Autohydrolysis Application on Vine Shoots and Grape Stalks to Obtain Extracts Enriched in Xylo-Oligosaccharides and Phenolic Compounds
Abstract
1. Introduction
2. Results and Discussion
2.1. Qualitative Analysis of Extracts
2.2. Quantitative Analysis of Extracts
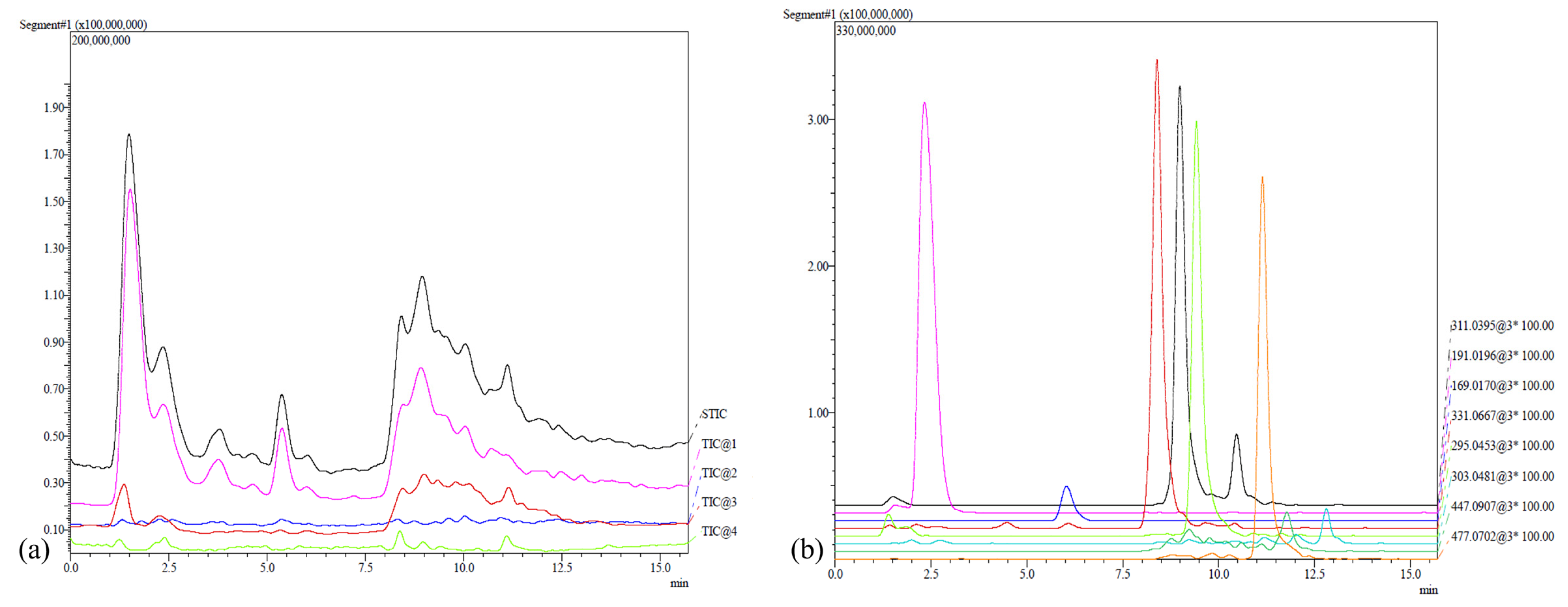
2.3. Phenolic Compounds Characterization
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Vine Shoots and Grape Stalks Preparation
3.3. Hydrothermal Treatments of Vine Shoots and Grape Stalks
3.4. Characterization of the Autohydrolysis Liquors
3.5. Determination and Quantification of Phenolic Profile
3.6. Antioxidant Activity Evaluation
3.7. Antioxidant Activity Evaluation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Vázquez, M.J.; Alonso, J.L.; Domínguez, H.; Parajó, J.C. Xylooligosaccharides: Manufacture and applications. Trends Food Sci. Technol. 2000, 11, 387–393. [Google Scholar] [CrossRef]
- Carvalho, A.F.A.; de Oliva Neto, P.; da Silva, D.F.; Pastore, G.M. Xylo-Oligosaccharides from lignocellulosic materials: Chemical structure, health benefits and production by chemical and enzymatic hydrolysis. Food Res. Int. 2013, 51, 75–85. [Google Scholar] [CrossRef]
- Santibáñez, L.; Henríquez, C.; Corro-Tejeda, R.; Bernal, S.; Armijo, B.; Salazar, O. Xylooligosaccharides from lignocellulosic biomass: A comprehensive review. Carbohydr. Polym. 2021, 251, 117118. [Google Scholar] [CrossRef]
- Capetti, C.C.D.M.; Vacilotto, M.M.; Dabul, A.N.G.; Sepulchro, A.G.V.; Pellegrini, V.O.A.; Polikarpov, I. Recent advances in the enzymatic production and applications of xylooligosaccharides. World J. Microbiol. Biotechnol. 2021, 37, 169. [Google Scholar] [CrossRef]
- de Freitas, C.; Carmona, E.; Brienzo, M. Xylooligosaccharides production process from lignocellulosic biomass and bioactive effects. Bioact. Carbohydr. Diet. Fibre 2019, 18, 100184. [Google Scholar] [CrossRef]
- Palaniappan, A.; Antony, U.; Emmambux, M.N. Current Status of Xylooligosaccharides: Production, characterization, health benefits and food application. Trends Food Sci. Technol. 2021, 111, 506–519. [Google Scholar] [CrossRef]
- Rashid, R.; Sohail, M. Xylanolytic bacillus species for xylooligosaccharides production: A critical review. Bioresour. Bioprocess. 2021, 8, 16. [Google Scholar] [CrossRef]
- Moure, A.; Gullón, P.; Domínguez, H.; Parajó, J.C. Advances in the manufacture, purification and applications of xylo-oligosaccharides as food additives and nutraceuticals. Process Biochem. 2006, 41, 1913–1923. [Google Scholar] [CrossRef]
- Global Xylo-Oligosaccharides Market Report. Available online: https://www.themarketreports.com/report/global-xylooligosaccharides-xos-market-research-report (accessed on 15 March 2023).
