Tetracyclic Bis-Piperidine Alkaloids: Structures, Bioinspired Synthesis, Synthesis, and Bioactivities
Abstract
1. Introduction
2. General Structural Features, Natural Occurrence, and Stereochemistry
2.1. General Structural Features
2.2. Natural Occurrence and Geographic Distribution of TcBPAs
2.3. Stereochemistry
3. Biosynthetic Hypotheses and Bioinspired Syntheses
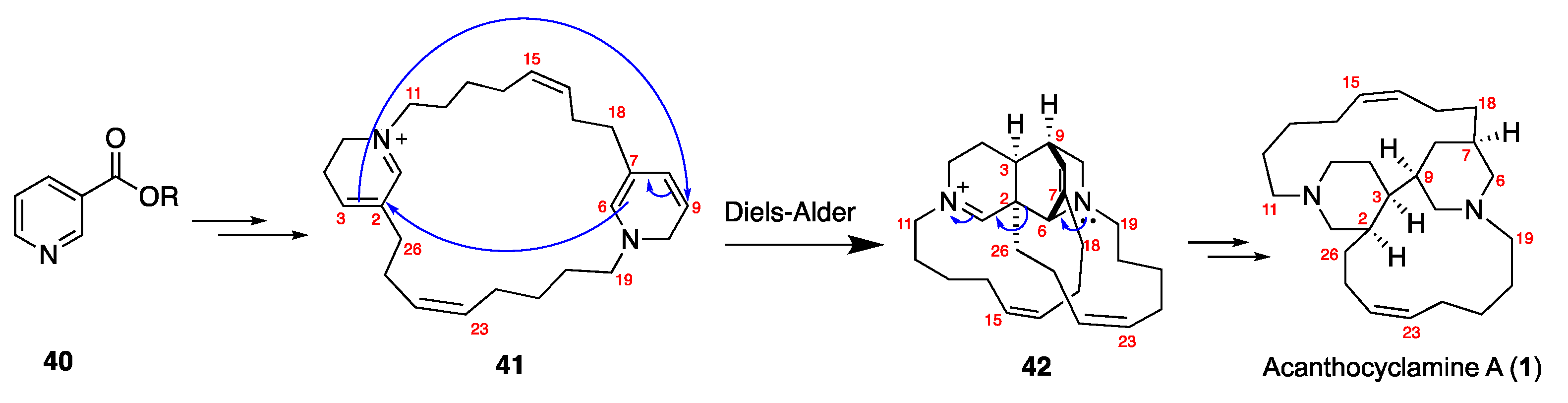
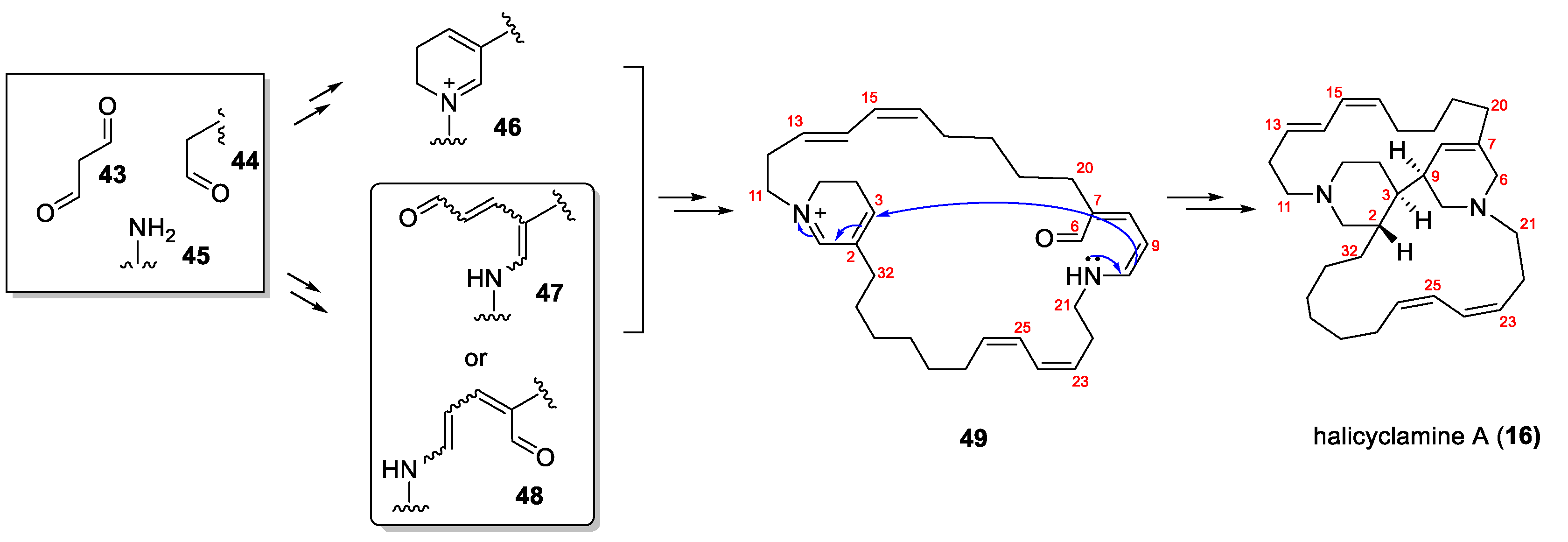
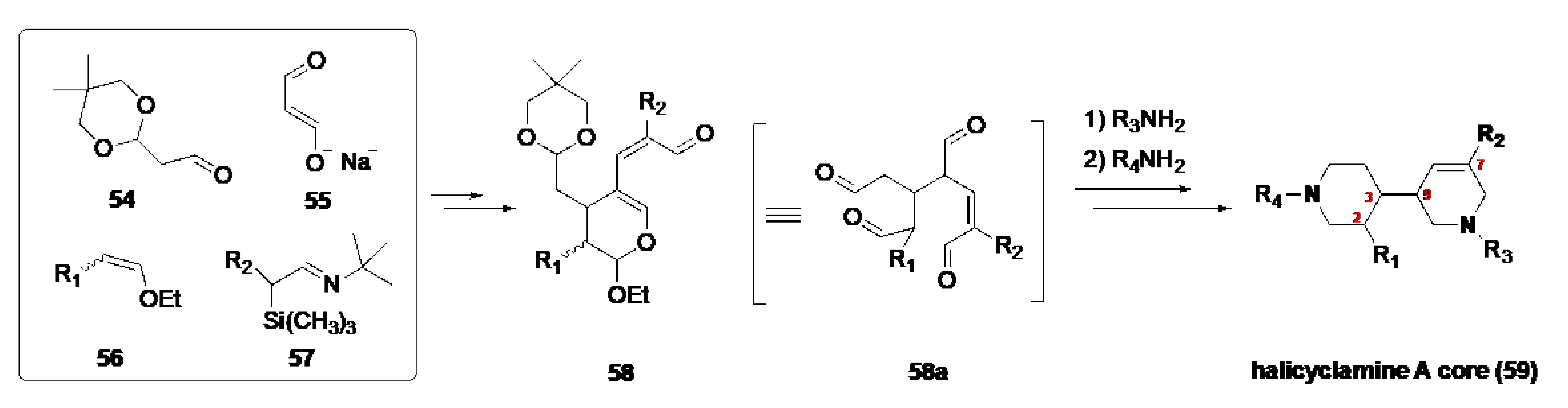
3.1. The Bioinspired Synthesis of TcBPAs Based on the Baldwin–Whitehead Hypothesis
3.2. Alternative Bioinspired Synthetic Model: The Marazano Modification
3.3. Stereochemical and Experimental Insights into TcBPA Bioinspired Synthesis
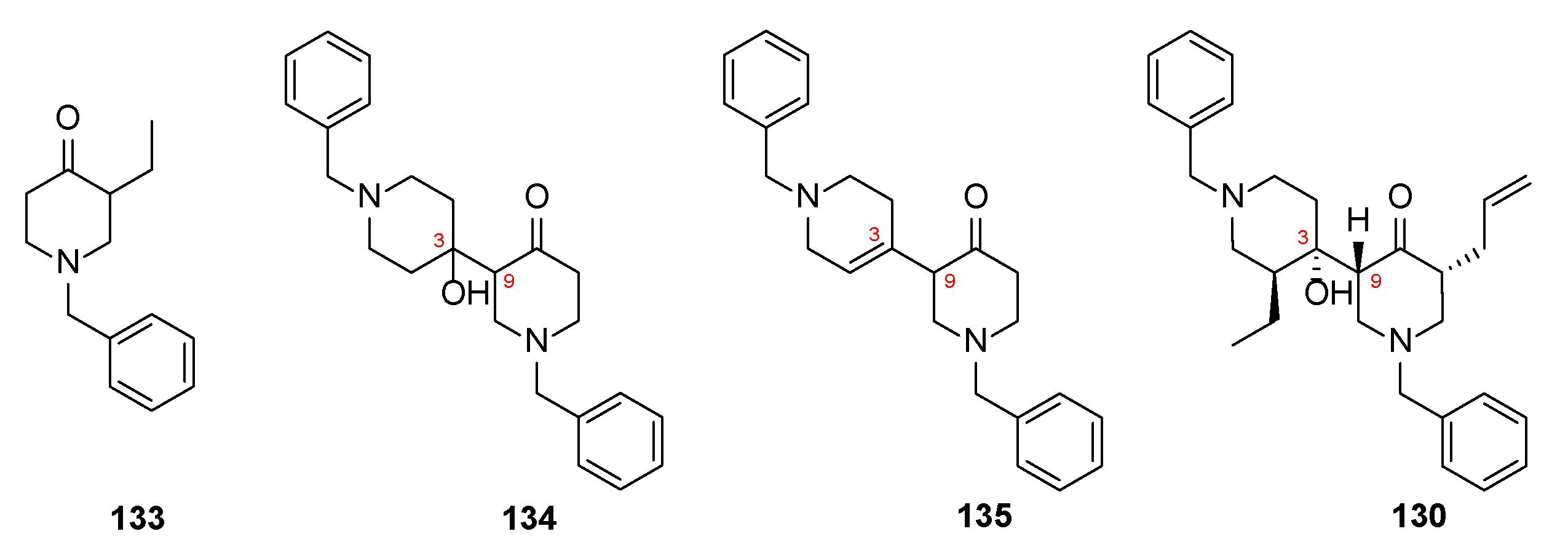
4. Chemical Synthesis
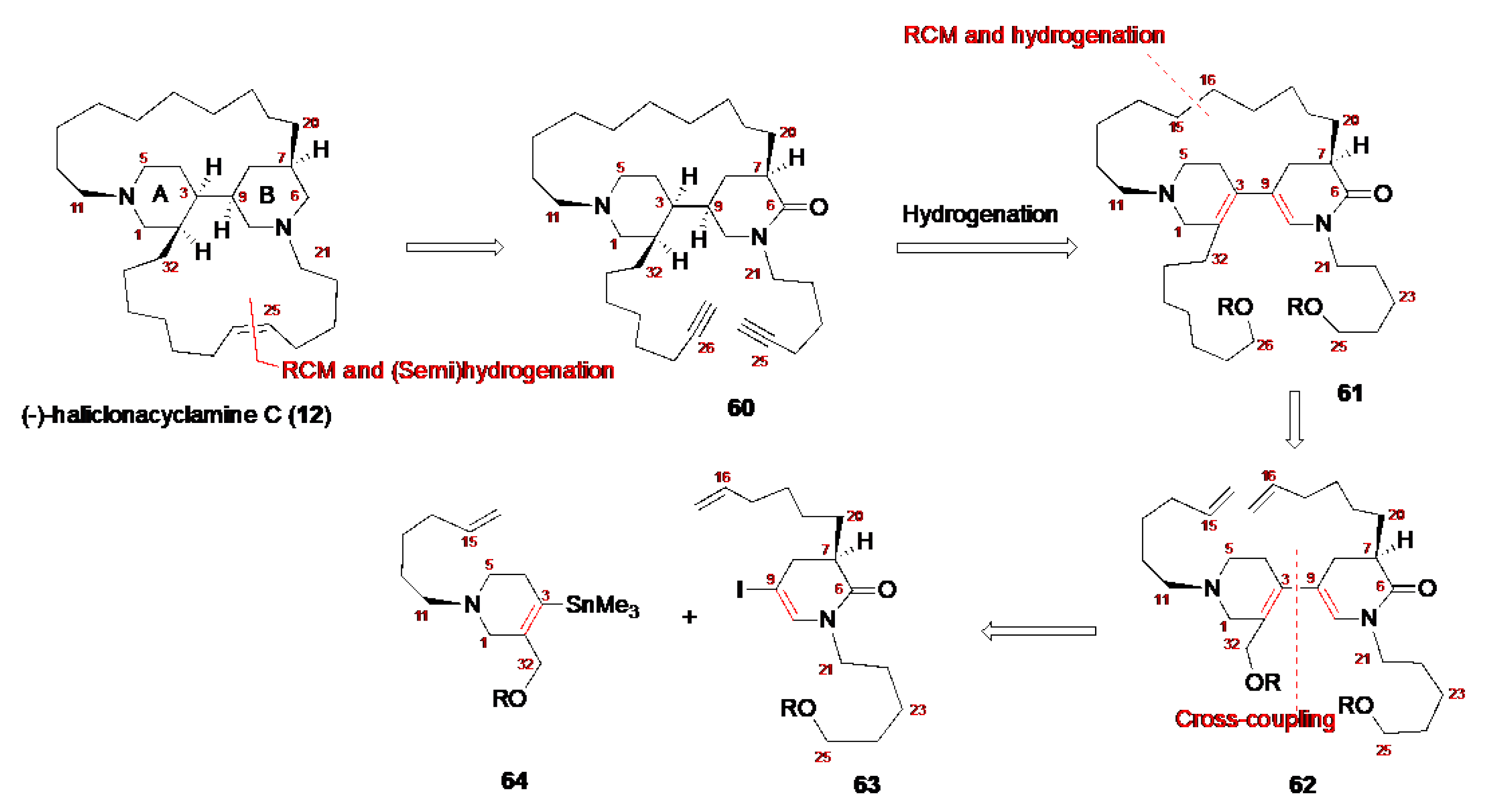
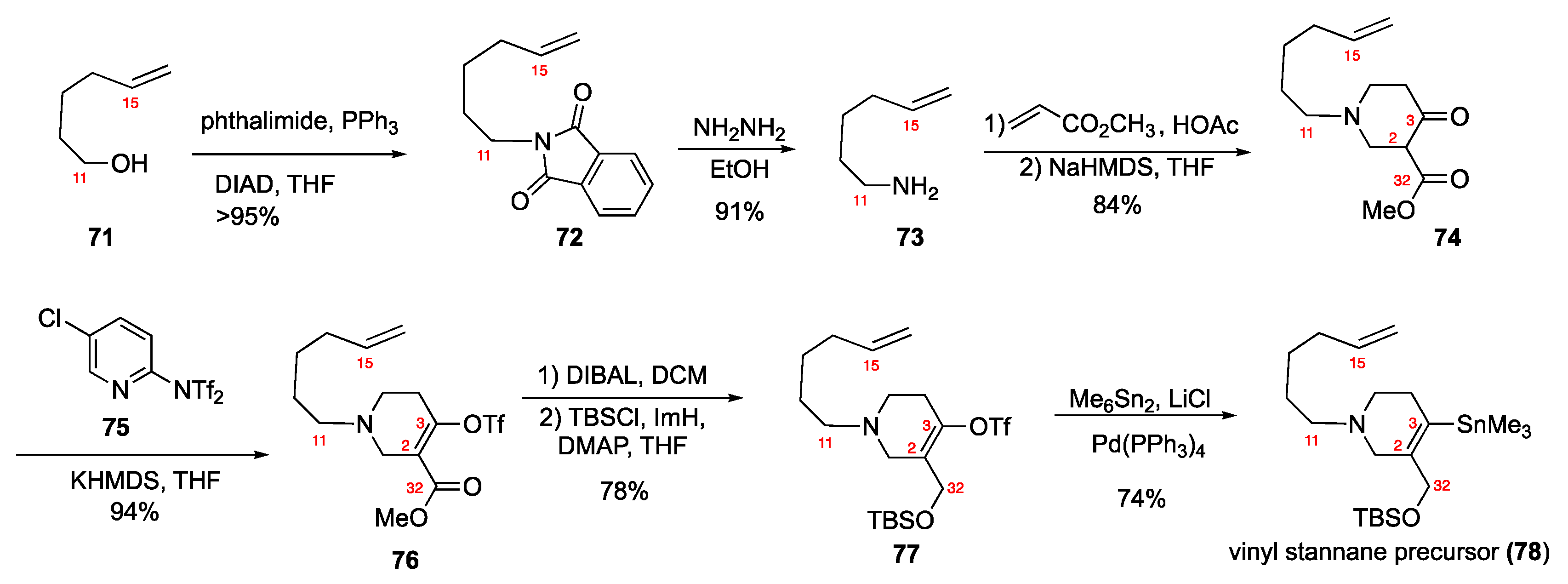
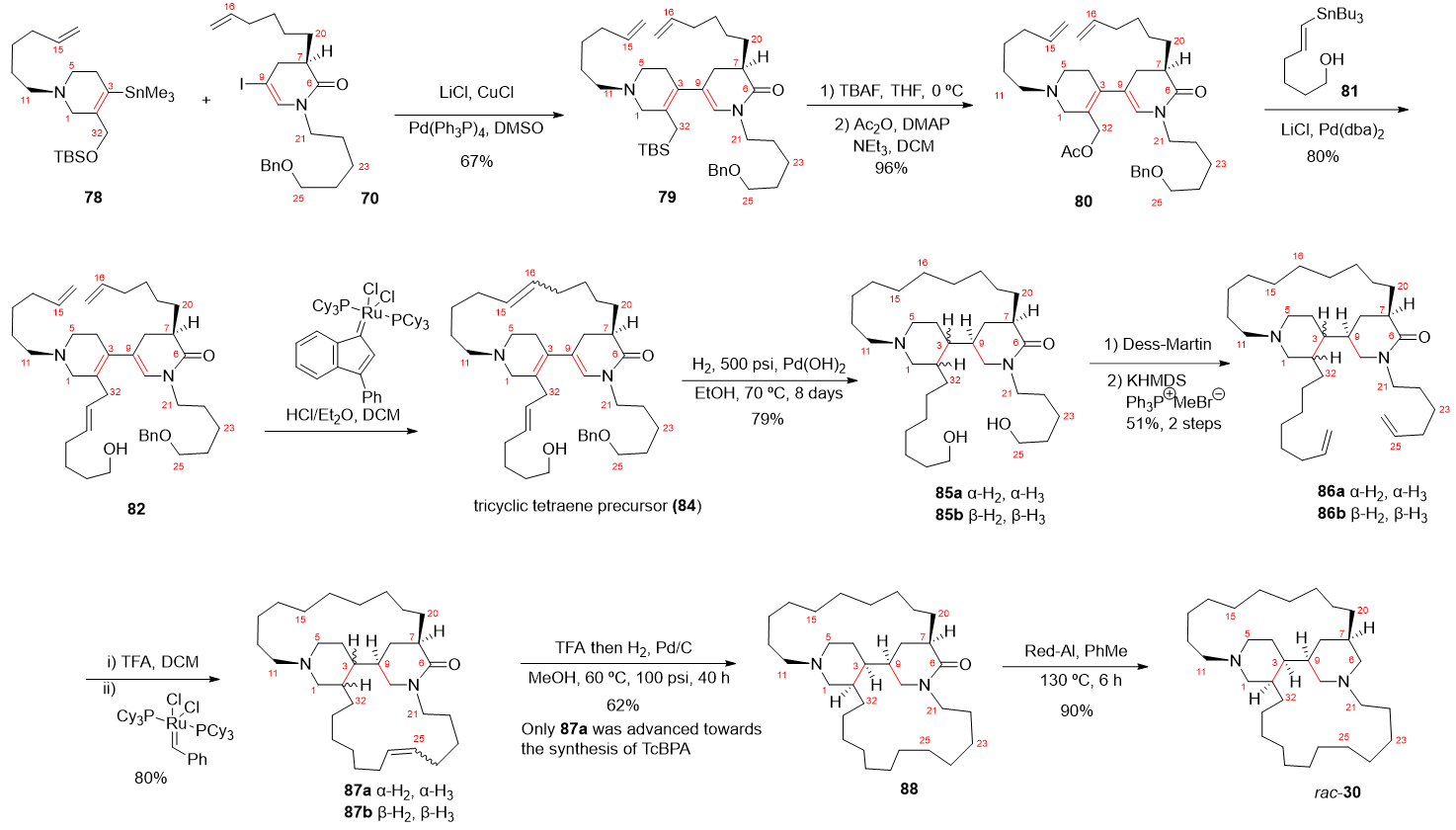
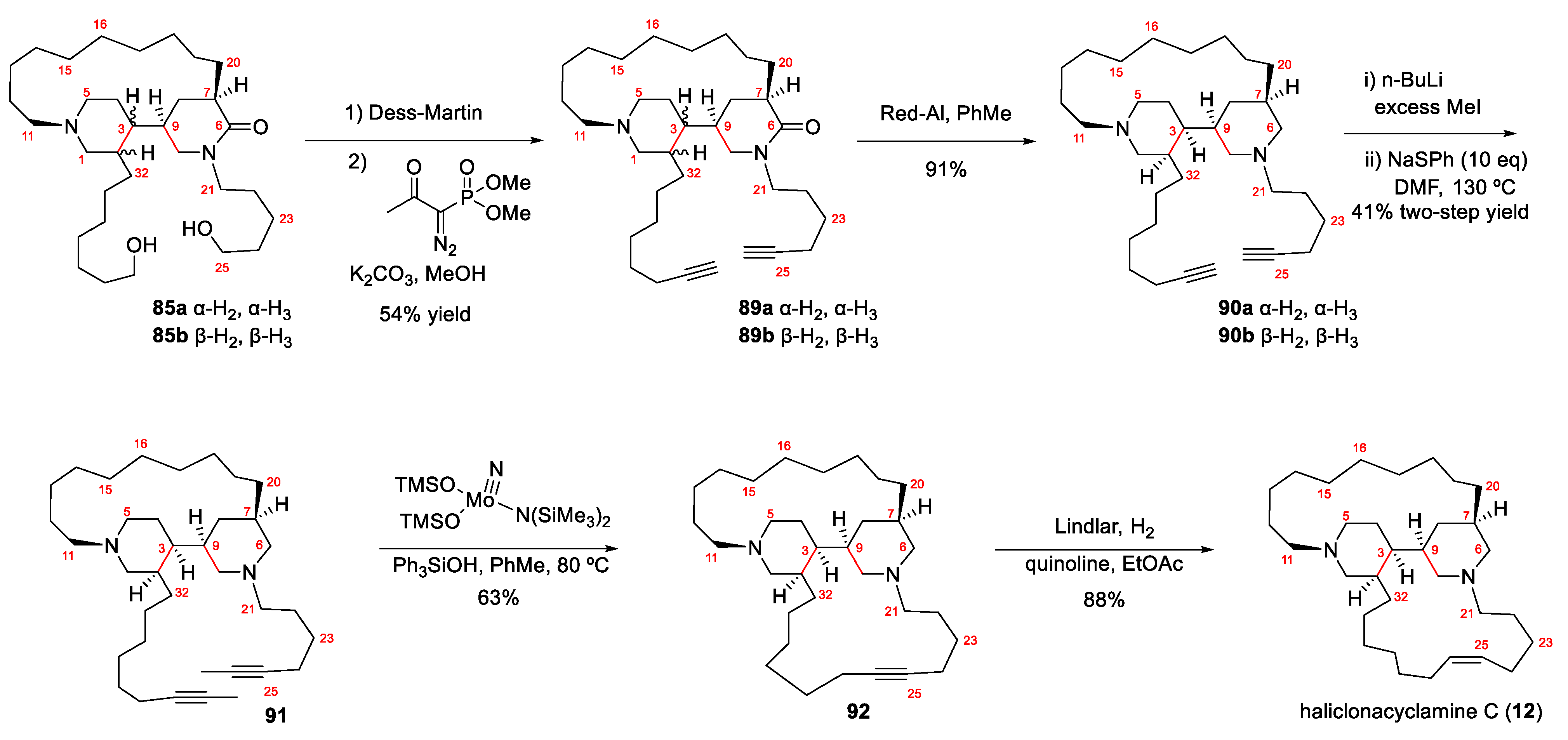
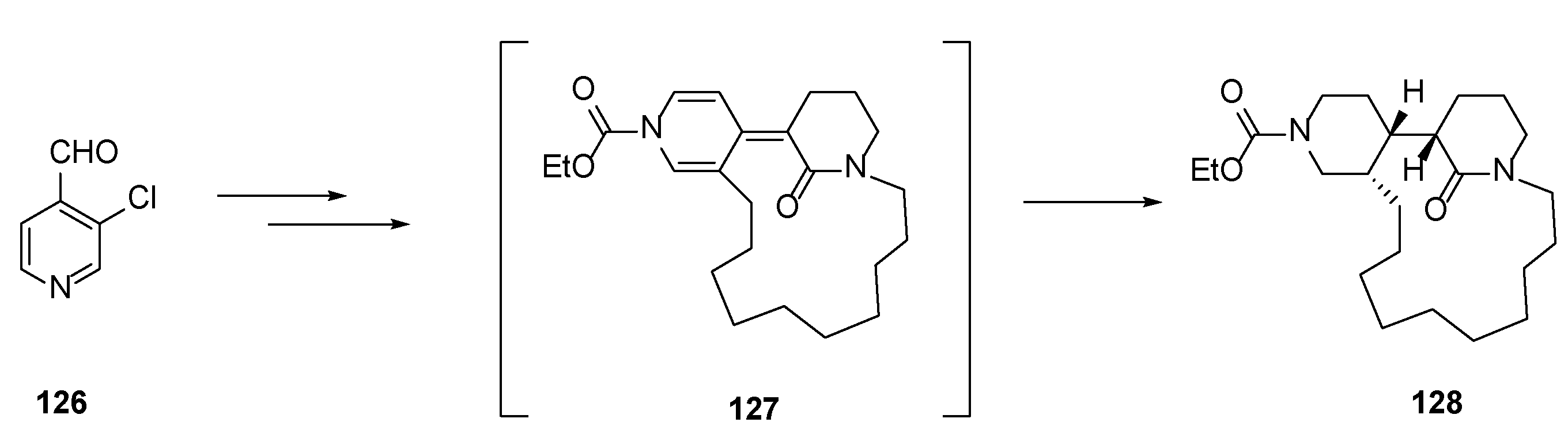
4.1. Total Synthesis of Halichonacyclamine C (12) and Tetrahydrohaliclonacyclamine A (89)
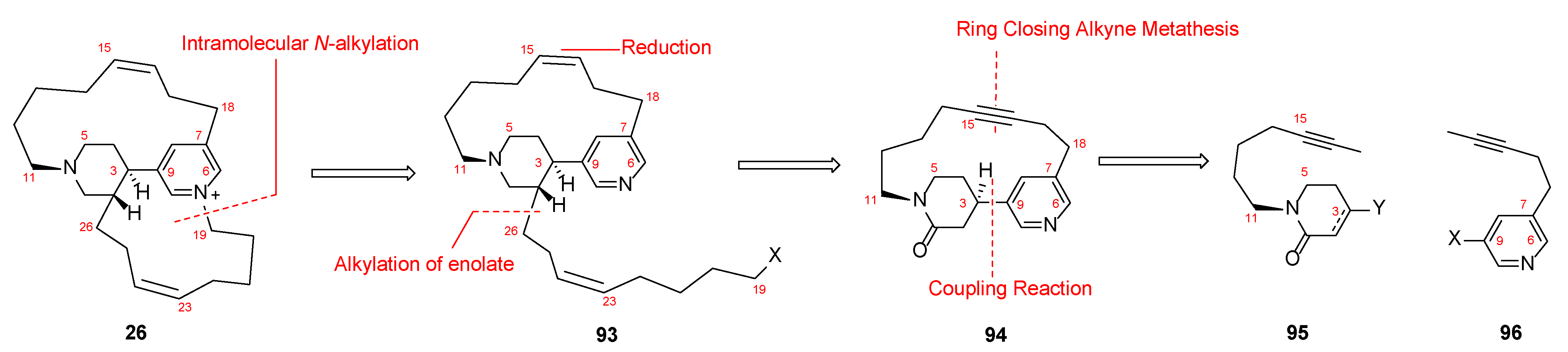
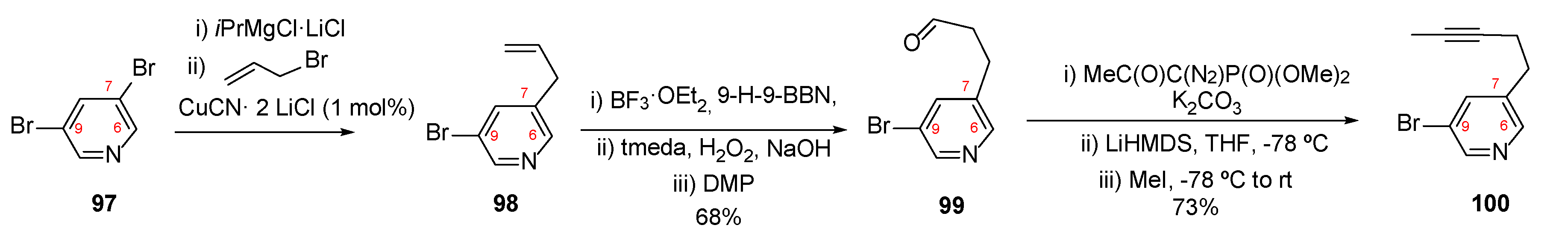
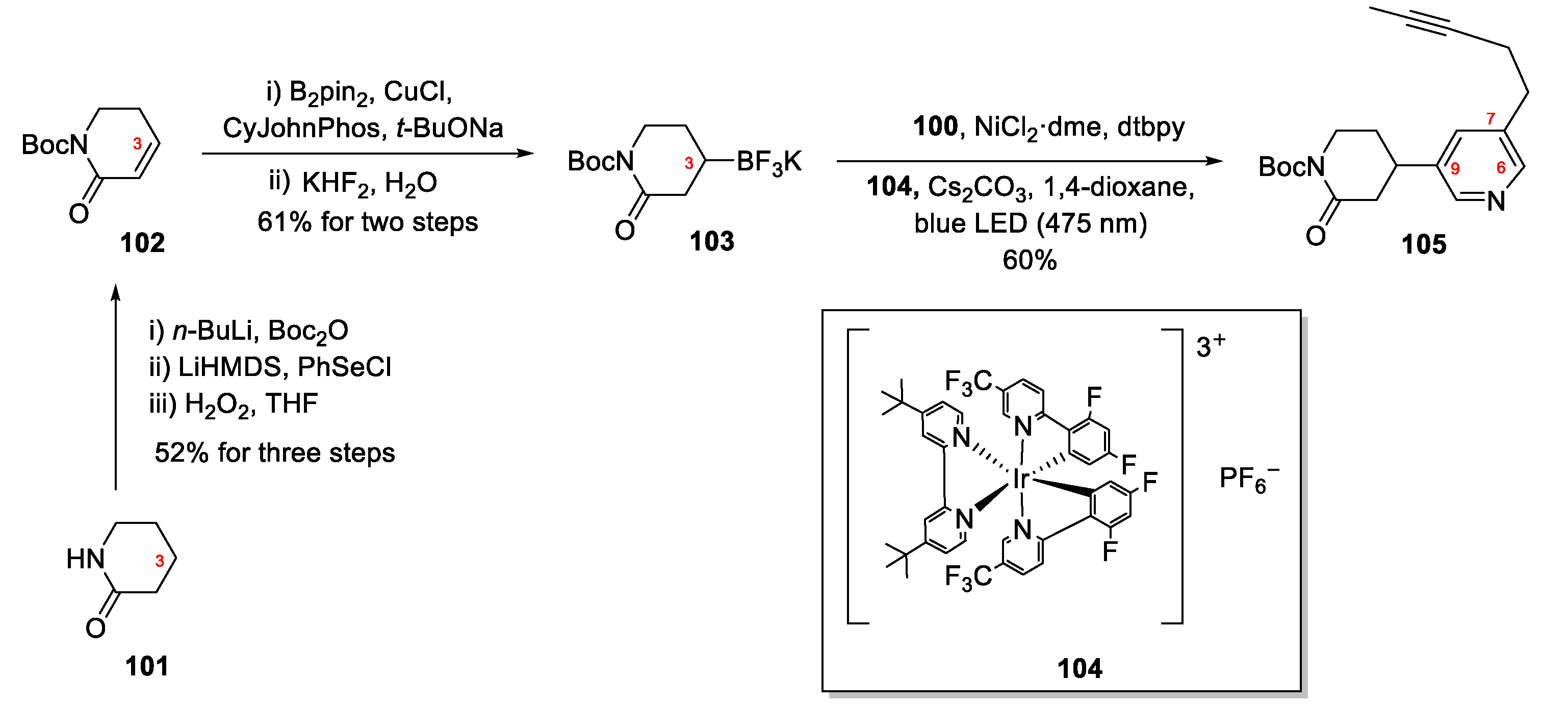
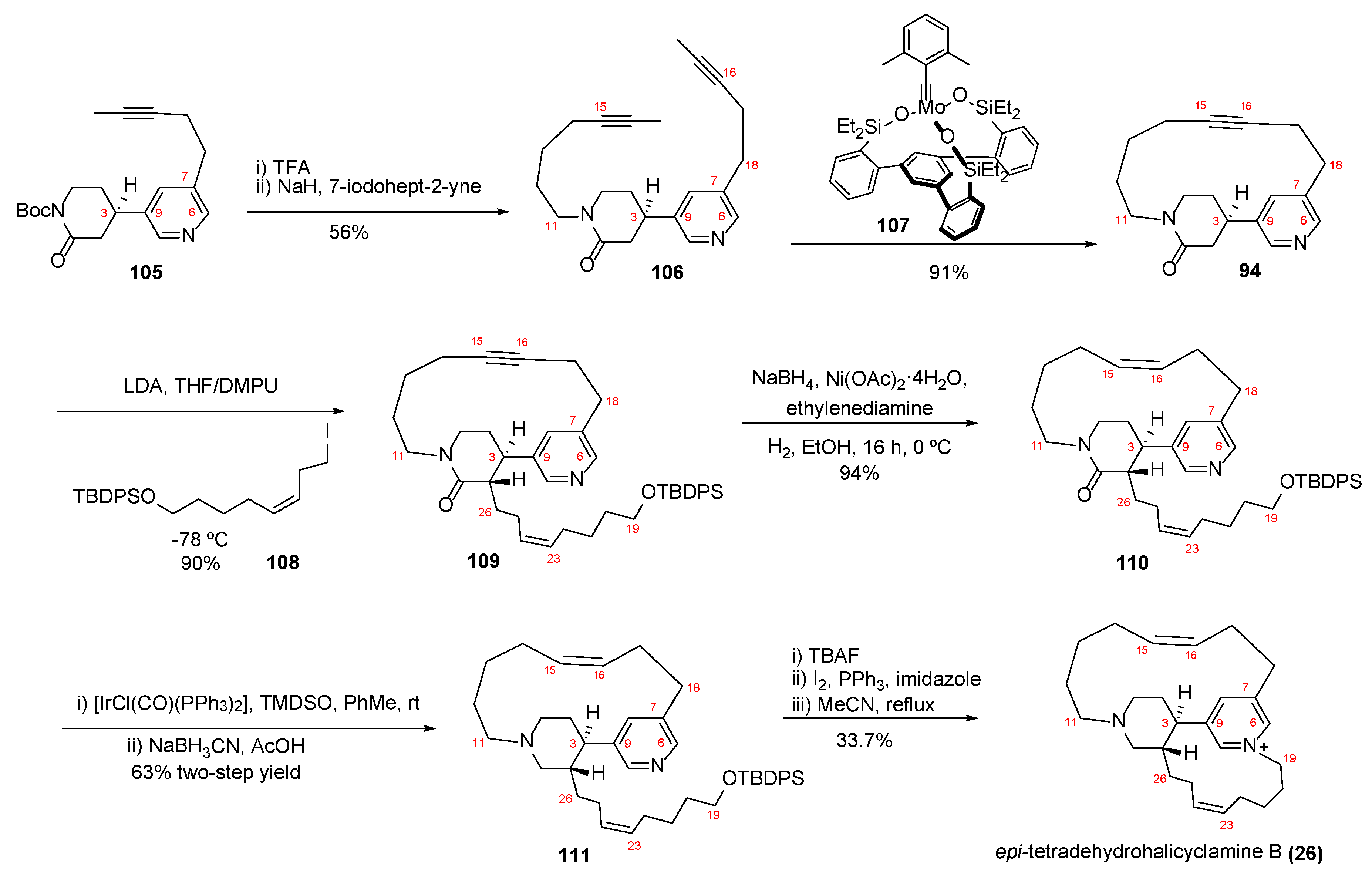
4.2. Synthesis of Epi-Tetradehydrohalicyclamine B (26)
4.3. Dimerization of 1,6-Dihydropyridines Towards Halicyclamine Mimics
4.4. Synthesis of Halicyclamine-like Scaffold via Transannular
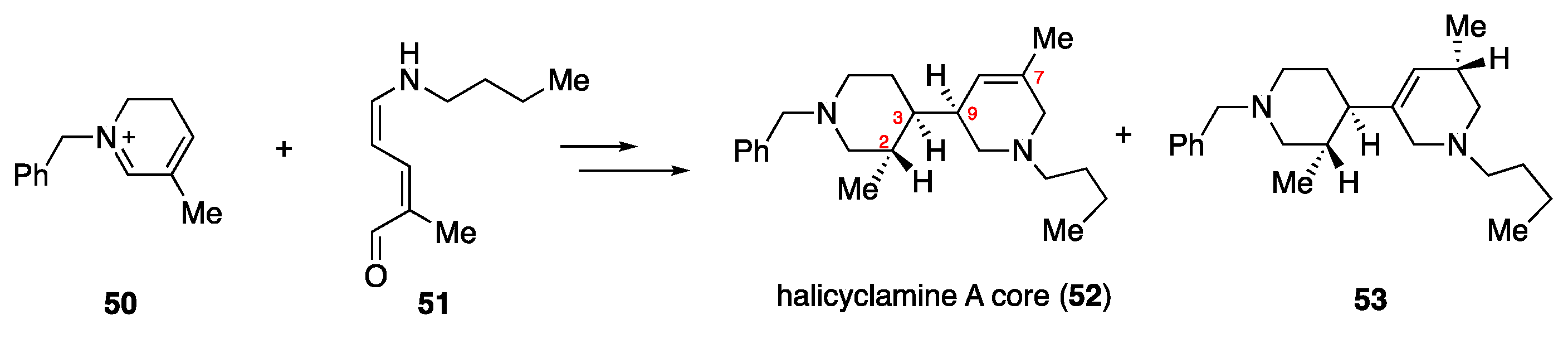
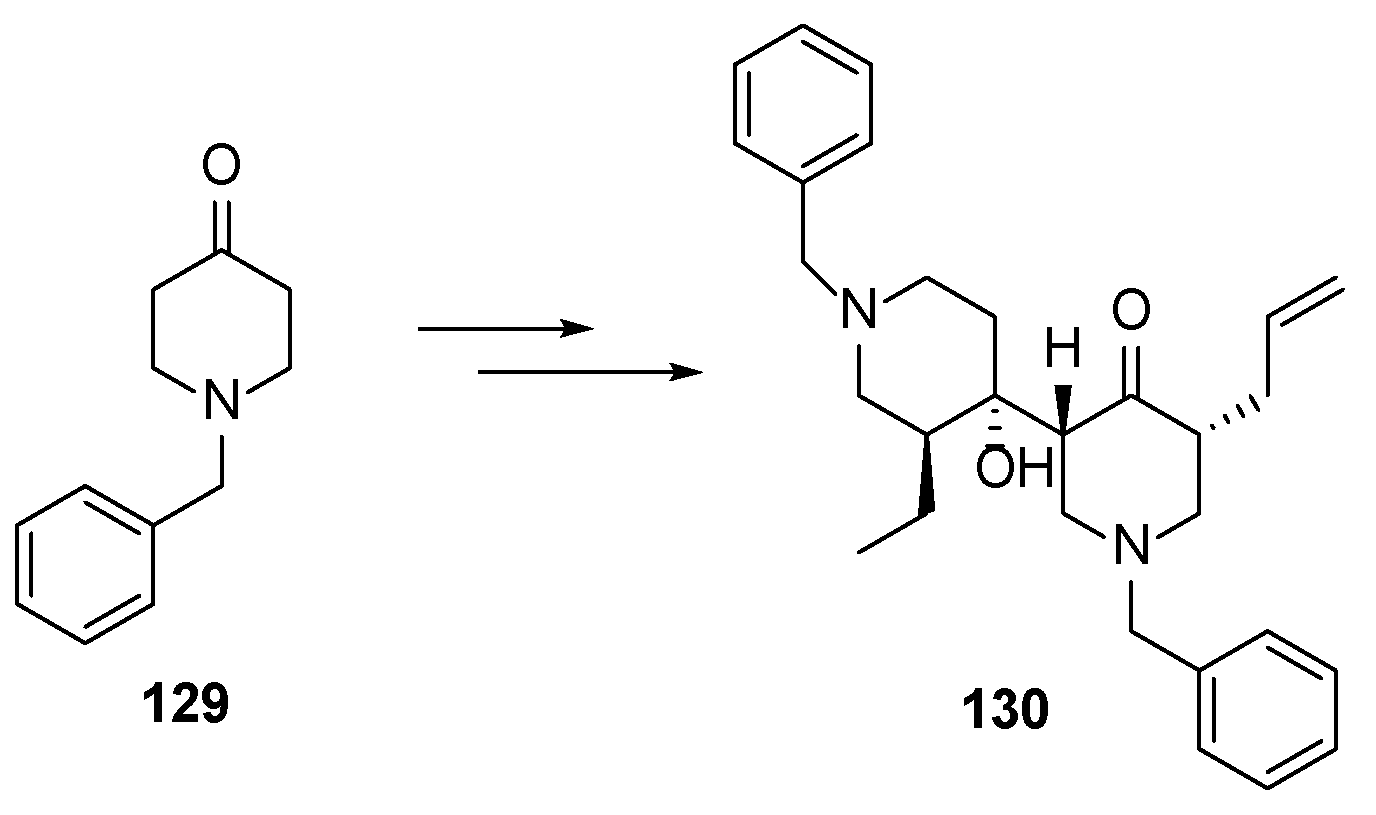
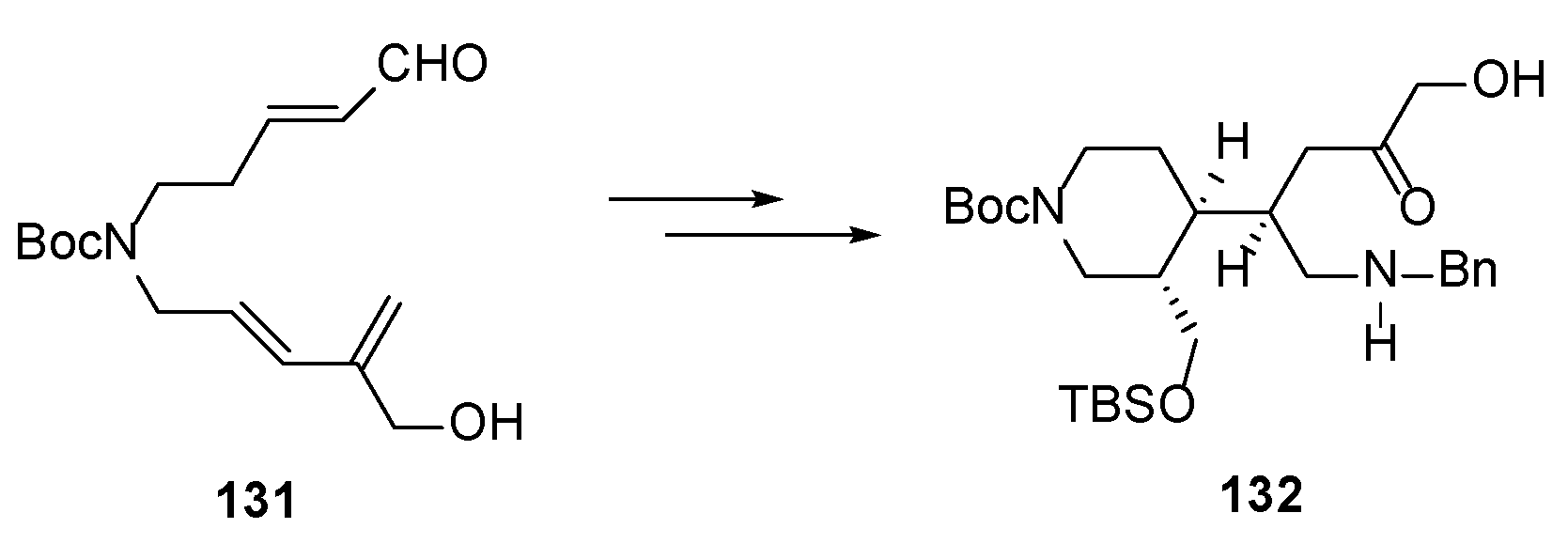
4.5. Model Synthetic Studies
5. Bioactivities and Pharmacological Potential
5.1. Anti-Proliferative Activity of TcBPAs on Cancer Cells
5.2. Antimicrobial Activities
5.3. Other Bioactivities of TcBPAs
6. Future Directions and Challenges
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CD | Circular dichroism |
| 1,6-DHP | 1,6-dihydropyridine |
| DIBAL-H | Diisobutylaluminum hydride |
| DMF | Dimethylformamide |
| DMPU | N,N’-dimethylpropyleneurea |
| ECD | Electronic circular dichroism |
| ECDD | Exciton-coupled circular dichroism |
| LDA | Lithium diisopropylamide |
| LiHMDS | Lithium hexamethyldisilazide |
| NaBH4 | Sodium borohydride |
| NaH | Sodium hydride |
| NaHMDS | Sodium hexamethyldisilazide |
| RCAM | Ring-closing alkyne metathesis |
| RCM | Ring-closing metathesis |
| SAR | Structure–activity relationship |
| TBAF | Tetra-n-butylammonium fluoride |
| TcBPAs | Tetracyclic bis-piperidine alkaloids |
| TFA | Trifluoacetic acid |
| TFAA | Trifluoroacetic anhydride |
| TMDSO | 1,1,3,3-tetramethyldisiloxane |
References
- Hajar, R. History of medicine timeline. Heart Views 2015, 16, 43–45. [Google Scholar] [PubMed]
- Heinrich, M.; Mah, J.; Amirkia, V. Alkaloids used as medicines: Structural phytochemistry meets biodiversity–An update and forward look. Molecules 2021, 26, 1836. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [PubMed]
