The Interfacial Dilational Rheology Properties of Betaine Solutions: Effect of Anionic Surfactant and Polymer
Abstract
1. Introduction
2. Results and Discussion
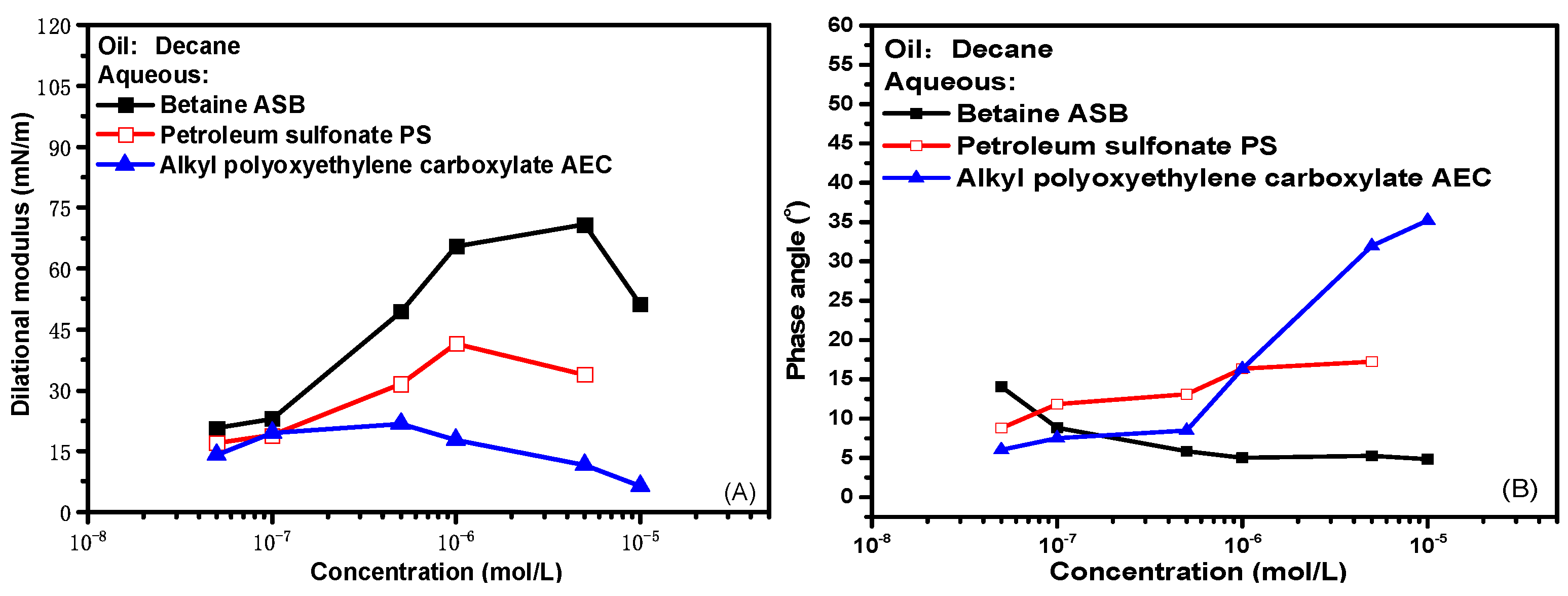
2.1. Interfacial Dilational Rheological Properties of Surfactant Solution
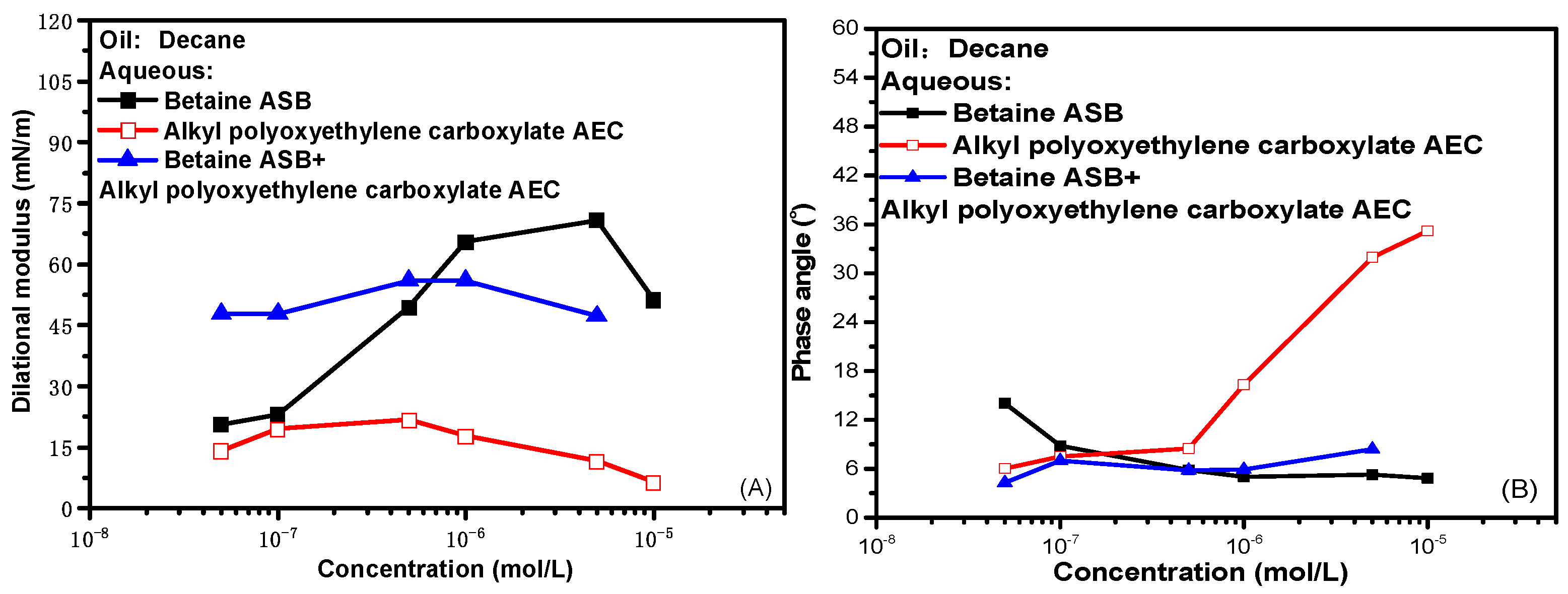
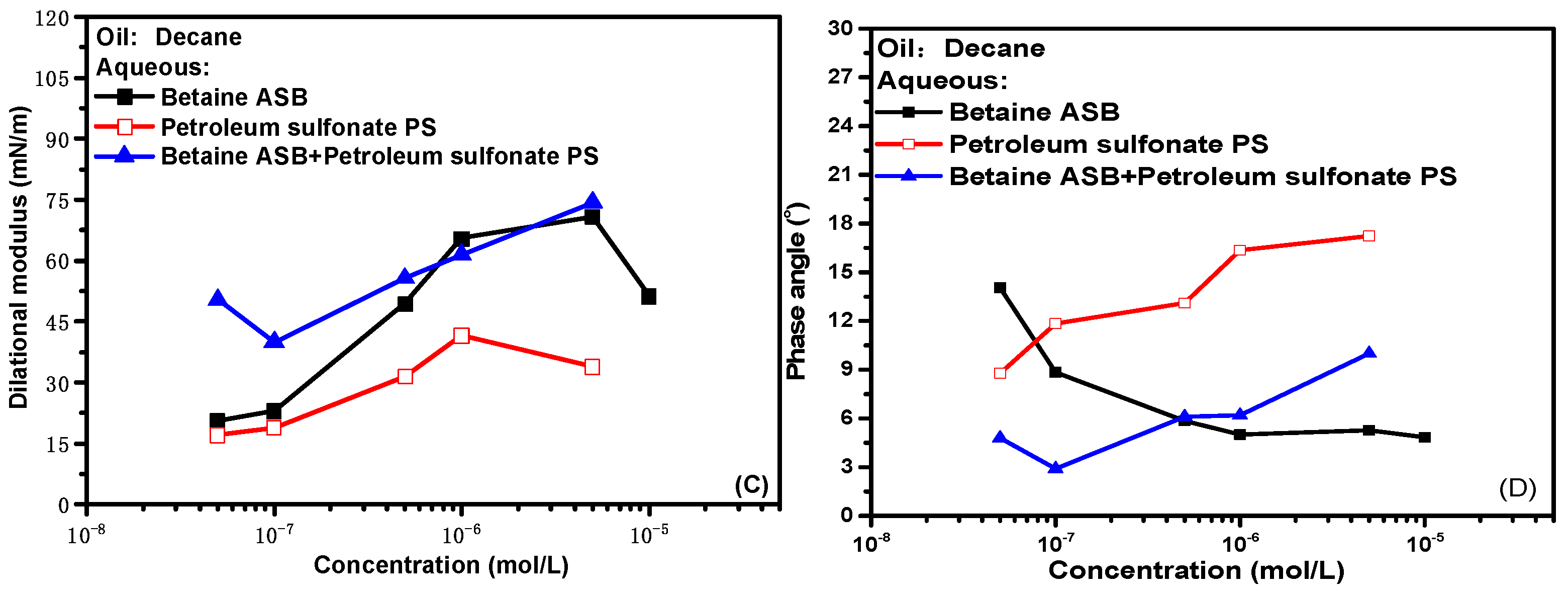
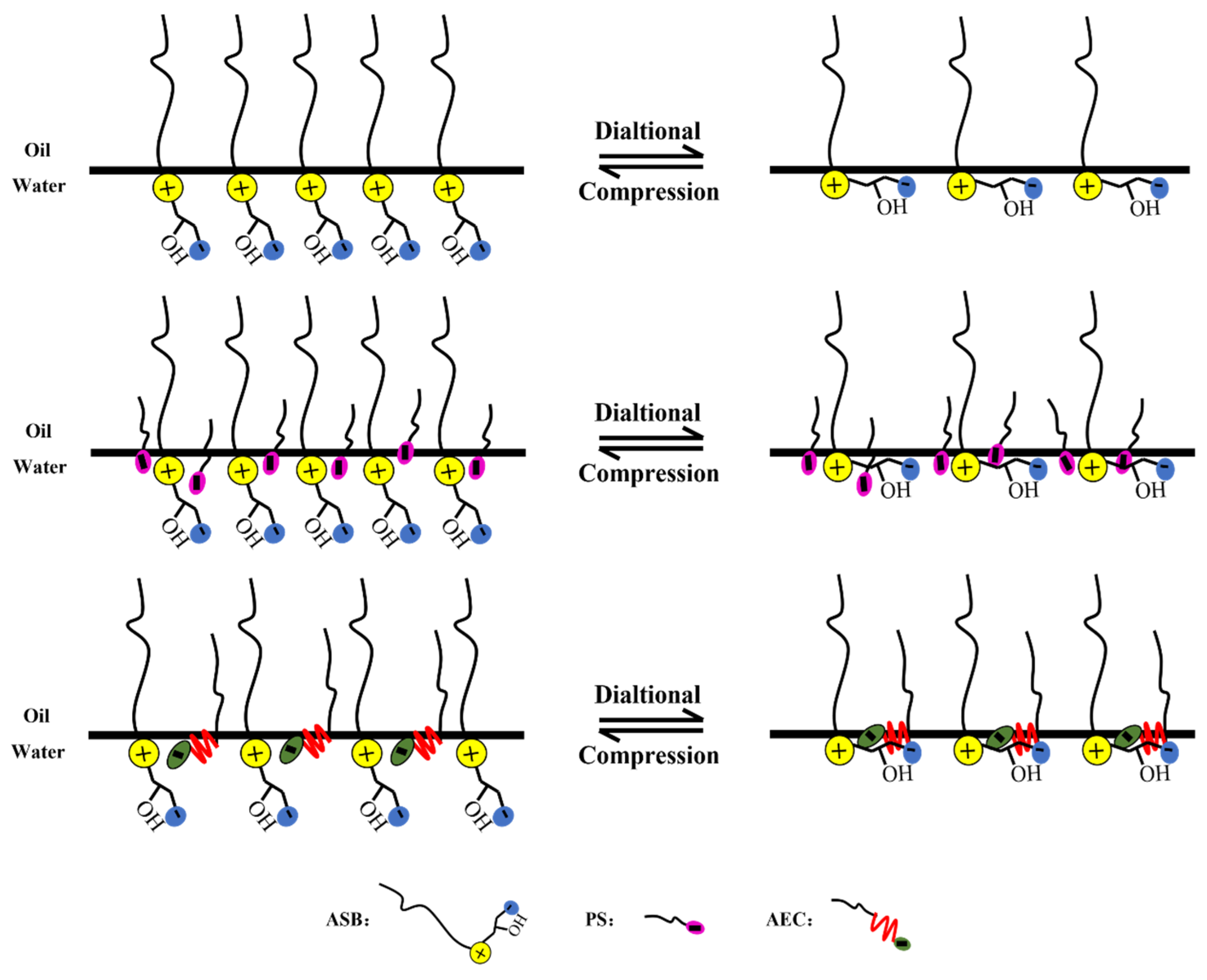
2.2. Dynamic Interfacial Dilational Rheological Properties of Surfactant Complex System
2.3. The Effect of Frequency on the Interfacial Dilational Rheology Properties of the Compound System
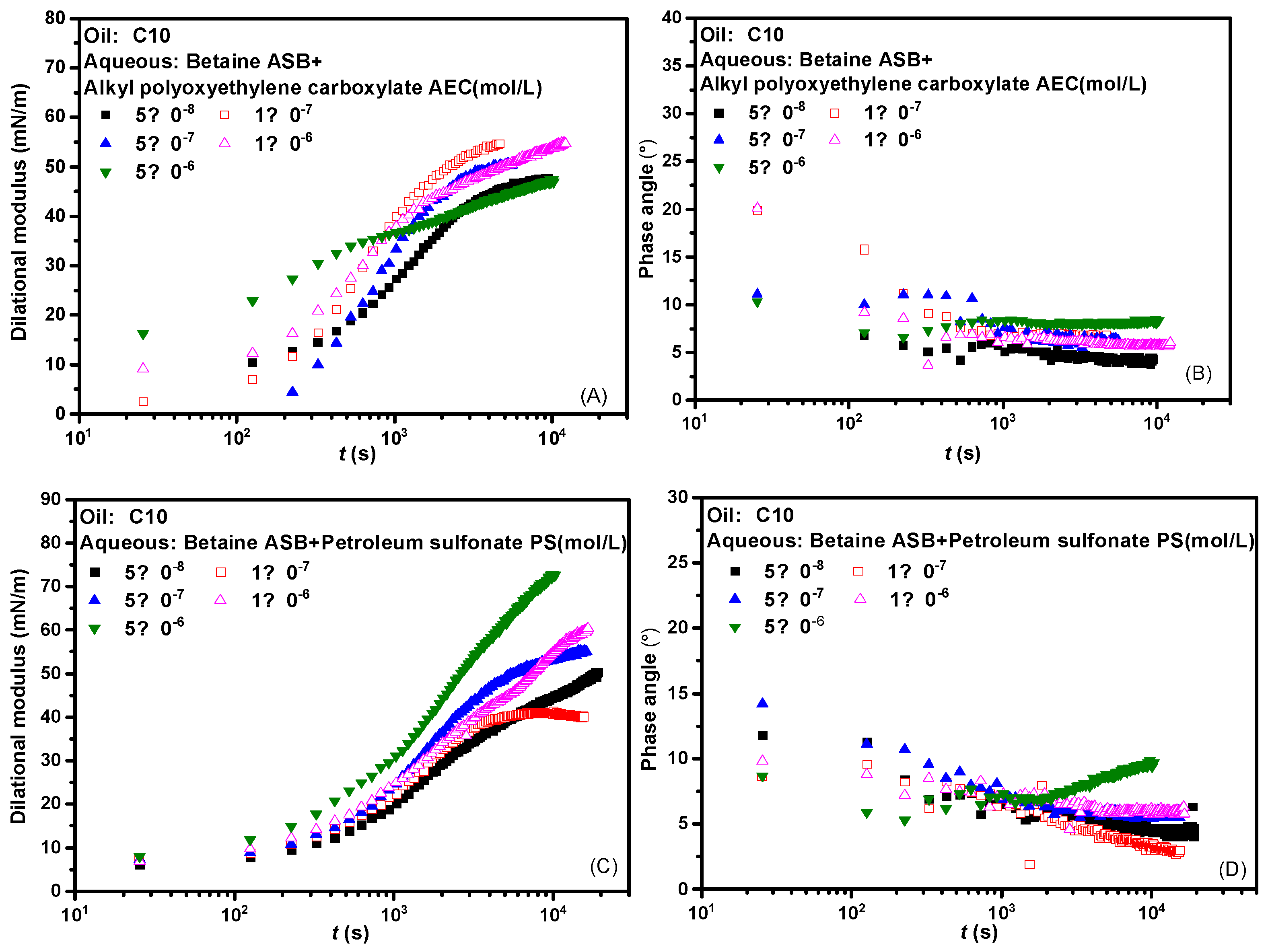
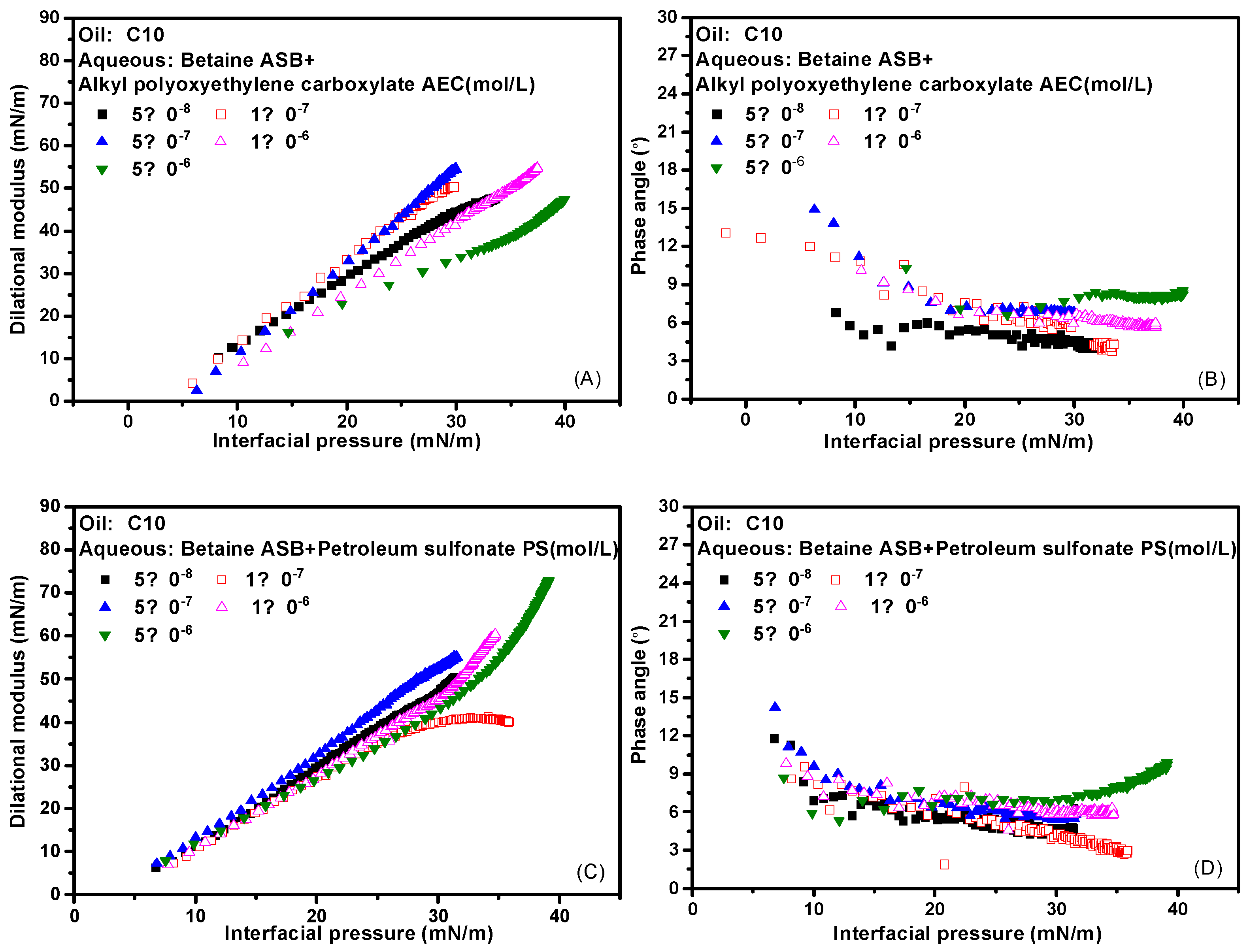
2.4. Effect of Concentration on Interfacial Dilational Rheology Properties of Complex System
2.5. Effect of Interfacial Pressure on Interfacial Dilational Rheology Properties of Mixed System
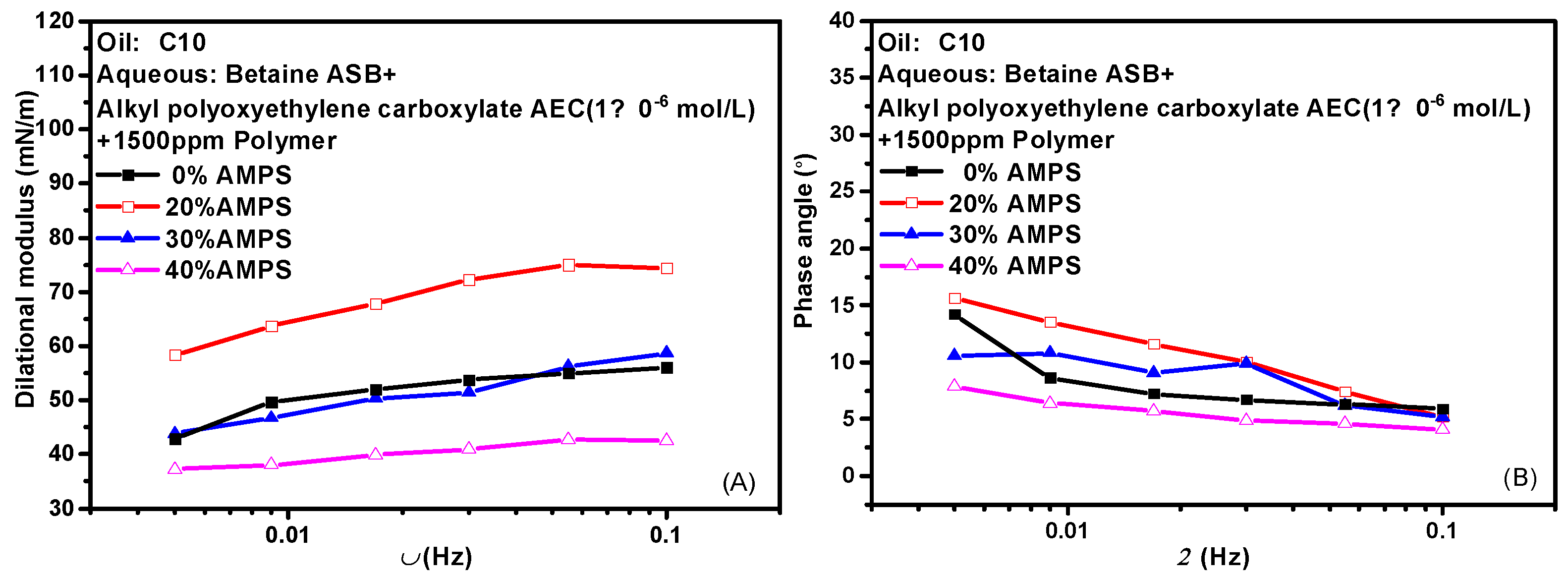
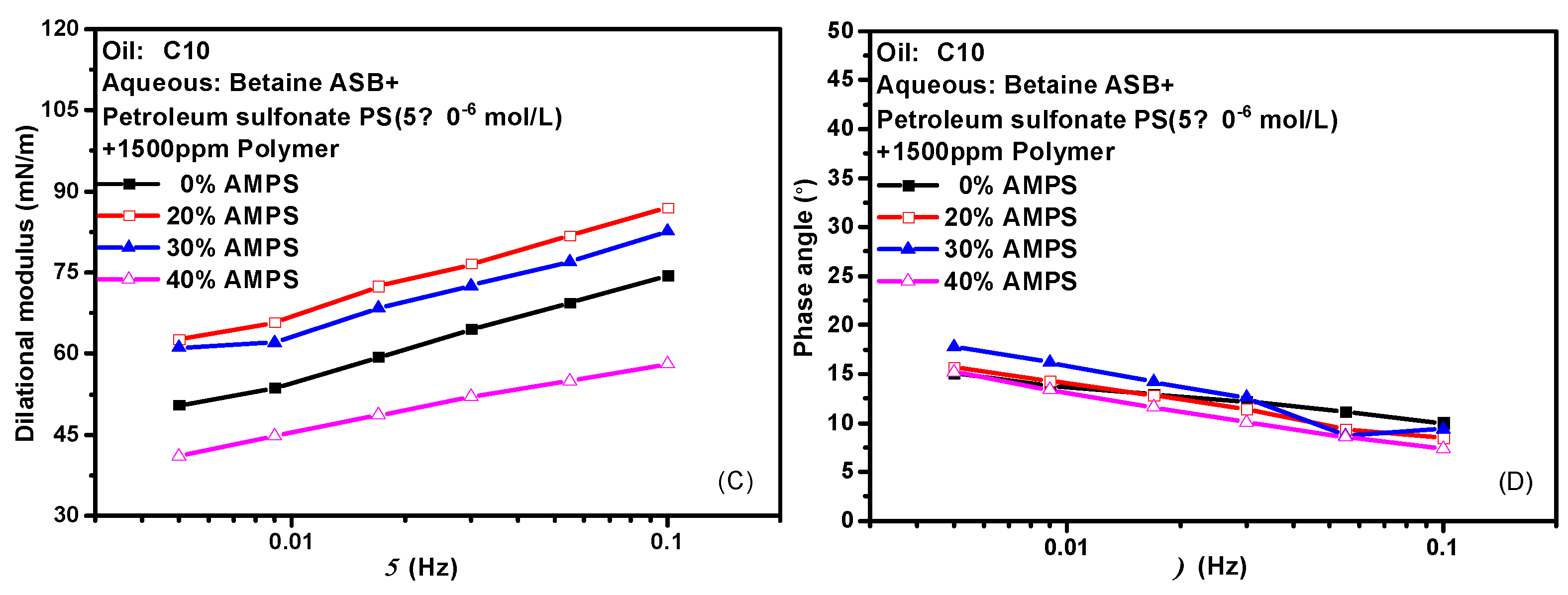
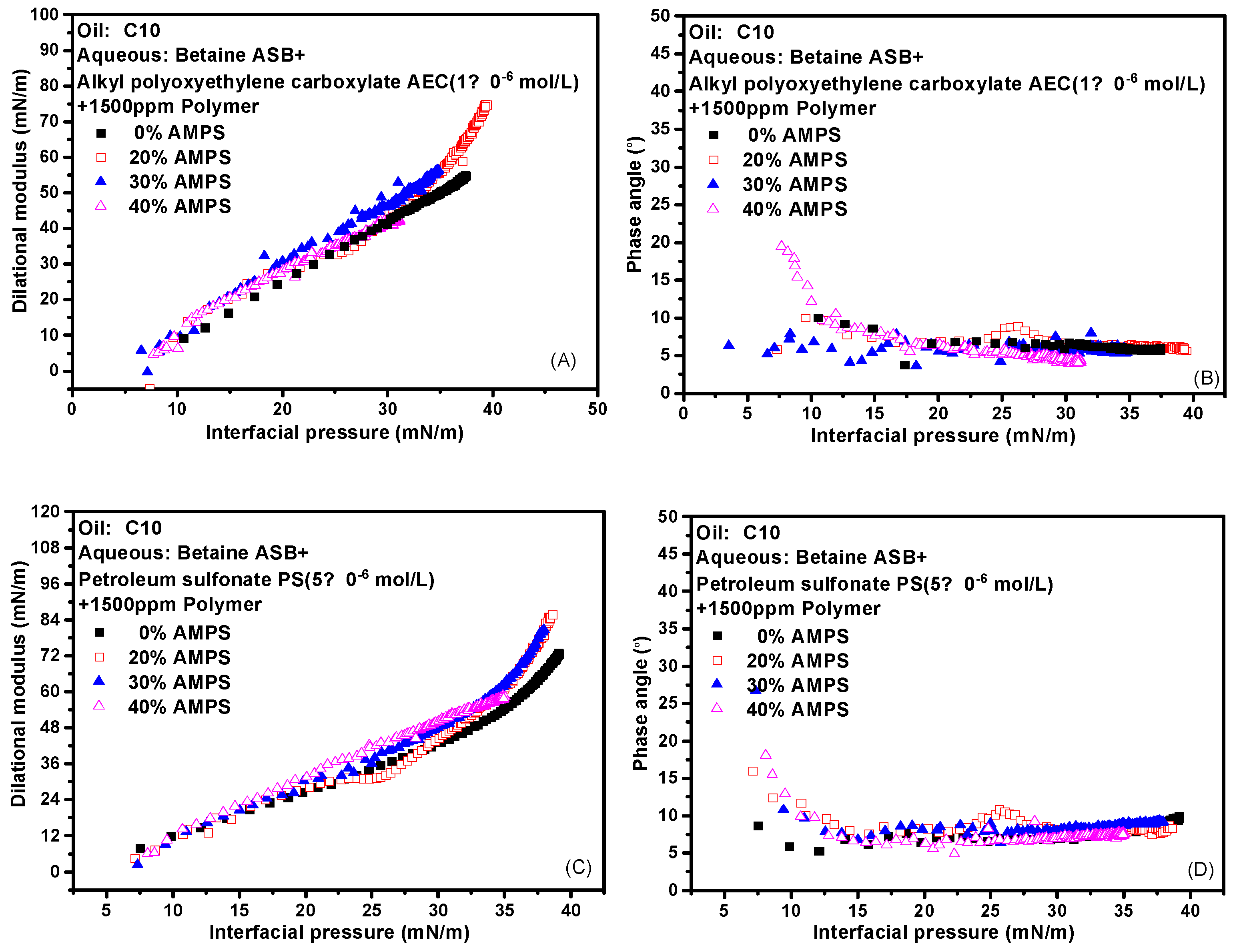
2.6. Effect of Polymer on Interfacial Dilational Rheology Properties of Compound System
3. Experiment Section
3.1. Materials
3.2. Experimental Method
3.3. Theoretical Background
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miller, R.; Ferri, J.K.; Javadi, A.; Krägel, J.; Mucic, N.; Wüstneck, R. Rheology of interfacial layers. Colloid Polym. Sci. 2010, 288, 937–950. [Google Scholar] [CrossRef]
- Rodriguez Patino, J.M.; Cejudo Fernandez, M.; Carrera Sanchez, C.; Rodriguez Nino, M.R. Structural and shear characteristics of adsorbed sodium caseinate and monoglyceride mixed monolayers at the air-water interface. J. Colloid Interf. Sci. 2007, 313, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yu, K.; Tsuji, T.; Jha, R.; Zuo, Y.Y. Determining the surface dilational rheology of surfactant and protein films with a droplet waveform generator. J. Colloid Interface Sci. 2019, 537, 547–553. [Google Scholar] [CrossRef]
