Allicin and Cancer Hallmarks
Abstract
1. Introduction
2. Allicin
2.1. Allicin Chemical Structure and Formation
2.2. Allicin Bioavailability
2.3. Allicin Toxicity
2.4. Biological Functions
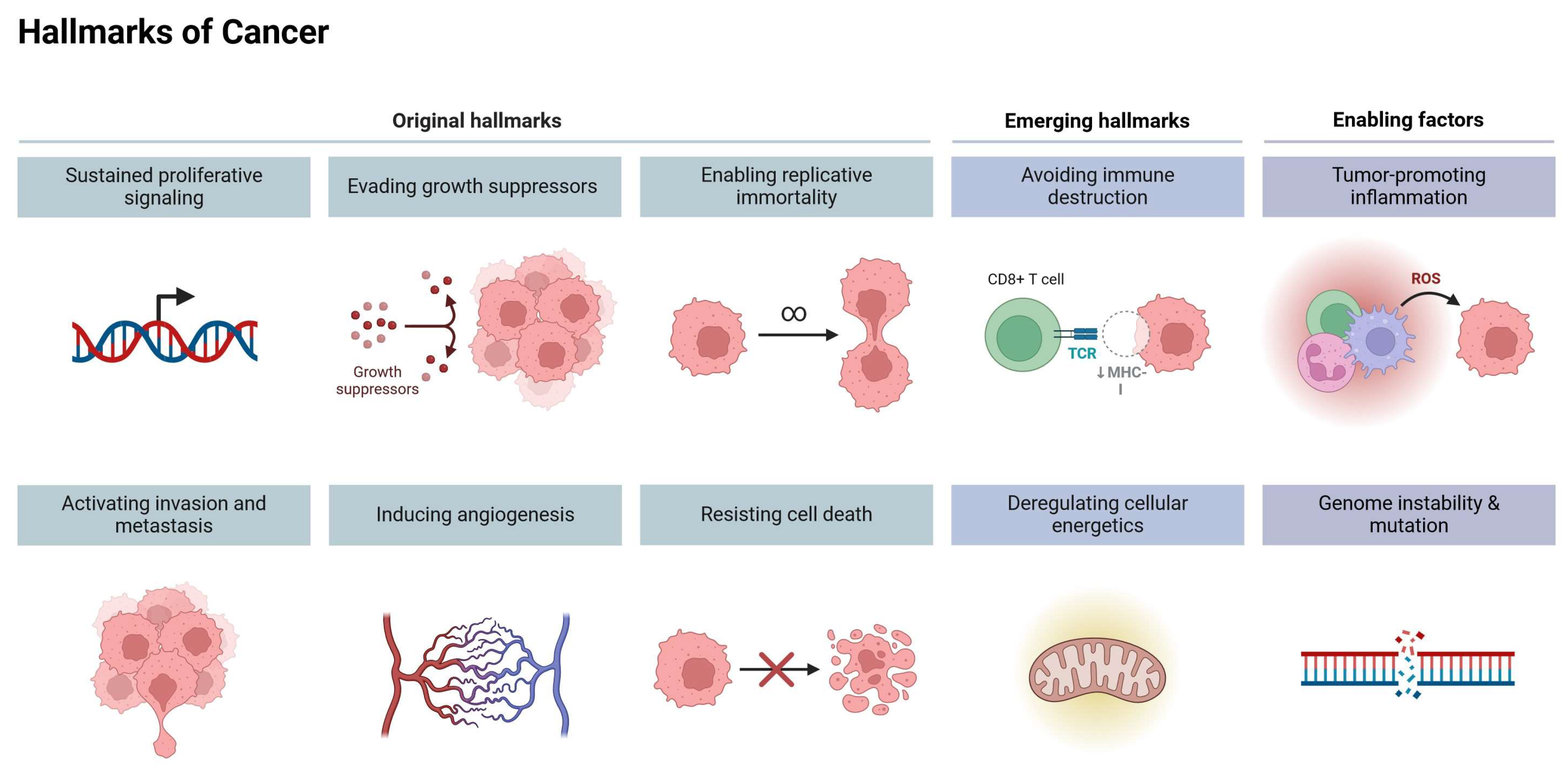
3. Cancer Hallmarks as Therapeutic Targets
4. Allicin and Cancer Hallmarks
4.1. Role of Allicin in Genomic Instability
4.2. Induction of Apoptosis
4.2.1. Caspase-Dependent Apoptosis
4.2.2. Caspase-Independent Apoptosis
4.3. Role of Allicin in Sustained Proliferative Signaling
4.4. Role of Allicin in Evasion of Anti-Growth Signaling
4.5. Role of Allicin in Replicative Immortality
4.6. Tumorigenesis and Carcinogen Activity Suppression
4.7. Role of Allicin in Tumor-Dysregulated Metabolism
4.8. Role of Allicin in Tumor-Promoting Inflammation
4.9. Role of Allicin in Angiogenesis Inhibition
4.10. Role of Allicin in Tissue Invasion and Metastasis
4.11. Clinical Trials: Allicin in Cancer Treatment
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mattiuzzi, C.; Lippi, G. Cancer statistics: A comparison between world health organization (WHO) and global burden of disease (GBD). Eur. J. Public Health 2020, 30, 1026–1027. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Choudhari, A.S.; Mandave, P.C.; Deshpande, M.; Ranjekar, P.; Prakash, O. Phytochemicals in cancer treatment: From preclinical studies to clinical practice. Front. Pharmacol. 2020, 10, 1614. [Google Scholar] [CrossRef]
- Catanzaro, E.; Canistro, D.; Pellicioni, V.; Vivarelli, F.; Fimognari, C. Anticancer potential of allicin: A review. Pharmacol. Res. 2022, 177, 106118. [Google Scholar] [CrossRef]
- Oommen, S.; Anto, R.J.; Srinivas, G.; Karunagaran, D. Allicin (from garlic) induces caspase-mediated apoptosis in cancer cells. Eur. J. Pharmacol. 2004, 485, 97–103. [Google Scholar] [CrossRef]
- Leontiev, R.; Hohaus, N.; Jacob, C.; Gruhlke, M.C.H.; Slusarenko, A.J. A comparison of the antibacterial and antifungal activities of thiosulfinate analogues of allicin. Sci. Rep. 2018, 8, 6763. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Cristina Cirone Silva, N.; Jantwal, A.; Bhatt, D.I.; Sharopov, F.; Cho, C.W.; Taheri, Y.; Martins, N. Therapeutic potential of allicin-rich garlic preparations: Emphasis on clinical evidence toward upcoming drugs formulation. Appl. Sci. 2019, 9, 5555. [Google Scholar] [CrossRef]
- Metwally, D.M.; Al-Olayan, E.M.; Alanazi, M.; Alzahrany, S.B.; Semlali, A. Antischistosomal and anti-inflammatory activity of garlic and allicin compared with that of praziquantel in vivo. BMC Complement. Altern. Med. 2018, 18, 135. [Google Scholar] [CrossRef]
- Ma, T.; Chen, D.; Tu, Y.; Zhang, N.; Si, B.; Deng, K.; Diao, Q. Effect of supplementation of allicin on methanogenesis and ruminal microbial flora in Dorper crossbred ewes. J. Anim. Sci. Biotechnol. 2016, 7, 1. [Google Scholar] [CrossRef] [PubMed]
- Cavallito, C.J.; Bailey, J.H. Allicin, the antibacterial principle of Allium sativum. I. Isolation, physical properties and antibacterial action. J. Am. Chem. Soc. 1944, 66, 1950–1951. [Google Scholar] [CrossRef]
- Lawson, L.D.; Hunsaker, S.M. Allicin Bioavailability and Bioequivalence from Garlic Supplements and Garlic Foods. Nutrients 2018, 10, 812. [Google Scholar] [CrossRef]
- Talib, W.H.; Daoud, S.; Mahmod, A.I.; Hamed, R.A.; Awajan, D.; Abuarab, S.F.; Odeh, L.H.; Khater, S.; Al Kury, L.T. Plants as a source of anticancer agents: From bench to bedside. Molecules 2022, 27, 4818. [Google Scholar] [CrossRef]
- Fouad, Y.A.; Aanei, C. Revisiting the hallmarks of cancer. Am. J. Cancer Res. 2017, 7, 1016–1036. [Google Scholar]
- Tu, G.; Zhang, Y.F.; Wei, W.; Li, L.; Zhang, Y.; Yang, J.; Xing, Y. Allicin attenuates H2O2-induced cytotoxicity in retinal pigmented epithelial cells by regulating the levels of reactive oxygen species. Mol. Med. Rep. 2016, 13, 2320–2326. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ren, D. Allicin protects traumatic spinal cord injury through regulating the HSP70/Akt/iNOS pathway in mice. Mol. Med. Rep. 2016, 14, 3086–3092. [Google Scholar] [CrossRef]
- Wang, W.; Du, Z.; Nimiya, Y.; Sukamtoh, E.; Kim, D.; Zhang, G. Allicin inhibits lymphangiogenesis through suppressing activation of vascular endothelial growth factor (VEGF) receptor. J. Nutr. Biochem. 2016, 29, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Omar, S.H.; Al-Wabel, N.A. Organosulfur compounds and possible mechanism of garlic in cancer. Saudi Pharm. J. 2010, 18, 51–58. [Google Scholar] [CrossRef]
- Marchese, A.; Barbieri, R.; Sanches-Silva, A.; Daglia, M.; Nabavi, S.F.; Jafari, N.J.; Izadi, M.; Ajami, M.; Nabavi, S.M. Antifungal and antibacterial activities of allicin: A review. Trends Food Sci. Technol. 2016, 52, 49–56. [Google Scholar] [CrossRef]
- Borlinghaus, J.; Albrecht, F.; Gruhlke, M.C.H.; Nwachukwu, I.D.; Slusarenko, A.J. Allicin: Chemistry and biological properties. Molecules 2014, 19, 12591–12618. [Google Scholar] [CrossRef]
