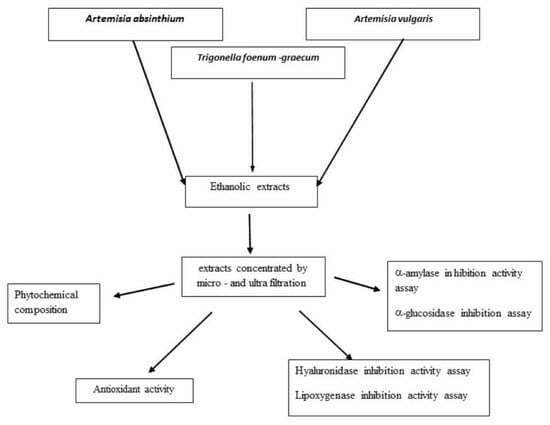
In Vitro Assessment of the Antidiabetic and Anti-Inflammatory Potential of Artemisia absinthium, Artemisia vulgaris and Trigonella foenum-graecum Extracts Processed Using Membrane Technologies
Abstract
:1. Introduction
2. Results and Discussion
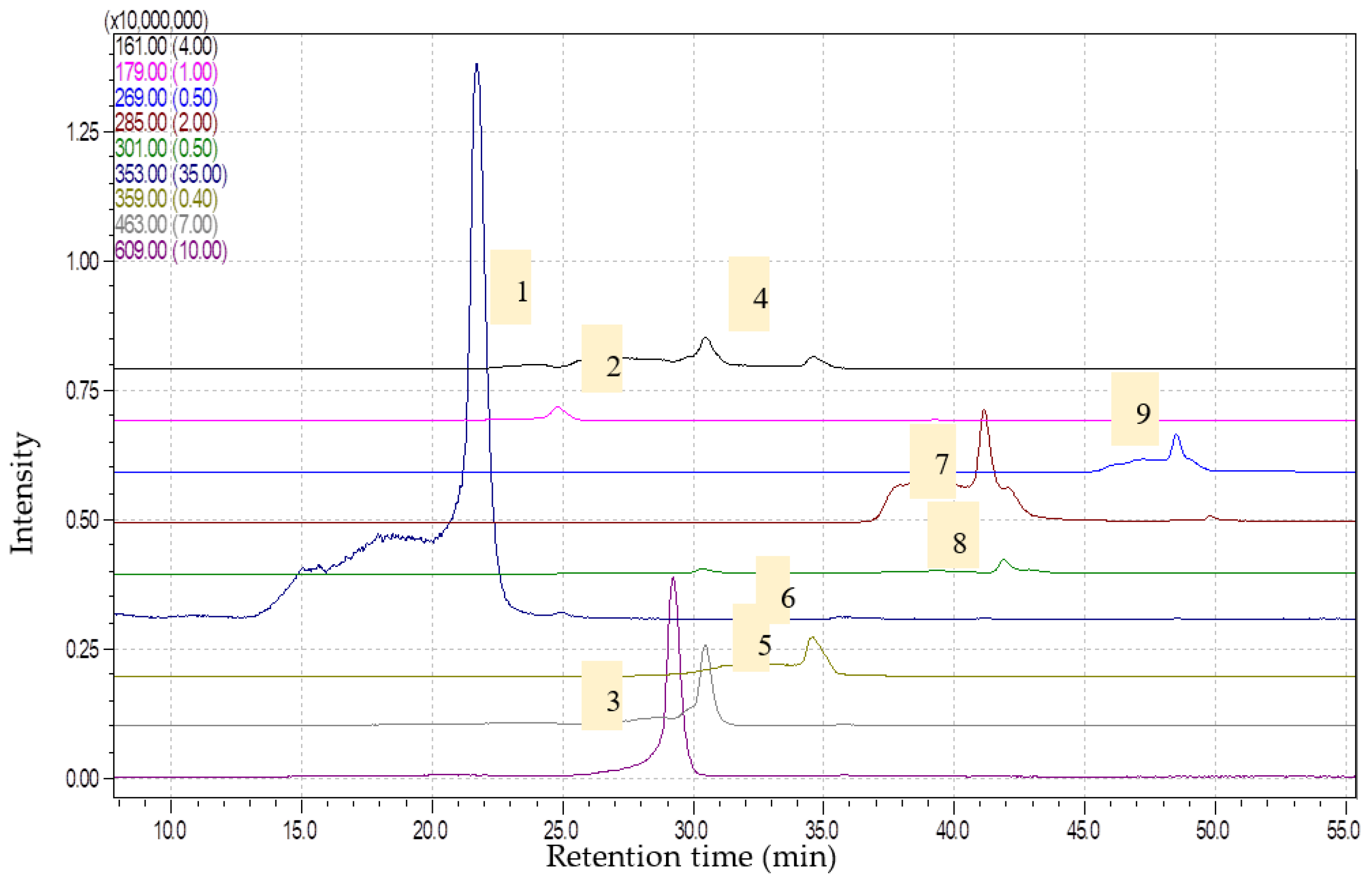
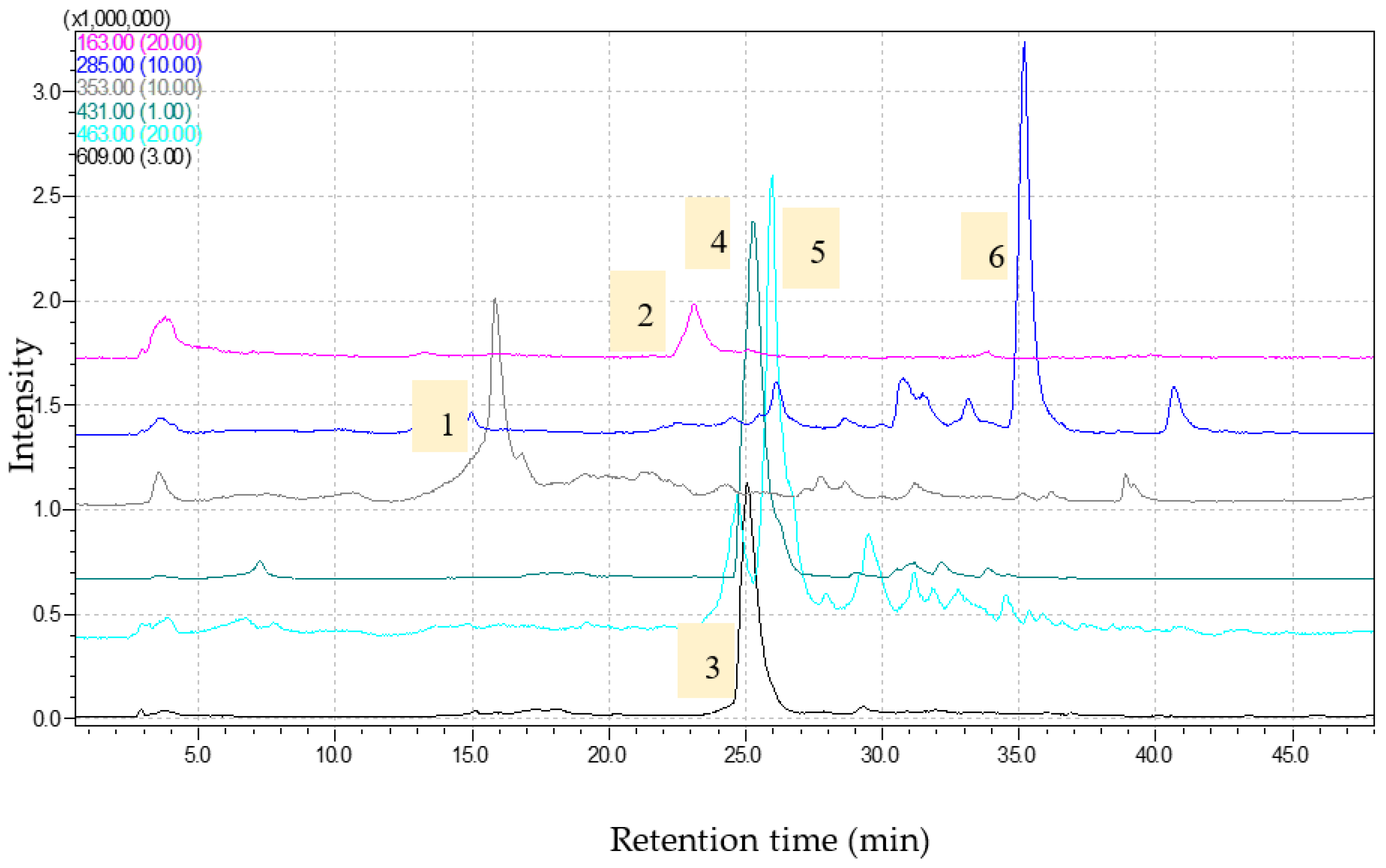
2.1. Phytochemical Analysis and Antioxidant Capacity
2.2. α-Amylase and α-Glucosidase Inhibition Activity
2.3. Lipoxygenase (LOX) and Hyaluronidase (HYA) Inhibition Activity
3. Materials and Methods
3.1. Chemicals
3.2. Obtaining the Extracts
3.3. Bioactive Compounds Determination
3.4. Antioxidant Assays
- The method was based on decreasing the maximum absorbance of ABTS to 731 nm in the presence of the antioxidant [63]; antioxidant activity was expressed in TEAC equivalents (Trolox Equivalent Antioxidant Capacity) using the formulawhere Ablank = control absorbance; ATrolox = Trolox absorbance. Asample = sample absorbance; f = dilution factor; CTrolox = Trolox concentration.
