L. var. citriodora (Becker) Balb. essential oil in mice. J. Health Sci. Inst. 2010, 28, 289–293. [Google Scholar]
- White Dead-Nettle (Lamium album). In Nonvitamin and Nonmineral Nutritional Supplements; Elsevier: Amsterdam, The Netherlands, 2019; pp. 455–459. ISBN 978-0-12-812491-8.
- Alipieva, K.I.; Taskova, R.M.; Jensen, S.R.; Handjieva, N.V. Iridoid glucosides from Lamium album and Lamium maculatum (Lamiaceae). Biochem. Syst. Ecol. 2006, 34, 88–91. [Google Scholar] [CrossRef]
- Alipieva, K.; Kokubun, T.; Taskova, R.; Evstatieva, L.; Handjieva, N. LC–ESI-MS analysis of iridoid glucosides in Lamium species. Biochem. Syst. Ecol. 2007, 35, 17–22. [Google Scholar] [CrossRef]
- Damtoft, S. Iridoid glucosides from Lamium album. Phytochemistry 1992, 31, 175–178. [Google Scholar] [CrossRef]
- Czerwińska, M.; Świerczewska, A.; Woźniak, M.; Kiss, A. Bioassay-Guided Isolation of Iridoids and Phenylpropanoids from Aerial Parts of Lamium album and Their Anti-inflammatory Activity in Human Neutrophils. Planta Med. 2017, 83, 1011–1019. [Google Scholar] [CrossRef]
- Paduch, R.; Wójciak-Kosior, M.; Matysik, G. Investigation of biological activity of Lamii albi flos extracts. J. Ethnopharmacol. 2007, 110, 69–75. [Google Scholar] [CrossRef]
- Khanaki, K.; Fekri, A.; Abedinzade, M.; Mohammadi, E.; Aghajanpour, F. Potential anti-inflammatory effect of Lamium album extract through caspase-3 and cyclooxygenase-2 genes expression in a rat model of middle cerebral artery occlusion. Folia Med. 2022, 64, 275–282. [Google Scholar] [CrossRef]
- Benedec, D.; Oniga, I.; Hanganu, D.; Vlase, A.-M.; Ielciu, I.; Crișan, G.; Fiţ, N.; Niculae, M.; Bab, T.; Pall, E.; et al. Revealing the Phenolic Composition and the Antioxidant, Antimicrobial and Antiproliferative Activities of Two Euphrasia sp. Extracts. Plants 2024, 13, 1790. [Google Scholar] [CrossRef]
- Teixeira, R.; Silva, L.R. Bioactive compounds and in vitro biological activity of Euphrasia rostkoviana Hayne extracts. Ind. Crops Prod. 2013, 50, 680–689. [Google Scholar] [CrossRef]
- Liu, Y.; Hwang, E.; Ngo, H.T.T.; Perumalsamy, H.; Kim, Y.J.; Li, L.; Yi, T.-H. Protective Effects of Euphrasia officinalis Extract against Ultraviolet B-Induced Photoaging in Normal Human Dermal Fibroblasts. Int. J. Mol. Sci. 2018, 19, 3327. [Google Scholar] [CrossRef] [PubMed]
- Paduch, R.; Woźniak, A.; Niedziela, P.; Rejdak, R. Assessment of Eyebright (Euphrasia officinalis L.) Extract Activity in Relation to Human Corneal Cells Using In Vitro Tests. Balk. Med. J. 2014, 33, 29–36. [Google Scholar] [CrossRef]
- Bigagli, E.; Cinci, L.; D’Ambrosio, M.; Luceri, C. Pharmacological activities of an eye drop containing Matricaria chamomilla and Euphrasia officinalis extracts in UVB-induced oxidative stress and inflammation of human corneal cells. J. Photochem. Photobiol. B 2017, 173, 618–625. [Google Scholar] [CrossRef]
- Porchezhian, E.; Ansari, S.H.; Shreedharan, N.K.K. Antihyperglycemic activity of Euphrasia officinale leaves. Fitoterapia 2000, 71, 522–526. [Google Scholar] [CrossRef]
- Ververis, A.; Kyriakou, S.; Paraskeva, H.; Panayiotidis, M.I.; Plioukas, M.; Christodoulou, K. Chemical Characterization and Assessment of the Neuroprotective Potential of Euphrasia officinalis. Int. J. Mol. Sci. 2024, 25, 12902. [Google Scholar] [CrossRef]
- Novy, P.; Davidova, H.; Serrano-Rojero, C.S.; Rondevaldova, J.; Pulkrabek, J.; Kokoska, L. Composition and Antimicrobial Activity of Euphrasia rostkoviana Hayne Essential Oil. Evid. Based Complement. Alternat. Med. 2015, 2015, 734101. [Google Scholar] [CrossRef]
- Park, Y.J.; Lim, J.Y.; Kwon, S.-H.; Shin, M.-S. Aucubin suppresses lipopolysaccharide-induced pro-inflammatory responses by blocking the NF-κB translocation signaling pathways in activated microglial cells. Phytochem. Lett. 2022, 48, 120–127. [Google Scholar] [CrossRef]
- Yuan, J.; Cheng, W.; Zhang, G.; Ma, Q.; Li, X.; Zhang, B.; Hu, T.; Song, G. Protective effects of iridoid glycosides on acute colitis via inhibition of the inflammatory response mediated by the STAT3/NF-кB pathway. Int. Immunopharmacol. 2020, 81, 106240. [Google Scholar] [CrossRef] [PubMed]
- Jaafar, A.; Zulkipli, M.A.; Mohd Hatta, F.H.; Jahidin, A.H.; Abdul Nasir, N.A.; Hazizul Hasan, M. Therapeutic potentials of iridoids derived from Rubiaceae against in vitro and in vivo inflammation: A scoping review. Saudi Pharm. J. 2024, 32, 101876. [Google Scholar] [CrossRef]
