Synthesis, Spectroscopic Characterization, Antibacterial Activity, and Computational Studies of Novel Pyridazinone Derivatives
Abstract
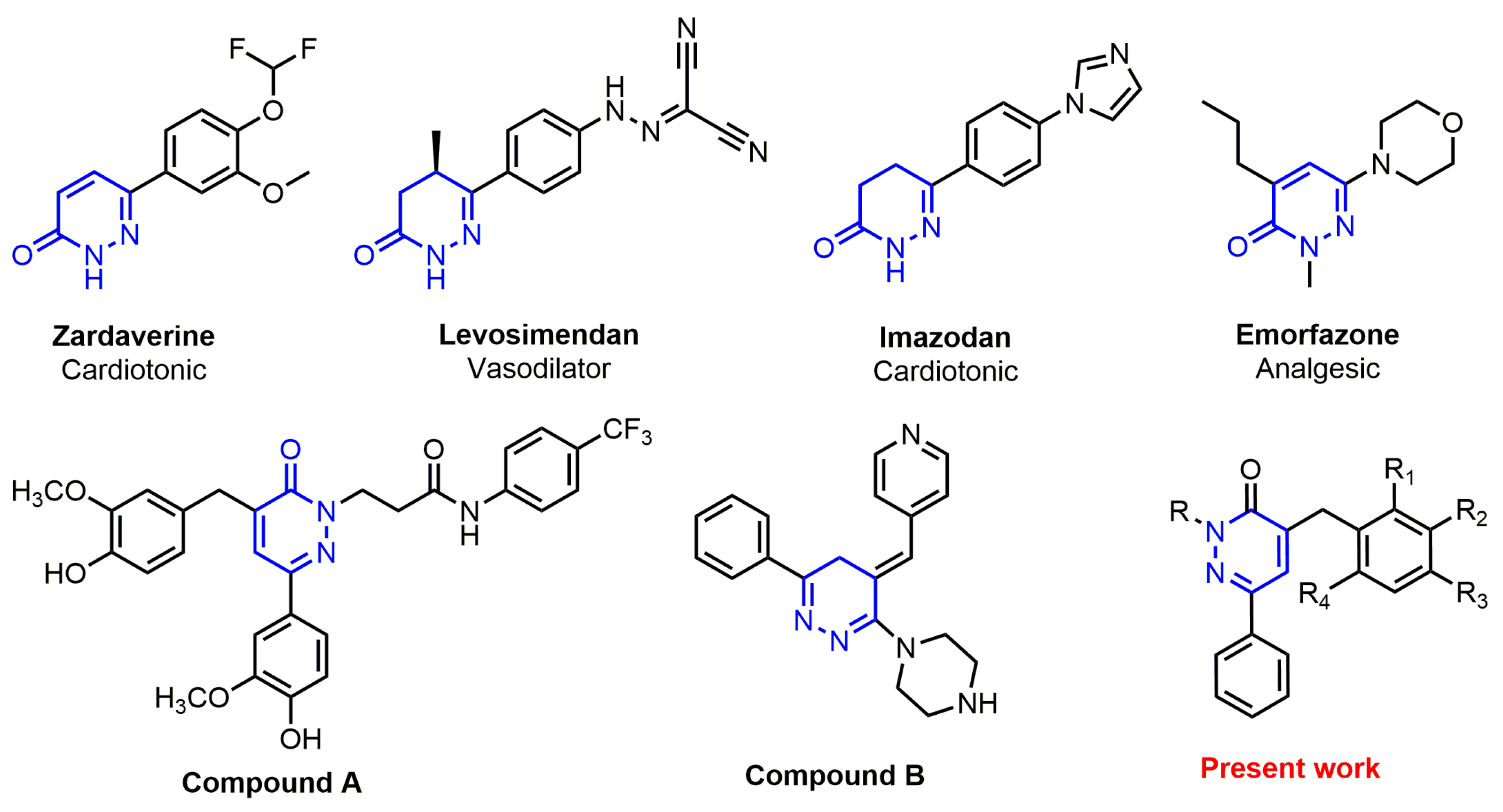
1. Introduction
2. Results and Discussion
2.1. Chemistry
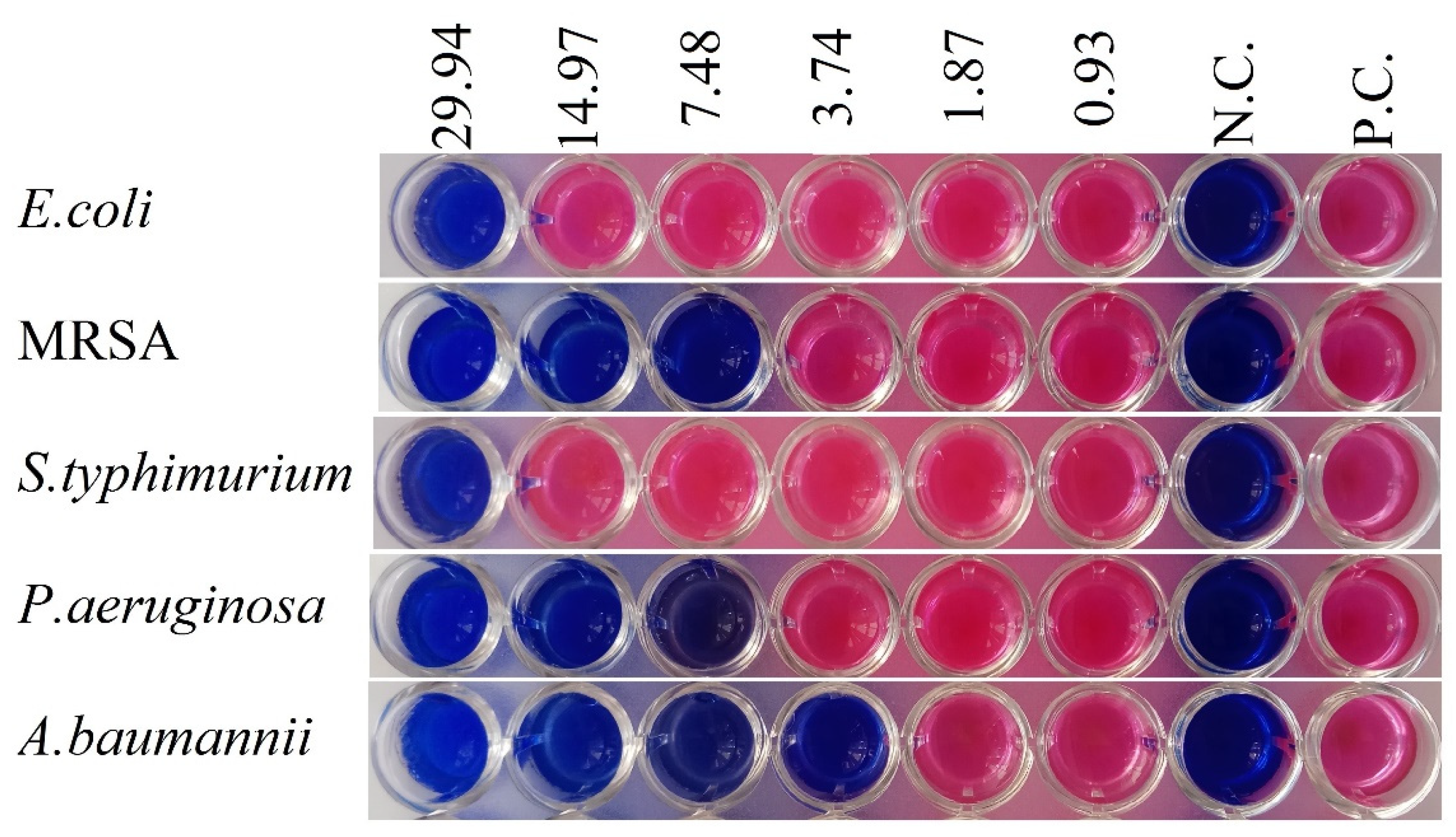
2.2. Antibacterial Activity
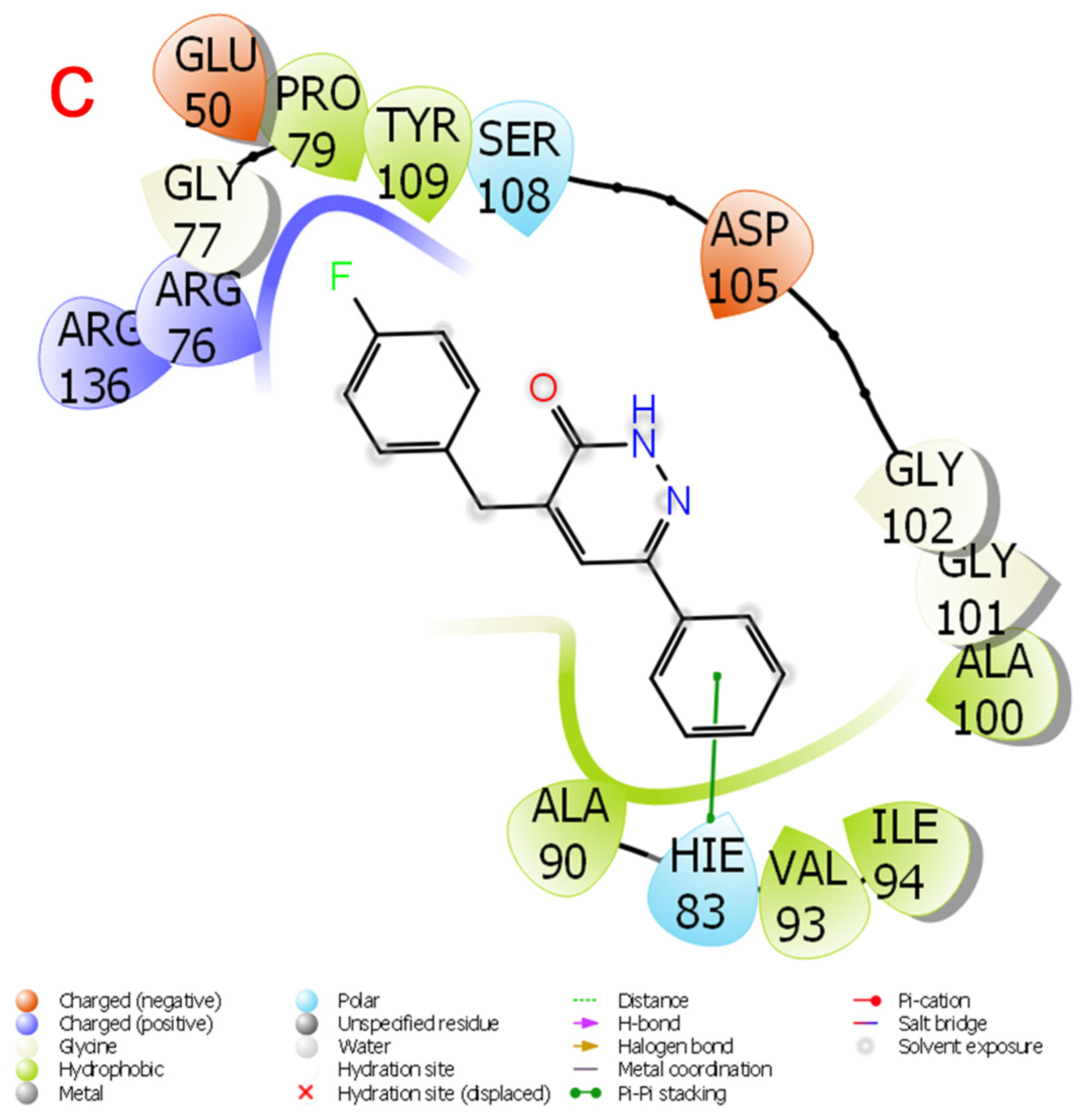
2.3. Theoretical Calculations
3. Materials and Methods
3.1. General Methods
3.2. Chemistry
3.2.1. General Procedure for the Synthesis of Compounds (3–7)
3.2.2. General Procedure for the Synthesis of Compounds (8–12)
3.2.3. General Procedure for the Synthesis of Compounds (13–17)
3.3. Theoretical Methods
3.4. Antibacterial Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADMET | Absorption, distribution, metabolism, excretion, and toxicity |
| B3LYP | Becke 3-parameter Lee–Yang–Parr |
| CLSI | Clinical and Laboratory Standards Institute |
| DFT | Density functional theory |
| ESI–MS | Electrospray ionisation–mass spectrometry |
| ESP | Electrostatic potential |
| FT-IR | Fourier-transform infrared |
| HOMO | Highest occupied molecular orbital |
| LUMO | Lowest unoccupied molecular orbital |
| MIC | Minimal inhibition concentration |
| MRSA | Methicillin-resistant Streptococcus aureus |
| NMR | Nuclear magnetic resonance |
| TBAB | Tetrabutylammonium bromide |
| THF | Tetrahydrofuran |
References
- Singh, S.R.; Krishnamurthy, N.B.; Mathew, B.B. A review on recent diseases caused by microbes. J. Appl. Environ. Microbiol. 2014, 2, 106–115. [Google Scholar]
- Jiang, Q.; Chen, J.; Yang, C.; Yin, Y.; Yao, K. Quorum sensing: A prospective therapeutic target for bacterial diseases. Biomed. Res. Int. 2019, 2019, 2015978. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, R.; Ahmed, S.A. Synthesis of some new thiazole, oxazole, pyrimidine and pyridazine derivatives from 2-cyano-N-octadecyl-acetamide as antimicrobial and surface active agents. J. Heter. Chem. 2016, 53, 121–128. [Google Scholar] [CrossRef]
- Mabkhot, Y.N.; Kaal, N.A.; Alterary, S.; Al-Showiman, S.S.; Farghaly, T.A.; Mubarak, M.S. Antimicrobial activity of thiophene derivatives derived from ethyl (E)-5-(3-(dimethylamino) acryloyl)-4-methyl-2-(phenylamino) thiophene-3-carboxylate. Chem. Cent. J. 2017, 11, 75. [Google Scholar] [CrossRef] [PubMed]
- Jacomini, A.P.; Silva, M.J.; Silva, R.G.; Gonçalves, D.S.; Volpato, H.; Basso, E.A.; Rosa, F.A. Synthesis and evaluation against Leishmania amazonensis of novel pyrazolo [3, 4-d] pyridazinone-N-acylhydrazone-(bi) thiophene hybrids. Eur. J. Med. Chem. 2016, 124, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, M.; Yao, X.; Li, Y.; Tan, J.; Wang, L.; Wang, Q. Design, synthesis and antiviral activity of novel pyridazines. Eur. J. Med. Chem. 2012, 54, 33–41. [Google Scholar] [CrossRef]