- Ariyanta, H.A.; Ivandini, T.A.; Yulizar, Y. A novel way of the synthesis of three-dimensional (3D) MoS2 cauliflowers using allicin. Chem. Phys. Lett. 2021, 767, 138345. [Google Scholar] [CrossRef]
- Putnik, P.; Gabrić, D.; Roohinejad, S.; Barba, F.J.; Granato, D.; Mallikarjunan, K.; Lorenzo, J.M.; Kovačević, D.B. An overview of organosulfur compounds from Allium spp.: From processing and preservation to evaluation of their bioavailability, antimicrobial, and anti-inflammatory properties. Food Chem. 2019, 276, 680–691. [Google Scholar] [CrossRef]
- Modem, S.; DiCarlo, S.E.; Reddy, T.R. Fresh garlic extract induces growth arrest and morphological differentiation of MCF7 breast cancer cells. Genes Cancer 2012, 3, 177–186. [Google Scholar] [CrossRef]
- Revtovich, S.; Lyfenko, A.; Tkachev, Y.; Kulikova, V.; Koval, V.; Puchkov, V.; Anufrieva, N.; Solyev, P.; Morozova, E. Anticandidal Activity of In Situ Methionine γ-Lyase-Based Thiosulfinate Generation System vs. Synthetic Thiosulfinates. Pharmaceuticals 2023, 16, 1695. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Ruan, J.; Zhuang, X.; Zhang, X.; Li, Z. Phytochemicals of garlic: Promising candidates for cancer therapy. Biomed. Pharmacother. 2020, 123, 109730. [Google Scholar] [CrossRef]
- Sarvizadeh, M.; Hasanpour, O.; Naderi Ghale-Noie, Z.; Mollazadeh, S.; Rezaei, M.; Pourghadamyari, H.; Masoud Khooy, M.; Aschner, M.; Khan, H.; Rezaei, N. Allicin and digestive system cancers: From chemical structure to its therapeutic opportunities. Front. Oncol. 2021, 11, 650256. [Google Scholar] [CrossRef]
- Pandey, P.; Khan, F.; Alshammari, N.; Saeed, A.; Aqil, F.; Saeed, M. Updates on the anticancer potential of garlic organosulfur compounds and their nanoformulations: Plant therapeutics in cancer management. Front. Pharmacol. 2023, 14, 1154034. [Google Scholar] [CrossRef]
- Chen, X.; Li, H.; Xu, W.; Huang, K.; Zhai, B.; He, X. Self-assembling cyclodextrin-based nanoparticles enhance the cellular delivery of hydrophobic allicin. J. Agric. Food Chem. 2020, 68, 11144–11150. [Google Scholar] [CrossRef]
- Alyasiri, F.J.; Ghobeh, M.; Tabrizi, M.H. Preparation and Characterization of Allicin-Loaded Solid Lipid Nanoparticles Surface-Functionalized with Folic Acid-Bonded Chitosan: In Vitro Anticancer and Antioxidant Activities. FBL 2023, 28, 135. [Google Scholar] [CrossRef] [PubMed]
- Ossama, M.; Hathout, R.M.; Attia, D.A.; Mortada, N.D. Enhanced Allicin Cytotoxicity on HEPG-2 Cells Using Glycyrrhetinic Acid Surface-Decorated Gelatin Nanoparticles. ACS Omega 2019, 4, 11293–11300. [Google Scholar] [CrossRef] [PubMed]
- Hashemy, S.I.; Amiri, H.; Hosseini, H.; Sadeghzadeh, F.; Jaseem, M.M.M.; Tabrizi, M.H. PEGylated lecithin–chitosan–folic acid nanoparticles as nanocarriers of allicin for in vitro controlled release and anticancer effects. Appl. Biochem. Biotechnol. 2023, 195, 4036–4052. [Google Scholar] [CrossRef] [PubMed]
- Alare, K.; Alare, T. Review of toxicity of allicin from garlic. Open Access J. Toxicol. 2020, 3. [Google Scholar] [CrossRef]
- Zhu, B.; Zou, L.; Qi, L.; Zhong, R.; Miao, X. Allium vegetables and garlic supplements do not reduce risk of colorectal cancer, based on meta-analysis of prospective studies. Clin. Gastroenterol. Hepatol. 2014, 12, 1991–2001. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Das, S.; Banik, S. Fabrication and physicochemical investigation of pH-responsive alginate/pectin hybrid network hydrogel for improved stability and controlled release of diallyl thiosulfinate. Mater. Today Commun. 2024, 38, 108235. [Google Scholar] [CrossRef]
- Talib, W.H.; Atawneh, S.; Shakhatreh, A.N.; Hamed, R.A.; Al-Yasari, I.H. Anticancer potential of garlic bioactive constituents: Allicin, Z-ajoene, and organosulfur compounds. Pharmacia 2024, 71, 1–23. [Google Scholar] [CrossRef]
- Țigu, A.B.; Moldovan, C.S.; Toma, V.-A.; Farcaș, A.D.; Moț, A.C.; Jurj, A.; Fischer-Fodor, E.; Mircea, C.; Pârvu, M. Phytochemical analysis and in vitro effects of Allium fistulosum L. and Allium sativum L. extracts on human normal and tumor cell lines: A comparative study. Molecules 2021, 26, 574. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Hanahan, D. Hallmarks of cancer: New dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Adverse effects of cancer chemotherapy: Anything new to improve tolerance and reduce sequelae? Front. Pharmacol. 2018, 9, 362658. [Google Scholar] [CrossRef]
- Harries, L.W. Dysregulated RNA processing and metabolism: A new hallmark of ageing and provocation for cellular senescence. FEBS J. 2023, 290, 1221–1234. [Google Scholar] [CrossRef]
- Sonugür, F.G.; Akbulut, H. The Role of Tumor Microenvironment in Genomic Instability of Malignant Tumors. Front. Genet. 2019, 10, 485489. [Google Scholar] [CrossRef]