- Biswas, S.K. Does the Interdependence between Oxidative Stress and Inflammation Explain the Antioxidant Paradox? Oxid. Med. Cell. Longev. 2016, 2016, 5698931. [Google Scholar] [CrossRef]
- Ramani, S.; Pathak, A.; Dalal, V.; Paul, A.; Biswas, S. Oxidative Stress in Autoimmune Diseases: An Under Dealt Malice. Curr. Protein Pept. Sci. 2020, 21, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Sun, Y.; Hu, J. Catalpol inhibits apoptosis in hydrogen peroxide-induced endothelium by activating the PI3K/Akt signaling pathway and modulating expression of Bcl-2 and Bax. Eur. J. Pharmacol. 2010, 628, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Li, D.-Q.; Duan, Y.-L.; Bao, Y.-M.; Liu, C.-P.; Liu, Y.; An, L.-J. Neuroprotection of catalpol in transient global ischemia in gerbils. Neurosci. Res. 2004, 50, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Wang, Y.; Liu, Z.; Wang, J.; Wan, D.; Feng, S.; Yang, X.; Wang, T. Antidiabetic and antioxidant effects of catalpol extracted from Rehmannia glutinosa (Di Huang) on rat diabetes induced by streptozotocin and high-fat, high-sugar feed. Chin. Med. 2016, 11, 25. [Google Scholar] [CrossRef]
- Zeng, X.; Guo, F.; Ouyang, D. A review of the pharmacology and toxicology of aucubin. Fitoterapia 2020, 140, 104443. [Google Scholar] [CrossRef]
- Ma, B.; Zhang, J.; Zhu, Z.; Bao, X.; Zhang, M.; Ren, C.; Zhang, Q. Aucubin, a natural iridoid glucoside, attenuates oxidative stress-induced testis injury by inhibiting JNK and CHOP activation via Nrf2 up-regulation. Phytomedicine 2019, 64, 153057. [Google Scholar] [CrossRef]
- Shen, B.; Zhao, C.; Wang, Y.; Peng, Y.; Cheng, J.; Li, Z.; Wu, L.; Jin, M.; Feng, H. Aucubin inhibited lipid accumulation and oxidative stress via Nrf2/HO-1 and AMPK signalling pathways. J. Cell. Mol. Med. 2019, 23, 4063–4075. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, S.; Lu, Y.; Chen, S.; Du, X.; Hou, S.; Huang, H.; Liang, J. Asperuloside suppressing oxidative stress and inflammation in DSS-induced chronic colitis and RAW 264.7 macrophages via Nrf2/HO-1 and NF-κB pathways. Chem. Biol. Interact. 2021, 344, 109512. [Google Scholar] [CrossRef]
- He, C.; Zhu, R.; He, L.; Chook, C.Y.B.; Li, H.; Leung, F.P.; Tse, G.; Chen, Z.-Y.; Huang, Y.; Wong, W.T. Asperuloside as a Novel NRF2 Activator to Ameliorate Endothelial Dysfunction in High Fat Diet-Induced Obese Mice. Antioxid. Redox Signal. 2025, 42, 77–96. [Google Scholar] [CrossRef]
- Xi, Y.; Hou, X.; Huang, Y.; Zhou, Y.; Chen, Y.; Wang, Y.; Cheng, H. Loganin attenuates the inflammation, oxidative stress, and apoptosis through the JAK2/STAT3 pathway in cerebral ischemia-reperfusion injury. J. Stroke Cerebrovasc. Dis. 2025, 34, 108114. [Google Scholar] [CrossRef]
- Liu, J.; Li, T.; Zhong, G.; Pan, Y.; Gao, M.; Su, S.; Liang, Y.; Ma, C.; Liu, Y.; Wang, Q.; et al. Exploring the therapeutic potential of natural compounds for Alzheimer’s disease: Mechanisms of action and pharmacological properties. Biomed. Pharmacother. 2023, 166, 115406. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.-B.; Wang, J.-Y.; Zhang, Y.-L.; Hu, Y.-G.; Yue, Z.-S.; Zeng, L.-R.; Zheng, W.-J.; Hou, Q.; Yan, S.-G.; Quan, R.-F. Mechanisms underlying the antiapoptotic and anti-inflammatory effects of monotropein in hydrogen peroxide-treated osteoblasts. Mol. Med. Rep. 2016, 14, 5377–5384. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wang, J.; Wang, G.; Wang, F.; Yang, Y.; Liu, Z.; Song, Q.; Chen, S.; Chen, H. Monotropein inhibits MMP9-mediated cardiac oxidative stress, inflammation, matrix degradation and apoptosis in a mouse and cell line models of septic cardiac injury. Mol. Biol. Rep. 2025, 52, 329. [Google Scholar] [CrossRef]
- Jia, Y. Monotropein Reduces Retinal Inflammation, Oxidative Stress, and Dyslipidemia in Streptozotocin-induced Diabetic Retinopathy in Rats. Pharmacogn. Mag. 2025, 21, 170–180. [Google Scholar] [CrossRef]
- Czerwińska, M.E.; Świerczewska, A.; Granica, S. Bioactive Constituents of Lamium album L. as Inhibitors of Cytokine Secretion in Human Neutrophils. Molecules 2018, 23, 2770. [Google Scholar] [CrossRef]
- Ghule, B.V.; Kotagale, N.R.; Patil, K.S. Inhibition of the pro-inflammatory mediators in rat neutrophils by shanzhiside methyl ester and its acetyl derivative isolated from Barleria prionitis. J. Ethnopharmacol. 2020, 249, 112374. [Google Scholar] [CrossRef]
- Makino, Y.; Kondo, S.; Nishimura, Y.; Tsukamoto, Y.; Huang, Z.-L.; Urade, Y. Hastatoside and verbenalin are sleep-promoting components in Verbena officinalis. Sleep Biol. Rhythms 2009, 7, 211–217. [Google Scholar] [CrossRef]
