Investigation of an Allosteric Deoxyhypusine Synthase Inhibitor in P. falciparum
Abstract
1. Introduction
2. Results
2.1. 6-Bromo-N-(1H-indol-4yl)-1-benzothiophene-2-carboxamide Has No Impact on Parasite Growth
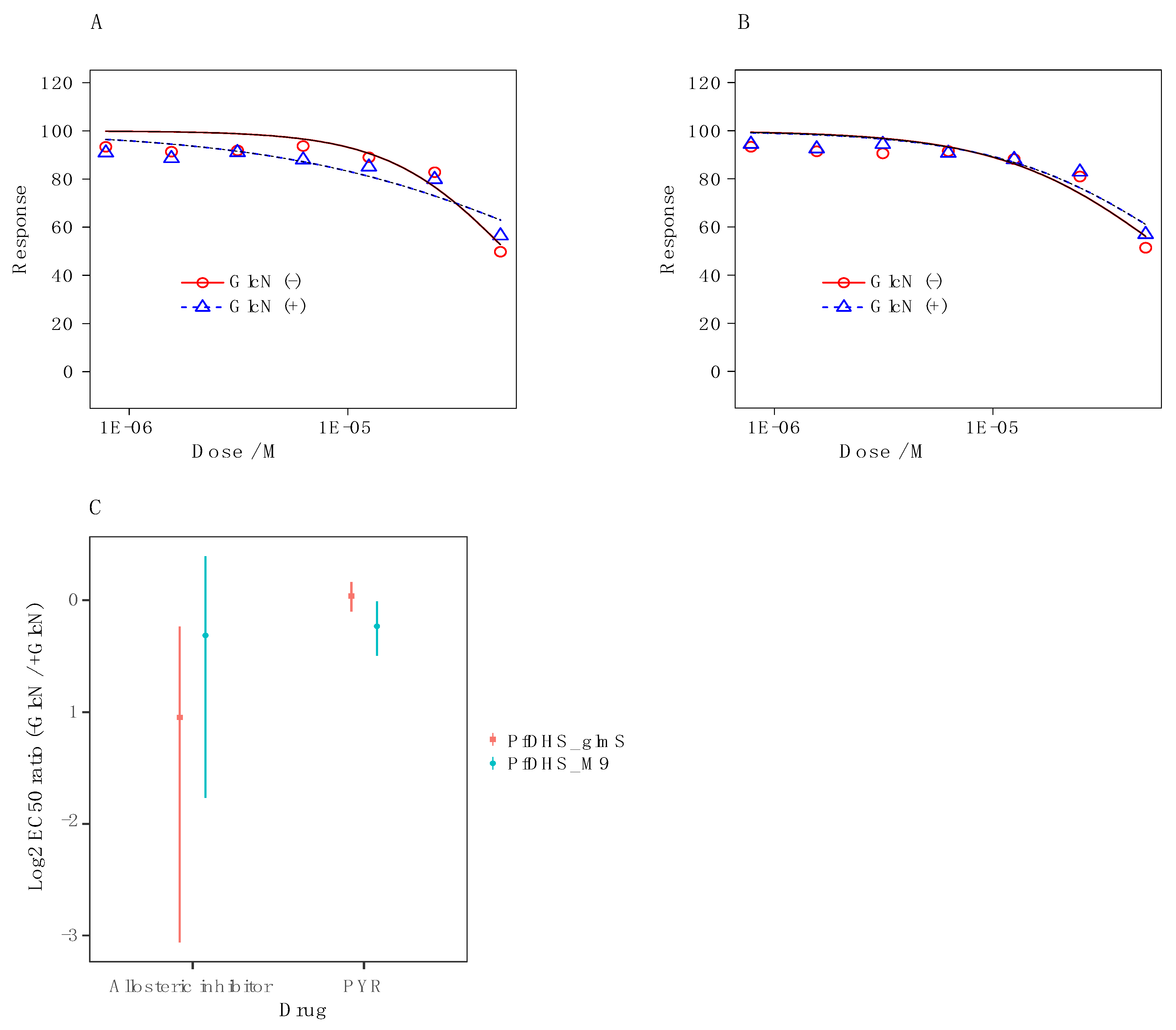
2.2. 6-Bromo-N-(1H-indol-4yl)-1-benzothiophene-2-carboxamide Does Not Primarily Target the DHS Protein in P. falciparum
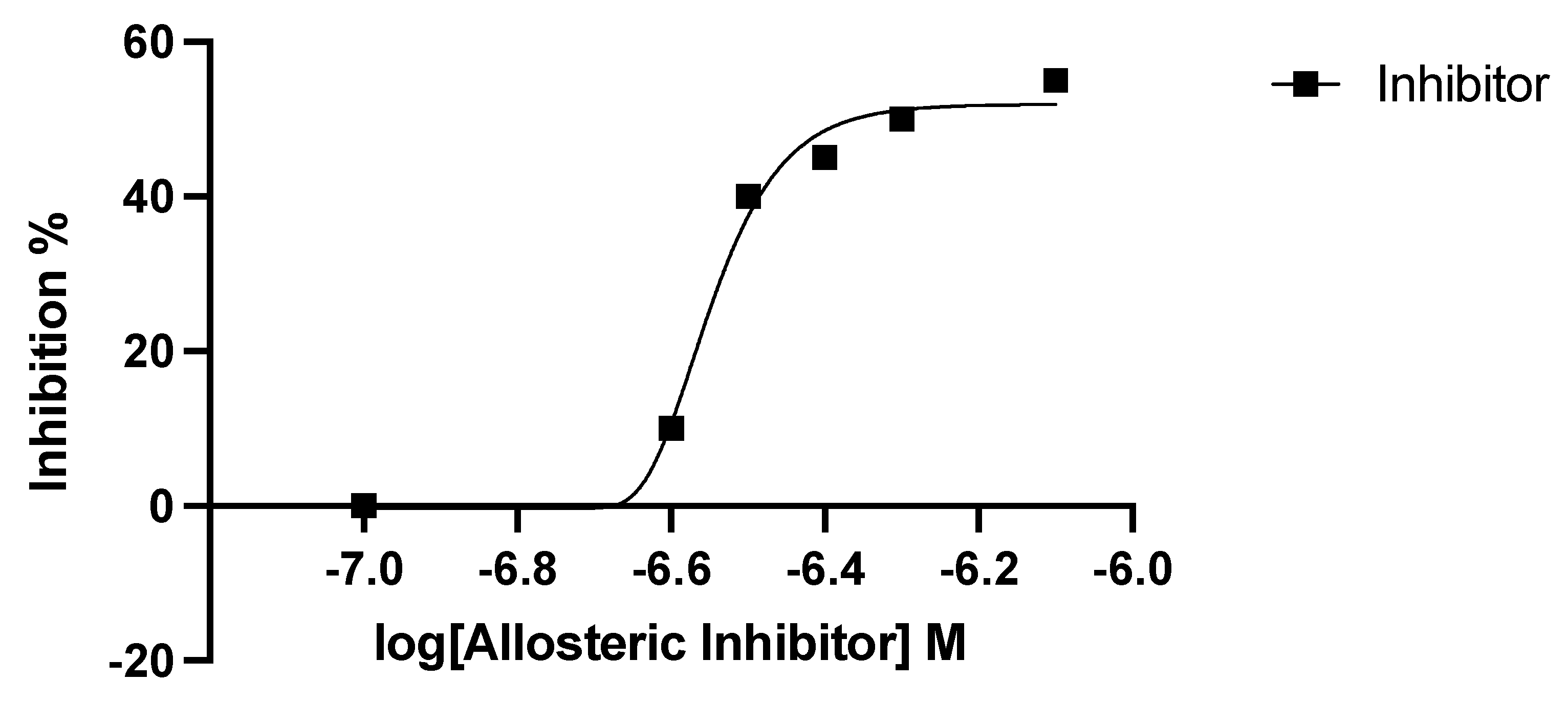
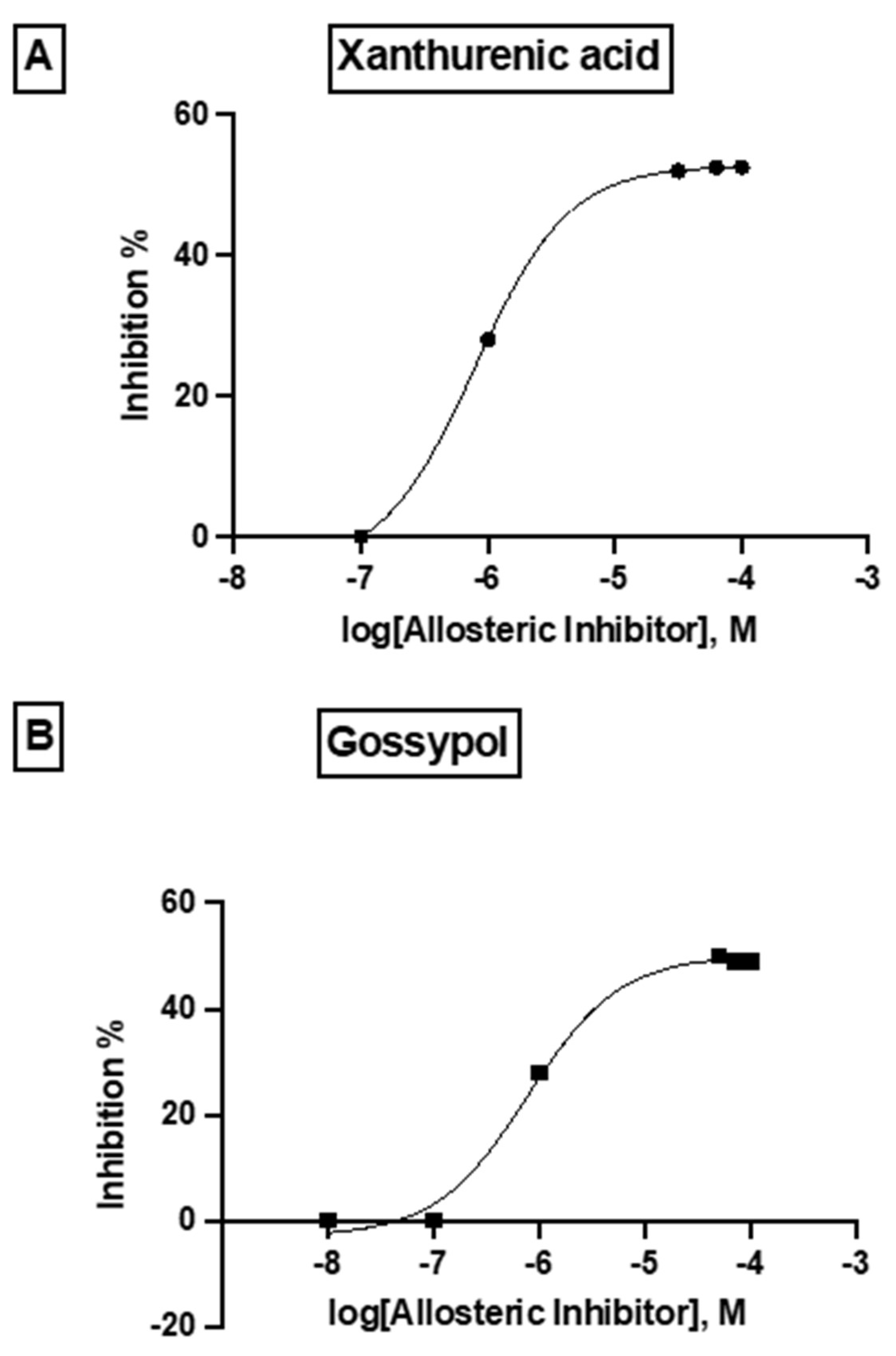
2.3. Inhibitor Studies with Recombinant, Purified dhs from P. falciparum Suggest a Different Allosteric Site in the Enzyme of the Parasite
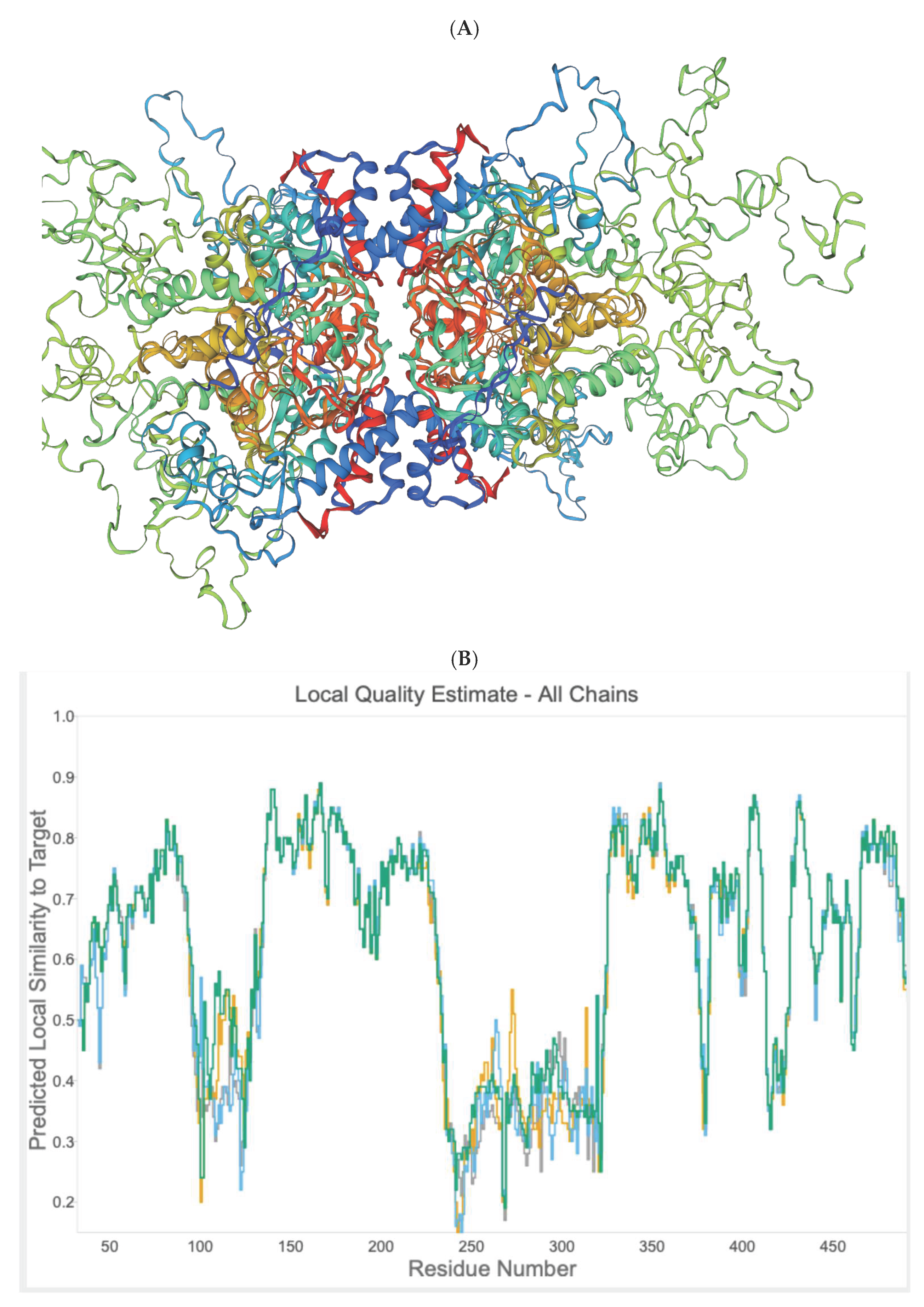
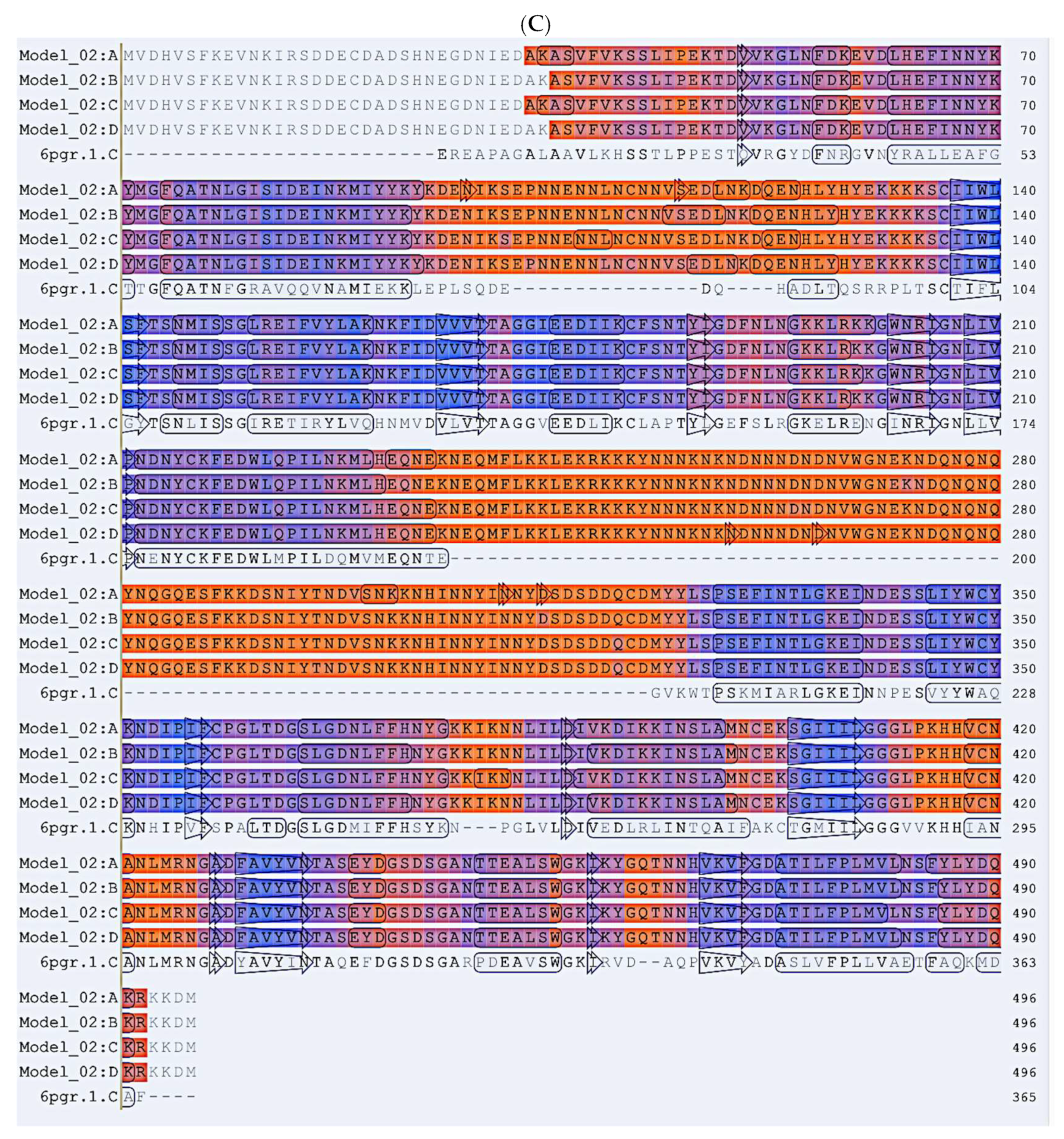
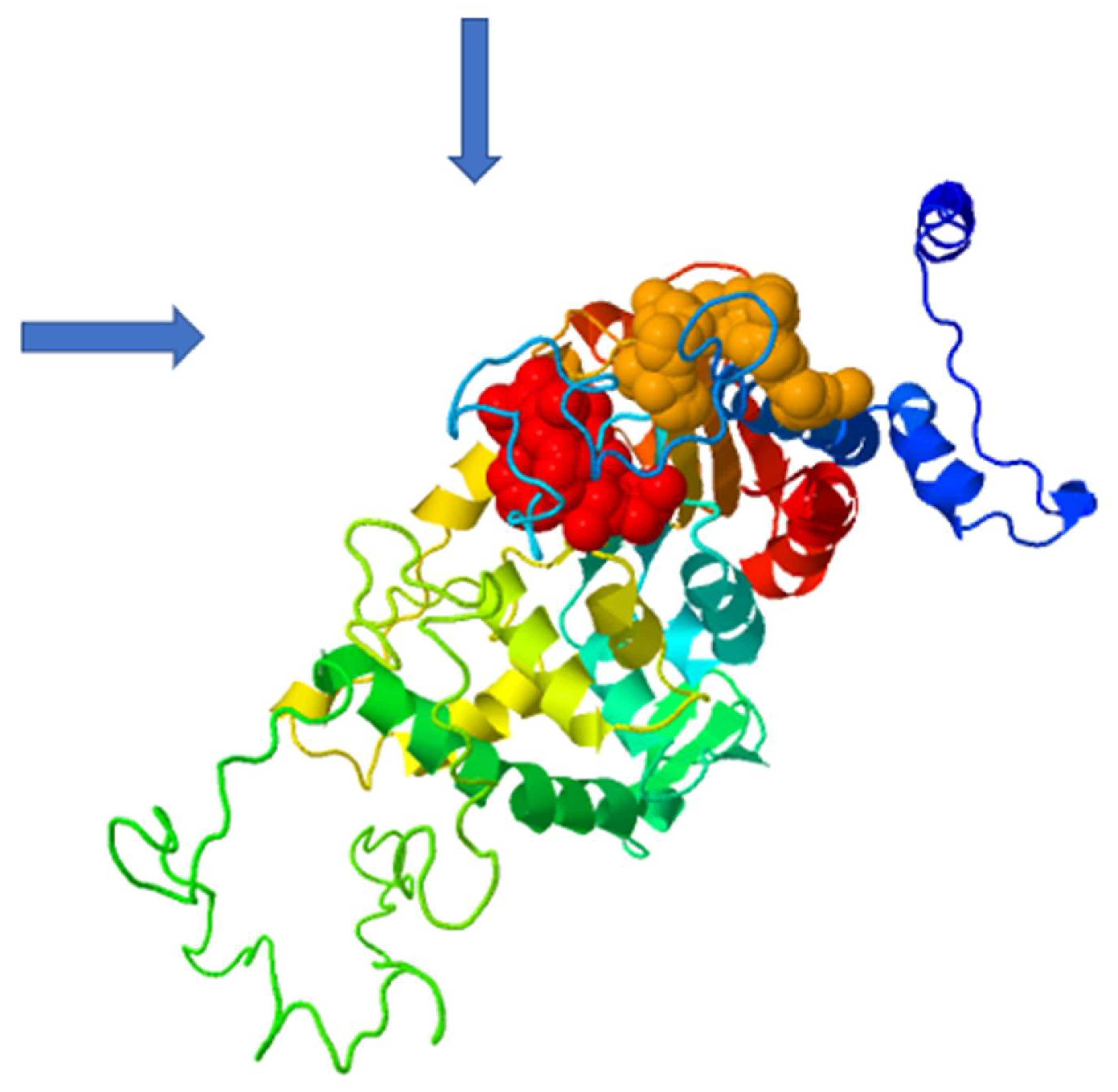
2.4. In Silico Prediction of Allosteric Sites in P. falciparum DHS

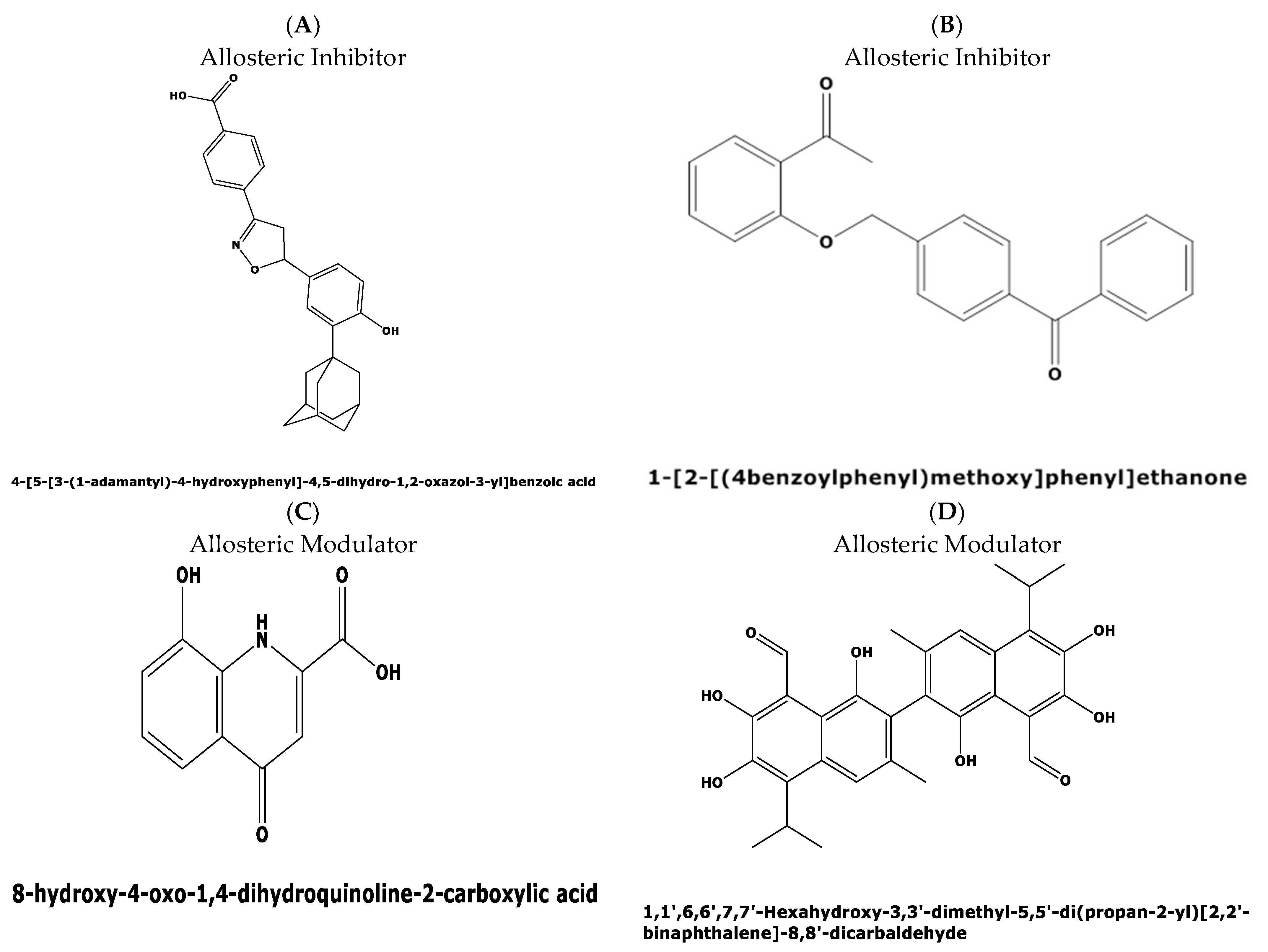
3. Discussion
4. Materials and Methods
4.1. Growth Inhibition of P. falciparum In Vitro Cultures and Determination of IC50 Values for Dd2 and 3D7 Strains Using a SYBR Green 1(SG)-Based In Vitro
4.2. Dose–Response Growth Inhibition Assays
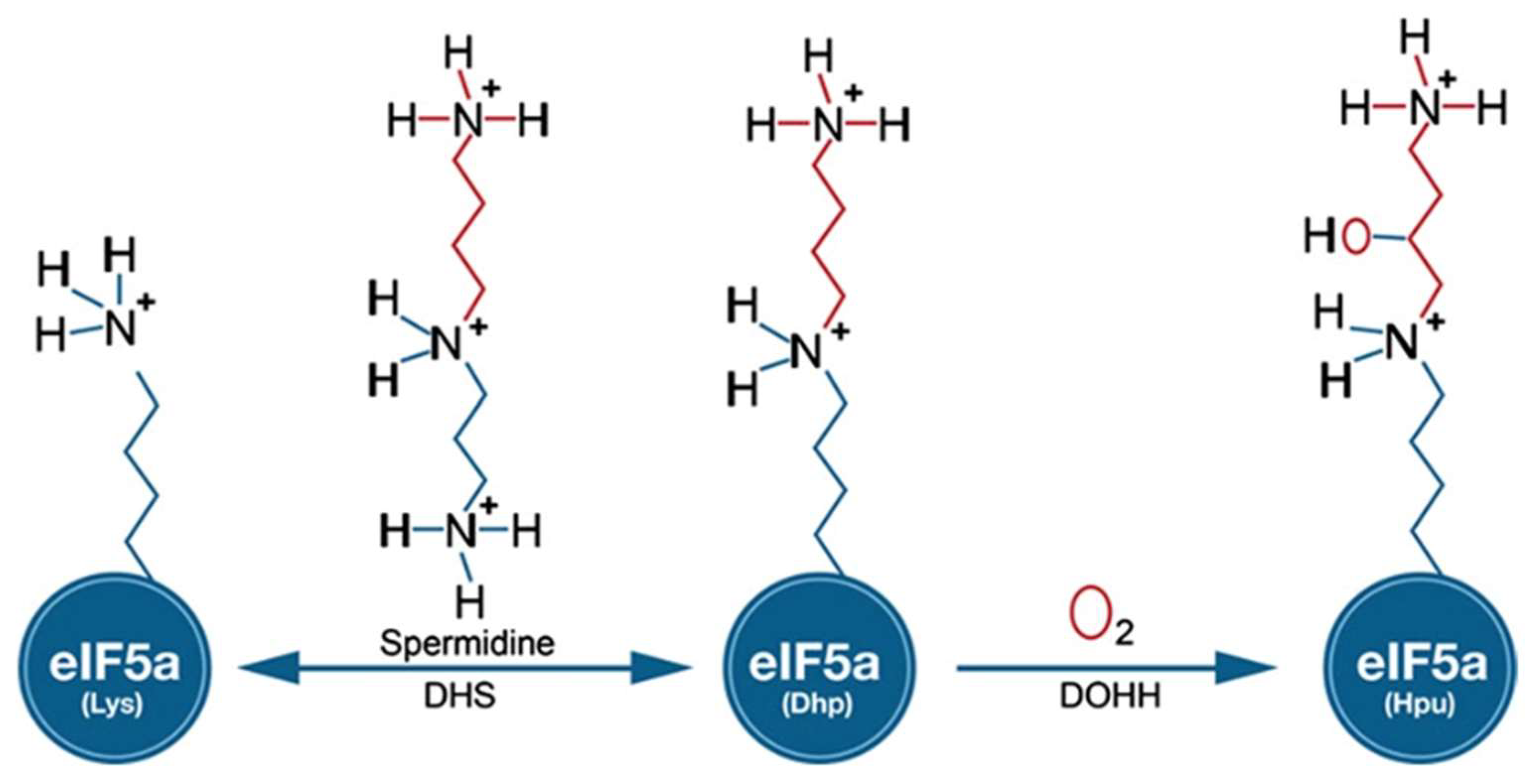
4.3. Enzymatic Synthesis of Deoxyhypusine
4.4. GC MS Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- WHO Strategic Advisory Group on Malaria Eradication. Malaria Eradication: Benefits, Future Scenarios and Feasibility; World Health Organization: Geneva, Switzerland, 2019; pp. 1–19. [Google Scholar]
- Chandramohan, D.; Zongo, I.; Sagara, I.; Cairns, M.; Yerbanga, R.S.; Diarra, M.; Nikièma, F.; Tapily, A.; Sompougdou, F.; Issiaka, D.; et al. Seasonal Malaria Vaccination with or without Seasonal Malaria Chemoprevention. N. Engl. J. Med. 2021, 385, 1015–1017. [Google Scholar] [CrossRef] [PubMed]