- Lin, S.H.; Chou, L.M.; Chien, Y.W.; Chang, J.S.; Lin, C.I. Prebiotic effects of xylooligosaccharides on the improvement of microbiota balance in human subjects. Gastroenterol. Res. Pract. 2016, 2016, 5789232. [Google Scholar] [CrossRef]
- Aragon, C.C.; Santos, A.F.; Ruiz-Matute, A.I.; Corzo, N.; Guisan, J.M.; Monti, R.; Mateo, C. Continuous production of xylooligosaccharides in a packed bed reactor with immobilized-stabilized biocatalysts of xylanase from aspergillus versicolor. J. Mol. Catal. B Enzym. 2013, 98, 8–14. [Google Scholar] [CrossRef]
- Mano, M.C.R.; Neri-Numa, I.A.; da Silva, J.B.; Paulino, B.N.; Pessoa, M.G.; Pastore, G.M. Oligosaccharide biotechnology: An approach of prebiotic revolution on the industry. Appl. Microbiol. Biotechnol. 2018, 102, 17–37. [Google Scholar] [CrossRef] [PubMed]
- Ayyappan, P.; Abirami, A.; Anbuvahini, N.A.; Tamil Kumaran, P.S.; Naresh, M.; Malathi, D.; Antony, U. Physicochemical properties of cookies enriched with xylooligosaccharides. Food Sci. Technol. Int. 2016, 22, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Ferrão, L.L.; Ferreira, M.V.S.; Cavalcanti, R.N.; Carvalho, A.F.A.; Pimentel, T.C.; Silva, R.; Esmerino, E.A.; Neto, R.P.C.; Tavares, M.I.B.; Freitas, M.Q.; et al. The Xylooligosaccharide addition and sodium reduction in requeijão cremoso processed cheese. Food Res. Int. 2018, 107, 137–147. [Google Scholar] [CrossRef]
- Zhang, L.; Zeng, X.; Fu, N.; Tang, X.; Sun, Y.; Lin, L. Maltodextrin: A consummate carrier for spray-drying of xylooligosaccharides. Food Res. Int. 2018, 106, 383–393. [Google Scholar] [CrossRef]
- Samanta, A.K.; Jayapal, N.; Jayaram, C.; Roy, S.; Kolte, A.P.; Senani, S.; Sridhar, M. Xylooligosaccharides as prebiotics from agricultural by-products: Production and applications. Bioact. Carbohydr. Diet. Fibre 2015, 5, 62–71. [Google Scholar] [CrossRef]
- Gullón, P.; González-Muñoz, M.J.; Parajó, J.C. Manufacture and prebiotic potential of oligosaccharides derived from industrial solid wastes. Bioresour. Technol. 2011, 102, 6112–6119. [Google Scholar] [CrossRef] [PubMed]
- Troilo, M.; Difonzo, G.; Paradiso, V.M.; Summo, C.; Caponio, F. Bioactive compounds from vine shoots, grape stalks, and wine lees: Their potential use in agro-food chains. Foods 2021, 10, 342. [Google Scholar] [CrossRef]
- Delgado-Torre, M.P.; Ferreiro-Vera, C.; Priego-Capote, F.; Pérez-Juan, P.M.; Luque De Castro, M.D. Comparison of accelerated methods for the extraction of phenolic compounds from different vine-shoot cultivars. J. Agric. Food Chem. 2012, 60, 3051–3060. [Google Scholar] [CrossRef]
- Dávila, I.; Gordobil, O.; Labidi, J.; Gullón, P. Assessment of suitability of vine shoots for hemicellulosic oligosaccharides production through aqueous processing. Bioresour. Technol. 2016, 211, 636–644. [Google Scholar] [CrossRef]