- Daley, S.; Cordell, G.A. Alkaloids in contemporary drug discovery to meet global disease needs. Molecules 2021, 26, 3800. [Google Scholar] [CrossRef]
- Croteau, R.; Kutchan, T.M.; Lewis, N.G. Natural products (secondary metabolites). In Biochemistry and molecular biology of plants; Buchanan, B., Gruissem, R., Jones, R., Eds.; John Wiley & Sons Inc.: Somerset, NJ, USA, 2000. [Google Scholar]
- Mondal, A.; Gandhi, A.; Fimognari, C.; Atanasov, A.G.; Bishayee, A. Alkaloids for cancer prevention and therapy: Current progress and future perspectives. Eur. J. Pharmacol. 2019, 858, 172472. [Google Scholar]
- Lansakara, A.I.; Mariappan, S.V.S.; Pigge, F.C. Alkylidene dihydropyridines as synthetic intermediates: Model studies toward the synthesis of the bis(piperidine) alkaloid xestoproxamine C. J. Org. Chem. 2016, 81, 10266–10278. [Google Scholar] [PubMed]
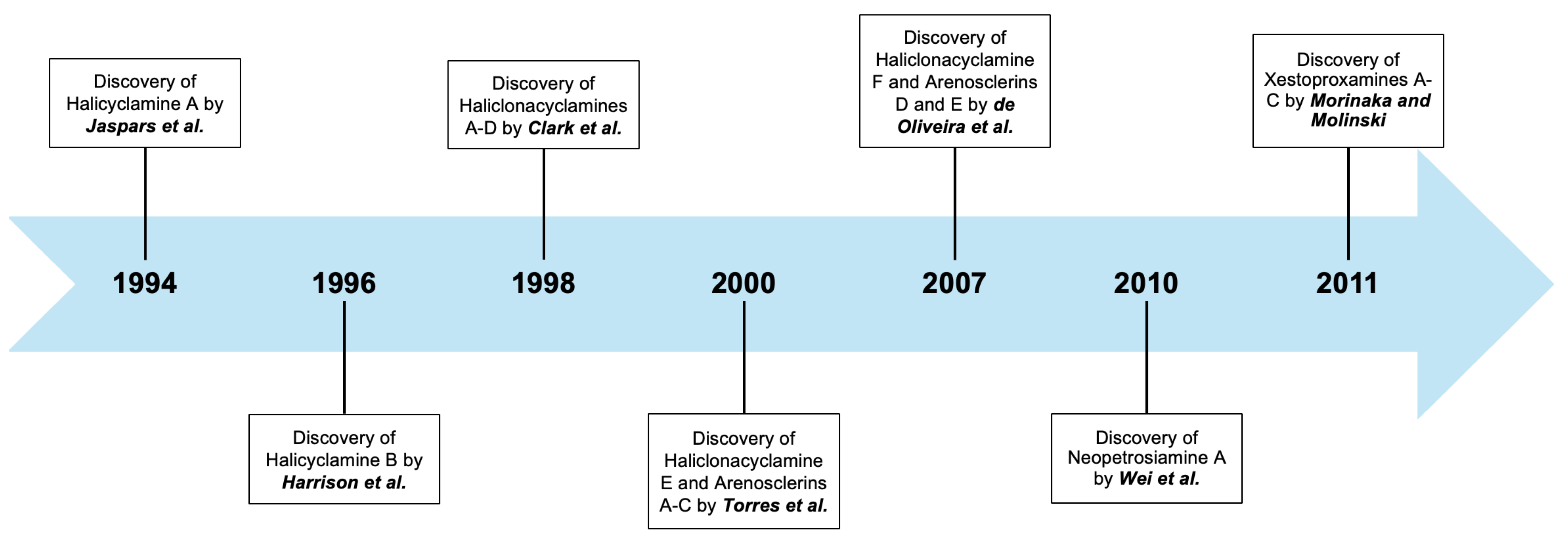
- Jaspars, M.; Pasupathy, V.; Crews, P.A. Tetracyclic diamine alkaloid, halicyclamine A, from the marine sponge Haliclona sp. J. Org. Chem. 1994, 59, 3253–3255. [Google Scholar]
- De Oliveira, J.H.L.; Nascimento, A.M.; Kossuga, M.H.; Cavalcanti, B.C.; Ressoa, C.O.; Moraes, M.O.; Ferreira, A.G.; Hajdu, E.; Pinheiro, U.S.; Berlinck, R.G.S. Cytotoxic alkylpiperidine alkaloids from the Brazilian marine sponge Pachychalina alcaloidifera. J. Nat. Prod. 2007, 70, 538–543. [Google Scholar]
- Torres, Y.R.; Berlinck, R.G.S.; Magalhães, A.; Schefer, A.B.; Ferreira, A.G.; Hajdu, E.; Muricy, G. Arenosclerins A−C and haliclonacyclamine E, new tetracyclic alkaloids from a Brazilian endemic Haplosclerid sponge Arenosclera Brasiliensis. J. Nat. Prod. 2000, 63, 1098–1105. [Google Scholar]
- Harrison, B.; Talapatra, S.; Lobkovsky, E.; Clardy, J.; Crews, P. The structure and biogenetic origin of (-) halicyclamine B from a Xestospongia Sponge. Tetrahedron Lett. 1996, 37, 9151–9154. [Google Scholar]
- Clark, R.J.; Field, K.L.; Charan, R.D.; Garson, M.J.; Brereton, M.; Willis, A.C. The Haliclonacyclamines, cytotoxic tertiary alkaloids from the tropical marine sponge Haliclona sp. Tetrahedron 1998, 54, 8811–8826. [Google Scholar]
- Morinaka, B.I.; Molinski, T.F. Xestoproxamines A-C from Neopetrosia proxima. Assignment of absolute stereostructure of bis-piperidine alkaloids by integrated degradation-CD analysis. J. Nat. Prod. 2011, 74, 430–440. [Google Scholar]
- Wei, X.; Nieves, K.; Rodriguez, A.D. Neopetrosiamine A, biologically active bis-piperidine alkaloid from the Caribbean Sea sponge Neopetrosia proxima. Bioorg. Med. Chem. Lett. 2010, 20, 5905–5908. [Google Scholar]
- Elissawy, A.M.; Soleiman Dehkordi, E.; Mehdinezhad, N.; Ashour, M.L.; Mohammadi Pour, P. Cytotoxic alkaloids derived from marine sponges: A comprehensive review. Biomolecules 2021, 11, 258. [Google Scholar] [CrossRef] [PubMed]
