Berberine, a Herbal Metabolite in the Metabolic Syndrome: The Risk Factors, Course, and Consequences of the Disease
Abstract
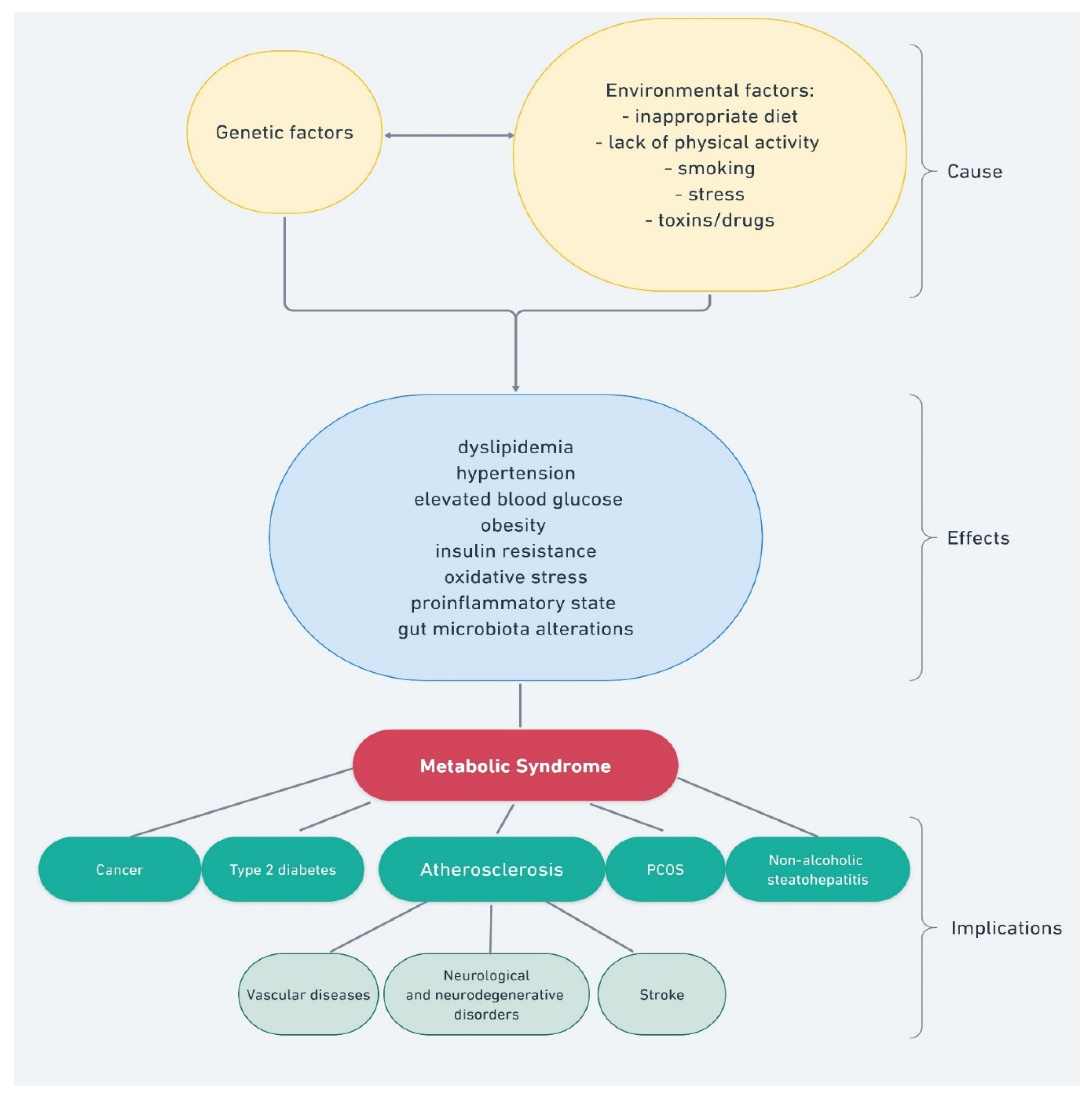
1. Introduction
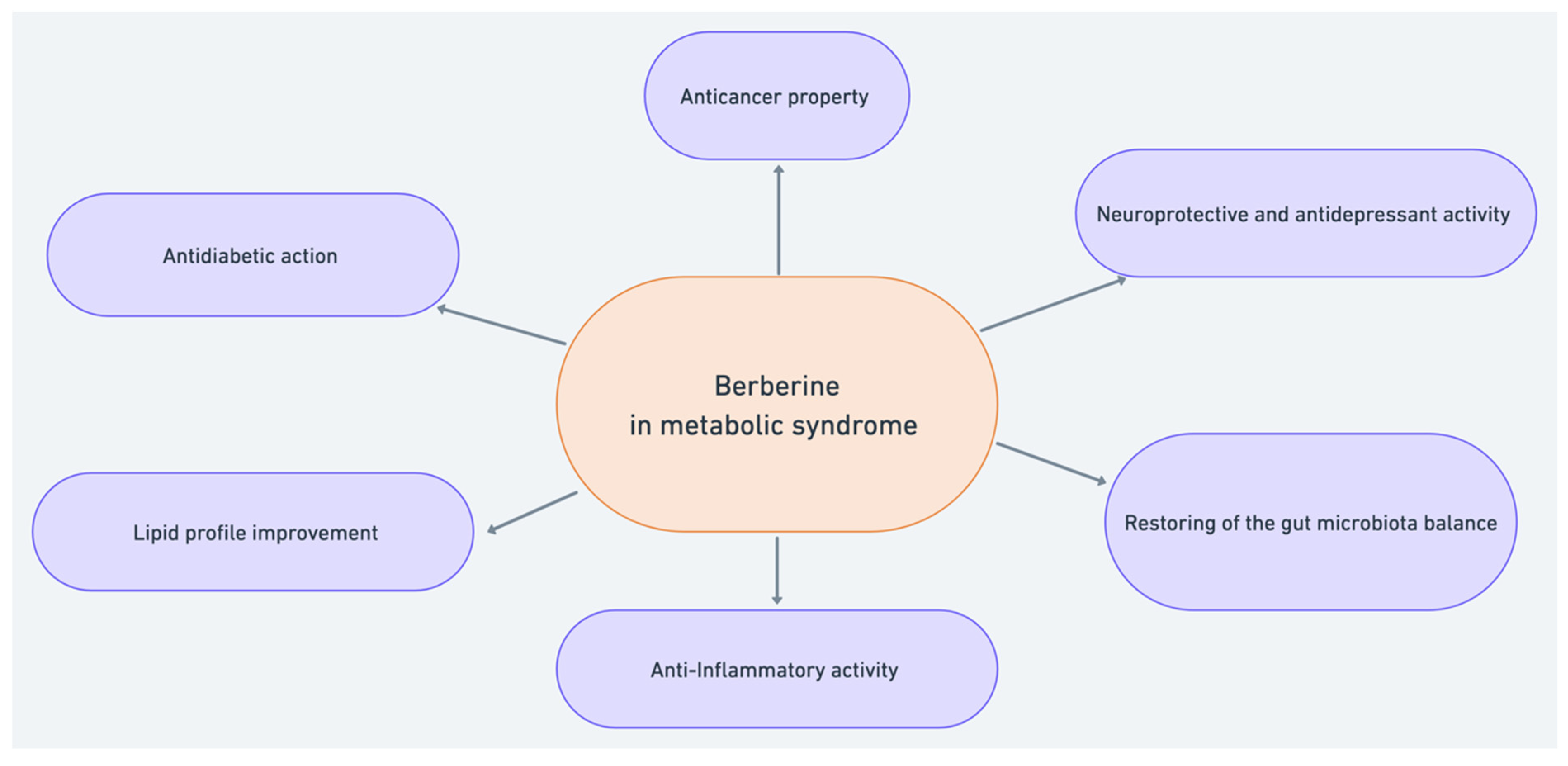
2. Antidiabetic Action of Berberine
3. The Cholesterol-Lowering Effect of Berberine
4. Berberine and Gut Microbiota
5. Anti-Inflammatory Activity of Berberine
6. Anticancer Activity of Berberine
7. Berberine in Mental Disorders within the Metabolic Syndrome
8. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The Metabolic Syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar] [CrossRef]
- Dietrich, P.; Hellerbrand, C. Non-Alcoholic Fatty Liver Disease, Obesity and the Metabolic Syndrome. Best Pract. Res. Clin. Gastroenterol. 2014, 28, 637–653. [Google Scholar] [CrossRef] [PubMed]
- Sherling, D.H.; Perumareddi, P.; Hennekens, C.H. Metabolic Syndrome. J. Cardiovasc. Pharmacol. Ther. 2017, 22, 365–367. [Google Scholar] [CrossRef] [PubMed]
- De la Iglesia, R.; Loria-Kohen, V.; Zulet, M.A.; Martinez, J.A.; Reglero, G.; Ramirez de Molina, A. Dietary Strategies Implicated in the Prevention and Treatment of Metabolic Syndrome. Int. J. Mol. Sci. 2016, 17, 1877. [Google Scholar] [CrossRef] [PubMed]
- Ganguli, R.; Strassnig, M. Prevention of Metabolic Syndrome in Serious Mental Illness. Psychiatr. Clin. 2011, 34, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Zarich, S.W. Metabolic Syndrome, Diabetes and Cardiovascular Events: Current Controversies and Recommendations. Minerva Cardioangiol. 2006, 54, 195–214. [Google Scholar]
- Och, A.; Podgórski, R.; Nowak, R. Biological Activity of Berberine—A Summary Update. Toxins 2020, 12, 713. [Google Scholar] [CrossRef]