- Negrini, S.; Gorgoulis, V.G.; Halazonetis, T.D. Genomic instability—An evolving hallmark of cancer. Nat. Rev. Mol. Cell Biol. 2010, 11, 220–228. [Google Scholar] [CrossRef]
- Rusin, M.; Zajkowicz, A.; Butkiewicz, D. Resveratrol induces senescence-like growth inhibition of U-2 OS cells associated with the instability of telomeric DNA and upregulation of BRCA1. Mech. Ageing Dev. 2009, 130, 528–537. [Google Scholar] [CrossRef]
- Talib, W.H. Anticancer and antimicrobial potential of plant-derived natural products. In Phytochemicals—Bioactivities and Impact on Health; Rasooli, I., Ed.; Intech Open: Rijeka, Croatia, 2011; pp. 141–158. [Google Scholar]
- Anand, S.K.; Sharma, A.; Singh, N.; Kakkar, P. Entrenching role of cell cycle checkpoints and autophagy for maintenance of genomic integrity. DNA Repair 2020, 86, 102748. [Google Scholar] [CrossRef]
- Aguilera, A.; García-Muse, T. Causes of genome instability. Annu. Rev. Genet. 2013, 47, 1–32. [Google Scholar] [CrossRef]
- Takeshima, H.; Ushijima, T. Accumulation of genetic and epigenetic alterations in normal cells and cancer risk. npj Precis. Oncol. 2019, 3, 7. [Google Scholar] [CrossRef]
- Block, K.I.; Gyllenhaal, C.; Lowe, L.; Amedei, A.; Amin, A.R.M.R.; Amin, A.; Aquilano, K.; Arbiser, J.; Arreola, A.; Arzumanyan, A. Designing a broad-spectrum integrative approach for cancer prevention and treatment. Semin. Cancer Biol. 2015, 35, S276–S304. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, D. Allicin suppresses the migration and invasion in cervical cancer cells mainly by inhibiting NRF2. Exp. Ther. Med. 2019, 17, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Guterres, A.N.; Villanueva, J. Targeting telomerase for cancer therapy. Oncogene 2020, 39, 5811–5824. [Google Scholar] [CrossRef] [PubMed]
- Almatroodi, S.A. Bioactive Compounds of Garlic: Role in The Management of Various Types of Cancer. Pharmanest Int. J. Adv. Pharm. Sci 2020, 11, 1–13. [Google Scholar]
- Sun, L.; Wang, X. Effects of allicin on both telomerase activity and apoptosis in gastric cancer SGC-7901 cells. World J. Gastroenterol. 2003, 9, 1930. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Pinto, J.T.; Gundersen, G.G.; Weinstein, I.B. Effects of a series of organosulfur compounds on mitotic arrest and induction of apoptosis in colon cancer cells. Mol. Cancer Ther. 2005, 4, 1388–1398. [Google Scholar] [CrossRef]
- Walag, A.M.P.; Ahmed, O.; Jeevanandam, J.; Akram, M.; Ephraim-Emmanuel, B.C.; Egbuna, C.; Semwal, P.; Iqbal, M.; Hassan, S.; Uba, J.O. Health benefits of organosulfur compounds. In Functional Foods and Nutraceuticals: Bioactive Components, Formulations and Innovations; Springer: Cham, Switzerland, 2020; pp. 445–472. [Google Scholar]
- Nagini, S. Cancer chemoprevention by garlic and its organosulfur compounds-panacea or promise? In Anti-Cancer Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Anti-Cancer Agents); Bentham Science Publishers: Potomac, MD, USA, 2008; Volume 8, pp. 313–321. [Google Scholar]
- Miron, T.; Wilchek, M.; Sharp, A.; Nakagawa, Y.; Naoi, M.; Nozawa, Y.; Akao, Y. Allicin inhibits cell growth and induces apoptosis through the mitochondrial pathway in HL60 and U937 cells. J. Nutr. Biochem. 2008, 19, 524–535. [Google Scholar] [CrossRef]
- Park, S.-Y.; Cho, S.-J.; Kwon, H.-c.; Lee, K.-R.; Rhee, D.-K.; Pyo, S. Caspase-independent cell death by allicin in human epithelial carcinoma cells: Involvement of PKA (vol 224, pg 123, 2005). Cancer Lett. 2019, 444, 188. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, H.Y.; Lee, J.H.; Yoon, K.H.; Chang, M.S.; Park, S.K. The age-dependent induction of apoptosis-inducing factor (AIF) in the human semitendinosus skeletal muscle. Cell. Mol. Biol. Lett. 2010, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bat-Chen, W.; Golan, T.; Peri, I.; Ludmer, Z.; Schwartz, B. Allicin purified from fresh garlic cloves induces apoptosis in colon cancer cells via Nrf2. Nutr. Cancer 2010, 62, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, Y.-i.; Kuranaga, E. Caspase-dependent non-apoptotic processes in development. Cell Death Differ. 2017, 24, 1422–1430. [Google Scholar] [CrossRef]
- Choe, J.-Y.; Park, K.-Y.; Kim, S.-K. Oxidative stress by monosodium urate crystals promotes renal cell apoptosis through mitochondrial caspase-dependent pathway in human embryonic kidney 293 cells: Mechanism for urate-induced nephropathy. Apoptosis 2015, 20, 38–49. [Google Scholar] [CrossRef]
- Rosas-González, V.C.; Téllez-Bañuelos, M.C.; Hernández-Flores, G.; Bravo-Cuellar, A.; Aguilar-Lemarroy, A.; Jave-Suárez, L.F.; Haramati, J.; Solorzano-Ibarra, F.; Ortiz-Lazareno, P.C. Differential effects of alliin and allicin on apoptosis and senescence in luminal A and triple-negative breast cancer: Caspase, ΔΨm, and pro-apoptotic gene involvement. Fundam. Clin. Pharmacol. 2020, 34, 671–686. [Google Scholar] [CrossRef]