- Maxmen, A. Scientists Hail Historic Malaria Vaccine Approval—But Point to Challenges Ahead. Nature. 2021. Available online: https://www.nature.com/articles/d41586-021-02755-5 (accessed on 13 January 2022).
- Mwakingwe-Omari, A.; Healy, S.A.; Lane, J.; Cook, D.M.; Kalhori, S.; Wyatt, C.; Kolluri, A.; Marte-Salcedo, O.; Imeru, A.; Nason, N.; et al. Two chemoattenuated PfSPZ malaria vaccines induce sterile hepatic immunity. Nature 2021, 595, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Fiddock, D.A. CRISPR-mediated genome editing of Plasmodium falciparum malaria parasites. Genome Med. 2014, 26, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Baum, J.; Papenfuss, A.T.; Mair, G.R.; Janse, C.J.; Vlachou, D.; Waters, A.P.; Cowman, A.F.; Crabb, B.S.; de Koning-Ward, T.F. Molecular genetics and comparative genomics reveal RNAi is not functional in malaria parasites. Nucleic Acids Res. 2009, 37, 3788–3798. [Google Scholar] [CrossRef]
- Shaw, P.J.; Aroonsri, A. Tools for attenuation of gene expression in malaria parasites. Int. J. Parasitol. 2017, 47, 385–389. [Google Scholar] [CrossRef]
- Knuepfer, E.; Napiorkowska, M.; Van Oij, C.; Holder, A. Generating conditional gene knockouts in Plasmodium—A toolkit to produce stable DiCre recombinase-expressing parasite lines using CRISPR/Cas9. Sci. Rep. 2017, 7, 3881–3893. [Google Scholar] [CrossRef]