- Cao, C.; Zhang, L.; Zhang, X.-X.; Du, F.-P. Effect of gum arabic on the surface tension and surface dilational rheology of trisiloxane surfactant. Food Hydrocoll. 2013, 30, 456–462. [Google Scholar] [CrossRef]
- Javadi, A.; Dowlati, S.; Shourni, S.; Miller, R.; Kraume, M.; Kopka, K.; Eckert, K. Experimental techniques to study protein-surfactant interactions: New insights into competitive adsorptions via drop subphase and interface exchange. Adv. Colloid. Interface Sci. 2022, 301, 102601. [Google Scholar] [CrossRef] [PubMed]
- Madhumitha, D.; Jaganathan, M.; Dhathathreyan, A.; Miller, R. Balancing soft elasticity and low surface polarity in films of charged BSA capsules at air/fluid interface. Colloids Surf. B Biointerf. 2016, 146, 161–170. [Google Scholar]
- Zhou, H.; Luo, Q.; Gong, Q.-T.; Liu, Z.-Y.; Liu, M.; Zhang, L.; Zhang, L.; Zhao, S. Interfacial dilational properties of di-substituted alkyl benzene sulfonates at kerosene/water and crude oil/water interfaces. Colloids Surf. A Physicochem. Eng. Asp. 2017, 520, 561–569. [Google Scholar] [CrossRef]
- Niu, Q.; Dong, Z.; Lv, Q.; Zhang, F.; Shen, H.; Yang, Z.; Lin, M.; Zhang, J.; Xiao, K. Role of interfacial and bulk properties of long-chain viscoelastic surfactant in stabilization mechanism of CO2 foam for CCUS. J. CO2 Utilizat. 2022, 66, 102297–102311. [Google Scholar] [CrossRef]
- Akanno, A.; Guzman, E.; Fernandez-Pena, L.; Ortega, F.; Gubio, R.G. Surfactant-Like Behavior for the Adsorption of Mixtures of a Polycation and Two Different Zwitterionic Surfactants at the Water/Vapor Interface. Molecules 2019, 24, 3442–3458. [Google Scholar] [CrossRef]
- Chen, T.; Zhang, G.; Jiang, P.; Ge, J. Dilational Rheology at Air/Water Interface and Molecular Dynamics Simulation Research of Hydroxyl Sulfobetaine Surfactant. J. Dispers. Sci. Technol. 2014, 35, 448–455. [Google Scholar] [CrossRef]