- Kang, Z.-C.; Jiang, W.-L.; Xu, Y.; Zhu, H.-B.; Hou, J. Cardioprotection with 8-O-acetyl shanzhiside methylester on experimental myocardial ischemia injury. Eur. J. Pharm. Sci. 2012, 47, 124–130. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Gaynor, R. Role of the NF-kB Pathway in the Pathogenesis of Human Disease States. Curr. Mol. Med. 2001, 1, 287–296. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Park, K.S. Aucubin, a naturally occurring iridoid glycoside inhibits TNF-α-induced inflammatory responses through suppression of NF-κB activation in 3T3-L1 adipocytes. Cytokine 2013, 62, 407–412. [Google Scholar] [CrossRef]
- Park, K.S.; Chang, I.-M. Anti-Inflammatory Activity of Aucubin by Inhibition of Tumor Necrosis Factor-α Production in RAW 264.7 Cells. Planta Med. 2004, 70, 778–779. [Google Scholar] [CrossRef]
- Huang, H.; Chang, Y.-H.; Xu, J.; Ni, H.-Y.; Zhao, H.; Zhai, B.-W.; Efferth, T.; Gu, C.-B.; Fu, Y.-J. Aucubin as a natural potential anti-acute hepatitis candidate: Inhibitory potency and hepatoprotective mechanism. Phytomedicine 2022, 102, 154170. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cheng, X.; Qi, B.; Wang, Y.; Zheng, Y.; Liang, X.; Chang, Y.; Ning, M.; Gao, W.; Li, T. Aucubin protects against myocardial ischemia-reperfusion injury by regulating STAT3/NF-κB/HMGB-1 pathway. Int. J. Cardiol. 2024, 400, 131800. [Google Scholar] [CrossRef] [PubMed]
- Villasenor, I. Bioactivities of Iridoids. Anti-Inflamm. Anti-Allergy Agents Med. Chem. 2007, 6, 307–314. [Google Scholar] [CrossRef]
- Hassan, M.-A.M.; Fahmy, M.I.; Azzam, H.N.; Ebrahim, Y.M.; El-Shiekh, R.A.; Aboulmagd, Y.M. Multifaceted therapeutic potentials of catalpol, an iridoid glycoside: An updated comprehensive review. Inflammopharmacology 2025. [Google Scholar] [CrossRef]
- Oh, E.S.; Ryu, H.W.; Kim, M.-O.; Lee, J.-W.; Song, Y.N.; Park, J.-Y.; Kim, D.-Y.; Ro, H.; Lee, J.; Kim, T.-D.; et al. Verproside, the Most Active Ingredient in YPL-001 Isolated from Pseudolysimachion rotundum var. subintegrum, Decreases Inflammatory Response by Inhibiting PKCδ Activation in Human Lung Epithelial Cells. Int. J. Mol. Sci. 2023, 24, 7229. [Google Scholar] [CrossRef]
- Lee, S.U.; Sung, M.H.; Ryu, H.W.; Lee, J.; Kim, H.-S.; In, H.J.; Ahn, K.-S.; Lee, H.-J.; Lee, H.-K.; Shin, D.-H.; et al. Verproside inhibits TNF-α-induced MUC5AC expression through suppression of the TNF-α/NF-κB pathway in human airway epithelial cells. Cytokine 2016, 77, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Chi, G.; Wu, Q.; Ren, Y.; Chen, C.; Feng, H. Pretreatment with the compound asperuloside decreases acute lung injury via inhibiting MAPK and NF-κB signaling in a murine model. Int. Immunopharmacol. 2016, 31, 109–115. [Google Scholar] [CrossRef]
- Chan, Y.; Ng, S.W.; Xin Tan, J.Z.; Gupta, G.; Tambuwala, M.M.; Bakshi, H.A.; Dureja, H.; Dua, K.; Ishaq, M.; Caruso, V.; et al. Emerging therapeutic potential of the iridoid molecule, asperuloside: A snapshot of its underlying molecular mechanisms. Chem. Biol. Interact. 2020, 315, 108911. [Google Scholar] [CrossRef]
- He, J.; Lu, X.; Wei, T.; Dong, Y.; Cai, Z.; Tang, L.; Liu, M. Asperuloside and Asperulosidic Acid Exert an Anti-Inflammatory Effect via Suppression of the NF-κB and MAPK Signaling Pathways in LPS-Induced RAW 264.7 Macrophages. Int. J. Mol. Sci. 2018, 19, 2027. [Google Scholar] [CrossRef]
- Zhang, L.; Kan, Z.; Zhang, X.; Fang, H.; Jiang, W. 8-O-Acetyl Shanzhiside Methylester Attenuates Cerebral Ischaemia/Reperfusion Injury through an Anti-Inflammatory Mechanism in Diabetic Rats. Basic Clin. Pharmacol. Toxicol. 2014, 115, 481–487. [Google Scholar] [CrossRef]
- Li, X.; Liu, T.; Liang, K.; Wang, R.; Yang, J.; Chen, Y.; Wang, R.; Li, M. Elucidation of the antipyretic and anti-inflammatory effect of 8-O-Acetyl Shanzhiside methyl ester based on intestinal flora and metabolomics analysis. Front. Pharmacol. 2025, 16, 1482323. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Bai, Y.; Qiao, Y.; Wang, J.; Li, M.-Y.; Wang, J.-W.; Jia, N.; Chen, T.; Li, Y.-Q.; Wen, A.-D. 8-O-Acetyl Shanzhiside Methylester From Lamiophlomis Rotata Reduces Neuropathic Pain by Inhibiting the ERK/TNF-α Pathway in Spinal Astrocytes. Front. Cell. Neurosci. 2018, 12, 54. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zhan, H.; Wang, C.; Guo, P. Shanzhiside methylester protects against depression by inhibiting inflammation via the miRNA-155-5p/SOCS1 axis. Psychopharmacology 2022, 239, 2201–2213. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.-W.; Tran, V.H.; Duke, R.K.; Tan, S.; Chrubasik, S.; Roufogalis, B.D.; Duke, C.C. Harpagoside suppresses lipopolysaccharide-induced iNOS and COX-2 expression through inhibition of NF-κB activation. J. Ethnopharmacol. 2006, 104, 149–155. [Google Scholar] [CrossRef]
