A Study of Greek Graviera Cheese by NMR-Based Metabolomics
Abstract
1. Introduction
2. Results and Discussion
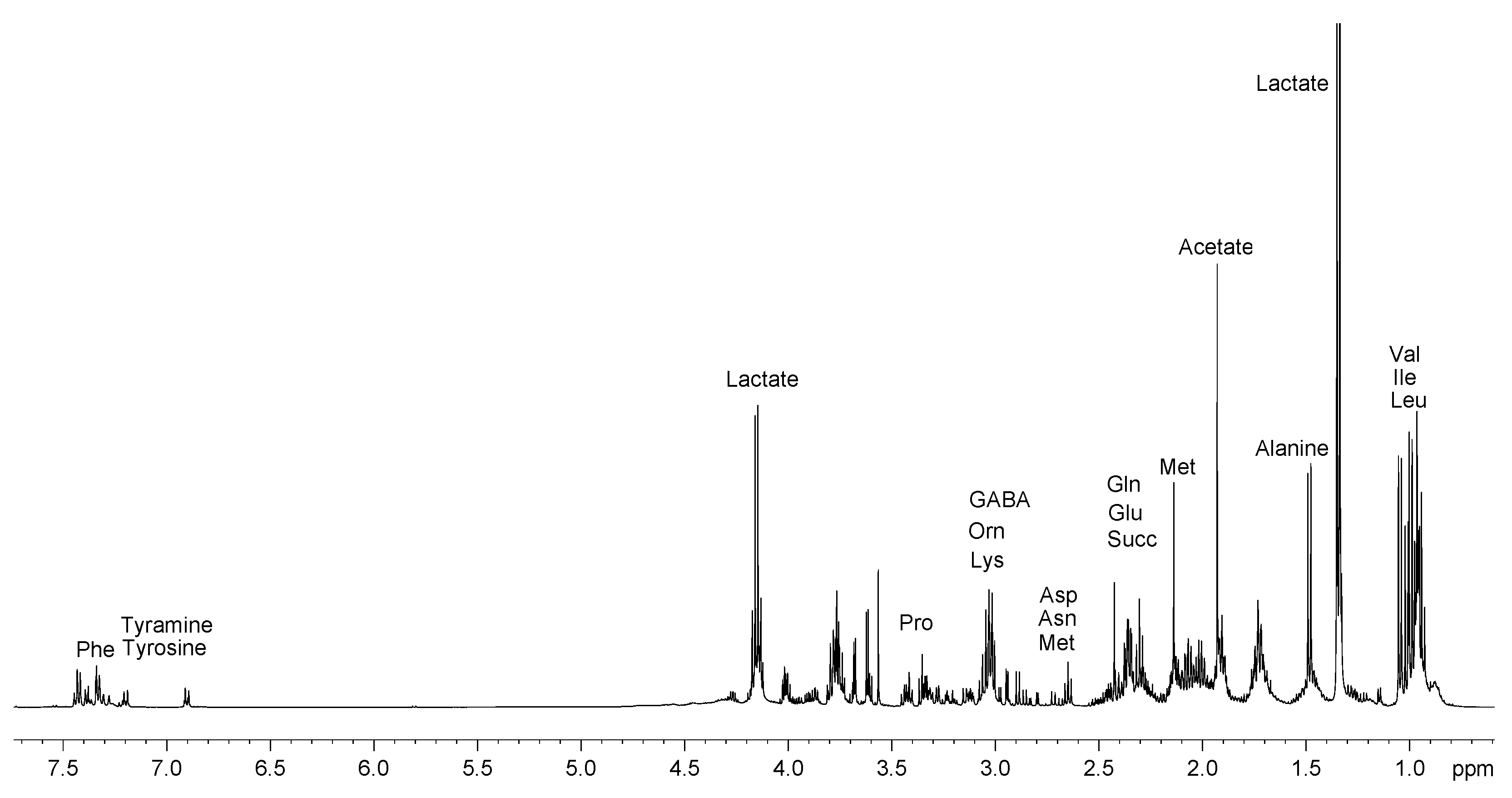
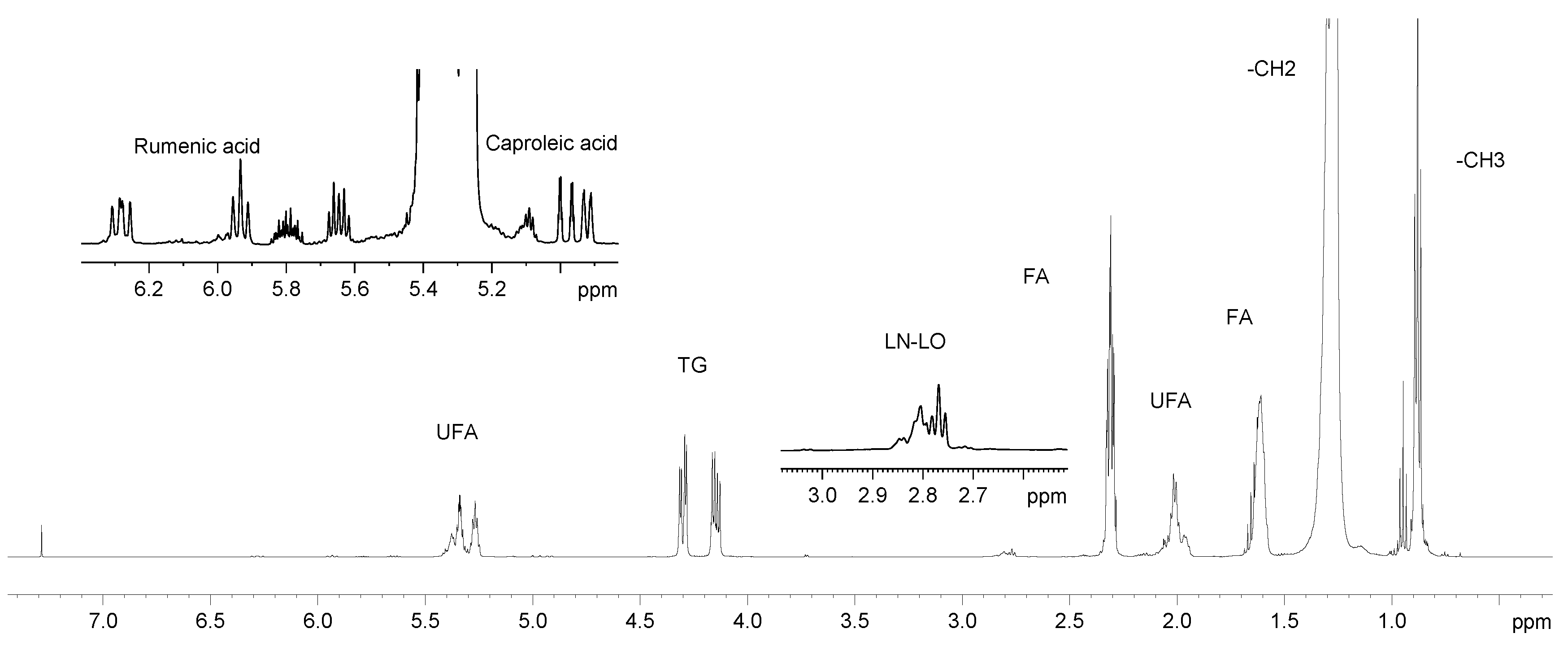
2.1. Analysis of 1H NMR Spectra
2.1.1. Polar Fraction
2.1.2. Lipid Fraction
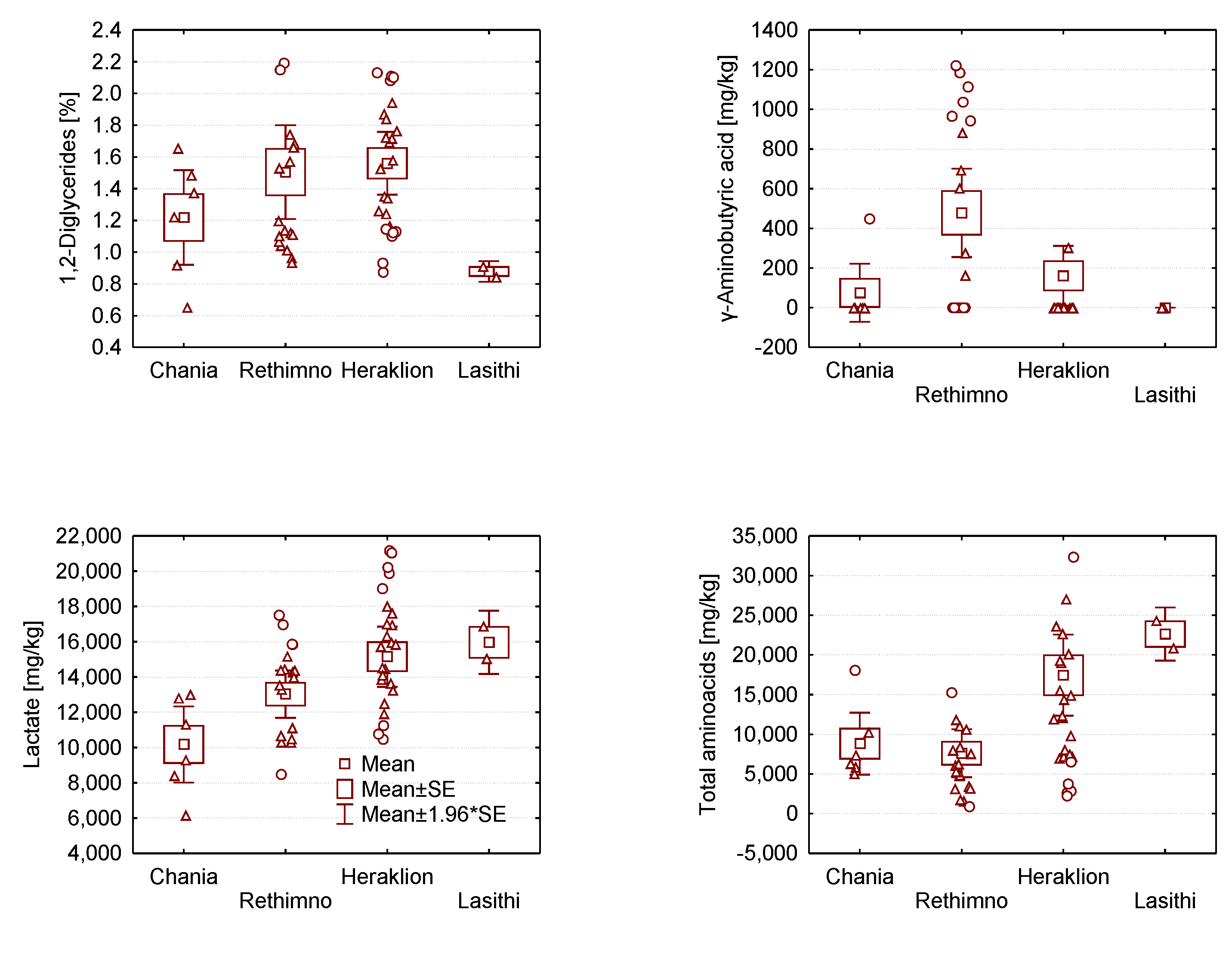
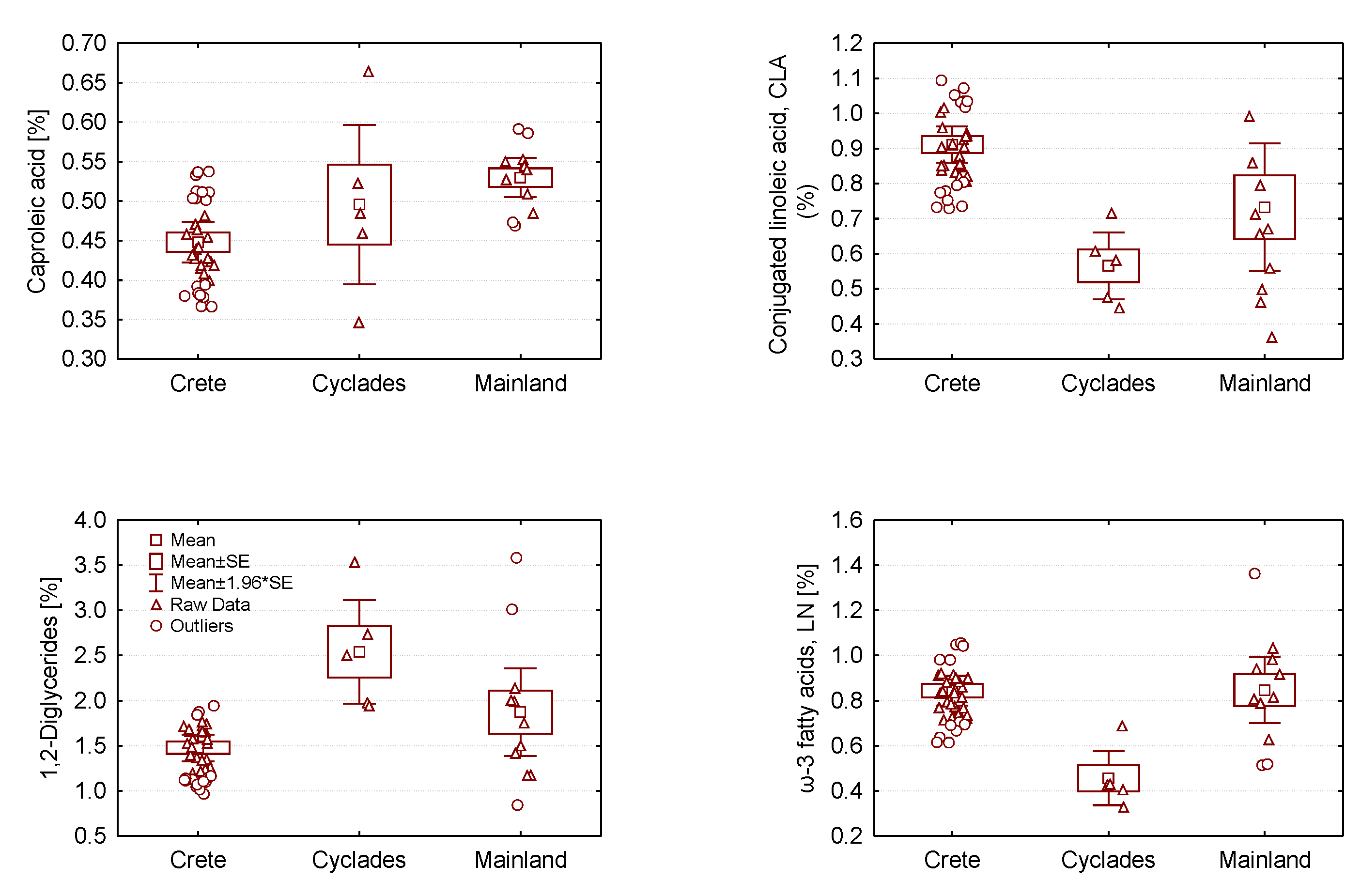
2.2. Analysis of Variance of Graviera Composition
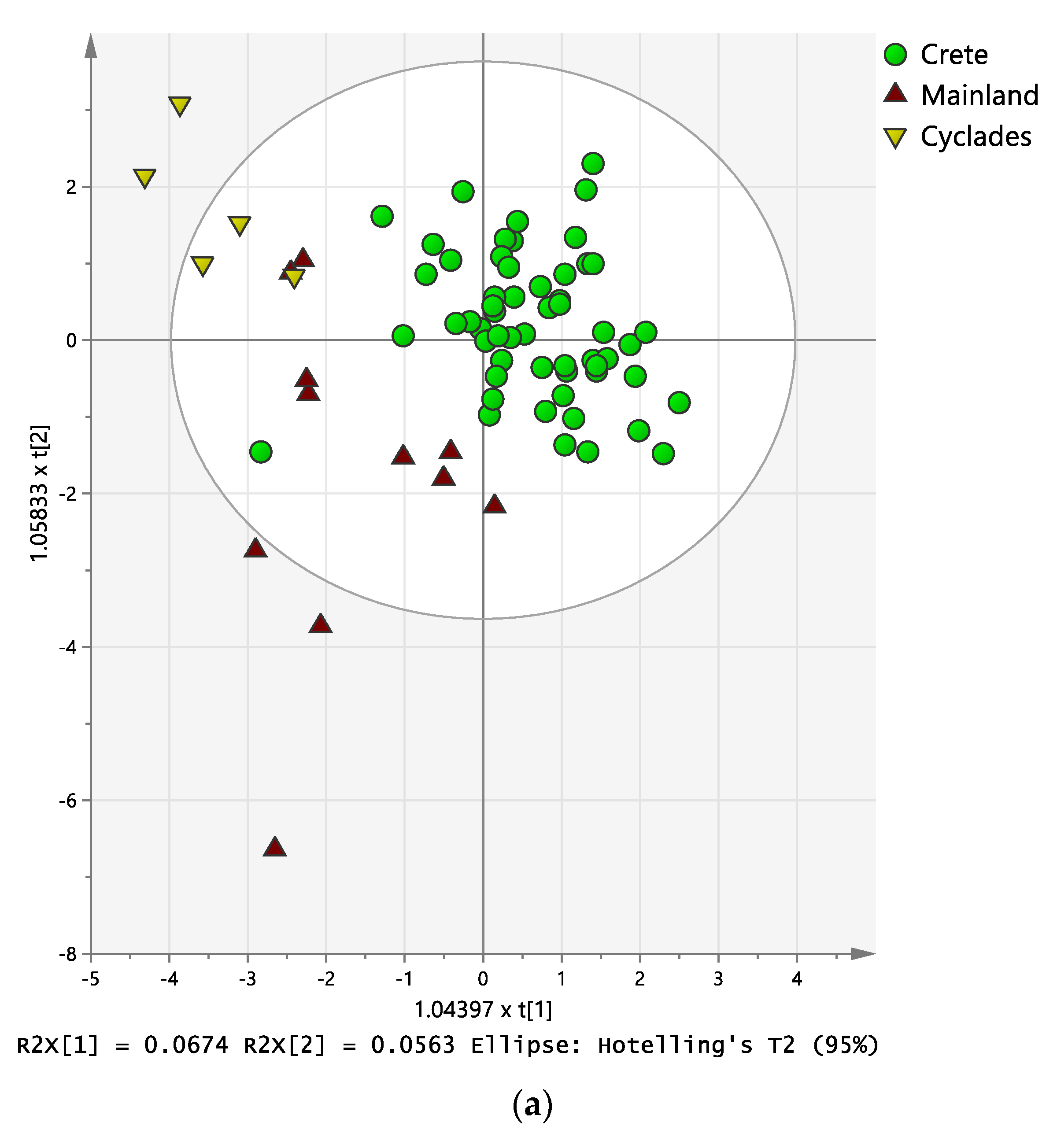
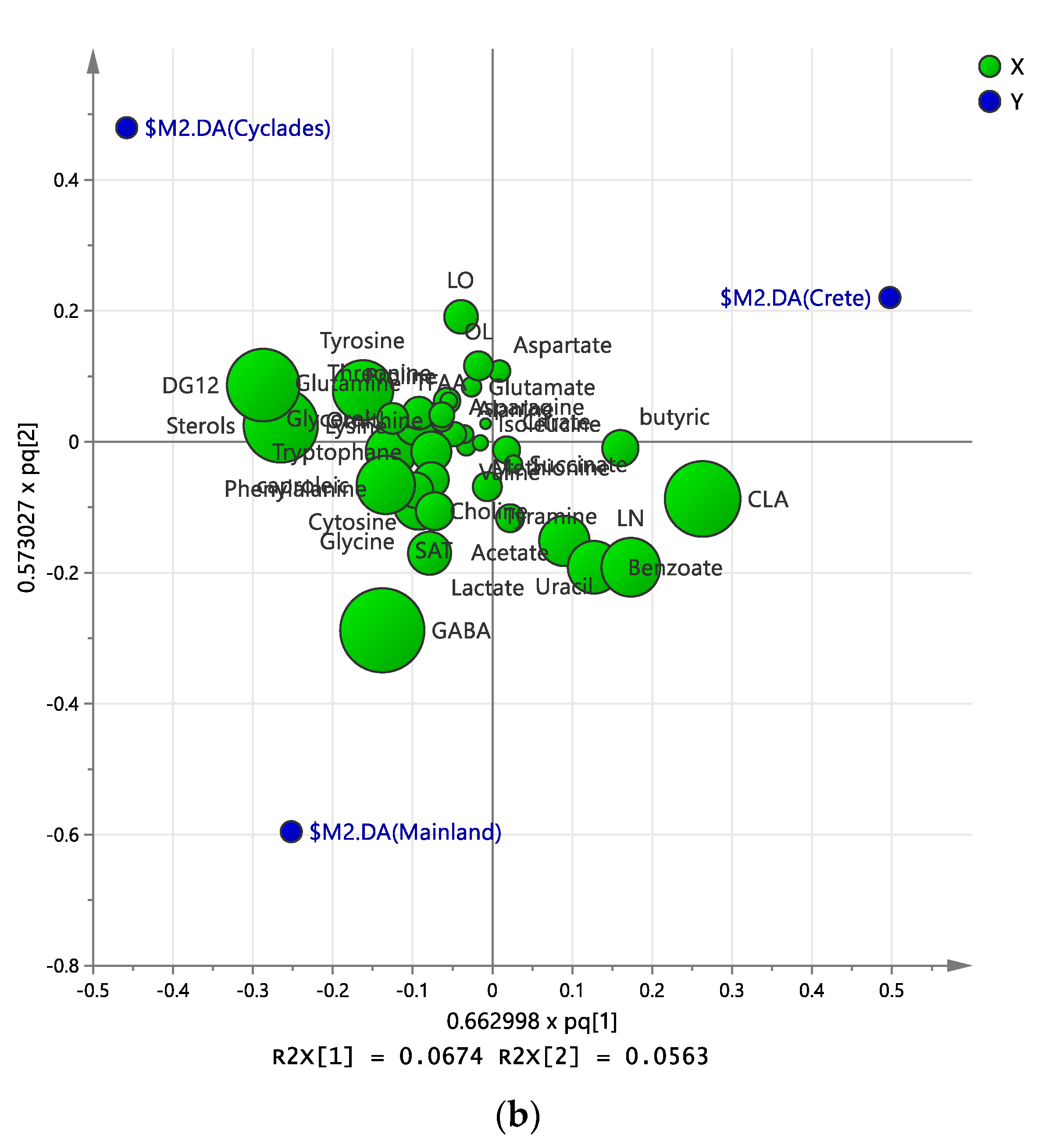
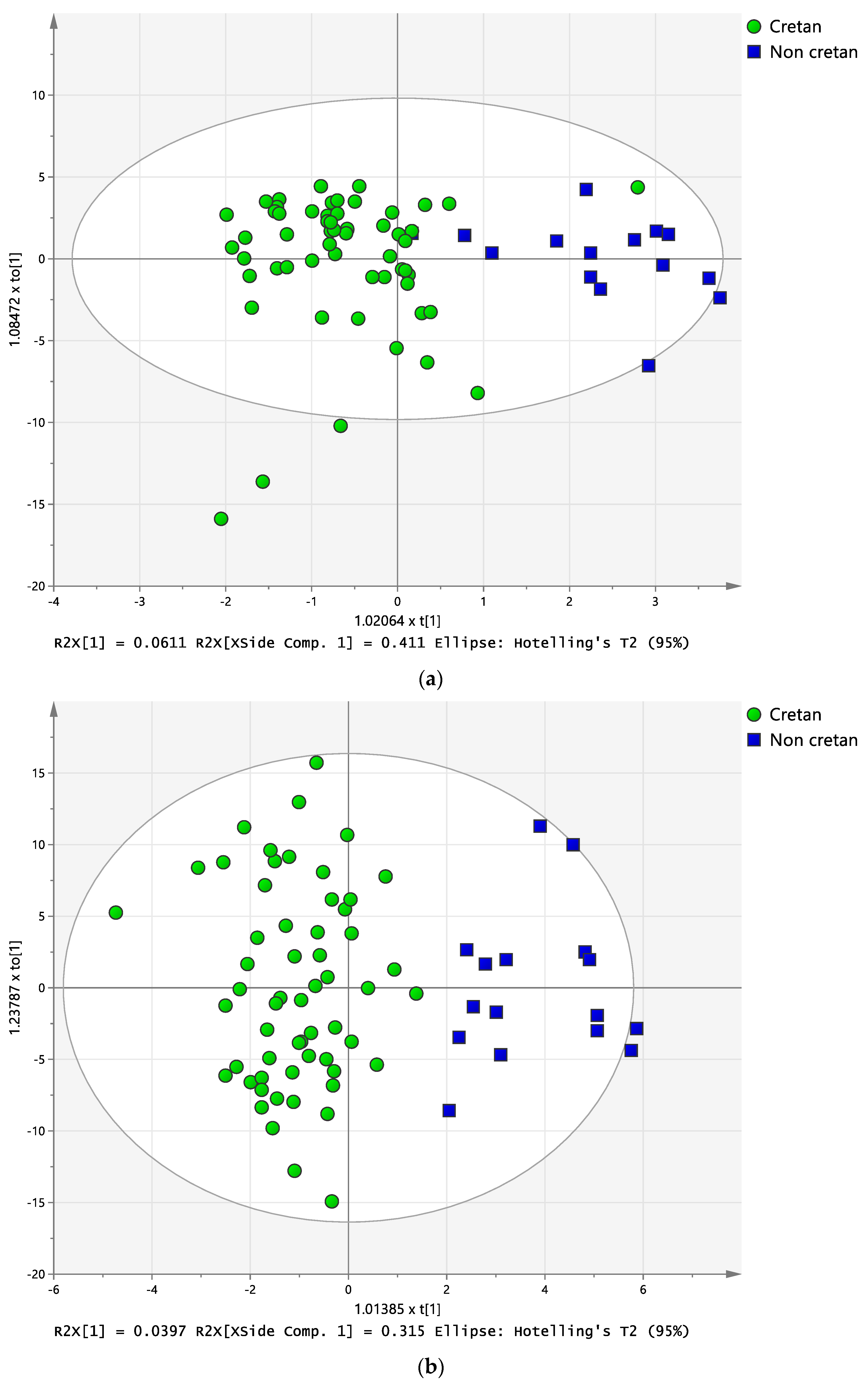
2.3. Discriminant Analysis
3. Materials and Methods
3.1. Chemicals
3.2. Samples
3.3. Sample Preparation: NMR Analysis
3.4. Spectra Integration and Multivariate Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- EC. Commission of the European Communities. Commission Regulation (EC) No 1107/96 of 12 June 1996 on the registration of geographical indications and designations of origin under the procedure laid down in Article 17 of Council Regulation (EEC) No 2081/92. Off. J. Eur. Commun. 1996, 39, 1–10. [Google Scholar]
- McSweeney, P.L.H.; Fox, P.F.; Ciocia, F. Chapter 16—Metabolism of Residual Lactose and of Lactate and Citrate. In Cheese, 4th ed.; McSweeney, P.L.H., Fox, P.F., Cotter, P.D., Everett, D.W., Eds.; Academic Press: San Diego, CA, USA, 2017; pp. 411–421. [Google Scholar] [CrossRef]
- Fox, P.F. Proteolysis during cheese manufacture and ripening. J. Dairy Sci. 1989, 72, 1379–1400. [Google Scholar] [CrossRef]
- Collins, Y.F.; McSweeney, P.L.H.; Wilkinson, M.G. Lipolysis and free fatty acid catabolism in cheese: A review of current knowledge. Int. Dairy J. 2003, 13, 841–866. [Google Scholar] [CrossRef]
- Tzora, A.; Nelli, A.; Voidarou, C.; Fotou, K.; Bonos, E.; Rozos, G.; Grigoriadou, K.; Papadopoulos, P.; Basdagianni, Z.; Giannenas, I.; et al. Impact of an Omega-3-Enriched Sheep Diet on the Microbiota and Chemical Composition of Kefalograviera Cheese. Foods 2022, 11, 843. [Google Scholar] [CrossRef] [PubMed]
