Synthesis and Biological Activity of New Hydrazones Based on N-Aminomorpholine
Abstract
1. Introduction
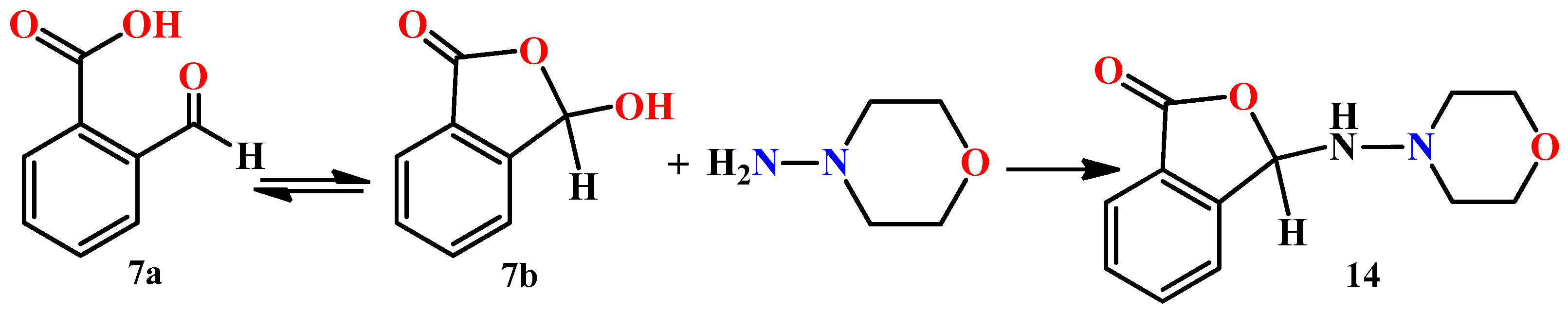
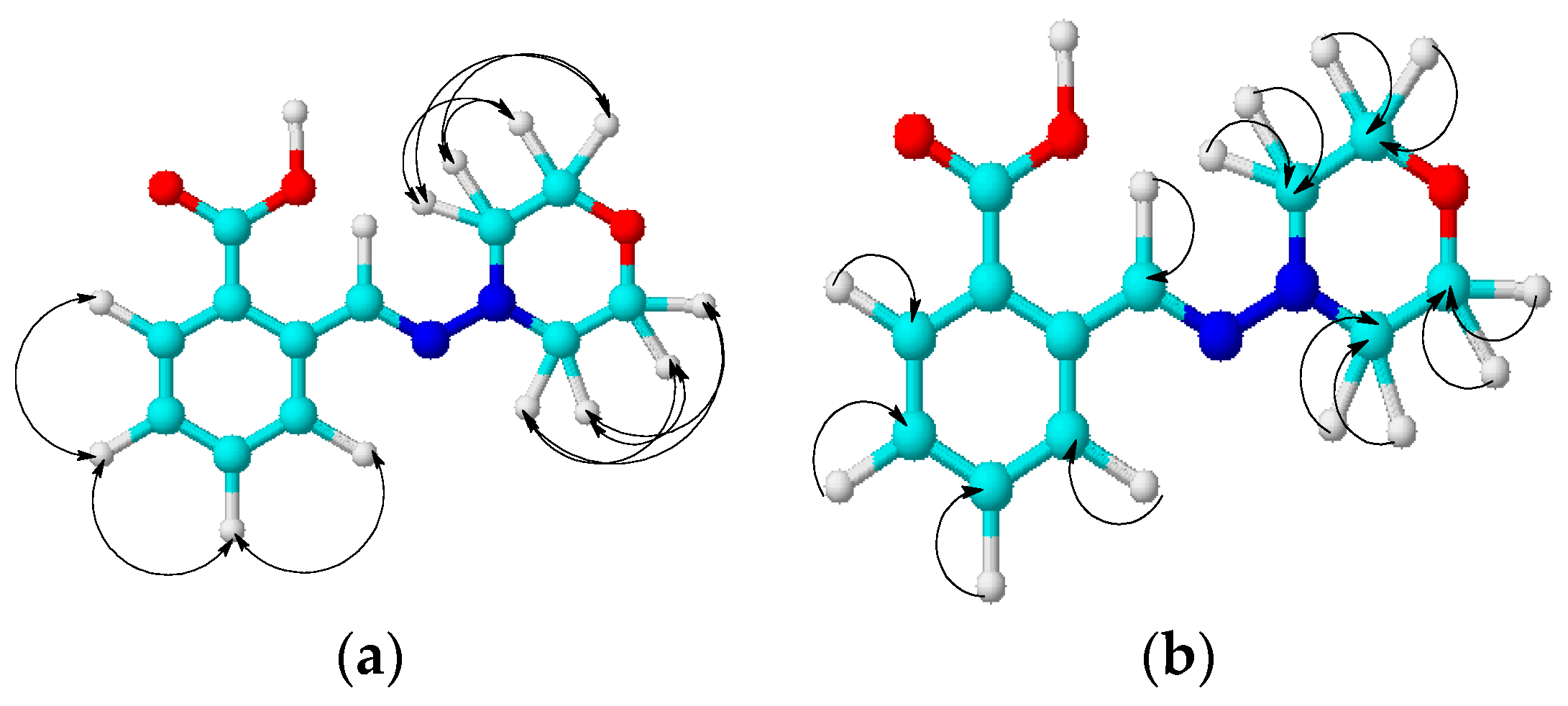
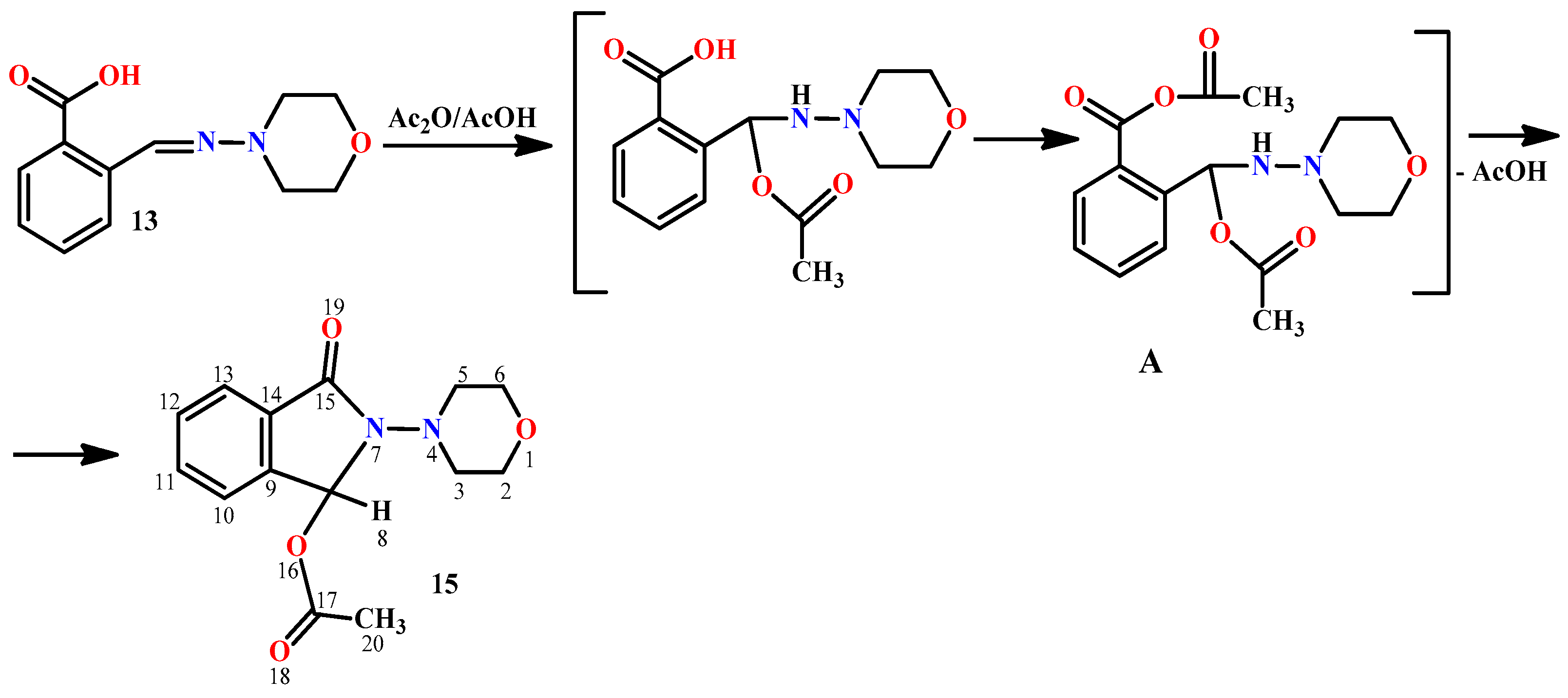
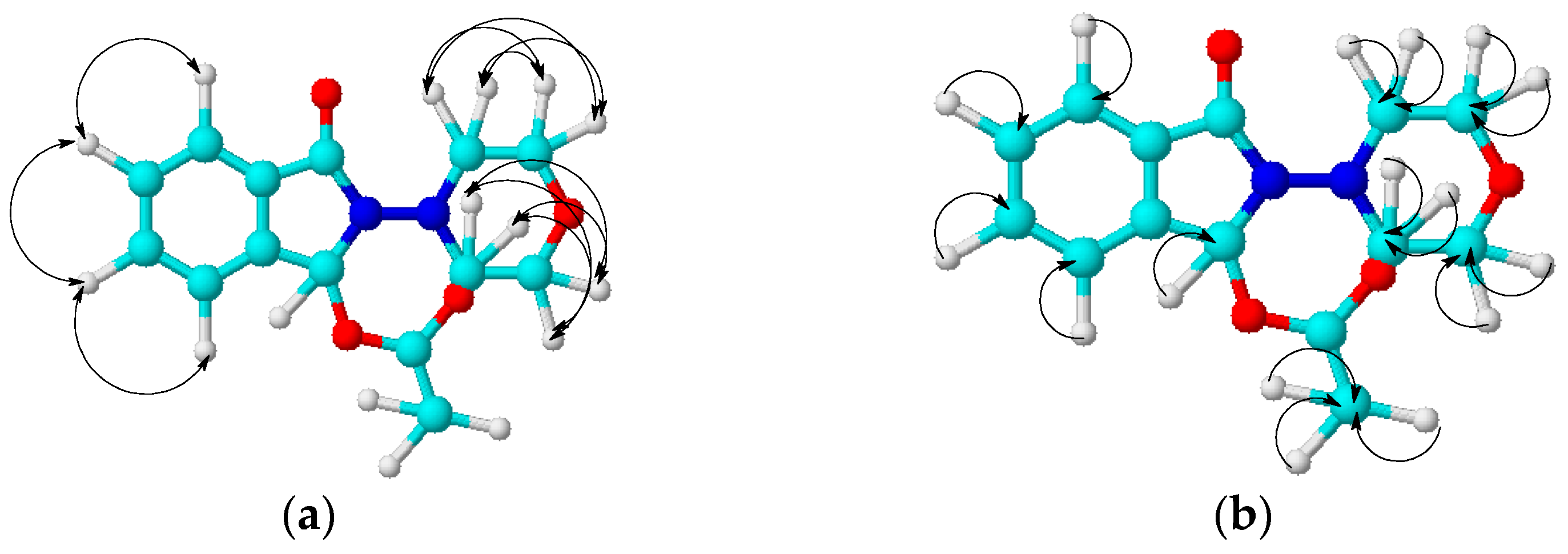
2. Results and Discussion
3. Materials and Methods
3.1. Synthesis of Hydrazones 8–13
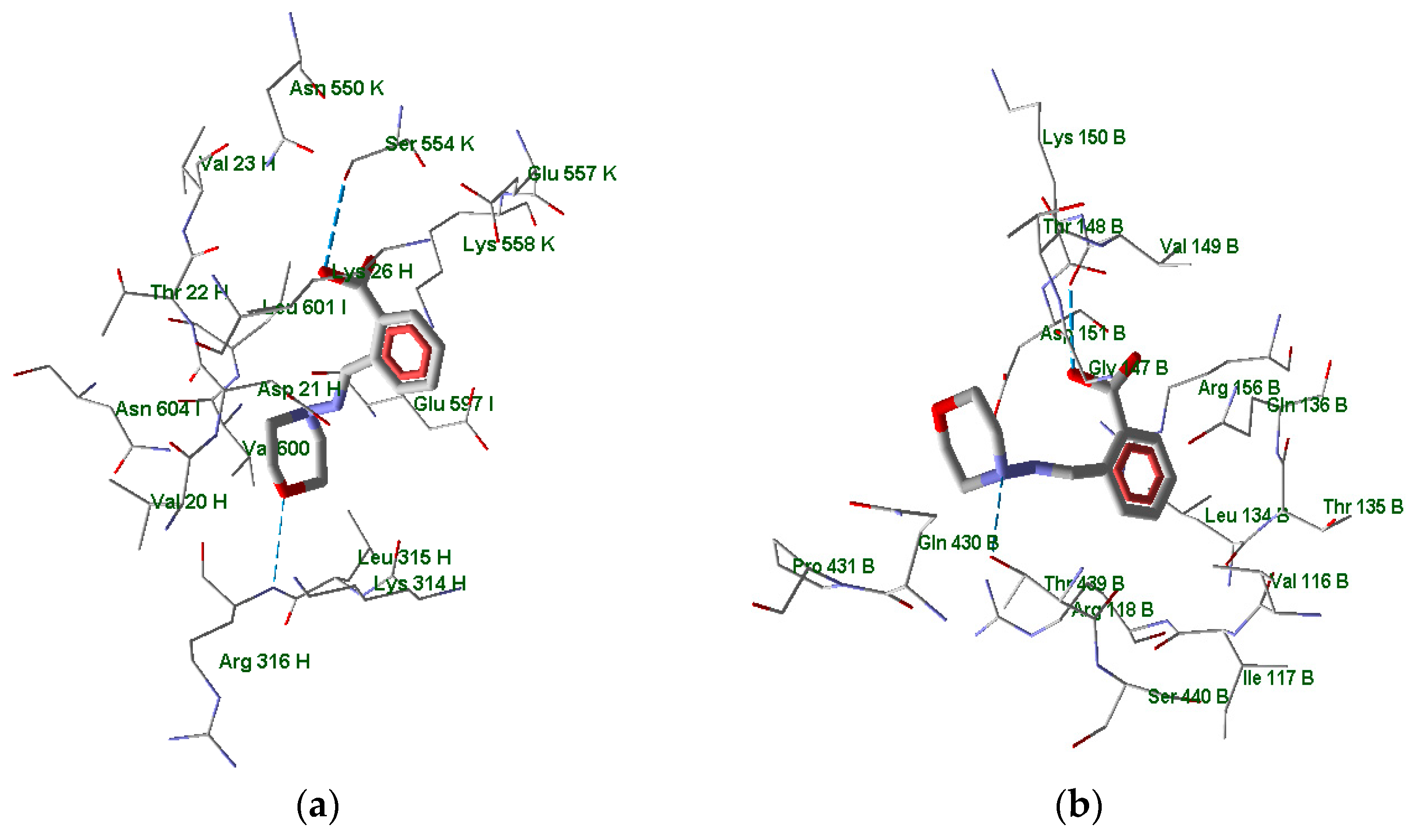
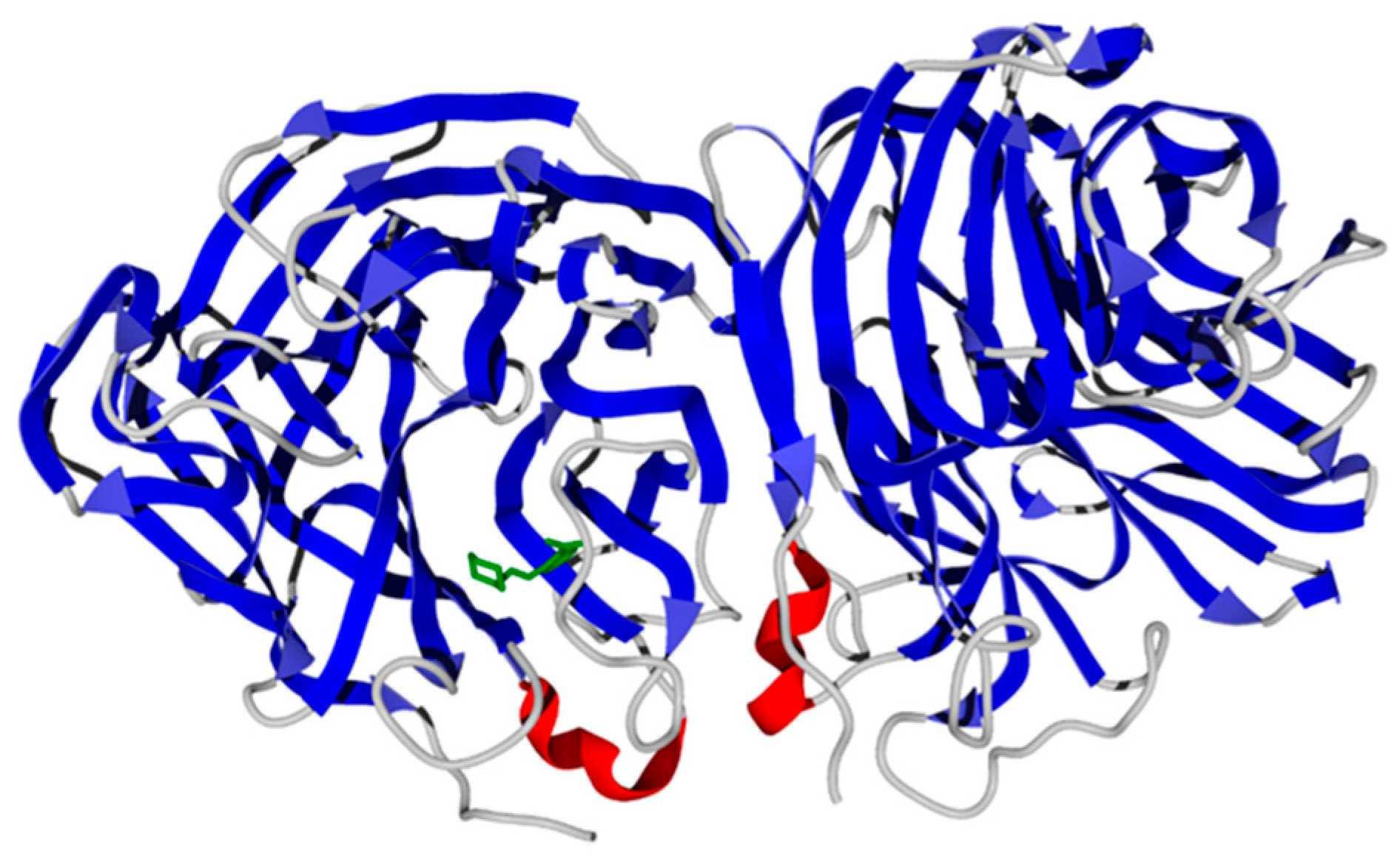
3.2. Molecular Docking
3.3. Research of Biological Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, G.Y.; Shi, X.; Phan, H. Efficient self-assembly of heterometallic triangular necklace with strong antibacterial activity. Nat. Commun. 2020, 11, 3178. [Google Scholar] [CrossRef] [PubMed]
- Khamitova, A.E.; Berillo, D.A. Overview of piperidine and morpholine derivatives as promising sources of biologically active compounds. Drug Dev. Regist. 2023, 12, 44–54. [Google Scholar] [CrossRef]
- De la Torre, B.G.; Albericio, F. The Pharmaceutical Industry in 2021. An Analysis of FDA Drug Approvals from the Perspective of Molecules. Molecules 2022, 27, 1075. [Google Scholar] [CrossRef] [PubMed]
- Kourounakis, A.P.; Xanthopoulos, D.; Tzara, A. Morpholine as a privileged structure: A review on the medicinal chemistry and pharmacological activity of morpholine containing bioactive molecules. Med. Res. Rev. 2020, 40, 709–752. [Google Scholar] [CrossRef]