- Barrick, J.E.; Corbino, K.A.; Winkler, W.C.; Mandal, A.N.M.; Collins, J.; Lee, M.; Roth, A.; Sudarsan, N.; Jona, I.; Wickiser, K.J.; et al. New RNA motifs suggest an expanded scope for riboswitches in bacterial genetic control. Proc. Natl. Acad. Sci. USA 2004, 101, 6421–6426. [Google Scholar] [CrossRef]
- Prommana, P.; Uthaipibull, C.; Wongsombat, C.; Kamchonwong Paisan, S.; Yuthavong, Y.; Knuepfer, E.; Holder, A.A.; Shaw, P.J. Inducible Knockdown of Plasmodium Gene Expression Using the glmS Ribozyme. PLoS ONE 2013, 8, e73783. [Google Scholar] [CrossRef]
- Park, M.H.; Wolff, E.C. Hypusine, a polyamine-derived amino acid critical for eukaryotic translation. J. Biol. Chem. 2018, 293, 18710–18718. [Google Scholar] [CrossRef]
- Kaiser, A. Targeting malaria. In Biological Aspects of Biogenic Amines, Polamines and Conjugates; Dendrifosse, G., Ed.; Transworld Research Network: Trivandrum, India, 2009; pp. 421–439. [Google Scholar]
- Kersting, D.; Krüger, M.; Sattler, J.M.; Mueller, A.K.; Kaiser, A. A suggested vital function for eIF-5A and dhs genes during murine malaria blood-stage infection. FEBS Open Bio 2016, 23, 860–872. [Google Scholar] [CrossRef]
- Aroonsri, A.; Posayapisit, N.; Kongsee, J.; Siripan, O.; Vitsupakorn, D.; Utaida, S.; Uthaipibull, C.; Kamchonwongpaisan, S.; Shaw, P.J. Validation of Plasmodium falciparum deoxyhypusine synthase as an antimalarial target. PeerJ 2019, 7, e6713. [Google Scholar] [CrossRef]
- Tanaka, Y.; Kurasawa, O.; Yokota, A.; Klein, M.G.; Ono, K.; Saito, B.; Matsumoto, S.; Okaniwa, M.; Ambrus-Aikelin, G.; Morishita, D.; et al. Discovery of Novel Allosteric Inhibitors of Deoxyhypusine Synthase. J. Med. Chem. 2021, 26, 3215–3226. [Google Scholar] [CrossRef]
