Optimization of Neferine Purification Based on Response Surface Methodology and Its Anti-Metastasis Mechanism on HepG2 Cells
Abstract
1. Introduction
2. Results
2.1. The Standard Curve of Neferine
2.2. Model Establishment and Significance Analysis
2.3. Interaction Effect of Each of the Two Parameters
2.4. Preliminary Analysis of Neferine
2.5. Neferine Inhibits the Proliferation of HepG2 Cells
2.6. Neferine Inhibits the Migration of HepG2 Cells
2.7. Changes of Related Protein Expression Induced by Different Dosage of Neferine
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation Process of Neferine
4.2.1. Preparation Process of Fatty Soluble Alkaloids from Plumula nelumbinis
4.2.2. Isolation and Purification of Neferine
4.3. Single-Factor Experiments
RSM Modeling and Experimental Design
4.4. Cell Culture
4.5. CCK-8 Assay
4.6. Wound Healing Assay
4.7. Transwell Assay
4.8. Western Blot Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, P.; Henshaw, C.; Youlden, D.R.; Clark, P.J.; Aitken, J.F.; Baade, P.D. Global trends in incidence rates of primary adult liver cancers: A systematic review and meta-Analysis. Front. Oncol. 2020, 10, 171. [Google Scholar] [CrossRef] [PubMed]
- Massarweh, N.N.; El-Serag, H.B. Epidemiology of hepatocellular carcinoma and intrahepatic cholangiocarcinoma. Cancer Control 2017, 24, 1073274817729245. [Google Scholar] [CrossRef]
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Ward, E.M.; Johnson, C.J.; Cronin, K.A.; Ma, J.; Ryerson, B.; Mariotto, A.; Lake, A.J.; Wilson, R.; Sherman, R.L.; et al. Annual report to the nation on the status of cancer, 1975–2014, featuring survival. J. Natl. Cancer Inst. 2017, 109, djx030. [Google Scholar] [CrossRef]
- Abdel-Rahman, O.; Lamarca, A. Development of sorafenib-related side effects in patients diagnosed with advanced hepatocellular carcinoma treated with sorafenib: A systematic-review and meta-analysis of the impact on survival. Expert Rev. Gastroenterol. Hepatol. 2017, 11, 75–83. [Google Scholar] [CrossRef]
- Nishimura, K.; Horii, S.; Tanahashi, T.; Sugimoto, Y.; Yamada, J. Synthesis and Pharmacological Activity of Alkaloids from Embryo of Lotus, Nelumbo nucifera. Chem. Pharm. Bull. 2013, 61, 59–68. [Google Scholar] [CrossRef]
