Exploration of Succinimide Derivative as a Multi-Target, Anti-Diabetic Agent: In Vitro and In Vivo Approaches
Abstract
1. Introduction
2. Results
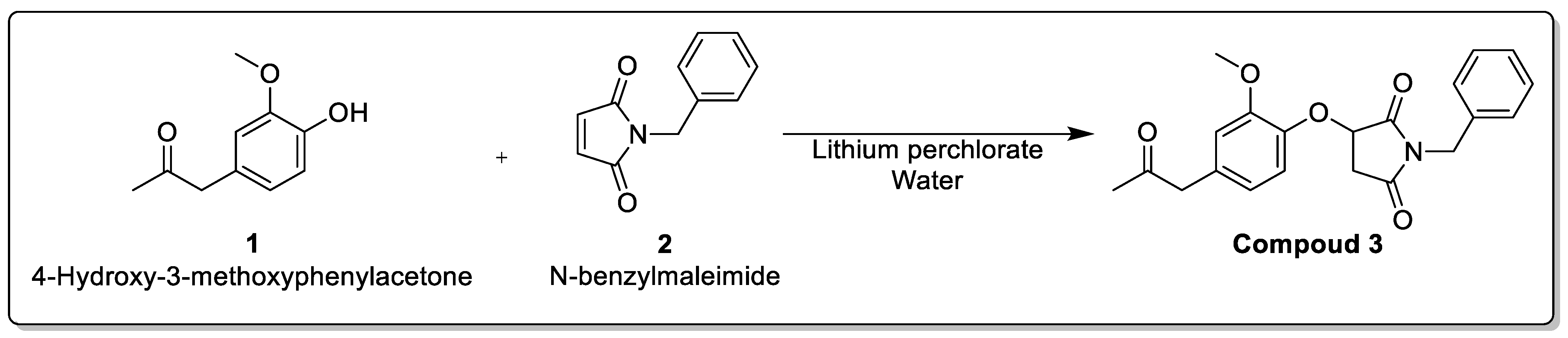
2.1. Chemistry of Compound
2.2. In Vitro Bioassays
2.3. In Vivo Assays
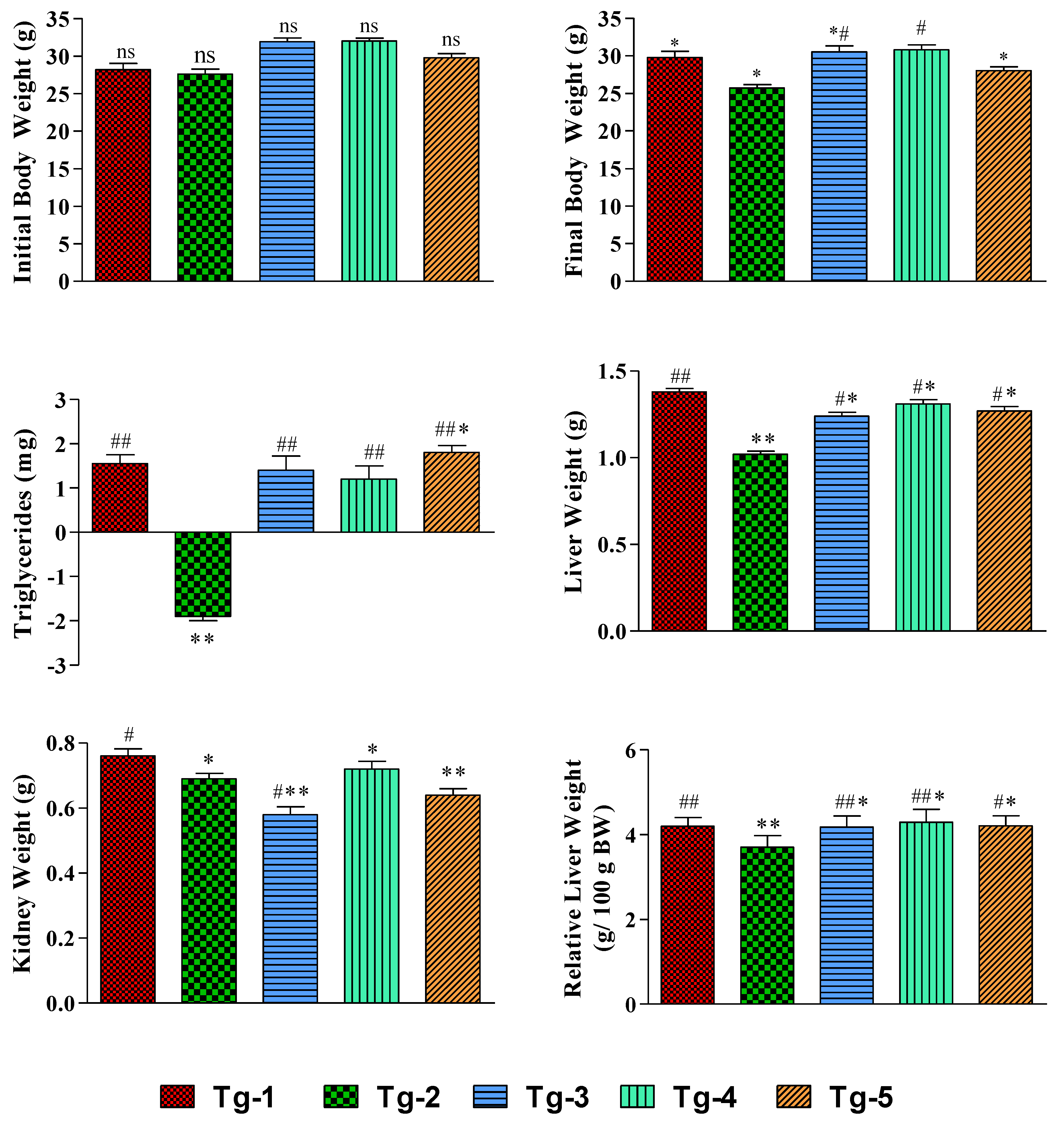
2.4. Effect of Compound’s 3 Body Weight and Organ Weight
2.5. Effect of Compound 3 on Lipid Profile
2.6. Serum Markers of the Kidneys and Liver
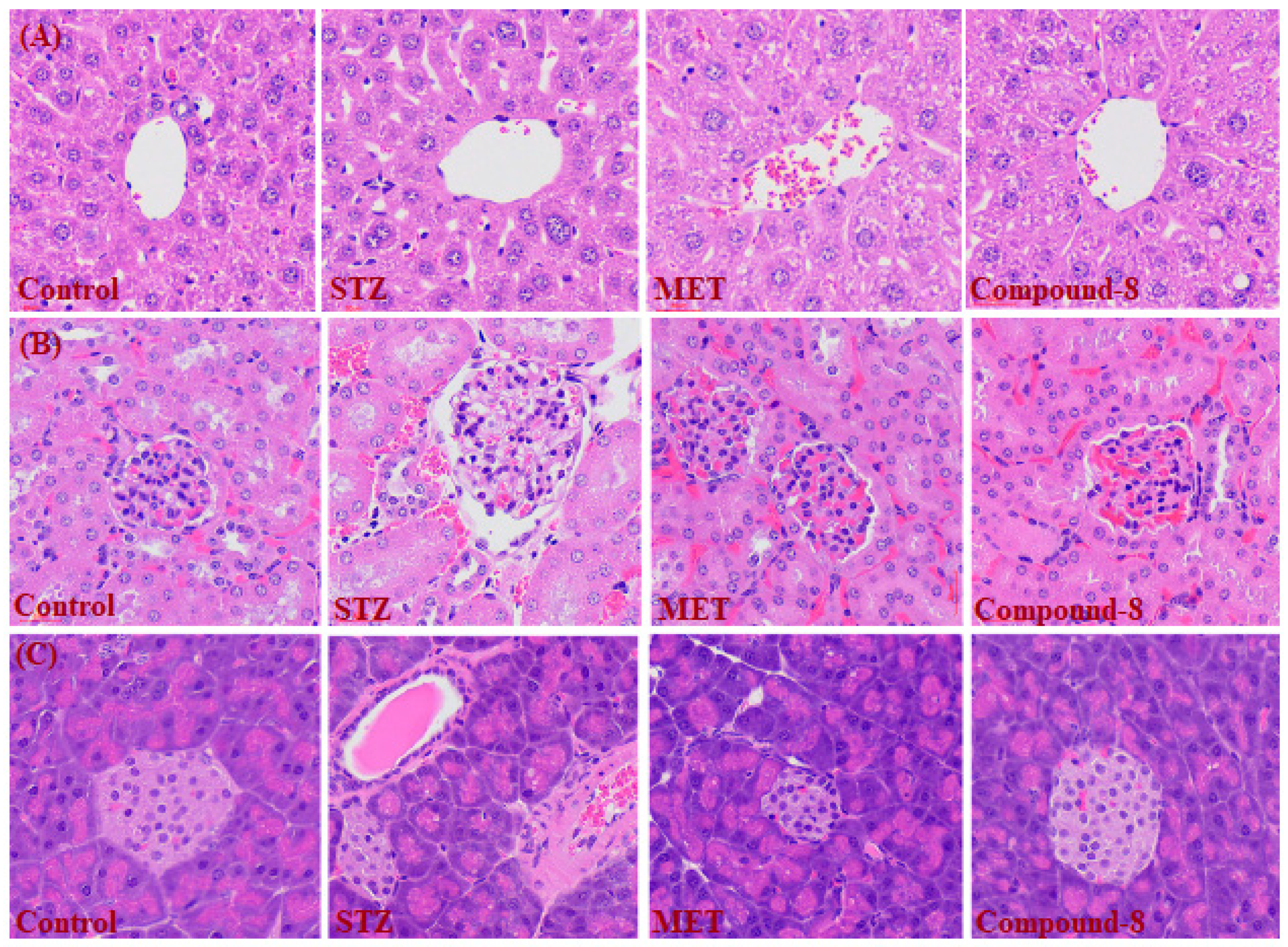
2.7. Effect of Compound 3 on Histopathology
3. Material and Methods
3.1. Chemical Used
3.2. Chemistry of Compound
3.3. Pharmacological Activities
3.3.1. Protein Tyrosine Phosphatase 1 B (PTP1 B) Test
3.3.2. Alpha-Glucosidase Test
3.3.3. Alpha-Amylase Test
3.3.4. Dipeptidyl Peptidase-4 (DPP-4) Test
3.3.5. Antioxidant (DPPH) Test
3.4. Experimental Animals and Ethical Guidelines
3.5. Acute Toxicity
3.6. Induction of Diabetes
3.7. In Vivo Experiment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Elsayed, M.S.; El Badawy, A.; Omar, R.; Mohamed, A. Serum cystatin C as an indicator for early detection of diabetic nephropathy in type 2 diabetes mellitus. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 374–381. [Google Scholar] [CrossRef]
- Lawal, M.; Verma, A.K.; Umar, I.A.; Gadanya, A.M.; Umar, B.; Yahaya, N.; Auwal, B. Analysis of New Potent Anti-Diabetic Molecules from Phytochemicals of PistiaStrateotes with Sglt1 and G6pc Proteins of Homo Sapiens for Treatment of Diabetes Mellitus. An In SilicoApproach. Silico Approach IOSR JPBS 2020, 15, 59–73. [Google Scholar]
- Mirzaei, F.; Khodadadi, I.; Vafaei, S.A.; Abbasi-Oshaghi, E.; Tayebinia, H.; Farahani, F. Importance of hyperglycemia in COVID-19 intensive-care patients: Mechanism and treatment strategy. Prim. Care Diabetes 2021, 15, 409–416. [Google Scholar] [CrossRef]
- Zhang, S.; Guo, F.; Yu, M.; Yang, X.; Yao, Z.; Li, Q.; Wei, Z.; Feng, K.; Zeng, P.; Zhao, D. Reduced Nogo expression inhibits diet-induced metabolic disorders by regulating ChREBP and insulin activity. J. Hepatol. 2020, 73, 1482–1495. [Google Scholar] [CrossRef]
- Zheng, W.; Chen, L.; Chen, J.-H.; Lin, X.; Tang, Y.; Lin, X.-J.; Wu, J.; Lin, Z.-M.; Lin, J.-Y. Hemichorea associated with non-ketotic hyperglycemia: A case report and literature review. Front. Neurol. 2020, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Liu, J.; Wang, D.; Qiu, S.; Yuan, Y.; Wang, F.; Wen, L.; Song, Q.; Sun, Z.-l. Efficacy of calcium dobesilate in treating Chinese patients with mild-to-moderate non-proliferative diabetic retinopathy (CALM-DR): Protocol for a single-blind, multicentre, 24-armed cluster-randomised, controlled trial. BMJ Open 2021, 11, e045256. [Google Scholar] [CrossRef] [PubMed]