- Cao, J.-H.; Zhou, Z.-H.; Xu, Z.-C.; Zhang, Q.; Li, S.-H.; Cui, H.-B.; Zhang, L.; Zhang, L. Synergism/Antagonism between Crude Oil Fractions and Novel Betaine Solutions in Reducing Interfacial Tension. Energy Fuels 2016, 30, 924–932. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhou, Z.-H.; Dong, L.-F.; Wang, H.-Z.; Cai, H.-Y.; Zhang, F.; Zhang, L.; Zhang, L.; Zhao, S. Dilational rheological properties of sulphobetaines at the water–decane interface: Effect of hydrophobic group. Colloids Surf. A Physicochem. Eng. Asp. 2014, 455, 97–103. [Google Scholar] [CrossRef]
- Gizzatov, A.; Mashat, A.; Kosynkin, D.; Alhazza, N.; Kmetz, A.; Eichmann, S.L.; Abdel-Fattah, A.I. Nanofluid of Petroleum Sulfonate Nanocapsules for Enhanced Oil Recovery in High-Temperature and High-Salinity Reservoirs. Energy Fuels 2019, 33, 11567–11573. [Google Scholar] [CrossRef]
- Li, H.; Zhu, W.; Song, Z. 2-D pore-scale oil recovery mechanisms of the anionic and nonionic surfactants. Colloids Surf. A Physicochem. Eng. Asp. 2022, 655, 130245–130253. [Google Scholar] [CrossRef]
- Somoza, A.; Tafur, N.; Arce, A.; Soto, A. Design and performance analysis of a formulation based on SDBS and ionic liquid for EOR in carbonate reservoirs. J. Pet. Sci. Eng. 2022, 209, 109856–109867. [Google Scholar] [CrossRef]
- Luan, H.; Zhou, Z.; Xu, C.; Bai, L.; Wang, X.; Han, L.; Zhang, Q.; Li, G. Study on the Synergistic Effects between Petroleum Sulfonate and a Nonionic–Anionic Surfactant for Enhanced Oil Recovery. Energies 2022, 15, 1177–1189. [Google Scholar] [CrossRef]
- Scerbacova, A.; Ivanova, A.; Grishin, P.; Cheremisin, A.; Tokareva, E.; Tkachev, I.; Sansiev, G.; Fedorchenko, G.; Afanasiev, I. Application of alkalis, polyelectrolytes, and nanoparticles for reducing adsorption loss of novel anionic surfactant in carbonate rocks at high salinity and temperature conditions. Colloids Surf. A Physicochem. Eng. Asp. 2022, 653, 129996–131000. [Google Scholar] [CrossRef]