- Esposito, G.; Bourguet-Kondracki, M.L.; Mai, L.H.; Longeon, A.; Teta, R.; Meijer, L.; Van Soest, R.; Mangoni, A.; Costantino, V. Chloromethylhalicyclamine B, a marine-derived protein kinase CK1δ/ε inhibitor. J. Nat. Prod. 2016, 79, 2953–2960. [Google Scholar]
- Dewi, A.S.; Hadi, T.A.; Fajarningsih, N.D.; Blanchfield, J.T.; Bernhardt, P.V.; Garson, M.J. Acanthocyclamine A from the Indonesian marine sponge Acanthostrongylophora ingens. Aust. J. Chem. 2014, 67, 1205. [Google Scholar]
- Esposito, G.; Mai, L.H.; Longeon, A.; Mangoni, A.; Durieu, E.; Meijer, L.; Van Soest, R.; Costantino, V.; Bourguet-Kondracki, M.-L. A collection of bioactive nitrogen-containing molecules from the marine sponge Acanthostrongylophora ingens. Mar. Drugs 2019, 17, 472. [Google Scholar]
- Chill, L.; Yosief, T.; Kashman, Y. Halichondramine, a new tetracyclic bispiperidine alkaloid from the marine sponge Halichondria Sp. J. Nat. Prod. 2002, 65, 1738–1741. [Google Scholar]
- Charan, R.D.; Garson, M.J.; Brereton, I.M.; Willis, A.C.; Hooper, J.N.A. Haliclonacyclamines A and B, cytotoxic alkaloids from the tropical marine sponge Haliclona sp. Tetrahedron 1996, 52, 9111–9120. Tetrahedron 1996, 52, 9111–9120. [Google Scholar]
- Matsunaga, S.; Miyata, Y.; van Soest, R.W.M.; Fusetani, N. Tetradehydrohalicyclamine A and 22-hydroxyhalicyclamine A, new cytotoxic bispiperidine alkaloids from a marine sponge Amphimedon sp. J. Nat. Prod. 2005, 67, 1758–1760. [Google Scholar]
- Kato, H.; El-Desoky, A.H.; Takeishi, Y.; Nehira, T.; Angkouw, E.D.; Mangindaan, R.E.P.; de Voogd, N.J.; Tsukamoto, S. Tetradehydrohalicyclamine B, a new proteasome inhibitor from the marine sponge Acanthostrongylophora ngens. Bioorg. Med. Chem. Lett. 2018, 29, 8–10. [Google Scholar] [PubMed]
- Arai, M.; Ishida, S.; Setiawan, A.; Kobayashi, M. Haliclonacyclamines, Tetracyclic alkylpiperidine alkaloids, as anti-dormant mycobacterial substances from a marine sponge of Haliclona sp. Chem. Pharm. Bull. 2009, 57, 1136–1138. [Google Scholar]
- Mudianta, I.W.; Katavic, P.L.; Lambert, L.K.; Hayes, P.Y.; Banwell, M.G.; Munro, M.H.G.; Bernhardt, P.V.; Garson, M.J. Structure and absolute configuration of 3-alkylpiperidine alkaloids from an Indonesian sponge of the genus Halichondria. Tetrahedron 2010, 66, 2752–2760. [Google Scholar]
- Parsons, S. Determination of absolute configuration using X-ray diffraction. Tetrahedron: Asymmetry 2017, 28, 1304–1313. [Google Scholar]
- Baldwin, J.E.; Whitehead, R.C. On the biosynthesis of manzamines. Tetrahedron Lett. 1992, 33, 2059–2062. [Google Scholar]
- Dalling, A.G.; Späth, G.; Fürstner, A. Total synthesis of the tetracyclic pyridinium alkaloid epi-tetradehydrohalicyclamine B. Angew. Chem. Int. Ed. 2022, 61, e202209651. [Google Scholar]
- Rodriguez, J.; Crews, P. Revised structure of xestocyclamine A and description of a new analogue. Tetrahedron Lett. 1994, 35, 4719–4722. [Google Scholar]
- Cutignano, A.; Tramice, A.; De Caro, S.; Villani, G.; Cimino, G.; Fontana, A. Biogenesis of 3-alkylpyridine alkaloids in the marine Mollusc Haminoea orbigyana. Angew. Chem. Int. Ed. 2003, 42, 2633. [Google Scholar]
