Accessible Eco-Friendly Method for Wastewater Removal of the Azo Dye Reactive Black 5 by Reusable Protonated Chitosan-Deep Eutectic Solvent Beads
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Synthesis of the Adsorbents
2.2.1. Unmodified Chitosan Beads
2.2.2. DES-Modified Chitosan Beads
2.2.3. Sulfuric Acid Treatment for Unmodified Chitosan (ST-un-Ch) and DES-Modified Chitosan Beads (ST-Ch-DES)
2.3. Characterization
2.4. Adsorption Assays
2.5. Adsorption Kinetics
2.6. Adsorption Isotherm
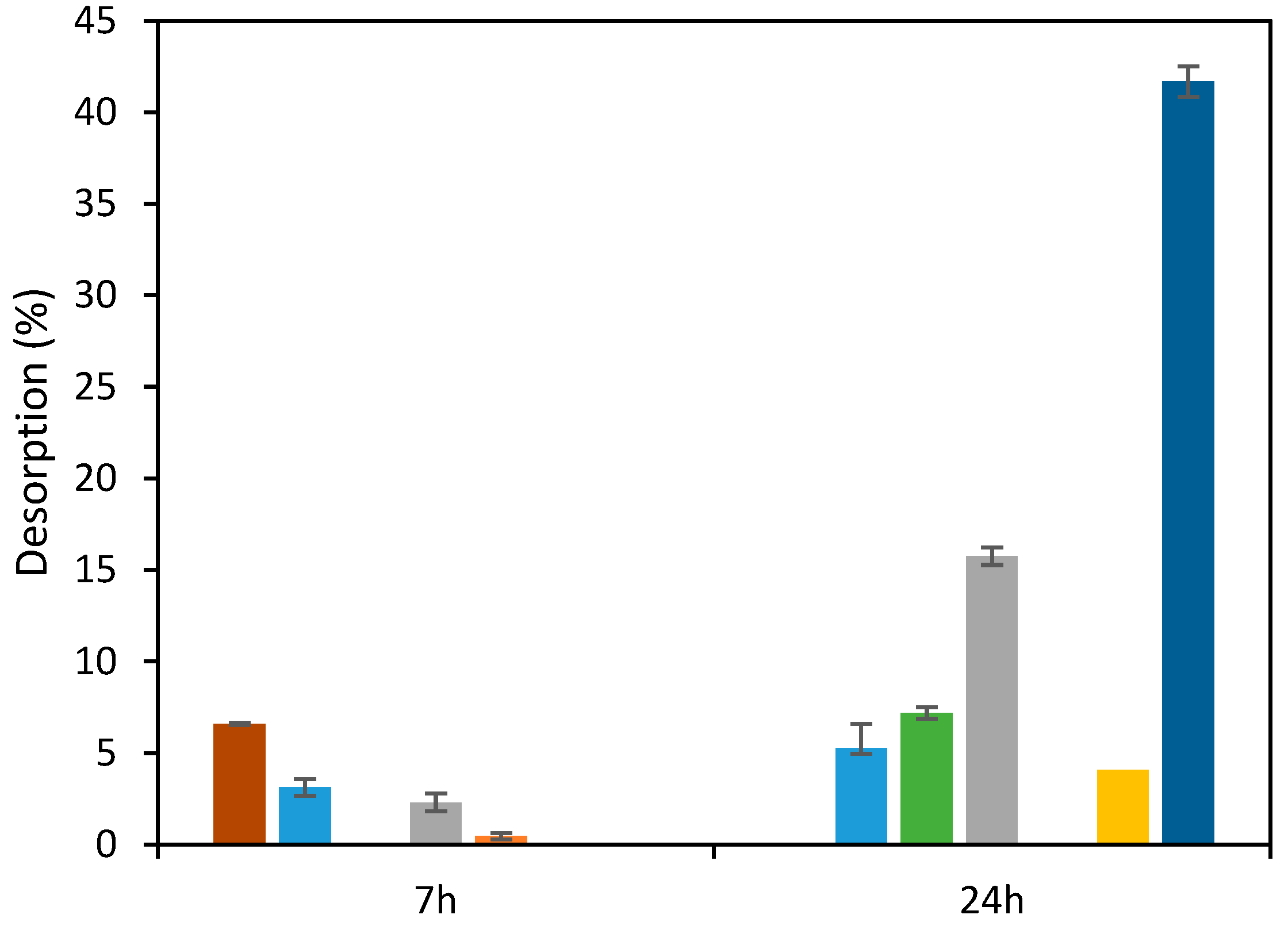
2.7. Desorption and Reusability Tests
3. Results and Discussion
3.1. Adsorbent Characterizations
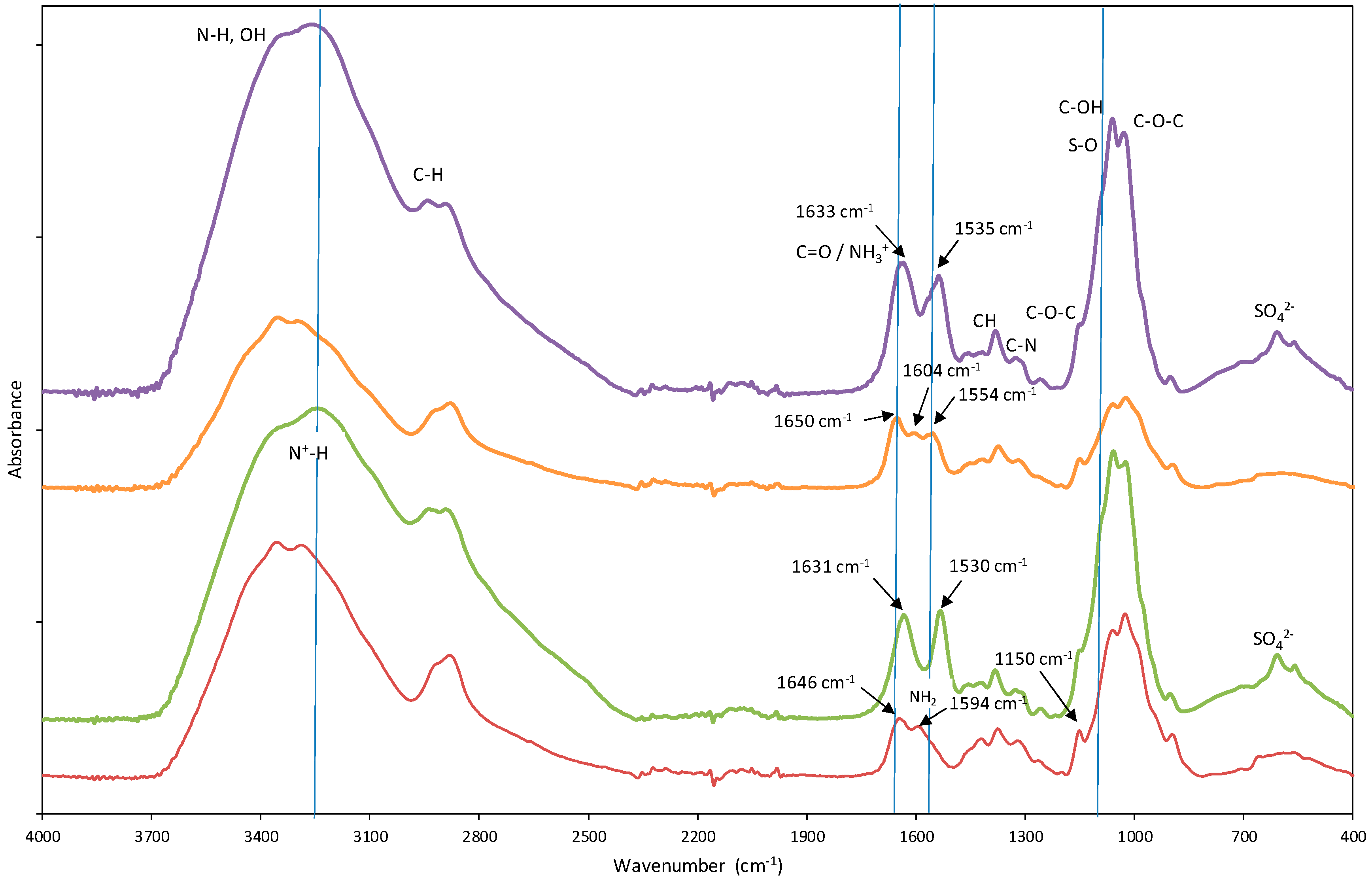
3.1.1. Chemical Characterization by Fourier-Transform Infrared (FTIR) Spectroscopy
3.1.2. Morphological Study by Scanning Electron Microscopy (SEM)
3.1.3. Brunauer–Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) Analysis
3.1.4. Swelling Behavior
3.2. Selection of the Best RB5 Adsorbent
3.3. Variables Impacting the Adsorption Process
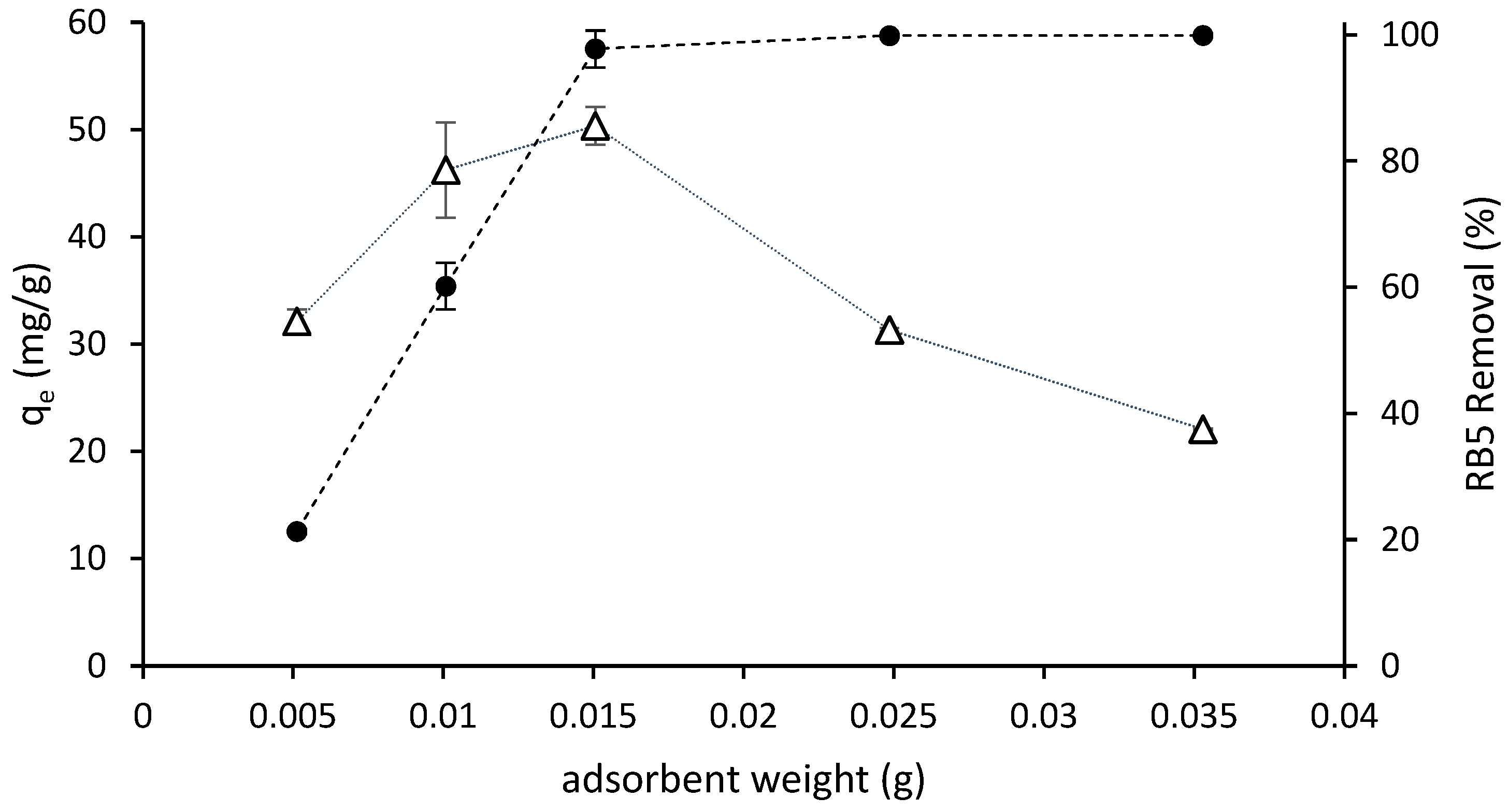
3.3.1. Effect of Adsorbent Dose
3.3.2. Effect of Initial Colorant Concentration and Contact Time
3.3.3. Effect of Temperature
3.3.4. Effect of pH
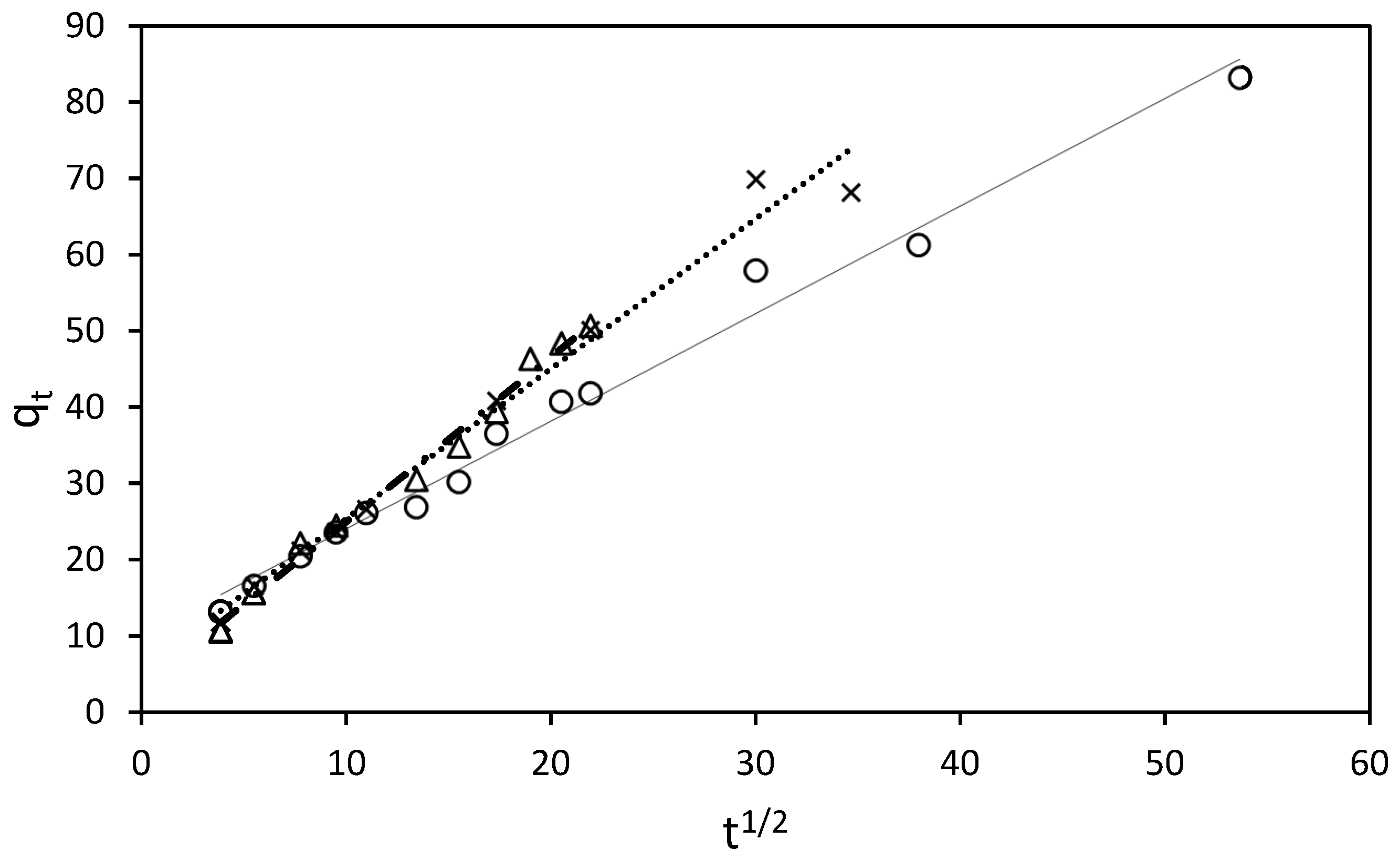
3.4. Adsorption Kinetic Models
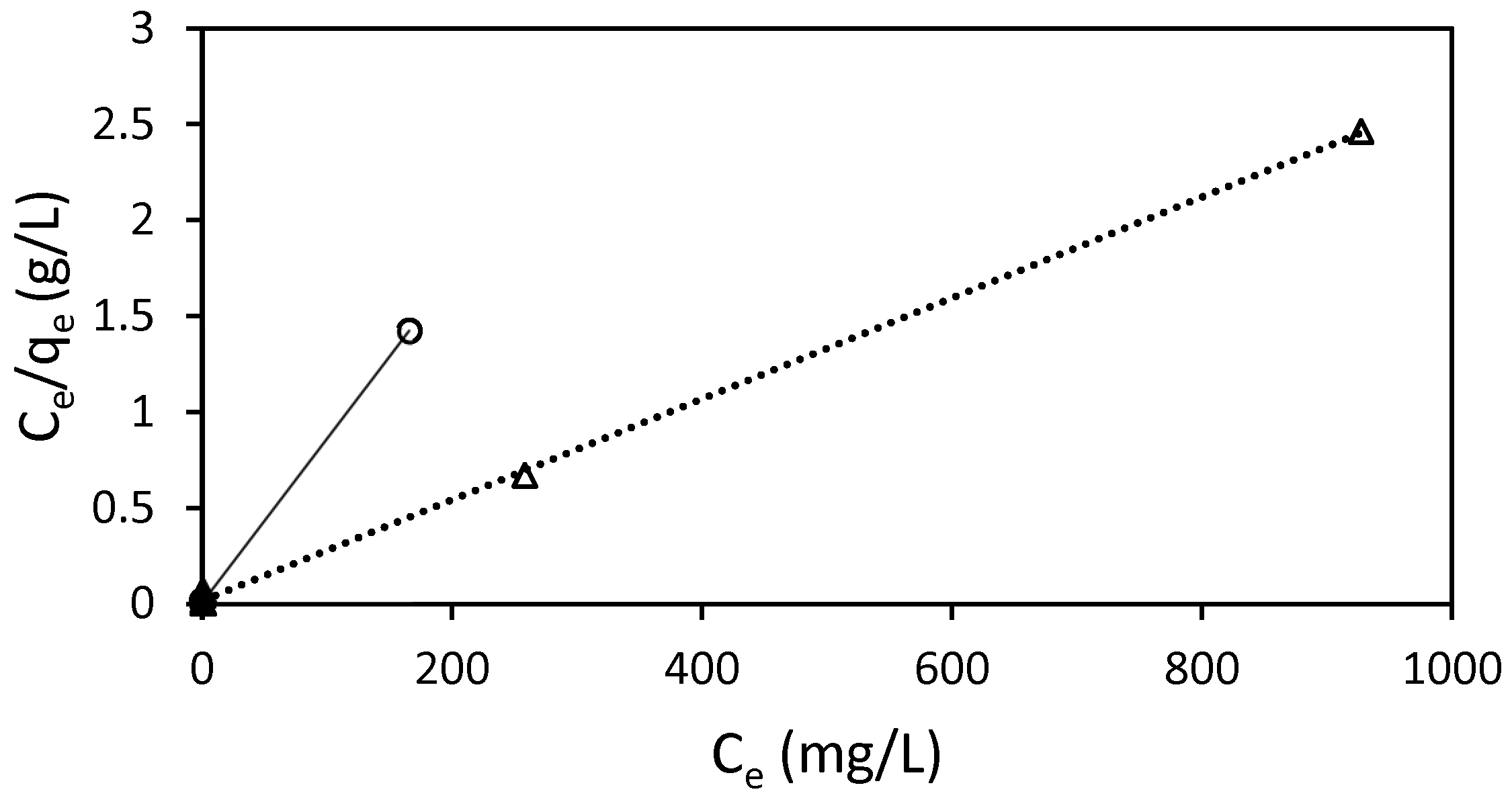
3.5. Adsorption Isotherms
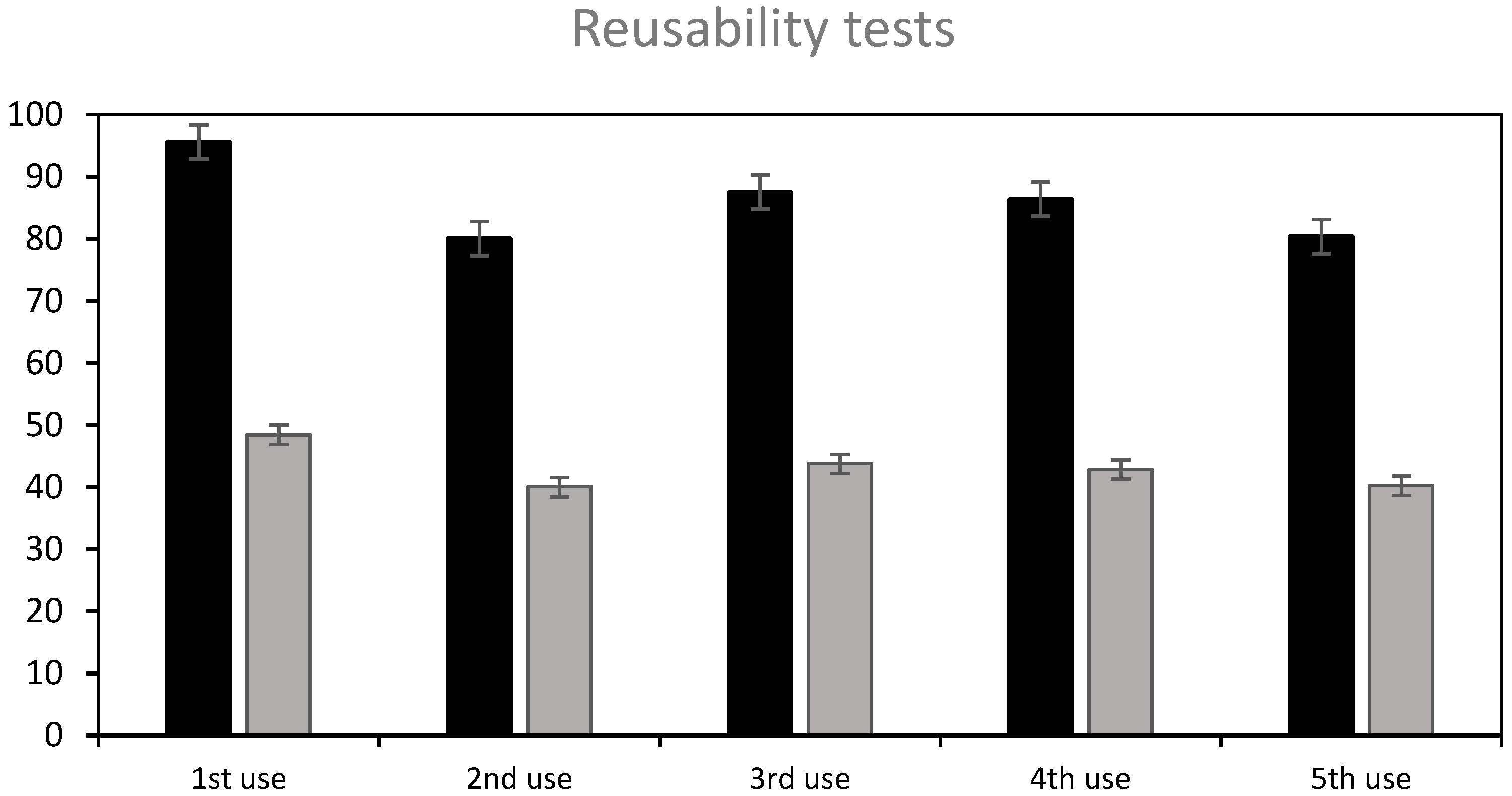
3.6. Reusability of the Adsorbent
3.6.1. Desorption
3.6.2. Regeneration
3.7. Possible Adsorption Mechanism
3.8. Comparison of Ch-DES Adsorption Capacities
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Kishor, R.; Purchase, D.; Saratale, G.D.; Saratale, R.G.; Ferreira, L.F.R.; Bilal, M.; Chandra, R.; Bharagava, R.N. Ecotoxicological and health concerns of persistent coloring pollutants of textile industry wastewater and treatment approaches for environmental safety. J. Environ. Chem. Eng. 2021, 9, 105012. [Google Scholar] [CrossRef]
- European Environment Agency. Available online: https://www.eea.europa.eu/publications/textiles-in-europes-circular-economy (accessed on 20 July 2022).
- Roth, J.; Zerger, B.; De Geeter, D.; Gómez Benavides, J.; Roudier, S. Best available Techniques (BAT) Reference Document for the Textiles Industry; Publications Office of the European Union: Luxembourg, 2023. [Google Scholar] [CrossRef]
- Arslan, S.; Eyvaz, M.; Gürbulak, E.; Yüksel, E. A Review of State-of-the-Art Technologies in Dye-Containing Wastewater Treatment—The Textile Industry Case. In Textile Wastewater Treatment; InTech: Rijeka, Croatia, 2016. [Google Scholar] [CrossRef]
- Aramesh, N.; Bagheri, A.R.; Bilal, M. Chitosan-based hybrid materials for adsorptive removal of dyes and underlying interaction mechanisms. Int. J. Biol. Macromol. 2021, 183, 399–422. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef]