- Irwin, G.M. Erectile dysfunction. Prim. Care Clin. Off. Pract. 2019, 46, 249–255. [Google Scholar] [CrossRef]
- Rasouli, H.; Yarani, R.; Pociot, F.; Popović-Djordjević, J. Anti-diabetic potential of plant alkaloids: Revisiting current findings and future perspectives. Pharmacol. Res. 2020, 155, 104723. [Google Scholar] [CrossRef] [PubMed]
- Deprince, A.; Haas, J.T.; Staels, B. Dysregulated lipid metabolism links NAFLD to cardiovascular disease. Mol. Metab. 2020, 42, 101092. [Google Scholar] [CrossRef] [PubMed]
- Farid, A.; Haridyy, H.; Ashraf, S.; Ahmed, S.; Safwat, G. Co-treatment with grape seed extract and mesenchymal stem cells in vivo regenerated beta cells of islets of Langerhans in pancreas of type I-induced diabetic rats. Stem Cell Res. Ther. 2022, 13, 528. [Google Scholar] [CrossRef] [PubMed]
- Docherty, F.M.; Sussel, L. Islet regeneration: Endogenous and exogenous approaches. Int. J. Mol. Sci. 2021, 22, 3306. [Google Scholar] [CrossRef]
- Wiggins, S.C.; Abuid, N.J.; Gattás-Asfura, K.M.; Kar, S.; Stabler, C.L. Nanotechnology approaches to modulate immune responses to cell-based therapies for type 1 diabetes. J. Diabetes Sci. Technol. 2020, 14, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, K.; Huang, Q.; Shu, S. Molecular Cloning And Characterization of An Alpha-Amylase Inhibitor (Tkaai) Gene From Trichosanthes Kirilowii Maxim. Biotechnol. Lett. 2022, 44, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Mahnashi, M.H.; Alyami, B.A.; Alqahtani, Y.S.; Jan, M.S.; Rashid, U.; Sadiq, A.; Alqarni, A.O. Phytochemical profiling of bioactive compounds, anti-inflammatory and analgesic potentials of Habenaria digitata Lindl.: Molecular docking based synergistic effect of the identified compounds. J. Ethnopharmacol. 2021, 273, 113976. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Su, L.; Wang, L.; Wu, J.; Chen, S. Alpha-glucanotransferase from the glycoside hydrolase family synthesizes α (1–6)-linked products from starch: Features and synthesis pathways of the products. Trends Food Sci. Technol. 2022, 128, 160–172. [Google Scholar] [CrossRef]
- Aslam, H.; Khan, A.-u.; Naureen, H.; Ali, F.; Ullah, F.; Sadiq, A. Potential application of Conyza canadensis (L) Cronquist in the management of diabetes: In vitro and in vivo evaluation. Trop. J. Pharm. Res. 2018, 17, 1287–1293. [Google Scholar] [CrossRef]
- Mahnashi, M.H.; Alqahtani, Y.S.; Alyami, B.A.; Alqarni, A.O.; Ahmed Alshrahili, M.; Abou-Salim, M.A.; Alqahtani, M.N.; Mushtaq, S.; Sadiq, A.; Jan, M.S. GC-MS Analysis and Various In Vitro and In Vivo Pharmacological Potential of Habenaria plantaginea Lindl. Evid.-Based Complement. Altern. Med. 2022, 2022, 7921408. [Google Scholar] [CrossRef]