- Kaiser, A.; Billot, X.; Gateau-Olesker, A.; Marazano, C.; Das, B.C. Selective Entry to the dimeric or oligomeric pyridinium sponge macrocycles via aminopentadienal derivatives. Possible biogenetic relevance with manzamine alkaloids. J. Am. Chem. Soc. 1998, 120, 8026–8034. [Google Scholar]
- Jakubowicz, K.; Abdeljelil, K.B.; Herdemann, M.; Martin, M.-T.; Gateau-Olesker, A.; Mourabit, A.A.; Marazano, C.; Das, B.C. Reactions of aminopentadienal derivatives with 5,6-dihydropyridinium salts as an approach to manzamine alkaloids based upon biogenetic considerations. J. Org. Chem. 1999, 64, 7381–7387. [Google Scholar]
- Tang, Y.; Zhu, L.; Hong, R. Madangamine alkaloids: Madness and tranquility. Tetrahedron Chem. 2022, 3, 100025. [Google Scholar]
- Sanchez-Salvatori, M.R.; Marazano, C. An access to the bicyclic nucleus of the sponge alkaloid halicyclamine A by successive condensation of malondialdehyde units, aldehyde derivatives, and primary amines. J. Org. Chem. 2003, 68, 8883–8889. [Google Scholar]
- Althagbi, H.I.; Alarif, W.M.; Al-Footy, K.O.; Abdel-Lateff, A. Marine-derived macrocyclic alkaloids (MDMAs): Chemical and biological diversity. Mar. Drugs 2020, 18, 368. [Google Scholar]
- Wayama, T.; Arai, Y.; Oguri, H. Regiocontrolled dimerization of densely functionalized 1,6-dihydropyridines for the biomimetic synthesis of a halicyclamine-type scaffold by preventing disproportionation. J. Org. Chem. 2022, 87, 5938–5951. [Google Scholar] [PubMed]
- Wayama, T.; Oguri, H. Synthesis of a halicyclamine-type macrocyclic scaffold via biomimetic transannular cyclization. Org. Lett. 2023, 25, 3596–3601. [Google Scholar] [PubMed]
- Smith, B.J.; Qu, T.; Mulder, M.; Noetzel, M.J.; Lindsley, C.W.; Sulikowski, G.A. Synthesis and bioactivity of (±)-tetrahydrohaliclonacyclamine A. Tetrahedron 2010, 66, 4805–4810. [Google Scholar]
- Smith, B.J.; Sulikowski, G.A. Total synthesis of (±)-haliclonacyclamine C. Angew. Chem. Int. Ed. 2010, 49, 1599–1602. [Google Scholar]
- Sinigagia, I.; Nguyen, T.M.; Wypych, J.-C.; Delpech, B.; Marazano, C. Concise access to a model of the marine alkaloid halicyclamine A through macrocycle-forming addition of a 5-aminopenta-2,4-dienal onto a 2,3-dihydropyridinium salt. Chem. Eur. J. 2010, 16, 3594–3597. [Google Scholar]
- Banwell, M.G.; Coster, M.J.; Hungerford, N.L.; Garson, M.J.; Su, S.; Kotze, A.C.; Munro, M.H.G. 3,4′-Linked bis(piperidines) related to the haliclonacyclamine class of marine alkaloids: Synthesis using crossed-aldol chemistry and preliminary biological evaluations. Org. Biomol. Chem. 2012, 10, 154–161. [Google Scholar]
- Molander, G.A.; Cadoret, F. Synthesis of the stereogenic triad of the halicyclamine A core. Tetrahedron Lett. 2011, 52, 2199–2202. [Google Scholar]