- Rafi, U.M.; Mahendiran, D.; Devi, V.G.; Doble, M.; Rahiman, A.K. Pyridazine-based heteroleptic copper (II) complexes as potent anticancer drugs by inducing apoptosis and S-phase arrest in breast cancer cell. Inorg. Chim. Acta 2018, 482, 160–169. [Google Scholar] [CrossRef]
- Mantu, D.; Luca, M.C.; Moldoveanu, C.; Zbancioc, G.; Mangalagiu, I.I. Synthesis and antituberculosis activity of some new pyridazine derivatives. Part II. Eur. J. Med. Chem. 2010, 45, 5164–5168. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, E.M.; Kassab, A.E.; El-Malah, A.A.; Hassan, M.S. Synthesis and biological evaluation of pyridazinone derivatives as selective COX-2 inhibitors and potential anti-inflammatory agents. Eur. J. Med. Chem. 2019, 171, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Barberot, C.; Moniot, A.; Allart-Simon, I.; Malleret, L.; Yegorova, T.; Laronze-Cochard, M.; Gérard, S. Synthesis and biological evaluation of pyridazinone derivatives as potential anti-inflammatory agents. Eur. J. Med. Chem. 2018, 146, 139–146. [Google Scholar] [CrossRef]
- Zerroug, A.; Belaidi, S.; BenBrahim, I.; Sinha, L.; Chtita, S. Virtual screening in drug-likeness and structure/activity relationship of pyridazine derivatives as Anti-Alzheimer drugs. J. King Saud Univ. Sci. 2019, 31, 595–601. [Google Scholar] [CrossRef]
- Demirayak, S.; Karaburun, A.C.; Beis, R. Some pyrrole substituted aryl pyridazinone and phthalazinone derivatives and their antihypertensive activities. Eur. J. Med. Chem. 2004, 39, 1089–1095. [Google Scholar] [CrossRef]
- Partap, S.; Akhtar, M.J.; Yar, M.S.; Hassan, M.Z.; Siddiqui, A.A. Pyridazinone hybrids: Design, synthesis and evaluation as potential anticonvulsant agents. Bioorg. Chem. 2018, 77, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Lümmen, P. Complex I inhibitors as insecticides and acaricides. Biochim. Biophys. Acta Bioenerg. 1998, 1364, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Dekeyser, M.A. Acaricide mode of action. Pest Manag. Sci. 2005, 61, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Weissmuller, J.; Tietjen, K.G.; Stendel, W.; Wachendorff-Neumann, U. Pyridazinones as Pesticides. U.S. Patent 5,004,744, 2 April 1991. [Google Scholar]
- Asif, M. Antifeedant, herbicidal and molluscicidal activities of pyridazinone compounds. Mini Rev. Org. Chem. 2013, 10, 113–122. [Google Scholar] [CrossRef]