- Soliman, N.; Moustafa, A. Industrial solid waste for heavy metals adsorption features and challenges; a review. J. Mater. Res. Technol. 2020, 9, 10235–10253. [Google Scholar] [CrossRef]
- da Silva Alves, D.C.; Healy, B.; Pinto, L.A.d.A.; Cadaval, T.R.S., Jr.; Breslin, C.B. Recent Developments in Chitosan-Based Adsorbents for the Removal of Pollutants from Aqueous Environments. Molecules 2021, 26, 594. [Google Scholar] [CrossRef]
- Blanco, L.; Martínez-Rico, O.; Domínguez, Á.; González, B. Removal of Acid Blue 80 from aqueous solutions using chitosan-based beads modified with choline chloride:urea Deep Eutectic Solvent and FeO. Water Resour. Ind. 2023, 29, 100195. [Google Scholar] [CrossRef]
- Blanco, L.V.; Sas, O.G.; Sánchez, P.B.; Santiago, Á.D.; de Prado, B.G. Congo red recovery from water using green extraction solvents. Water Resour. Ind. 2022, 27, 100170. [Google Scholar] [CrossRef]
- Sadiq, A.C.; Rahim, N.Y.; Suah, F.B.M. Adsorption and desorption of malachite green by using chitosan-deep eutectic solvents beads. Int. J. Biol. Macromol. 2020, 164, 3965–3973. [Google Scholar] [CrossRef]
- Sadiq, A.C.; Olasupo, A.; Rahim, N.Y.; Ngah, W.S.W.; Suah, F.B.M. Comparative removal of malachite green dye from aqueous solution using deep eutectic solvents modified magnetic chitosan nanoparticles and modified protonated chitosan beads. J. Environ. Chem. Eng. 2021, 9, 106281. [Google Scholar] [CrossRef]
- Lawal, I.A.; Dolla, T.H.; Pruessner, K.; Ndungu, P. Synthesis and characterization of deep eutectic solvent functionalized CNT/ZnCo2O4 nanostructure: Kinetics, isotherm and regenerative studies on Eosin Y adsorption. J. Environ. Chem. Eng. 2019, 7, 102877. [Google Scholar] [CrossRef]
- Dai, Y.; Row, K.H. Magnetic chitosan functionalized with β-cyclodextrin as ultrasound-assisted extraction adsorbents for the removal of methyl orange in wastewater coupled with high-performance liquid chromatography. J. Sep. Sci. 2018, 41, 3397–3403. [Google Scholar] [CrossRef]
- Li, G.; Row, K.H. Deep eutectic solvents skeleton typed molecularly imprinted chitosan microsphere coated magnetic graphene oxide for solid-phase microextraction of chlorophenols from environmental water. J. Sep. Sci. 2020, 43, 1063–1070. [Google Scholar] [CrossRef]
- Viswanathan, N.; Sundaram, C.S.; Meenakshi, S. Removal of fluoride from aqueous solution using protonated chitosan beads. J. Hazard. Mater. 2009, 161, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.-H.; Li, S.-Y.; Wang, F.; Liu, G.-L. Adsorption of perchlorate from water by protonated cross-linked chitosan gel beads. Huan Jing Ke Xue Huanjing Kexue 2009, 30, 2580–2585. [Google Scholar]