- Wicha, P.; Onsa-Ard, A.; Chaichompoo, W.; Suksamrarn, A.; Tocharus, C. Vasorelaxant and antihypertensive effects of neferine in rats: An in vitro and in vivo study. Planta Med. 2020, 86, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.P.; Zhou, Y.J.; Song, W.; Yin, Z.; He, A.D.; Ming, Z.Y. Pharmacological actions of neferine in the modulation of human platelet function. Eur. J. Pharmacol. 2019, 862, 172626. [Google Scholar] [CrossRef]
- Xu, T.; Singh, D.; Liu, J.; Li, H.; Peng, S.; Rizzolo, L.J.; Wang, S.B. Neferine, is not inducer but blocker for macroautophagic flux targeting on lysosome malfunction. Biochem. Biophys. Res. Commun. 2018, 495, 1516–1521. [Google Scholar] [CrossRef]
- Wan, J.; Zhao, L.; Xu, C.; Zhang, S.; Zhang, Z.; Zeng, C.; Chang, M.; Xiao, J.; Wang, J. Effects of neferine on the pharmacokinetics of amiodarone in rats. Biomed. Chromatogr. 2011, 25, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Erdogan, S.; Turkekul, K. Neferine inhibits proliferation and migration of human prostate cancer stem cells through p38 MAPK/JNK activation. J. Food Biochem. 2020, 44, e13253. [Google Scholar] [CrossRef] [PubMed]
- Poornima, P.; Quency, R.S.; Padma, V.V. Neferine induces reactive oxygen species mediated intrinsic pathway of apoptosis in HepG2 cells. Food Chem. 2013, 136, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dong, Y.; Li, Q. Neferine induces mitochondrial dysfunction to exert anti-proliferative and anti-invasive activities on retinoblastoma. Exp. Biol. Med. 2020, 245, 1385–1394. [Google Scholar] [CrossRef]
- Xiong, R.; Guo, L.; Luo, J.; Niu, J.; Li, J. Optimization of extraction technology for lotus plumuleby multiple–Index Comprehensive Detection. Chin. J. Vet. Drug 2017, 51, 44–48. [Google Scholar]
- Zhong, J.J.; Li, W.L.; Fu, Y.; Dai, H.L. Study on Extraction of alkaloids from lotus plumule. Technol. Dev. Chem. Ind. 2013, 42, 11–13. [Google Scholar]
- Li, Z.; Li, F.; Qiao, Z. New development of the methods of extracting and separating for alkaloids. Chem. Ind. Eng. Prog. 2010, 29, 293–299. [Google Scholar]
- Tao, R.; Pan, Y.; Jiang, Y.P.; Liu, L.J. Study on non-phenolic alkaloids and non-alkaloid components of nelumbo nucifera Gaertn. J. Nanjing TCM Univ. 2008, 24, 173–175. [Google Scholar]
- Sui, Y.; Wang, S.C. Quantitative Assay and optimization of ultrasonic-assisted extraction of three main alkaloids from Plumula nelumbinis. Chin. Pharm. J. 2018, 24, 1901–1907. [Google Scholar]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef]