- 2.
- DPPH radical scavenging activity
- ○
- Reducing Power Activity (Iron (III) to iron (II) reduction)
3.5. Enzyme Inhibitory Activity Assay
3.5.1. Testing the Antidiabetic Capacity of the Extracts
- α Amylase inhibition assay
- α-Glucosidase inhibition assay
3.5.2. Testing the Anti-Inflammatory Capacity of the Extracts
- ○
- Hyaluronidase inhibition assay
- ○
- Lipoxygenase inhibition assay
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- IDF (International Diabetes Federation). IDF Diabetes Atlas, 8th ed.; 2017; Available online: https://www.idf.org/e-library/epidemiology-research/diabetes-atlas.html (accessed on 14 November 2017).
- Ogurtsova, K.; Da Rocha Fernandes, J.D.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.H.; Makaroff, L.E. IDF diabetes atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef]
- Vadivelan, R.; Krishnan, R.G.; Kannan, R. Antidiabetic potential of Asparagus racemosus, Willd leaf extracts through inhibition of a-amylase and a-glucosidase. J. Trad. Complement Med. 2019, 9, 1–4. [Google Scholar] [CrossRef]
- Chester, K.; Zahiruddin, S.; Ahmad, A.; Khan, W.; Paliwal, S.; Ahmad, S. Anti-inflammatory activity of total alkaloids from hypecoum leptocarpum hook f. et Thoms. Pharmacogn. Mag. 2017, 13, 179–188. [Google Scholar]
- Bribi, N.; Algieri, F.; Rodriguez-Nogales, A.; Garrido-Mesa, J.; Vezza, T.; Maiza, F.; Utrilla, M.P.; Rodriguez-Cabezas, M.E.; Galvez, J. Antinociceptive and anti-inflammatory effects of total alkaloid extract from fumaria capreolata. Evid. Based Complement Altern. Med. 2015, 2015, 1–7. [Google Scholar] [CrossRef]
- Hermanto, F.E.; Soewondo, A.; Tsuboi, H.; Ibrahim, M.; Rifa’i, M. The hepatoprotective effect of Cheral as anti-oxidant and anti-inflammation on mice (Mus musculus) with breast cancer. J. Herbmed. Pharmacol. 2020, 9, 153–160. [Google Scholar] [CrossRef]
- Girish, K.S.; Kemparaju, K.; Nagaraju, S.; Vishwanath, B.S. Hyaluronidase inhibitors: A biological and therapeutic perspective. Curr. Med. Chem. 2009, 16, 2261–2288. [Google Scholar] [CrossRef] [PubMed]
- Loncaric, M.; Strelec, I.; Moslavac, T.; Subaric, D.; Pavic, V.; Molnar, M. Lipoxygenase inhibition by plant extracts. Biomolecules 2021, 11, 152. [Google Scholar] [CrossRef] [PubMed]
- González-Peña, D.; Colina-Coca, C.; Char, C.D.; Cano, M.P.; Ancos, B.; Sánchez-Moreno, C. Hyaluronidase inhibiting activity and radical scavenging potential of flavonols in processed onion. J. Agric. Food Chem. 2013, 61, 4862–4872. [Google Scholar] [CrossRef] [PubMed]
- Sarmiento-Salinas, F.L.; Perez-Gonzalez, A.; Acosta-Casique, A.; Ix-Ballote, A.; Diaz, A.; Treviño, S.; Rosas-Murrieta, N.H.; Millán-Perez-Peña, L.M.; Maycotte, P. Reactive oxygen species: Role in carcinogenesis, cancer cell signaling and tumor progression. Life Sci. 2021, 284, 119942. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, D.; Freitas, M.; Tomé, S.M.; Silva, A.M.; Porto, G.; Cabrita, E.J.; Marques, M.; Manuel, B.; Fernandes, E. Inhibition of LOX by flavonoids: A structure activity relationship study. Eur. J. Med. Chem. 2014, 72, 137–145. [Google Scholar] [CrossRef]
- Sudha, P.N.; Rose, M.H. Beneficial effects of hyaluronic acid. Adv. Food Nutr. Res. 2014, 72, 137–176. [Google Scholar] [PubMed]
- Ölgen, S.; Kaessler, A.; Zühal Kiliç-Kurt, Z.; Jose, J. Investigation of aminomethyl indole derivatives as hyaluronidase inhibitors. Z. Für Naturfors C 2014, 65, 445–450. [Google Scholar] [CrossRef]