- Zhang, T.; Li, Z.; Gao, M.; Xu, Z.; Adenutsi, C.D.; You, Q. New insights into the synergism between silica nanoparticles and surfactants on interfacial properties: Implications for spontaneous imbibition in tight oil reservoirs. J. Pet. Sci. Eng. 2022, 215, 110647–110661. [Google Scholar] [CrossRef]
- Chen, D.; Li, F.; Gao, Y.; Yang, M. Pilot Performance of Chemical Demulsifier on the Demulsification of Produced Water from Polymer/Surfactant Flooding in the Xinjiang Oilfield. Water 2018, 10, 1874–1883. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, L.; Zhang, L.; Luo, L.; Zhao, S.; Yu, J.-Y. Effect of Oxyethylene Numbers on Surface Dilational Properties of Alkyl Secondary Alcohol Ethoxylates. J. Dispers. Sci. Technol. 2011, 32, 490–497. [Google Scholar] [CrossRef]
- Khoramian, R.; Kharrat, R.; Golshokooh, S. The development of novel nanofluid for enhanced oil recovery application. Fuel 2022, 311, 122558–122577. [Google Scholar] [CrossRef]
- Mohamed, A.I.A.; Khishvand, M.; Piri, M. The role of injection fluid elasticity in microscopic displacement efficiency of residual non-wetting phase: An in-situ experimental investigation. Fuel 2023, 333, 126180–126195. [Google Scholar] [CrossRef]
- Sun, Q.; Hu, F.T.; Han, L.; Zhu, X.Y.; Zhang, F.; Ma, G.Y.; Zhang, L.; Zhou, Z.H.; Zhang, L. The Synergistic Effects between Sulfobetaine and Hydrophobically Modified Polyacrylamide on Properties Related to Enhanced Oil Recovery. Molecules 2023, 28, 1787–1804. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Dai, Y.-H.; Zhang, L.; Luo, L.; Chu, Y.-P.; Zhao, S.; Li, M.-Z.; Wang, E.-J.; Yu, J.-Y. Hydrophobically Modified Associating Polyacrylamide Solutions: Relaxation Processes and Dilational Properties at the Oil-Water Interface. Macromolecules 2004, 37, 2930–2937. [Google Scholar] [CrossRef]