- Li, C.; Jing, H.; Ma, G.; Liang, P. Allicin induces apoptosis through activation of both intrinsic and extrinsic pathways in glioma cells. Mol. Med. Rep. 2018, 17, 5976–5981. [Google Scholar] [CrossRef]
- Xiang, Y.; Zhao, J.; Zhao, M.; Wang, K. Allicin activates autophagic cell death to alleviate the malignant development of thyroid cancer. Exp. Ther. Med. 2018, 15, 3537–3543. [Google Scholar] [CrossRef]
- Pandey, N.; Tyagi, G.; Kaur, P.; Pradhan, S.; Rajam, M.V.; Srivastava, T. Allicin overcomes hypoxia mediated cisplatin resistance in lung cancer cells through ROS mediated cell death pathway and by suppressing hypoxia inducible factors. Cell. Physiol. Biochem. 2020, 54, 748–766. [Google Scholar]
- Chen, H.; Zhu, B.; Zhao, L.; Liu, Y.; Zhao, F.; Feng, J.; Jin, Y.; Sun, J.; Geng, R.; Wei, Y. Allicin inhibits proliferation and invasion in vitro and in vivo via SHP-1-mediated STAT3 signaling in cholangiocarcinoma. Cell. Physiol. Biochem. 2018, 47, 641–653. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, Z.; Cao, Z.; Li, L. Allicin induces apoptosis in EL-4 cells in vitro by activation of expression of caspase-3 and-12 and up-regulation of the ratio of Bax/Bcl-2. Nat. Prod. Res. 2012, 26, 1033–1037. [Google Scholar] [CrossRef]
- Luo, Q.; Wu, X.; Zhao, P.; Nan, Y.; Chang, W.; Zhu, X.; Su, D.; Liu, Z. OTUD1 activates caspase-independent and caspase-dependent apoptosis by promoting AIF nuclear translocation and MCL1 degradation. Adv. Sci. 2021, 8, 2002874. [Google Scholar] [CrossRef] [PubMed]
- Joza, N.; Susin, S.A.; Daugas, E.; Stanford, W.L.; Cho, S.K.; Li, C.Y.J.; Sasaki, T.; Elia, A.J.; Cheng, H.Y.M.; Ravagnan, L. Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death. Nature 2001, 410, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Luo, R.; Fang, D.; Hang, H.; Tang, Z. The mechanism in gastric cancer chemoprevention by allicin. In Anti-Cancer Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Anti-Cancer Agents); Bentham Science Publishers: Potomac, MD, USA, 2016; Volume 16, pp. 802–809. [Google Scholar]
- Wu, X.; Cai, J.; Li, X.; Li, H.; Li, J.; Bai, X.; Liu, W.; Han, Y.; Xu, L.; Zhang, D. Allicin protects against cisplatin-induced vestibular dysfunction by inhibiting the apoptotic pathway. Eur. J. Pharmacol. 2017, 805, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Khakbaz, P.; Panahizadeh, R.; Vatankhah, M.A.; Najafzadeh, N. Allicin reduces 5-fluorouracil-resistance in gastric cancer cells through modulating MDR1, DKK1, and WNT5A expression. Drug Res. 2021, 71, 448–454. [Google Scholar] [CrossRef]
- Chen, F.; Li, H.; Wang, Y.; Gao, M.; Cheng, Y.; Liu, D.; Jia, M.; Zhang, J. Inhibition of allicin in Eca109 and EC9706 cells via G2/M phase arrest and mitochondrial apoptosis pathway. J. Funct. Foods 2016, 25, 523–536. [Google Scholar] [CrossRef]
- Zhang, W.; Ha, M.; Gong, Y.; Xu, Y.; Dong, N.; Yuan, Y. Allicin induces apoptosis in gastric cancer cells through activation of both extrinsic and intrinsic pathways. Oncol. Rep. 2010, 24, 1585–1592. [Google Scholar] [CrossRef]
- Yang, Z.; Du, J.; Zhu, J.; Rong, Y.; Chen, S.; Yu, L.; Deng, X.; Zhang, X.; Sheng, H.; Yang, L. Allicin inhibits proliferation by decreasing IL-6 and IFN-β in HCMV-infected glioma cells. Cancer Manag. Res. 2020, 12, 7305–7317. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.l.; Wu, S.f.; Xu, S.t.; Ma, Y.c.; Wang, R.; Jin, S.; Zhou, S. Allicin enhances the radiosensitivity of colorectal cancer cells via inhibition of NF-κB signaling pathway. J. Food Sci. 2020, 85, 1924–1931. [Google Scholar] [CrossRef]
- Lv, Q.; Xia, Q.; Li, J.; Wang, Z. Allicin suppresses growth and metastasis of gastric carcinoma: The key role of microRNA-383-5p-mediated inhibition of ERBB4 signaling. Biosci. Biotechnol. Biochem. 2020, 84, 1997–2004. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Liu, H.; Chen, Y.; Yan, W. The effect of allicin on cell proliferation and apoptosis compared to blank control and cis-platinum in oral tongue squamous cell carcinoma. OncoTargets Ther. 2020, 12, 13183–13189. [Google Scholar] [CrossRef]
- Amin, A.R.M.R.; Karpowicz, P.A.; Carey, T.E.; Arbiser, J.; Nahta, R.; Chen, Z.G.; Dong, J.-T.; Kucuk, O.; Khan, G.N.; Huang, G.S. Evasion of anti-growth signaling: A key step in tumorigenesis and potential target for treatment and prophylaxis by natural compounds. Semin. Cancer Biol. 2015, 35, S55–S77. [Google Scholar] [CrossRef]
- Chen, J. The cell-cycle arrest and apoptotic functions of p53 in tumor initiation and progression. Cold Spring Harb. Perspect. Med. 2016, 6, a026104. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.-L.; Ho, C.-T.; Chung, J.-G.; Raghu, R.; Lo, Y.-C.; Sheen, L.-Y. Allicin induces anti-human liver cancer cells through the p53 gene modulating apoptosis and autophagy. J. Agric. Food Chem. 2013, 61, 9839–9848. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.S.; Henley, D.C.; Bukovsky, A.; Seth, P.; Wimalasena, J. Multifaceted regulation of cell cycle progression by estrogen: Regulation of Cdk inhibitors and Cdc25A independent of cyclin D1-Cdk4 function. Mol. Cell. Biol. 2001, 21, 794–810. [Google Scholar] [CrossRef]