- Michalea, R.; Stathopoulou, K.; Panagiotisa, P.; Dimitrab, B.; Emmanuel, M. Efficient identification of Acetylcholinesterase and Hyaluronidase inhibitors from Paeonia parnassica extracts through a Hetero Covariance Approach, Aligiannis Nektariosa. J. Ethnopharmacol. 2020, 257, 111547. [Google Scholar] [CrossRef]
- Bazinet, L.; Doyen, A. Antioxidants, mechanisms, and recovery by membrane processes. Crit. Rev. Food Sci. Nutr. 2017, 57, 677–700. [Google Scholar] [CrossRef] [PubMed]
- Conidi, C.; Drioli, E.; Cassano, A. Membrane-based agro-food production processes for polyphenol separation, purification and concentration. Curr. Opin. Food Sci. 2018, 17, 149–164. [Google Scholar] [CrossRef]
- Gerke, I.B.; Hamerski, F.; Scheer, A.P.; Silva, V.R. Clarification of crude extract of yerba mate (Ilex paraguariensis) by membrane processes: Analysis of fouling and loss of bioactive compounds. Food Bioprod. Process. 2017, 102, 204–212. [Google Scholar] [CrossRef]
- Dos Santos, L.F.; Vargas, B.K.; Bertol, C.D.; Biduski, B.; Bertolina, T.E.; Dos Santos, L.R.; Brião, V.B. Clarification and concentration of yerba mate extract by membrane technology to increase shelf life. Food Bioprod. Process. 2020, 122, 22–30. [Google Scholar] [CrossRef]
- Pandey, A.K.; Singh, P. The genus artemisia: A 2012–2017 literature review on chemical composition, antimicrobial, insecticidal and antioxidant activities of essential oils. Medicines 2017, 4, 68. [Google Scholar] [CrossRef]
- Ekiert, H.; Pajor, J.; Klin, P.; Rzepiela, A.; Ślesak, H.; Szopa, A. Significance of Artemisia vulgaris L. (common Mugwort) in the history of medicine and its possible contemporary applications substantiated by phytochemical and pharmacological studies. Molecules 2020, 25, 4415. [Google Scholar] [CrossRef]
- Taleghani, A.; Emami, S.A.; Tayarani-Najaran, Z. Artemisia: A promising plant for the treatment of cancer. Bioorg. Med. Chem. 2020, 28, 10. [Google Scholar] [CrossRef]
- Ben Nasr, S.; Aazza, S.; Mnif, W.; Miguel, M. In-vitro antioxidant and anti-inflamatory activities of Pituranthos chloranthus and Artemisia vulgaris from Tunisia. Int. J. Appl. Pharm. Sci. Res. 2020, 11, 605–614. [Google Scholar]
- Liu, T.; Chen, X.; Hu, Y.; Li, M.; Wu, Y.; Dai, M.; Huang, Z.; Sun, P.; Zheng, J.; Ren, Z.; et al. Sesquiterpenoids and triterpenoids with anti-inflammatory effects from Artemisia vulgaris L. Phytochemistry 2022, 204, 113428. [Google Scholar] [CrossRef]
- Ahamad, J.; Mir, S.R.; Amin, S. A pharmacognostic review on Artemisia absinthium. Int. Res. J. Pharm. 2019, 10, 25–31. [Google Scholar] [CrossRef]
- Szopa, A.; Pajor, J.; Klin, P.; Rzepiela, A.; Elansary, H.O.; Al-Mana, F.A.; Mattar, M.A.; Ekiert, H. Artemisia absinthium L.—Importance in the history of medicine, the latest advances in phytochemistry and therapeutical, cosmetological and culinary uses. Plants 2020, 9, 1063. [Google Scholar] [CrossRef] [PubMed]
- Al-Malki, A.L. Shikimic acid from Artemisia absinthium inhibits protein glycation in diabetic rats. Int. J. Biol. Macromol. 2019, 122, 1212–1216. [Google Scholar] [CrossRef]
- Srivastava, R.; Srivastava, P. Hepatotoxicity and the role of some herbal hepatoprotective plants in present scenario. Glob. J. Dig. Dis. 2018, 3. [Google Scholar] [CrossRef]
- Singh, N.; Yadav, S.S.; Kumar, S.; Narashiman, B. Ethnopharmacological, phytochemical and clinical studies on Fenugreek (Trigonella foenum-graecum L.). Food Biosci. 2022, 46, 101546. [Google Scholar] [CrossRef]