- Buday, L.; Vas, V. Novel regulation of Ras proteins by direct tyrosine phosphorylation and dephosphorylation. Cancer Metastasis Rev. 2020, 39, 1067–1073. [Google Scholar] [CrossRef]
- Mahnashi, M.H.; Alqahtani, Y.S.; Alqarni, A.O.; Alyami, B.A.; Jan, M.S.; Ayaz, M.; Ullah, F.; Rashid, U.; Sadiq, A. Crude extract and isolated bioactive compounds from Notholirion thomsonianum (Royale) Stapf as multitargets antidiabetic agents: In-vitro and molecular docking approaches. BMC Complement. Med. Ther. 2021, 21, 270. [Google Scholar] [CrossRef]
- Sadiq, A.; Nugent, T.C. Catalytic access to succinimide products containing stereogenic quaternary carbons. ChemistrySelect 2020, 5, 11934–11938. [Google Scholar] [CrossRef]
- Berger, J.P.; SinhaRoy, R.; Pocai, A.; Kelly, T.M.; Scapin, G.; Gao, Y.D.; Pryor, K.A.D.; Wu, J.K.; Eiermann, G.J.; Xu, S.S. A comparative study of the binding properties, dipeptidyl peptidase-4 (DPP-4) inhibitory activity and glucose-lowering efficacy of the DPP-4 inhibitors alogliptin, linagliptin, saxagliptin, sitagliptin and vildagliptin in mice. Endocrinol. Diabetes Metab. 2018, 1, e00002. [Google Scholar] [CrossRef] [PubMed]
- Hedrington, M.S.; Davis, S.N. Considerations when using alpha-glucosidase inhibitors in the treatment of type 2 diabetes. Expert Opin. Pharmacother. 2019, 20, 2229–2235. [Google Scholar] [CrossRef]
- Mahnashi, M.H.; Alyami, B.A.; Alqahtani, Y.S.; Alqarni, A.O.; Jan, M.S.; Hussain, F.; Zafar, R.; Rashid, U.; Abbas, M.; Tariq, M. Antioxidant Molecules Isolated from Edible Prostrate Knotweed: Rational Derivatization to Produce More Potent Molecules. Oxidative Med. Cell. Longev. 2022, 2022, 3127480. [Google Scholar] [CrossRef] [PubMed]
- Huneif, M.A.; Alshehri, D.B.; Alshaibari, K.S.; Dammaj, M.Z.; Mahnashi, M.H.; Majid, S.U.; Javed, M.A.; Ahmad, S.; Rashid, U.; Sadiq, A. Design, synthesis and bioevaluation of new vanillin hybrid as multitarget inhibitor of α-glucosidase, α-amylase, PTP-1B and DPP4 for the treatment of type-II diabetes. Biomed. Pharmacother. 2022, 150, 113038. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.-f.; Zhu, L.-l.; Chen, M.; Xu, H.-m.; Wang, H.-f.; Feng, X.-q.; Zhu, X.-p.; Zhou, Q. The optimal choice of medication administration route regarding intravenous, intramuscular, and subcutaneous injection. Patient Prefer. Adherence 2015, 9, 923. [Google Scholar]
- Javed, M.A.; Jan, M.S.; Shbeer, A.M.; Al-Ghorbani, M.; Rauf, A.; Wilairatana, P.; Mannan, A.; Sadiq, A.; Farooq, U.; Rashid, U. Evaluation of pyrimidine/pyrrolidine-sertraline based hybrids as multitarget anti-Alzheimer agents: In-vitro, in-vivo, and computational studies. Biomed. Pharmacother. 2023, 159, 114239. [Google Scholar] [CrossRef]