- Andersen, M.L.; Santos, E.H.R.; Seabra, M.D.L.V.; Da Silva, A.A.B.; Tufik, S. Evaluation of acute and chronic treatments with Harpagophytum procumbens on Freund’s adjuvant-induced arthritis in rats. J. Ethnopharmacol. 2004, 91, 325–330. [Google Scholar] [CrossRef]
- Chrubasik, S. Effectiveness of Harpagophytum extract WS 1531 in the treatment of exacerbation of low back pain: A randomized, placebo-controlled, double-blind study. Eur. J. Anaesthesiol. 1999, 16, 118. [Google Scholar] [CrossRef]
- Mei, C.; Wang, X.; Meng, F.; Zhang, X.; Chen, L.; Yan, S.; Xue, J.; Sun, X.; Wang, Y. Aucuboside Inhibits the Generation of Th17 Cells in Mice Colitis. Front. Pharmacol. 2021, 12, 696599. [Google Scholar] [CrossRef]
- Wen, H.; Xing, L.; Sun, K.; Xiao, C.; Meng, X.; Yang, J. Loganin attenuates intestinal injury in severely burned rats by regulating the toll-like receptor 4/NF-κB signaling pathway. Exp. Ther. Med. 2020, 20, 591–598. [Google Scholar] [CrossRef]
- Xu, J.; Li, R.; Zhang, Z.; Yang, C.; Liu, S.; Li, Y.; Chen, M.; Wang, W.; Zhang, G.; Song, G.; et al. Loganin Inhibits Angiotensin II–Induced Cardiac Hypertrophy Through the JAK2/STAT3 and NF-κB Signaling Pathways. Front. Pharmacol. 2021, 12, 678886. [Google Scholar] [CrossRef]
- He, J.; Li, J.; Liu, H.; Yang, Z.; Zhou, F.; Wei, T.; Dong, Y.; Xue, H.; Tang, L.; Liu, M. Scandoside Exerts Anti-Inflammatory Effect Via Suppressing NF-κB and MAPK Signaling Pathways in LPS-Induced RAW 264.7 Macrophages. Int. J. Mol. Sci. 2018, 19, 457. [Google Scholar] [CrossRef]
- Wang, F.; Wu, L.; Li, L.; Chen, S. Monotropein exerts protective effects against IL-1β-induced apoptosis and catabolic responses on osteoarthritis chondrocytes. Int. Immunopharmacol. 2014, 23, 575–580. [Google Scholar] [CrossRef]
- Li, Z.; Chen, Z.; Chen, J.; Liu, Z.; Li, Z.; Sun, H.; Wang, X.; Wei, J.; Cao, X.; Zheng, D. Monotropein attenuates apoptosis and pyroptosis in chondrocytes and alleviates osteoarthritis progression in mice. Chin. Med. 2023, 18, 42. [Google Scholar] [CrossRef]
- Zhang, Q.; Hu, S.; He, Y.; Song, Z.; Shen, Y.; Zhao, Z.; Zhang, Q.; Qin, L.; Zhang, Q. Monotropein Protects against Inflammatory Bone Loss and Suppresses Osteoclast Formation and Bone Resorption by Inhibiting NFATc1 via NF-κB and Akt/GSK-3β Pathway. Nutrients 2022, 14, 3978. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Ye, B.; Tian, J.; Wang, B.; Zha, Y.; Zheng, S.; Ma, T.; Zhuang, W.; Park, W.S.; Liang, J. Monotropein resists atherosclerosis by reducing inflammation, oxidative stress, and abnormal proliferation and migration of vascular smooth muscle cells. Korean J. Physiol. Pharmacol. 2025, 29, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Garcia, D.; Shaw, R.J. AMPK: Mechanisms of Cellular Energy Sensing and Restoration of Metabolic Balance. Mol. Cell 2017, 66, 789–800. [Google Scholar] [CrossRef] [PubMed]
- Dodington, D.W.; Desai, H.R.; Woo, M. JAK/STAT—Emerging Players in Metabolism. Trends Endocrinol. Metab. 2018, 29, 55–65. [Google Scholar] [CrossRef]
- Ruderman, N.B.; Carling, D.; Prentki, M.; Cacicedo, J.M. AMPK, insulin resistance, and the metabolic syndrome. J. Clin. Investig. 2013, 123, 2764–2772. [Google Scholar] [CrossRef]
- Liu, C.; Li, X. Role of leptin and adiponectin in immune response and inflammation. Int. Immunopharmacol. 2025, 161, 115082. [Google Scholar] [CrossRef]
- Kubota, N.; Yano, W.; Kubota, T.; Yamauchi, T.; Itoh, S.; Kumagai, H.; Kozono, H.; Takamoto, I.; Okamoto, S.; Shiuchi, T.; et al. Adiponectin Stimulates AMP-Activated Protein Kinase in the Hypothalamus and Increases Food Intake. Cell Metab. 2007, 6, 55–68. [Google Scholar] [CrossRef] [PubMed]
- AL-Suhaimi, E.A.; Shehzad, A. Leptin, resistin and visfatin: The missing link between endocrine metabolic disorders and immunity. Eur. J. Med. Res. 2013, 18, 12. [Google Scholar] [CrossRef]
- Kadowaki, T. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J. Clin. Investig. 2006, 116, 1784–1792. [Google Scholar] [CrossRef]
- Fievet, C.; Fruchart, J.; Staels, B. PPARα and PPARγ dual agonists for the treatment of type 2 diabetes and the metabolic syndrome. Curr. Opin. Pharmacol. 2006, 6, 606–614. [Google Scholar] [CrossRef]