- Yang, J.; Zhou, K. NMR spectroscopic analysis of neferine and isoliensinine. Magn. Reson. Chem. 2004, 42, 994–997. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Sun, C.; Cao, X.; Zhou, H.; Hong, Z.; Pan, Y. Preparative counter-current chromatography isolation of liensinine and its analogues from embryo of the seed of Nelumbo nucifera GAERTN. using upright coil planet centrifuge with four multilayer coils connected in series. J. Chromatogr. A 2004, 1041, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Fan, G.; Wu, H.; Wu, Y.; Mitchell, A. Separation, identification and rapid determination of liensine, isoliensinine and neferine from embryo of the seed of Nelumbo nucifera Gaertn. by liquid chromatography coupled to diode array detector and tandem mass spectrometry. J. Pharm. Biomed. Anal. 2007, 43, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, B.; Li, X.-Z.; Qi, L.-F.; Liang, Y.-Z. Preparative separation and purification of liensinine, isoliensinine and neferine from seed embryo of Nelumbo nucifera Gaertn using high-speed counter-current chromatography. J. Sep. Sci. 2009, 32, 2476–2481. [Google Scholar] [CrossRef]
- Duanmu, Q.; Li, A.; Sun, A.; Liu, R.; Li, X. Semi-preparative high-speed counter-current chromatography separation of alkaloids from embryo of the seed of Nelumbo nucifera Gaertn by pH-gradient elution. J. Sep. Sci. 2010, 33, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, L.; Zhou, H.; Guo, X.; Wu, S. K-targeted strategy for isolation of phenolic alkaloids of Nelumbo nucifera Gaertn by counter-current chromatography using lysine as a pH regulator. J. Chromatogr. A 2017, 1490, 115–125. [Google Scholar] [CrossRef]
- Leitão, G.G.; Leal, C.M.; Mendonça, S.C.; Pereda-Miranda, R. Purification of Alkaloids by Countercurrent Chromatography. Rev. Bras. Farmacogn. 2021, 31, 625–647. [Google Scholar] [CrossRef]
- Wang, X.; Liu, J.; Geng, Y.; Wang, D.; Dong, H.; Zhang, T. Preparative separation of alkaloids from Nelumbo nucifera Gaertn by pH-zone-refining counter-current chromatography. J. Sep. Sci. 2010, 33, 539–544. [Google Scholar] [CrossRef]
- Ryu, G.; Weon, J.B.; Yang, W.S.; Ma, C.J. Simultaneous Determination of Four Compounds in a Nelumbo nucifera Seed Embryo by HPLC-DAD. J. Spectrosc. 2017, 2017, 6426394. [Google Scholar] [CrossRef]
- Russell, J.O.; Monga, S.P. Wnt/beta-Catenin signaling in liver development, homeostasis, and pathobiology. Annu. Rev. Pathol. 2018, 13, 351–378. [Google Scholar] [CrossRef]