- Amorim, C.; Silvério, S.C.; Prather, K.L.J.; Rodrigues, L.R. From lignocellulosic residues to market: Production and commercial potential of xylooligosaccharides. Biotechnol. Adv. 2019, 37, 107397. [Google Scholar] [CrossRef]
- Cotana, F.; Buratti, C.; Barbanera, M.; Lascaro, E. Optimization of the steam explosion and enzymatic hydrolysis for sugars production from oak woods. Bioresour. Technol. 2015, 198, 470–477. [Google Scholar] [CrossRef]
- Tekin, K.; Hao, N.; Karagoz, S.; Ragauskas, A.J. Ethanol: A promising green solvent for the deconstruction of lignocellulose. ChemSusChem 2018, 11, 3559–3575. [Google Scholar] [CrossRef]
- Casiello, M.; Catucci, L.; Fracassi, F.; Fusco, C.; Laurenza, A.G.; Di Bitonto, L.; Pastore, C.; D’accolti, L.; Nacci, A. ZnO/Ionic liquid catalyzed biodiesel production from renewable and waste lipids as feedstocks. Catalysts 2019, 9, 71. [Google Scholar] [CrossRef]
- Laurenza, A.G.; Losito, O.; Casiello, M.; Fusco, C.; Nacci, A.; Pantone, V.; D’Accolti, L. Valorization of cigarette butts for synthesis of levulinic acid as top value-added chemicals. Sci. Rep. 2021, 11, 15775. [Google Scholar] [CrossRef]
- Amendola, D.; De Faveri, D.M.; Egües, I.; Serrano, L.; Labidi, J.; Spigno, G. Autohydrolysis and organosolv process for recovery of hemicelluloses, phenolic compounds and lignin from grape stalks. Bioresour. Technol. 2012, 107, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Gullón, B.; Eibes, G.; Dávila, I.; Vila, C.; Labidi, J.; Gullón, P. Valorization of vine shoots based on the autohydrolysis fractionation optimized by a kinetic approach. Ind. Eng. Chem. Res. 2017, 56, 14164–14171. [Google Scholar] [CrossRef]
- Jesus, M.S.; Romaní, A.; Genisheva, Z.; Teixeira, J.A.; Domingues, L. Integral valorization of vine pruning residue by sequential autohydrolysis stages. J. Clean. Prod. 2017, 168, 74–86. [Google Scholar] [CrossRef]
- De Leo, V.; Casiello, M.; Deluca, G.; Cotugno, P.; Catucci, L.; Nacci, A.; Fusco, C.; D’Accolti, L. Concerning snthesis of new biobased polycarbonates with curcumin in replacement of bisphenol a and recycled diphenyl carbonate as example of circular economy. Polymers 2021, 13, 361. [Google Scholar] [CrossRef] [PubMed]
- Sanz, M.L.; Sanz, J.; Martínez-Castro, I. Gas chromatographic-mass spectrometric method for the qualitative and quantitative determination of disaccharides and trisaccharides in honey. J. Chromatogr. A 2004, 1059, 143–148. [Google Scholar] [CrossRef]
- Sweeley, C.C.; Bentley, R.; Makita, M.; Well, W.W. Gas-liquid chromatography of trimethylsilyl derivatives of sugars and related substances. J. Am. Chem. Soc. 1963, 85, 2497–2507. [Google Scholar] [CrossRef]
