Advancements in the Cultivation, Active Components, and Pharmacological Activities of Taxus mairei
Abstract
1. Introduction
2. Origin and Cultivation
3. Phytochemical Components
3.1. Chemical Composition of Different Parts of T. mairei
3.1.1. Chemical Components of Leaves and Twigs of T. mairei
3.1.2. Chemical Components of the Bark of T. mairei
3.1.3. Chemical Components of the Fruit and Seeds of T. mairei
3.2. Factors Influencing the Active Components of T. mairei
3.3. Comparison of Chemical Components between T. mairei and Other Taxus Species
4. Pharmacological Activity Studies
4.1. Anticancer Activity
4.1.1. Anticancer Activity of Extracts
4.1.2. Anticancer Activity of Monomers and Major Effective Components
4.2. Antidiabetic and Antihypertensive Effects
4.3. Anti-Inflammatory Effects
4.4. Antimicrobial Effects
5. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ning, F.; Zhiming, L. “Plant gold”—Taxus. For. Shanxi 2007, 1, 32. (In Chinese) [Google Scholar] [CrossRef]
- Weiwen, L.; Quan, H. “Plant gold”—Taxus. Middle Sch. Sci. Technol. 2007, 4, 26–27. (In Chinese) [Google Scholar]
- Chen, X.; Xu, X.; Yuan, H.; Luo, T.; Cui, H.; Zhu, T.; Li, S.; Liu, Y.; Lin, T.; Qiao, T.; et al. Nigrospora musae Causing T. mairei stem blight in Sichuan, China. Plant Dis. 2023, 107, 4032. [Google Scholar] [CrossRef]
- Shao, F.; Wilson, I.W.; Qiu, D. The Research Progress of Taxol in Taxus. Curr. Pharm. Biotechnol. 2021, 22, 360–366. [Google Scholar] [CrossRef]
- Hao, D.C.; Xiao, P.G.; Peng, Y.; Liu, M.; Huo, L. Research progress and trend analysis of biology and chemistry of Taxus medicinal resources. Yao Xue Xue Bao 2012, 47, 827–835. [Google Scholar]
- Nižnanský, Ľ.; Osinová, D.; Kuruc, R.; Hengerics Szabó, A.; Szórádová, A.; Masár, M.; Nižnanská, Ž. Natural Taxanes: From Plant Composition to Human Pharmacology and Toxicity. Int. J. Mol. Sci. 2022, 23, 15619. [Google Scholar] [CrossRef]
- Truus, K.; Vaher, M.; Borissova, M.; Robal, M.; Levandi, T.; Tuvikene, R.; Toomike, P.; Kaljurand, M. Characterization of yew tree (Taxus) varieties by fingerprint and principal component analyses. Nat. Prod. Commun. 2012, 7, 1143–1146. [Google Scholar] [CrossRef]
- Gallego-Jara, J.; Lozano-Terol, G.; Sola-Martínez, R.A.; Cánovas-Díaz, M.; de Diego Puente, T. A Compressive Review about Taxol(®): History and Future Challenges. Molecules 2020, 25, 5986. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Chen, Z. Taxus resources in China. J. Northwest For. Coll. 2006, 21, 5. (In Chinese) [Google Scholar]
- Fang, C. Content Determination method and Time variation of the Year of Taxanes, Flavonoids and Polysaccharide in the Needles of T. mairei; Xinjiang Agricultural University: Urumchi, China, 2014. [Google Scholar]
- Wang, Z.; Peng, H.; Li, D. Flora of China; Germplasm Bank of Wild Life in Southwest China: Kunming, China, 2004; Volume 24, p. 4. [Google Scholar]
- Wen, Y.; Xie, W.; Han, W.; Zhou, H.; Cao, J.; Chen, G. Resource status and distribution characteristics of Taxus chinensis in southern Nanling Mountains. J. Cent. South Univ. For. Technol. 2012, 32, 5. (In Chinese) [Google Scholar]
- Wu, N.; Guo, M.; Zhang, T.; Shu, F.; You, J.; Xu, Y.; Liu, J. Status Quo of Species Resources and Develop ment Countermeasures in Taxus wallichiana var. mairei Nature Reserve e of Ruyuan, Guangdong. Cent. South For. Inventory Plan. 2022, 41, 48–52. [Google Scholar]
- Fei, Y.; Zhou, Y.; Yong, L.; Shen, J.; Qi, R. A New Forma of the Genus Taxus L. from Hubei, China. Acta Bot. Boreali Occident. Sin. 2016, 36, 1707–1709. (In Chinese) [Google Scholar]
- Li, C.; Liu, Y.; Dong, M.; Shi, Q. Advances in studies on chemical constituents in Taxus chinensis var. mairei. Chin. Tradit. Herb. Drugs 2007, 38, 12. (In Chinese) [Google Scholar]