- Malerba, F.; Paoletti, F.; Capsoni, S.; Cattaneo, A. Intranasal delivery of therapeutic proteins for neurological diseases. Expert Opin. Drug Deliv. 2011, 8, 1277–1296. [Google Scholar] [CrossRef]
- Deacon, C.; Lebovitz, H.E. Comparative review of dipeptidyl peptidase-4 inhibitors and sulphonylureas. Diabetes Obes. Metab. 2016, 18, 333–347. [Google Scholar] [CrossRef]
- Nugent, T.C.; Sadiq, A.; Bibi, A.; Heine, T.; Zeonjuk, L.L.; Vankova, N.; Bassil, B.S. Noncovalent bifunctional organocatalysts: Powerful tools for contiguous quaternary-tertiary stereogenic carbon formation, scope, and origin of enantioselectivity. Chem.–A Eur. J. 2012, 18, 4088–4098. [Google Scholar] [CrossRef]
- Makhoba, X.H.; Viegas Jr, C.; Mosa, R.A.; Viegas, F.P.; Pooe, O.J. Potential impact of the multi-target drug approach in the treatment of some complex diseases. Drug Des. Dev. Ther. 2020, 14, 3235. [Google Scholar] [CrossRef]
- Qayyum, M.I.; Ullah, S.; Rashid, U.; Mahnashi, M.H.; Alshahrani, M.M.; Al Ali, A.; Asiri, A.; Al Awadh, A.A.; Alshehri, O.M.; Sadiq, A. Design, synthesis and preclinical evaluations of (s)-2-((s)-1-benzyl-2, 5-dioxopyrrolidin-3-yl)-3-(4-isopropylphenyl)-2-methylpropanal (succ-5) as cardioprotective, hepatoprotective and lipid lowering molecule. in-vivo and in-silico approaches. Arab. J. Chem. 2023, 16, 104504. [Google Scholar] [CrossRef]
- Vieira, R.; Souto, S.B.; Sánchez-López, E.; López Machado, A.; Severino, P.; Jose, S.; Santini, A.; Fortuna, A.; García, M.L.; Silva, A.M. Sugar-lowering drugs for type 2 diabetes mellitus and metabolic syndrome—Review of classical and new compounds: Part-I. Pharmaceuticals 2019, 12, 152. [Google Scholar] [CrossRef] [PubMed]
- Waheed, B.; Mukarram Shah, S.M.; Hussain, F.; Khan, M.I.; Zeb, A.; Jan, M.S. Synthesis, antioxidant, and antidiabetic activities of ketone derivatives of succinimide. Evid.-Based Complement. Altern. Med. 2022, 2022, 1445604. [Google Scholar] [CrossRef]
- Pervaiz, A.; Jan, M.S.; Hassan Shah, S.M.; Khan, A.; Zafar, R.; Ansari, B.; Shahid, M.; Hussain, F.; Ijaz Khan, M.; Zeb, A. Comparative in-vitro anti-inflammatory, anticholinesterase and antidiabetic evaluation: Computational and kinetic assessment of succinimides cyano-acetate derivatives. J. Biomol. Struct. Dyn. 2022, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hussain, F.; Khan, Z.; Jan, M.S.; Ahmad, S.; Ahmad, A.; Rashid, U.; Ullah, F.; Ayaz, M.; Sadiq, A. Synthesis, in-vitro α-glucosidase inhibition, antioxidant, in-vivo antidiabetic and molecular docking studies of pyrrolidine-2, 5-dione and thiazolidine-2, 4-dione derivatives. Bioorganic Chem. 2019, 91, 103128. [Google Scholar] [CrossRef] [PubMed]