- Segato, S.; Caligiani, A.; Contiero, B.; Galaverna, G.; Bisutti, V.; Cozzi, G. 1H NMR metabolic profile to discriminate pasture based alpine asiago PDO cheeses. Animals 2019, 9, 722. [Google Scholar] [CrossRef] [PubMed]
- Danezis, G.; Theodorou, C.; Massouras, T.; Zoidis, E.; Hadjigeorgiou, I.; Georgiou, C.A. Greek graviera cheese assessment through elemental metabolomics-implications for authentication, safety and nutrition. Molecules 2019, 24, 670. [Google Scholar] [CrossRef]
- Bozoudi, D.; Pavlidou, S.; Kotzamanidis, C.; Georgakopoulos, P.; Torriani, S.; Kondyli, E.; Claps, S.; Belibasaki, S.; Litopoulou-Tzanetaki, E. “Graviera Naxou and Graviera Kritis Greek PDO cheeses: Discrimination based on microbiological and physicochemical criteria and volatile organic compounds profile”. Small Ruminant Res. 2016, 136, 161–172. [Google Scholar] [CrossRef]
- EC. Commission of the European Communities. Council Regulation (EC) No 510/2006 of 20 March 2006 on the protection of geographical indications and designations of origin for agricultural products and foodstuffs. Off. J. Eur. Commun. 2006, 93, 12–25. [Google Scholar]
- Vatavali, K.A.; Kosma, I.S.; Louppis, A.P.; Badeka, A.V.; Kontominas, M.G. Physicochemical, spectroscopic, and chromatographic analyses in combination with chemometrics for the discrimination of the geographical origin of Greek Graviera Cheeses. Molecules 2020, 25, 3507. [Google Scholar] [CrossRef]
- Danezis, G.P.; Tsiplakou, E.; Pappa, E.C.; Pappas, A.C.; Mavrommatis, A.; Sotirakoglou, K.; Georgiou, C.A.; Zervas, G. Fatty acid profile and physicochemical properties of Greek protected designation of origin cheeses, implications for authentication. Eur. Food Res. Technol. 2020, 246, 1741–1753. [Google Scholar] [CrossRef]
- Georgala, A.; Kaminarides, S.; Anifantakis, E.M. Free fatty acid content of some traditional Greek cheese varieties. Aust. J. Dairy Technol. 2006, 61, 26–31. [Google Scholar]