- Belhaj, A.F.; Aris, B.M.; Shuhli, J.; Elraies, K.A.; Mahmood, S.M.; Maulianda, B.; Alnarabiji, M.S. Partitioning behaviour of novel surfactant mixture for high reservoir temperature and high salinity conditions. Energy 2020, 198, 117319–117333. [Google Scholar] [CrossRef]
- Kurnia, I.; Zhang, G.; Han, X.; Yu, J. Zwitterionic-anionic surfactant mixture for chemical enhanced oil recovery without alkali. Fuel 2020, 259, 116236–116245. [Google Scholar] [CrossRef]
- Towesend, V.d.J.; Creatto, E.J.; Pedroni, L.G.; Pérez-Gramatges, A. Synergism in binary surfactant mixtures containing a pH-responsive surfactant towards enhanced foam stability in brine at high pressure and high temperature conditions. Colloids Surf. A Physicochem. Eng. Asp. 2022, 648, 129433–129445. [Google Scholar] [CrossRef]
- Sun, Q.; Zhou, Z.-H.; Zhang, Q.; Zhang, F.; Ma, G.-Y.; Zhang, L.; Zhang, L. Effect of Electrolyte on Synergism for Reducing Interfacial Tension between Betaine and Petroleum Sulfonate. Energy Fuels 2020, 34, 3188–3198. [Google Scholar] [CrossRef]
- Sun, H.-Q.; Guo, Z.-Y.; Cao, X.-L.; Zhu, Y.-W.; Pan, B.-L.; Liu, M.; Zhang, L.; Zhang, L. Interfacial interactions between oleic acid and betaine molecules at decane-water interface: A study of dilational rheology. J. Mol. Liq. 2020, 316, 113784–113792. [Google Scholar] [CrossRef]
- Fainerman, V.B.; Petkov, J.T.; Miller, R. Surface Dilational Viscoelasticity of C14EO8 Micellar Solution Studied by Bubble Profile Analysis Tensiometry. Langmuir 2008, 24, 6447–6452. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.-L.; Feng, J.; Guo, L.-L.; Zhu, Y.-w.; Zhang, L.; Zhang, L.; Luo, L.; Zhao, S. Dynamic surface dilational properties of anionic Gemini surfactants with polyoxyethylene spacers. Colloids Surf. A Physicochem. Eng. Asp. 2016, 490, 41–48. [Google Scholar] [CrossRef]