- Li, L.; Yang, X.; Cai, Z.; Yin, B.; Jiang, W. Aucubin Regulates Allergic Airway Inflammation in Ovalbumin-induced Allergic Asthma in Mice by Controlling Th2 Cytokines. Pharmacogn. Mag. 2025, 21, 885–894. [Google Scholar] [CrossRef]
- Yan, J.; Wang, C.; Jin, Y.; Meng, Q.; Liu, Q.; Liu, Z.; Liu, K.; Sun, H. Catalpol ameliorates hepatic insulin resistance in type 2 diabetes through acting on AMPK/NOX4/PI3K/AKT pathway. Pharmacol. Res. 2018, 130, 466–480. [Google Scholar] [CrossRef]
- Bhattamisra, S.K.; Koh, H.M.; Lim, S.Y.; Choudhury, H.; Pandey, M. Molecular and Biochemical Pathways of Catalpol in Alleviating Diabetes Mellitus and Its Complications. Biomolecules 2021, 11, 323. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Du, Q.; Jiao, N.; Shu, A.; Gao, Y.; Chen, J.; Lv, G.; Lu, J.; Chen, Y.; Xu, H. Catalpol ameliorates diabetes-induced testicular injury and modulates gut microbiota. Life Sci. 2021, 267, 118881. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Xu, G.; Ma, S.; Li, F.; Yuan, M.; Xu, H.; Huang, K. Catalpol ameliorates high-fat diet-induced insulin resistance and adipose tissue inflammation by suppressing the JNK and NF-κB pathways. Biochem. Biophys. Res. Commun. 2015, 467, 853–858. [Google Scholar] [CrossRef]
- Zou, G.; Zhong, W.; Wu, F.; Wang, X.; Liu, L. Catalpol attenuates cardiomyocyte apoptosis in diabetic cardiomyopathy via Neat1/miR-140–5p/HDAC4 axis. Biochimie 2019, 165, 90–99. [Google Scholar] [CrossRef]
- Ishaq, M.; Tran, D.; Yang, C.; Ng, M.; Kackanattil, A.; Tata, K.; Deans, B.; Bleasel, M.; Vicenzi, S.; Randall, C.; et al. The Anti-Obesity Compound Asperuloside Reduces Inflammation in the Liver and Hypothalamus of High-Fat-Fed Mice. Endocrines 2022, 3, 641–653. [Google Scholar] [CrossRef]
- Fujikawa, T.; Hirata, T.; Hosoo, S.; Nakajima, K.; Wada, A.; Yurugi, Y.; Soya, H.; Matsui, T.; Yamaguchi, A.; Ogata, M.; et al. Asperuloside stimulates metabolic function in rats across several organs under high-fat diet conditions, acting like the major ingredient of Eucommia leaves with anti-obesity activity. J. Nutr. Sci. 2012, 1, e10. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Husain, A.; Abuzinadah, M.F.; Alghamdi, A.A.; Ahmad, V.; Shaik, R.A.; Eid, B.G. The molecular effects of Asperuloside against thermogenesis and anti-inflammatory process through multiple recent obesity pathways: An anti-obesity drug discovery by in-silico analysis. J. King Saud Univ.-Sci. 2023, 35, 102897. [Google Scholar] [CrossRef]
- Jeon, H.; Lee, C.-G.; Jeong, H.; Yun, S.-H.; Kim, J.; Uprety, L.P.; Oh, K.-I.; Singh, S.; Yoo, J.; Park, E.; et al. Inhibitory Effects of Loganin on Adipogenesis In Vitro and In Vivo. Int. J. Mol. Sci. 2023, 24, 4752. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Ye, Z.; Tian, T.; Zhu, R.; Liu, C.; Fang, X.; Zhang, D.; Fu, M.; Gao, S.; Zhao, D. Loganin regulates glycolipid metabolism by influencing intestinal microbiota and AMPK signaling in obese mice. J. Tradit. Chin. Med. Sci. 2022, 9, 321–329. [Google Scholar] [CrossRef]
- Wiman, K.G.; Zhivotovsky, B. Understanding cell cycle and cell death regulation provides novel weapons against human diseases. J. Intern. Med. 2017, 281, 483–495. [Google Scholar] [CrossRef]
- Xiong, Y.; Shi, L.; Wang, L.; Zhou, Z.; Wang, C.; Lin, Y.; Luo, D.; Qiu, J.; Chen, D. Activation of sirtuin 1 by catalpol-induced down-regulation of microRNA-132 attenuates endoplasmic reticulum stress in colitis. Pharmacol. Res. 2017, 123, 73–82. [Google Scholar] [CrossRef]
- Dong, J.; Du, C.; Xu, C.; Wang, Q.; Wang, Z.; Zhu, Q.; Lv, X.; Zhang, L.; Li, J.; Huang, C.; et al. Verbenalin attenuates hepatic damage and mitochondrial dysfunction in alcohol-associated steatohepatitis by regulating MDMX/PPARα-mediated ferroptosis. J. Ethnopharmacol. 2023, 307, 116227. [Google Scholar] [CrossRef]
- Chao, G.; Tian, X.; Zhang, W.; Ou, X.; Cong, F.; Song, T. Blocking Smad2 signalling with loganin attenuates SW10 cell cycle arrest induced by TNF-α. PLoS ONE 2017, 12, e0176965. [Google Scholar] [CrossRef]
- Kong, X.; Zhao, Y.; Wang, X.; Yu, Y.; Meng, Y.; Yan, G.; Yu, M.; Jiang, L.; Song, W.; Wang, B.; et al. Loganin reduces diabetic kidney injury by inhibiting the activation of NLRP3 inflammasome-mediated pyroptosis. Chem. Biol. Interact. 2023, 382, 110640. [Google Scholar] [CrossRef]