- Wu, J.; Chen, J. Adsorption characteristics of tannic acid onto the novel protonated palygorskite/chitosan resin microspheres. J. Appl. Polym. Sci. 2013, 127, 1765–1771. [Google Scholar] [CrossRef]
- Guo, Y.-J.; Xue, J.-Q.; Zhang, J.; Ma, J.; Quan, X.-T.; Liu, Y. Adsorption behaviors of protonation modified chitosan and the analysis of spectra. Spectrosc. Spectr. Anal. 2014, 34, 78–81. [Google Scholar]
- Xueting, Q.; Linbo, L.; Yingjuan, G.; Yao, L.; Juanqin, X. Preparation of protonated modified-chitosan and its adsorption properties for sulfate. Chin. J. Environ. Eng. 2014, 8, 3228–3232. [Google Scholar]
- Padilla-Rodríguez, A.; Hernández-Viezcas, J.A.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L.; Perales-Pérez, O.; Román-Velázquez, F.R. Adsorption of arsenic(V) oxyanion from aqueous solutions by using protonated chitosan flakes. Sep. Sci. Technol. 2015, 50, 2099–2111. [Google Scholar] [CrossRef]
- Huang, R.; Yang, B.; Liu, Q.; Gong, N. Adsorption of a model anionic dye on protonated crosslinked chitosan. Desalination Water Treat. 2014, 52, 7693–7700. [Google Scholar] [CrossRef]
- Huang, R.; Liu, Q.; Huo, J.; Yang, B. Adsorption of methyl orange onto protonated cross-linked chitosan. Arab. J. Chem. 2013, 10, 24–32. [Google Scholar] [CrossRef]
- Ngah, W.W.; Fatinathan, S.; Yosop, N. Isotherm and kinetic studies on the adsorption of humic acid onto chitosan-H2SO4 beads. Desalination 2011, 272, 293–300. [Google Scholar] [CrossRef]
- Patiño-Ruiz, D.A.; De Ávila, G.; Alarcón-Suesca, C.; González-Delgado, Á.D.; Herrera, A. Ionic Cross-Linking Fabrication of Chitosan-Based Beads Modified with FeO and TiO2 Nanoparticles: Adsorption Mechanism toward Naphthalene Removal in Seawater from Cartagena Bay Area. ACS Omega 2020, 5, 26463–26475. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Chen, W.; Mao, Q.; Bai, Y.; Ma, H. Facile synthesis of chitosan/gelatin filled with graphene bead adsorbent for orange II removal. Chem. Eng. Res. Des. 2019, 144, 35–46. [Google Scholar] [CrossRef]
- Soliman, N.K.; Moustafa, A.F.; El-Mageed, H.R.A.; Abdel-Gawad, O.F.; Elkady, E.T.; Ahmed, S.A.; Mohamed, H.S. Experimentally and theoretically approaches for disperse red 60 dye adsorption on novel quaternary nanocomposites. Sci. Rep. 2021, 11, 10000. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.-C.; Tseng, R.-L.; Juang, R.-S. Initial behavior of intraparticle diffusion model used in the description of adsorption kinetics. Chem. Eng. J. 2009, 153, 1–8. [Google Scholar] [CrossRef]
- Weber, W.J.; Morris, J.C. Kinetics of Adsorption on Carbon from Solution. J. Sanit. Eng. Div. 1963, 89, 31–60. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Über die Adsorption in Lösungen. Zeitschrift Phys. Chemie 1907, 57U, 385–470. [Google Scholar] [CrossRef]