- Colvin, S.C.; Maier, B.; Morris, D.L.; Tersey, S.A.; Mirmira, R.G. Deoxyhypusine synthase promotes differentiation and proliferation of T helper type 1 (Th1) cells in autoimmune diabetes. J. Biol. Chem. 2013, 288, 36226–36335. [Google Scholar] [CrossRef]
- Njuguna, J.T.; von Koschitzky, I.; Gerhardt, H.; Lämmerhofer, M.; Choucry, A.; Pink, M.; Schmitz-Spahnke, S.; Bakheit, M.A.; Strube, C.; Kaiser, A. Target evaluation of deoxyhypusine synthase from Theileria parva the neglected animal parasite and its relationship to Plasmodium. Bioorg. Med. Chem. 2014, 22, 4338–4346. [Google Scholar] [CrossRef]
- Umland, T.C.; Wolff, E.C.; Park, M.H.; Davies, D.R. A new crystal structure of deoxyhypusine synthase reveals the configuration of the active enzyme and of an enzyme.NAD.inhibitor ternary complex. J. Biol. Chem. 2014, 2, 28697–28705. [Google Scholar] [CrossRef]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 2, 296–303. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Tian, H.; Jiang, X.; Tao, P. PASSer: Prediction of Allosteric Site Server. Mach. Learn. Sci. Technol. 2021, 2, 035015. [Google Scholar] [CrossRef]
- Sheik Amamuddy, O.; Veldman, W.; Manyumwa, C.; Khairallah, A.; Agajanian, S.; Oluyemi, O.; Verkhivker, G.; Tastan Bishop, O. Integrated computational approaches and tools for allosteric drug discovery. Int. J Mol. Sci. 2020, 21, 847. [Google Scholar] [CrossRef]
- Yokomizo, T.; Nakamura, M.; Shimizu, T. Leukotriene receptors as potential therapeutic targets. J. Clin. Investig. 2018, 128, 2691–2701. [Google Scholar] [CrossRef]