- Gianferri, R.; Maioli, M.; Delfini, M.; Brosio, E. A low-resolution and high-resolution nuclear magnetic resonance integrated approach to investigate the physical structure and metabolic profile of Mozzarella di Bufala Campana cheese. Int. Dairy J. 2007, 17, 167–176. [Google Scholar] [CrossRef]
- Scano, P.; Anedda, R.; Melis, M.P.; Dessi, M.A.; Lai, A.; Roggio, T. 1H- and 13C-NMR characterization of the molecular components of the lipid fraction of Pecorino Sardo cheese. J. Am. Oil Chem. Soc. 2011, 88, 1305–1316. [Google Scholar] [CrossRef]
- Scano, P.; Cagliani, L.R.; Consonni, R. 1H NMR characterisation of the lipid fraction and the metabolite profiles of Fossa (pit) cheese. Int. Dairy J. 2019, 90, 39–44. [Google Scholar] [CrossRef]
- De Angelis Curtis, S.; Curini, R.; Delfini, M.; Brosio, E.; D’Ascenzo, F.; Bocca, B. Amino acid profile in the ripening of Grana Padano cheese: A NMR study. Food Chem. 2000, 71, 495–502. [Google Scholar] [CrossRef]
- Piras, C.; Marincola, F.C.; Savorani, F.; Engelsen, S.B.; Cosentino, S.; Viale, S.; Pisano, M.B. A NMR metabolomics study of the ripening process of the Fiore Sardo cheese produced with autochthonous adjunct cultures. Food Chem. 2013, 141, 2137–2147. [Google Scholar] [CrossRef]
- Consonni, R.; Cagliani, L.R. Ripening and geographical characterization of Parmigiano Reggiano cheese by 1H NMR spectroscopy. Talanta 2008, 76, 200–205. [Google Scholar] [CrossRef]
- Brescia, M.A.; Monfreda, M.; Buccolieri, A.; Carrino, C. Characterisation of the geographical origin of buffalo milk and mozzarella cheese by means of analytical and spectroscopic determinations. Food Chem. 2005, 89, 139–147. [Google Scholar] [CrossRef]
- Schievano, E.; Pasini, G.; Cozzi, G.; Mammi, S. Identification of the production chain of Asiago d’Allevo cheese by nuclear magnetic resonance spectroscopy and principal component analysis. J. Agric. Food Chem. 2008, 56, 7208–7214. [Google Scholar] [CrossRef]
- Shintu, L.; Caldarelli, S. Toward the determination of the geographical origin of emmental(er) cheese via high resolution MAS NMR: A preliminary investigation. J. Agric. Food Chem. 2006, 54, 4148–4154. [Google Scholar] [CrossRef]