- Lepère, J.F.; Collet, L.; Henry, S.; Rabarison, P. Breaking news: Interruption de la transmission indigène du paludisme à Mayotte. Méd. Mal. Infect. 2020, 50, S145–S146. [Google Scholar] [CrossRef]
- Yao, Y.; Luo, Z.P.; Li, H.W.; Wang, S.X.; Wu, Y.C.; Hu, Y.; Hu, S.; Yang, C.C.; Yang, J.F.; Wang, J.P.; et al. P38gamma modulates the lipid metabolism in non-alcoholic fatty liver disease by regulating the JAK-STAT signaling pathway. FASEB J. 2023, 37, e22716. [Google Scholar] [CrossRef]
- Liu, J.; Wang, F.; Luo, F. The role of JAK/STAT pathway in fibrotic diseases: Molecular and cellular mechanisms. Biomolecules 2023, 13, 119. [Google Scholar] [CrossRef]
- Apisarnthanarax, S.; Barry, A.; Cao, M.; Czito, B.; DeMatteo, R.; Drinane, M.; Hallemeier, C.L.; Koay, E.J.; Lasley, F.; Meyer, J.; et al. External beam radiation therapy for primary liver cancers: An ASTRO clinical practice guideline. Pract. Radiat. Oncol. 2022, 12, 28–51. [Google Scholar] [CrossRef] [PubMed]
- Thorvardur, R.; Halfdanarson, M.L.K.; Grothey, A. The role of chemotherapy in managing patients with resectable liver metastases. Cancer J. 2010, 16, 125–131. [Google Scholar]
- Greten, T.F.; Sangro, B. Targets for immunotherapy of liver cancer. J. Hepatol. 2017, 68, 157–166. [Google Scholar] [CrossRef]
- Krstic, J.; Reinisch, I.; Schindlmaier, K.; Galhuber, M.; Riahi, Z.; Berger, N.; Kupper, N.; Moyschewitz, E.; Auer, M.; Michenthaler, H.; et al. Fasting improves therapeutic response in hepatocellular carcinoma through p53-dependent metabolic synergism. Sci. Adv. 2022, 8, eabh2635. [Google Scholar] [CrossRef]
- Su, G.L.; Altayar, O.; O’Shea, R.; Shah, R.; Estfan, B.; Wenzell, C.; Sultan, S.; Falck-Ytter, Y. AGA clinical practice guideline on systemic therapy for hepatocellular carcinoma. Gastroenterology 2022, 162, 920–934. [Google Scholar] [CrossRef]
- Niu, Y.; Liu, F.; Wang, X.; Chang, Y.; Song, Y.; Chu, H.; Bao, S.; Chen, C. miR-183-5p promotes HCC migration/invasion via increasing aerobic glycolysis. OncoTargets Ther. 2021, 14, 3649–3658. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, X.; Feng, L.; Liu, G.; Yao, L.; Wang, Z.; Liu, S.; Xiao, Y.; Zhang, G. Androgen receptor (AR) decreases HCC cells migration and invasion via miR-325/ACP5 signaling. J. Cancer 2021, 12, 1915–1925. [Google Scholar] [CrossRef]
