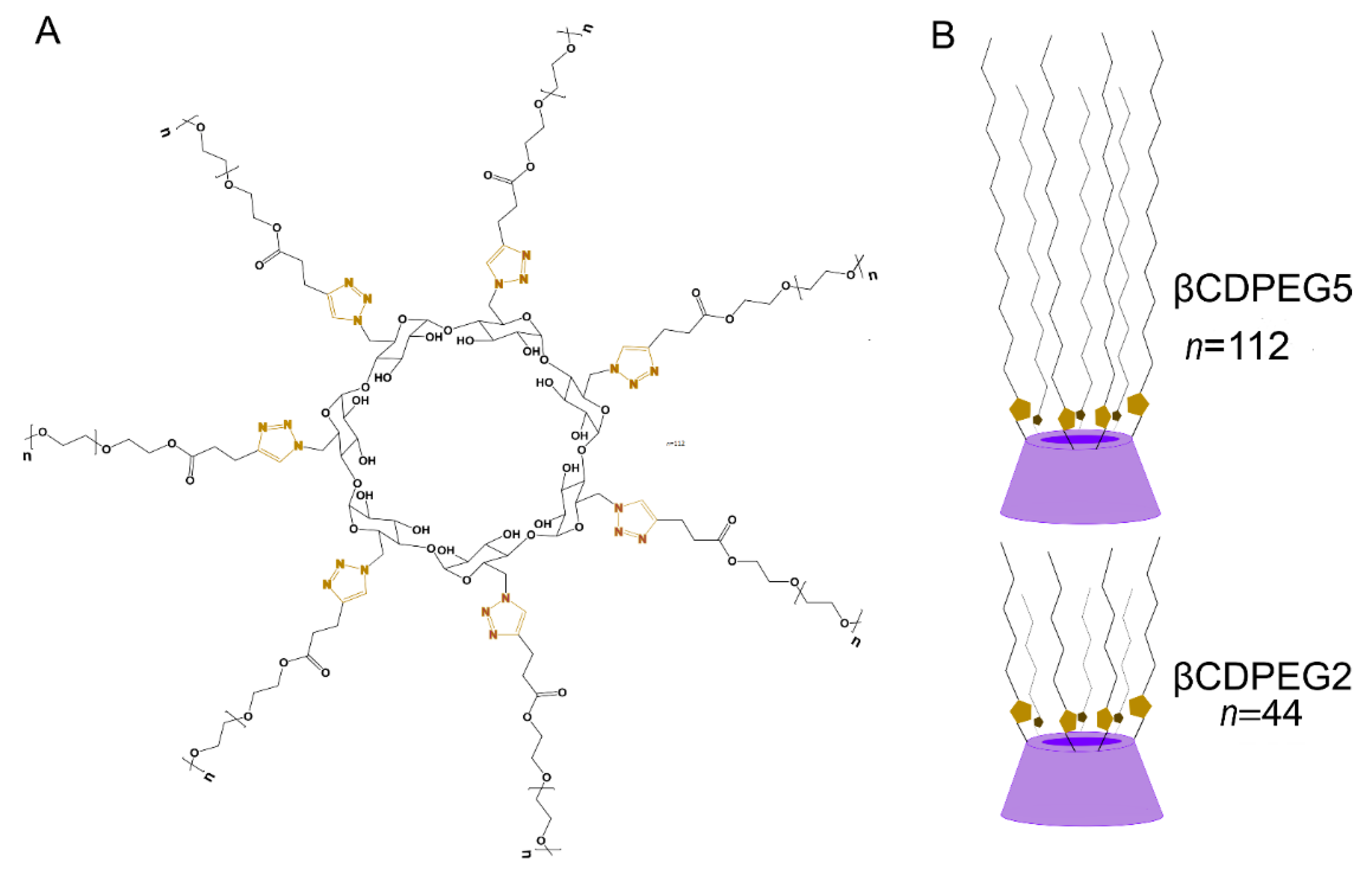
Exploring In Vitro Biological Cellular Responses of Pegylated β-Cyclodextrins
Abstract
1. Introduction
2. Results and Discussion
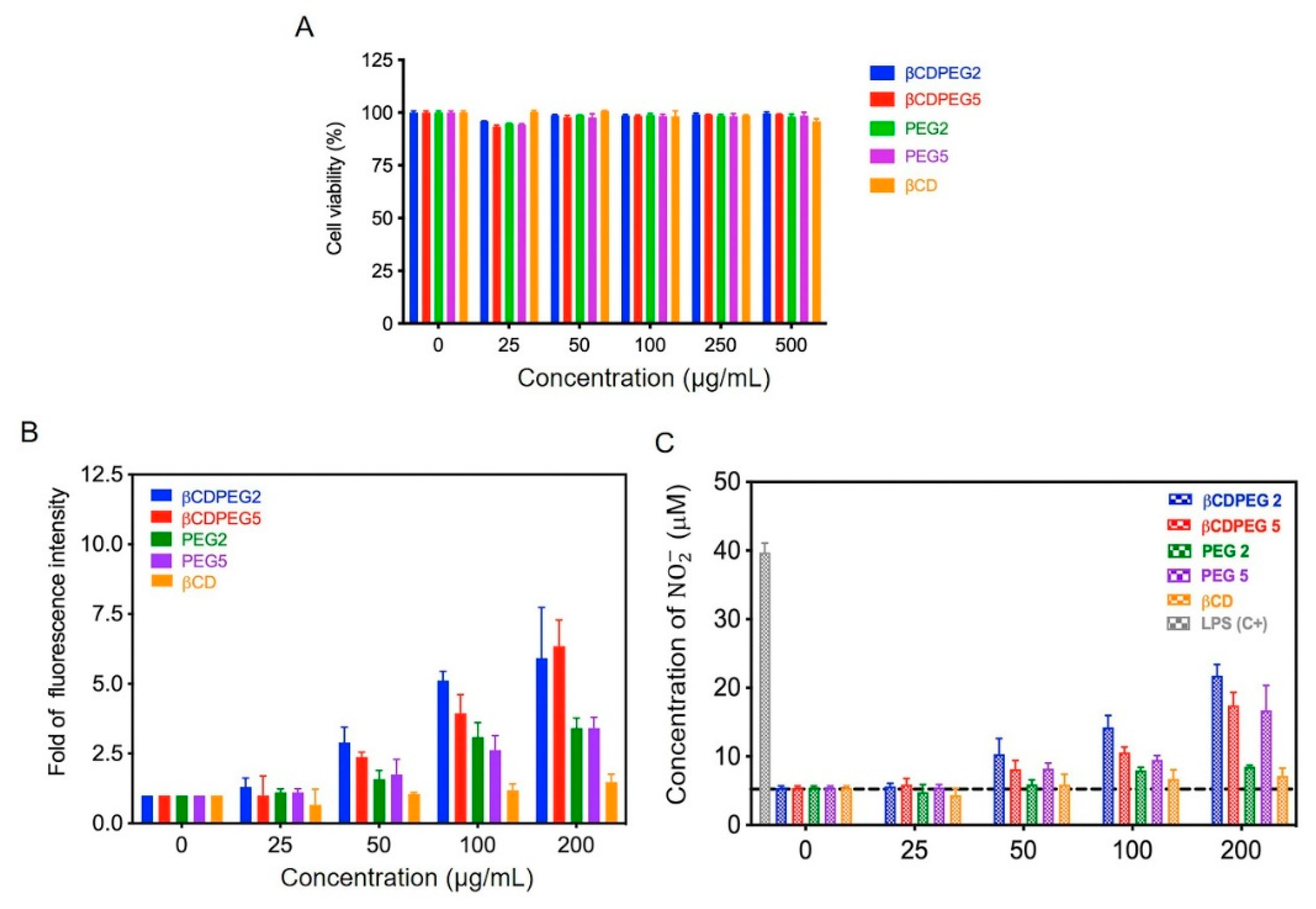
2.1. RAW 264.7 Macrophages
2.1.1. Cell Viability
2.1.2. ROS and Nitrite (NO2−) Generation
2.2. MC3T3-E1 Osteoblastic Cell Line
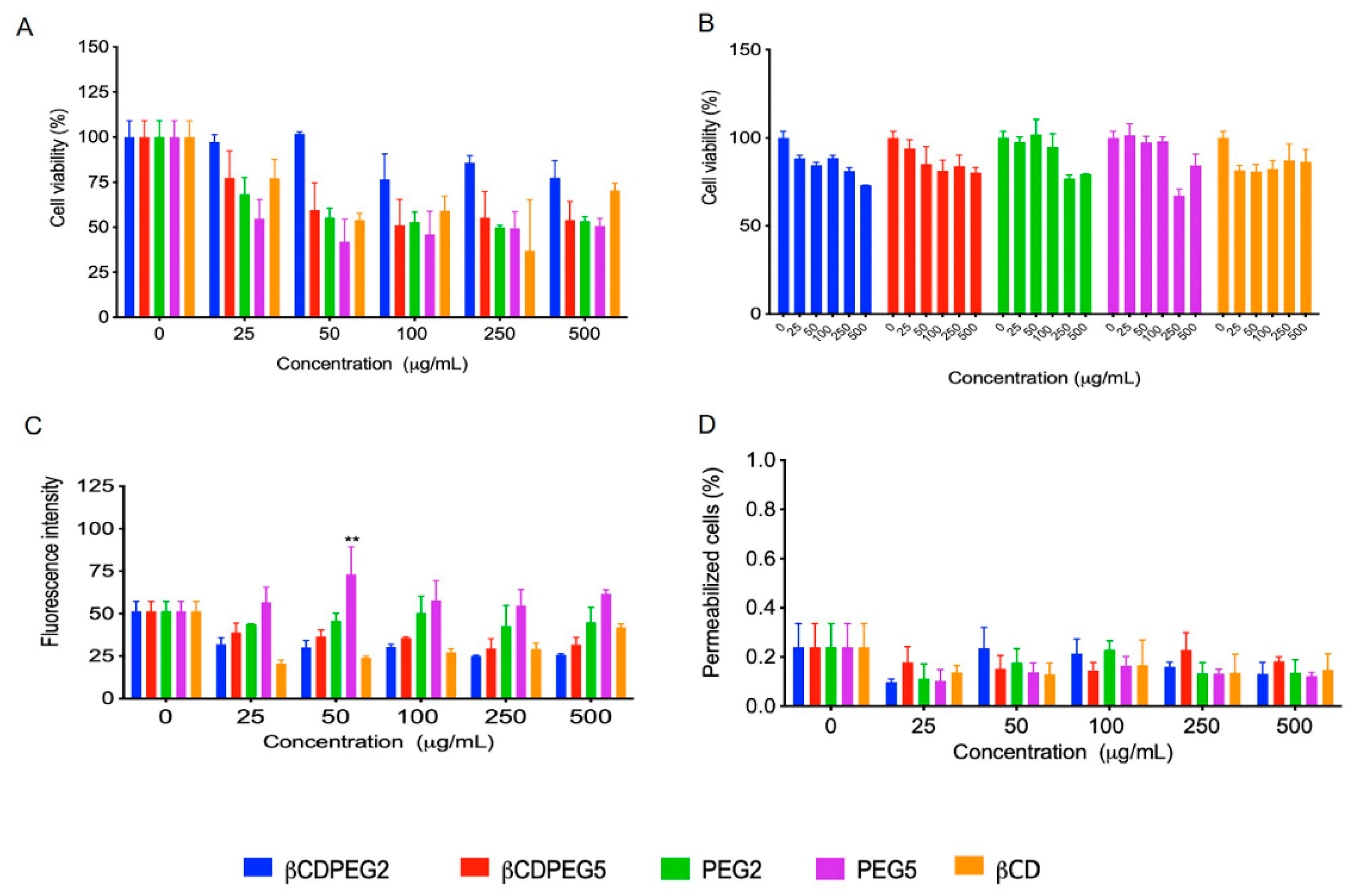
2.2.1. Cell Viability
2.2.2. Post-Treatment Recovery Assay
2.2.3. ROS Generation
2.2.4. Membrane Permeability
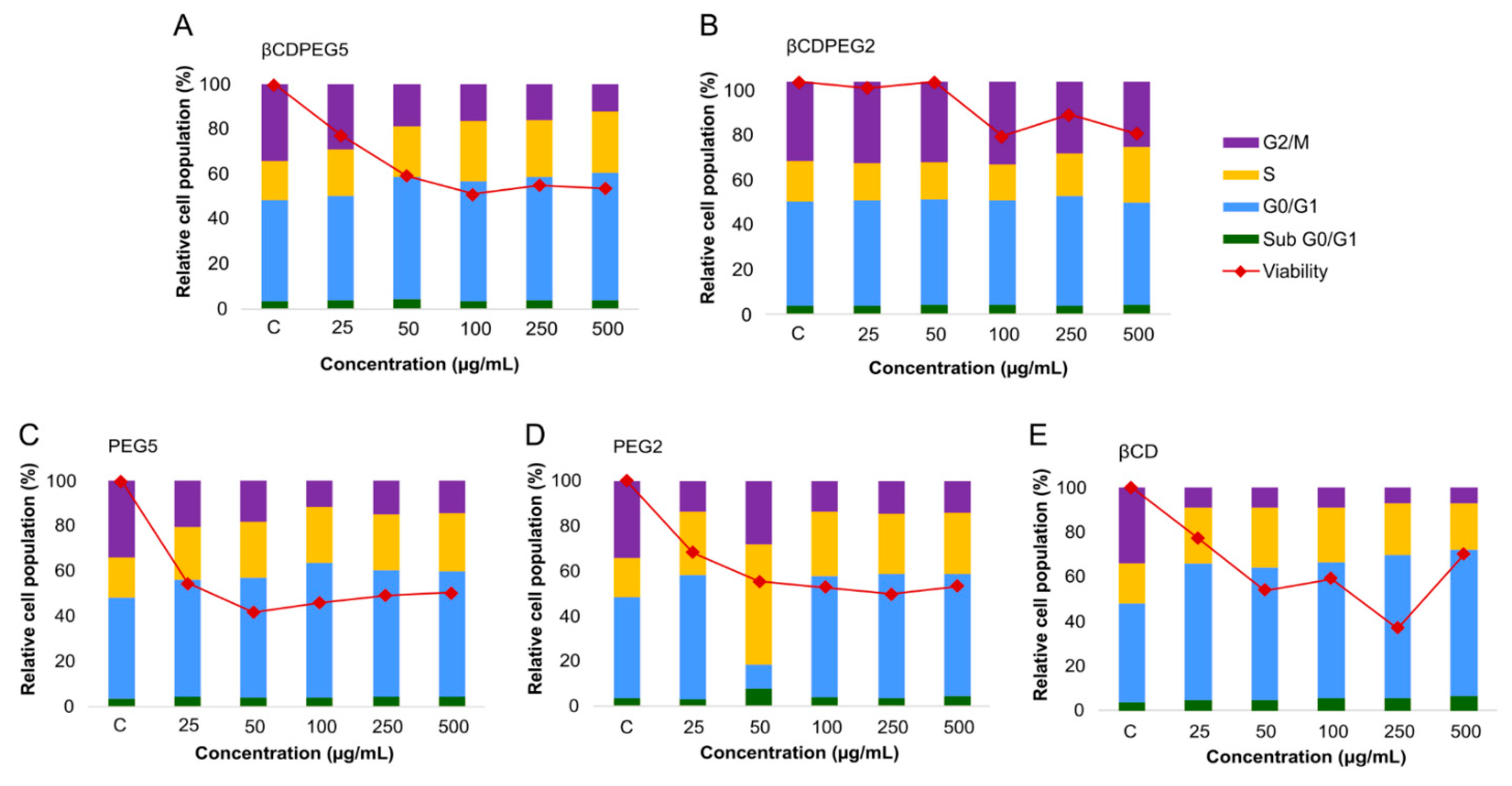
2.2.5. Cell Cycle
2.2.6. Cell Migration
2.3. MDCK Cells
2.3.1. Cell Viability
2.3.2. Post-Treatment Recovery Assay
2.3.3. ROS Generation
2.3.4. Membrane Permeability
2.3.5. Cell Cycle
2.3.6. Cell Migration
3. Methodology
3.1. Materials
3.2. Cell Culture
3.3. Instrumentation
3.4. Cell Viability Assay
3.5. Production of Nitrites by Macrophages
3.6. Reactive Oxygen Species Production
3.7. Post-Treatment Recovery Assay (Re-Cultivation Assay)
3.8. Cell Membrane Permeability Assay
3.9. Cell Cycle Progression Measurements by Flow Cytometry
3.10. Cell Migration Assay (Scratch Assay)
3.11. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| αCD | α-cyclodextrin |
| βCD | β-cyclodextrin |
| HPβCD | 2-Hydroxypropyl-β-cyclodextrin |
| IC | Inclusion complex |
| MβCD | Methyl-β-cyclodextrin |
| MW | Molecular weight |
| PEG | Polyethylene glycol |
| ROS | Reactive oxygen species |
| SBEβCD | Sulfobutylether-β-cyclodextrin |
References
- Dodziuk, H. Molecules with Holes—Cyclodextrins. In Cyclodextrins and Their Complexes; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006; pp. 1–30. ISBN 9783527608980. [Google Scholar]
- Rincón-López, J.; Almanza-Arjona, Y.C.; Riascos, A.P.; Rojas-Aguirre, Y. Technological Evolution of Cyclodextrins in the Pharmaceutical Field. J. Drug Deliv. Sci. Technol. 2020, 61, 102156. [Google Scholar] [CrossRef] [PubMed]
- Rincón-López, J.; Almanza-Arjona, Y.C.; Riascos, A.P.; Rojas-Aguirre, Y. When Cyclodextrins Met Data Science: Unveiling Their Pharmaceutical Applications through Network Science and Text-Mining. Pharmaceutics 2021, 13, 1297. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Flores, F.; Castro-Lugo, A.; Ramírez, O.T. Understanding Cellular Interactions with Nanomaterials: Towards a Rational Design of Medical Nanodevices. Nanotechnology 2020, 31, 132002. [Google Scholar] [CrossRef]