- Temkin, M.I.; Pyzhev, V. Kinetic of Ammonia Synthesis on Promoted Iron Catalysts. J. Phys. Chem. USSR. 1939, 13, 851–857. [Google Scholar]
- Dubinin, M.M.; Radushkevich, L.V. Equation of the Characteristic Curve of Activated Charcoal. Proc. Acad. Sci. USSR Phys. Chem Sect. 1947, 55, 331–333. [Google Scholar]
- Elovich, S.Y.; Larinov, O.G. Theory of adsorption from solutions of non electrolytes on solid (I) equation adsorption from solutions and the analysis of its simplest form, (II) verification of the equation of adsorption isotherm from solutions. Izv. Akad. Nauk. SSSR, Otd. Khim. Nauk. 1962, 2, 209–216. [Google Scholar]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies. In Tables and Charts, 3rd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2001. [Google Scholar]
- Cui, Z.; Xiang, Y.; Si, J.; Yang, M.; Zhang, Q.; Zhang, T. Ionic interactions between sulfuric acid and chitosan membranes. Carbohydr. Polym. 2008, 73, 111–116. [Google Scholar] [CrossRef]
- Jakubowska, E.; Gierszewska, M.; Nowaczyk, J.; Olewnik-Kruszkowska, E. Physicochemical and storage properties of chitosan-based films plasticized with deep eutectic solvent. Food Hydrocoll. 2020, 108, 106007. [Google Scholar] [CrossRef]
- el Nahrawy, A.M.; Bakr, A.; Hammad, A.B.A.; Mansour, A.-F.M. Nano-architecture of CaO/Ag-chitosan nanocomposite by sol gel process: Formation and characterization. Egypt. J. Chem. 2021, 64, 7393–7406. [Google Scholar] [CrossRef]
- Rehman, M.S.U.; Kim, I.; Han, J.-I. Adsorption of methylene blue dye from aqueous solution by sugar extracted spent rice biomass. Carbohydr. Polym. 2012, 90, 1314–1322. [Google Scholar] [CrossRef]
- Subramaniam, S.; Foo, K.; Yusof, E.M.; Jawad, A.H.; Wilson, L.; Sabar, S. Hydrothermal synthesis of phosphorylated chitosan and its adsorption performance towards Acid Red 88 dye. Int. J. Biol. Macromol. 2021, 193, 1716–1726. [Google Scholar] [CrossRef]
- Barreiro-Iglesias, R.; Coronilla, R.; Concheiro, A.; Alvarez-Lorenzo, C. Preparation of chitosan beads by simultaneous cross-linking/insolubilisation in basic pH: Rheological optimisation and drug loading/release behaviour. Eur. J. Pharm. Sci. 2005, 24, 77–84. [Google Scholar] [CrossRef]
- Sadiq, A.C.; Olasupo, A.; Ngah, W.S.W.; Rahim, N.Y.; Suah, F.B.M. A decade development in the application of chitosan-based materials for dye adsorption: A short review. Int. J. Biol. Macromol. 2021, 191, 1151–1163. [Google Scholar] [CrossRef]
- Silva, L.S.; Lima, L.C.B.; Ferreira, F.J.L.; Silva, M.S.; Osajima, J.A.; Bezerra, R.D.S.; Filho, E.C.S. Sorption of the anionic reactive red RB dye in cellulose: Assessment of kinetic, thermodynamic, and equilibrium data. Open Chem. 2014, 13, 000010151520150079. [Google Scholar] [CrossRef]