- Taormina, C.R.; Baca, J.T.; Asher, S.A.; Grabowski, J.J.; Finegold, D.N. Analysis of tear glucose concentration with electrospray ionization mass spectrometry. J. Am. Soc. Mass Spectrom. 2007, 18, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Chen, H. Fractionation and characterization of lignin from steam-exploded corn stalk by sequential dissolution in ethanol-water solvent. Sep. Purif. Technol. 2013, 120, 402–409. [Google Scholar] [CrossRef]
- Hu, X.; Wu, L.; Wang, Y.; Song, Y.; Mourant, D.; Gunawan, R.; Gholizadeh, M.; Li, C.Z. Acid-catalyzed conversion of mono- and poly-sugars into platform chemicals: Effects of molecular structure of sugar substrate. Bioresour. Technol. 2013, 133, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Elango, D.; Rajendran, K.; Van der Laan, L.; Sebastiar, S.; Raigne, J.; Thaiparambil, N.A.; El Haddad, N.; Raja, B.; Wang, W.; Ferela, A.; et al. Raffinose family oligosaccharides: Friend or foe for human and plant health? Front. Plant Sci. 2022, 13, 829118. [Google Scholar] [CrossRef]
- Shimelis, E.A.; Kumar, R.S. Antinutritional factors and in vitro protein digestibility of improved haricot bean (Phaseolus vulgaris L.) varieties grown in ethiopia. Int. J. Food Sci. Nutr. 2005, 56, 377–387. [Google Scholar] [CrossRef]
- Banti, M.; Bajo, W. Review on nutritional importance and anti-nutritional factors of legumes. Int. J. Food Sci. Nutr. 2020, 9, 138–149. [Google Scholar] [CrossRef]
- Bharath, A.P.; Kathalsar, A.K.; Chandrashekhar, S.; Prabhasankar, P. Influence of tetraploid wheat (Triticum dicoccum) on low clycaemic index pizza base processing and its starch digestibility. Int. J. Food Sci. Technol. 2021, 56, 2273–2281. [Google Scholar] [CrossRef]
- Cardoso, B.B.; Amorim, C.; Silvério, S.C.; Rodrigues, L.R. Novel and emerging prebiotics: Advances and opportunities. Adv. Food Nutr. Res. 2021, 95, 41–95. [Google Scholar] [CrossRef]
- Spigno, G.; Pizzorno, T.; De Faveri, D.M. Cellulose and hemicelluloses recovery from grape stalks. Bioresour. Technol. 2008, 99, 4329–4337. [Google Scholar] [CrossRef]
- Dávila, I.; Gullón, B.; Alonso, J.L.; Labidi, J.; Gullón, P. Vine shoots as new source for the manufacture of prebiotic oligosaccharides. Carbohydr. Polym. 2019, 207, 34–43. [Google Scholar] [CrossRef]
- Teixeira, A.; Baenas, N.; Dominguez-Perles, R.; Barros, A.; Rosa, E.; Moreno, D.A.; Garcia-Viguera, C. Natural bioactive compounds from winery by-products as health promoters: A Review. Int. J. Mol. Sci. 2014, 15, 15638–15678. [Google Scholar] [CrossRef]
- Vazquez-Armenta, F.J.; Bernal-Mercado, A.T.; Lizardi-Mendoza, J.; Silva-Espinoza, B.A.; Cruz-Valenzuela, M.R.; Gonzalez-Aguilar, G.A.; Nazzaro, F.; Fratianni, F.; Ayala-Zavala, J.F. Phenolic extracts from grape stems inhibit Listeria Monocytogenes motility and adhesion to food contact surfaces. J. Adhes. Sci. Technol. 2018, 32, 889–907. [Google Scholar] [CrossRef]
- Jiménez-Moreno, N.; Volpe, F.; Moler, J.A.; Esparza, I.; Ancín-Azpilicueta, C. Impact of extraction conditions on the phenolic composition and antioxidant capacity of grape stem extracts. Antioxidants 2019, 8, 597. [Google Scholar] [CrossRef]