- Kwon, S.-H.; Kim, J.-A.; Hong, S.-I.; Jung, Y.-H.; Kim, H.-C.; Lee, S.-Y.; Jang, C.-G. Loganin protects against hydrogen peroxide-induced apoptosis by inhibiting phosphorylation of JNK, p38, and ERK 1/2 MAPKs in SH-SY5Y cells. Neurochem. Int. 2011, 58, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, C.; Kang, K.; Liu, H.; Liu, X.; Jia, X.; Yu, K. Loganin Attenuates Septic Acute Renal Injury with the Participation of AKT and Nrf2/HO-1 Signaling Pathways. Drug Des. Devel. Ther. 2021, 15, 501–513. [Google Scholar] [CrossRef]
- Zou, K.; Ying, J.; Xu, H.; Zeng, Q.; Huang, H.; Chen, W.; Li, X.; Wang, P.; Jin, H.; Li, J.; et al. Aucubin Alleviates Intervertebral Disc Degeneration by Repressing NF-κB-NLRP3 Inflammasome Activation in Endplate Chondrocytes. J. Inflamm. Res. 2023, 16, 5899–5913. [Google Scholar] [CrossRef]
- Bao, X.; Li, J.; Ren, C.; Wei, J.; Lu, X.; Wang, X.; Du, W.; Jin, X.; Ma, B.; Zhang, Q.; et al. Aucubin ameliorates liver fibrosis and hepatic stellate cells activation in diabetic mice via inhibiting ER stress-mediated IRE1α/TXNIP/NLRP3 inflammasome through NOX4/ROS pathway. Chem. Biol. Interact. 2022, 365, 110074. [Google Scholar] [CrossRef]
- Chiu, M.-H.; Hou, T.-Y.; Fan, C.-K.; Chang, J.-H.; Lin, C.-L.; Huang, S.-C.; Lee, Y.-L. Catalpol exerts antiallergic effects in IgE/ovalbumin-activated mast cells and a murine model of ovalbumin-induced allergic asthma. Int. Immunopharmacol. 2021, 96, 107782. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Yang, X. Effects of catalpol on bronchial asthma and its relationship with cytokines. J. Cell. Biochem. 2019, 120, 8992–8998. [Google Scholar] [CrossRef] [PubMed]
- Iftikhar, I.H.; Schimmel, M.; Bender, W.; Swenson, C.; Amrol, D. Comparative Efficacy of Anti IL-4, IL-5 and IL-13 Drugs for Treatment of Eosinophilic Asthma: A Network Meta-analysis. Lung 2018, 196, 517–530. [Google Scholar] [CrossRef]
- Yang, L.; Liu, T.; Zhuo, Y.; Li, D.; Li, D.; Liu, J.; Gao, H.; Zhang, L.; Lin, J.; Wang, X. Verbenalin alleviates acute lung injury induced by sepsis and IgG immune complex through GPR18 receptor. Cell. Signal. 2023, 109, 110768. [Google Scholar] [CrossRef]
- Sun, Q.; Zhao, R.; Li, S.; Zhou, W.; Zhang, J.; Pang, B.; Ding, S.; Bao, L.; Geng, Z.; Xie, R.; et al. Verbenalin protects against coronavirus pneumonia by promoting host immune homeostasis: Evidences for its mechanism of action. Phytomedicine 2025, 143, 156820. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Deng, Q.; Li, X.; Xian, L.; Xian, D.; Zhong, J. Natural Plant Extract—Loganin: A Hypothesis for Psoriasis Treatment Through Inhibiting Oxidative Stress and Equilibrating Immunity via Regulation of Macrophage Polarization. Clin. Cosmet. Investig. Dermatol. 2023, 16, 407–417. [Google Scholar] [CrossRef]
- Liu, S.; Shen, H.; Li, J.; Gong, Y.; Bao, H.; Zhang, J.; Hu, L.; Wang, Z.; Gong, J. Loganin inhibits macrophage M1 polarization and modulates sirt1/NF-κB signaling pathway to attenuate ulcerative colitis. Bioengineered 2020, 11, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.H.; Yang, G.; Seok, J.K.; Kang, H.C.; Cho, Y.-Y.; Lee, H.S.; Lee, J.Y. Loganin Prevents Hepatic Steatosis by Blocking NLRP3 Inflammasome Activation. Biomol. Ther. 2023, 31, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Dong, J.; Wang, Q.; Xu, C.; Feng, S.; Feng, R.; Lv, X.; Li, J.; Zhang, L.; Huang, C.; et al. Hastatoside attenuatescarbon tetrachloride-induced liver fibrosis by targeting glycogen synthase kinase-3β. Phytomedicine 2023, 109, 154585. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Qing, Y.; Zhang, Q.; Han, Z. The proteomic analysis of anti-inflammatory effects of geniposidic acid in LPS induced THP1 monocytic cells. Sci. Asia 2023, 49, 411. [Google Scholar] [CrossRef]
- Bai, Y.; Zhu, R.; Tian, Y.; Li, R.; Chen, B.; Zhang, H.; Xia, B.; Zhao, D.; Mo, F.; Zhang, D.; et al. Catalpol in Diabetes and its Complications: A Review of Pharmacology, Pharmacokinetics, and Safety. Molecules 2019, 24, 3302. [Google Scholar] [CrossRef]
- Fei, B.; Dai, W.; Zhao, S. Efficacy, Safety, and Cost of Therapy of the Traditional Chinese Medicine, Catalpol, in Patients Following Surgical Resection for Locally Advanced Colon Cancer. Med. Sci. Monit. 2018, 24, 3184–3192. [Google Scholar] [CrossRef]
- Santoro, A.; Bianco, G.; Picerno, P.; Aquino, R.P.; Autore, G.; Marzocco, S.; Gazzerro, P.; Lioi, M.B.; Bifulco, M. Verminoside- and verbascoside-induced genotoxicity on human lymphocytes: Involvement of PARP-1 and p53 proteins. Toxicol. Lett. 2008, 178, 71–76. [Google Scholar] [CrossRef]