- Jesus, S.; Schmutz, M.; Som, C.; Borchard, G.; Wick, P.; Borges, O. Hazard Assessment of Polymeric Nanobiomaterials for Drug Delivery: What Can We Learn From Literature So Far. Front. Bioeng. Biotechnol. 2019, 7, 261. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Aguirre, Y.; Torres-Mena, M.A.; López-Méndez, L.J.; Alcaraz-Estrada, S.L.; Guadarrama, P.; Urucha-Ortíz, J.M. PEGylated β-Cyclodextrins: Click Synthesis and in Vitro Biological Insights. Carbohydr. Polym. 2019, 223, 115113. [Google Scholar] [CrossRef] [PubMed]
- Rincón-López, J.; Ramírez-Rodríguez, N.J.; Luviano, A.S.; Costas, M.; López-Cervantes, J.L.; García-Figueroa, A.A.; Domínguez, H.; Mendoza-Cruz, R.; Guadarrama, P.; López-Morales, S.; et al. Experimental and Theoretical Studies of Pegylated-β-Cyclodextrin: A Step Forward to Understand Its Tunable Self-Aggregation Abilities. J. Drug Deliv. Sci. Technol. 2021, 67, 102975. [Google Scholar] [CrossRef]
- Farace, C.; Sánchez-Moreno, P.; Orecchioni, M.; Manetti, R.; Sgarrella, F.; Asara, Y.; Peula-García, J.M.; Marchal, J.A.; Madeddu, R.; Delogu, L.G. Immune Cell Impact of Three Differently Coated Lipid Nanocapsules: Pluronic, Chitosan and Polyethylene Glycol. Sci. Rep. 2016, 6, 18423. [Google Scholar] [CrossRef] [PubMed]
- Kelley, W.J.; Fromen, C.A.; Lopez-Cazares, G.; Eniola-Adefeso, O. PEGylation of Model Drug Carriers Enhances Phagocytosis by Primary Human Neutrophils. Acta Biomater. 2018, 79, 283–293. [Google Scholar] [CrossRef]
- Knop, K.; Hoogenboom, R.; Fischer, D.; Schubert, U.S. Poly(Ethylene Glycol) in Drug Delivery: Pros and Cons as Well as Potential Alternatives. Angew. Chem. Int. Ed. 2010, 49, 6288–6308. [Google Scholar] [CrossRef]
- Yang, Q.; Jones, S.W.; Parker, C.L.; Zamboni, W.C.; Bear, J.E.; Lai, S.K. Evading Immune Cell Uptake and Clearance Requires PEG Grafting at Densities Substantially Exceeding the Minimum for Brush Conformation. Mol. Pharm. 2014, 11, 1250–1258. [Google Scholar] [CrossRef]
- Sanchez, L.; Yi, Y.; Yu, Y. Effect of Partial PEGylation on Particle Uptake by Macrophages. Nanoscale 2016, 9, 288–297. [Google Scholar] [CrossRef]