- Ferreyra, S.; Bottini, R.; Fontana, A. Assessment of grapevine stems as source of phenolics with antioxidant properties. Rev. Fac. Cienc. Agrar. 2019, 51, 1853–8665. [Google Scholar]
- Gouvinhas, I.; Pinto, R.; Santos, R.; Saavedra, M.J.; Barros, A.I. Enhanced phytochemical composition and biological activities of grape (Vitis vinifera L.) stems growing in low altitude regions. Sci. Hortic. 2020, 265, 109248. [Google Scholar] [CrossRef]
- Difonzo, G.; Antonino, C.; Squeo, G.; Caponio, F.; Faccia, M. Application of Agri-Food By-Products in Cheesemaking. Antioxidants 2023, 12, 660. [Google Scholar] [CrossRef]
- Teixeira, N.; Mateus, N.; de Freitas, V.; Oliveira, J. Wine Industry By-Product: Full Polyphenolic characterization of grape stalks. Food Chem. 2018, 268, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.H. Effect of extraction solvent on total phenol content, Total flavonoid content, and antioxidant activity of Limnophila Aromatica. J. Food Drug Anal. 2014, 22, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Vural, N.; Algan Cavuldak, Ö.; Akay, M.A.; Anlı, R.E. Determination of the various extraction solvent effects on polyphenolic profile and antioxidant activities of selected tea samples by chemometric approach. J. Food Meas. Charact. 2020, 14, 1286–1305. [Google Scholar] [CrossRef]
- Bhebhe, M.; Füller, T.N.; Chipurura, B.; Muchuweti, M. Effect of solvent type on total phenolic content and free radical ccavenging activity of black tea and herbal infusions. Food Anal. Methods 2016, 9, 1060–1067. [Google Scholar] [CrossRef]
- Sun, J.; Liang, F.; Bin, Y.; Li, P.; Duan, C. Screening non-colored phenolics in red wines using liquid chromatography/ultraviolet and mass spectrometry/mass spectrometry libraries. Molecules 2007, 12, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Di Lecce, G.; Arranz, S.; Jáuregui, O.; Tresserra-Rimbau, A.; Quifer-Rada, P.; Lamuela-Raventós, R.M. Phenolic profiling of the skin, pulp and seeds of albariño grapes using hybrid quadrupole time-of-flight and triple-quadrupole mass spectrometry. Food Chem. 2014, 145, 874–882. [Google Scholar] [CrossRef]
- Ghareeb, M.A.; Mohamed, T.; Saad, A.M.; Refahy, L.A.G.; Sobeh, M.; Wink, M. HPLC-DAD-ESI-MS/MS analysis of fruits from Firmiana simplex (L.) and evaluation of their antioxidant and antigenotoxic properties. J. Pharm. Pharmacol. 2018, 70, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Spinola, V.; Pinto, J.; Castilho, P.C. Identification and quantification of phenolic compounds of selected fruits from Madeira Island by HPLC-DAD–ESI-MSn and screening for their antioxidant activity. J. Food Chem. 2015, 173, 14–30. [Google Scholar] [CrossRef]