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP); Bampidis, V.; Azimonti, G.; Bastos, M.d.L.; Christensen, H.; Fašmon Durjava, M.; Kouba, M.; López-Alonso, M.; López Puente, S.; Marcon, F.; et al. Safety of a feed additive consisting of a tincture derived from Verbascum thapsus L. (great mullein tincture) for use in all animal species (MANGHEBATI SAS). EFSA J. 2021, 19, e06711. [Google Scholar] [CrossRef]
- Alsaraf, K.M.; Mohammad, M.H.; Al-Shammari, A.M.; Abbas, I.S. Selective cytotoxic effect of Plantago lanceolata L. against breast cancer cells. J. Egypt. Natl. Cancer Inst. 2019, 31, 10. [Google Scholar] [CrossRef]
- Kartini; Piyaviriyakul, S.; Thongpraditchote, S.; Siripong, P.; Vallisuta, O. Effects of Plantago major extracts and its chemical compounds on proliferation of cancer cells and cytokines production of lipopolysaccharide-activated THP-1 macrophages. Pharmacogn. Mag. 2017, 13, 393. [Google Scholar] [CrossRef]
- Zhou, L.; Feng, Y.; Jin, Y.; Liu, X.; Sui, H.; Chai, N.; Chen, X.; Liu, N.; Ji, Q.; Wang, Y.; et al. Verbascoside promotes apoptosis by regulating HIPK2–p53 signaling in human colorectal cancer. BMC Cancer 2014, 14, 747. [Google Scholar] [CrossRef] [PubMed]
- Daneshforouz, A.; Nazemi, S.; Gholami, O.; Kafami, M.; Amin, B. The cytotoxicity and apoptotic effects of verbascoside on breast cancer 4T1 cell line. BMC Pharmacol. Toxicol. 2021, 22, 72. [Google Scholar] [CrossRef]
- Park, E.J.; Lee, H.S.; Oh, S.-R.; Lee, H.-K.; Lee, H.S. Pharmacokinetics of verproside after intravenous and oral administration in rats. Arch. Pharm. Res. 2009, 32, 559–564. [Google Scholar] [CrossRef]
- Bhattamisra, S.K.; Yap, K.H.; Rao, V.; Choudhury, H. Multiple Biological Effects of an Iridoid Glucoside, Catalpol, and Its Underlying Molecular Mechanisms. Biomolecules 2019, 10, 32. [Google Scholar] [CrossRef]
- Zhang, F.; Yan, Y.; Zhang, J.; Li, L.; Wang, Y.; Xia, C.; Lian, W.; Peng, Y.; Zheng, J.; He, J.; et al. Phytochemistry, synthesis, analytical methods, pharmacological activity, and pharmacokinetics of loganin: A comprehensive review. Phytother. Res. 2022, 36, 2272–2299. [Google Scholar] [CrossRef] [PubMed]
- Bartholomaeus, A. Inhibition of P-450 by aucubin: Is the biological activity of aucubin due to its glutaraldehyde-like aglycone? Toxicol. Lett. 1995, 80, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Cao, X.; Li, T.; Li, X. Effects of catalpol on the activity of human liver cytochrome P450 enzymes. Xenobiotica 2019, 49, 1289–1295. [Google Scholar] [CrossRef]
- Dąbrowska, M.; Souto, E.B.; Nowak, I. Lipid Nanoparticles Loaded with Iridoid Glycosides: Development and Optimization Using Experimental Factorial Design. Molecules 2021, 26, 3161. [Google Scholar] [CrossRef]
- Dąbrowska, M.; Nowak, I. Lipid Nanoparticles Loaded with Selected Iridoid Glycosides as Effective Components of Hydrogel Formulations. Materials 2021, 14, 4090. [Google Scholar] [CrossRef]
- Su, Y.; Zhang, B.; Sun, R.; Liu, W.; Zhu, Q.; Zhang, X.; Wang, R.; Chen, C. PLGA-based biodegradable microspheres in drug delivery: Recent advances in research and application. Drug Deliv. 2021, 28, 1397–1418. [Google Scholar] [CrossRef]
- Shah, L.; Yadav, S.; Amiji, M. Nanotechnology for CNS delivery of bio-therapeutic agents. Drug Deliv. Transl. Res. 2013, 3, 336–351. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Xiang, H.; Ding, K.; Wei, S.; Wang, X.; Pan, X.; Yin, W.; Zhang, W.; He, J.; Xu, J. Modification of iridoids in focus: Bridging structural complexity with biological innovation. Bioorganic Chem. 2025, 161, 108519. [Google Scholar] [CrossRef] [PubMed]
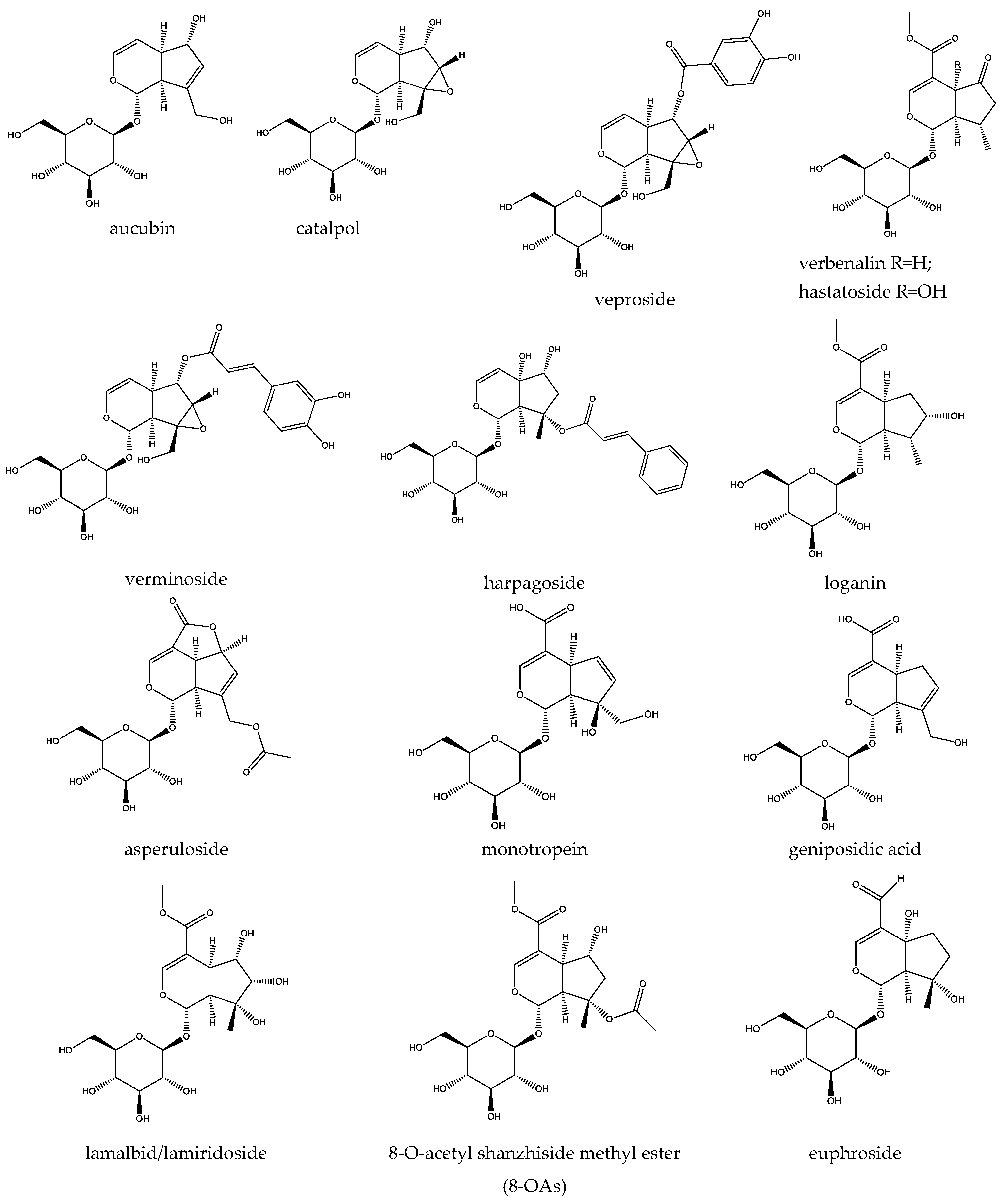
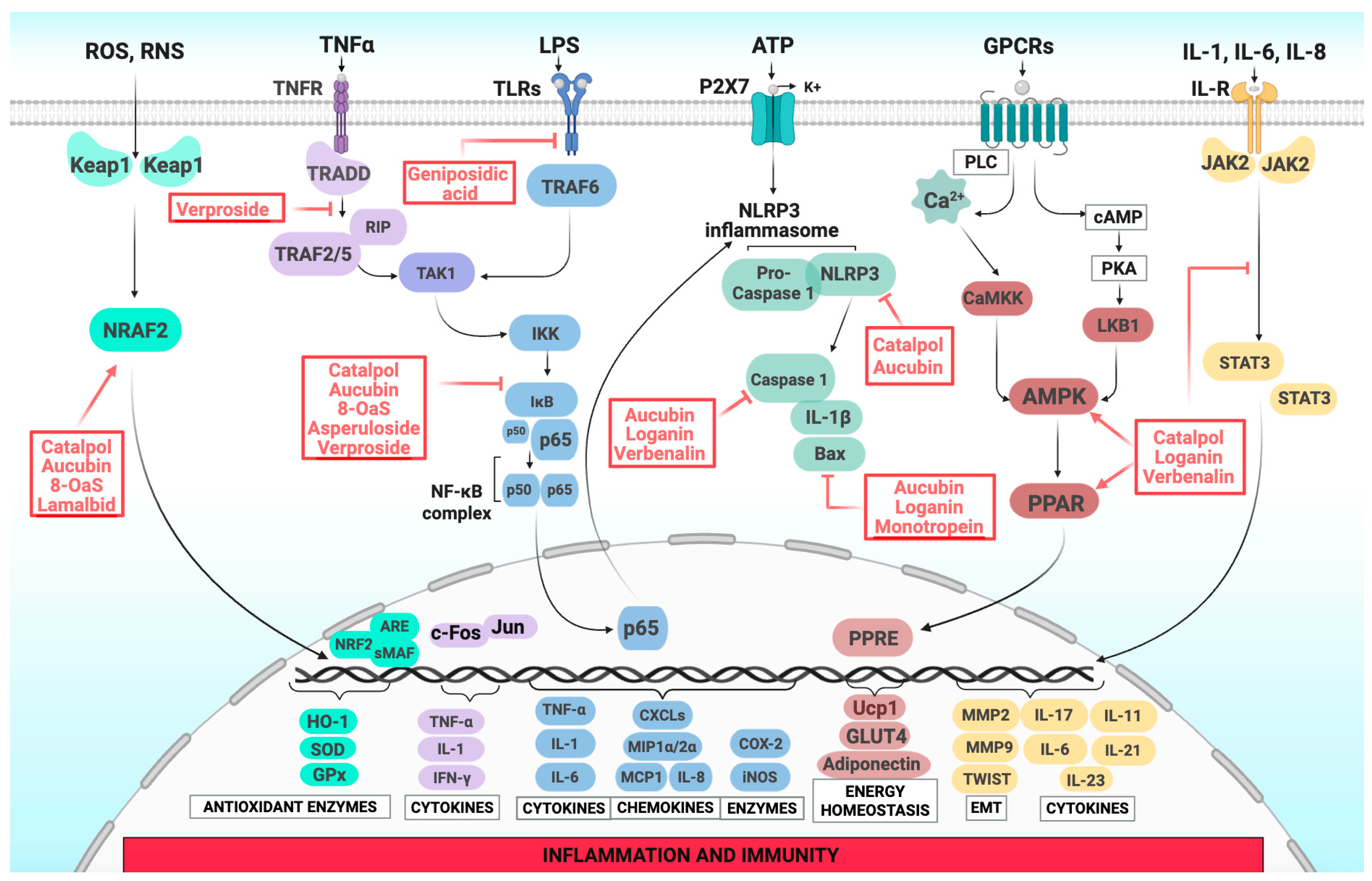
| Iridoid | Plant Species | Molecular Mechanisms | Biological Effects | Ref. |
|---|---|---|---|---|
| Aucubin | Veronica officinalis, Plantago spp., Verbena officinalis, Verbascum phlomoides, Euphrasia officinalis | - scavenges ROS, MDA, 4-HNE - enhances SOD, CAT, GSH-Px - activates Nrf2/HO-1 pathway - inhibits NF-κB and STAT3 phosphorylation - downregulates TNF-α, IL-6, iNOS - modulates tight junction proteins (ZO-1, Occludin, Claudin-11, Cx43) - PPARα and PPARγ activation - NLRP3 inflammasome inhibition - suppressed caspase-1 activation - caspase-3 and Bax down-regulation | Antioxidant, cardioprotective, hepatoprotective, anti-allergic in airway inflammation, chondroprotective | [95,98,114,146,164] |
| Catalpol | Veronica officinalis, Plantago spp., Scrophularia nodosa, Verbascum phlomoides, Euphrasia officinalis | - reduces ROS and iNOS - increases SOD, CAT, GSH-Px - inhibits NF-κB and NLRP3 - reduces IL-1β, TNF-α, COX-2, LOX-1 - suppresses mast cell degranulation - GLP-1R activation - activates AMPK pathway | Antioxidant, anti-inflammatory, bronchoprotective, nephroprotective, hepatoprotective, alleviating effects in diabetes and its related multiorgan complications, neuroprotective | [11,94,117,118,148,158,166] |