- Verma, G.; Marella, A.; Shaquiquzzaman, M.; Akhtar, M.; Ali, M.R.; Alam, M.M. A review exploring biological activities of hydrazones. J. Pharm. Bioallied Sci. 2014, 6, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Siddiqui, S.P.; Tarannum, N.A. Systematic review on the synthesis and biological activity of hydrazide derivatives. Hygeia J. Drug Med. 2017, 9, 61–79. [Google Scholar] [CrossRef]
- Mali, S.N.; Thorat, B.R.; Gupta, D.R.; Pandey, A. Mini-review of the importance of hydrazides and their derivatives. Synth. Biol. Act. Eng. Proc. 2021, 11, 21. [Google Scholar] [CrossRef]
- Nurkenov, O.A.; Satpaeva, Z.B.; Shchepetkin, I.A.; Fazylov, S.D.; Seilkhanov, T.M.; Akhmetova, S.B. Synthesis of new hydrazones based on o- and p-hydroxybenzohydrazides. Russ. J. Gen. Chem. 2017, 87, 1707–1710. [Google Scholar] [CrossRef]
- Popiolek, L.; Biernasiuk, A. Synthesis and investigation of antimicrobial activities of nitrofurazone analogues containing hydrazidehydrazone moiety. Saudi Pharm. J. 2017, 25, 1097–1102. [Google Scholar] [CrossRef]
- Nurkenov, O.A.; Fazylov, S.D.; Satpaeva, Z.B.; Seilkhanov, T.M.; Turdybekov, D.M.; Mendibayeva, A.Z.; Akhmetova, S.B.; Shulgau, Z.T.; Alkhimova, L.E.; Kulakov, I.V. Synthesis, structure and biological activity of hydrazones derived from 2- and 4-hydroxybenzoic acid hydrazides. Chem. Data Collect. 2023, 48, 101089. [Google Scholar] [CrossRef]
