A Superior Two-Dimensional Phosphorus Flame Retardant: Few-Layer Black Phosphorus
Abstract
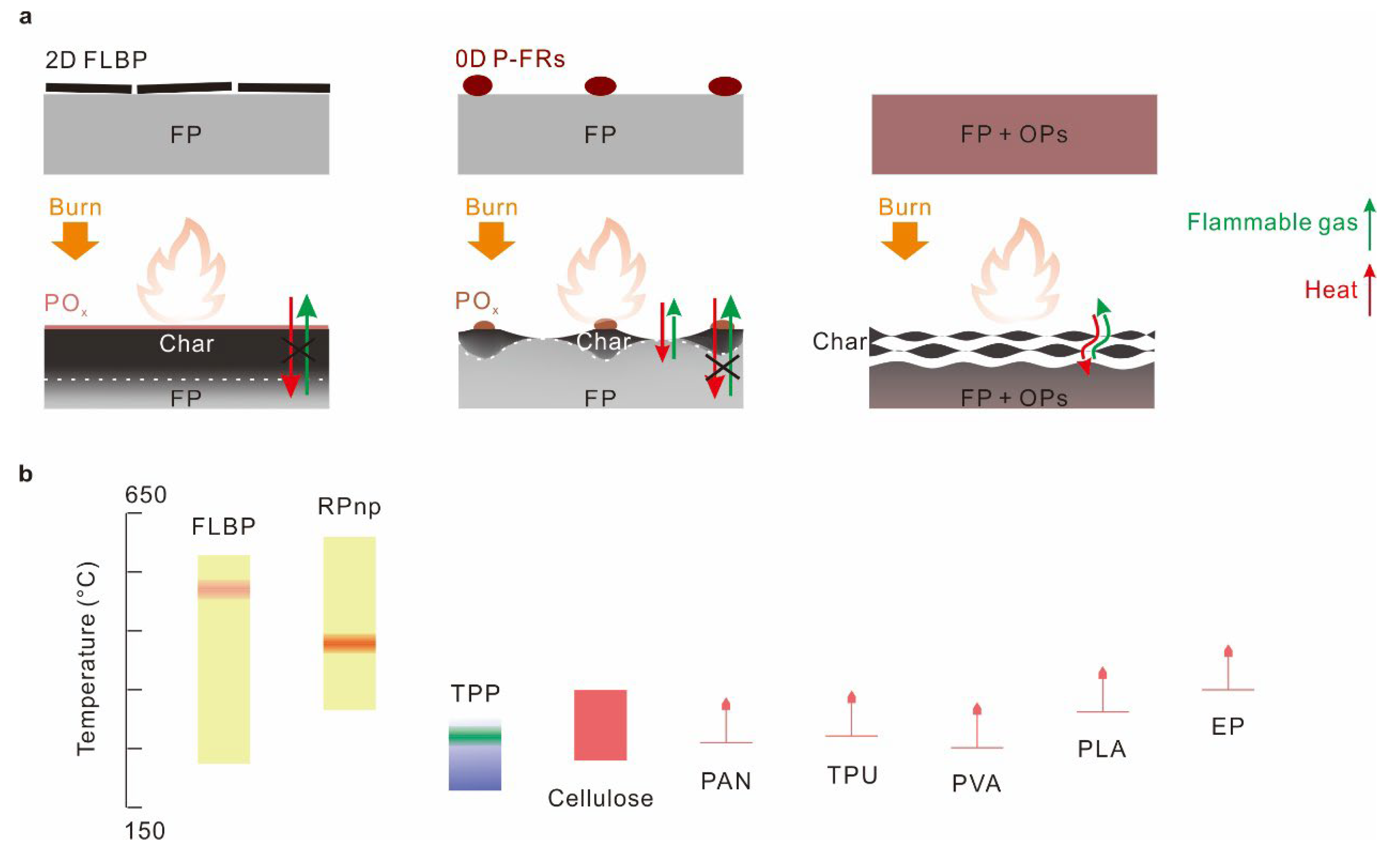
1. Introduction
2. Result and Discussion
3. Materials and Methods
3.1. Preparation of the Bulk Black Phosphorus
3.2. Preparation of the Few-Layer Black Phosphorus and Red Phosphorus Nanoparticles
3.3. Preparations of the Papers Coated with Different P-FRs
3.4. Preparations of the PAN Fiber Films with Different P-FRs
3.5. Characterizations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Liu, B.-W.; Zhao, H.-B.; Wang, Y.-Z. Advanced Flame-Retardant Methods for Polymeric Materials. Adv. Mater. 2022, 34, 2107905. [Google Scholar] [CrossRef] [PubMed]
- Velencoso, M.M.; Battig, A.; Markwart, J.C.; Schartel, B.; Wurm, F.R. Molecular Firefighting-How Modern Phosphorus Chemistry Can Help Solve the Challenge of Flame Retardancy. Angew. Chem. Int. Ed. 2018, 57, 10450–10467. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Liu, W.; Qiu, Y.; Kong, B.; Sun, Y.; Chen, Z.; Zhuo, D.; Lin, D.; Cui, Y. Electrospun core-shell microfiber separator with thermal-triggered flame-retardant properties for lithium-ion batteries. Sci. Adv. 2017, 3, e1601978. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Xue, J.; Dong, L.; Jiang, Y.; Wang, X.; Qu, L.; Dai, L. Scalable Preparation of Multifunctional Fire-Retardant Ultralight Graphene Foams. ACS Nano 2016, 10, 1325–1332. [Google Scholar] [CrossRef]
- Xia, D.; Shen, Z.; Huang, G.; Wang, W.; Yu, J.C.; Wong, P.K. Red Phosphorus: An Earth-Abundant Elemental Photocatalyst for “Green” Bacterial Inactivation under Visible Light. Environ. Sci. Technol. 2015, 49, 6264–6273. [Google Scholar] [CrossRef]
- Some, S.; Shackery, I.; Kim, S.J.; Jun, S.C. Phosphorus-Doped Graphene Oxide Layer as a Highly Efficient Flame Retardant. Chem. A Eur. J. 2015, 21, 15480–15485. [Google Scholar] [CrossRef]
- Savas, L.A.; Deniz, T.K.; Tayfun, U.; Dogan, M. Effect of microcapsulated red phosphorus on flame retardant, thermal and mechanical properties of thermoplastic polyurethane composites filled with huntite&hydromagnesite mineral. Polym. Degrad. Stab. 2017, 135, 121–129. [Google Scholar]
- Kim, M.-J.; Jeon, I.-Y.; Seo, J.-M.; Dai, L.; Baek, J.-B. Graphene phosphonic acid as an efficient flame retardant. ACS Nano 2014, 8, 2820–2825. [Google Scholar] [CrossRef]
- Schartel, B.; Kunze, R.; Neubert, D. Red phosphorus–controlled decomposition for fire retardant PA 66. J. Appl. Polym. Sci. 2002, 83, 2060–2071. [Google Scholar] [CrossRef]