- Dotto, G.; Pinto, L. Adsorption of food dyes acid blue 9 and food yellow 3 onto chitosan: Stirring rate effect in kinetics and mechanism. J. Hazard. Mater. 2011, 187, 164–170. [Google Scholar] [CrossRef]
- García, E.R.; Medina, R.L.; Lozano, M.M.; Pérez, I.H.; Valero, M.J.; Franco, A.M.M. Adsorption of Azo-Dye Orange II from Aqueous Solutions Using a Metal-Organic Framework Material: Iron- Benzenetricarboxylate. Materials 2014, 7, 8037–8057. [Google Scholar] [CrossRef]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and Interpretation of Adsorption Isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Hamd, A.; Shaban, M.; AlMohamadi, H.; Dryaz, A.R.; Ahmed, S.A.; Abu Al-Ola, K.A.; El-Mageed, H.R.A.; Soliman, N.K. Novel Wastewater Treatment by Using Newly Prepared Green Seaweed–Zeolite Nanocomposite. ACS Omega 2022, 7, 11044–11056. [Google Scholar] [CrossRef]
- Puentes-Cárdenas, I.J.; Chávez-Camarillo, G.M.; Flores-Ortiz, C.M.; Cristiani-Urbina, M.d.C.; Netzahuatl-Muñoz, A.R.; Salcedo-Reyes, J.C.; Pedroza-Rodríguez, A.M.; Cristiani-Urbina, E. Adsorptive Removal of Acid Blue 80 Dye from Aqueous Solutions by Cu-TiO2. J. Nanomater. 2016, 2016, 3542359. [Google Scholar] [CrossRef]
- Balakrishnan, A.; Appunni, S.; Chinthala, M.; Jacob, M.M.; Vo, D.-V.N.; Reddy, S.S.; Kunnel, E.S. Chitosan-based beads as sustainable adsorbents for wastewater remediation: A review. Environ. Chem. Lett. 2023, 21, 1881–1905. [Google Scholar] [CrossRef]
- Alaguprathana, M.; Poonkothai, M.; Ameen, F.; Bhat, S.A.; Mythili, R.; Sudhakar, C. Sodium hydroxide pre-treated Aspergillus flavus biomass for the removal of reactive black 5 and its toxicity evaluation. Environ. Res. 2022, 214, 113859. [Google Scholar] [CrossRef]
- Damdib, S.; Vanichsetakul, N.; Pimpapoat, P.; Mikhalovsky, S.; Busquets, R.; Siyasukh, A.; Tonanon, N. Removal of Reactive Black Dye in Water by Magnetic Mesoporous Carbon from Macadamia Nutshell. Adsorpt. Sci. Technol. 2022, 2022, 9884474. [Google Scholar] [CrossRef]
- Nascimento, B.F.D.; de Araujo, C.M.B.; Nascimento, A.C.D.; da Costa, G.R.B.; Gomes, B.F.M.L.; da Silva, M.P.; Santos, R.K.d.S.; Sobrinho, M.A.d.M. Adsorption of Reactive Black 5 and Basic Blue 12 using biochar from gasification residues: Batch tests and fixed-bed breakthrough predictions for wastewater treatment. Bioresour. Technol. Rep. 2021, 15, 100767. [Google Scholar] [CrossRef]
- Ben Dassi, R.; Chamam, B.; Méricq, J.P.; Heran, M.; Faur, C.; El Mir, L.; Tizaoui, C.; Trabelsi, I. Pb doped ZnO nanoparticles for the sorption of Reactive Black 5 textile azo dye. Water Sci. Technol. 2020, 82, 2576–2591. [Google Scholar] [CrossRef]