- Escobar-Avello, D.; Lozano-Castellón, J.; Mardones, C.; Pérez, A.J.; Saéz, V.; Riquelme, S.; Von Baer, D.; Vallverdú-Queralt, A. Phenolic profile of grape canes: Novel compounds identified by LC-ESI-LTQ-orbitrap-MS. Molecules 2019, 24, 3763. [Google Scholar] [CrossRef]
- Piccolella, S.; Crescente, G.; Volpe, M.G.; Paolucci, M.; Pacifico, S. UHPLC-HR-MS/MS-Guided recovery of bioactive. Molecules 2019, 24, 3630. [Google Scholar] [CrossRef] [PubMed]
- Goufo, P.; Singh, R.K.; Cortez, I. A Reference List of phenolic compounds (including stilbenes) in grapevine (Vitis vinifera L.) roots, woods, canes, stems, and leaves. Antioxidants 2020, 9, 398. [Google Scholar] [CrossRef] [PubMed]
- López-fernández, O.; Domínguez, R.; Pateiro, M.; Munekata, P.E.S.; Rocchetti, G.; Lorenzo, J.M. Determination of polyphenols using liquid chromatography–tandem mass spectrometry technique (LC–MS/MS): A review. Antioxidants 2020, 9, 479. [Google Scholar] [CrossRef]
- Stój, A.; Kapusta, I.; Domagała, D. Classification of red wines produced from zweigelt and rondo grape varieties based on the analysis of phenolic compounds by UPLC-PDA-MS/MS. Molecules 2020, 25, 1342. [Google Scholar] [CrossRef] [PubMed]
- Escobar-Avello, D.; Mardones, C.; Saéz, V.; Riquelme, S.; von Baer, D.; Lamuela-Raventós, R.M.; Vallverdú-Queralt, A. Pilot-plant ccale extraction of phenolic compounds from grape canes: Comprehensive characterization by LC-ESI-LTQ-Orbitrap-MS. Food Res. Int. 2021, 143, 110265. [Google Scholar] [CrossRef] [PubMed]
- Myrtsi, E.D.; Koulocheri, S.D.; Iliopoulos, V.; Haroutounian, S.A. High-throughput quantification of 32 bioactive antioxidant phenolic compounds in grapes, wines and vinification byproducts by LC–MS/MS. Antioxidants 2021, 10, 1174. [Google Scholar] [CrossRef] [PubMed]
- Razgonova, M.; Zakharenko, A.; Pikula, K.; Manakov, Y.; Ercisli, S.; Derbush, I.; Kislin, E.; Seryodkin, I.; Sabitov, A.; Kalenik, T.; et al. Lc-ms/Ms screening of phenolic compounds in wild and cultivated grapes vitis Amurensis Rupr. Molecules 2021, 26, 3650. [Google Scholar] [CrossRef]
- Iorio, D.; Gambacorta, G.; Tarricone, L.; Vilanova, M.; Paradiso, V.M. Effect of early basal leaf removal on phenolic and volatile composition and sensory properties of aglianico red wines. Plants 2022, 11, 591. [Google Scholar] [CrossRef]
- Troilo, M.; Difonzo, G.; Paradiso, V.M.; Pasqualone, A.; Caponio, F. Grape pomace as innovative flour for the formulation of functional muffins: How particle size affects the nutritional, textural and sensory properties. Foods 2022, 11, 1799. [Google Scholar] [CrossRef]
- Li, S.; Lin, Z.; Jiang, H.; Tong, L.; Wang, H.; Chen, S. Rapid identification and assignation of the active ingredients in fufang banbianlian injection using. J. Chromatogr. Sci. 2016, 54, 1225–1237. [Google Scholar] [CrossRef]