- Matthews, H.K.; Bertoli, C.; de Bruin, R.A.M. Cell cycle control in cancer. Nat. Rev. Mol. Cell Biol. 2022, 23, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Gutierrez, D.; Bauer, M.A.; Zimmermann, A.; Aguilera, A.; Austriaco, N.; Ayscough, K.; Balzan, R.; Bar-Nun, S.; Barrientos, A.; Belenky, P.; et al. Guidelines and recommendations on yeast cell death nomenclature. Microb. Cell 2018, 5, 4–31. [Google Scholar] [CrossRef]
- Ganesh, K.; Massague, J. Targeting metastatic cancer. Nat. Med. 2021, 27, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Hellum, B.H.; Liang, A.; Nilsen, O.G. The in vitro inhibition of human CYP1A2, CYP2D6 and CYP3A4 by tetrahydropalmatine, neferine and berberine. Phytother. Res. 2012, 26, 277–283. [Google Scholar] [CrossRef]
- Tian, Y.; Qian, S.; Jiang, Y.; Shen, Q.; Zheng, J.; Zhou, H.; Zeng, S. The interaction between human breast cancer resistance protein (BCRP) and five bisbenzylisoquinoline alkaloids. Int. J. Pharm. 2013, 453, 371–379. [Google Scholar] [CrossRef]
- Yoon, J.S.; Kim, H.M.; Yadunandam, A.K.; Kim, N.H.; Jung, H.A.; Choi, J.S.; Kim, C.Y.; Kim, G.D. Neferine isolated from Nelumbo nucifera enhances anti-cancer activities in Hep3B cells: Molecular mechanisms of cell cycle arrest, ER stress induced apoptosis and anti-angiogenic response. Phytomedicine 2013, 20, 1013–1022. [Google Scholar] [CrossRef]
- Esposito, M.; Ganesan, S.; Kang, Y. Emerging strategies for treating metastasis. Nat. Cancer 2021, 2, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Espigares, J.; Nieto, D.; Moroni, L.; Jimenez, G.; Marchal, J.A. Evolution of metastasis study models toward metastasis-on-a-chip: The ultimate model? Small 2021, 17, e2006009. [Google Scholar] [CrossRef]
- Deng, G.; Zeng, S.; Ma, J.; Zhang, Y.; Qu, Y.; Han, Y.; Yin, L.; Cai, C.; Guo, C.; Shen, H. The anti-tumor activities of Neferine on cell invasion and oxaliplatin sensitivity regulated by EMT via Snail signaling in hepatocellular carcinoma. Sci. Rep 2017, 7, 41616. [Google Scholar] [CrossRef]
- Wang, T.; Rao, D.; Yu, C.; Sheng, J.; Luo, Y.; Xia, L.; Huang, W. RHO GTPase family in hepatocellular carcinoma. Exp. Hematol. Oncol. 2022, 11, 91–102. [Google Scholar] [CrossRef]
- Hu, P.; Wan, P.; Xu, A.; Yan, B.; Liu, C.; Xu, Q.; Wei, Z.; Xu, J.; Liu, S.; Yang, G.; et al. Neferine, a novel ROCK1-targeting inhibitor, blocks EMT process and induces apoptosis in non-small cell lung cancer. J. Cancer Res. Clin. Oncol. 2023, 149, 553–566. [Google Scholar] [CrossRef]