- Martiz, R.M.; Patil, S.M.; Abdulaziz, M.; Babalghith, A.; Al-Areefi, M.; Al-Ghorbani, M.; Mallappa Kumar, J.; Prasad, A.; Mysore Nagalingaswamy, N.P.; Ramu, R. Defining the Role of Isoeugenol from Ocimum tenuiflorum against Diabetes Mellitus-Linked Alzheimer’s Disease through Network Pharmacology and Computational Methods. Molecules 2022, 27, 2398. [Google Scholar] [CrossRef]
- Martiz, R.M.; Patil, S.M.; Thirumalapura Hombegowda, D.; Shbeer, A.M.; Alqadi, T.; Al-Ghorbani, M.; Ramith, R.; Prasad, A. Phyto-Computational Intervention of Diabetes Mellitus at Multiple Stages Using Isoeugenol from Ocimum tenuiflorum: A Combination of Pharmacokinetics and Molecular Modelling Approaches. Molecules 2022, 27, 6222. [Google Scholar] [CrossRef]
- Maradesha, T.; Patil, S.M.; Al-Mutairi, K.A.; Ramu, R.; Madhunapantula, S.V.; Alqadi, T. Inhibitory effect of polyphenols from the whole green jackfruit flour against α-glucosidase, α-amylase, aldose reductase and glycation at multiple stages and their interaction: Inhibition kinetics and molecular simulations. Molecules 2022, 27, 1888. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. I. The effect of the exchange-only gradient correction. J. Chem. Phys. 1992, 96, 2155–2160. [Google Scholar] [CrossRef]
- Vautherin, D.; Brink, D.T. Hartree-Fock calculations with Skyrme’s interaction. I. Spherical nuclei. Phys. Rev. C 1972, 5, 626. [Google Scholar] [CrossRef]
- Hohenstein, E.G.; Chill, S.T.; Sherrill, C.D. Assessment of the performance of the M05− 2X and M06− 2X exchange-correlation functionals for noncovalent interactions in biomolecules. J. Chem. Theory Comput. 2008, 4, 1996–2000. [Google Scholar] [CrossRef]
- Karrouchi, K.; Fettach, S.; Tüzün, B.; Radi, S.; Alharthi, A.I.; Ghabbour, H.A.; Garcia, Y. Synthesis, crystal structure, DFT, α-glucosidase and α-amylase inhibition and molecular docking studies of (E)-N′-(4-chlorobenzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide. J. Mol. Struct. 2021, 1245, 131067. [Google Scholar] [CrossRef]
- Dadou, S.; Altay, A.; Koudad, M.; Türkmenoğlu, B.; Yeniçeri, E.; Çağlar, S.; Karrouchi, K. Design, synthesis, anticancer evaluation and molecular docking studies of new imidazo [2, 1-b] thiazole-based chalcones. Med. Chem. Res. 2022, 31, 1369–1383. [Google Scholar] [CrossRef]
- Fettach, S.; Thari, F.Z.; Hafidi, Z.; Tachallait, H.; Karrouchi, K.; El Achouri, M.; Faouzi, M.E.A. Synthesis, α-glucosidase and α-amylase inhibitory activities, acute toxicity and molecular docking studies of thiazolidine-2, 4-diones derivatives. J. Biomol. Struct. Dyn. 2021, 40, 8340–8351. [Google Scholar] [CrossRef]