- Auburn, S.; Fry, A.E.; Clark, T.G.; Campino, S.; Diakite, M.; Green, A.; Richardson, A.; Jallow, M.; Sisay-Joof, F.P.; Pinder, M.; et al. Further evidence supporting a role for gs signal transduction in severe malaria pathogenesis. PLoS ONE 2010, 5, e10017. [Google Scholar] [CrossRef] [PubMed]
- Billker, O.; Lindo, V.; Panico, M.; Etienne, A.E.; Paxton, T.; Dell, A.; Rogers, M.; Sinden, R.E.; Morris, H.R. Identification of xanthurenic acid as the putative inducer of malaria development in the mosquito. Nature 1998, 392, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Conners, R.; Schambach, F.; Read, J.; Cameron, A.; Sessions, R.B.; Vivas, L.; Easton, A.; Croft, S.L.; Brady, R.L. Mapping the binding site for gossypol-like inhibitors of Plasmodium falciparum lactate dehydrogenase. Mol. Biochem. Parasitol. 2005, 142, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Magid, A.F. Allosteric Modulators: An Emerging Concept in DrugDiscovery. ACS Med. Chem. Lett. 2015, 6, 104–107. [Google Scholar] [CrossRef]
- Huang, M.; Song, K.; Liu, X.; Lu, S.; Shen, Q.; Wang, R.; Gao, J.; Hong, Y.; Li, Q.; Ni, D.; et al. AlloFinder: A strategy for allosteric modulator discovery and allosterome analyses. Nucleic Acids Res. 2018, 46, 451–458. [Google Scholar] [CrossRef]
- Song, K.; Zhang, J.; Lu, S. Progress in Allosteric Database. Adv. Exp. Med. Biol. 2019, 116, 65–87. [Google Scholar] [CrossRef]
- Hernández Alvarez, L.; Barreto Gomes, D.E.; Hernández González, J.E.; Pascutti, P.G. Dissecting a novel allosteric mechanism of cruzain: A computer-aided approach. PLoS ONE 2019, 14, e0211227. [Google Scholar] [CrossRef]