- Jian, Z.; Meng, L.; Hu, X. An endophytic fungus efficiently producing paclitaxel isolated from Taxus wallichiana var. mairei. Medicine 2017, 96, e7406. [Google Scholar] [CrossRef]
- Yu, S.S.; Sun, Q.W.; Zhang, X.P.; Tian, S.N.; Bo, P.L. Content and distribution of active components in cultivated and wild Taxus chinensis var. mairei plants. Ying Yong Sheng Tai Xue Bao 2012, 23, 2641–2647. [Google Scholar] [PubMed]
- Xie, H.; Qi, H.; Hu, X.; Yang, G. Diterpenoids From Taxus wallichiana var. mairei Cultivated in Hubei province. J. Green Sci. Technol. 2023, 25, 141–146. (In Chinese) [Google Scholar]
- Dong, Q.F.; Liu, J.J.; Yu, R.M. Taxol content comparison in different parts of Taxus madia and Taxus chinensis var. mairei by HPLC. Zhong Yao Cai 2010, 33, 1048–1051. [Google Scholar]
- Yan, J.; Chen, S.; Zhang, D.; Qi, Y. Comparison of taxanes in Taxus. Zhejiang Agric. Sci. 2018, 59, 4. (In Chinese) [Google Scholar]
- Zhang, H.; Zhang, W.; Wang, Y.; Fan, X.; Zhang, X. Determination and Analysis of Taxanes Content in the Branches of Different Taxus Species. J. Shanghai Jiaotong Univ. Agric. Sci. 2010, 28, 9–13. (In Chinese) [Google Scholar]
- Zhang, D. Study on the Taxol Content in Different Parts of Taxus Growing in China. J. For. Environ. 2003, 23, 160–163. (In Chinese) [Google Scholar]
- Xiaoyu, H.; Liuting, Z.; Liwei, L. Determination of 10-deacetylbacatine III in four varieties of Taxus chinensis. J. Jingchu Univ. Technol. 2017, 32, 4. (In Chinese) [Google Scholar]
- Ye, B.; Hua, C.; Liang, S.; Wang, Y. Accumulation of taxane content in different parts of taxus chinensis. Shaanxi J. Tradit. Chin. Med. 2020, 41, 3. (In Chinese) [Google Scholar]
- Yang, X.; Wang, R.; Zhang, J.; Li, Y.; Qin, Y. Content analysis on 6 kinds of taxanes in branches, leaves and fruits of Taxus chinensis var. mairei. J. Hunan Agric. Univ. Nat. Sci. 2016, 5. [Google Scholar]
- Li, S.; Zhang, C.; Nian, H.; Xiong, Y. Study on microwave extraction technology of flavonoids from Taxus chinensis by response surface analysis. J. Yunnan Univ. Tradit. Chin. Med. 2012, 35, 6–9. (In Chinese) [Google Scholar]
- Ruan, X.; Yan, L.Y.; Li, X.X.; Liu, B.; Zhang, H.; Wang, Q. Optimization of process parameters of extraction of amentoflavone, quercetin and ginkgetin from Taxus chinensis using supercritical CO2 plus co-solvent. Molecules 2014, 19, 17682–17696. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Y.; Huang, X.; Feng, K.; Ma, D.; Sun, J.; Shen, J.; Xiong, Y. Study on the content of polysaccharides in different parts of Taxus chinensis var. extracts. Chin. J. Tradit. Chin. Med. 2014, 29, 6. (In Chinese) [Google Scholar]
- Bin, T. Study on Material Basis of Antidiabetic Effect of Branches and Leaves of Southern Taxus chinensis; Nanjing University of Traditional Chinese Medicine: Nanjing, China, 2014. [Google Scholar]
- Wei, Q.; Sun, T. Review on Anti-tumor Components from Taxus and Their Derivatives. Nat. Prod. Res. 2016, 28, 13. (In Chinese) [Google Scholar]
- Zhou, M.; Wang, D.; Zhang, M.; Luo, H.; Yang, X.; Qin, Z.; Qin, T. Study on Volatile Components of Taxuswallic hianavar. marirei in Guizhou. Guangzhou Chem. Ind. 2019, 47, 6. (In Chinese) [Google Scholar]
- Li, L.; Chen, Y.; Ma, Y.; Wang, Z.; Wang, T.; Xie, Y. Optimization of Taxol Extraction Process Using Response Surface Methodology and Investigation of Temporal and Spatial Distribution of Taxol in Taxus mairei. Molecules 2021, 26, 5485. [Google Scholar] [CrossRef]
- Lun, X.; Lin, Y.; Chai, F.; Fan, C.; Li, H.; Liu, J. Reviews of emission of biogenic volatile organic compounds (BVOCs) in Asia. J. Environ. Sci. 2020, 95, 266–277. [Google Scholar] [CrossRef]
- Yu, H.; Holopainen, J.K.; Kivimäenpää, M.; Virtanen, A.; Blande, J.D. Potential of Climate Change and Herbivory to Affect the Release and Atmospheric Reactions of BVOCs from Boreal and Subarctic Forests. Molecules 2021, 26, 2283. [Google Scholar] [CrossRef]