- Fettach, S.; Thari, F.Z.; Hafidi, Z.; Karrouchi, K.; Bouathmany, K.; Cherrah, Y.; Faouzi, M.E.A. Biological, toxicological and molecular docking evaluations of isoxazoline-thiazolidine-2, 4-dione analogues as new class of anti-hyperglycemic agents. J. Biomol. Struct. Dyn. 2021, 1–13. [Google Scholar] [CrossRef]
- Karrouchi, K.; Fettach, S.; Radi, S.; Taoufik, J.; Mabkhot, Y.N.; Alterary, S.; Ansar, M. Synthesis, characterization, free-radical scavenging capacity and antioxidant activity of novel series of hydrazone, 1, 3, 4-oxadiazole and 1, 2, 4-triazole derived from 3, 5-dimethyl-1H-pyrazole. Lett. Drug Des. Discov. 2019, 16, 712–720. [Google Scholar] [CrossRef]
- Karrouchi, K.; Chemlal, L.; Taoufik, J.; Cherrah, Y.; Radi, S.; Faouzi, M.E.A.; Ansar, M. Synthesis, antioxidant and analgesic activities of Schiff bases of 4-amino-1, 2, 4-triazole derivatives containing a pyrazole moiety. Ann. Pharm. Fr. 2016, 74, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Curran, W.V.; Ross, A. 6-Phenyl-4, 5-dihydro-3 (2H)-pyridazinones. Series of hypotensive agents. J. Med. Chem. 1974, 17, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Daoui, S.; Cinar, E.B.; Dege, N.; Chelfi, T.; El Kalai, F.; Abudunia, A.; Benchat, N. Crystal structure and Hirshfeld surface analysis of 4-(2, 6-dichlorobenzyl)-6-[(E)-2-phenylethenyl] pyridazin-3 (2H)-one. Acta Crystallogr. Sect. E 2021, 77, 23–27. [Google Scholar] [CrossRef]
- El Kalai, F.; Baydere, C.; Daoui, S.; Saddik, R.; Dege, N.; Karrouchi, K.; Benchat, N. Crystal structure and Hirshfeld surface analysis of ethyl 2-[5-(3-chlorobenzyl)-6-oxo-3-phenyl-1, 6-dihydropyridazin-1-yl] acetate. Acta Crystallogr. Sect. E 2019, 75, 892–895. [Google Scholar] [CrossRef] [PubMed]
- Daoui, S.; Baydere, C.; El Kalai, F.; Mahi, L.; Dege, N.; Karrouchi, K.; Benchat, N. Crystal structure, Hirshfeld surface analysis and DFT studies of 2-[5-(4-methylbenzyl)-6-oxo-3-phenyl-1, 6-dihydropyridazin-1-yl] acetic acid. Acta Crystallogr. Sect. E 2019, 75, 1925–1929. [Google Scholar] [CrossRef] [PubMed]
- Daoui, S.; Baydere, C.; Chelfi, T.; El Kalai, F.; Dege, N.; Karrouchi, K.; Benchat, N. Polymorphism of 2-(5-benzyl-6-oxo-3-phenyl-1, 6-dihydropyridazin-1-yl) acetic acid with two monoclinic modifications: Crystal structures and Hirshfeld surface analyses. Acta Crystallogr. Sect. E 2020, 76, 432–437. [Google Scholar] [CrossRef]
- Günsel, A.; Kırbaç, E.; Tüzün, B.; Erdoğmuş, A.; Bilgiçli, A.T.; Yarasir, M.N. Selective chemosensor phthalocyanines for Pd2+ ions; synthesis, characterization, quantum chemical calculation, photochemical and photophysical properties. J. Mol. Struct. 2019, 1180, 127–138. [Google Scholar] [CrossRef]
