Correction: Marrero et al. Kallopterolides A–I, a New Subclass of seco-Diterpenes Isolated from the Southwestern Caribbean Sea Plume Antillogorgia kallos. Molecules 2024, 29, 2493
- Text Correction
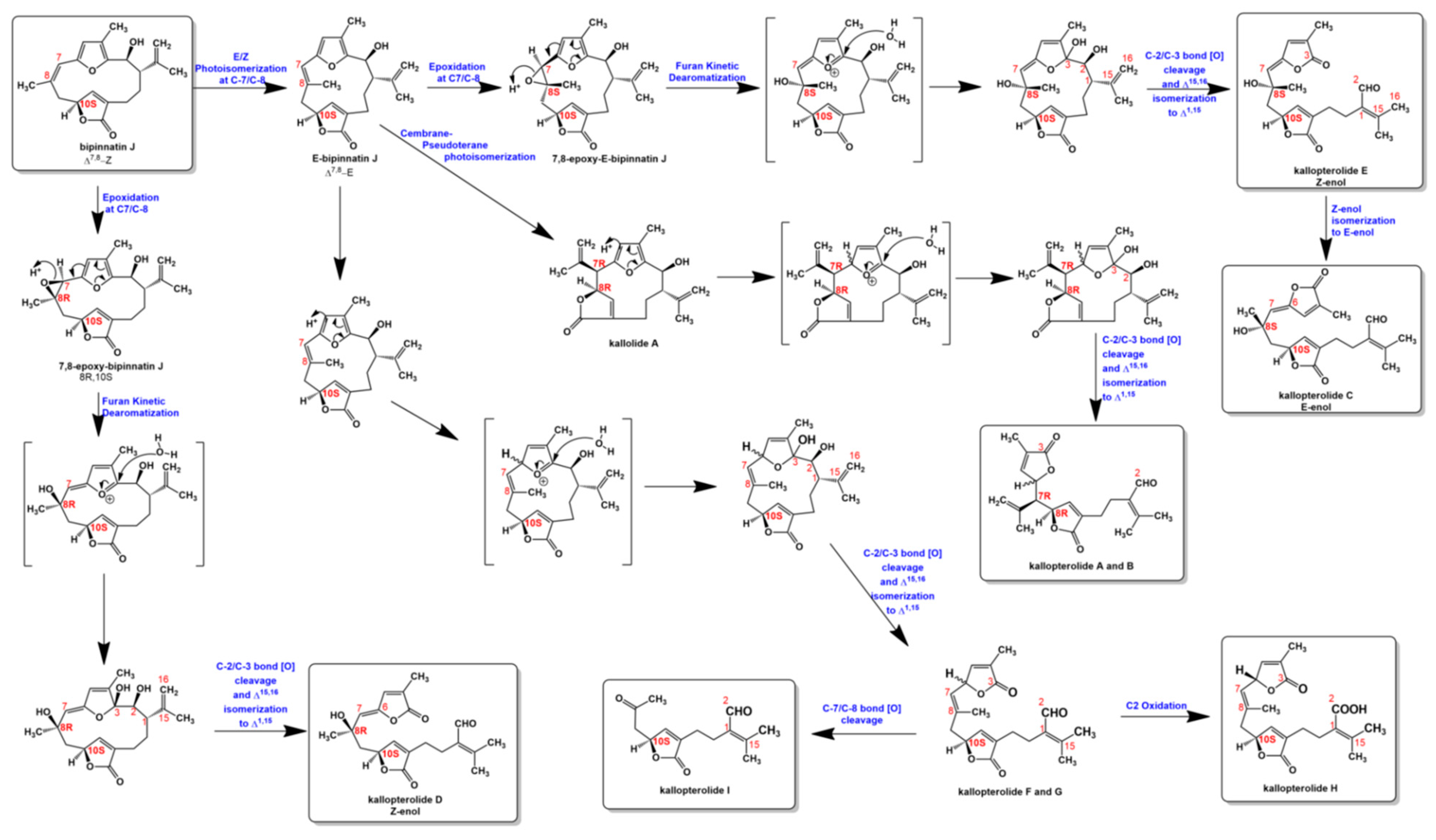
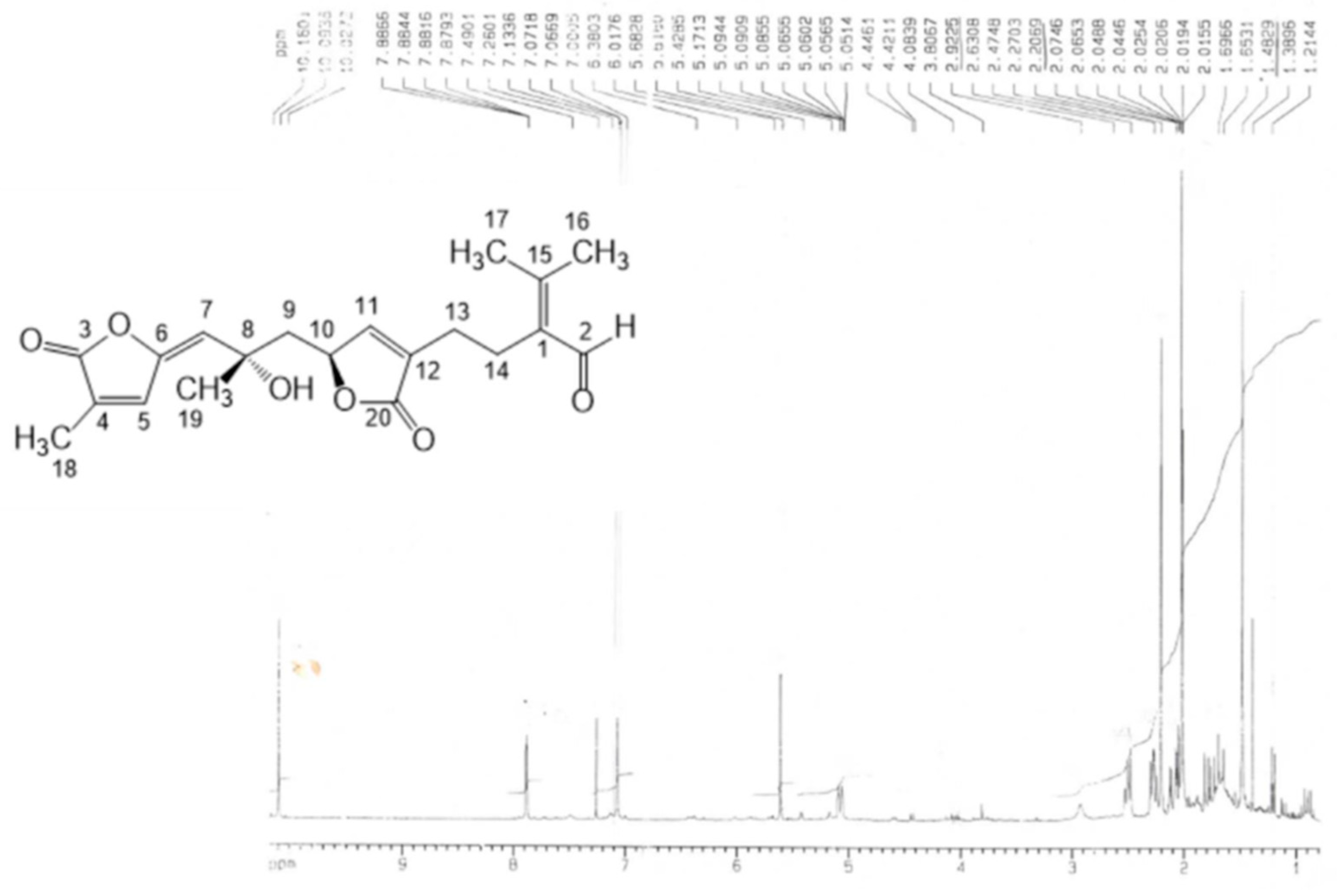
- Error in Figure
Reference
- Marrero, J.; Amador, L.A.; Novitskiy, I.M.; Kutateladze, A.G.; Rodríguez, A.D. Kallopterolides A–I, a New Subclass of seco-Diterpenes Isolated from the Southwestern Caribbean Sea Plume Antillogorgia kallos. Molecules 2024, 29, 2493. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marrero, J.; Amador, L.A.; Novitskiy, I.M.; Kutateladze, A.G.; Rodríguez, A.D. Correction: Marrero et al. Kallopterolides A–I, a New Subclass of seco-Diterpenes Isolated from the Southwestern Caribbean Sea Plume Antillogorgia kallos. Molecules 2024, 29, 2493. Molecules 2025, 30, 2929. https://doi.org/10.3390/molecules30142929