- Lin, J.; Jian, Y.; Li, J.; Wang, X.; He, J.; Luo, S. Study on releasing characteristics of fendolinium from living branches and leaves of Taxus chinensis. J. Sichuan For. Sci. Technol. 2021, 42, 92–97. [Google Scholar]
- Shi, Q.W.; Oritani, T.; Sugiyama, T.; Kiyota, H. Two new taxanes from Taxus chinensis var. mairei. Planta Med. 1998, 64, 766–769. [Google Scholar] [CrossRef] [PubMed]
- Fan, X. Study on Extraction and Enrichment of Main Taxanes from Needles of Taxus chinensis; Beijing Forestry University: Beijing, China, 2021. [Google Scholar]
- Fang, C. Methods for Determination of Taxanes, Flavonoids and Polysaccharides in Taxus Conifer and Their Seasonal Changes; Xinjiang Agricultural University: Urumchi, China, 2017. [Google Scholar]
- Chen, F.; Zhang, C.; Yang, Y.; Xiong, Y.K. Dynamic Change of Polysaccharide Content in South Chinese Yew under Storage Conditions of Different Humidity. Mod. Chin. Med. 2016, 772–775. (In Chinese) [Google Scholar]
- Su, J.; Shi, H.; Wang, L.; Guo, R.; Ren, T.; Wu, Y. Chemical Constituents of Bark of Taxus chinensis var. mairei. J. Chin. Med. Mater. 2014, 37, 243–251. (In Chinese) [Google Scholar]
- Shen, Y.C.; Prakash, C.V.; Chen, Y.J.; Hwang, J.F.; Kuo, Y.H.; Chen, C.Y. Taxane diterpenoids from the stem bark of Taxus mairei. J. Nat. Prod. 2001, 64, 950–952. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Ge, F. Studies on constituents from Taxus mairei Bark. Zhong Yao Cai 2004, 27, 566–568. [Google Scholar] [PubMed]
- Cui, H.; Zhen, W.; Zhang, Z.; Fu, Y.; Li, X.; Fu, Y.; Gu, C. Determination and Analysis of Seven Taxanes in Different Taxus Species by HPLC Method. For. Eng. 2022, 38, 118–124. (In Chinese) [Google Scholar]
- Liu, G.M.; Fang, W.S.; Qian, J.F.; Lan, H. Distribution of paclitaxel and its congeners in Taxus mairei. Fitoterapia 2001, 72, 743–746. [Google Scholar] [CrossRef] [PubMed]
- Ke, C.; Quan, C.; Wang, Y.; Huang, J.; Ni, J.; Yang, H. Taxol and 10-DAB contents of different prov enance Taxus chinensis var. marei and related affecting factors. In Proceedings of the 5th Symposium of Chinese Young Ecologists, Kaifeng, China, 16 May 2008. [Google Scholar]
- Li, C.; Ou, C.; Zhou, G. Analysis of paclitaxel, 10DABⅢ and 7-xylitaxel in various parts of Taxus chinensis. Chin. Med. Innov. 2012, 9, 157–159. (In Chinese) [Google Scholar]
- Shi, C.; Jiang, Y.; Ma, C.; Xie, X.; Li, F.; Zhang, Y. The medicinal properties and clinical application of Taxus chinensis in Traditional Chinese Medicine. Pharm. Clin. Chin. Mater. Med. 2018, 3. (In Chinese) [Google Scholar]
- Li, N.; Pan, Z.; Zhang, D.; Wang, H.X.; Yu, B.; Zhao, S.P.; Guo, J.J.; Wang, J.W.; Yao, L.; Cao, W.G. Chemical Components, Biological Activities, and Toxicological Evaluation of the Fruit (Aril) of Two Precious Plant Species from Genus Taxus. Chem. Biodivers. 2017, 14, e1700305. [Google Scholar] [CrossRef]
- Zhao, C.; Li, Z.; Li, C.; Yang, L.; Yao, L.; Fu, Y.; He, X.; Shi, K.; Lu, Z. Optimized extraction of polysaccharides from Taxus chinensis var. mairei fruits and its antitumor activity. Int. J. Biol. Macromol. 2015, 75, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Zuo, F.; Sheng, J.; Chang, Q.; Fan, J.; Zhang, X. Comparison of 1000-grain weight, viability and nutrient chemical content of Taxus chinensis seeds from different provenances and their relationship with ecological factors. Flor. Stud. 2016, 36, 8. (In Chinese) [Google Scholar]
- Huang, R.; Fan, G.X.; Guo, X.; Zhen, K.; Luo, L. Chemical composition analysis of Taxus chinensis seeds. J. Appl. Environ. Biol. 2002, 8, 3. (In Chinese) [Google Scholar]