- Nam, S.; Lee, Y.; Kim, J.H. RhoA protein expression correlates with clinical features in gastric cancer: A systematic review and meta-analysis. BMC Cancer 2022, 22, 798. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Sun, Y. Overexpression of myosin phosphatase target subunit 1 (MYPT1) inhibits tumor progression and metastasis of gastric cancer. Med. Sci. Monit. 2018, 24, 2508–2517. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Lai, Y.; Zhang, H.; Ren, K.; Liu, W.; An, Y.; Yao, J.; Fan, H. Hesperetin inhibits TGF-beta1-Induced migration and invasion of triple negative breast cancer MDA-MB-231 cells via suppressing Fyn/Paxillin/RhoA pathway. Integr. Cancer Ther. 2022, 21, 15347354221086900. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhu, X.; Xu, C.; Min, F.; Yu, G.; Chen, C. Ulinastatin ameliorates the malignant progression of prostate cancer cells by blocking the RhoA/ROCK/NLRP3 pathway. Drug Dev. Res. 2023, 84, 36–44. [Google Scholar] [CrossRef]
- Xia, N.; Yang, N.; Shan, Q.; Wang, Z.; Liu, X.; Chen, Y.; Lu, J.; Huang, W.; Wang, Z. HNRNPC regulates RhoA to induce DNA damage repair and cancer-associated fibroblast activation causing radiation resistance in pancreatic cancer. J. Cell. Mol. Med. 2022, 26, 2322–2336. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, D.; Peng, Z.; Wang, R.; Li, K.; Ren, H.; Sun, X.; Du, N.; Tang, S.C. LINC00891 regulated by miR-128-3p/GATA2 axis impedes lung cancer cell proliferation, invasion and EMT by inhibiting RhoA pathway. Acta Biochim. Biophys. Sin. 2022, 54, 378–387. [Google Scholar] [CrossRef]
- Shin, Y.; Jung, W.; Kim, M.Y.; Shin, D.; Kim, G.H.; Kim, C.H.; Park, S.H.; Cho, E.H.; Choi, D.W.; Han, C.J.; et al. NPFFR2 contributes to the malignancy of hepatocellular carcinoma development by activating RhoA/YAP signaling. Cancers 2022, 14, 5850. [Google Scholar] [CrossRef]
- Yadav, S.; Kashaninejad, N.; Nguyen, N.T. RhoA and Rac1 in liver cancer cells: Induction of overexpression using mechanical stimulation. Micromachines 2020, 11, 729. [Google Scholar] [CrossRef]
- Lee, E.; Stafford, W.F., 3rd. Interaction of myosin phosphatase target subunit (MYPT1) with myosin phosphatase-RhoA Interacting protein (MRIP): A role of glutamic acids in the interaction. PLoS ONE 2015, 10, e0139875. [Google Scholar]