- Rodrigues, F.A.R.; Oliveira, A.C.A.; Cavalcanti, B.C.; Pessoa, C.; Pinheiro, A.C.; de Souza, M.V.N. Biological evaluation of isoniazid derivatives as an anticancer class. Sci. Pharm. 2014, 82, 21–28. [Google Scholar] [CrossRef]
- Naveen Kumar, H.; Jummat, F.; Asmawi, M.Z. Synthesis and evaluation of isonicotinoyl hydrazone derivatives as antimycobacterial and anticancer agents. Med. Chem. Res. 2014, 23, 269–279. [Google Scholar] [CrossRef]
- Hu, Y.-Q.; Zhang, S.; Zhao, F.; Gao, C.; Feng, L.-S.; Lv, Z.-S.; Xu, Z.; Wu, X. Isoniazid derivatives and their anti-tubercular activity. Eur. J. Med. Chem. 2017, 133, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Turgunalieva, D.M.; Dilbaryan, D.S.; Vasilchenko, A.S.; Nurkenov, O.A.; Fazylov, S.D.; Karipova, G.Z.; Seilkhanov, T.M.; Kulakov, I.V. Synthesis and antibacterial activity of hydrazones of isonicotinic and salicylic acids based on acetyl derivatives of coumarin and benzo[g][1,3,5]oxadiazocine. Bulletin of the Karaganda university. Chem. Ser. 2022, 4, 25–34. [Google Scholar] [CrossRef]
- Nurkenov, O.A.; Fazylov, S.D.; Seilkhanov, T.M.; Abulyaissova, L.K.; Turdybekov, K.M.; Zhivotova, T.S.; Kabieva, S.K.; Mendibayeva, A.Z. Interaction of Isonicotinic Acid Hydrazide with Carboxylic Acid Anhydrides. Eurasian J. Chem. 2023, 2, 29–35. [Google Scholar] [CrossRef]