- Shi, Q.; Oritani, T.; Sugiyama, T.; Murakami, R.; Wei, H. Six new taxane diterpenoids from the seeds of Taxus chinensis var. mairei and Taxus yunnanensis. J. Nat. Prod. 1999, 62, 1114–1118. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.W.; Oritani, T.; Sugiyama, T.; Zhao, D.; Murakami, R. Three new taxane diterpenoids from seeds of the Chinese yew, Taxus yunnanensis and T. chinensis var. mairei. Planta Med. 1999, 65, 767–770. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.C.; Chen, C.Y.; Hung, M.C. Taxane diterpenoids from seeds of Taxus mairei. Chem. Pharm. Bull. 2000, 48, 1344–1346. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.P.; Wang, C.L.; Gu, J.S.; Shi, Q.W. Studies on chemical constituents in seeds of Taxus mairei II. Zhongguo Zhong Yao Za Zhi 2005, 30, 1260–1263. [Google Scholar] [PubMed]
- Nadeem, M.; Rikhari, H.C.; Kumar, A.; Palni, L.M.; Nandi, S.K. Taxol content in the bark of Himalayan Yew in relation to tree age and sex. Phytochemistry 2002, 60, 627–631. [Google Scholar] [CrossRef]
- Bai, P.; Sun, Q.; Jiang, J.; Yan, H.; Yu, S.; Zhang, X. Comparative analyses of taxol content and fingerprint of branch and leaf of Taxus chinensis var. mairei from different locations. J. Plant Resour. Environ. 2012, 21, 6. (In Chinese) [Google Scholar]
- Wu, C.; Jiang, L.; Yang, Y.; Tang, Q.; Lu, Y.; Duan, C.; Qiu, D. Comparative, regression and cluster analysis on contents of six taxanes in Taxus spp. Chin. J. Chin. Mater. Medica 2021, 52, 6. (In Chinese) [Google Scholar]
- Zhu, L. Study on Chemical Constituents and Biological Activities of Endophytic Fungi of Bitter Bamboo and Taxus chinensis; Zhejiang A&F University: Hangzhou, China, 2014. [Google Scholar]
- Yang, F.; Pang, H.; Zu, Y.; Zhang, X.; Gao, Y. Growth and development of Taxus chinensis and the relationship between taxol content and environmental factors. Flor. Stud. 2010, 742–746. [Google Scholar]
- Wang, Z.; Zhou, R.; Liu, X.; Tong, Q.; Liu, P.; Wang, S. Dynamic change of taxol content in Taxis chinensis var. mairei in Hunan Province. Cent. South Pharm. 2010, 3, 15–17. (In Chinese) [Google Scholar]
- Dianjie, C.; Guoshen, Z.; Qiang, W.; Zhi, L.; Tiantian, W.; Shuaii, X.; Chenchen, H. Comparative study on taxol content in bark of Taxus chinensis with three different color fruits. Genom. Appl. Biol. 2018, 37, 6. (In Chinese) [Google Scholar]
- Guo, T.; Zhong, H.; Li, X.; Mu, J.; Liu, T.; Yu, N.; Zhao, Y.; Liang, X.J.; Guo, S. Drug content on anticancer efficacy of self-assembling ketal-linked dextran-paclitaxel conjugates. J. Control. Release 2023, 359, 175–187. [Google Scholar] [CrossRef]
- Kutne, J.; Shanlin, G. Contents of paclitaxel and brevis in seven Taxus plants from East Asia and North America. J. China Pharm. Univ. 1995, 26, 3. (In Chinese) [Google Scholar]
- Veselá, D.; Šaman, D., IV; Vanĕk, T. Seasonal variations in the content of taxanes in the bark of Taxus baccata L. Phytochem. Anal. 1999, 10, 319–321. [Google Scholar] [CrossRef]
- Wei, L.; Yang, L.; Wang, J.; Wang, W.; Zhao, Y. Determination of taxanes in taxus taxus from different regions by HPLC. In Proceedings of the 10th National Symposium on Medicinal Plants and Plant Drugs, Kunming, China, 10 August 2011. [Google Scholar]
- Van Rozendaal, E.L.; Lelyveld, G.P.; van Beek, T.A. Screening of the needles of different yew species and cultivars for paclitaxel and related taxoids. Phytochemistry 2000, 53, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lv, J.; Wang, L.; Shao, H. Comparison of Pharmacokinetics and Biodistribution of 10-Deacetylbaccatin III after Oral Administration as Pure Compound or in Taxus chinensis Extract: A Pilot Study. Planta Med. 2016, 82, 230–237. [Google Scholar] [CrossRef]
- Tao, J.; Yan, R.; Zhao, L.; Wang, D.; Xu, X. Separation and purification of two taxanes and one xylosyl-containing taxane from Taxus wallichiana Zucc.: A comparison between high-speed countercurrent chromatography and reversed-phase flash chromatography. J. Sep. Sci. 2017, 40, 1273–1282. [Google Scholar] [CrossRef]