- Sofi, S.; Mehraj, U.; Qayoom, H.; Aisha, S.; Asdaq, S.M.B.; Almilaibary, A.; Mir, M.A. Cyclin-dependent kinases in breast cancer: Expression pattern and therapeutic implications. Med. Oncol. 2022, 39, 106. [Google Scholar] [CrossRef]
- Malla, R.; Marni, R.; Chakraborty, A.; Kamal, M.A. Diallyl disulfide and diallyl trisulfide in garlic as novel therapeutic agents to overcome drug resistance in breast cancer. J. Pharm. Anal. 2022, 12, 221–231. [Google Scholar] [CrossRef]
- Kiesel, V. Diallyl Trisulfide Modulates Notch Pathway Components in Breast Cancer Cells. Master’s Thesis, Purdue University, West Lafayette, IN, USA, 2016. [Google Scholar]
- Li, Y.; Zhang, J.; Zhang, L.; Si, M.; Yin, H.; Li, J. Diallyl trisulfide inhibits proliferation, invasion and angiogenesis of osteosarcoma cells by switching on suppressor microRNAs and inactivating of Notch-1 signaling. Carcinogenesis 2013, 34, 1601–1610. [Google Scholar] [CrossRef] [PubMed]
- Yaswen, P.; MacKenzie, K.L.; Keith, W.N.; Hentosh, P.; Rodier, F.; Zhu, J.; Firestone, G.L.; Matheu, A.; Carnero, A.; Bilsland, A.; et al. Therapeutic targeting of replicative immortality. Semin. Cancer Biol. 2015, 35, S104–S128. [Google Scholar] [CrossRef] [PubMed]
- Patra, S.; Nayak, R.; Patro, S.; Pradhan, B.; Sahu, B.; Behera, C.; Bhutia, S.K.; Jena, M. Chemical diversity of dietary phytochemicals and their mode of chemoprevention. Biotechnol. Rep. 2021, 30, e00633. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Song, Y.; Lian, J.; Wang, Z. Allicin inhibits the invasion of lung adenocarcinoma cells by altering tissue inhibitor of metalloproteinase/matrix metalloproteinase balance via reducing the activity of phosphoinositide 3-kinase/AKT signaling. Oncol. Lett. 2017, 14, 468–474. [Google Scholar] [CrossRef][Green Version]
- Cao, Y. Tumorigenesis as a process of gradual loss of original cell identity and gain of properties of neural precursor/progenitor cells. Cell Biosci. 2017, 7, 61. [Google Scholar] [CrossRef]
- Swetha, M.; Keerthana, C.; Rayginia, T.P.; Anto, R.J. Cancer chemoprevention: A strategic approach using phytochemicals. Front. Pharmacol. 2022, 12, 809308. [Google Scholar]
- Li, X.; Ni, J.; Tang, Y.; Wang, X.; Tang, H.; Li, H.; Zhang, S.; Shen, X. Allicin inhibits mouse colorectal tumorigenesis through suppressing the activation of STAT3 signaling pathway. Nat. Prod. Res. 2019, 33, 2722–2725. [Google Scholar] [CrossRef]
- Ferguson, D.T.; Taka, E.; Tilghman, S.L.; Womble, T.; Redmond, B.V.; Gedeon, S.; Flores-Rozas, H.; Reed, S.L.; Soliman, K.F.; Kanga, K.J. The Anticancer Effects of the Garlic Organosulfide Diallyl Trisulfide through the Attenuation of B [a] P-Induced Oxidative Stress, AhR Expression, and DNA Damage in Human Premalignant Breast Epithelial (MCF-10AT1) Cells. Int. J. Mol. Sci. 2024, 25, 923. [Google Scholar] [CrossRef]
- Bassal, M.A. The Interplay between Dysregulated Metabolism and Epigenetics in Cancer. Biomolecules 2023, 13, 944. [Google Scholar] [CrossRef] [PubMed]
- Isa, A. Dysregulated Metabolism in Cancer. Biomed. Res. Ther. 2022, 9, 5201–5208. [Google Scholar] [CrossRef]
- Gruhlke, M.C.; Antelmann, H.; Bernhardt, J.; Kloubert, V.; Rink, L.; Slusarenko, A.J. The human allicin-proteome: S-thioallylation of proteins by the garlic defence substance allicin and its biological effects. Free Radic. Biol. Med. 2019, 131, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, L.; Sandhu, J.K.; Harper, M.-E.; Cuperlovic-Culf, M. Role of Glutathione in Cancer: From Mechanisms to Therapies. Biomolecules 2020, 10, 1429. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, X.; Luo, W.; Zhu, J.; Zhao, J.; Wang, M.; Sang, L.; Chang, B.; Wang, B. Allicin in Digestive System Cancer: From Biological Effects to Clinical Treatment. Front. Pharmacol. 2022, 13, 903259. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Chang, W.; Wang, X.; Liu, F.; Wang, X.; Yuan, D.; Zhang, Y. Allicin inhibits osteosarcoma growth by promoting oxidative stress and autophagy via the inactivation of the lncRNA MALAT1-miR-376a-Wnt/β-catenin signaling pathway. Oxidative Med. Cell. Longev. 2022, 2022, 4857814. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ding, L.; Liu, J.; Savarin, P.; Wang, X.; Zhao, K.; Ding, W.; Hou, Y. Allicin ameliorates glucose and lipid metabolism via modulation of gut microbiota and bile acid profile in diabetic rats. J. Funct. Foods 2023, 111, 105899. [Google Scholar] [CrossRef]
- Arias, J.-I.; Aller, M.-A.; Prieto, I.; Arias, A.; De Julian, Z.; Yang, H.; Arias, J. The amazing power of cancer cells to recapitulate extraembryonic functions: The Cuckoo’s tricks. J. Oncol. 2012, 2012, 521284. [Google Scholar] [CrossRef]