- Nurkenov, O.A.; Fazylov, S.D.; Gazaliev, A.M.; Satpaeva, Z.B.; Amerkhanova, S.K.; Karipova, G.Z. Synthesis and properties of isonicotinic acid hydrazide derivatives. Rep. Natl. Acad. Sci. Repub. Kazakhstan 2017, 1, 68–78. [Google Scholar]
- Ukhin, L.Y.; Suponitsky, K.Y.; Belousova, L.V.; Orlova, J.I. A new synthesis of phthalimidines. Russ. Chem. Bull. 2009, 12, 2399–2407. [Google Scholar] [CrossRef]
- Ukhin, L.Y.; Kuzmina, L.G.; Gribanova, T.N.; Belousova, L.V.; Orlova, J.I. Anthranilic acid hydrazide in the synthesis of condensed polycyclic compounds with quinazoline fragments. Russ. Chem. Bull. 2008, 11, 2294–2302. [Google Scholar] [CrossRef]
- Ukhin, L.Y.; Shepelenko, E.N.; Belousova, L.V.; Orlova, Z.I.; Borodkin, G.S.; Suponitsky, K.Y. Derivatives of 2-aminothiophene in the new synthesis of phthalimidines. Russ. Chem. Bull. 2011, 2, 345–352. [Google Scholar] [CrossRef]
- Ukhin, L.Y.; Krasnikov, V.V.; Zaichenko, S.B.; Borodkin, G.S.; Gribanova, T.N.; Shepelenko, E.N.; Etmetchenko, L.N. Reaction of derivatives of 2-aminopyrrolo with o-formyl benzoic acid. Russ. Chem. Bull. 2015, 2, 410–414. [Google Scholar] [CrossRef]