- Zhang, W.; Barrio, J.; Gervais, C.; Kocjan, A.; Yu, A.; Wang, X.; Shalom, M. Synthesis of Carbon-Nitrogen-Phosphorous Materials with an Unprecedented High Amount of Phosphorous toward an Efficient Fire-Retardant Material. Angew. Chem. Int. Ed. 2018, 57, 9764–9769. [Google Scholar] [CrossRef]
- Nguyen, T.-M.D.; Chang, S.; Condon, B.; Uchimiya, M.; Graves, E.; Smith, J.; Easson, M.; Wakelyn, P. Synthesis and characterization of a novel phosphorus–nitrogen-containing flame retardant and its application for textile. Polym. Adv. Technol. 2012, 23, 1036–1044. [Google Scholar] [CrossRef]
- Wang, J.; Ma, C.; Wang, P.; Qiu, S.; Cai, W.; Hu, Y. Ultra-low phosphorus loading to achieve the superior flame retardancy of epoxy resin. Polym. Degrad. Stab. 2018, 149, 119–128. [Google Scholar] [CrossRef]
- Ji, W.; Yao, Y.; Guo, J.; Fei, B.; Gu, X.; Li, H.; Sun, J.; Zhang, S. Toward an understanding of how red phosphorus and expandable graphite enhance the fire resistance of expandable polystyrene foams. J. Appl. Polym. Sci. 2020, 137, 49045. [Google Scholar] [CrossRef]
- Dogan, M.; Murat Unlu, S. Flame retardant effect of boron compounds on red phosphorus containing epoxy resins. Polym. Degrad. Stab. 2014, 99, 12–17. [Google Scholar] [CrossRef]
- Pecht, M.; Deng, Y. Electronic device encapsulation using red phosphorus flame retardants. Microelectron. Reliab. 2006, 46, 53–62. [Google Scholar] [CrossRef]
- Bridgman, P.W. Two New Modifications of Phosphorus. J. Am. Chem. Soc. 1914, 36, 1344–1363. [Google Scholar] [CrossRef]
- Li, L.; Yu, Y.; Ye, G.J.; Ge, Q.; Ou, X.; Wu, H.; Feng, D.; Chen, X.H.; Zhang, Y. Black phosphorus field-effect transistors. Nat. Nanotechnol. 2014, 9, 372–377. [Google Scholar] [CrossRef]
- Zeng, L.; Zhang, X.; Liu, Y.; Yang, X.; Wang, J.; Liu, Q.; Luo, Q.; Jing, C.; Yu, X.-F.; Qu, G.; et al. Surface and interface control of black phosphorus. Chem 2022, 8, 632–662. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, C.; Xie, J.; Ågren, H.; Zhang, H. Black Phosphorus/Polymers: Status and Challenges. Adv. Mater. 2021, 33, 2100113. [Google Scholar] [CrossRef]
- Zhang, X.; Donskyi, I.; Tang, W.; Deng, S.; Liu, D.; Zhang, S.; Zhao, Q.; Xing, B. Biological Effects of Black Phosphorus Nanomaterials on Mammalian Cells and Animals. Angew. Chem. Int. Ed. 2023, 62, e202213336. [Google Scholar] [CrossRef]