- Torres, Y.R.; Berlinck, R.G.S.; Nascimento, G.G.F.; Fortier, S.C.; Pessoa, C.; de Moraes, M.O. Antibacterial activity against resistant bacteria and cytotoxicity of four alkaloid toxins isolated from the marine sponse Arenosclera brasiliensis. Toxicon 2002, 40, 885–891. [Google Scholar] [PubMed]
- Prasad, V.V.; Gopalan, R.O. Continued use of MDA-MB-435, a melanoma cell line, as a model for human breast cancer, even in Year 2014. NPJ Breast Cancer 2015, 1, 15002. [Google Scholar] [PubMed]
- Garcia Jimenez, D.; Poongavanam, V.; Kihlberg, J. Macrocycles in drug discovery─learning from the past for the future. J. Med. Chem. 2023, 66, 5377–5396. [Google Scholar] [PubMed]
- Chambers, A.F. MDA-MB-435 and M14 Cell Lines. Identical but not M14 Melanoma. Cancer Res. 2009, 69, 5292–5293. [Google Scholar]
- Mani, L.; Petek, S.; Valentin, A.; Chevalley, S.; Folcher, E.; Aalbersberg, W.; Debitus, C. The in vivo anti-plasmodial activity of haliclonacyclamine A, an alkaloid from the marine sponge, Haliclona sp. Nat. Prod. Res. 2011, 25, 1923–1930. [Google Scholar]







| Comp. Number | Name | Source Organism | Collection Location | Ref |
|---|---|---|---|---|
| 1 | Acanthocyclamine A | Acanthostrongylophora ingens | South Sulawesi, Indonesia | [17,18] |
| 2 | Arenosclerin A | Arenosclera brasiliensis | Endemic to the Southeastern Brazilian coast | [10] |
| 3 | Arenosclerin B | Arenosclera brasiliensis | Endemic to the Southeastern Brazilian coast | [10] |
| 4 | Arenosclerin C | Arenosclera brasiliensis | Endemic to the Southeastern Brazilian coast | [10] |
| 5 | Arenosclerin D | Pachychalina alcaloidifera | Brazilian | [9] |
| 6 | Arenosclerin E | Pachychalina alcaloidifera | Brazilian | [9] |
| 7 | Chloromethyltetradehydrohalicyclamine B | Acanthostrongylophora ingens | South Sulawesi, Indonesia | [18] |
| 8 | Chloromethylhalicyclamine B | Acanthostrongylophora ingens | South Sulawesi, Indonesia | [16,18] |
| 9 | Halichondramine | Halichondria sp., Unknown species | Red Sea, Dahlak archipelago, Eritrea | [19] |
| 10 | (−)-Haliclonacyclamine A | Haliclona sp. Unknown species | Heron Island, Great Barrier Reef | [20] |
| 11 | (+)-Haliclonacyclamine B | Haliclona sp. Unknown species | Heron Island, Great Barrier Reef | [20] |
| 12 | Haliclonacyclamine C | Haliclona sp. Unknown species | Heron Island, Great Barrier Reef | [12] |
| 13 | Haliclonacyclamine D | Haliclona sp. Unknown species | Heron Island, Great Barrier Reef | [12] |
| 14 | Haliclonacyclamine E | Arenosclera brasiliensis | Endemic to the Southeastern Brazilian coast | [10] |
| 15 | Haliclonacyclamine F | Pachychalina alcaloidifera | IIha do Pai, Niteroi, Rio de Janerio | [9] |
| 16 | Halicyclamine A | Haliclona sp. Unknown species Amphimedon sp. Unknown species | Biak, Indonesia Southern Japan | [8,21] |
| 17 | (−)-Halicyclamine B | Xestospongia sp. Acanthostrongylophora ingens | Sangihe Islands, Indonesia South Sulawesi, Indonesia | [11,18,22] |