- Guiterrez, E.; Shin, B.S.; Woolstenhulme, C.J.; Kim, J.R.; Saini, P.; Buskirk, A.R.; Dever, T.E. EIF-5A promotes translation of polyproline motifs. Mol. Cell 2013, 51, 35–45. [Google Scholar] [CrossRef]
- Liao, D.I.; Wolff, E.C.; Park, M.H.; Davies, D.R. Crystal structure of the NAD complex of human deoxyhypusine synthase: An enzyme with a ball-and-chain mechanism for blocking the active site. Structure 1998, 15, 23–32. [Google Scholar] [CrossRef]
- Specht, S.; Sarite, S.R.; Hauber, I.; Hauber, J.; Görbig, U.F.; Meier, C.; Bevec, D.; Hoerauf, A.; Kaiser, A. The guanylhydrazone CNI-1493: An inhibitor with dual activity against malaria-inhibition of host cell pro-inflammatory cytokine release and parasitic deoxyhypusine synthase. Parasitol. Res. 2008, 102, 1177–1184. [Google Scholar] [CrossRef]
- Farfán-López, M.; Espinoza-Culupú, A.; García-de-la-Guarda, R.; Serral, F.; Sosa, E.; Palomino, M.M.; Fernández Do Porto, D.A. Prioritisation of potential drug targets against Bartonella bacilliformis by an integrative in-silico approach. Mem. Inst. Oswaldo Cruz Rio J. 2020, 115, e200184. [Google Scholar] [CrossRef]
- Dery, V.; Duah, N.O.; Ayanful-Torgby, R.; Matrevi, S.A.; Anto, F.; Quashie, N.B. An improved SYBR Green-1-based fluorescence method for the routine monitoring of Plasmodium falciparum resistance to anti-malarial drugs. Malar. J. 2015, 14, 481. [Google Scholar] [CrossRef]
- Aroonsri, A.; Akinola, O.; Posayapisit, N.; Songsungthong, W.; Uthaipibull, C.; Kamchonwongpaisan, S.; Gbotosho, G.O.; Yuthavong, Y.; Shaw, P.J. Identifying antimalarial compounds targeting dihydrofolate reductase-thymidylate synthase (DHFR-TS) by chemogenomic profiling. Int. J. Parasitol. 2016, 46, 527–535. [Google Scholar] [CrossRef]