| Verproside | Veronica officinalis, Verbascum phlomoides | - inhibition of NF-κB signaling - reduced MUC5AC expression - inhibited TNF-α signaling | Anti-inflammatory, pulmoprotective | [118,119] |
| Verbenalin | Verbena officinalis | - ROS detoxification - regulation of the MDMX/PPARα axis - caspase-1 inhibition - NLRP3 inflammasome suppression and “phytoresolving” properties - AMPK activation | Antioxidant, antiapoptotic, hepatoprotective, pulmoprotective, antiviral | [159,169,170] |
| Hastatoside | Verbena officinalis | - antioxidant activity - reduced expression of fibrosis-related proteins | Antioxidant, anti-inflammatory, reduces airway inflammation and mucus hypersecretion in COPD, neuroprotective, antimicrobial | [108,174] |
| Harpagoside | Scrophularia nodosa | - inhibits COX-2 and iNOS expression - prevents IκB-α degradation - blocks NF-κB nuclear translocation | Anti-inflammatory | [127,128] |
| Aucuboside | Scrophularia nodosa | - inhibits LTC4 release - reduces TXB2 synthesis - modulates leukotriene and thromboxane pathways | Anti-inflammatory, anti-colitis | [50,130] |
| Loganin | Galium spp. | - reduces ROS, MDA - enhances SOD and NRf2 activity - inhibits TLR4/MyD88/NF-κB - Downregulates TNF-α, IL-1β, IL-6, iNOS, COX-2 - inhibits NLRP3 inflammasome - modulates JAK/STAT and AMPK signaling | Antioxidant, anti-inflammatory, neuroprotective, cardioprotective, anti-obesity effects, nephroprotective, antipsoriatic, anti-colitis | [101,102,131,132,155,161,163,171,172,173] |
| Asperuloside | Galium spp. Plantago spp. | - activates Nrf2/HO-1 - suppresses NF-κB and MAPK (ERK, JNK, p38) - reduces TNF-α, IL-1β, IL-6 - inhibits IκBα phosphorylation - reduced leptin secretion -up-regulates GLUt-4 expression | Antioxidant, vasoprotective, anti-colitis, pulmoprotective, anti-obesity | [99,100,120,121,152] |
| Monotropein | Galium spp. | - scavenges ROS - enhances mitochondrial potential, SIRT1, GSH, CAT - suppresses NF-κB and AP-1 - inhibits caspase-3/9, RANKL/TRAF6 - reduced MMP activity - antiapoptotic action | Antioxidant, retinoprotective, ameliorates joint and cartilage inflammation, antiosteoporotic, anti-atherosclerotic | [103,105,134,135,136,137] |
| Scandoside | Galium spp. | - inhibits NF-κB - decreases NO, PGE2, TNF-α, IL-6, iNOS, COX-2 - reduced MAPK signaling | In vitro anti-inflammatory | [133] |
| Geniposidic acid | Galium spp. | - reduced expression of IL-1, IL-6, IL-8, and TNF-α; - chemoattractants (CXCL10) and Toll-like receptors (TLR5) | In vitro anti-inflammatory | [175] |
| Lamalbid/ | Lamium album | - reduces ROS levels - inhibits IL-8 and TNF-α secretion | Antioxidant, anti-inflammatory | [106,109] |
| lamiridoside | ||||
| 8-O-acetyl shanzhiside methyl ester (8-OaS) | Lamium album | - NF-κB pathway inhibition - reduced LTB4 secretion - modulates TLR4/NF-κB, HSP70/NF-κB pathways - modulates gut–brain axis - modulates JAK2/STAT3 axis in neuroinflammation and depression - GLP-1R agonists | Anti-inflammatory, neuroprotective, antidepressant | [123,126] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mihaylova, R.; Elincheva, V.; Gevrenova, R.; Zheleva-Dimitrova, D.; Momekov, G.; Simeonova, R. Targeting Inflammation with Natural Products: A Mechanistic Review of Iridoids from Bulgarian Medicinal Plants. Molecules 2025, 30, 3456. https://doi.org/10.3390/molecules30173456
Mihaylova R, Elincheva V, Gevrenova R, Zheleva-Dimitrova D, Momekov G, Simeonova R. Targeting Inflammation with Natural Products: A Mechanistic Review of Iridoids from Bulgarian Medicinal Plants. Molecules. 2025; 30(17):3456. https://doi.org/10.3390/molecules30173456
Chicago/Turabian StyleMihaylova, Rositsa, Viktoria Elincheva, Reneta Gevrenova, Dimitrina Zheleva-Dimitrova, Georgi Momekov, and Rumyana Simeonova. 2025. "Targeting Inflammation with Natural Products: A Mechanistic Review of Iridoids from Bulgarian Medicinal Plants" Molecules 30, no. 17: 3456. https://doi.org/10.3390/molecules30173456
APA StyleMihaylova, R., Elincheva, V., Gevrenova, R., Zheleva-Dimitrova, D., Momekov, G., & Simeonova, R. (2025). Targeting Inflammation with Natural Products: A Mechanistic Review of Iridoids from Bulgarian Medicinal Plants. Molecules, 30(17), 3456. https://doi.org/10.3390/molecules30173456