- Ruwali, P.; Pandey, N.; Jindal, K.; Singh, R.V. Fenugreek (Trigonella foenum-graecum): Nutraceutical values, phytochemical, ethnomedicinal and pharmacological overview. S. Afr. J. Bot. 2022, 151, 423–431. [Google Scholar] [CrossRef]
- Singh, P.; Bajpai, V.; Gond, V.; Kumar, A.; Tadigoppula, N.; Kumar, B. Determination of bioactive compounds of fenugreek (Trigonella foenum-graecum) seeds using LC-MS techniques. Methods Mol. Biol. 2020, 2107, 377–393. [Google Scholar]
- Chandrasekara, A.; Shahidi, F. Determination of antioxidant activity in free and hydrolyzed fractions of millet grains and characterization of their phenolic profiles by HPLC-DAD-ESI-MSn. J. Funct. Foods 2011, 3, 144–158. [Google Scholar] [CrossRef]
- Seasotiya, L.; Siwach, P.; Bai, S.; Malik, A.; Bharti, P.; Dalal, S. Free radical scavenging activity, phenolic contents and phytochemical analysis of seeds of Trigonella foenum graecum. Asian Pac. J. Health Sci. 2014, 1, 219–226. [Google Scholar]
- Mohammad, A. Light-induced fluctuations in biomass accumulation, secondary metabolites production and antioxidant activity in cell suspension cultures of Artemisia absinthium L. J. Photochem. Photobiol. B Biol. 2014, 140, 223–227. [Google Scholar]
- Nguyen, H.T.; Radacsi, P.; Gosztola, B.; Nemeth, E.Z. Effects of temperature and light intensity on morphological and phytochemical characters and antioxidant potential of wormwood (Artemisia absinthium L.). Biochem. Sys. Ecol. 2018, 79, 1–7. [Google Scholar] [CrossRef]
- Lee, Y.J.; Thiruvengadam, M.; Chung, I.M.; Nagella, P. Polyphenol composition and antioxidant activity from the vegetable plant Artemisia absinthium L. Aust. J. Crop Sci. 2013, 7, 1921–1926. [Google Scholar]
- Ferreira, J.F.; Luthria, D.L.; Sasaki, T.; Heyerick, A. Flavonoids from Artemisia annua L. as antioxidants and their potential synergism with artemisinin against malaria and cancer. Molecules 2010, 15, 3135–3170. [Google Scholar] [CrossRef]
- Casazza, A.A.; Aliakbarian, B.; De Faveri, D.; Fiori, L.; Perego, P. Antioxidants from winemaking wastes: A study on extraction parameters using response surface methodology. J. Food Biochem. 2012, 36, 28–37. [Google Scholar] [CrossRef]
- Aliakbarian, B.; Fathi, A.; Perego, P.; Dehghani, F. Extraction of antioxidants from winery wastes using subcritical water. J. Supercrit. Fluids 2012, 65, 18–24. [Google Scholar] [CrossRef]
- Melguizo-Melguizo, D.; Diaz-de-Cerio, E.; Quirantes-Piné, R.; Švarc-Gajic´, J.; Segura-Carretero, A. The potential of Artemisia vulgaris leaves as a source of antioxidant phenolic compounds. J. Func. Foods 2014, 10, 192–200. [Google Scholar] [CrossRef]
- Haniya, A.K.; Padma, P. Phytochemical investigation of methanolic extract of Artemisia vulgaris L. leaves. Int. J. Pharma Bio Sci. 2014, 5, 184–195. [Google Scholar]
- Orhan, E.I.; Belhattab, R.; Şenol, F.S.; Gülpinar, A.R.; Hoşbaş, S.; Kartal, M. Profiling of cholinesterase inhibitory and antioxidant activities of Artemisia absinthium, A. herba-alba, A. fragrans, Marrubium vulgare, M. astranicum, Origanum vulgare subsp. glandulossum and essential oil analysis of two Artemisia species. Ind. Crops Prod. 2010, 32, 566–571. [Google Scholar] [CrossRef]
- Craciunescu, O.; Constantin, D.; Gaspar, A.; Toma, L.; Utoiu, E.; Moldovan, L. Evaluation of antioxidant and cytoprotective activities of Arnica montana L. and Artemisia absinthium L. ethanolic extracts. Chem. Cent. J. 2012, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Magalingam, K.B.; Radhakrishnan, A.K.; Haleagrahara, N. Protective mechanisms of flavonoids in Parkinson’s disease. Oxid. Med. Cell. Longev. 2015, 2015, 314560. [Google Scholar] [CrossRef] [PubMed]