| 18 | 22-hydroxyhaliclonacyclamine B | Halichondria sp. Unknown species | Flores Island, Indonesia | [23] |
| 19 | 22-hydroxyhalicyclamine A | Amphimedon sp. Unknown species | Southern Japan | [21] |
| 20 | Neopetrosiamine A | Neopetrosia proxima | Mona Island, Puerto Rico | [14] |
| 21 | Tetradehydrohalicyclamine A | Amphimedon niphatidae | Lojima Island, Southern Japan | [21] |
| 22 | 2-epi-Tetradehydrohaliclonacyclamine A | Halichondria sp. Unknow species | Tulamben Bay, Bali, Indonesia | [24] |
| 23 | Tetradehydrohaliclonacyclamine A mono-N-oxide | Halichondria sp. Unknown species | Tulamben Bay, Bali, Indonesia | [24] |
| 24 | Tetrahydrohaliclonacyclamine A | Halichondria sp. Unknown species | Tulamben Bay, Bali, Indonesia | [24] |
| 25 | Tetradehydrohalicyclamine B | Acanthostrongylophora ingens | South Sulawesi, Indonesia | [18,22] |
| 26 | epi-Tetradehydrohalicyclamine B | Acanthostrongylophora ingens | South Sulawesi, Indonesia | [18] |
| 27 | Xestoproxamine A | Neopetrosia proxima | Bahamas | [13] |
| 28 | Xestoproxamine B | Neopetrosia proxima | Bahamas | [13] |
| 29 | Xestoproxamine C | Neopetrosia proxima | Bahamas | [13] |
| Bis-piperidine Alkaloids | [a]D Value | C, g/100 mL | Solvent | Absolute Configurations | Reference |
|---|---|---|---|---|---|
| Acanthocylamine A (1) | −26.5 | 0.073 | CHCl3 | 2R,3R,7R,9R (X-ray anomalous dispersion) | [17,18] |
| Arenosclerin A (2) | −3.0 | 0.015 | MeOH | 2R,3S,7S,9S (ECCD of bis-P-Br-phenacyl derivative and linked its configuration to that of (-)-perhaliclonacyclamine) | [10,13] |
| Arenosclerin B (3) | +8.6 | 0.008 | MeOH | 2S*,3S*,7R*,9R* (Relative configuration) | [10] |
| Arenosclerin C (4) | −17 | 0.02 | MeOH | 2S*,3R*,7S*,9S* (Relative configuration) | [10] |
| Arenosclerin D (5) | +6.9 | 0.014 | MeOH | 2R*,3S*,9R* (Tentative Relative configuration) The configuration at C7 was not defined. | [9] |
| Arenosclerin E (6) | +14.5 | 0.015 | MeOH | 2R*,3S*,7S*,9S* (Relative configuration) | [9] |
| Chloromethyltetradehydro-halicyclamine B (7) | N/A | N/A | N/A | N/A | [18] |
| Chloromethylhalicyclamine B (8) | −94 | 0.05 | MeOH | 2S,3R,7R (ECD) | [16] |
| Halichondramine (9) | +3.3 | 0.54 | MeOH | 2R*,3S*,7R*,9R* Or 2R*,3S*,7S*,9S* (Relative configuration) | [19] |
| Haliclonacyclamine A (10) | −3.4 | 1.21 | CH2Cl2 | 2R,3R,7R,9R (X-Ray anomalous dispersion) | [20,24] |
| Haliclonacyclamine B (11) | +3.4 | 0.55 | CH2Cl2 | 2R,3R,7R,9R (X-ray anomalous dispersion) | [20,24] |
| Haliclonacyclamine C (12) | +4.8 | 0.30 | CH2Cl2 | 2R,3R,7R,9R (inferred from optical rotation comparison with hydrogenated analogs.) | [12] |
| Haliclonacyclamine D (13) | +16.1 | 0.30 | CH2Cl2 | 2R,3R,7R,9R (NOESY data suggested the configurations; insufficient material for hydrogenation. | [12] |
| Haliclonacyclamine E (14) | +14 | 0.02 | MeOH | 2R,3S,7S,9S (ECCD of bis-P-Br-phenacyl derivative and linked its configuration to that of (-)-perhaliclonacyclamine) | [10,13] |