- Singh, N.; Baby, D.; Rajguru, J.P.; Patil, P.B.; Thakkannavar, S.S.; Pujari, V.B. Inflammation and cancer. Ann. Afr. Med. 2019, 18, 121–126. [Google Scholar] [CrossRef]
- Karin, M. Nuclear factor-κB in cancer development and progression. Nature 2006, 441, 431–436. [Google Scholar] [CrossRef]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef]
- Greten, F.R.; Grivennikov, S.I. Inflammation and cancer: Triggers, mechanisms, and consequences. Immunity 2019, 51, 27–41. [Google Scholar] [CrossRef]
- Lang, A.; Lahav, M.; Sakhnini, E.; Barshack, I.; Fidder, H.H.; Avidan, B.; Bardan, E.; Hershkoviz, R.; Bar-Meir, S.; Chowers, Y. Allicin inhibits spontaneous and TNF-α induced secretion of proinflammatory cytokines and chemokines from intestinal epithelial cells. Clin. Nutr. 2004, 23, 1199–1208. [Google Scholar] [CrossRef]
- Li, C.; Lun, W.; Zhao, X.; Lei, S.; Guo, Y.; Ma, J.; Zhi, F. Allicin alleviates inflammation of trinitrobenzenesulfonic acid-induced rats and suppresses P38 and JNK pathways in Caco-2 cells. Mediat. Inflamm. 2015, 2015, 434692. [Google Scholar] [CrossRef]
- Savairam, V.D.; Patil, N.A.; Borate, S.R.; Ghaisas, M.M.; Shete, R.V. Allicin: A review of its important pharmacological activities. Pharmacol. Res. Mod. Chin. Med. 2023, 8, 100283. [Google Scholar] [CrossRef]
- Lugano, R.; Ramachandran, M.; Dimberg, A. Tumor angiogenesis: Causes, consequences, challenges and opportunities. Cell. Mol. Life Sci. CMLS 2020, 77, 1745–1770. [Google Scholar] [CrossRef]
- Sela, U.; Brill, A.; Kalchenko, V.; Dashevsky, O.; Hershkoviz, R. Allicin inhibits blood vessel growth and downregulates Akt phosphorylation and actin polymerization. Nutr. Cancer 2008, 60, 412–420. [Google Scholar] [CrossRef]
- Sun, H.-H.; Feng, X.-M.; Wang, J.-C.; Cai, J. Allicin can suppress the activity of vascular endothelial cells probably by regulating JAK2/STAT3 pathway. Mol. Cell. Biochem. 2021, 476, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Flournoy, J.; Ashkanani, S.; Chen, Y. Mechanical regulation of signal transduction in angiogenesis. Front. Cell Dev. Biol. 2022, 10, 933474. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Shu, Y.; Cui, T.; Fu, P. Allicin inhibits human renal clear cell carcinoma progression via suppressing HIF pathway. Int. J. Clin. Exp. Med. 2015, 8, 20573–20580. [Google Scholar]
- Alhasan, L.; Addai, Z.R. Allicin-induced modulation of angiogenesis in lung cancer cells (A549). Trop. J. Pharm. Res. 2018, 17, 2129–2134. [Google Scholar] [CrossRef]
- Veterini, L.; Savitri, A.D.; Widyaswari, M.S.; Muhammad, A.R.; Fairus, A.; Zulfikar, M.Q.B.; Astri, M.; Ramasima, N.A.; ANGGRAENI, D.P.; Nainatika, R.S. In silico study of the potential of garlic allicin compound as anti-angiogenesis in breast cancer. Trop. J. Nat. Prod. Res. 2021, 5, 1995–1996. [Google Scholar]
- Matsuura, N.; Miyamae, Y.; Yamane, K.; Nagao, Y.; Hamada, Y.; Kawaguchi, N.; Katsuki, T.; Hirata, K.; Sumi, S.; Ishikawa, H. Aged garlic extract inhibits angiogenesis and proliferation of colorectal carcinoma cells. J. Nutr. 2006, 136, 842s–846s. [Google Scholar] [CrossRef] [PubMed]
- Shang, A.; Cao, S.-Y.; Xu, X.-Y.; Gan, R.-Y.; Tang, G.-Y.; Corke, H.; Mavumengwana, V.; Li, H.-B. Bioactive Compounds and Biological Functions of Garlic (Allium sativum L.). Foods 2019, 8, 246. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Liu, Y.Q.; Cao, W.K.; Chen, X.F.; Wan, Y.Y.; Heng, C.; Xu, L.J. Effects of allicin on invasion and metastasis of colon cancer LoVo cell line in vitro. Zhonghua Yi Xue Za Zhi 2009, 89, 1382–1386. [Google Scholar] [PubMed]
- Lee, C.G.; Lee, H.-W.; Kim, B.-O.; Rhee, D.-K.; Pyo, S. Allicin inhibits invasion and migration of breast cancer cells through the suppression of VCAM-1: Regulation of association between p65 and ER-α. J. Funct. Foods 2015, 15, 172–185. [Google Scholar] [CrossRef]
- Jiang, X.; Zhang, Y.; Li, F.; Zhu, Y.; Chen, Y.; Yang, S.; Sun, G. Allicin as a possible adjunctive therapeutic drug for stage II oral submucous fibrosis: A preliminary clinical trial in a Chinese cohort. Int. J. Oral Maxillofac. Surg. 2015, 44, 1540–1546. [Google Scholar] [CrossRef]
- Bala, R.; Madaan, R.; Chauhan, S.; Gupta, M.; Dubey, A.K.; Zahoor, I.; Brijesh, H.; Calina, D.; Sharifi-Rad, J. Revitalizing allicin for cancer therapy: Advances in formulation strategies to enhance bioavailability, stability, and clinical efficacy. Naunyn-Schmiedebergs Arch. Pharmacol. 2024, 397, 703–724. [Google Scholar] [CrossRef]
- Lee, G.-Y.; Lee, J.J.; Lee, S.-M. Antioxidant and anticoagulant status were improved by personalized dietary intervention based on biochemical and clinical parameters in cancer patients. Nutr. Cancer 2015, 67, 1083–1092. [Google Scholar] [CrossRef]

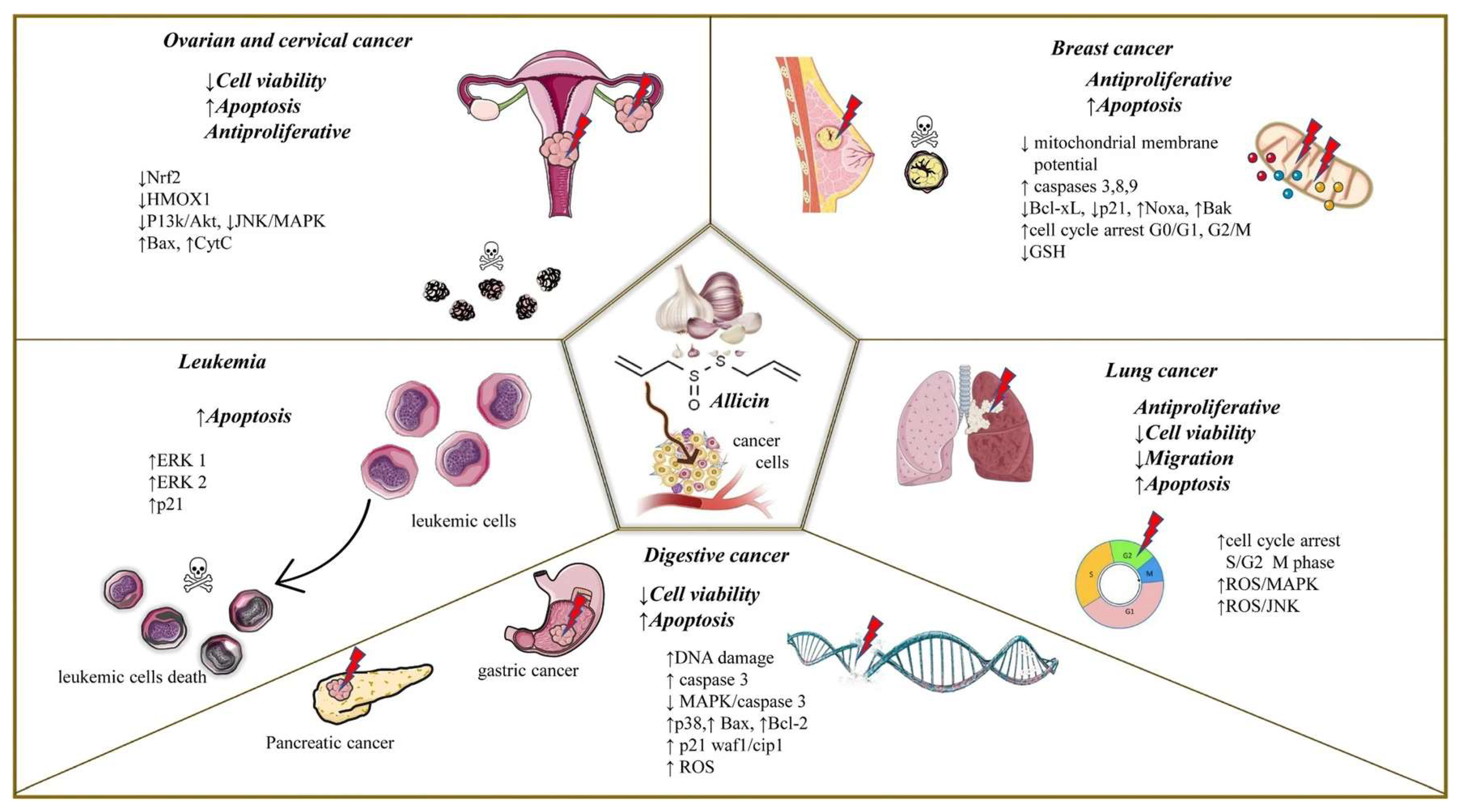
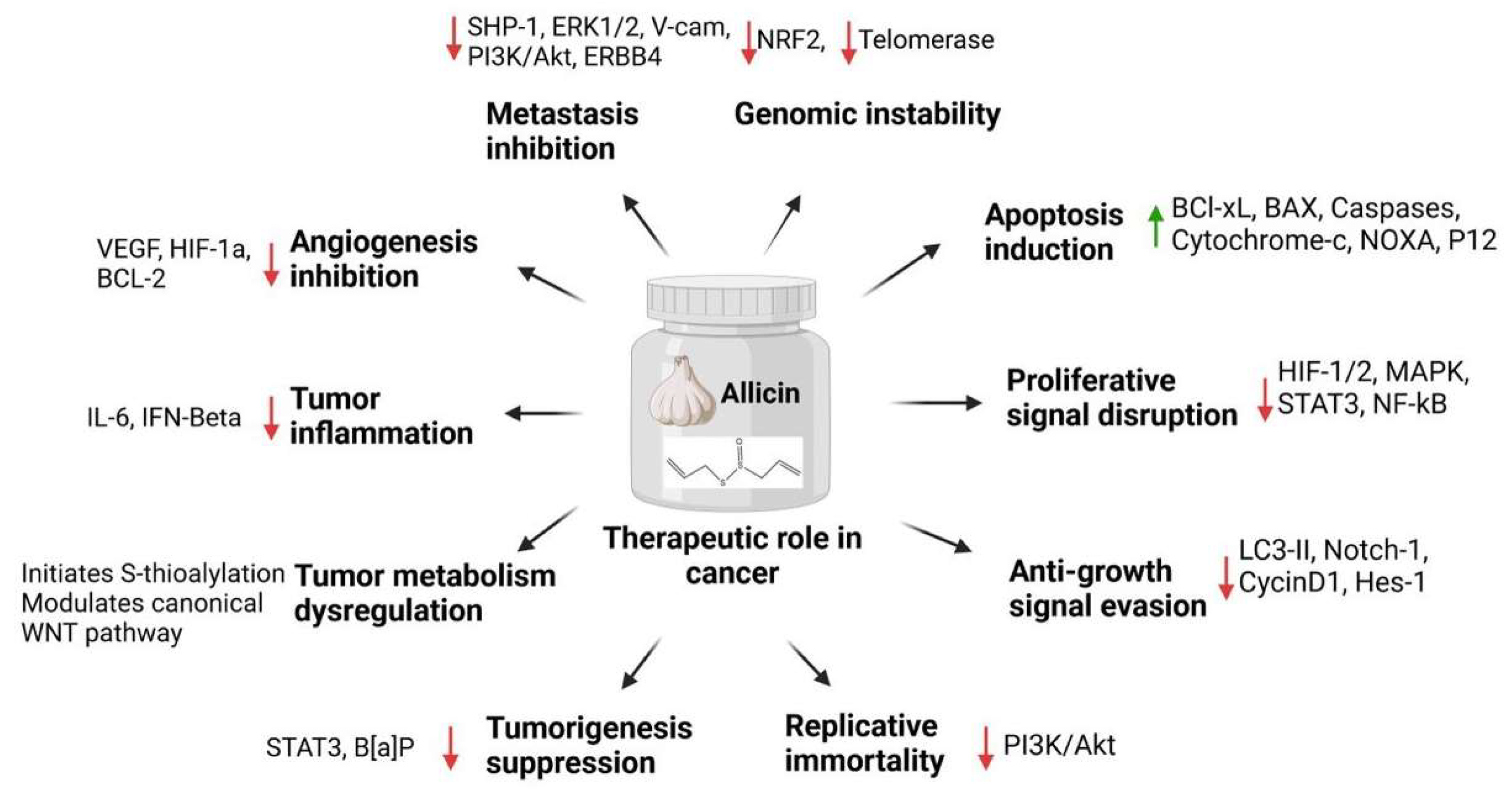
| Cancer Hallmark | Concentration Used | Type of Cells | Experimental Model | Outcomes of the Combination | Reference |