- Bondonno, N.P.; Dalgaard, F.; Kyrø, C.; Murray, K.; Bondonno, C.P.; Lewis, J.R.; Croft, K.D.; Gislason, G.; Scalbert, A.; Cassidy, A.; et al. Flavonoid intake is associated with lower mortality in the Danish Diet Cancer and Health Cohort. Nat. Commun. 2019, 10, 3651. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Sang, W.; Zhou, M.; Ren, G. Antioxidant and a-glucosidase inhibitory activity of colored grains in China. J. Agric. Food Chem. 2010, 58, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Cruz, E.C.; Andrade-Cetto, A. Ethnopharmacological field study of the plants used to treat type 2 diabetes among the Cakchiquels in Guatemala. J. Ethnopharmacol. 2015, 159, 238–244. [Google Scholar] [CrossRef]
- Awad, N.E.; Seida, A.A.; Shaffie, Z.E.N.; El-Aziz, A.M. Hypoglycemic activity of Artemisia herba-alba (Asso.) used in Egyptian traditional medicine as hypoglycemic remedy. J. Appl. Pharm. Sci. 2012, 2, 30–39. [Google Scholar]
- Ahmad, W.I.; Khan, M.A.; Khan, M.; Ahmad, F.; Subhan, F.; Karim, N. Evaluation of antidiabetic and antihyperlipidemic activity of Artemisia indica linn (aeriel parts) in Streptozotocin induced diabetic rats. J. Ethnopharmacol. 2014, 151, 618–623. [Google Scholar] [CrossRef]
- Ghazanfar, K.; Ganai, B.A.; Akbar, S.; Mubashir, K.; Dar, S.A.; Dar, M.Y.; Tantry, M.A. Antidiabetic activity of Artemisia amygdalina Decne in streptozotocin induced diabetic rats. BioMed Res. Int. 2014, 2014, 185676. [Google Scholar] [CrossRef]
- Nathar, V.N.; Yatoo, G.M. Micropropagation of an antidiabetic medicinal plant Artemisia pallens. Turk. J. Bot. 2014, 38, 491–498. [Google Scholar] [CrossRef]
- Herrera, T.; Del Hierro, J.N.; Fornari, T.; Reglero, G.; Martin, D. Inhibitory effect of quinoa and fenugreek extracts on pancreatic lipase and α-amylase under in vitro traditional conditions or intestinal simulated conditions. Food Chem. 2019, 270, 509–517. [Google Scholar] [CrossRef]
- Alamgeer Uttra, A.M.; Ahsan, H.; Hasan, U.H.; Chaudhary, M.A. Traditional medicines of plant origin used for the treatment of in-flammatory disorders in Pakistan: A review. J. Tradit. Chin. Med. 2018, 38, 636–656. [Google Scholar] [CrossRef]
- Afsar, S.; Kumar, K.R.; Gopal, J.V.; Raveesha, P. Assessment of anti-inflammatory activity of Artemisia vulgaris leaves by cotton pellet granuloma method in Wistar albino rats. J. Pharm. Res. 2013, 7, 463–467. [Google Scholar] [CrossRef]
- Arundina, I.; Budhy, S.T.I.; Nirwanaa, I.; Indrawatia, R.; Luthfi, M. Toxicity Test n-Hexane: Ethyl Acetate (3:7) Fraction of Sudamala (Artemisia vulgaris L.) Molecular and Cellular Life Sciences: Infectious Diseases, Biochemistry and Structural Biology 2015 Conference, MCLS 2015. Procedia Chem. 2016, 18, 174–178. [Google Scholar] [CrossRef]
- Dib, I.; El Alaoui-Faris, E.F. Artemisia campestris L.: Review on taxonomical aspects, cytogeography, biological activities and bioactive compounds. Biomed. Pharmacother. 2019, 109, 1884–1906. [Google Scholar] [CrossRef]
- Parameswari, P.; Devika, R. Phytochemical screening of bioactive compounds of Artemisia nilagirica (clarke) pamp. J. Chem. Pharmaceut. Sci. 2014, 7, 351–353. [Google Scholar]