- Jin, H.; Xin, S.; Chuang, C.; Li, W.; Wang, H.; Zhu, J.; Xie, H.; Zhang, T.; Wan, Y.; Qi, Z.; et al. Black phosphorus composites with engineered interfaces for high-rate high-capacity lithium storage. Science 2020, 370, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Viscardi, L.; Intonti, K.; Kumar, A.; Faella, E.; Pelella, A.; Giubileo, F.; Sleziona, S.; Kharsah, O.; Schleberger, M.; Di Bartolomeo, A. Black Phosphorus Nanosheets in Field Effect Transistors with Ni and NiCr Contacts. Phys. Status Solidi B 2023, 2200537. [Google Scholar] [CrossRef]
- Telesio, F.; le Gal, G.; Serrano-Ruiz, M.; Prescimone, F.; Toffanin, S.; Peruzzini, M.; Heun, S. Ohmic contact engineering in few–layer black phosphorus: Approaching the quantum limit. Nanotechnology 2020, 31, 334002. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Faella, E.; Durante, O.; Giubileo, F.; Pelella, A.; Viscardi, L.; Intonti, K.; Sleziona, S.; Schleberger, M.; Di Bartolomeo, A. Optoelectronic memory in 2D MoS2 field effect transistor. J. Phys. Chem. Solids 2023, 179, 111406. [Google Scholar] [CrossRef]
- Kumar, A.; Viscardi, L.; Faella, E.; Giubileo, F.; Intonti, K.; Pelella, A.; Sleziona, S.; Kharsah, O.; Schleberger, M.; Di Bartolomeo, A. Black phosphorus unipolar transistor, memory, and photodetector. J. Mater. Sci. 2023, 58, 2689–2699. [Google Scholar] [CrossRef]
- Ling, X.; Wang, H.; Huang, S.; Xia, F.; Dresselhaus, M.S. The renaissance of black phosphorus. Proc. Natl. Acad. Sci. USA 2015, 112, 4523–4530. [Google Scholar] [CrossRef]
- Dinh, K.N.; Zhang, Y.; Sun, W. The synthesis of black phosphorus: From zero- to three-dimensional nanostructures. J. Phys. Energy 2021, 3, 032007. [Google Scholar] [CrossRef]
- Chaudhary, V.; Neugebauer, P.; Mounkachi, O.; Lahbabi, S.; El Fatimy, A. Phosphorene—An emerging two-dimensional material: Recent advances in synthesis, functionalization, and applications. 2d Mater. 2022, 9, 032001. [Google Scholar] [CrossRef]
- Ren, X.; Mei, Y.; Lian, P.; Xie, D.; Yang, Y.; Wang, Y.; Wang, Z. A Novel Application of Phosphorene as a Flame Retardant. Polymers 2018, 10, 227. [Google Scholar] [CrossRef]
- Qiu, S.; Zhou, Y.; Zhou, X.; Zhang, T.; Wang, C.; Yuen, R.K.K.; Hu, W.; Hu, Y. Air-Stable Polyphosphazene-Functionalized Few-Layer Black Phosphorene for Flame Retardancy of Epoxy Resins. Small 2019, 15, 1805175. [Google Scholar] [CrossRef]
- Qu, Z.; Wu, K.; Jiao, E.; Chen, W.; Hu, Z.; Xu, C.; Shi, J.; Wang, S.; Tan, Z. Surface functionalization of few-layer black phosphorene and its flame retardancy in epoxy resin. Chem. Eng. J. 2020, 382, 122991. [Google Scholar] [CrossRef]
- Zou, B.; Qiu, S.; Ren, X.; Zhou, Y.; Zhou, F.; Xu, Z.; Zhao, Z.; Song, L.; Hu, Y.; Gong, X. Combination of black phosphorus nanosheets and MCNTs via phosphoruscarbon bonds for reducing the flammability of air stable epoxy resin nanocomposites. J. Hazard. Mater. 2020, 383, 121069. [Google Scholar] [CrossRef] [PubMed]