- Alhujaily, A.; Yu, H.; Zhang, X.; Ma, F. Adsorptive removal of anionic dyes from aqueous solutions using spent mushroom waste. Appl. Water Sci. 2020, 10, 183. [Google Scholar] [CrossRef]
- Correia, J.; Vasques Mendonça, A.R.; de Souza, S.M.D.A.G.U.; Valle, J.A.B. Adsorbents made from textile scraps: Preparation, characterization and application for removal of reactive dye. Clean Technol. Environ. Policy 2018, 20, 839–853. [Google Scholar] [CrossRef]
- Munagapati, V.S.; Yarramuthi, V.; Kim, Y.; Lee, K.M.; Kim, D.-S. Removal of anionic dyes (Reactive Black 5 and Congo Red) from aqueous solutions using Banana Peel Powder as an adsorbent. Ecotoxicol. Environ. Saf. 2018, 148, 601–607. [Google Scholar] [CrossRef]
- Ye, X.; Wu, L.; Zhu, M.; Wang, Z.; Huang, Z.-H.; Wang, M.-X. Lotus pollen-derived hierarchically porous carbons with exceptional adsorption performance toward Reactive Black 5: Isotherms, kinetics and thermodynamics investigations. Sep. Purif. Technol. 2022, 300, 121899. [Google Scholar] [CrossRef]
- Vojnović, B.; Cetina, M.; Franjković, P.; Sutlović, A. Influence of Initial pH Value on the Adsorption of Reactive Black 5 Dye on Powdered Activated Carbon: Kinetics, Mechanisms, and Thermodynamics. Molecules 2022, 27, 1349. [Google Scholar] [CrossRef] [PubMed]
- Nordin, A.H.; Wong, S.; Ngadi, N.; Zainol, M.M.; Latif, N.A.F.A.; Nabgan, W. Surface functionalization of cellulose with polyethyleneimine and magnetic nanoparticles for efficient removal of anionic dye in wastewater. J. Environ. Chem. Eng. 2020, 9, 104639. [Google Scholar] [CrossRef]
- Santos, R.; Silva, É.F.M.; Dantas, E.J.M.; Oliveira, E.D.C.; Simões, T.B.; Araújo, Í.R.S.; Ribeiro, A.T.S.; Oliveira, L.P.S.; Garcia, R.R.P.; Almeida, L.C. Potential Reuse of PET Waste Bottles as a Green Substrate/Adsorbent for Reactive Black 5 Dye Removal. Water, Air, Soil Pollut. 2020, 231, 533. [Google Scholar] [CrossRef]
- Omaç, F.; Karakurt, H.; Kartal, E. Removal of Reactive Black 5 by UV-C assisted peroxydisulfate process: Effect of parameters, kinetic study, and phytotoxicity assessment. Int. J. Environ. Anal. Chem. 2024, 1, 21. [Google Scholar] [CrossRef]
- Phan, K.-H.; Le, L.-T.; Tran, T.-D.; Vo, T.-K.; Tra, V.-T.; Nguyen, T.-Y.; Tran, C.-S.; Mai, T.-P.; Bui, X.-T. Anaerobic biodegradation of mixed azo dyes in thermophilic and mesophilic conditions. Case Stud. Chem. Environ. Eng. 2024, 9, 100667. [Google Scholar] [CrossRef]

 ) Ch-DES, (∆) ST-un-Ch, (
) Ch-DES, (∆) ST-un-Ch, ( ) ST-Ch-DES; at 298.16 K with a ratio adsorbent mass/dye solution volume of 3 g/L.
) ST-Ch-DES; at 298.16 K with a ratio adsorbent mass/dye solution volume of 3 g/L.
 ) Ch-DES, (∆) ST-un-Ch, (
) Ch-DES, (∆) ST-un-Ch, ( ) ST-Ch-DES; at 298.16 K with a ratio adsorbent mass/dye solution volume of 3 g/L.