- Mazzei, P.; Piccolo, A. 1H HRMAS-NMR metabolomic to assess quality and traceability of mozzarella cheese from Campania buffalo milk. Food Chem. 2012, 132, 1620–1627. [Google Scholar] [CrossRef]
- Rodrigues, D.; Santos, C.H.; Rocha-Santos, T.A.P.; Gomes, A.M.; Goodfellow, B.J.; Freitas, A.C. Metabolic profiling of potential probiotic or synbiotic cheeses by Nuclear Magnetic Resonance (NMR) spectroscopy. J. Agric. Food Chem. 2011, 59, 4955–4961. [Google Scholar] [CrossRef] [PubMed]
- Dudley, E.G.; Steele, J.L. Succinate production and citrate catabolism by Cheddar cheese nonstarter lactobacilli. J. Appl. Microbiol. 2005, 98, 14–23. [Google Scholar] [CrossRef]
- Warmke, R.; Belitz, H.D.; Grosch, W. Evaluation of taste compounds of Swiss cheese (Emmentaler). Z. Für Lebensm. Unters. Und Forsch. 1996, 203, 230–235. [Google Scholar] [CrossRef]
- Fox, P.F.; Lucey, J.A.; Cogan, T.M. Glycolysis and related reactions during cheese manufacture and ripening. Crit. Rev. Food Sci. Nutr. 1990, 29, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Yerlikaya, O.; Gucer, L.; Akan, E.; Meric, S.; Aydin, E.; Kinik, O. Benzoic acid formation and its relationship with microbial properties in traditional Turkish cheese varieties. Food Biosci. 2021, 41, 101040. [Google Scholar] [CrossRef]
- Moatsou, G.A.; Kandarakis, I.G.; Georgala, A.K.; Alichanidis, E.S.; Anifantakis, E.M. Effect of starters on proteolysis of Graviera Kritis cheese. Lait 1999, 79, 303–315. [Google Scholar] [CrossRef]
- Hogenboom, J.A.; D’Incecco, P.; Fuselli, F.; Pellegrino, L. Ion-Exchange Chromatographic Method for the Determination of the Free Amino Acid Composition of Cheese and Other Dairy Products: An Inter-Laboratory Validation Study. Food Anal. Methods 2017, 10, 3137–3148. [Google Scholar] [CrossRef]
- Poveda, J.M.; Cabezas, L.; McSweeney, P.L.H. Free amino acid content of Manchego cheese manufactured with different starter cultures and changes throughout ripening. Food Chem. 2004, 84, 213–218. [Google Scholar] [CrossRef]