- Huang, Y.-P.; Zhang, L.; Zhang, L.; Luo, L.; Zhao, S.; Yu, J.-Y. Dynamic Interfacial Dilational Properties of Hydroxy-Substituted Alkyl Benzenesulfonates. J. Phys. Chem. B 2007, 111, 5640–5647. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, X.C.; Gong, Q.T.; Zhang, L.; Luo, L.; Zhao, S.; Yu, J.Y. Interfacial dilational properties of tri-substituted alkyl benzene sulfonates at air/water and decane/water interfaces. J. Colloid Interface Sci. 2008, 327, 451–458. [Google Scholar] [CrossRef] [PubMed]
- He, H.-J.; Xiao, H.; Zhou, Z.-H.; Zhang, Q.; Gao, M.; Chen, X.-F.; Zhang, L.; Zhang, L. Effect of oxyethylene and oxypropylene groups on the interfacial structure and property of extended surfactants: Molecular simulation and experimental study. J. Mol. Liq. 2023, 382, 121944–121955. [Google Scholar] [CrossRef]
- Wang, Z.-S.; Zhou, Z.-H.; Han, L.; Chen, X.; He, H.-J.; Zhang, Q.; Xu, Z.-c.; Gong, Q.-t.; Zhang, L.; Ma, G.-Y.; et al. The mechanism for lowering interfacial tension by extended surfactant containing ethylene oxide and propylene oxide groups. J. Mol. Liq. 2022, 359, 119364–119374. [Google Scholar] [CrossRef]
- Fainerman, V.B.; Kovalchuk, V.I.; Aksenenko, E.V.; Zinkovych, I.I.; Makievski, A.V.; Nikolenko, M.V.; Miller, R. Dilational Viscoelasticity of Proteins Solutions in Dynamic Conditions. Langmuir 2018, 34, 6678–6686. [Google Scholar] [CrossRef]
- Ulaganathan, V.; Retzlaff, I.; Won, J.Y.; Gochev, G.; Gunes, D.Z.; Gehin-Delval, C.; Leser, M.; Noskov, B.A.; Miller, R. β-Lactoglobulin adsorption layers at the water/air surface: 2. Dilational rheology: Effect of pH and ionic strength. Colloids Surf. A Physicochem. Eng. Asp. 2017, 521, 167–176. [Google Scholar] [CrossRef]
- Cao, C.; Zhou, Z.-L.; Zheng, L.; Huang, Q.-L.; Du, F.-P. Dilational rheology of different globular protein with imidazolium-based ionic liquid surfactant adsorption layer at the decane/water interface. J. Mol. Liq. 2017, 233, 344–351. [Google Scholar] [CrossRef]
- Akbari, S.; Mahmood, S.M.; Tan, I.M.; Ghaedi, H.; Ling, O.L. Assessment of Polyacrylamide Based Co-Polymers Enhanced by Functional Group Modifications with Regards to Salinity and Hardness. Polymers 2017, 9, 647–663. [Google Scholar] [CrossRef]