- Jing, Z. Construction of Administrative Talents in Zhejiang Food and Drug Administration. Chin. Pharm. Aff. 2016. [Google Scholar]
- Wang, K.; Pei, J.; Xie, C.; Dai, F.; Sun, X. Taxus chinensis var. mairei (AETC) Inhibiting HER2 Positive Human Gastric Cancer Cells NCI–N87 Transplantation Tumor and Inducing Apoptosis. Chin. Arch. Tradit. Chin. Med. 2016, 34, 5. (In Chinese) [Google Scholar]
- Dai, S.; Liu, Y.; Zhao, F.; Wang, H.; Shao, T.; Xu, Z.; Shou, L.; Chen, S.; Zhang, G.C.; Shu, Q. Aqueous extract of Taxus chinensis var. mairei targeting CD47 enhanced antitumor effects in non-small cell lung cancer. Biomed. Pharmacother. 2022, 154, 113628. [Google Scholar] [CrossRef] [PubMed]
- Cui, Q.L.; Shao, M.; Shu, Q.J. Study on inhibitory effect of aqueous extract of Taxus chinensis var. mairei on growth of A549 lung cancer xenografts in nude mice and its mechanism. Zhongguo Zhong Yao Za Zhi 2013, 38, 3549–3553. [Google Scholar]
- Cui, Q.L.; Ye, P.; Shu, Q.J.; Shao, M. Study on Inhibitory Effect of Aqueous Extract of Taxus chinensis var. mairei Combined Erlotnib on A549 Xenograft in Nude Mice and Its Mechanism. Zhongguo Zhong Xi Yi Jie He Za Zhi 2015, 35, 572–577. [Google Scholar]
- Zhang, J.; Shu, Q.-J.; Gao, J.-L.; Zhang, L. Study on Inhibitory Effects of Taxus chinensis var. mairei Aqueous Extract on the Proliferation of Tumor Cells. Chin. J. Integr. Tradit. West. Med. 2013, 33, 0805–0809. (In Chinese) [Google Scholar]
- Zhang, G.; Dai, S.; Chen, Y.; Wang, H.; Chen, T.; Shu, Q.; Chen, S.; Shou, L.; Cai, X. Aqueous extract of Taxus chinensis var. mairei regulates the Hippo-YAP pathway and promotes apoptosis of non-small cell lung cancer via ATF3 in vivo and in vitro. Biomed. Pharmacother. 2021, 138, 111506. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Ding, S.; Luo, Q.; Wang, P.; Yang, X.; Wu, L.; Chen, Y.; Zheng, X.; Zhang, H.; Yuan, L.; et al. Taxus wallichiana var. chinensis (Pilg.) Florin Aqueous Extract Suppresses the Proliferation and Metastasis in Lung Carcinoma via JAK/STAT3 Signaling Pathway. Front. Pharmacol. 2021, 12, 736442. [Google Scholar] [CrossRef]
- Shu, Q.J.; Li, P.; Wang, B.B. Experimental study on apoptosis induced by aqueous extract of Taxus chinensis in human pulmonary carcinoma cell A549 and its molecular mechanisms. Zhongguo Zhong Xi Yi Jie He Za Zhi 2011, 31, 1243–1247. [Google Scholar]
- Bingyi, W. Study on Effective Components of Taxus chinensis; Suzhou University: Suzhou, China, 2013. [Google Scholar]
- Zhang, S.; Lu, X.; Zheng, T.; Guo, X.; Tang, Z. Investigation of bioactivities of Taxus chinensis, Taxus cuspidata, and Taxus × media by gas chromatography-mass spectrometry. Open Life Sci. 2021, 16, 287–296. [Google Scholar] [CrossRef]
- Yang, C.H.; Horwitz, S.B. Taxol(®): The First Microtubule Stabilizing Agent. Int. J. Mol. Sci. 2017, 18, 1733. [Google Scholar] [CrossRef]
- Kingston, D.G. Taxol: The chemistry and structure-activity relationships of a novel anticancer agent. Trends Biotechnol. 1994, 12, 222–227. [Google Scholar] [CrossRef]
- Guchelaar, H.J.; Ten Napel, C.H.; de Vries, E.G.; Mulder, N.H. Clinical, toxicological and pharmaceutical aspects of the antineoplastic drug taxol: A review. Clin. Oncol. R. Coll. Radiol. 1994, 6, 40–48. [Google Scholar] [CrossRef]
- Liebmann, J.E.; Cook, J.A.; Lipschultz, C.; Teague, D.; Fisher, J.; Mitchell, J.B. Cytotoxic studies of paclitaxel (Taxol) in human tumour cell lines. Br. J. Cancer 1993, 68, 1104–1109. [Google Scholar] [CrossRef]
- Weaver, B.A. How Taxol/paclitaxel kills cancer cells. Mol. Biol. Cell 2014, 25, 2677–2681. [Google Scholar] [CrossRef]
- Zhu, L.; Chen, L. Progress in research on paclitaxel and tumor immunotherapy. Cell. Mol. Biol. Lett. 2019, 24, 40. [Google Scholar] [CrossRef] [PubMed]