| Haliclonacyclamine F (15) | +5.4 | 0.0041 | MeOH | 2R*,3S*,7R*,9R* (Tentative Relative configuration) The configuration for ring B was proposed. | [9] |
| Halicyclamine A (16) | −7.3 | 0.72 | CH2Cl2 | 2R*,3S*,9R* (Relative configuration) | [8] |
| −24.0 | 0.1 | MeOH | 2R*,3S*,9R* (Relative configuration) | [21] | |
| Halicyclamine B (17) | −143.5 | 0.65 | N/A | 2S,3R,7R Relative configuration (X-Ray); Absolute configuration (ECD) | [11,22] |
| 22-Hydroxyhaliclonacyclamie B (18) | +11.8 | 0.1 | MeOH | 2R,3R,7R,9R (relative configurations were assigned based on ROESY data.) The configuration at C-22 (22-OH) remains undetermined. | [23] |
| 22-Hydroxyhalicyclamine A (19) | +21.0 | 0.1 | MeOH | 2R*,3S*,9R* (Relative configuration only) No ROESY. Only comparison with 1H and 13C NMR data. | [21] |
| Neopetrosiamine A (20) | −10.0 | 1.0 | CHCl3 | 2R*,3R*,7R*,9R* (Relative configuration) | [14] |
| Tetradehydrohaliclonacyclamine A (21) | +19.4 | 0.515 | CHCl3 | 2S,3S,7S,9S (X-ray anomalous dispersion) | [24] |
| 2-epi-Tetradehydrohaliclonacyclamine A (22) | +6.6 | 0.083 | CHCl3 | 2R,3S,7S,9S | [24] |
| Tetradehydrohaliclonacyclamine A mono-N-oxide (23) | +9.2 | 0.035 | CHCl3 | 2S,3S,7S,9S | [24] |
| Tetradehydrohalicyclamine A (24) | −14.7 | 0.1 | MeOH | 2R*,3S* (Relative configuration) | [21] |
| Tetradehydrohalicyclamine B (25) | N/A | N/A | N/A | 2S, 3R Bioinspired relationship with halicyclamine B ECD | [18,22] |
| 3-epi-Tetradehydrohalicyclamine B (26) a | N/A | N/A | N/A | 2S, 3S Based on comparison with 26. | [18] |
| Xestoproxamine A (27) | +4.4 | 2.0 | MeOH | 2R,3S,7S,9S ECCD | [13] |
| Xestoproxamine B (28) | +2.7 | 2.4 | MeOH | 2R,3S,7S,9S ECCD | [13] |
| Xestoproxamine C (29) | −18.5 | 0.67 | CHCl3 | 2S,3S,7S,9S,23S ECCD | [13] |
| Perhydrohaliclonacyclamine (30) b | −20.9 | 0.205 | CHCl3 | 2S,3S,7S,9S (X-ray of its parent compound) | [13] |
| Compound | IC50 Value in µM (Cell Line) | Cell model | Ref. |
|---|---|---|---|
| Acanthocyclamine A (1) | ND | ND | [17,18] |
| Arenosclerin A (2) | 8.9 (HL60) | leukemia | [10,42] |
| 3.6 (B16) | melanoma | ||
| 2.1 4.8 (L929) | fibrosarcoma | ||
| 7.9 (U138) | glioblastoma | ||
| 4.7 (HL60) | leukemia | ||
| Arenosclerin B (3) | 8.4 (HL60) | leukemia | [10,42] |
| 3.6 (B16) | melanoma | ||
| 4.6 (L929) | fibrosarcoma | ||
| 7.5 (U138) | glioblastoma | ||
| Arenosclerin C (4) | 7.5 (HL60) | leukemia | [10,42] |
| 3.5 (B16) | melanoma | ||
| 4.5 (L929) | fibrosarcoma | ||
| 7.4 (U138) | glioblastoma | ||
| Arenosclerin D (5) | 12.2 (SF295) | glioblastoma | [9] |
| 2.4 (MDA-MB435) | reported as breast cancer a | ||
| 12.8 (HCT8) | colon cancer | ||
| 4.3 (HL60) | leukemia | ||
| Arenosclerin E (6) | 18.0 (SF295) | glioblastoma | |
| 6.4 (MDA-MB435) | reported as breast cancer a | ||
| >51.8 (HCT8) | colon cancer | ||
| 14.3 (HL60) | leukemia | ||
| Chloromethyltetradehydro- halicyclamine B (7) | ND | ND | [18] |