) ST-Ch-DES; at 298.16 K with a ratio adsorbent mass/dye solution volume of 3 g/L.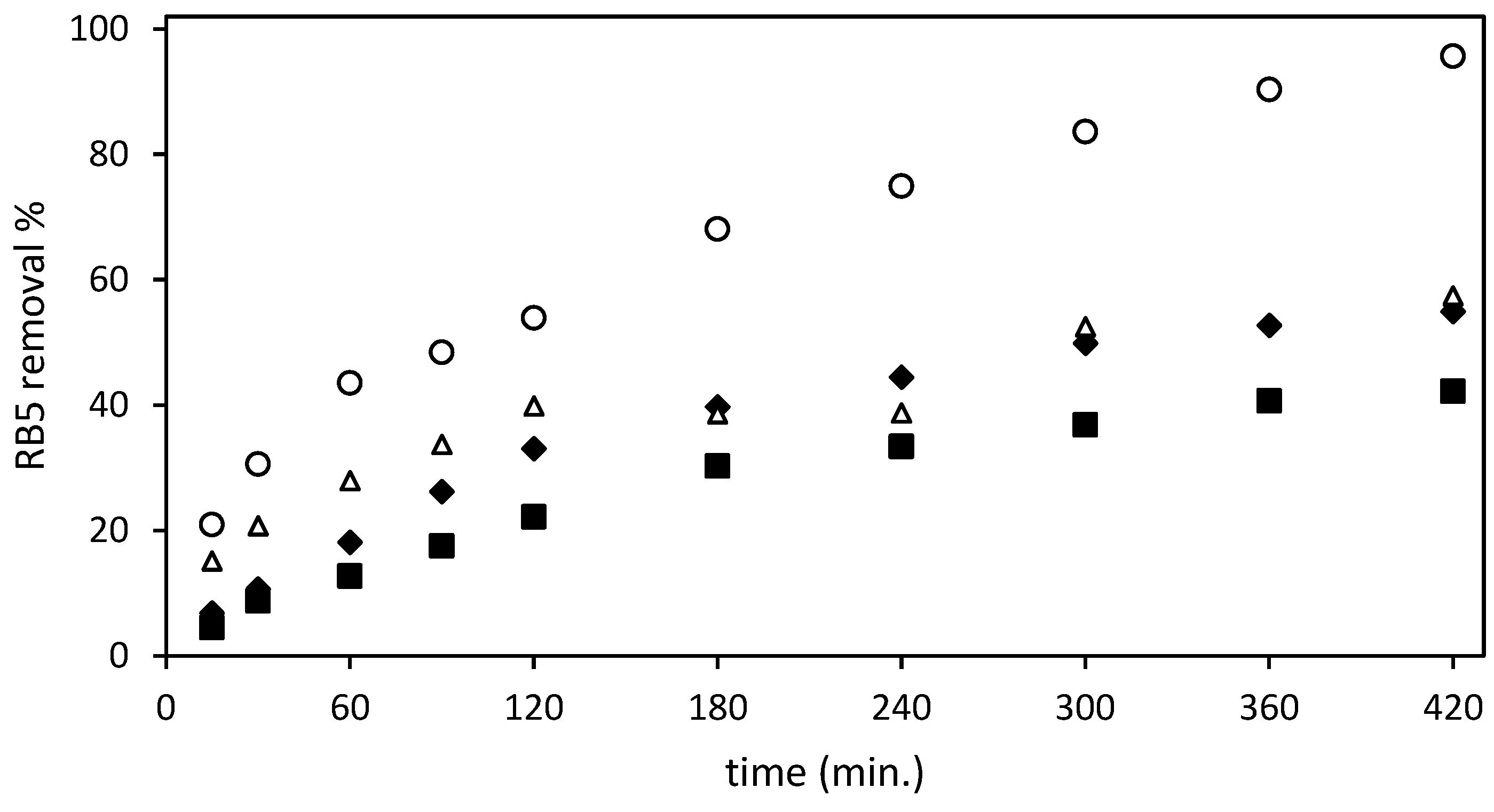
 ) 150 mg/L, (●) 250 mg/L.
) 150 mg/L, (●) 250 mg/L.
 ) 150 mg/L, (●) 250 mg/L.
) 150 mg/L, (●) 250 mg/L.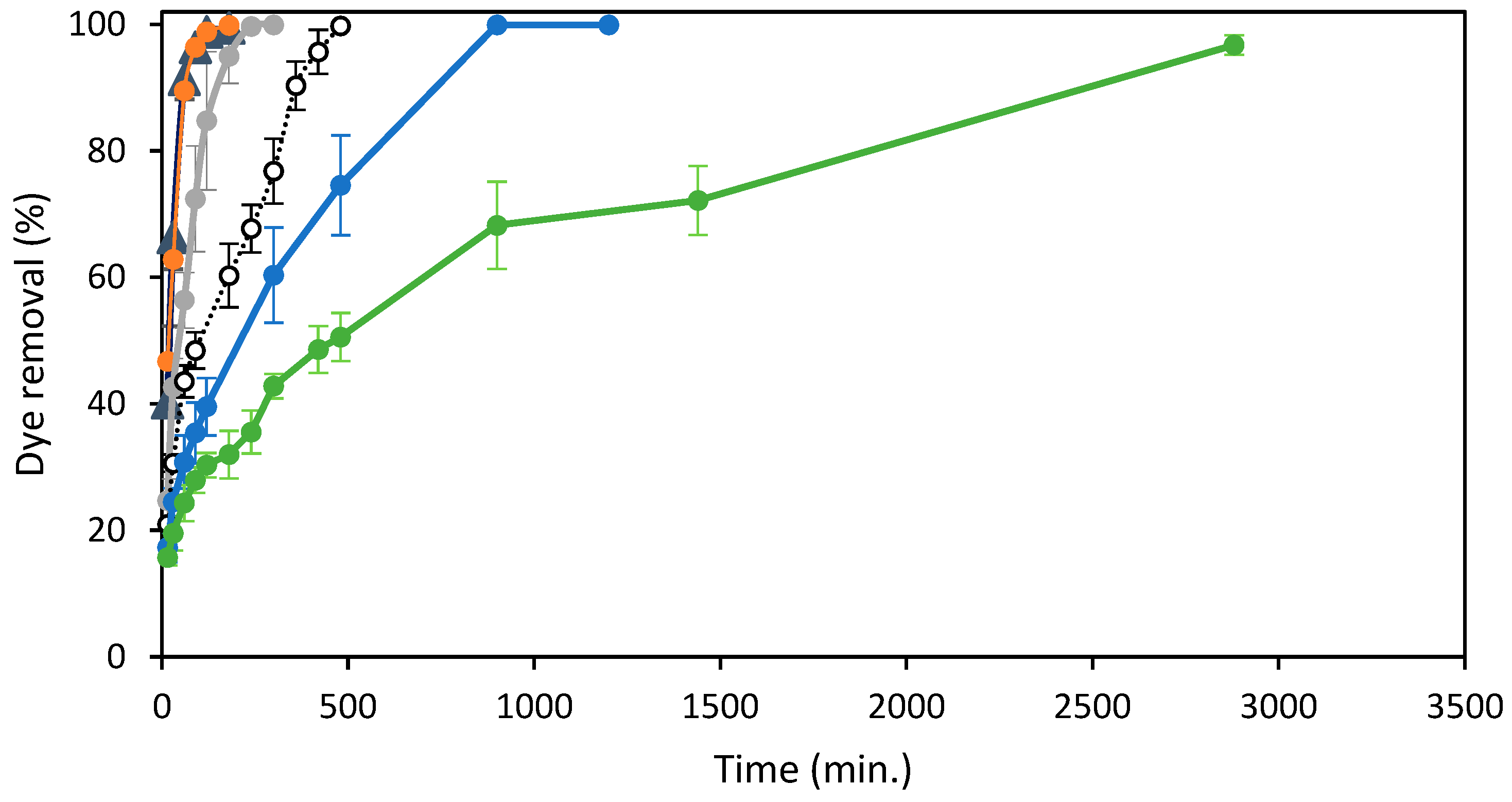

| Series | Dye Concentration (mg/L) | Adsorbent | Adsorbent Weight (g) | Temperature (K) | pH |
|---|---|---|---|---|---|
| 0 | 150 | un-Ch Ch-DES ST-un-Ch ST-Ch-DES | 0.015 | 298.16 | 6.0 |
| 1 | 150 | ST-Ch-DES | 0.005, 0.010, 0.015, 0.025, 0.035 | 298.16 | 6.0 |
| 2 | 25, 50, 100, 150, 200, 250 | ST-Ch-DES | 0.015 | 298.16 | 6.0 |
| 3 | 25, 50, 100, 150, 200, 250 | ST-Ch-DES | 0.015 | 298.16, 318.16 | 6.0 |
| 4 | 150 | ST-Ch-DES | 0.015 | 298.16 | 3.6, 5.2, 6.0, 8.2, 9.2 |
| un-Ch | Ch-DES | ST-un-Ch | ST-Ch-DES | |
|---|---|---|---|---|
| BET surface area (m2/g) | 0.34 | 0.15 | 2.27 | 1.97 |
| pore volume (cm3/g) | 3.6 × 10−4 | 1.5 × 10−4 | 20.4 × 10−4 | 18.4 × 10−4 |
| pore size (nm) | 4.16 | 4.05 | 3.60 | 3.73 |
| micropore surface area (m2/g) | 0.54 | 0.23 | 3.14 | 2.82 |
| Ch-DES | ||
|---|---|---|
| Treatment | Swelling Ratio (%) | Diameter Increase (%) |
| W (H2O) | 109.3 ± 2.1 | 22.4 ± 0.6 |
| A (H2SO4) | 149.4 ± 1.5 | 29.3 ± 0.9 |
| W-A (H2O + H2SO4) | 226.5 ± 2.5 | 44.2 ± 0.4 |
| un-Ch | ||
| W (H2O) | 80.8 ± 1.2 | 26.8 ± 2.2 |
| A (H2SO4) | 128.5 ± 2.6 | 29.2 ± 1.5 |
| W-A (H2O + H2SO4) | 118.7 ± 2.0 | 29.9 ± 0.9 |
| Dye | RB5 | |||||
| Concentration (mg/L) | 25 | 50 | 100 | 150 | 200 | 250 |
| qe, exp (mg/g) | 8.57 | 16.92 | 34.73 | 50.76 | 69.45 | 83.35 |
| Pseudo-first-order kinetic model | ||||||
| k1 (1/min) | 41.9 × 10−3 | 37.9 × 10−3 | 13.6 × 10−3 | 6.2 × 10−3 | 2.3 × 10−3 | 0.8 × 10−3 |
| qe (mg/g) | 9.56 | 17.15 | 32.71 | 48.51 | 56.77 | 64.50 |
| R2 | 0.9968 | 0.9873 | 0.9957 | 0.9158 | 0.9960 | 0.9502 |
| Pseudo-second-order kinetic model | ||||||
| k2 (mg/(g min)) | 4.7 × 10−3 | 2.7 × 10−3 | 0.4 × 10−3 | 0.2 × 10−3 | 0.7 × 10−4 | 0.4 × 10−4 |
| qe (mg/g) | 9.91 | 19.13 | 42.07 | 58.65 | 78.03 | 86.34 |
| R2 | 0.9938 | 0.9933 | 0.9972 | 0.9549 | 0.9738 | 0.9587 |
| Elovich kinetic model | ||||||
| β (g/mg) | 0.4626 | 0.2488 | 0.109 | 0.0877 | 0.0718 | 0.0789 |