- Patel, D.; Kumari, P.; Patel, N. Synthesis of 3-{4-[4-dimethylamino-6-(4-methyl-2-oxo-2H-chromen-7-yloxy)-[1,3,5] triazin-2-ylamino]-phenyl}-2-phenyl-5-(4-pyridin-2-yl-piperazin-1-ylmethyl)-thiazolidin-4-one and their biological evaluation. Med. Chem. Res. 2012, 21, 2926–2944. [Google Scholar] [CrossRef]
- Nurkenov, O.A.; Fazylov, S.D.; Kulakov, I.V.; Seilkhanov, T.M.; Mendibayeva, A.Z.; Syzdykov, A.K.; Kabieva, S.K. Reaction of o-formylbenzoic acid with hydrazides of (iso)niconitic and hydroxybenzoic acids. Rus. J. Gen. Chem. 2023, 93, 1326–1334. [Google Scholar] [CrossRef]
- Allen, F.H.; Kennard, O.; Watson, D.G.; Brammer, L.; Orpen, A.G.; Taylor, R. Tables of bond lengths determined by X-ray and neutron diffraction. Part 1. Bond lengths in organic compounds. J. Chem. Soc. Perkin Trans. II 1987, 2, 1–19. [Google Scholar] [CrossRef]
- Mamatha, S.V.; Mahesh, B.; Sagar, B.K.; Meenakshi, S.K. Synthesis, characterization, crystal structure biological activity of 4-{2-[5-(4-fluoro-phenyl)-[1,3,4]oxadiazol-2-ylsulfanyl]-ethyl}-morpholine. J. Molec. Struc. 2019, 1196, 186–193. [Google Scholar] [CrossRef]
- Aiello, T.F.; García-Vidal, C.; Soriano, A. Antiviral drugs against SARS CoV-2. Rev. Esp. Quimioter. 2022, 35, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.K.P.; Hui, D.S.C. Antiviral therapies for influenza. Curr. Opin. Infect. Dis. 2023, 36, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Tian, S.; Yang, X.; Liu, Z. Synthesis methods of 1,2,3- and 1,2,4-triazoles: A Review. Front. Chem. 2022, 26, 891484. [Google Scholar] [CrossRef]