- Yi, F.; Huang, B.; Wang, C.; Tang, X.; Wang, X.; Liu, Q.; Su, Y.; Chen, S.; Wu, X.; Chen, B.; et al. Hydrophobically Associating Polymers Dissolved in Seawater for Enhanced Oil Recovery of Bohai Offshore Oilfields. Molecules 2022, 27, 2744–2759. [Google Scholar] [CrossRef]
- Jouenne, S. Polymer flooding in high temperature, high salinity conditions: Selection of polymer type and polymer chemistry, thermal stability. J. Pet. Sci. Eng. 2020, 195, 107545–107560. [Google Scholar] [CrossRef]
- Noskov, B.A.; Akentiev, A.V.; Bilibin, A.Y.; Zorin, I.M.; Miller, R. Dilational surface viscoelasticity of polymer solutions. Adv. Colloid Interface Sci. 2003, 104, 245–271. [Google Scholar] [CrossRef] [PubMed]
- Noskov, B.A.; Alexandrov, D.A.; Loglio, G.; Miller, R. Characterisation of adsorbed polymer film structure by dynamic surface tension and dilational elasticity. Colloids Surf. A Physicochem. Eng. Asp. 1999, 156, 307–313. [Google Scholar] [CrossRef]
- Noskov, B.A.; Akentiev, A.V.; Loglio, G.; Miller, R. Dynamic Surface Properties of Solutions of Poly(ethylene oxide) and Polyethylene Glycols. J. Phys. Chem. B 2000, 104, 7923–7931. [Google Scholar] [CrossRef]
- Noskov, B.A.; Akentiev, A.V.; Miller, R. Dynamic Surface Properties of Poly(vinylpyrrolidone) Solutions. J. Colloid Interface Sci 2002, 255, 417–424. [Google Scholar] [CrossRef]
- Wang, D.X.; Luo, L.; Zhang, L.; Zhao, S.; Wang, L.; Gong, Q.T.; Liao, L.; Chu, Y.p.; Yu, J.Y. The Fast Relaxation Process between Hydrophobically Modified Associating Polyacrylamide and Different Surfactants at the Water-Octane Interface. J. Dispers. Sci. Technol. 2007, 28, 725–736. [Google Scholar] [CrossRef]
- Amani, P.; Miller, R.; Ata, S.; Hurter, S.; Rudolph, V.; Firouzi, M. Dynamics of interfacial layers for sodium dodecylbenzene sulfonate solutions at different salinities. J. Ind. Eng. Chem. 2020, 92, 174–183. [Google Scholar] [CrossRef]