- Duan, Z.; Wang, Y.; Bian, S.; Liu, D.; Zhang, Y.; Zhang, X.; He, R.; Wang, J.; Qu, G.; Chu, P.K.; et al. Size-dependent flame retardancy of black phosphorus nanosheets. Nanoscale 2022, 14, 2599–2604. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Zhou, Y.; Ren, X.; Zou, B.; Guo, W.; Song, L.; Hu, Y. Construction of hierarchical functionalized black phosphorus with polydopamine: A novel strategy for enhancing flame retardancy and mechanical properties of polyvinyl alcohol. Chem. Eng. J. 2020, 402, 126212. [Google Scholar] [CrossRef]
- Xiao, Y.; Li, J.; Wu, J.; Song, L.; Han, Y.; Wang, Z.; Yu, Q. Glass fiber reinforced PET modified by few-layer black phosphorus. Polym. Adv. Technol. 2021, 32, 3515–3522. [Google Scholar] [CrossRef]
- Qiu, S.; Zou, B.; Sheng, H.; Guo, W.; Wang, J.; Zhao, Y.; Wang, W.; Yuen, R.K.K.; Kan, Y.; Hu, Y. Electrochemically Exfoliated Functionalized Black Phosphorene and Its Polyurethane Acrylate Nanocomposites: Synthesis and Applications. ACS Appl. Mater. Interfaces 2019, 11, 13652–13664. [Google Scholar] [CrossRef]
- Ren, X.; Mei, Y.; Lian, P.; Xie, D.; Deng, W.; Wen, Y.; Luo, Y. Fabrication and Application of Black Phosphorene/Graphene Composite Material as a Flame Retardant. Polymers 2019, 11, 193. [Google Scholar] [CrossRef]
- Cai, W.; Cai, T.; He, L.; Chu, F.; Mu, X.; Han, L.; Hu, Y.; Wang, B.; Hu, W. Natural antioxidant functionalization for fabricating ambient-stable black phosphorus nanosheets toward enhancing flame retardancy and toxic gases suppression of polyurethane. J. Hazard. Mater. 2020, 387, 121971. [Google Scholar] [CrossRef]
- Yin, S.; Ren, X.; Lian, P.; Zhu, Y.; Mei, Y. Synergistic Effects of Black Phosphorus/Boron Nitride Nanosheets on Enhancing the Flame-Retardant Properties of Waterborne Polyurethane and Its Flame-Retardant Mechanism. Polymers 2020, 12, 1487. [Google Scholar] [CrossRef]
- Zhou, Y.; Huang, J.; Wang, J.; Chu, F.; Xu, Z.; Hu, W.; Hu, Y. Rationally designed functionalized black phosphorus nanosheets as new fire hazard suppression material for polylactic acid. Polym. Degrad. Stab. 2020, 178, 109194. [Google Scholar] [CrossRef]
- Cai, W.; Hu, Y.; Pan, Y.; Zhou, X.; Chu, F.; Han, L.; Mu, X.; Zhuang, Z.; Wang, X.; Xing, W. Self-assembly followed by radical polymerization of ionic liquid for interfacial engineering of black phosphorus nanosheets: Enhancing flame retardancy, toxic gas suppression and mechanical performance of polyurethane. J. Colloid Interface Sci. 2020, 561, 32–45. [Google Scholar] [CrossRef]