- De, S.; Aamna, B.; Sahu, R.; Parida, S.; Behera, S.K.; Dan, A.K. Seeking heterocyclic scaffolds as antivirals against dengue virus. Eur. J. Med. Chem. 2022, 240, 114576. [Google Scholar] [CrossRef] [PubMed]
- Dinodia, M. Recent Advances in N-Heterocycles for COVID-19 Treatment—A Mini Review. Med. Chem. 2023, 19, 717–729. [Google Scholar] [CrossRef]
- Kaushik, S.; Paliwal, S.K.; Iyer, M.R.; Patil, V.M. Promising Schiff bases in antiviral drug design and discovery. Med. Chem. Res. 2023, 32, 1063–1076. [Google Scholar] [CrossRef]
- Moskalik, M.Y. Sulfonamides with heterocyclic periphery as antiviral agents. Molecules 2022, 28, 51. [Google Scholar] [CrossRef] [PubMed]
- Nikitina, P.A.; Zakharova, A.M.; Serova, O.A.; Bormotov, N.I.; Mazurkov, O.Y.; Shishkina, L.N.; Koldaeva, T.Y.; Basanova, E.I.; Perevalov, V.P. Synthesis, cytotoxicity and antiviral activity against vaccinia virus of 2-(3-Coumarinyl)-1-Hydroxyimidazoles. Med. Chem. 2023, 19, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Utreja, D. Synthesis and antiviral activity of diverse heterocyclic scaffolds. Chem. Biol. Drug Des. 2022, 100, 870–920. [Google Scholar] [CrossRef] [PubMed]