- Aksenenko, E.V.; Kovalchuk, V.I.; Fainerman, V.B.; Miller, R. Surface Dilational Rheology of Mixed Surfactants Layers at Liquid Interfaces. J. Phys. Chem. C 2007, 111, 14713–14719. [Google Scholar] [CrossRef]
- Feng, J.; Liu, X.P.; Zhang, L.; Zhao, S.; Yu, J.Y. Dilational properties of anionic gemini surfactants with polyoxyethylene spacers at water-air and water-decane interfaces. Langmuir 2010, 26, 11907–11914. [Google Scholar] [CrossRef]
- Zhu, Y.-W.; Zhang, L.; Song, X.-W.; Luo, L.; Zhang, L.; Zhao, S.; Yu, J.-Y. Effect of electrolyte on interfacial dilational properties of chemical flooding systems by relaxation measurements. Fuel 2011, 90, 3172–3178. [Google Scholar] [CrossRef]

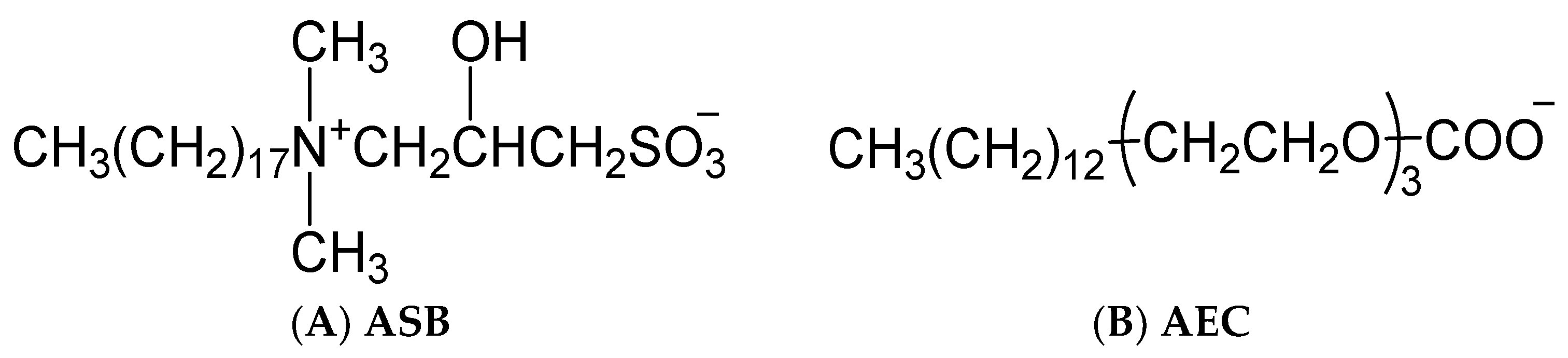
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Cui, C.; Cao, X.; Yuan, F.; Xu, Z.; Zhang, L.; Zhang, L. The Interfacial Dilational Rheology Properties of Betaine Solutions: Effect of Anionic Surfactant and Polymer. Molecules 2023, 28, 5436. https://doi.org/10.3390/molecules28145436
Li H, Cui C, Cao X, Yuan F, Xu Z, Zhang L, Zhang L. The Interfacial Dilational Rheology Properties of Betaine Solutions: Effect of Anionic Surfactant and Polymer. Molecules. 2023; 28(14):5436. https://doi.org/10.3390/molecules28145436
Chicago/Turabian StyleLi, Haitao, Chuanzhi Cui, Xulong Cao, Fuqing Yuan, Zhicheng Xu, Lei Zhang, and Lu Zhang. 2023. "The Interfacial Dilational Rheology Properties of Betaine Solutions: Effect of Anionic Surfactant and Polymer" Molecules 28, no. 14: 5436. https://doi.org/10.3390/molecules28145436
APA StyleLi, H., Cui, C., Cao, X., Yuan, F., Xu, Z., Zhang, L., & Zhang, L. (2023). The Interfacial Dilational Rheology Properties of Betaine Solutions: Effect of Anionic Surfactant and Polymer. Molecules, 28(14), 5436. https://doi.org/10.3390/molecules28145436