- Malachowski, T.; Hassel, A. Engineering Nanoparticles to Overcome Immunological Barriers for Enhanced Drug Delivery. Eng. Regen. 2020, 1, 35–50. [Google Scholar] [CrossRef]
- Chamberlain, L.M.; Godek, M.L.; Gonzalez-Juarrero, M.; Grainger, D.W. Phenotypic Non-Equivalence of Murine (Monocyte-) Macrophage Cells in Biomaterial and Inflammatory Models. J. Biomed. Mater. Res. Part A 2009, 88, 858–871. [Google Scholar] [CrossRef] [PubMed]
- Quarles, L.D.; Yohay, D.A.; Lever, L.W.; Caton, R.; Wenstrup, R.J. Distinct Proliferative and Differentiated Stages of Murine MC3T3-E1 Cells in Culture: An in Vitro Model of Osteoblast Development. J. Bone Miner. Res. 1992, 7, 683–692. [Google Scholar] [CrossRef] [PubMed]
- Terauchi, M.; Tamura, A.; Arisaka, Y.; Masuda, H.; Yoda, T.; Yui, N. Cyclodextrin-Based Supramolecular Complexes of Osteoinductive Agents for Dental Tissue Regeneration. Pharmaceutics 2021, 13, 136. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Lorenzo, C.; García-González, C.A.; Concheiro, A. Cyclodextrins as Versatile Building Blocks for Regenerative Medicine. J. Control. Release 2017, 268, 269–281. [Google Scholar] [CrossRef]
- He, B.; Jia, Z.; Du, W.; Yu, C.; Fan, Y.; Dai, W.; Yuan, L.; Zhang, H.; Wang, X.; Wang, J.; et al. The Transport Pathways of Polymer Nanoparticles in MDCK Epithelial Cells. Biomaterials 2013, 34, 4309–4326. [Google Scholar] [CrossRef] [PubMed]
- Dukes, J.D.; Whitley, P.; Chalmers, A.D. The MDCK Variety Pack: Choosing the Right Strain. BMC Mol. Cell Biol. 2011, 12, 43. [Google Scholar] [CrossRef]
- Volpe, D.A. Drug-Permeability and Transporter Assays in Caco-2 and MDCK Cell Lines. Future Med. Chem. 2011, 3, 2063–2077. [Google Scholar] [CrossRef]
- Rusznyák, Á.; Malanga, M.; Fenyvesi, É.; Szente, L.; Váradi, J.; Bácskay, I.; Vecsernyés, M.; Vasvári, G.; Haimhoffer, Á.; Fehér, P.; et al. Investigation of the Cellular Effects of Beta-Cyclodextrin Derivatives on Caco-2 Intestinal Epithelial Cells. Pharmaceutics 2021, 13, 157. [Google Scholar] [CrossRef]