- Ojha, L.K.; Tüzün, B.; Bhawsar, J. Experimental and theoretical study of effect of allium sativum extracts as corrosion inhibitor on mild steel in 1 M HCl medium. J. Bio-Tribo-Corros. 2020, 6, 39. [Google Scholar] [CrossRef]
- Günsel, A.; Bilgiçli, A.T.; Pişkin, H.; Tüzün, B.; Yarasir, M.N.; Gündüz, B. Synthesis of non-peripherally tetra-substituted copper (ii) phthalocyanines: Characterization, optical and surface properties, fabrication and photo-electrical properties of a photosensitive diode. Dalton Trans. 2019, 48, 14839–14852. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, D.; Tüzün, B.; Pal, T.K.; Saini, R.V.; Bankura, K.; Mishra, D. Structurally diverse heterobimetallic Pb (II)-Salen complexes mechanistic notion of cytotoxic activity against neuroblastoma cancer cell: Synthesis, characterization, protein–ligand interaction profiler, and intuitions from DFT. Polyhedron 2021, 210, 115504. [Google Scholar] [CrossRef]
- Khalilov, A.N.; Tüzün, B.; Taslimi, P.; Tas, A.; Tuncbilek, Z.; Cakmak, N.K. Cytotoxic effect, spectroscopy, DFT, enzyme inhibition, and moleculer docking studies of some novel mesitylaminopropanols: Antidiabetic and anticholinergics and anticancer potentials. J. Mol. Liq. 2021, 344, 117761. [Google Scholar] [CrossRef]
- Rbaa, M.; Haida, S.; Tuzun, B.; El Hassane, A.; Kribii, A.; Lakhrissi, Y.; Berdimurodov, E. Synthesis, characterization and bioactivity of novel 8-hydroxyquinoline derivatives: Experimental, molecular docking, DFT and POM analyses. J. Mol. Struct. 2022, 1258, 132688. [Google Scholar] [CrossRef]
- Lakhrissi, Y.; Rbaa, M.; Tuzun, B.; Hichar, A.; Ounine, K.; Almalki, F.; Lakhrissi, B. Synthesis, structural confirmation, antibacterial properties and bio-informatics computational analyses of new pyrrole based on 8-hydroxyquinoline. J. Mol. Struct. 2022, 1259, 132683. [Google Scholar] [CrossRef]
- Al-Janabi, I.A.S.; Yavuz, S.Ç.; Köprü, S.; Tapera, M.; Kekeçmuhammed, H.; Akkoç, S.; Sarıpınar, E. Antiproliferative activity and molecular docking studies of new 4-oxothiazolidin-5-ylidene acetate derivatives containing guanylhydrazone moiety. J. Mol. Struct. 2022, 1258, 132627. [Google Scholar] [CrossRef]
- Koçyiğit, Ü.M.; Doğan, M.; Muğlu, H.; Taslimi, P.; Tüzün, B.; Yakan, H.; Gülçin, İ. Determination of biological studies and molecular docking calculations of isatin-thiosemicarbazone hybrid compounds. J. Mol. Struct. 2022, 1264, 133249. [Google Scholar] [CrossRef]