- Mahnashi, M.H.; Alqahtani, Y.S.; Alqarni, A.O.; Alyami, B.A.; Alqahtani, O.S.; Jan, M.S.; Hussain, F.; Islam, Z.U.; Ullah, F.; Ayaz, M. Phytochemistry, anti-diabetic and antioxidant potentials of Allium consanguineum Kunth. BMC Complement. Med. Ther. 2022, 22, 154. [Google Scholar] [CrossRef]
- Finch, S.C.; Boundy, M.J.; Harwood, D.T. The acute toxicity of tetrodotoxin and tetrodotoxin–saxitoxin mixtures to mice by various routes of administration. Toxins 2018, 10, 423. [Google Scholar] [CrossRef]
- Assadi, S.; Shafiee, S.M.; Erfani, M.; Akmali, M. Antioxidative and antidiabetic effects of Capparis spinosa fruit extract on high-fat diet and low-dose streptozotocin-induced type 2 diabetic rats. Biomed. Pharmacother. 2021, 138, 111391. [Google Scholar] [CrossRef]
- Campbell, J.E.; Newgard, C.B. Mechanisms controlling pancreatic islet cell function in insulin secretion. Nat. Rev. Mol. Cell Biol. 2021, 22, 142–158. [Google Scholar] [CrossRef]
- Napolitano, A.; Miller, S.; Nicholls, A.W.; Baker, D.; Van Horn, S.; Thomas, E.; Rajpal, D.; Spivak, A.; Brown, J.R.; Nunez, D.J. Novel gut-based pharmacology of metformin in patients with type 2 diabetes mellitus. PLoS ONE 2014, 9, e100778. [Google Scholar] [CrossRef]
- Jaishree, V.; Narsimha, S. Swertiamarin and quercetin combination ameliorates hyperglycemia, hyperlipidemia and oxidative stress in streptozotocin-induced type 2 diabetes mellitus in wistar rats. Biomed. Pharmacother. 2020, 130, 110561. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-X.; Chen, W.; Liu, X.; Liu, Y.-R.; Zhu, S.-L. A review of the open charm and open bottom systems. Rep. Prog. Phys. 2017, 80, 076201. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Aljadhey, H.; Kesterson, J.; Murray, M.D.; Hall, S.D. Patients with elevated liver enzymes are not at higher risk for statin hepatotoxicity. Gastroenterology 2004, 126, 1287–1292. [Google Scholar] [CrossRef] [PubMed]
- Rashid, Y.; Rashid, A.; Warraich, M.A.; Sabir, S.S.; Waseem, A. Case study method: A step-by-step guide for business researchers. Int. J. Qual. Methods 2019, 18, 1609406919862424. [Google Scholar] [CrossRef]
- Al-Owaisi, M.; Al-Hadiwi, N.; Khan, S.A. GC-MS analysis, determination of total phenolics, flavonoid content and free radical scavenging activities of various crude extracts of Moringa peregrina (Forssk.) Fiori leaves. Asian Pac. J. Trop. Biomed. 2014, 4, 964–970. [Google Scholar] [CrossRef]
- Fagbohun, O.F.; Awoniran, P.O.; Babalola, O.O.; Agboola, F.K.; Msagati, T.A. Changes in the biochemical, hematological and histopathological parameters in STZ-Induced diabetic rats and the ameliorative effect of Kigelia africana fruit extract. Heliyon 2020, 6, e03989. [Google Scholar] [CrossRef]