- Mu, K.; Jiang, K.; Wang, Y.; Zhao, Z.; Cang, S.; Bi, K.; Li, Q.; Liu, R. The Biological Fate of Pharmaceutical Excipient β-Cyclodextrin: Pharmacokinetics, Tissue Distribution, Excretion, and Metabolism of β-Cyclodextrin in Rats. Molecules 2022, 27, 1138. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Li, Y.; Yang, L.; Wei, Y.; Wang, X.; Wang, Z.; Tao, L. Cytotoxicity Study of Polyethylene Glycol Derivatives. RSC Adv. 2017, 7, 18252–18259. [Google Scholar] [CrossRef]
- Grenier, P.; Maíra, I.; Viana, D.O.; Martins, E.; Bertrand, N. Anti-Polyethylene Glycol Antibodies Alter the Protein Corona Deposited on Nanoparticles and the Physiological Pathways Regulating Their Fate in Vivo. J. Control. Release 2018, 287, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Goda, S.; Morita, S. The Effect of Methyl-β-Cyclodextrin on the Differentiation of RAW264 Cells into Osteoclasts. Oral Sci. Int. 2008, 5, 15–23. [Google Scholar] [CrossRef][Green Version]
- He, J.; Yang, Y.; Zhou, X.; Zhang, W.; Liu, J. Shuttle/Sink Model Composed of -Cyclodextrin and Simvastatin-Loaded Discoidal Reconstituted High-Density Lipoprotein for Enhanced Cholesterol Efflux and Drug Uptake in Macrophage/Foam Cells. J. Mater. Chem. B 2020, 8, 1496–1506. [Google Scholar] [CrossRef]
- Balaraman, K.; Vieira, N.C.; Moussa, F.; Vacus, J.; Cojean, S.; Pomel, S.; Bories, C.; Figadère, B.; Kesavan, V.; Loiseau, P.M. In Vitro and in Vivo Antileishmanial Properties of a 2-n-Propylquinoline Hydroxypropyl β-Cyclodextrin Formulation and Pharmacokinetics via Intravenous Route. Biomed. Pharmacother. 2015, 76, 127–133. [Google Scholar] [CrossRef]
- Shibaguchi, K.; Tamura, A.; Terauchi, M.; Matsumura, M.; Miura, H.; Yui, N. Mannosylated Polyrotaxanes for Increasing Cellular Uptake Efficiency in Macrophages through Receptor-Mediated Endocytosis. Molecules 2019, 24, 439. [Google Scholar] [CrossRef]
- Davaatseren, M.; Jo, Y.-J.; Hong, G.-P.; Hur, H.J.; Park, S.; Choi, M.-J. Studies on the Anti-Oxidative Function of Trans-Cinnamaldehyde-Included β-Cyclodextrin Complex. Molecules 2017, 22, 1868. [Google Scholar] [CrossRef]
- Giacoppo, S.; Rajan, T.S.; Iori, R.; Rollin, P.; Bramanti, P.; Mazzon, E. The α-Cyclodextrin Complex of the Moringa Isothiocyanate Suppresses Lipopolysaccharide-Induced Inflammation in RAW 264.7 Macrophage Cells through Akt and P38 Inhibition. Inflamm. Res. 2017, 66, 487–503. [Google Scholar] [CrossRef]