- Michaelidou, A.; Katsiari, M.; Voutsinas, L.; Kondyli, E.; Alichanidis, E. Effect of commercial adjunct cultures on proteolysis in low-fat Kefalograviera-type cheese. Int. Dairy J. 2003, 13, 743–753. [Google Scholar] [CrossRef]
- Carafa, I.; Stocco, G.; Nardin, T.; Larcher, R.; Bittante, G.; Tuohy, K.; Franciosi, E. Production of Naturally γ-Aminobutyric Acid-Enriched Cheese Using the Dairy Strains Streptococcus thermophilus 84C and Lactobacillus brevis DSM 32386. Front. Microbiol. 2019, 10, 93. [Google Scholar] [CrossRef]
- Valsamaki, K.; Michaelidou, A.; Polychroniadou, A. Biogenic amine production in Feta cheese. Food Chem. 2000, 71, 259–266. [Google Scholar] [CrossRef]
- Diana, M.; Rafecas, M.; Arco, C.; Quílez, J. Free amino acid profile of Spanish artisanal cheeses: Importance of gamma-aminobutyric acid (GABA) and ornithine content. J. Food Compos. Anal. 2014, 35, 94–100. [Google Scholar] [CrossRef]
- Dhiman, T.R.; Anand, G.R.; Satter, L.D.; Pariza, M.W. Conjugated Linoleic Acid Content of Milk from Cows Fed Different Diets1. J. Dairy Sci. 1999, 82, 2146–2156. [Google Scholar] [CrossRef] [PubMed]
- Mollica, M.P.; Trinchese, G.; Cimmino, F.; Penna, E.; Cavaliere, G.; Tudisco, R.; Musco, N.; Manca, C.; Catapano, A.; Monda, M.; et al. Milk Fatty Acid Profiles in Different Animal Species: Focus on the Potential Effect of Selected PUFAs on Metabolism and Brain Functions. Nutrients 2021, 13, 1111. [Google Scholar] [CrossRef]
- Vatavali, K.; Kosma, I.; Louppis, A.; Gatzias, I.; Badeka, A.V.; Kontominas, M.G. Characterisation and differentiation of geographical origin of Graviera cheeses produced in Greece based on physico-chemical, chromatographic and spectroscopic analyses, in combination with chemometrics. Int. Dairy J. 2020, 110, 104799. [Google Scholar] [CrossRef]
- Zlatanos, S.; Laskaridis, K. Variation in the conjugated linoleic acid content of three traditional greek cheeses during a 1-year period. J. Food Qual. 2009, 32, 84–95. [Google Scholar] [CrossRef]