- Du, X.; Qiu, J.; Deng, S.; Du, Z.; Cheng, X.; Wang, H. Flame-retardant and form-stable phase change composites based on black phosphorus nanosheets/cellulose nanofiber aerogels with extremely high energy storage density and superior solar-thermal conversion efficiency. J. Mater. Chem. A 2020, 8, 14126–14134. [Google Scholar] [CrossRef]
- Qu, Z.; Wu, K.; Xu, C.-a.; Li, Y.; Jiao, E.; Chen, B.; Meng, H.; Cui, X.; Wang, K.; Shi, J. Facile Construction of a Flexible Film with Ultrahigh Thermal Conductivity and Excellent Flame Retardancy for a Smart Fire Alarm. Chem. Mater. 2021, 33, 3228–3240. [Google Scholar] [CrossRef]
- Zhang, T.; Wan, Y.; Xie, H.; Mu, Y.; Du, P.; Wang, D.; Wu, X.; Ji, H.; Wan, L. Degradation Chemistry and Stabilization of Exfoliated Few-Layer Black Phosphorus in Water. J. Am. Chem. Soc. 2018, 140, 7561–7567. [Google Scholar] [CrossRef]
- Zhou, Q.; Chen, Q.; Tong, Y.; Wang, J. Light-Induced Ambient Degradation of Few-Layer Black Phosphorus: Mechanism and Protection. Angew. Chem. Int. Ed. 2016, 55, 11437–11441. [Google Scholar] [CrossRef] [PubMed]
- Favron, A.; Gaufrès, E.; Fossard, F.; Phaneuf-L’Heureux, A.-L.; Tang, N.Y.W.; Lévesque, P.L.; Loiseau, A.; Leonelli, R.; Francoeur, S.; Martel, R. Photooxidation and quantum confinement effects in exfoliated black phosphorus. Nat. Mater. 2015, 14, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, K.; Li, X.; Xu, Q.; Weng, J.; Xu, J. Electron Matters: Recent Advances in Passivation and Applications of Black Phosphorus. Adv. Mater. 2021, 33, 2005924. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, T.; Jiang, D.; Duan, H.; Sun, Z.; Zhang, M.; Jin, H.; Guan, R.; Liu, Y.; Chen, M.; et al. Stabilizing black phosphorus nanosheets via edge-selective bonding of sacrificial C60 molecules. Nat. Commun. 2018, 9, 4177. [Google Scholar] [CrossRef]
- Okram, G.S.; Singh, J.; Kaurav, N.; Lalla, N.P. Trioctylphosphine as self-assembly inducer. Faraday Discuss. 2015, 181, 211–223. [Google Scholar] [CrossRef]
- Wang, J.; Yamada, Y.; Sodeyama, K.; Watanabe, E.; Takada, K.; Tateyama, Y.; Yamada, A. Fire-extinguishing organic electrolytes for safe batterie. Nat. Energy 2018, 3, 22–29. [Google Scholar] [CrossRef]
- Aizenshtein, E.M. Global Output of Chemical Fibres in 2014. Fibre Chem. 2016, 48, 90–93. [Google Scholar] [CrossRef]