Marrero J, Amador LA, Novitskiy IM, Kutateladze AG, Rodríguez AD. Correction: Marrero et al. Kallopterolides A–I, a New Subclass of seco-Diterpenes Isolated from the Southwestern Caribbean Sea Plume Antillogorgia kallos. Molecules 2024, 29, 2493. Molecules. 2025; 30(14):2929. https://doi.org/10.3390/molecules30142929
Chicago/Turabian StyleMarrero, Jeffrey, Luis A. Amador, Ivan M. Novitskiy, Andrei G. Kutateladze, and Abimael D. Rodríguez. 2025. "Correction: Marrero et al. Kallopterolides A–I, a New Subclass of seco-Diterpenes Isolated from the Southwestern Caribbean Sea Plume Antillogorgia kallos. Molecules 2024, 29, 2493" Molecules 30, no. 14: 2929. https://doi.org/10.3390/molecules30142929
APA StyleMarrero, J., Amador, L. A., Novitskiy, I. M., Kutateladze, A. G., & Rodríguez, A. D. (2025). Correction: Marrero et al. Kallopterolides A–I, a New Subclass of seco-Diterpenes Isolated from the Southwestern Caribbean Sea Plume Antillogorgia kallos. Molecules 2024, 29, 2493. Molecules, 30(14), 2929. https://doi.org/10.3390/molecules30142929