- Jay Forman, H.; Torres, M. Redox Signaling in Macrophages. Mol. Asp. Med. 2001, 22, 189216. [Google Scholar] [CrossRef]
- Herb, M.; Schramm, M. Functions of Ros in Macrophages and Antimicrobial Immunity. Antioxidants 2021, 10, 313. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Chawla, A.; Pollard, J.W. Macrophage Biology in Development, Homeostasis and Disease. Nature 2013, 496, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, L.; Nie, H.; Lei, L. Dexamethasone-Loaded β-Cyclodextrin for Osteogenic Induction of Mesenchymal Stem/Progenitor Cells and Bone Regeneration. J. Biomed. Mater. Res. Part A 2021, 109, 1125–1135. [Google Scholar] [CrossRef]
- Pan, P.; Chen, X.; Metavarayuth, K.; Su, J.; Wang, Q. Self-Assembled Supramolecular Systems for Bone Engineering Applications. Curr. Opin. Colloid Interface Sci. 2018, 35, 104–111. [Google Scholar] [CrossRef]
- Du, J.; Gan, S.; Bian, Q.; Fu, D.; Wei, Y.; Wang, K.; Lin, Q.; Chen, W.; Huang, D. Preparation and Characterization of Porous Hydroxyapatite/β-Cyclodextrin-Based Polyurethane Composite Scaffolds for Bone Tissue Engineering. J. Biomater. Appl. 2018, 33, 402–409. [Google Scholar] [CrossRef]
- Palomino-Durand, C.; Lopez, M.; Cazaux, F.; Martel, B.; Blanchemain, N.; Chai, F. Influence of the Soluble-Insoluble Ratios of Cyclodextrins Polymers on the Viscoelastic Properties of Injectable Chitosan-Based Hydrogels for Biomedical Application. Polymer 2019, 11, 214. [Google Scholar] [CrossRef] [PubMed]
- Kamel, R.; El-Wakil, N.A.; Abdelkhalek, A.F.A.; Elkasabgy, N.A. Nanofibrillated Cellulose/Cyclodextrin Based 3D Scaffolds Loaded with Raloxifene Hydrochloride for Bone Regeneration. Int. J. Biol. Macromol. 2020, 156, 704–716. [Google Scholar] [CrossRef]
- Trajano, V.C.C.; Costa, K.J.R.; Lanza, C.R.M.; Sinisterra, R.D.; Cortés, M.E. Osteogenic Activity of Cyclodextrin-Encapsulated Doxycycline in a Calcium Phosphate PCL and PLGA Composite. Mater. Sci. Eng. C 2016, 64, 370–375. [Google Scholar] [CrossRef]
- Liu, X.; Wei, Y.; Xuan, C.; Liu, L.; Lai, C.; Chai, M.; Zhang, Z.; Wang, L.; Shi, X. A Biomimetic Biphasic Osteochondral Scaffold with Layer-Specific Release of Stem Cell Differentiation Inducers for the Reconstruction of Osteochondral Defects. Adv. Healthc. Mater. 2020, 9, 2000076. [Google Scholar] [CrossRef]