- Yan, C. Ertraction Technology Optimization and Content Detemination of Taxol in the Braches and Leaves of Taxus madia; Nanchang University: Nanchang, China, 2015. [Google Scholar]
- Joly, F.; Hilpert, F.; Okamoto, A.; Stuart, G.; Ochiai, K.; Friedlander, M. Fifth Ovarian Cancer Consensus Conference of the Gynecologic Cancer InterGroup: Recommendations on incorporating patient-reported outcomes in clinical trials in epithelial ovarian cancer. Eur. J. Cancer 2017, 78, 133–138. [Google Scholar] [CrossRef]
- Xu, X.; Jin, S.; Ma, Y.; Fan, Z.; Yan, Z.; Li, W.; Song, Q.; You, W.; Lyu, Z.; Song, Y.; et al. miR-30a-5p enhances paclitaxel sensitivity in non-small cell lung cancer through targeting BCL-2 expression. J. Mol. Med. 2017, 95, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Sun, C. Study on Dynamic Changes of Biomass and 10-DAB Content of Taxus media var CL; Sichuan Agricultural University: Yaan, China, 2017. [Google Scholar]
- Ullah, A.; Leong, S.W.; Wang, J.; Wu, Q.; Ghauri, M.A.; Sarwar, A.; Su, Q.; Zhang, Y. Cephalomannine inhibits hypoxia-induced cellular function via the suppression of APEX1/HIF-1α interaction in lung cancer. Cell Death Dis. 2021, 12, 490. [Google Scholar] [CrossRef] [PubMed]
- Helson, L. Cephalomannine and 10-deacetyltaxol cytotoxicity in human glial and neuroblastoma cell-lines. Int. J. Oncol. 1993, 2, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Hosoyama, H.; Shigemori, H.; Tomida, A.; Tsuruo, T.; Kobayashi, J. Modulation of multidrug resistance in tumor cells by taxinine derivatives. Bioorg. Med. Chem. Lett. 1999, 9, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, J.; Hirose, K.; Ando, M. An efficient conversion of taxinine to taxinine NN-1, an anticancer agent and a modulator of multidrug-resistant tumor cells. J. Nat. Prod. 2002, 65, 1786–1792. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Lee, Y.R.; Park, C.S.; Im, S.A.; Song, S.; Hong, J.T.; Whang, B.Y.; Kim, K.; Lee, C.K. Baccatin III, a precursor for the semisynthesis of paclitaxel, inhibits the accumulation and suppressive activity of myeloid-derived suppressor cells in tumor-bearing mice. Int. Immunopharmacol. 2014, 21, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Leong, C.N.; Tako, M.; Hanashiro, I.; Tamaki, H. Antioxidant flavonoid glycosides from the leaves of Ficus pumila L. Food Chem. 2008, 109, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.W.; Hu, J.J.; Fu, R.Q.; Liu, X.; Zhang, Y.H.; Li, J.; Liu, L.; Li, Y.N.; Deng, Q.; Luo, Q.S.; et al. Flavonoids inhibit cell proliferation and induce apoptosis and autophagy through downregulation of PI3Kγ mediated PI3K/AKT/mTOR/p70S6K/ULK signaling pathway in human breast cancer cells. Sci. Rep. 2018, 8, 11255. [Google Scholar] [CrossRef]
- Xie, Z.; Du, L.; Li, X.; Xiong, Y. New progress in the study of Taxus australis. Chin. Pharm. Ind. 2009, 18, 3–5. (In Chinese) [Google Scholar]
- Yan, S.; Jie, Y. Effects of Taxus Chinensis Polysaccharide on Proliferation and Apoptosis of Cervical Cancer Cells. Shenzhen J. Integr. Tradit. Chin. West. Med. 2021, 31, 3. (In Chinese) [Google Scholar]
- Cai, W.; Yu, H.; Luo, Y.; Liu, S. Study on the mechanism of Taxus chinensis polysaccharides enhancing paclitaxel on tumor inhibition in S180 tumor-bearing mice. Mod. Chin. Med. Res. Pract. 2021, 035, 38–42. (In Chinese) [Google Scholar]
- Cai, W.; Xia, M.; Xiong, Y. Study on Enhancing Efficacy and Reducing Toxicity of Total Flavonoids, Total Polysaccharides in Taxus mairei Compatibility with Taxol. Mod. Tradit. Chin. Med. Mater. Med. World Sci. Technol. 2015, 17, 556–562. [Google Scholar]