- Zlatanos, S.; Laskaridis, K.; Feist, C.; Sagredos, A. CLA content and fatty acid composition of Greek Feta and hard cheeses. Food Chem. 2002, 78, 471–477. [Google Scholar] [CrossRef]
- Vigli, G.; Philippidis, A.; Spyros, A.; Dais, P. Classification of edible oils by employing 31P and 1H NMR spectroscopy in combination with multivariate statistical analysis. A proposal for the detection of seed oil adulteration in virgin olive oils. J. Agric. Food Chem. 2003, 51, 5715–5722. [Google Scholar] [CrossRef]
- Tsafrakidou, P.; Bozoudi, D.; Pavlidou, S.; Kotzamanidis, C.; Hatzikamari, M.; Zdragas, A.; Litopoulou-Tzanetaki, E. Technological, phenotypic and genotypic characterization of lactobacilli from Graviera Kritis PDO Greek cheese, manufactured at two traditional dairies. Food Sci. Technol. Leb. 2016, 68, 681–689. [Google Scholar] [CrossRef]
- Abd El-Salam, M.H.; El-Shibiny, S. Conjugated linoleic acid and vaccenic acid contents in cheeses: An overview from the literature. J. Food Compos. Anal. 2014, 33, 117–126. [Google Scholar] [CrossRef]
- Ralli, E.; Amargianitaki, M.; Manolopoulou, E.; Misiak, M.; Markakis, G.; Tachtalidou, S.; Kolesnikova, A.; Dais, P.; Spyros, A. NMR Spectroscopy Protocols for Food Metabolomics Applications. Methods Mol. Biol. 2018, 1738, 203–211. [Google Scholar] [CrossRef] [PubMed]
| Compound | Assignment | 1H ppm | 13C ppm | |
|---|---|---|---|---|
| 1 | Acetate | α-CH3 | 1.92 | 26.1 |
| 2 | Alanine | β-CH3 | 1.48 | 18.9 |
| α-CH | 3.78 | 53.7 | ||
| 3 | Arginine | γ-CH2 | 1.68 | 26.8 |
| β,β′-CH2 | 1.90 | 30.1 | ||
| δ,δ′-CH2 | 3.23 | 43.1 | ||
| α-CH | 3.74 | 57.1 | ||
| 4 | Asparagine | β-CH2 | 2.89 | 37.4 |
| α-CH | 4.00 | 51.5 | ||
| 5 | Aspartate | β′-CH2 | 2.80 | 39.0 |
| α-CH | 3.90 | 53.6 | ||
| 6 | Benzoate | 2,6-CH | 7.87 | 131.8 |
| 7 | Choline | α-CH | 3.21 | 56.7 |
| 8 | Citrate | 2,4-CH2 | 2.72/2.54 | 47.6 |
| 9 | Cytosine | 2-CH2 | 6.00 | - |
| 10 | Formate | HCOO | 8.50 | 173.5 |
| 11 | GABA | β-CH2 | 1.91 | 26.4 |
| α-CH2 | 2.30 | 36.4 | ||
| γ-CH2 | 3.02 | 42.0 | ||