- Tratrat, C.; Petrou, A.; Fesatidou, M.; Haroun, M.; Athina, G.; Venugopala, K.; Sreeharsha, N.; Chemali, J. 5-Membered heterocyclic compounds as antiviral agents. Curr. Top. Med. Chem. 2023, 23, 520–538. [Google Scholar] [CrossRef] [PubMed]
- Tret’yakova, E.V.; Ma, X.; Kazakova, O.B.; Shtro, A.A.; Petukhova, G.D.; Klabukov, A.M.; Dyatlov, D.S.; Smirnova, A.A.; Xu, H.; Xiao, S. Synthesis and evaluation of diterpenic Mannich bases as antiviral agents against influenza A and SARS-CoV-2. Phytochem. Lett. 2022, 51, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Gamblin, S.J.; Haire, L.F.; Russell, R.J.; Stevens, D.J.; Xiao, B.; Ha, Y.; Vasisht, N.; Steinhauer, D.A.; Daniels, R.S.; Elliot, A.; et al. The structure and receptor binding properties of the 1918 influenza hemagglutinin. Science 2004, 303, 1838–1842. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhu, X.; Dwek, R.A.; Stevens, J.; Wilson, I.A. Structural characterization of the 1918 influenza virus H1N1 neuraminidase. J. Virol. 2008, 82, 10493–10501. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.; Olson, A.J.; Goodsell, D.S. Automated prediction of ligand-binding sites in proteins. Proteins 2008, 70, 1506–1517. [Google Scholar] [CrossRef]
- SMART V5.051 and SAINT V5.00, Area Detector Control and Integration Software; Bruker AXS Inc.: Madison, WI, USA, 1998.