- Makhlouf, F.Z.; Squeo, G.; Barkat, M.; Trani, A.; Caponio, F. Antioxidant activity, tocopherols and polyphenols of acornoil obtained from quercus species grown in algeria. Food Res. Int. 2018, 114, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Difonzo, G.; Troilo, M.; Allegretta, I.; Pasqualone, A.; Caponio, F. Grape skin and seed flours as functional ingredients of pizza: Potential and drawbacks related to nutritional, physicochemical and sensory attributes. LWT 2023, 175, 114494. [Google Scholar] [CrossRef]
- Difonzo, G.; Aresta, A.; Cotugno, P.; Ragni, R.; Squeo, G.; Summo, C.; Massari, F.; Pasqualone, A.; Faccia, M.; Zambonin, C.; et al. Supercritical CO2 extraction of phytocompounds from olive pomace subjected to different drying methods. Molecules 2021, 26, 598. [Google Scholar] [CrossRef] [PubMed]
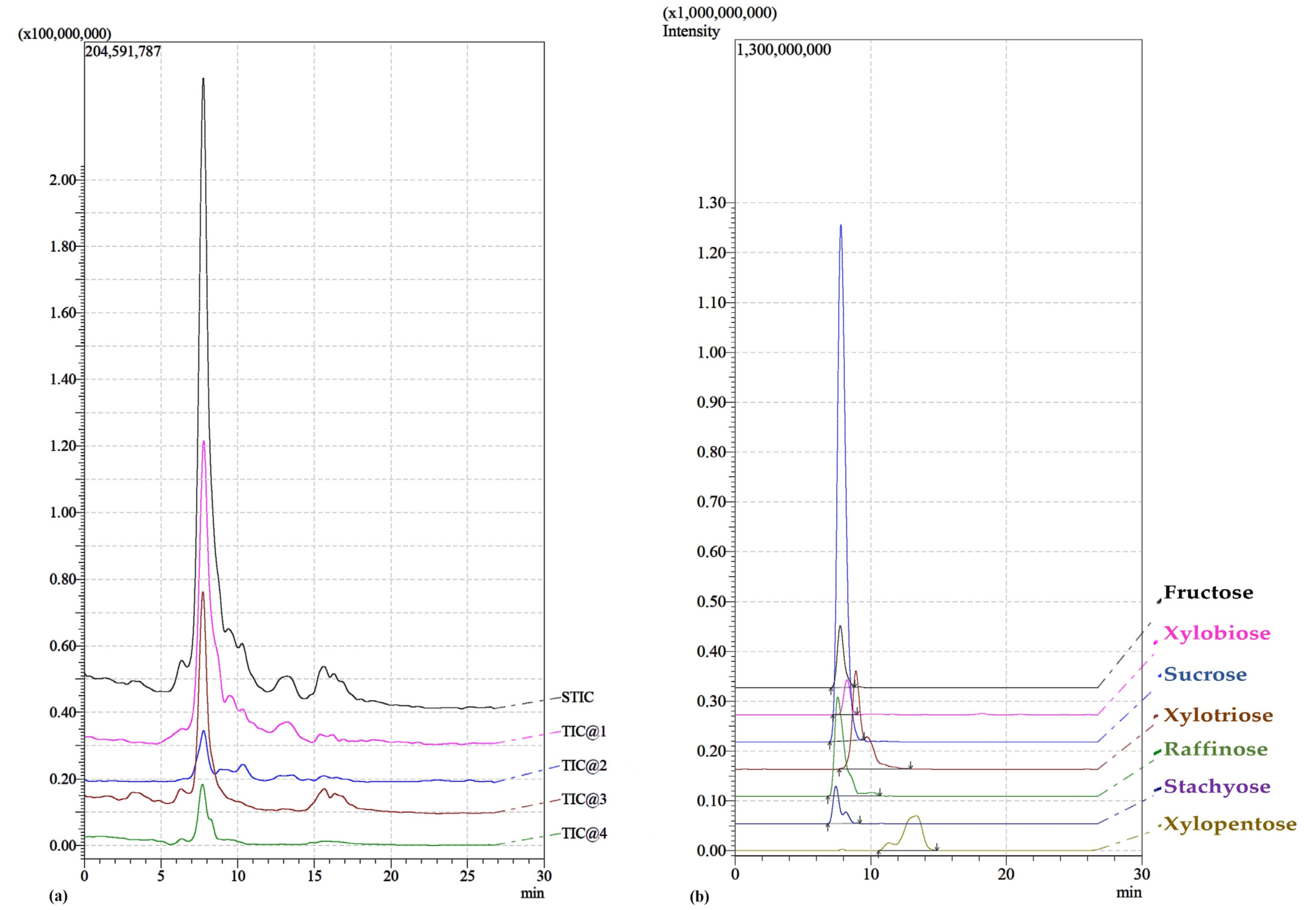


| Sugars | Molecular Formula | Exact Mass (Molecular Ion) | Predicted Mass (Na Adduct) | Measured Mass | Diff PPM |
|---|---|---|---|---|---|
| Fructose | C6H12O6 | 180.0634 | 203.0526 | 203.0501 | 12.3121 |
| Stachyose | C24H42O21 | 666.2219 | 689.2111 | 689.2101 | 1.4509 |
| Xylobiose | C10H18O9 | 282.0951 | 305.0843 | 305.0848 | 1.6389 |
| Raffinose | C18H32O16 | 504.1690 | 527.1583 | 527.1541 | 7.9673 |
| Sucrose | C12H22O11 | 342.1162 | 365.1054 | 365.1083 | 7.9429 |
| Xylotriose | C15H26O13 | 414.1373 | 437.1266 | 437.1247 | 4.3466 |
| Xylotetraose | C20H34O17 | 546.1796 | 569.1688 | 569.1651 | 6.5007 |
| Xylopentose | C25H42O21 | 678.2219 | 701.2111 | 701.2090 | 2.9948 |
| Compounds | Vine Shoots (VS) | ||
|---|---|---|---|
| H2O | H2O-EtOH | H2O-Amberlyst | |
| Fructose | 3.98 ± 0.12 b | 8.51 ± 0.04 a | 3.64 ± 0.54 b |
| Sucrose | 1.21 ± 0.03 a | 0.36 ± 0.03 b | 0.41 ± 0.13 b |
| Xylobiose | 2.67 ± 0.31 b | 3.53 ± 0.32 a | 3.69 ± 0.04 a |
| Xylotriose | 2.65 ± 0.02 a | 2.56 ± 0.00 a | 2.32 ± 0.06 b |
| Xylotetraose | 7.30 ± 0.71 c | 21.35 ± 0.47 a | 9.68 ± 0.78 b |
| Xylopentose | 32.84 ± 1.67 c | 45.98 ± 0.85 b | 51.02 ± 0.98 a |
| Raffinose | 15.86 ± 0.16 a | 4.19 ± 0.57 c | 11.01 ± 0.16 b |
| Stachyose | 32.45 ± 1.63 a | 12.82 ± 0.18 c | 17.03 ± 0.46 b |
| Total xylo-oligosaccarides | 45.46 ± 1.70 c | 73.42 ± 1.38 a | 66.72 ± 0.36 b |
| Other compounds | 1.03 | 1.34 | 1.19 |
| Compounds | Grape Stalks (GS) | ||
|---|---|---|---|
| H2O | H2O-EtOH | H2O-Amberlyst | |
| Fructose | 17.77 ± 0.49 b | 33.19 ± 0.37 a | 17.13 ± 0.44 c |
| Sucrose | 0.22 ± 0.03 b | 0.29 ± 0.01 a | 0.28 ± 0.01 a |
| Xylobiose | 5.29 ± 0.45 b | 15.59 ± 0.36 a | 5.90 ± 0.17 b |
| Xylotriose | 1.40 ± 0.06 b | 5.14 ± 0.26 a | 1.69 ± 0.08 b |
| Xylotetraose | 21.53 ± 1.35 a | 9.37 ± 0.64 c | 13.04 ± 1.12 b |
| Xylopentose | 39.90 ± 1.17 a | 25.65 ± 0.77 b | 39.86 ± 0.70 a |
| Raffinose | 5.42 ± 0.27 a | 4.19 ± 0.07 b | 4.54 ± 0.15 b |