- Erdogan, M.K.; Gundogdu, R.; Yapar, Y.; Gecibesler, I.H.; Kirici, M.; Behcet, L.; Taslimi, P. The Evaluation of Anticancer, Antioxidant, Antidiabetic and Anticholinergic Potentials of Endemic Rhabdosciadium microcalycinum Supported by Molecular Docking Study. Chem. Select 2022, 7, e202200400. [Google Scholar] [CrossRef]
- Mermer, A.; Bulbul, M.V.; Kalender, S.M.; Keskin, I.; Tuzun, B.; Eyupoglu, O.E. Benzotriazole-oxadiazole Hybrid Compounds: Synthesis, Anticancer Activity, Molecular Docking and ADME Profiling Studies. J. Mol. Liq. 2022, 359, 119264. [Google Scholar] [CrossRef]
- Lipinski, C.A. Lead-and drug-like compounds: The rule-of-five revolution. Drug Discov. Today Technol. 2004, 1, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Jorgensen, W.J.; Duffy, E.M. Prediction of drug solubility from structure. Adv. Drug Deliv. Rev. 2002, 54, 355–366. [Google Scholar] [CrossRef]
- Dennington, R.; Keith, T.A.; Millam, J.M. GaussView, version 6.0. 16; Semichem Inc.: Shawnee Mission, KS, USA, 2016.
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian, version 09, revision D.01; Gaussian Inc.: Wallingford, UK, 2009.
- Tüzün, B. Theoretical evaluation of six indazole derivatives as corrosion inhibitors based on DFT. Turk. Comput. Theoret. Chem. 2018, 2, 12–22. [Google Scholar]
- Günsel, A.; Bilgiçli, A.T.; Tüzün, B.; Pişkin, H.; Atmaca, G.Y.; Erdoğmuş, A.; Yarasir, M.N. Synthesis of tetra-substituted phthalocyanines bearing 2-(ethyl (m-tolyl) amino) ethanol: Computational and photophysicochemical studies. J. Photochem. Photobiol. 2019, 373, 77–86. [Google Scholar] [CrossRef]
- Güzel, E.; Günsel, A.; Tüzün, B.; Atmaca, G.Y.; Bilgiçli, A.T.; Erdoğmuş, A.; Yarasir, M.N. Synthesis of tetra-substituted metallophthalocyanines: Spectral, structural, computational studies and investigation of their photophysical and photochemical properties. Polyhedron 2019, 158, 316–324. [Google Scholar] [CrossRef]
- Schrödinger Release 2021-3: Maestro; Schrödinger, LLC: New York, NY, USA, 2021.
- Schrödinger Release 2019-4: Protein Preparation Wizard; Epik, Schrödinger, LLC: New York, NY, USA, 2016; Impact, Schrödinger, LLC: New York, NY, USA, 2016; Prime, Schrödinger, LLC: New York, NY, USA, 2019.
- Schrödinger Release 2021-3: LigPrep; Schrödinger, LLC: New York, NY, USA, 2021.
- Poustforoosh, A.; Hashemipour, H.; Tüzün, B.; Pardakhty, A.; Mehrabani, M.; Nematollahi, M.H. Evaluation of potential anti-RNA-dependent RNA polymerase (RdRP) drugs against the newly emerged model of COVID-19 RdRP using computational methods. Biophys. Chem. 2021, 272, 106564. [Google Scholar] [CrossRef] [PubMed]
- Schrödinger Release 2021-3: QikProp; Schrödinger, LLC: New York, NY, USA, 2021.