- Sheldrick, G.M. SADABS; Bruker AXS Inc.: Madison, WI, USA, 2008. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Valdes-Tresanco, M.S.; Valdes-Tresanco, M.E.; Valiente, P.A.; Moreno, E. AMDock: A versatile graphical tool for assisting molecular docking with Autodock Vina and Autodock4. Biol. Direct 2020, 15, 12. [Google Scholar] [CrossRef] [PubMed]


 | |
| Compound | R |
| 2, 8 | 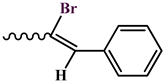 |
| 3, 9 | 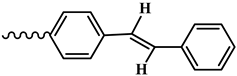 |
| 4, 10 |  |
| 5, 11 | 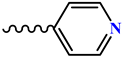 |
| 6, 12 | 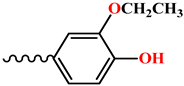 |
| 7, 13 |  |
| Compound | Chemical Therapeutic Index | |
|---|---|---|
| A/Almaty/8/98 (H3N2) | A/Vladivostok/2/09 (H1N1) | |
| 13 | 25.0 | 20.0 |
| 15 | <1.0 | <1.0 |
| Tamiflu | 29.9 | 30 |
| Remantadine | 10.3 | 11 |
| Compound | Staphylococcus aureus ATCC 6538 | Bacillus subtilis ATCC 6633 | Escherichia coli ATCC 25922 | Pseudomonas aeruginosa ATCC 27853 | Candida albicans ATCC 10231 |
|---|---|---|---|---|---|
| 2-((morpholino-amino) methyl)-benzoic acid (13) | 18 ± 0.1 | 15 ± 0.1 * | 21 ± 0.1 * | - | 14 ± 0.1 |
| Benzylpenicillin sodium salt | 16 ± 0.1 | 14 ± 0.1 | 15 ± 0.1 | - | - |
| Gentamicin | 24 ± 0.1 | 21 ± 0.2 | 26 ± 0.1 | 27 ± 0.1 | - |
| Nystatin | - | - | - | 21 ± 0.2 |
| Compound | MSC (mcg/mL) | ||||
|---|---|---|---|---|---|
| Staphylococcus aureus ATCC 6538 | Bacillus subtilis ATCC 6633 | Escherichia coli ATCC 25922 | Pseudomonas aeruginosa ATCC 27853 | Candida albicans ATCC 10231 | |
| 2-((morpholino-amino) methyl)-benzoic acid (13) | 12.5 | 25 | 6.3 | - | 50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nurkenov, O.A.; Zhautikova, S.B.; Khlebnikov, A.I.; Syzdykov, A.K.; Fazylov, S.D.; Seilkhanov, T.M.; Kabieva, S.K.; Turdybekov, K.M.; Mendibayeva, A.Z.; Zhumanazarova, G.M. Synthesis and Biological Activity of New Hydrazones Based on N-Aminomorpholine. Molecules 2024, 29, 3606. https://doi.org/10.3390/molecules29153606
Nurkenov OA, Zhautikova SB, Khlebnikov AI, Syzdykov AK, Fazylov SD, Seilkhanov TM, Kabieva SK, Turdybekov KM, Mendibayeva AZ, Zhumanazarova GM. Synthesis and Biological Activity of New Hydrazones Based on N-Aminomorpholine. Molecules. 2024; 29(15):3606. https://doi.org/10.3390/molecules29153606
Chicago/Turabian StyleNurkenov, Oralgazy A., Saule B. Zhautikova, Andrei I. Khlebnikov, Ardak K. Syzdykov, Serik D. Fazylov, Tulegen M. Seilkhanov, Saule K. Kabieva, Kobylandy M. Turdybekov, Anel Z. Mendibayeva, and Gaziza M. Zhumanazarova. 2024. "Synthesis and Biological Activity of New Hydrazones Based on N-Aminomorpholine" Molecules 29, no. 15: 3606. https://doi.org/10.3390/molecules29153606
APA StyleNurkenov, O. A., Zhautikova, S. B., Khlebnikov, A. I., Syzdykov, A. K., Fazylov, S. D., Seilkhanov, T. M., Kabieva, S. K., Turdybekov, K. M., Mendibayeva, A. Z., & Zhumanazarova, G. M. (2024). Synthesis and Biological Activity of New Hydrazones Based on N-Aminomorpholine. Molecules, 29(15), 3606. https://doi.org/10.3390/molecules29153606