- Farahani, Z.B.; Mirzaie, A.; Ashrafi, F.; Hesari, M.R.; Chitgar, A.; Noorbazargan, H.; Rahimi, A. Phytochemical composition and biological activities of Artemisia quettensis Podlech ethanolic extract. Nat. Prod. Res. 2017, 31, 2554–2558. [Google Scholar] [CrossRef]
- Khan, N.; Ahmed, M.; Ahmed, A.; Shaukat, S.S.; Wahab, M.; Ajaib, M.; Nasir, M. Important medicinal plants of Chitral Gol National Park (CGNP) Pakistan. Pak. J. Bot. 2011, 43, 797–809. [Google Scholar]
- Bano, A.; Ahmad, M.; Hadda, T.B.; Saboor, A.; Sultana, S.; Zafar, M.; Khan, M.P.Z.; Arshad, M.; Ashraf, M.A. Quantitative ethno-medicinal study of plants used in the Skardu valley at high altitude of Karakoram-Himalayan range, Pakistan. J. Ethnobiol. Ethnomed. 2014, 10, 43. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substances by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Lin, J.-Y.; Tang, C.-Y. Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chem. 2007, 101, 140–147. [Google Scholar] [CrossRef]
- Alecu, A.; Albu, C.; Litescu, S.C.; Eremia, S.A.V.; Radu, G.L. Phenolic Anthocyanin Profile of Valea Calugareasca Red Wines by HPLC-PDA-MS, MALDI-TOF Analysis. Food Analyt. Meth. 2016, 9, 300–310. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure–antioxidant activity relationships of flavonoids and phenolic acids. Free Radical Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Bondet, V.; Brand-Williams, W.; Berset, C. Kinetics and mechanism of antioxidant activity using the DPPH free radical method. Lebensm. Wiss Technol. 1997, 30, 609–615. [Google Scholar] [CrossRef]
- Berker, K.; Guclu, K.; Tor, I.; Apak, R. Comparative evaluation of Fe (III) reducing power-based antioxidant capacity assays in the presence of phenanthroline, batho-phenanthroline, tripyridyltriazine (FRAP) and ferricyanide reagents. Talanta 2007, 72, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Ranilla, L.G.; Kwon, Y.I.; Apostolidis, E.; Shetty, K. Phenolic compounds, antioxidant activity and in vitro inhibitory potential against key enzymes relevant for hyperglycemia and hypertension of commonly used medicinal plants, herbs and spices in Latin America. Bioresour. Technol. 2010, 101, 4676–4689. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, D.P.K.; Ferreira, A.G.; Lima, A.S.; Lima, E.S.; Lima, M.P. Isolation and identification of α-glucosidase, α-amylase and lipase inhibitors from hortia longifolia. Int. J. Pharm. Pharm. Sci. 2013, 5, 336–339. [Google Scholar]
- Sahasrabudhe, A.; Deodhar, M. Anti-hyaluronidase, antielastase activity of Garcinia indica. Int. J. Bot. 2010, 6, 299–303. [Google Scholar] [CrossRef]
- Hamberg, M.; Samuelsson, B. On the specificity of the oxygenation of unsaturated fatty acids catalyzed by soybean lipoxidase. J. Biol. Chem. 1967, 242, 5329–5335. [Google Scholar] [CrossRef]

| Sample | Polyphenol Content (mg CA/mL) | Flavone Content (mg RE/mL) | Reducing Power % | % DPPH Inhibition | TEAC_ABTS mg/mL | |
|---|---|---|---|---|---|---|
| Artemisia absinthium | MF | 3232.5 ± 140.3 | 389.1 ± 9.5 | 50.7 ± 1.3 | 36.1 ± 1.1 | 204.1 ± 6.3 |
| concentrate | 4777.5 ± 125.5 | 501.8 ± 26.9 | 73.5 ± 2.3 | 65.9 ± 2.3 | 483.2 ± 3.8 | |
| Artemisia vulgaris | MF | 7877.5 ± 260.9 | 505.2 ± 30.1 | 52.9 ± 1.3 | 63.6 ± 2.5 | 316.5 ± 9.5 |
| concentrate | 11,440.2 ± 49.9 | 1020.1 ± 45.1 | 93.1 ± 3.2 | 77.5 ± 2.4 | 541.5 ± 15.3 | |
| Trigonella foenum-graecum | MF | 1094.3 ± 62.4 | 395.8 ± 11.6 | 63.8 ± 2.4 | 75.4 ± 5.8 | 420.7 ± 17.3 |
| concentrate | 2636.8 ± 52.7 | 777.6 ± 2.7 | 97.1 ± 5.2 | 82.2 ± 3.7 | 591.2 ± 22.3 | |