- Sadiq, A.; Mahnashi, M.H.; Rashid, U.; Jan, M.S.; Alshahrani, M.A.; Huneif, M.A. 3-(((1S, 3S)-3-((R)-Hydroxy (4-(trifluoromethyl) phenyl) methyl)-4-oxocyclohexyl) methyl) pentane-2, 4-dione: Design and Synthesis of New Stereopure Multi-Target Antidiabetic Agent. Molecules 2022, 27, 3265. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Ullah, F.; Sadiq, A.; Ayaz, M.; Jan, M.S.; Shahid, M.; Wadood, A.; Mahmood, F.; Rashid, U.; Ullah, R. Comparative cholinesterase, α-glucosidase inhibitory, antioxidant, molecular docking, and kinetic studies on potent succinimide derivatives. Drug Des. Dev. Ther. 2020, 14, 2165. [Google Scholar] [CrossRef] [PubMed]
- Zafar, R.; Zubair, M.; Ali, S.; Shahid, K.; Waseem, W.; Naureen, H.; Haider, A.; Jan, M.S.; Ullah, F.; Sirajuddin, M. Zinc metal carboxylates as potential anti-Alzheimer’s candidate: In vitro anticholinesterase, antioxidant and molecular docking studies. J. Biomol. Struct. Dyn. 2021, 39, 1044–1054. [Google Scholar] [CrossRef]
- Mahmood, F.; Khan, J.A.; Mahnashi, M.H.; Jan, M.S.; Javed, M.A.; Rashid, U.; Sadiq, A.; Hassan, S.S.U.; Bungau, S. Anti-inflammatory, analgesic and antioxidant potential of new (2 S, 3 S)-2-(4-isopropylbenzyl)-2-methyl-4-nitro-3-phenylbutanals and their Corresponding carboxylic acids through in vitro, in silico and in vivo studies. Molecules 2022, 27, 4068. [Google Scholar] [CrossRef]
- Jan, M.S.; Ahmad, S.; Hussain, F.; Ahmad, A.; Mahmood, F.; Rashid, U.; Ullah, F.; Ayaz, M.; Sadiq, A. Design, synthesis, in-vitro, in-vivo and in-silico studies of pyrrolidine-2, 5-dione derivatives as multitarget anti-inflammatory agents. Eur. J. Med. Chem. 2020, 186, 111863. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.; Islam, M.S.; Zannah, S.; Sadik, G.; Rashid, M. Momordica charantia (Bitter melon) in combination with metformin potentiates hypoglycemic and hypolipidemic effects in alloxan-induced diabetic rats. Bangladesh Pharm. J. 2018, 21, 109–117. [Google Scholar] [CrossRef]
- Ul Haq, M.N.; Shah, G.M.; Menaa, F.; Khan, R.A.; Althobaiti, N.A.; Albalawi, A.E.; Alkreathy, H.M. Green Silver Nanoparticles Synthesized from Taverniera couneifolia Elicits Effective Anti-Diabetic Effect in Alloxan-Induced Diabetic Wistar Rats. Nanomaterials 2022, 12, 1035. [Google Scholar] [CrossRef] [PubMed]
- Alshehri, O.M.; Mahnashi, M.H.; Sadiq, A.; Zafar, R.; Jan, M.S.; Ullah, F.; Alshehri, M.A.; Alshamrani, S.; Hassan, E.E. Succinimide Derivatives as Antioxidant Anticholinesterases, Anti-α-Amylase, and Anti-α-Glucosidase: In Vitro and In Silico Approaches. Evid.-Based Complement. Altern. Med. 2022, 2022, 6726438. [Google Scholar] [CrossRef] [PubMed]
- Bibi, A.; Shah, T.; Sadiq, A.; Khalid, N.; Ullah, F.; Iqbal, A. L-isoleucine-catalyzed michael synthesis of N-alkylsuccinimide derivatives and their antioxidant activity assessment. Russ. J. Org. Chem. 2019, 55, 1749–1754. [Google Scholar] [CrossRef]