| α (mg/(g min)) | 0.940 | 2.131 | 1.484 | 1.425 | 1.261 | 1.054 |
| R2 | 0.9165 | 0.9198 | 0.9888 | 0.9424 | 0.9330 | 0.8938 |
| Intraparticle diffusion kinetic model | ||||||
| k3 (mg/(g min1/2)) | 0.526 | 0.9889 | 1.957 | 2.1609 | 1.9665 | 1.4123 |
| I | 2.624 | 5.585 | 3.931 | 3.364 | 5.692 | 9.935 |
| R2 | 0.7955 | 0.8159 | 0.9403 | 0.9887 | 0.9830 | 0.9867 |
| Bead Type: ST-Ch-DES | |||
| Model | Parameters | 298.16 K | 318.16 K |
| Langmuir isotherm | kL (L/mg) | 1.61 | 0.17 |
| qm (mg/g) | 116.78 | 379.90 | |
| RL | 0.0024 | 0.0055 | |
| R2 | 0.9999 | 0.9991 | |
| Freundlich isotherm | n | 5.13 | 3.37 |
| kF | 46.48 | 63.89 | |
| R2 | 0.2988 | 0.4149 | |
| Temkin isotherm | B (J/mol) | 10.89 | 44.00 |
| kT (L/mol) | 303.16 | 14.70 | |
| R2 | 0.5453 | 0.6713 | |
| Elovic isotherm | ke | 10.084 | 0.845 |
| qm (mg/g) | 36.589 | 134.572 | |
| R2 | 0.2205 | 0.2574 | |
| Dubinin–Radushkevich isotherm | β ×10−8 (kJ/mol) | 5.888 | 16.659 |
| E (kJ/mol) | 0.291 | 0.173 | |
| qm (mg/g) | 123.05 | 226.21 | |
| R2 | 0.4087 | 0.3317 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Rico, O.; Blanco, L.; Domínguez, Á.; González, B. Accessible Eco-Friendly Method for Wastewater Removal of the Azo Dye Reactive Black 5 by Reusable Protonated Chitosan-Deep Eutectic Solvent Beads. Molecules 2024, 29, 1610. https://doi.org/10.3390/molecules29071610
Martínez-Rico O, Blanco L, Domínguez Á, González B. Accessible Eco-Friendly Method for Wastewater Removal of the Azo Dye Reactive Black 5 by Reusable Protonated Chitosan-Deep Eutectic Solvent Beads. Molecules. 2024; 29(7):1610. https://doi.org/10.3390/molecules29071610
Chicago/Turabian StyleMartínez-Rico, Oscar, Lucía Blanco, Ángeles Domínguez, and Begoña González. 2024. "Accessible Eco-Friendly Method for Wastewater Removal of the Azo Dye Reactive Black 5 by Reusable Protonated Chitosan-Deep Eutectic Solvent Beads" Molecules 29, no. 7: 1610. https://doi.org/10.3390/molecules29071610
APA StyleMartínez-Rico, O., Blanco, L., Domínguez, Á., & González, B. (2024). Accessible Eco-Friendly Method for Wastewater Removal of the Azo Dye Reactive Black 5 by Reusable Protonated Chitosan-Deep Eutectic Solvent Beads. Molecules, 29(7), 1610. https://doi.org/10.3390/molecules29071610