| Ascorbic acid | 34.5 ± 1.3 | 96.9 ± 3.6 | ||||
| Compound | A. absinthium MF µg/mL | A. absinthium Concentrate µg/mL | A. vulgaris MF µg/mL | A. vulgaris Concentrate µg/mL | T. foenum-graecum MF µg/mL | T. foenum-graecum Concentrate µg/mL |
|---|---|---|---|---|---|---|
| Chlorogenic acid | 259.4 | 297.9 | 301.1 | 350.6 | 11.1 | 68.1 |
| Caffeic acid | 2.6 | 3.2 | 2.4 | 3.1 | - | - |
| Rosmarinic acid | 21.0 | 22.6 | 3.3 | 3.1 | - | - |
| Coumaric acid | - | - | - | - | 7.3 | 7.7 |
| Umbelifferone | 6.9 | 7.9 | 7.1 | 8.9 | - | - |
| Quercetol | 0.6 | 0.7 | 0.8 | 0.1 | - | - |
| Luteolin | 4.7 | 5.5 | 12.4 | 14.5 | 3.5 | 3.9 |
| Apigenin | 0.7 | 1.1 | 2.5 | 2.6 | - | - |
| Rutin | 12.6 | 15.6 | 18.4 | 19.6 | u.d.l | 84.9 |
| Ellagic acid | 4.7 | 5.1 | - | - | - | - |
| Isoquercitrin | 1.5 | 1.9 | 3.7 | 3.7 | 7.3 | 20.1 |
| Genistin | - | - | - | - | 1286.2 | 2032.9 |
| Samples | Inhibition of α-Amylase IC50 (μg/mL) | Inhibition of α-Glucosidase IC50 (μg/mL) | |
|---|---|---|---|
| A. absinthium extracts | MF | 22.2 ± 0.9 | 45.16 ± 1.4 |
| concentrate | 19.4 ± 0.5 | 31.90 ± 1.8 | |
| A. vulgaris extracts | MF | 17.0 ± 0.9 | 96.04 ± 3.2 |
| concentrate | 8.5 ± 2.3 | 77.13 ± 2.3 | |
| T. foenum-graecum extracts | MF | 24.1 ± 1.4 | 28.19 ± 1.8 |
| concentrate | 3.2 ± 0.3 | 11.14 ± 0.9 | |
| Rosmarinic acid | 0.9 ± 0.1 | 0.18 ± 0.1 | |
| Chlorogenic acid | 1.9 ± 0.1 | 0.59 ± 0.2 | |
| Acarbose | 3.5 ± 0.1 | 5.90 ± 0.3 | |
| Samples | Inhibition of HYA IC50 (μg/mL) | Inhibition of LOX IC50 (μg/mL) | |
|---|---|---|---|
| Artemisia absinthium extracts | MF | 78.1 ± 2.3 | 52.9 ± 2.1 |
| concentrate | 34.7 ± 1.2 | 19.7 ± 0.8 | |
| Artemisia vulgaris extracts | MF | 74.1 ± 2.6 | 182.4 ± 10.5 |
| concentrate | 17.2 ± 1.2 | 112.7 ± 8.5 | |
| T. foenum-graecum extracts | MF | 67.4 ± 2.6 | 31.1 ± 1.3 |
| concentrate | 17.6 ± 1.2 | 19.7 ± 0.5 | |
| Ibuprofen | 5.7 ± 0.2 | 20.2 ± 1.2 | |
| Rosmarinic acid | - | 30.3 ± 1.2 | |
| Chlorogenic acid | - | 26.1 ± 1.3 | |
| Rutin | - | 22.34 ± 1.9 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neagu, E.; Paun, G.; Albu, C.; Apreutesei, O.T.; Radu, G.L. In Vitro Assessment of the Antidiabetic and Anti-Inflammatory Potential of Artemisia absinthium, Artemisia vulgaris and Trigonella foenum-graecum Extracts Processed Using Membrane Technologies. Molecules 2023, 28, 7156. https://doi.org/10.3390/molecules28207156
Neagu E, Paun G, Albu C, Apreutesei OT, Radu GL. In Vitro Assessment of the Antidiabetic and Anti-Inflammatory Potential of Artemisia absinthium, Artemisia vulgaris and Trigonella foenum-graecum Extracts Processed Using Membrane Technologies. Molecules. 2023; 28(20):7156. https://doi.org/10.3390/molecules28207156
Chicago/Turabian StyleNeagu, Elena, Gabriela Paun, Camelia Albu, Oana Teodora Apreutesei, and Gabriel Lucian Radu. 2023. "In Vitro Assessment of the Antidiabetic and Anti-Inflammatory Potential of Artemisia absinthium, Artemisia vulgaris and Trigonella foenum-graecum Extracts Processed Using Membrane Technologies" Molecules 28, no. 20: 7156. https://doi.org/10.3390/molecules28207156
APA StyleNeagu, E., Paun, G., Albu, C., Apreutesei, O. T., & Radu, G. L. (2023). In Vitro Assessment of the Antidiabetic and Anti-Inflammatory Potential of Artemisia absinthium, Artemisia vulgaris and Trigonella foenum-graecum Extracts Processed Using Membrane Technologies. Molecules, 28(20), 7156. https://doi.org/10.3390/molecules28207156