- Zhao, J.V.; Yeung, W.-F.; Chan, Y.-H.; Vackova, D.; Leung, J.Y.Y.; Ip, D.K.M.; Zhao, J.; Ho, W.-K.; Tse, H.-F.; Schooling, C.M. Effect of Berberine on Cardiovascular Disease Risk Factors: A Mechanistic Randomized Controlled Trial. Nutrients 2021, 13, 2550. [Google Scholar] [CrossRef]
- Gonnelli, S.; Caffarelli, C.; Stolakis, K.; Cuda, C.; Giordano, N.; Nuti, R. Efficacy and Tolerability of a Nutraceutical Combination (Red Yeast Rice, Policosanols, and Berberine) in Patients with Low-Moderate Risk Hypercholesterolemia: A Double-Blind, Placebo-Controlled Study. Curr. Ther. Res. 2015, 77, 1–6. [Google Scholar] [CrossRef]
- Affuso, F.; Mercurio, V.; Ruvolo, A.; Pirozzi, C.; Micillo, F.; Carlomagno, G.; Grieco, F.; Fazio, S. A Nutraceutical Combination Improves Insulin Sensitivity in Patients with Metabolic Syndrome. World J. Cardiol. 2012, 4, 77–83. [Google Scholar] [CrossRef]
- Pirro, M.; Mannarino, M.R.; Ministrini, S.; Fallarino, F.; Lupattelli, G.; Bianconi, V.; Bagaglia, F.; Mannarino, E. Effects of a Nutraceutical Combination on Lipids, Inflammation and Endothelial Integrity in Patients with Subclinical Inflammation: A Randomized Clinical Trial. Sci. Rep. 2016, 6, 23587. [Google Scholar] [CrossRef] [PubMed]
- Spigoni, V.; Aldigeri, R.; Antonini, M.; Micheli, M.M.; Fantuzzi, F.; Fratter, A.; Pellizzato, M.; Derlindati, E.; Zavaroni, I.; Bonadonna, R.C.; et al. Effects of a New Nutraceutical Formulation (Berberine, Red Yeast Rice and Chitosan) on Non-HDL Cholesterol Levels in Individuals with Dyslipidemia: Results from a Randomized, Double Blind, Placebo-Controlled Study. Int. J. Mol. Sci. 2017, 18, 1498. [Google Scholar] [CrossRef] [PubMed]