- Çetin Altındal, D.; Gümüşderelioğlu, M. Dual-Functional Melatonin Releasing Device Loaded with PLGA Microparticles and Cyclodextrin Inclusion Complex for Osteosarcoma Therapy. J. Drug Deliv. Sci. Technol. 2019, 52, 586–596. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, J.; Chen, J.; Zeng, Y.; Zhu, Z.; Wan, Y. Fabrication of Curcumin-Modified TiO2 Nanoarrays via Cyclodextrin Based Polymer Functional Coatings for Osteosarcoma Therapy. Adv. Healthc. Mater. 2019, 8, 1901031. [Google Scholar] [CrossRef] [PubMed]
- Federico, S.; Pitarresi, G.; Palumbo, F.S.; Fiorica, C.; Yang, F.; Giammona, G. Hyaluronan Alkyl Derivatives-Based Electrospun Membranes for Potential Guided Bone Regeneration: Fabrication, Characterization and in Vitro Osteoinductive Properties. Colloids Surf. B Biointerfaces 2021, 197, 111438. [Google Scholar] [CrossRef] [PubMed]
- Schoonraad, S.A.; Trombold, M.L.; Bryant, S.J. The Effects of Stably Tethered BMP-2 on MC3T3-E1 Preosteoblasts Encapsulated in a PEG Hydrogel. Biomacromolecules 2021, 22, 1065–1079. [Google Scholar] [CrossRef] [PubMed]
- Scaffaro, R.; Lopresti, F.; Maio, A.; Botta, L.; Rigogliuso, S.; Ghersi, G. Electrospun PCL/GO-g-PEG Structures: Processing-Morphology-Properties Relationships. Compos. Part A Appl. Sci. Manuf. 2017, 92, 97–107. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, P.; Mao, H.; Zhang, Y.; Zheng, L.; Yu, P.; Guo, Z.; Li, L.; Jiang, Q. PEGylated Gold Nanoparticles Promote Osteogenic Differentiation in In Vitro and In Vivo Systems. Mater. Des. 2021, 197, 109231. [Google Scholar] [CrossRef]
- Fatokun, A.A.; Stone, T.W.; Smith, R.A. Responses of Differentiated MC3T3-E1 Osteoblast-like Cells to Reactive Oxygen Species. Eur. J. Pharmacol. 2008, 587, 35–41. [Google Scholar] [CrossRef]
- Hammoud, Z.; Khreich, N.; Auezova, L.; Fourmentin, S.; Elaissari, A.; Greige-Gerges, H. Cyclodextrin-Membrane Interaction in Drug Delivery and Membrane Structure Maintenance. Int. J. Pharm. 2019, 564, 59–76. [Google Scholar] [CrossRef]
- Parnaud, G.; Corpet, D.E.; Gamet-Payrastre, L. Cytostatic Effect of Polyethylene Glycol on Human Colonic Adenocarcinoma Cells. Int. J. Cancer 2001, 92, 63–69. [Google Scholar] [CrossRef]