- Sadiq, A.; Mahnashi, M.H.; Alyami, B.A.; Alqahtani, Y.S.; Alqarni, A.O.; Rashid, U. Tailoring the substitution pattern of Pyrrolidine-2, 5-dione for discovery of new structural template for dual COX/LOX inhibition. Bioorganic Chem. 2021, 112, 104969. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, B.; Yang, J.; Liu, T.; Sun, J.; Wang, X. Synthesis and biological evaluation of 3-arylcoumarin derivatives as potential anti-diabetic agents. J. Enzym. Inhib. Med. Chem. 2019, 34, 15–30. [Google Scholar] [CrossRef]
- Asgari, M.S.; Mohammadi-Khanaposhtani, M.; Kiani, M.; Ranjbar, P.R.; Zabihi, E.; Pourbagher, R.; Rahimi, R.; Faramarzi, M.A.; Biglar, M.; Larijani, B. Biscoumarin-1, 2, 3-triazole hybrids as novel anti-diabetic agents: Design, synthesis, in vitro α-glucosidase inhibition, kinetic, and docking studies. Bioorganic Chem. 2019, 92, 103206. [Google Scholar] [CrossRef]
- Miethke, M.; Pieroni, M.; Weber, T.; Brönstrup, M.; Hammann, P.; Halby, L.; Arimondo, P.B.; Glaser, P.; Aigle, B.; Bode, H.B. Towards the sustainable discovery and development of new antibiotics. Nat. Rev. Chem. 2021, 5, 726–749. [Google Scholar] [CrossRef]
- Ahmad, A.; Ullah, F.; Sadiq, A.; Ayaz, M.; Rahim, H.; Rashid, U.; Ahmad, S.; Jan, M.S.; Ullah, R.; Shahat, A.A. Pharmacological evaluation of aldehydic-pyrrolidinedione against HCT-116, MDA-MB231, NIH/3T3, MCF-7 cancer cell lines, antioxidant and enzyme inhibition studies. Drug Des. Dev. Ther. 2019, 13, 4185. [Google Scholar] [CrossRef]
- Qayyum, M.I.; Ullah, S.; Rashid, U.; Sadiq, A.; Mahnashi, M.H.; Alshehri, O.M.; Jalal, M.M.; Alzahrani, K.J.; Halawani, I.F. Synthesis, Molecular Docking, and Preclinical Evaluation of a New Succinimide Derivative for Cardioprotective, Hepatoprotective and Lipid-Lowering Effects. Molecules 2022, 27, 6199. [Google Scholar] [CrossRef]
- Genovese, M.; Nesi, I.; Caselli, A.; Paoli, P. Natural α-glucosidase and protein tyrosine phosphatase 1B inhibitors: A source of scaffold molecules for synthesis of new multitarget antidiabetic drugs. Molecules 2021, 26, 4818. [Google Scholar] [CrossRef] [PubMed]
- Huneif, M.A.; Alqahtani, S.M.; Abdulwahab, A.; Almedhesh, S.A.; Mahnashi, M.H.; Riaz, M.; Ur-Rahman, N.; Jan, M.S.; Ullah, F.; Aasim, M. α-glucosidase, α-amylase and antioxidant evaluations of isolated bioactives from wild strawberry. Molecules 2022, 27, 3444. [Google Scholar] [CrossRef] [PubMed]
- Al-Joufi, F.A.; Jan, M.; Zahoor, M.; Nazir, N.; Naz, S.; Talha, M.; Sadiq, A.; Nawaz, A.; Khan, F.A. Anabasis articulata (Forssk.) Moq: A good source of phytochemicals with antibacterial, antioxidant, and antidiabetic potential. Molecules 2022, 27, 3526. [Google Scholar] [CrossRef]