- Smilkstein, M.; Sriwilaijaroen, R.; Kelly, J.X.; Wilairat, P.; Riscoe, M. Simple and inexpensive fluorescence-based technique for high-throughput antimalarial drug screening. Antimicrob. Agents Chemother. 2004, 48, 1803–1806. [Google Scholar] [CrossRef]
- Ritz, C.; Streibig, J.C. Bioassay analysis using R. J. Stat. Softw. 2005, 12, 1–22. [Google Scholar] [CrossRef]
- Hammels, I.; Gottwald, A.; Nassar, M.; Zaghloul, M.S.; Motaal, B.A.; Hauber, J.; Hoerauf, A. Modification of eukaryotic initiation factor 5A from Plasmodium vivax by a truncated deoxyhypusine synthase from Plasmodium falciparum: An enzyme with dual enzymatic properties. Bioorg. Med. Chem. 2007, 15, 6200–6207. [Google Scholar] [CrossRef]
- Frommholz, D.; Kusch, P.; Blavid, R.; Scheer, H.; Tu, J.M.; Marcus, K.; Zhao, K.H.; Atemnkeng, V.; Marciniak, J.; Kaiser, A.E. Completing the hypusine pathway in Plasmodium. FEBS J. 2009, 276, 5881–5891. [Google Scholar] [CrossRef]
- Atemnkeng, V.A.; Pink, M.; Schmitz-Spanke, S.; Wu, X.J.; Dong, L.L.; Zhao, M.C.; Laufer, S.; Langer, B.; Kaiser, A. Deoxyhypusine Hydroxylase from Plasmodium vivax, the Neglected Human Malaria Parasite: Molecular Cloning, Expression and Specific Inhibition by the 5-LOX Inhibitor Zileuton. PLoS ONE 2013, 8, e58318. [Google Scholar] [CrossRef]
- Von Koschitzky, I.; Gerhardt, H.; Lämmerhofer, M.; Kohout, M.; Gehringer, M.; Laufer, S.; Pink, M.; Schmitz-Spanke, S.; Strube, C.; Kaiser, A. New insights into novel inhibitors against deoxyhypusine hydroxylase from Plasmodium falciparum: Compounds with an iron chelating potential. Amino Acids 2015, 476, 1155–1166. [Google Scholar] [CrossRef]