- Choi, Y.-A.; Rho Chin, B.; Hoon Rhee, D.; Choi, H.-G.; Chang, H.-W.; Kim, J.-H.; Baek, S.-H. Methyl-β-Cyclodextrin Inhibits Cell Growth and Cell Cycle Arrest via a Prostaglandin E(2) Independent Pathway. Exp. Mol. Med. 2004, 36, 78–84. [Google Scholar] [CrossRef]
- Poon, R.Y.C. Cell Cycle Control: A System of Interlinking Oscillators. In Cell Cycle Oscillators. Methods in Molecular Biology; Humana Press: New York, NY, USA, 2016; ISBN 978-1-4939-2957-3. [Google Scholar]
- Maki, M.A.A.; Cheah, S.-C.; Bayazeid, O.; Kumar, P.V. Cyclodextrin Inclusion Complex Inhibits Circulating Galectin-3 and FGF-7 and Affects the Reproductive Integrity and Mobility of Caco-2 Cells. Sci. Rep. 2020, 10, 17468. [Google Scholar] [CrossRef]
- Guerra, F.S.; da Silva Sampaio, L.; Konig, S.; Bonamino, M.; Rossi, M.I.D.; Costa, M.L.; Fernandes, P.; Mermelstein, C. Membrane Cholesterol Depletion Reduces Breast Tumor Cell Migration by a Mechanism That Involves Non-Canonical Wnt Signaling and IL-10 Secretion. Transl. Med. Commun. 2016, 1, 3–10. [Google Scholar] [CrossRef]
- Verhoef, J.J.F.; Anchordoquy, T.J. Questioning the Use of PEGylation for Drug Delivery. Drug Deliv. Transl. Res. 2013, 3, 499–503. [Google Scholar] [CrossRef]
- Hailstones, D.; Sleer, L.S.; Parton, R.G.; Stanley, K.K. Regulation of Caveolin and Caveolae by Cholesterol in MDCK Cells. J. Lipid Res. 1998, 39, 369–379. [Google Scholar] [CrossRef]
- Francis, S.A.; Kelly, J.M.; McCormack, J.; Rogers, R.A.; Jean, L.; Schneeberger, E.E.; Lynch, R.D. Rapid Reduction of MDCK Cell Cholesterol by Methyl-β-Cyclodextrin Alters Steady State Transepithelial Electrical Resistance. Eur. J. Cell Biol. 1999, 78, 473–484. [Google Scholar] [CrossRef]
- Tian, Z.; Si, L.; Meng, K.; Zhou, X.; Zhang, Y.; Zhou, D.; Xiao, S. Inhibition of Influenza Virus Infection by Multivalent Pentacyclic Triterpene-Functionalized per- O -Methylated Cyclodextrin Conjugates. Eur. J. Med. Chem. 2017, 134, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, X.; Zhu, Y.; Si, L.; Zhang, B.; Zhang, Y.; Zhang, L.; Zhou, D.; Xiao, S. Synthesis of a Hexavalent Betulinic Acid Derivative as a Hemagglutinin-Targeted Influenza Virus Entry Inhibitor. Mol. Pharm. 2020, 17, 2546–2554. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Guo, Y.; He, Y.; Ren, T.; Yin, L.; Fawcett, J.P.; Gu, J.; Sun, H. Impact of Molecular Weight on the Mechanism of Cellular Uptake of Polyethylene Glycols (PEGs) with Particular Reference to P-Glycoprotein. Acta Pharm. Sin. B 2020, 10, 2002–2009. [Google Scholar] [CrossRef] [PubMed]