- Solà, R.; Valls, R.-M.; Puzo, J.; Calabuig, J.-R.; Brea, A.; Pedret, A.; Moriña, D.; Villar, J.; Millán, J.; Anguera, A. Effects of Poly-Bioactive Compounds on Lipid Profile and Body Weight in a Moderately Hypercholesterolemic Population with Low Cardiovascular Disease Risk: A Multicenter Randomized Trial. PLoS ONE 2014, 9, e101978. [Google Scholar] [CrossRef] [PubMed]
- Trimarco, V.; Izzo, R.; Stabile, E.; Rozza, F.; Santoro, M.; Manzi, M.V.; Serino, F.; Schiattarella, G.G.; Esposito, G.; Trimarco, B. Effects of a New Combination of Nutraceuticals with Morus Alba on Lipid Profile, Insulin Sensitivity and Endotelial Function in Dyslipidemic Subjects. A Cross-over, Randomized, Double-Blind Trial. High Blood Press. Cardiovasc. Prev. 2015, 22, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-X.; Gao, Q.-Y.; Zou, T.-H.; Wang, B.-M.; Liu, S.-D.; Sheng, J.-Q.; Ren, J.-L.; Zou, X.-P.; Liu, Z.-J.; Song, Y.-Y.; et al. Berberine versus Placebo for the Prevention of Recurrence of Colorectal Adenoma: A Multicentre, Double-Blinded, Randomised Controlled Study. Lancet Gastroenterol. Hepatol. 2020, 5, 267–275. [Google Scholar] [CrossRef]
- Li, M.; Liu, Y.; Qiu, Y.; Zhang, J.; Zhang, Y.; Zhao, Y.; Jia, Q.; Li, J. The Effect of Berberine Adjunctive Treatment on Glycolipid Metabolism in Patients with Schizophrenia: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Psychiatry Res. 2021, 300, 113899. [Google Scholar] [CrossRef]
- Zhang, L.-S.; Zhang, J.-H.; Feng, R.; Jin, X.-Y.; Yang, F.-W.; Ji, Z.-C.; Zhao, M.-Y.; Zhang, M.-Y.; Zhang, B.-L.; Li, X.-M. Efficacy and Safety of Berberine Alone or Combined with Statins for the Treatment of Hyperlipidemia: A Systematic Review and Meta-Analysis of Randomized Controlled Clinical Trials. Am. J. Chin. Med. 2019, 47, 751–767. [Google Scholar] [CrossRef]