- Cho, H.S.; Suzuki, T.; Dohmae, N.; Hayami, S.; Unoki, M.; Yoshimatsu, M.; Toyokawa, G.; Takawa, M.; Chen, T.; Kurash, J.K.; et al. Demethylation of RB regulator MYPT1 by histone demethylase LSD1 promotes cell cycle progression in cancer cells. Cancer Res. 2011, 71, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, L.; Guo, J.; Zuo, S.; Wang, Z.; Hua, S. MYPT1, regulated by miR-19b-3p inhibits the progression of non-small cell lung cancer via inhibiting the activation of wnt/beta-catenin signaling. Life Sci. 2021, 278, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Torres, A.; Banuelos-Villegas, E.G.; Martinez-Acuna, N.; Sulpice, E.; Gidrol, X.; Alvarez-Salas, L.M. MYPT1 is targeted by miR-145 inhibiting viability, migration and invasion in 2D and 3D HeLa cultures. Biochem. Biophys. Res. Commun. 2018, 507, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Feng, Y.; Lau, E.P.; Tsang, C.; Ching, Y.; Man, K.; Tong, Y.; Nagamatsu, T.; Su, W.; Tsao, S. F-actin reorganization and inactivation of rho signaling pathway involved in the inhibitory effect of Coptidis Rhizoma on hepatoma cell migration. Integr. Cancer Ther. 2010, 9, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, Q.; Tang, Q.; Hu, C.; Huang, J.; Liu, Y.; Lu, Y.; Wang, Q.; Li, G.; Zhang, R. Lycorine inhibits cell proliferation and migration by inhibiting ROCK1/cofilin-induced actin dynamics in HepG2 hepatoblastoma cells. Oncol. Rep. 2018, 40, 2298–2306. [Google Scholar] [CrossRef] [PubMed]

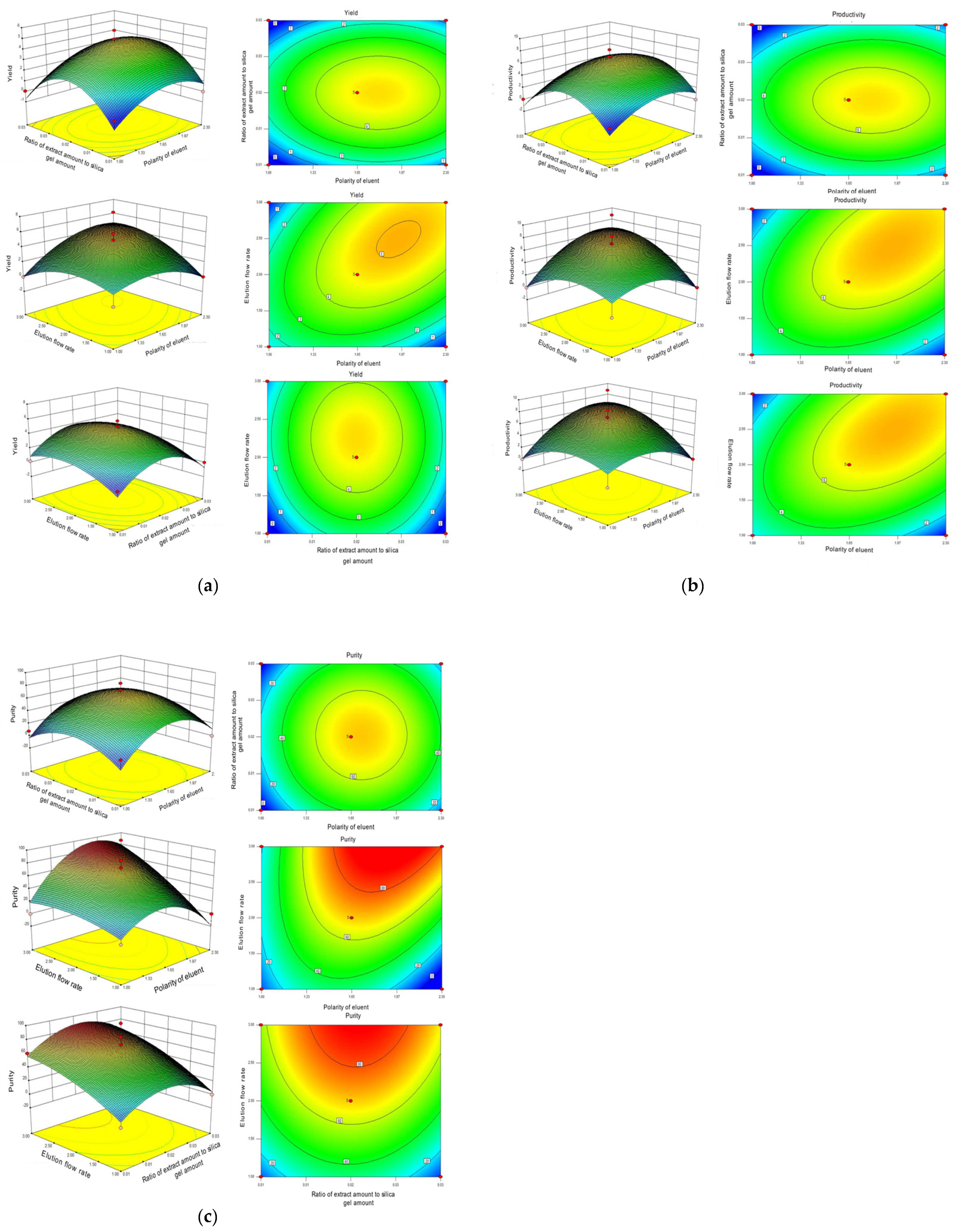
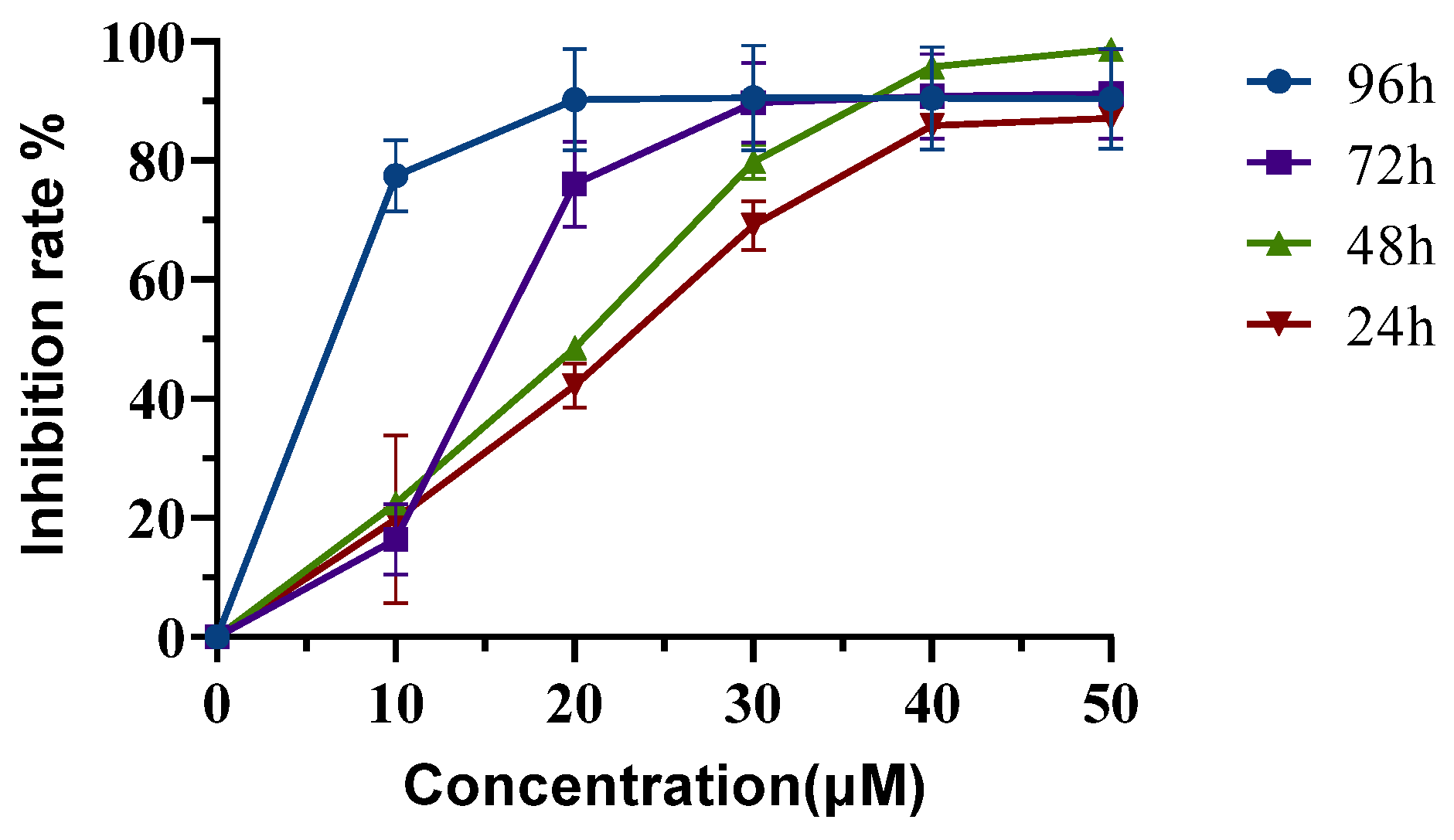

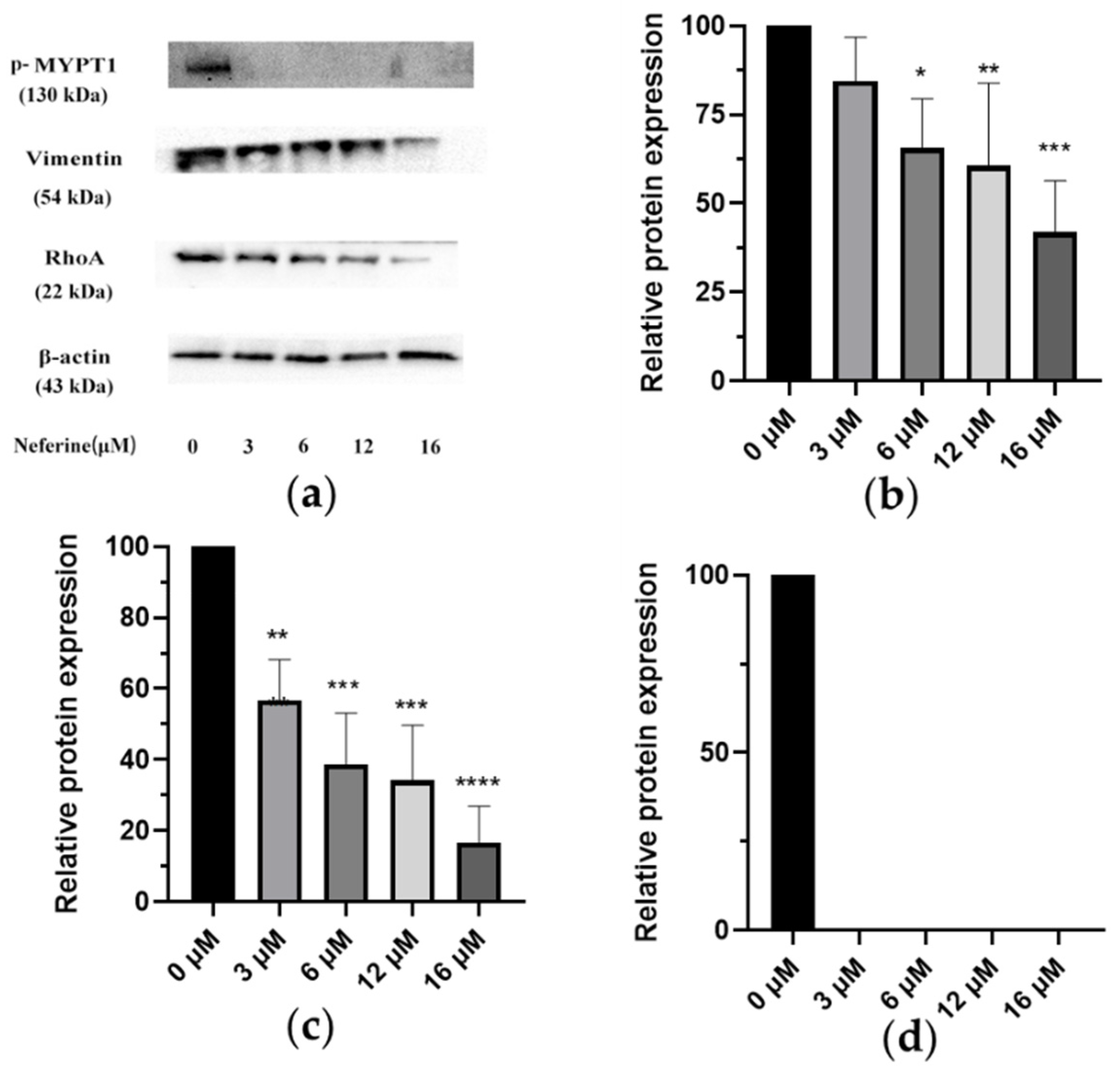
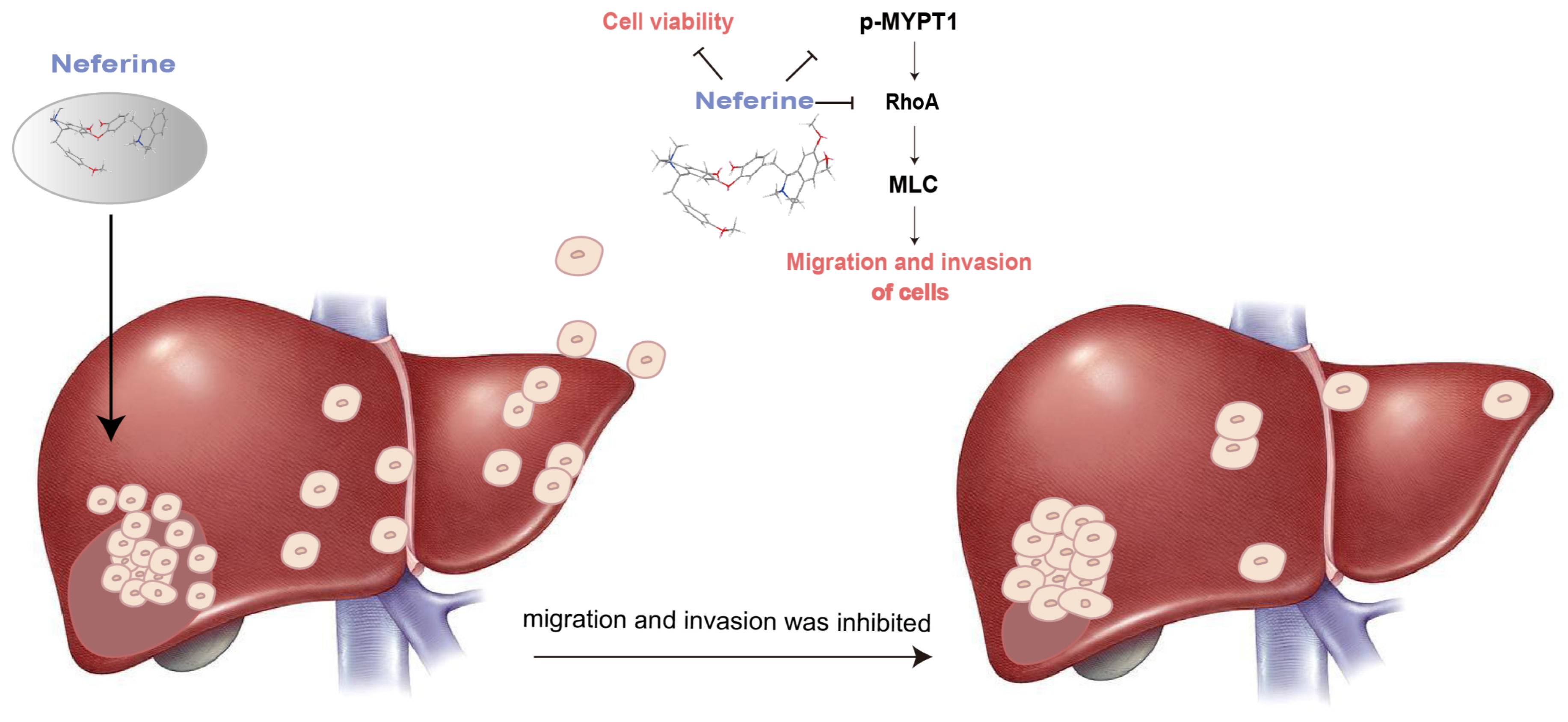
| No. | Flow Matching Ratio (mL/mL) | Extract Amount: Silica Gel Amount (g: g) | Elution Flow Rate (mL/min) | Yield of Neferine (mg) | Productivity of Neferine (%) | Purity of Neferine (%) |
|---|---|---|---|---|---|---|
| 1 | 7:3 | 1:40 | 2 | 0 | 0 | 0 |
| 2 | 6:4 | 1:60 | 2 | 5.76 | 8.23 | 58.32 |
| 3 | 7:3 | 1:60 | 1 | 0 | 0 | 0 |
| 4 | 7:3 | 1:80 | 2 | 0 | 0 | 0 |
| 5 | 5:5 | 1:60 | 3 | 0 | 0 | 0 |
| 6 | 6:4 | 1:40 | 3 | 0.19 | 0.19 | 73.68 |
| 7 | 6:4 | 1:80 | 3 | 0.17 | 0.34 | 60.49 |
| 8 | 5:5 | 1:40 | 2 | 0.05 | 0.05 | 7.79 |
| 9 | 6:4 | 1:60 | 2 | 4.49 | 6.42 | 72.95 |
| 10 | 6:4 | 1:40 | 1 | 0 | 0 | 0 |
| 11 | 7:3 | 1:60 | 3 | 6.13 | 8.76 | 87.04 |
| 12 | 5:5 | 1:60 | 1 | 0 | 0 | 0 |