| Chloromethylhalicyclamine B (8) | ND | ND | [16,18] |
| Halichondramine (9) | ND | ND | [19] |
| Haliclonacyclamine A (10) | 1.71 (P388) | lymphocytic leukemia | [12,20,24] |
| Haliclonacyclamine B (11) | 1.28 (P388) | lymphocytic leukemia | |
| Haliclonacyclamine C (12) | 1.49 (P388) | lymphocytic leukemia | [12,24] |
| Haliclonacyclamine D (13) | N/A | N/A | |
| Haliclonacyclamine E (14) | 9.0 (HL60) | leukemia | [10,42] |
| 3.9 (B16) | melanoma | ||
| 8.3 (L929) | fibrosarcoma | ||
| 13.0 (U138) | glioblastoma | ||
| Haliclonacyclamine F (15) | 4.5 (SF295) | glioblastoma | [9] |
| 1.0 (MDA-435) | reported as breast cancer a | ||
| 2.2 (HL-60) | Leukemia | ||
| 8.6 (HCT8) | Colon | ||
| Halicyclamine A (16) | 0.97 (P388) | lymphocytic leukemia | [21] |
| (-)-Halicyclamine B (17) | 12 (HeLa) | Cervical cancer | [22] |
| 22-Hydroxyhaliclonacyclamine B (18) | ND | ND | [23] |
| 22-Hydroxyhalicyclamine A (19) | 0.94 (P388) | lymphocytic leukemia | [21] |
| Neopetrosiamine A (20) | 1.5 (MALME-3M) | melanoma | [14] |
| 2.0 (CCRF-CEM) | leukemia | ||
| 3.5 (MCF) | breast cancer | ||
| 96 (VERO) | African green monkey kidney (normal cell model) | ||
| Tetradehydrohaliclonacyclamine A (21) | 3.88 (P388) | Lymphocytic leukemia | [24] |
| 2-Epi-tetradehydrohalicyclamien A (22) | ND | ND | [21] |
| Tetradehydrohalicyclamine A mono-N-oxide (23) | ND | ND | [21] |
| Tetradehydrohalicyclamine A (24) | 4.8 (P388) | Lymphocytic leukemia | [21] |
| Tetradehydrohalicyclamine B (25) | >50 (HeLa) | Cervical cancer | [22] |
| Epi-Tetradehydrohalicyclamine B (26) | ND | ND | [18] |
| Xestoproxamine A (27) | 21.2 (HCT-116) | Colorectal cancer | [16] |
| Xestoproxamine B (28) | 6.3 (HCT-116) | Colorectal cancer | [16] |
| Xestoproxamine C (29) | 5.4 (HCT-116) | Colorectal cancer | [16] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez, S.I.; Oceguera Nava, E.I.; Dadawalla, A.; Ashong, D.; Chen, G.; Chen, Q.-H. Tetracyclic Bis-Piperidine Alkaloids: Structures, Bioinspired Synthesis, Synthesis, and Bioactivities. Molecules 2025, 30, 2907. https://doi.org/10.3390/molecules30142907
Gómez SI, Oceguera Nava EI, Dadawalla A, Ashong D, Chen G, Chen Q-H. Tetracyclic Bis-Piperidine Alkaloids: Structures, Bioinspired Synthesis, Synthesis, and Bioactivities. Molecules. 2025; 30(14):2907. https://doi.org/10.3390/molecules30142907
Chicago/Turabian StyleGómez, Stan Iridio, Esveidy Isabel Oceguera Nava, Abbas Dadawalla, Dennis Ashong, Guanglin Chen, and Qiao-Hong Chen. 2025. "Tetracyclic Bis-Piperidine Alkaloids: Structures, Bioinspired Synthesis, Synthesis, and Bioactivities" Molecules 30, no. 14: 2907. https://doi.org/10.3390/molecules30142907
APA StyleGómez, S. I., Oceguera Nava, E. I., Dadawalla, A., Ashong, D., Chen, G., & Chen, Q.-H. (2025). Tetracyclic Bis-Piperidine Alkaloids: Structures, Bioinspired Synthesis, Synthesis, and Bioactivities. Molecules, 30(14), 2907. https://doi.org/10.3390/molecules30142907