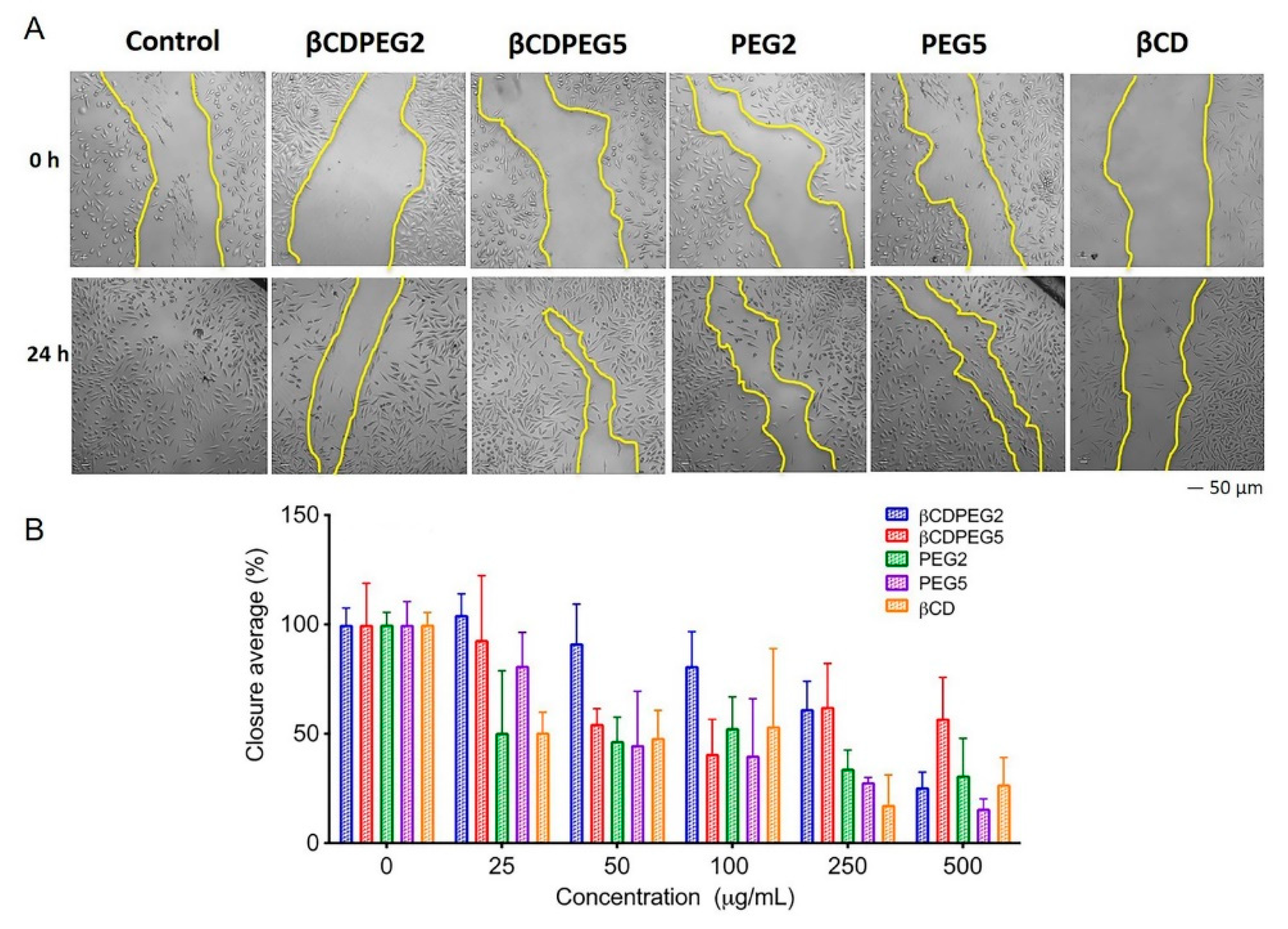
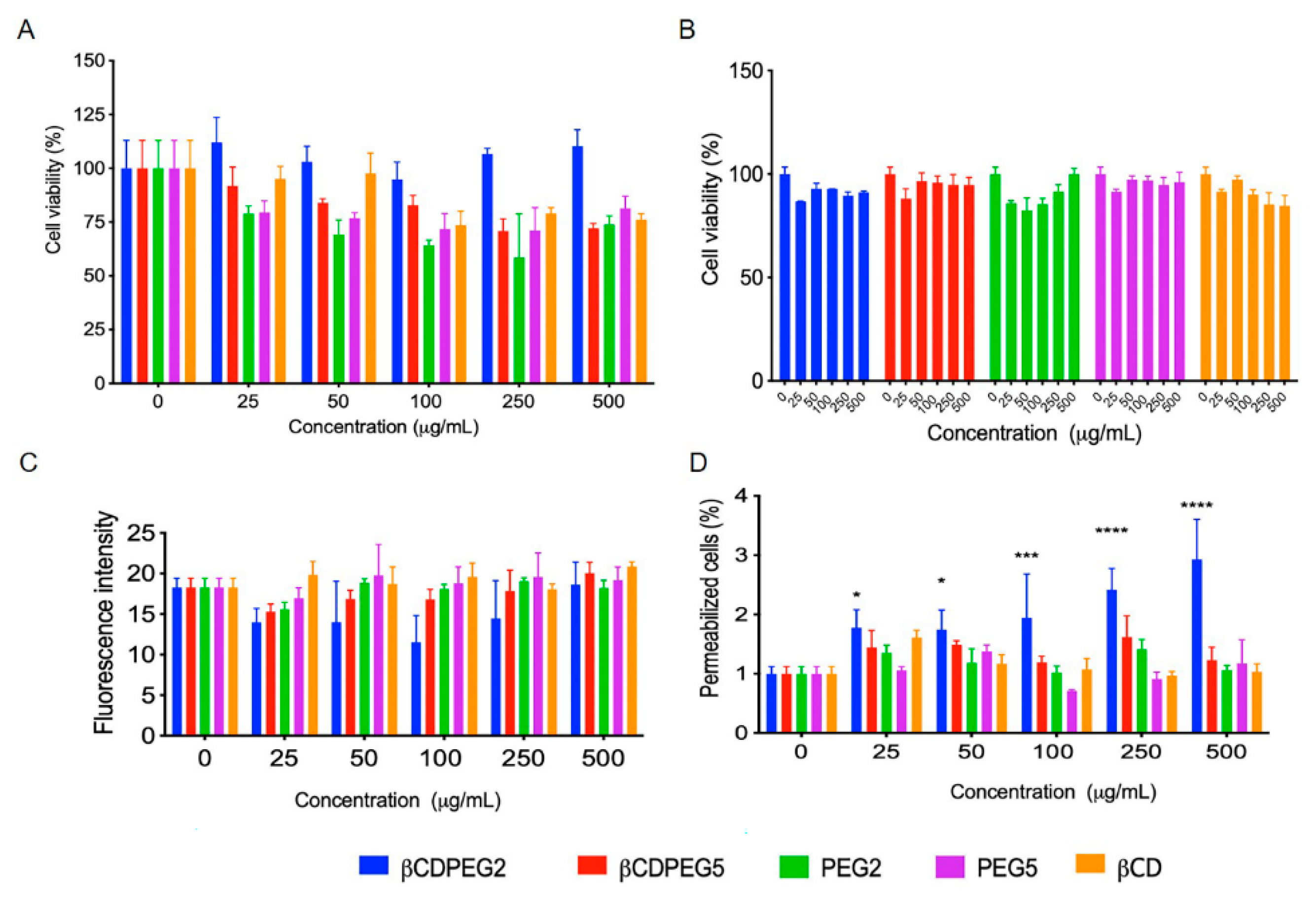
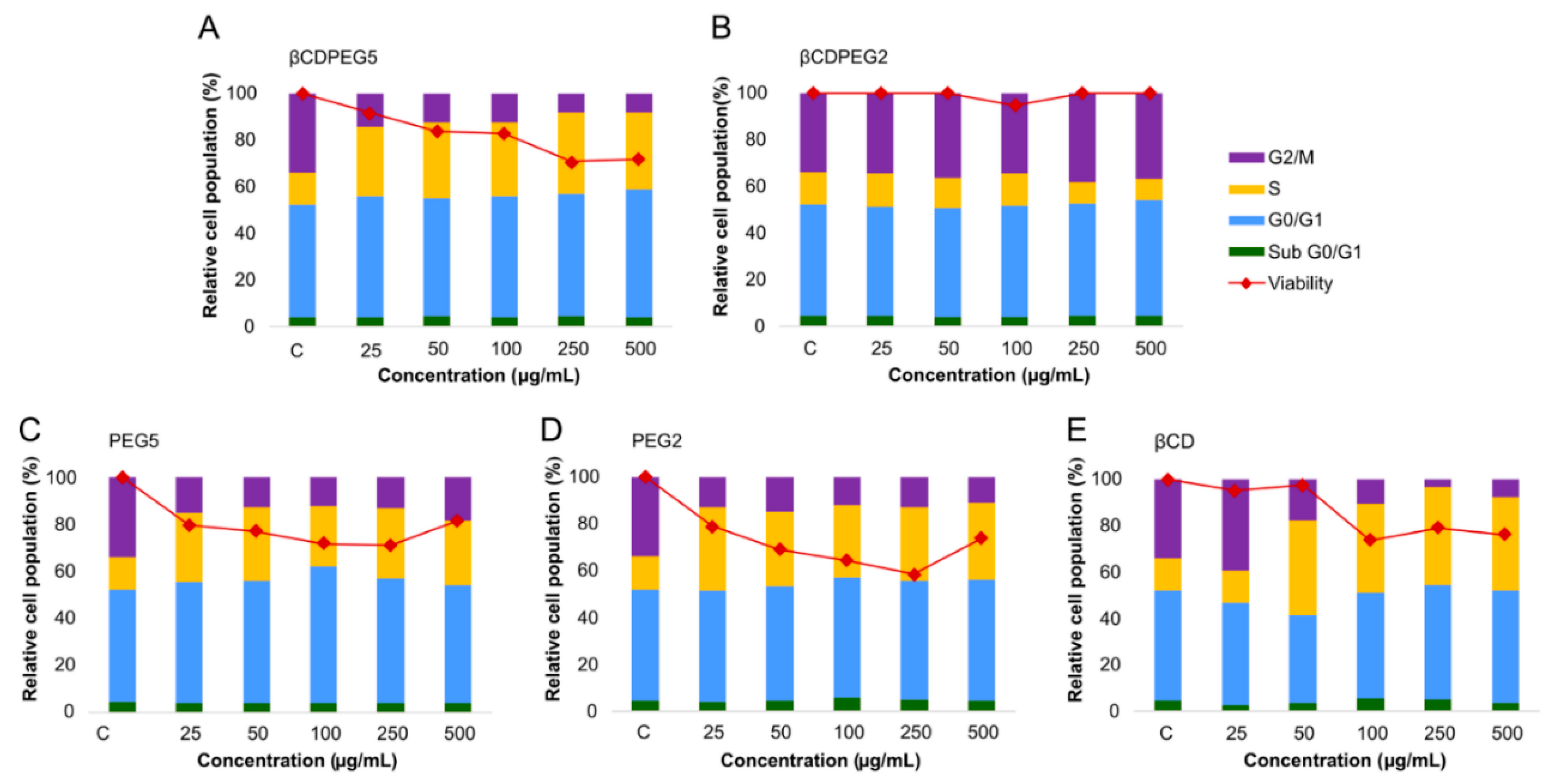
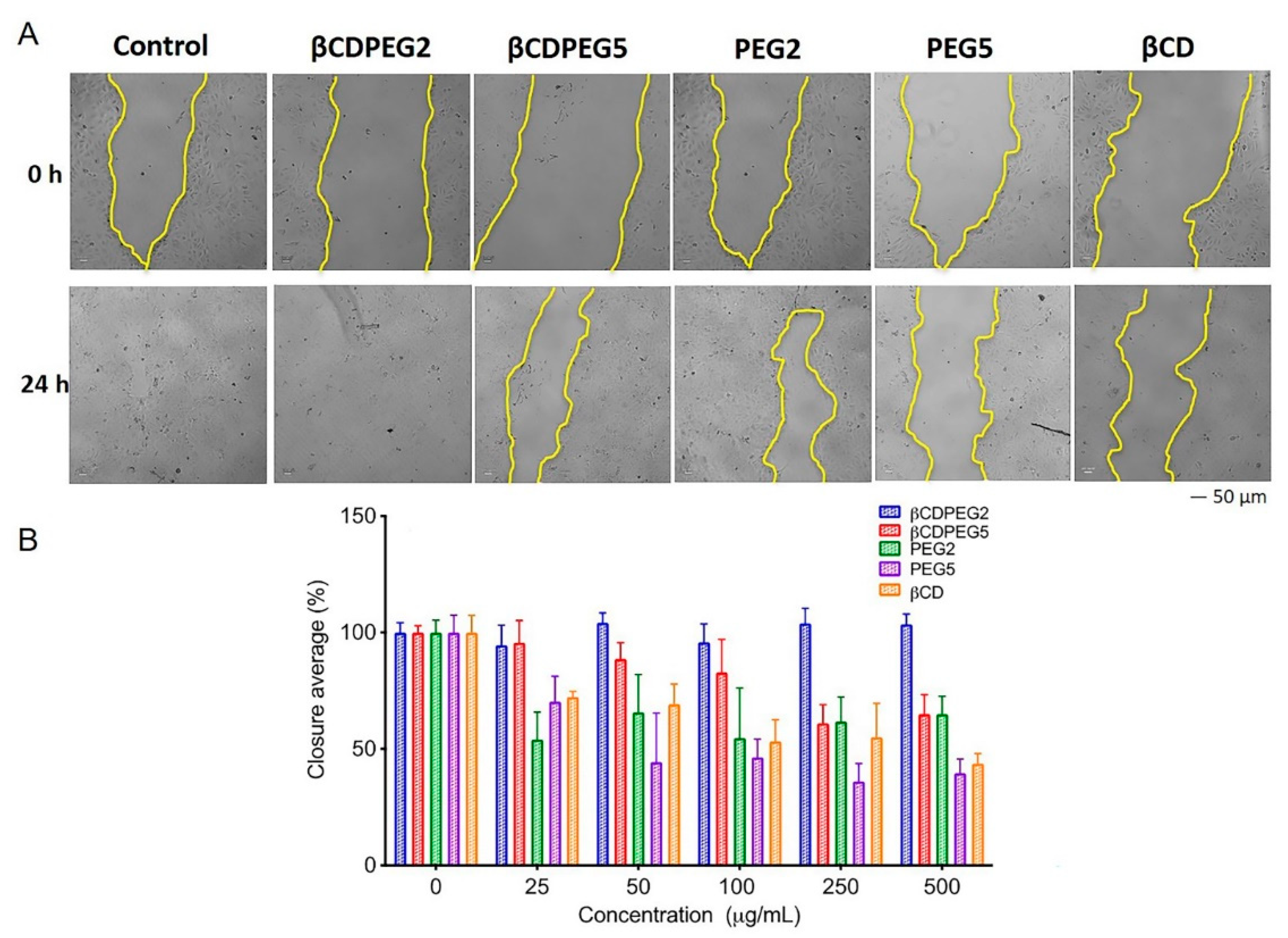
| Cellular Response | MC3T3-E1 Osteoblasts | MDCK Cells | RAW 264.7 Macrophages |
|---|---|---|---|
| Cell viability | βCDPEG5 > βCDPEG2 PEG2 > βCDPEG2 PEG5 = βCDPEG5 | βCDPEG5 > βCDPEG2 PEG2 > βCDPEG2 PEG5 = βCDPEG5 | βCDPEG5 = PEG5 = βCDPEG2 = PEG2 |
| ROS generation | PEG5 > βCDPEG5 = βCDPEG2 = PEG2 | βCDPEG5 = PEG5 = βCDPEG2 = PEG2 | βCDPEG5 = βCDPEG2 PEG5 = PEG2 βCDPEGs > PEGs |
| Cell cycle | βCDPEG5 ** > βCDPEG2 * PEG2 * > βCDPEG2 * PEG5 ** = βCDPEG5 ** | βCDPEG5 * > βCDPEG2 * PEG2 * > βCDPEG2 * PEG5 * < βCDPEG5 * | N/A |
| § Cell migration | βCDPEG5 < βCDPEG2 PEG2 < βCDPEG2 PEG5 > βCDPEG5 | βCDPEG5 > βCDPEG2 PEG2 > βCDPEG2 PEG5 > βCDPEG5 | N/A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rincón-López, J.; Martínez-Aguilera, M.; Guadarrama, P.; Juarez-Moreno, K.; Rojas-Aguirre, Y. Exploring In Vitro Biological Cellular Responses of Pegylated β-Cyclodextrins. Molecules 2022, 27, 3026. https://doi.org/10.3390/molecules27093026
Rincón-López J, Martínez-Aguilera M, Guadarrama P, Juarez-Moreno K, Rojas-Aguirre Y. Exploring In Vitro Biological Cellular Responses of Pegylated β-Cyclodextrins. Molecules. 2022; 27(9):3026. https://doi.org/10.3390/molecules27093026
Chicago/Turabian StyleRincón-López, Juliana, Miguelina Martínez-Aguilera, Patricia Guadarrama, Karla Juarez-Moreno, and Yareli Rojas-Aguirre. 2022. "Exploring In Vitro Biological Cellular Responses of Pegylated β-Cyclodextrins" Molecules 27, no. 9: 3026. https://doi.org/10.3390/molecules27093026
APA StyleRincón-López, J., Martínez-Aguilera, M., Guadarrama, P., Juarez-Moreno, K., & Rojas-Aguirre, Y. (2022). Exploring In Vitro Biological Cellular Responses of Pegylated β-Cyclodextrins. Molecules, 27(9), 3026. https://doi.org/10.3390/molecules27093026