- Smith, M.W.; Dallmeyer, I.; Johnson, T.J.; Brauer, C.S.; McEwen, J.-S.; Espinal, J.F.; Garcia-Perez, M. Structural analysis of char by Raman spectroscopy: Improving band assignments through computational calculations from first principles. Carbon 2016, 100, 678–692. [Google Scholar] [CrossRef]
- Cançado, L.G.; Takai, K.; Enoki, T.; Endo, M.; Kim, Y.A.; Mizusaki, H.; Jorio, A.; Coelho, L.N.; Magalhães-Paniago, R.; Pimenta, M.A. General equation for the determination of the crystallite size La of nanographite by Raman spectroscopy. Appl. Phys. Lett. 2006, 88, 163106. [Google Scholar] [CrossRef]
- Bernard, S.; Beyssac, O.; Benzerara, K.; Findling, N.; Tzvetkov, G.; Brown, G.E. XANES, Raman and XRD study of anthracene-based cokes and saccharose-based chars submitted to high-temperature pyrolysis. Carbon 2010, 48, 2506–2516. [Google Scholar] [CrossRef]
- Gómez-Pérez, J.; Barna, B.; Tóth, I.Y.; Kónya, Z.; Kukovecz, Á. Quantitative Tracking of the Oxidation of Black Phosphorus in the Few-Layer Regime. ACS Omega 2018, 3, 12482–12488. [Google Scholar] [CrossRef]
- Gibertini, E.; Carosio, F.; Aykanat, K.; Accogli, A.; Panzeri, G.; Magagnin, L. Silica-encapsulated red phosphorus for flame retardant treatment on textile. Surf. Interfaces 2021, 25, 101252. [Google Scholar] [CrossRef]
- Arseneau, D.F. Competitive Reactions in the Thermal Decomposition of Cellulose. Can. J. Chem. 1971, 49, 632–638. [Google Scholar] [CrossRef]
- Soares, S.; Camino, G.; Levchik, S.V. Comparative study of the thermal decomposition of pure cellulose and pulp paper. Polym. Degrad. Stab. 1995, 49, 275–283. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, T.; Xie, H.; Xie, S.; Hu, A.; Liu, J.; Kang, J.; Hou, J.; Hao, Q.; Liu, H.; Ji, H. A Superior Two-Dimensional Phosphorus Flame Retardant: Few-Layer Black Phosphorus. Molecules 2023, 28, 5062. https://doi.org/10.3390/molecules28135062
Zhang T, Xie H, Xie S, Hu A, Liu J, Kang J, Hou J, Hao Q, Liu H, Ji H. A Superior Two-Dimensional Phosphorus Flame Retardant: Few-Layer Black Phosphorus. Molecules. 2023; 28(13):5062. https://doi.org/10.3390/molecules28135062
Chicago/Turabian StyleZhang, Taiming, Huanyu Xie, Shuai Xie, Ajuan Hu, Jie Liu, Jian Kang, Jie Hou, Qing Hao, Hong Liu, and Hengxing Ji. 2023. "A Superior Two-Dimensional Phosphorus Flame Retardant: Few-Layer Black Phosphorus" Molecules 28, no. 13: 5062. https://doi.org/10.3390/molecules28135062
APA StyleZhang, T., Xie, H., Xie, S., Hu, A., Liu, J., Kang, J., Hou, J., Hao, Q., Liu, H., & Ji, H. (2023). A Superior Two-Dimensional Phosphorus Flame Retardant: Few-Layer Black Phosphorus. Molecules, 28(13), 5062. https://doi.org/10.3390/molecules28135062