|---|---|---|---|---|---|
| Genomic Instability | 40 nM | Cervical cancer | vitro | Allicin inhibited the expression of NRF2 in cervical cancer SiHa cells. | [49] |
| 0.016 mg/mL, 0.05 mg/mL, and 0.1 mg/mL | Gastric cancer | vitro | Allicin can suppress telomerase activity and induce apoptosis of SGC-7901 cells. | [52] | |
| Inducing Apoptosis | (15–120 μg/mL) | Gastric cancer | vitro | Treatment with allicin reduced the proliferation of SGC-7901 cancer cells and caused them to undergo apoptosis. After allicin treatment, SGC-7901 cells underwent synchronous apoptosis through both intrinsic mitochondrial and extrinsic Fas/FasL-mediated mechanisms. | [74] |
| 18.2 mg/kg | Testicular, ovarian, bladder, head and neck, lung, and cervical cancer | vivo | Allicin protected against vestibular dysfunction caused by cisplatin and potentially avoided oto-vestibulotoxicity caused by cisplatin. | [71] | |
| 30 and 60 µg/mL | Brain cancer | vitro | Allicin can effectively inhibit proliferation and induce apoptosis of both intrinsic and extrinsic pathways in U251 glioma cells. | [63] | |
| allicin (10 µM) and Cisplatin (10 µM) | Thyroid cancer | vitro | The combination of allicin (10 µM) and Cisplatin (10 µM) caused autophagy-dependent cell death in SW1736 and HTh-7 cells. | [64] | |
| 45 μM (MCF-7) and 12 μM, 20 μM, and 45 μM (HCC-70) | Breast cancer | vitro | Allicin decreased cell viability and antiproliferative effects through activation of caspase -3, -8, and -9, leading to (MCF-7 and HCC-70) cell apoptosis through upregulation of P21, NOXA, BAK, and BCL-XL. | [62] | |
| 40 µg/mL | Lung cancer (NSCLC) | vitro | Allicin caused G2-M arrest and death in A549 cells through ROS-dependent changes to p53, p21, and other downstream effectors. | [65] | |
| 10 and 20 mg/kg | vivo | Enhanced the apoptotic caspases by suppressing STAT3 signaling, such as cleaved caspase-3 and cleaved caspase-9 | [66] | ||
| 4 μg/mL and 8 μg/mL (EL-4 cells) | Lymphoma | vitro | Increased caspase-3, -12, and Bax/Bcl2 and cytochrome-c expression and reduced mitochondrial membrane potential. | [67] | |
| Caspase-independent apoptosis | 40 nM | Cervical cancer | vitro | Allicin primarily inhibited NRF2 in cervical cancer cells. | [49] |
| allicin (2–64 g/mL) and 5-FU (10–480 g/mL) | Gastric cancer | vitro | The combination could reverse multidrug resistance in the GC cells and lower the expression of WNT5A, DKK1, MDR1, P-gp, and CD44 levels. | [72] | |
| 40–100 µg/mL | Oesophageal squamous cell carcinoma | vitro | Decreased cell viability and triggered G2/M phase arrest via p53-p21- CDK1/cyclinB and induced apoptosis via mitochondrial signaling pathways. | [73] | |
| Sustained Proliferative Signaling | 40 µg/mL | Lung cancer (NSCLC) | vitro | Allicin reduced the expression of HIF-1 and HIF-2 in hypoxic cells by inhibiting the ROS/MAPK pathway. | [65] |
| 40 µM | Liver cancer | vitro | Allicin inhibited CCA cell proliferation and invasion through suppressing STAT3 signaling. | [66] | |
| 60 μg/mL | Central nervous tumor | vitro | Allicin concentration may boost p53 expression by lowering IE2 protein levels. | [75] | |
| 10 µg/mL | Colorectal cancer | vitro | Allicin inhibited cell proliferation and migration and promoted apoptosis in HCT116 cells. | [76] | |
| 30 mg/kg | vivo | Allicin inhibited colorectal cancer cell proliferation by suppressing NF-κB signalling. | |||
| 10 µg/mL | Gastric cancer | vitro | Allicin efficiently reduced the growth and metastasis by enhancing the expression of miR-383-5p while reducing the expressions of ERBB4, p-PI3K, and p-Akt. | [77] | |
| 50 μg/mL | Oral cancer | vitro | Allicin was shown to be highly effective at inhibiting cell growth and promoting cell death when compared to cis-platinum in OTSCC patients. | [78] | |
| Evasion of Anti-Growth Signalling | 35 µM | Liver cancer | vitro | After allicin exposure for 24 h, Hep G2 and Hep 3B cells were knocked down by p53 and exhibited inhibition of LC3-II protein expression but increased caspase-3 production. | [81] |
| 4 mg/mL dry mass | Breast cancer | vitro | Retinoblastoma (Rb) in breast cancer cell line MCF7 was completely dephosphorylated after 16 h of allicin exposure. | [22] | |
| DATS (50 μM) | Osteosarcoma cells | vitro | Diallyl trisulfide (DATS) downregulated the expression of Notch-1 protein, Hes-1, and cyclin D1. | [86] | |
| Replicative Immortality | 15.0 and 20.0 µM H1299 and A549 | Lung adenocarcinoma | vitro | Allicin reduced cell proliferation by modulating PI3K/AKT signalling. | [89] |
| Tumorigenesis and Carcinogen Activity Suppression | AOM/DSS mice | Colon cancer | vivo | Suppressed the tumorigenesis of colon tumors by inhibition of STAT3 signalling activation. | [92] |