- Ranjitkar, S.; Zhang, D.; Sun, F.; Salman, S.; He, W.; Venkitanarayanan, K.; Tulman, E.R.; Tian, X. Cytotoxic effects on cancerous and non-cancerous cells of trans-cinnamaldehyde, carvacrol, and eugenol. Sci. Rep. 2021, 11, 16281. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Sun, R.; Zhao, Z.; Wang, Y. Auricularia polytricha polysaccharides induce cell cycle arrest and apoptosis in human lung cancer A549 cells. Int. J. Biol. Macromol. Struct. Funct. Interact. 2014, 68, 67–71. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, W.; Wang, Y.; Zhang, C. Clinical nursing of ovarian cancer treated by paclitaxel combined with cisplatin. Chin. Foreign Womens Health Res. 2020, 1. 53+55 (In Chinese) [Google Scholar]
- Ming, N. Clinical nursing observation of paclitaxel combined with cisplatin in treatment of ovarian cancer. Chin. J. Mod. Drug Appl. 2014, 8, 2. (In Chinese) [Google Scholar]
- Lida, F.; Shang, W.; Lizhi, J. Clinical effect of paclitaxel combined with cisplatin concurrent radiotherapy and chemotherapy in the treatment of advanced non-small cell lung cancer. Clin. Med. Res. Pract. 2020, 5, 2. (In Chinese) [Google Scholar]
- Chen, S.; Zhang, Z.; Zhang, J. Emodin enhances antitumor effect of paclitaxel on human non-small-cell lung cancer cells in vitro and in vivo. Drug Des. Devel. Ther. 2019, 13, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Cao, J. Research progress of water extracts of Taxus australis in tumor. Zhejiang Clin. Med. 2019, 21, 1729–1731. (In Chinese) [Google Scholar]
- Shang, S.; Shu, Q. Effects of Taxus chinensis aqueous extract and erlotinib on expression of COX-2 and MMP-2 in human lung cancer A549 cells. J. Xinjiang Med. Univ. 2013, 36, 789–792. (In Chinese) [Google Scholar] [CrossRef]
- Yang, W.X.; Zhao, Z.G.; Wang, L.H.; Yu, S.J.; Liang, Z.S. Control of hypertension in rats using volatile components of leaves of Taxus chinensis var. mairei. J. Ethnopharmacol. 2012, 141, 309–313. [Google Scholar] [CrossRef]
- Kumari, N.; Anand, S.; Shah, K.; Chauhan, N.S.; Sethiya, N.K.; Singhal, M. Emerging Role of Plant-Based Bioactive Compounds as Therapeutics in Parkinson’s Disease. Molecules 2023, 28, 7588. [Google Scholar] [CrossRef]
- Prasher, P.; Sharma, M.; Chellappan, D.K.; Gupta, G.; Jha, N.K.; Singh, S.K.; MacLoughlin, R.; Terezinha, J.A.P.; Löbenberg, R.; Dua, K. Advanced drug delivery systems targeting NF-κB in respiratory diseases. Future Med. Chem. 2021, 13, 1087–1090. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zhang, F.; Zhou, C.; Chen, M.; Yang, C. Effects of ethyl acetate extract of Taxus chinensis fruit on phenotype and inflammatory cytokines in rats with depression. World Chin. Med. 2023, 1–5. (In Chinese) [Google Scholar]
- Zhang, S.; Li, L.; Hu, J.; Ma, P.; Zhu, H. Polysaccharide of Taxus chinensis var. mairei Cheng et L.K.Fu attenuates neurotoxicity and cognitive dysfunction in mice with Alzheimer’s disease. Pharm. Biol. 2020, 58, 959–968. [Google Scholar] [CrossRef]
- Tong, L.; Jiang, Y.; Guo, W.; Zhu, J. Experimental study on analgesic effect of stems and leaves of Taxus chinensis. Zhejiang Clin. Med. J. 2008, 10, 439–440. (In Chinese) [Google Scholar]
- Lou, P.; Xia, A.; Ying, Y. Experimental study on analgesic and anti-inflammatory effects of stem and leaf extracts of Taxus australis. Zhejiang J. Tradit. Chin. Med. 2015, 2, 556–567. (In Chinese) [Google Scholar]
- Wang, N.; Cai, T.; Tong, Y.; Liu, X.; Zhu, W.; Jiang, S.; Zhao, G. Study on the selection of anti-airway inflammation active ingredients in Taxus chinensis by HTRF method based on NF-κB signaling pathway. Zhejiang J. Tradit. Chin. Med. 2023, 58, 553–556. (In Chinese) [Google Scholar]
- Gupta, M.B.; Bhalla, T.N.; Gupta, G.P.; Mitra, C.R.; Bhargava, K.P. Anti-inflammatory activity of taxifolin. Jpn. J. Pharmacol. 1971, 21, 377–382. [Google Scholar] [CrossRef]
- Li, W.; Zhang, L.; Xu, Q.; Yang, W.; Zhao, J.; Ren, Y.; Yu, Z.; Ma, L. Taxifolin Alleviates DSS-Induced Ulcerative Colitis by Acting on Gut Microbiome to Produce Butyric Acid. Nutrients 2022, 14, 1069. [Google Scholar] [CrossRef]