| 12 | Glutamate | β,β′-CH2 | 2.09 | 30.1 |
| γ,γ′-CH2 | 2.36 | 36.3 | ||
| α-CH | 3.76 | 57.2 | ||
| 13 | Glutamine | β-CH2 | 2.14 | 28.9 |
| γ-CH2 | 2.46 | 33.6 | ||
| 14 | Glycerol | 2-CH | 3.79 | 74.9 |
| 1,3-CH2 | 3.65/3.56 | 65.4 | ||
| 15 | Glycine | α-CH2 | 3.56 | 44.2 |
| 16 | Isoleucine | δ-CH3 | 0.94 | 13.8 |
| β′-CH3 | 1.02 | 17.6 | ||
| γ′-CH | 1.27 | 27.1 | ||
| γ-CH | 1.46 | 27.1 | ||
| β-CH | 1.99 | 38.5 | ||
| α-CH | 3.68 | 62.4 | ||
| 17 | Lactate | β-CH3 | 1.34 | 23.0 |
| α-CH | 4.16 | 71.5 | ||
| 18 | Leucine | δ,δ′-CH3 | 0.96 | 20.9/24.6 |
| β-CH2 | 1.72 | 42.5 | ||
| γ-CH | 1.72 | 27.1 | ||
| α-CH | 3.74 | 56.2 | ||
| 20 | Lysine | γ-CH2 | 1.49 | 24.4 |
| β-CH2 | 1.72 | 29.2 | ||
| δ-CH2 | 1.90 | 33.1 | ||
| ε-CH2 | 3.03 | 41.9 | ||
| α-CH | 3.75 | 57.1 | ||
| 21 | Methionine | δ-CH3 | 2.14 | 16.7 |
| γ-CH2 | 2.65 | 31.5 | ||
| β-CH2 | 2.14 | 32.4 | ||
| α-CH | 3.85 | 56.7 | ||
| 22 | Ornithine | δ-CH2 | 3.06 | 39.3 |
| γ-CH2 | 1.77 | 25.5 | ||
| β-CH2 | 1.95 | 30.2 | ||
| α-CH | 3.79 | 57.3 | ||
| 23 | Phenylalanine | β-CH2 | 3.14/3.29 | 39.2 |
| α-CH2 | 4.00 | 58.9 | ||
| 2,6-CH | 7.33 | 131.8 | ||
| 4-CH | 7.37 | 131.9 | ||
| 3,5-CH | 7.43 | 132.1 | ||
| 24 | Proline | 3-CH2 | 2.01 | 27.3 |
| 2-CH2 | 2.35 | 32.0 | ||
| 4-CH | 3.35 | 48.9 | ||
| 4′-CH | 3.41 | 48.9 | ||
| 1-CH | 4.14 | 64.1 | ||
| 25 | Serine | α-CH | 3.86 | 56.0 |
| β,β′-CH2 | 3.97 | 63.1 | ||
| 26 | Succinate | 2,3 CH2 | 2.41 | 36.2 |
| 27 | Threonine | γ-CH3 | 1.33 | 19.5 |
| α-CH | 3.62 | 62.2 | ||
| β-CH | 4.27 | 66.0 | ||
| 28 | Tryptophan | 4-CH | 7.73 | 118.0 |
| 29 | Tyramine | α,β-CH2 | 2.93/3.27 | 34.8 |
| 3,5-H | 6.90 | 118.8 | ||
| 2,6-H | 7.22 | 133.7 | ||
| 30 | Tyrosine | 3,5-H | 6.90 | 118.8 |
| 2,6-H | 7.19 | 133.7 | ||
| 31 | Uracil | 2-CH | 5.80 | 103.8 |
| 1-CH | 7.54 | 146.3 | ||
| 32 | Valine | γ-CH3 | 1.00 | 19.5 |
| γ′-CH3 | 1.05 | 20.7 | ||
| β-CH | 2.28 | 31.9 | ||
| α-CH | 3.63 | 63.2 |
| Compound | Assignment | 1H ppm | 13C ppm | Letter Code | ||
|---|---|---|---|---|---|---|