| DATS (MCF7-10AT1 cells) | Breast cancer cells | vitro | DATS suppressed B[a]P (ubiquitous environmental pollutant) carcinogenic activity in normal and cancerous breast cells. | [93] | |
| Tumor Dysregulated Metabolism | 100 µM allicin (Jurkat cells) | Acute T cell leukemia | vitro | Allicin initiated the S-thioallylation of enzymes involved in the catabolism of glucose to pyruvate. | [96] |
| Allicin Saos-2 and U2OS cells | Osteosarcoma | vitro | Allicin encouraged oxidative stress and autophagy in osteosarcoma cells by modulating MALATI-miR-376a-Wnt and β-catenin cascade. | [99] | |
| Allicin (60 mg/kg) Wistar rats (6 weeks) | Diabetes | vivo | Allicin reduced glucose and lipid levels in diabetic rats’ blood, thus improving glucose tolerance. | [100] | |
| Tumor- Inflammation | Allicin 60 µg/mL U87MG cells | Human cytomegalovirus (HCMV) | vitro | Allicin significantly inhibited the expression of IL-6 and IFN-β inflammatory factors which are overexpressed in human cytomegalovirus (HCMV)-infected glioblastoma multiforme (GBM). | [75] |
| Angiogenesis inhibition | Allicin 0.1 mg/mL RCC-9863 cells | Renal carcinoma | vitro | Allicin resulted in a considerable reduction in the expression of HIF-1α in human renal clear cell carcinoma (RCC-9863) cells, also inhibiting VEGF and Bcl-2. | [113] |
| 50 and 100 µg/mL (A549 cells) | Lung cancer | vitro | Allicin can also inhibit angiogenesis in lung cancer cells (A549) by reducing VEGF-A protein expression, suppressing VEGF-A gene expression, targeting the HIF pathway, and stimulating the immune system. | [114] | |
| Breast cancer | silico | Allicin can substantially inhibit the VEGFR-2 receptors in breast cancer, according to an in silico study, which could limit the growth of breast cancer cells. | [115] | ||
| Allicin 10 μM and 10 μg | Lymphangiogenesis | vivo | Allicin effectively inhibited VEGF-C-induced lymphangiogenesis and infiltration of inflammatory cells in a Matrigel plug assay in C57BL/6 mice. | [16] | |
| Tissue Invasion and Metastasis | Allicin 10 µg/mL | Gastric cancer | vitro | Allicin increased the expression of miR-383-5p, which in turn suppressed the activity of the ERBB4/PI3 K/Akt signalling pathway, which has a role in promoting cancer. | [77] |
| Allicin 7.5 and 10.0 µM | Lung adenocarcinoma | vitro | Allicin has the potential to impede the invasion of lung adenocarcinoma cells (A549 and H1299) by modulating the balance of TIMP/MMP via suppressing the activity of the PI3K/AKT signalling pathway. | [89] | |
| Allicin 3 and 6 µg/mL | Colon cancer | vitro | Allicin has demonstrated the ability to hinder the spread and secondary growth of LoVo human colon cancer cells by reducing the expression of VEGF, u-PAR, and HPA mRNA. | [118] | |
| Allicin 20 µM | Cholangiocarcinoma | vitro | Allicin has the potential to inhibit the proliferation, invasion, and metastasis of human CCA cell lines (HuCCT-1 and QBC939). The study suggests this inhibition may be achieved by targeting the SHP-1-mediated STAT3 signalling pathway. | [66] | |
| Allicin 0.1–10 ng/mL | Breast cancer | vitro | Allicin suppressed the invasion and metastasis of MCF-7 cells effectively by inhibiting the activation of ERK1/2 induced by TNF-α. Based on the findings, allicin can hinder the activation of VCAM-1 caused by TNF-α. This is achieved by blocking the ERK1/2 and NF-κB signalling pathways and enhancing the interaction between ER-α and p65. | [119] | |
| Allicin 20 nM | Cervical cancer | vitro | Allicin may potentially inhibit tumor invasion and metastasis in SiHa cells by inhibiting the PI3K/AKT pathway, which is essential for promoting cell growth and survival, and by suppressing NRF2 expression. NRF2 has been linked to the progression of cervical cancer. | [49] | |
| 0, 0.1, 1, and 10 mg/mL | Colorectal cancer | vitro | AGEs could hinder the invasiveness of SW480 and SW620 cells by enhancing the adhesion of endothelial cells to collagen and fibronectin, which can inhibit their ability to move and invade surrounding tissues. | [98] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Talib, W.H.; Baban, M.M.; Azzam, A.O.; Issa, J.J.; Ali, A.Y.; AlSuwais, A.K.; Allala, S.; AL Kury, L.T. Allicin and Cancer Hallmarks. Molecules 2024, 29, 1320. https://doi.org/10.3390/molecules29061320
Talib WH, Baban MM, Azzam AO, Issa JJ, Ali AY, AlSuwais AK, Allala S, AL Kury LT. Allicin and Cancer Hallmarks. Molecules. 2024; 29(6):1320. https://doi.org/10.3390/molecules29061320
Chicago/Turabian StyleTalib, Wamidh H., Media Mohammed Baban, Aya O. Azzam, Jenan J. Issa, Alaa Y. Ali, Alia Kh. AlSuwais, Sana Allala, and Lina T. AL Kury. 2024. "Allicin and Cancer Hallmarks" Molecules 29, no. 6: 1320. https://doi.org/10.3390/molecules29061320
APA StyleTalib, W. H., Baban, M. M., Azzam, A. O., Issa, J. J., Ali, A. Y., AlSuwais, A. K., Allala, S., & AL Kury, L. T. (2024). Allicin and Cancer Hallmarks. Molecules, 29(6), 1320. https://doi.org/10.3390/molecules29061320