| Stachyose | 6.15 ± 0.35 b | 3.69 ± 0.09 c | 15.09 ± 0.44 a |
| Total xylo-oligosaccarides | 68.13 ± 1.73 a | 55.45 ± 0.83 c | 60.49 ± 0.83 b |
| Other compounds | 2.32 | 2.89 | 2.47 |
| Vine Shoots (VS) | Grape Stalks (GS) | |||||
|---|---|---|---|---|---|---|
| Parameters | H2O | H2O-EtOH | H2O-Amberlyst | H2O | H2O-EtOH | H2O-Amberlyst |
| ABTS (µmol TE/g) | 65.67 ± 1.80 a | 36.41 ± 0.50 b | 23.81 ± 0.32 c | 58.35 ± 1.25 c | 128.04 ± 1.25 a | 73.60 ± 0.79 b |
| DPPH (µmol TE/g) | 250.14 ± 0.56 a | 148.17 ± 0.56 b | 107.71 ± 4.12 c | 132.98 ± 4.19 c | 425.66 ± 9.99 a | 249.45 ± 2.41 b |
| TPC (mg GAE/g) | 58.03 ± 1.80 a | 35.80 ± 0.97 b | 28.76 ± 0.52 c | 49.99 ± 0.07 c | 90.92 ± 1.76 a | 56.62 ± 0.26 b |
| Compounds | RT (min) | [M − H]− | MS/MS Ion | Molecular Formula | Reference |
|---|---|---|---|---|---|
| Citric acid | 2.342 | 191.0197 | 111.0123; 173.0107; 191.0202 | C6H8O7 | [55,63] |
| Gallic acid | 6.032 | 169.0170 | 125.0278 | C7H6O5 | [52,53,54,56,57,59,61,62,63,64] |
| Monogalloyl glucose | 8.417 | 331.0669 | 313.0569; 169.0167; 168.0093; 125.0293 | C13H16O10 | [53,57,60,62,64] |
| Caftaric acid | 9.002 | 311.0409 | 111.0113; 173.0120 | C13H12O9 | [52,54,56,58,59,61,63,64] |
| Coutaric acid | 9.407 | 295.0453 | 112.9935; 149.0132; 163.0425; 251.0447 | C13H12O8 | [53,56,58,60,61,63] |
| Quercetin-3-O-glucuronide | 11.150 | 477.0702 | 175.0280; 301.0357; 413.0928 | C21H18O13 | [56,58,60,61,63] |
| Kaempferol-3-O-glucoside | 11.842 | 447.0907 | 150.9993; 255.032; 285.0396; 32.0492 | C20H20O11 | [58,59,61] |
| Taxifolin | 12.787 | 303.0481 | 125.0428; 177.019; 285.0536 | C15H12O7 | [56,58,59,61] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Difonzo, G.; Troilo, M.; Casiello, M.; D’Accolti, L.; Caponio, F. Autohydrolysis Application on Vine Shoots and Grape Stalks to Obtain Extracts Enriched in Xylo-Oligosaccharides and Phenolic Compounds. Molecules 2023, 28, 3760. https://doi.org/10.3390/molecules28093760
Difonzo G, Troilo M, Casiello M, D’Accolti L, Caponio F. Autohydrolysis Application on Vine Shoots and Grape Stalks to Obtain Extracts Enriched in Xylo-Oligosaccharides and Phenolic Compounds. Molecules. 2023; 28(9):3760. https://doi.org/10.3390/molecules28093760
Chicago/Turabian StyleDifonzo, Graziana, Marica Troilo, Michele Casiello, Lucia D’Accolti, and Francesco Caponio. 2023. "Autohydrolysis Application on Vine Shoots and Grape Stalks to Obtain Extracts Enriched in Xylo-Oligosaccharides and Phenolic Compounds" Molecules 28, no. 9: 3760. https://doi.org/10.3390/molecules28093760
APA StyleDifonzo, G., Troilo, M., Casiello, M., D’Accolti, L., & Caponio, F. (2023). Autohydrolysis Application on Vine Shoots and Grape Stalks to Obtain Extracts Enriched in Xylo-Oligosaccharides and Phenolic Compounds. Molecules, 28(9), 3760. https://doi.org/10.3390/molecules28093760