| 1 | Sterols | –CH3 | 0.68 | 11.7 | ||
| 2 | FA except n-3/Butyric | ω1 | –CH3 | 0.88 | 14.1 | F |
| 3 | Butyric acid | H4 | –CH3 | 0.94 | 13.8 | I |
| 4 | ω-3 FA | ω1 | –CH3 | 0.97 | 14.1 | Ε |
| 5 | All FA | –(CH2)n- | 1.26 | 29.4 | ||
| 6 | All FA | H3 | –O–CO–CH2–CH2− | 1.61 | 24.9 | D |
| 7 | UFA-cis | –CH2–CH=CH– | 1.97 | 32.4 | C | |
| 8 | UFA-trans | –CH2–CH=CH– | 2.02 | 27.1 | C | |
| 9 | All FA | H2 | –O–CO–CH2–CH2 | 2.30 | 34.0 | B |
| 10 | PUFA (Linoleic) | H11 | =CH–CH2–CH= | 2.77 | 25.6 | A |
| 11 | PUFA (Linolenic) | H11 H14 | =CH–CH2–CH= | 2.80 | 25.6 | A |
| 12 | 1,2-Diglycerides | HO–CH2–CH– | 3.72 | 61.6 | ||
| 13 | 1,3-Diglycerides | –CH2–O–CO– | 3.98 | 64.3 | ||
| 14 | Triglycerides | –CH2–O–CO– | 4.14 | 62.1 | ||
| 15 | Triglycerides | –CH2–O–OC– | 4.30 | 62.1 | ||
| 16 | Caproleic acid | H10a | =CH | 4.91 | 114.3 | H |
| 17 | Caproleic acid | H10b | =CH | 4.97 | 114.3 | H |
| 18 | 1,2-Diglycerides | –CH–O–CO– | 5.09 | 72.0 | ||
| 19 | Triglycerides | –CH–O–CO | 5.26 | 68.7 | ||
| 20 | UFA-cis | –CH=CH– | 5.33 | 129.8 | ||
| 21 | UFA-trans | –CH=CH– | 5.37 | 130.3 | ||
| 22 | CLA | H12 | –CH= | 5.63 | 134.6 | |
| 23 | Caproleic acid | H9 | –CH=CH2 | 5.78 | 139.0 | |
| 24 | CLA | H10 | –CH= | 5.92 | 128.8 | |
| 25 | CLA | H11 | –CH= | 6.27 | 125.6 | G |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ralli, E.; Spyros, A. A Study of Greek Graviera Cheese by NMR-Based Metabolomics. Molecules 2023, 28, 5488. https://doi.org/10.3390/molecules28145488
Ralli E, Spyros A. A Study of Greek Graviera Cheese by NMR-Based Metabolomics. Molecules. 2023; 28(14):5488. https://doi.org/10.3390/molecules28145488
Chicago/Turabian StyleRalli, Evangelia, and Apostolos Spyros. 2023. "A Study of Greek Graviera Cheese by NMR-Based Metabolomics" Molecules 28, no. 14: 5488. https://doi.org/10.3390/molecules28145488
APA StyleRalli, E., & Spyros, A. (2023). A Study of Greek Graviera Cheese by NMR-Based Metabolomics. Molecules, 28(14), 5488. https://doi.org/10.3390/molecules28145488