| Strain | Compound | Molecular Weight | IC50 (µM) |
|---|---|---|---|
| Dd2 | 6-bromo-N-(1H-indol-4yl)-1-benzothiophene-2-carboxamide | 371.30 | 46.1 |
| Dd2 | Amodiaquine | 464.80 | 0.031 |
| Dd2 | Chloroquine | 515.90 | 0.66 |
| 3D7 | 6-bromo-N-(1H-indol-4yl)-1-benzothiophene-2-carboxamide | 371.30 | 51.5 |
| 3D7 | Amodiaquine | 464.80 | 0.066 |
| 3D7 | Chloroquine | 515.90 | 0.03 |
| Organism | Protein | IC50 |
|---|---|---|
| P. falciparum | DHS | 0.340 µM |
| Human | DHS | 0.062 µM [15] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aroonsri, A.; Wongsombat, C.; Shaw, P.; Franke, S.; Przyborski, J.; Kaiser, A. Investigation of an Allosteric Deoxyhypusine Synthase Inhibitor in P. falciparum. Molecules 2022, 27, 2463. https://doi.org/10.3390/molecules27082463
Aroonsri A, Wongsombat C, Shaw P, Franke S, Przyborski J, Kaiser A. Investigation of an Allosteric Deoxyhypusine Synthase Inhibitor in P. falciparum. Molecules. 2022; 27(8):2463. https://doi.org/10.3390/molecules27082463
Chicago/Turabian StyleAroonsri, Aiyada, Chayaphat Wongsombat, Philip Shaw, Siegrid Franke, Jude Przyborski, and Annette Kaiser. 2022. "Investigation of an Allosteric Deoxyhypusine Synthase Inhibitor in P. falciparum" Molecules 27, no. 8: 2463. https://doi.org/10.3390/molecules27082463
APA StyleAroonsri, A., Wongsombat, C., Shaw, P., Franke, S., Przyborski, J., & Kaiser, A. (2022). Investigation of an Allosteric Deoxyhypusine Synthase Inhibitor in P. falciparum. Molecules, 27(8), 2463. https://doi.org/10.3390/molecules27082463