- Zhang, Z.; Sun, T.; Niu, J.G.; He, Z.Q.; Liu, Y.; Wang, F. Amentoflavone protects hippocampal neurons: Anti-inflammatory, antioxidative, and antiapoptotic effects. Neural Regen. Res. 2015, 10, 1125–1133. [Google Scholar] [CrossRef]
- Nie, Y.; Zhang, D.; Qian, F.; Wu, Y. Baccatin III ameliorates bleomycin-induced pulmonary fibrosis via suppression of TGF-β1 production and TGF-β1-induced fibroblast differentiation. Int. Immunopharmacol. 2019, 74, 105696. [Google Scholar] [CrossRef]
- Wei, Q.; Li, Q.Z.; Wang, R.L. Flavonoid Components, Distribution, and Biological Activities in Taxus: A review. Molecules 2023, 28, 1713. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.J.; Sung, W.S.; Yeo, S.H.; Kim, H.S.; Lee, I.S.; Woo, E.R.; Lee, D.G. Antifungal effect of amentoflavone derived from Selaginella tamariscina. Arch. Pharm. Res. 2006, 29, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Hwang, I.S.; Lee, J.; Jin, H.G.; Woo, E.R.; Lee, D.G. Amentoflavone stimulates mitochondrial dysfunction and induces apoptotic cell death in Candida albicans. Mycopathologia 2012, 173, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.S.; Zhao, L.P.; Huang, Z.H.; Chen, X.Y.; Xu, J.T.; Tai, W.C.; Tsim, K.W.K.; Chen, Y.T.; Xie, T. Ginkgetin derived from Ginkgo biloba leaves enhances the therapeutic effect of cisplatin via ferroptosis-mediated disruption of the Nrf2/HO-1 axis in EGFR wild-type non-small-cell lung cancer. Phytomedicine 2021, 80, 153370. [Google Scholar] [CrossRef]
- Matsuse, I.T.; Lim, Y.A.; Hattori, M.; Correa, M.; Gupta, M.P. A search for anti-viral properties in Panamanian medicinal plants: The effects on HIV and its essential enzymes. J. Ethnopharmacol. 1998, 64, 15–22. [Google Scholar] [CrossRef]
- Wilson, C.R.; Sauer, J.M.; Hooser, S.B. Taxines: A review of the mechanism and toxicity of yew (Taxus spp.) alkaloids. Toxicon 2001, 39, 175–185. [Google Scholar] [CrossRef]
- Arens, A.M.; Anaebere, T.C.; Horng, H.; Olson, K. Fatal Taxus baccata ingestion with perimortem serum taxine B quantification. Clin. Toxicol. 2016, 54, 878–880. [Google Scholar] [CrossRef]
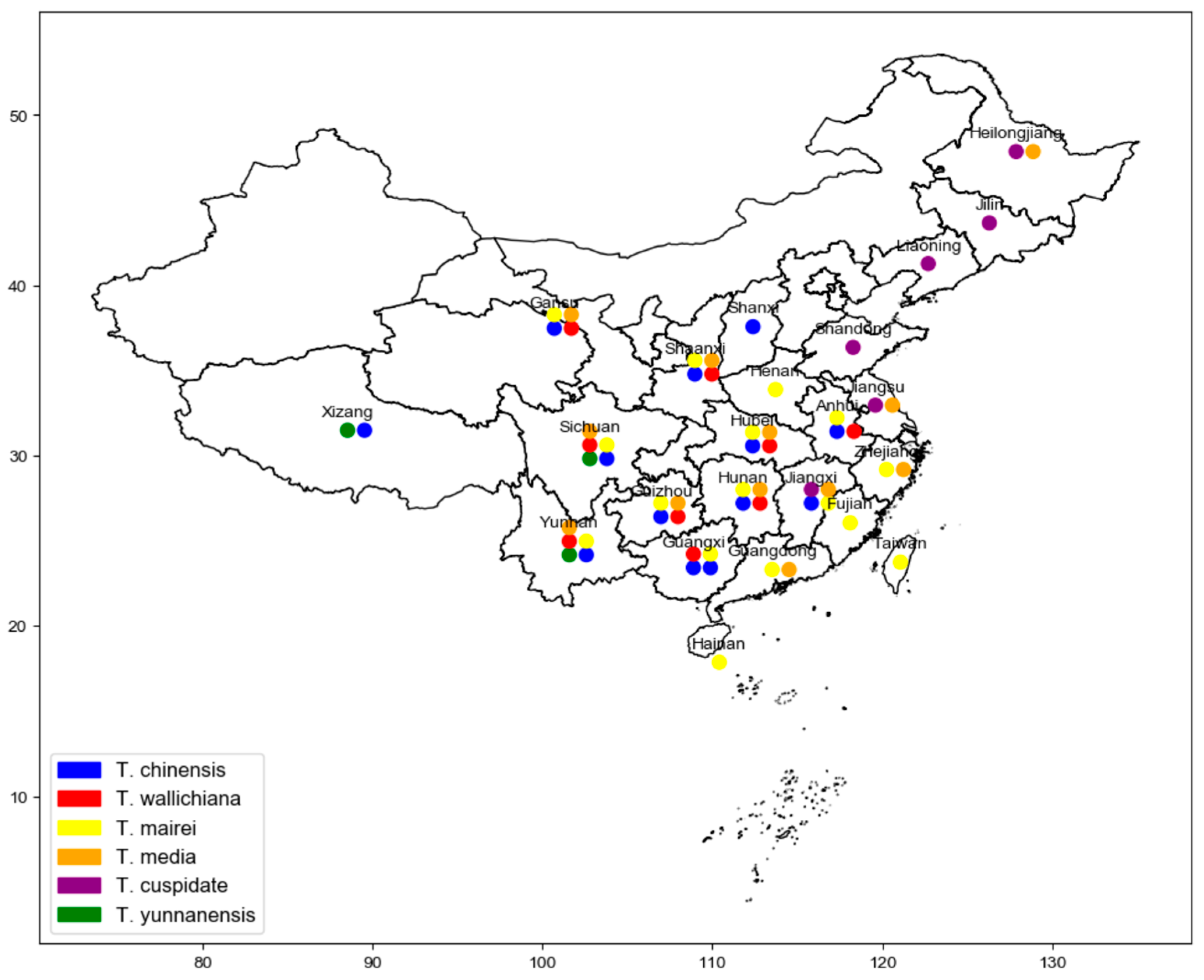
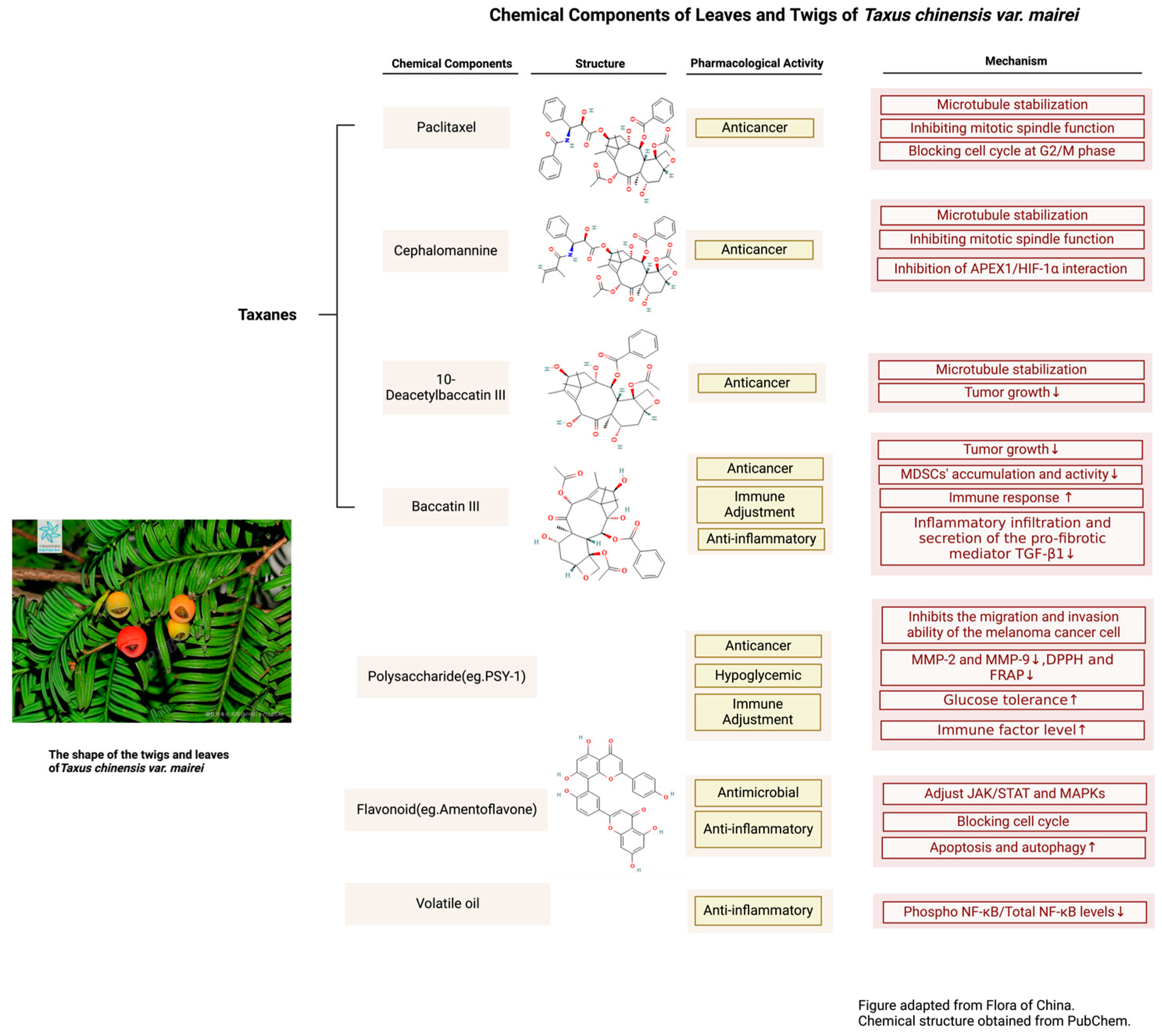
| Species | Paclitaxel Content (mg/g) | 10-DAB Content (mg/g) | Baccatin III Content (mg/g) | Cephalomannine Content (mg/g) |
|---|---|---|---|---|
| T. wallichiana | 0.10–0.30 [20,63] | 0.71 [20] | 0.19 [20] | 0.18 [20] |
| T.yunnanensis | 0.52–1.00 [20,64] | 0.700–0.821 [20,65] | 0.83 [20] | 0.01–0.12 [20,66] |
| T. mairei | 0.11–0.15 [20,21,22] | 0.74 [20] | 0.01–0.44 [20,21] | 0.23 [20] |
| T. cuspidata | 0.14–1.67 [20,21] | 0.77 [20] | 0.16–0.77 [20,21] | 0.25–0.84 [20,66] |
| T. media | 0.60–1.20 [20,58,64,67] | 0.34–0.75 [20,58] | 0.29–0.30 [20,67] | 0.53–0.60 [20,58] |
| T. chinensis | 0.60 [68] | 0.70–1.20 [68,69] | 0.21–0.35 [68,69] | 0.40 [68] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, X.; Zhang, N.; Xie, W. Advancements in the Cultivation, Active Components, and Pharmacological Activities of Taxus mairei. Molecules 2024, 29, 1128. https://doi.org/10.3390/molecules29051128
Gao X, Zhang N, Xie W. Advancements in the Cultivation, Active Components, and Pharmacological Activities of Taxus mairei. Molecules. 2024; 29(5):1128. https://doi.org/10.3390/molecules29051128
Chicago/Turabian StyleGao, Xinyu, Ni Zhang, and Weidong Xie. 2024. "Advancements in the Cultivation, Active Components, and Pharmacological Activities of Taxus mairei" Molecules 29, no. 5: 1128. https://doi.org/10.3390/molecules29051128
APA StyleGao, X., Zhang, N., & Xie, W. (2024). Advancements in the Cultivation, Active Components, and Pharmacological Activities of Taxus mairei. Molecules, 29(5), 1128. https://doi.org/10.3390/molecules29051128