- Ustabaş, R.; Süleymanoğlu, N.; Ünver, Y.; Direkel, Ş. 5-(4-Bromobenzyl)-4-(4-(5-phenyl-1, 3, 4-oxadiazole-2-yl) phenyl)-2, 4-dihydro-3H-1, 2, 4-triazole-3-one: Synthesis, characterization, DFT study and antimicrobial activity. J. Mol. Struct. 2020, 1214, 128217. [Google Scholar] [CrossRef]



| Compound | R1 | R2 | R3 | R4 | Molecular Formula | Mol. Wt (g/mol) | Yield (%) | m.p. (°C) |
|---|---|---|---|---|---|---|---|---|
| 3 | H | H | CH3 | H | C18H16N2O | 276.13 | 78 | 275–297 |
| 4 | H | H | Cl | H | C17H13ClN2O | 296.07 | 72 | 295–297 |
| 5 | Cl | H | H | Cl | C17H12Cl2N2O | 330.03 | 94 | 164–166 |
| 6 | NO2 | H | H | H | C17H13N3O3 | 307.10 | 76 | 170–172 |
| 7 | H | H | F | H | C17H13FN2O | 280.10 | 58 | 279–281 |
| 8 | H | H | CH3 | H | C22H22N2O2 | 346.17 | 71 | 120–122 |
| 9 | H | H | Cl | H | C21H19ClN2O3 | 382.11 | 92 | 142–144 |
| 10 | Cl | H | H | Cl | C21H18Cl2N2O3 | 416.07 | 78 | 80–82 |
| 11 | NO2 | H | H | H | C21H19N3O5 | 393.13 | 85 | 175–177 |
| 12 | H | H | F | H | C21H19FN2O3 | 366.14 | 88 | 73–75 |
| 13 | H | H | CH3 | H | C20H18N2O3 | 334.13 | 90 | 170–172 |
| 14 | H | H | Cl | H | C19H15ClN2O3 | 354.08 | 84 | 174–176 |
| 15 | Cl | H | H | Cl | C19H14Cl2N2O3 | 388.04 | 89 | 140–142 |
| 16 | NO2 | H | H | H | C19H15N3O5 | 365.10 | 80 | 102–104 |
| 17 | H | H | F | H | C19H15FN2O3 | 338.11 | 86 | 187–189 |
| Compound | MIC (µM/mL) | ||||
|---|---|---|---|---|---|
| E. coli | S. aureus (MRSA) | S. typhimurium | P. aeruginosa | A. baumannii | |
| 3 | >36.21 | 4.52 | >36.21 | 36.21 | >36.21 |
| 4 | >33.77 | > 33.77 | >33.77 | >33.77 | >33.77 |
| 5 | >30.30 | >30.30 | >30.30 | >30.30 | >30.30 |
| 6 | >32.56 | >32.56 | >32.56 | >32.56 | >32.56 |
| 7 | 8.92 | 8.92 | 8.92 | 17.85 | 8.92 |
| 8 | >28.88 | >28.88 | >28.88 | 28.88 | 28.88 |
| 9 | >27.32 | >27.32 | >27.32 | >27.32 | >27.32 |
| 10 | >25.00 | >25.00 | 25.00 | >25.00 | 25.00 |
| 11 | >26.51 | >26.51 | >26.51 | 26.51 | 26.51 |
| 12 | >28.57 | >28.57 | >28.57 | >28.57 | >28.57 |
| 13 | 29.94 | 7.48 | 29.94 | 7.48 | 3.74 |
| 14 | >28.24 | 28.24 | 28.24 | 28.24 | >28.24 |
| 15 | >25.77 | 25.77 | >25.77 | 6.44 | 6.44 |
| 16 | NA a | NA | NA | NA | NA |
| 17 | >29.57 | 29.57 | >29.57 | 14.78 | >29.57 |
| Amikacin | <0.53 | <0.53 | <0.53 | <0.53 | <0.53 |
| Compound | Quantum Chemical Descriptors (e.V) | |||||||
|---|---|---|---|---|---|---|---|---|
| EHOMO | ELUMO | ΔE | η | μ | ω | ε | χ | |
| 7 | −6.20 | −1.90 | 4.30 | 2.15 | 0.47 | 3.81 | 0.26 | 4.05 |
| 13 | −6.06 | −1.76 | 4.29 | 2.14 | 0.47 | 3.58 | 0.28 | 3.91 |
| Compound | Docking Parameters (kcal/mol) | |||||||
|---|---|---|---|---|---|---|---|---|
| Docking Score | Glide Evdw | Glide Ecoul | Glide Emodel | Glide Einternal | Glide Einternal | Glide Einternal | Glide Posenum | |
| 1JIJ | ||||||||
| 7 | −7.12 | −43.07 | −1.87 | −65.11 | 1.23 | 1.23 | 1.23 | 351 |
| 13 | −7.31 | −45.05 | −3.71 | −68.49 | 5.12 | 5.12 | 5.12 | 1 |
| Amikacin | −6.07 | −28.77 | −23.49 | −78.46 | 10.46 | 10.46 | 10.46 | 372 |
| 2UV0 | ||||||||
| 7 | −4.52 | −25.49 | −0.40 | −32.81 | 0.30 | 0.30 | 0.30 | 214 |