| 13 | 6:4 | 1:80 | 1 | 0 | 0 | 0 |
| 14 | 5:5 | 1:80 | 2 | 0.05 | 0.10 | 9.22 |
| 15 | 6:4 | 1:60 | 2 | 4.96 | 7.09 | 64.14 |
| 16 | 6:4 | 1:60 | 2 | 4.51 | 6.44 | 67.87 |
| 17 | 6:4 | 1:60 | 2 | 3.78 | 5.41 | 83.65 |
| Sum of | Mean | F | p-Value | |||
|---|---|---|---|---|---|---|
| Source | Squares | df | Square | Value | Prob > F | |
| Model | 19,082.55 | 9 | 2120.28 | 7.325 | 0.008 | significant |
| A | 612.87 | 1 | 612.87 | 2.117 | 0.189 | |
| B | 17.27 | 1 | 17.26 | 0.060 | 0.814 | |
| C | 6116.56 | 1 | 6116.56 | 21.131 | 0.003 | significant |
| AB | 0.51 | 1 | 0.51 | 0.002 | 0.968 | |
| AC | 1893.96 | 1 | 1893.92 | 6.543 | 0.038 | significant |
| BC | 43.47 | 1 | 43.47 | 0.150 | 0.710 | |
| A2 | 6227.51 | 1 | 6227.51 | 21.514 | 0.002 | significant |
| B2 | 2996.16 | 1 | 2996.16 | 10.351 | 0.015 | significant |
| C2 | 354.09 | 1 | 354.09 | 1.223 | 0.305 | |
| Residual | 2026.22 | 7 | 289.46 | |||
| Lack of Fit | 1657.83 | 3 | 552.61 | 6.000 | 0.058 | not significant |
| Pure Error | 368.40 | 4 | 92.10 | |||
| Cor Total | 21,108.77 | 16 |
| Level | A (mL/mL) | B (g:g) | C (mL/min) |
|---|---|---|---|
| −1 | 5:5 | 1:80 | 1 |
| 0 | 6:4 | 1:60 | 2 |
| 1 | 7:3 | 1:40 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Wei, Z.; Hu, P.; Xia, W.; Liao, Z.; Assani, I.; Yang, G.; Pan, Y. Optimization of Neferine Purification Based on Response Surface Methodology and Its Anti-Metastasis Mechanism on HepG2 Cells. Molecules 2023, 28, 5086. https://doi.org/10.3390/molecules28135086
Wang X, Wei Z, Hu P, Xia W, Liao Z, Assani I, Yang G, Pan Y. Optimization of Neferine Purification Based on Response Surface Methodology and Its Anti-Metastasis Mechanism on HepG2 Cells. Molecules. 2023; 28(13):5086. https://doi.org/10.3390/molecules28135086
Chicago/Turabian StyleWang, Xinzhu, Zhenhuan Wei, Po Hu, Weibo Xia, Zhixin Liao, Israa Assani, Guangming Yang, and Yang Pan. 2023. "Optimization of Neferine Purification Based on Response Surface Methodology and Its Anti-Metastasis Mechanism on HepG2 Cells" Molecules 28, no. 13: 5086. https://doi.org/10.3390/molecules28135086
APA StyleWang, X., Wei, Z., Hu, P., Xia, W., Liao, Z., Assani, I., Yang, G., & Pan, Y. (2023). Optimization of Neferine Purification Based on Response Surface Methodology and Its Anti-Metastasis Mechanism on HepG2 Cells. Molecules, 28(13), 5086. https://doi.org/10.3390/molecules28135086