- Newsholme, P.; Keane, K.N.; Carlessi, R.; Cruzat, V. Oxidative stress pathways in pancreatic β-cells and insulin-sensitive cells and tissues: Importance to cell metabolism, function, and dysfunction. Am. J. Physiol.-Cell Physiol. 2019, 317, C420–C433. [Google Scholar] [CrossRef] [PubMed]


| Test Sample/Standard | IC50 (µM) ± SEM | Antioxidant (DPPH) IC50 (µM) | |||
|---|---|---|---|---|---|
| PTP1 B | α-Glucosidase | α-Amylase | DPP4 | ||
| Compound 3 | 13.20 ± 0.11 | 7.20 ± 0.92 | 15.40 ± 0.15 | 0.07 ± 0.03 | 3.31 ± 0.65 |
| Ursolic acid | 2.77 ± 0.93 | ---- | ---- | ---- | ---- |
| Sitagliptin | ---- | ---- | ---- | 0.005 ± 0.03 | ---- |
| Acarbose | ---- | 8.76 ± 0.60 | 12.30 ± 0.90 | ---- | ---- |
| Ascorbic acid | 1.44 ± 0.20 | ||||
| Sample/Standard | Dose (IP) | Blood Glucose Level (mmole per Liter) | |||
|---|---|---|---|---|---|
| 0 Day | 7th Day | 14th Day | 21st Day | ||
| Group-I (NS) | 4.29 | 4.24 | 4.28 | 4.27 | |
| Group-II (Tween 80) | 21.3 | 21.2 | 21.6 | 21.7 | |
| Group III (Glibenclamide) | 250 µM/kg | 21.5 | 17.7 | 14.5 | 11.1 |
| Group-IV (Compound 3) | 250 µM/kg | 22.4 | 18.4 | 15.7 | 12.2 |
| 125 µM/kg | 22.6 | 18.9 | 16.2 | 12.8 | |
| 62.5 µM/kg | 23.2 | 19.3 | 16.7 | 13.1 | |
| 31.25 µM/kg | 22.9 | 19.1 | 17.1 | 13.6 | |
| 15.625 µM/kg | 22.7 | 19.7 | 17.6 | 15.1 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahnashi, M.H.; Alam, W.; Huneif, M.A.; Abdulwahab, A.; Alzahrani, M.J.; Alshaibari, K.S.; Rashid, U.; Sadiq, A.; Jan, M.S. Exploration of Succinimide Derivative as a Multi-Target, Anti-Diabetic Agent: In Vitro and In Vivo Approaches. Molecules 2023, 28, 1589. https://doi.org/10.3390/molecules28041589
Mahnashi MH, Alam W, Huneif MA, Abdulwahab A, Alzahrani MJ, Alshaibari KS, Rashid U, Sadiq A, Jan MS. Exploration of Succinimide Derivative as a Multi-Target, Anti-Diabetic Agent: In Vitro and In Vivo Approaches. Molecules. 2023; 28(4):1589. https://doi.org/10.3390/molecules28041589
Chicago/Turabian StyleMahnashi, Mater H., Waqas Alam, Mohammed A. Huneif, Alqahtani Abdulwahab, Mohammed Jamaan Alzahrani, Khaled S. Alshaibari, Umar Rashid, Abdul Sadiq, and Muhammad Saeed Jan. 2023. "Exploration of Succinimide Derivative as a Multi-Target, Anti-Diabetic Agent: In Vitro and In Vivo Approaches" Molecules 28, no. 4: 1589. https://doi.org/10.3390/molecules28041589
APA StyleMahnashi, M. H., Alam, W., Huneif, M. A., Abdulwahab, A., Alzahrani, M. J., Alshaibari, K. S., Rashid, U., Sadiq, A., & Jan, M. S. (2023). Exploration of Succinimide Derivative as a Multi-Target, Anti-Diabetic Agent: In Vitro and In Vivo Approaches. Molecules, 28(4), 1589. https://doi.org/10.3390/molecules28041589