| 13 | −5.69 | −19.45 | −5.00 | −29.44 | 2.70 | 2.70 | 2.70 | 157 |
| Amikacin | −4.13 | −22.71 | −30.13 | −65.36 | 21.50 | 21.50 | 21.50 | 252 |
| 4WUB | ||||||||
| 7 | −5.74 | −30.15 | −0.69 | −39.21 | 1.72 | 1.72 | 1.72 | 357 |
| 13 | −4.65 | −28.96 | −1.29 | −37.10 | 3.39 | 3.39 | 3.39 | 49 |
| Amikacin | −5.07 | −16.41 | −29.44 | −70.26 | 6.21 | 6.21 | 6.21 | 362 |
| Parameters | 7 | 13 | Reference Range |
|---|---|---|---|
| mol_MW | 280 | 334 | 130–725 |
| Dipole (D) | 3.4 | 8.0 | 1.0–12.5 |
| SASA | 539 | 626 | 300–1000 |
| FOSA | 36 | 159 | 0–750 |
| FISA | 113 | 150 | 7–330 |
| PISA | 344 | 317 | 0–450 |
| WPSA | 47 | 0 | 0–175 |
| Volume (A3) | 918 | 1096 | 500–2000 |
| donorHB | 1 | 1 | 0–6 |
| accptHB | 2 | 4 | 2.0–20.0 |
| glob (sphere = 1) | 0.8 | 0.8 | 0.75–0.95 |
| QPpolrz (A3) | 31.9 | 37.5 | 13.0–70.0 |
| QPlogPC16 | 9.8 | 11.9 | 4.0–18.0 |
| QPlogPoct | 13.4 | 16.9 | 8.0–35.0 |
| QPlogPw | 6.9 | 8.7 | 4.0–45.0 |
| QPlogPo/w | 4.0 | 4.3 | −2.0–6.5 |
| QPlogS | −5.1 | −5.4 | −6.5–0.5 |
| CIQPlogS | −5.1 | −5.5 | −6.5–0.5 |
| QPlogHERG | −5.6 | −4.1 | * |
| QPPCaco (nm/sec) | 846 | 95 | ** |
| QPlogBB | −0.6 | −1.2 | −3.0–1.2 |
| QPPMDCK (nm/sec) | 744 | 49 | ** |
| QPlogKp | −2.1 | −2.7 | Kp in cm/hr |
| IP (ev) | 9.8 | 9.7 | 7.9–10.5 |
| EA (eV) | 1.2 | 1.2 | −0.9–1.7 |
| #metab | 1 | 3 | 1–8 |
| QPlogKhsa | 0.6 | 0.4 | −1.5–1.5 |
| Human Oral Absorption | 3 | 3 | - |
| Percent Human Oral Absorption | 100 | 88 | *** |
| PSA | 60 | 89 | 7–200 |
| RuleOfFive | 0 | 0 | Maximum is 4 |
| RuleOfThree | 0 | 0 | Maximum is 3 |
| Jm | 0.0 | 0.0 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daoui, S.; Direkel, Ş.; Ibrahim, M.M.; Tüzün, B.; Chelfi, T.; Al-Ghorbani, M.; Bouatia, M.; Karbane, M.E.; Doukkali, A.; Benchat, N.; et al. Synthesis, Spectroscopic Characterization, Antibacterial Activity, and Computational Studies of Novel Pyridazinone Derivatives. Molecules 2023, 28, 678. https://doi.org/10.3390/molecules28020678
Daoui S, Direkel Ş, Ibrahim MM, Tüzün B, Chelfi T, Al-Ghorbani M, Bouatia M, Karbane ME, Doukkali A, Benchat N, et al. Synthesis, Spectroscopic Characterization, Antibacterial Activity, and Computational Studies of Novel Pyridazinone Derivatives. Molecules. 2023; 28(2):678. https://doi.org/10.3390/molecules28020678
Chicago/Turabian StyleDaoui, Said, Şahin Direkel, Munjed M. Ibrahim, Burak Tüzün, Tarik Chelfi, Mohammed Al-Ghorbani, Mustapha Bouatia, Miloud El Karbane, Anass Doukkali, Noureddine Benchat, and et al. 2023. "Synthesis, Spectroscopic Characterization, Antibacterial Activity, and Computational Studies of Novel Pyridazinone Derivatives" Molecules 28, no. 2: 678. https://doi.org/10.3390/molecules28020678
APA StyleDaoui, S., Direkel, Ş., Ibrahim, M. M., Tüzün, B., Chelfi, T., Al-Ghorbani, M., Bouatia, M., Karbane, M. E., Doukkali, A., Benchat, N., & Karrouchi, K. (2023). Synthesis, Spectroscopic Characterization, Antibacterial Activity, and Computational Studies of Novel Pyridazinone Derivatives. Molecules, 28(2), 678. https://doi.org/10.3390/molecules28020678