- Zhao, M.-M.; Lu, J.; Li, S.; Wang, H.; Cao, X.; Li, Q.; Shi, T.-T.; Matsunaga, K.; Chen, C.; Huang, H.; et al. Berberine Is an Insulin Secretagogue Targeting the KCNH6 Potassium Channel. Nat. Commun. 2021, 12, 5616. [Google Scholar] [CrossRef]
- Yin, J.; Xing, H.; Ye, J. Efficacy of Berberine in Patients with Type 2 Diabetes Mellitus. Metabolism 2008, 57, 712–717. [Google Scholar] [CrossRef]
- Wang, H.; Zhu, C.; Ying, Y.; Luo, L.; Huang, D.; Luo, Z. Metformin and Berberine, Two Versatile Drugs in Treatment of Common Metabolic Diseases. Oncotarget 2017, 9, 10135–10146. [Google Scholar] [CrossRef]
- Wu, N.; Sarna, L.K.; Siow, Y.L.; O, K. Regulation of Hepatic Cholesterol Biosynthesis by Berberine during Hyperhomocysteinemia. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2011, 300, R635–R643. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yi, X.; Ghanam, K.; Zhang, S.; Zhao, T.; Zhu, X. Berberine Decreases Cholesterol Levels in Rats through Multiple Mechanisms, Including Inhibition of Cholesterol Absorption. Metabolism 2014, 63, 1167–1177. [Google Scholar] [CrossRef] [PubMed]
- Home-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ (accessed on 22 January 2022).
- An, Y.; Sun, Z.; Zhang, Y.; Liu, B.; Guan, Y.; Lu, M. The Use of Berberine for Women with Polycystic Ovary Syndrome Undergoing IVF Treatment. Clin. Endocrinol. 2014, 80, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Ko, B.-S.; Choi, S.B.; Park, S.K.; Jang, J.S.; Kim, Y.E.; Park, S. Insulin Sensitizing and Insulinotropic Action of Berberine from Cortidis Rhizoma. Biol. Pharm. Bull. 2005, 28, 1431–1437. [Google Scholar] [CrossRef]
- Chang, W.; Zhang, M.; Li, J.; Meng, Z.; Wei, S.; Du, H.; Chen, L.; Hatch, G.M. Berberine Improves Insulin Resistance in Cardiomyocytes via Activation of 5′-Adenosine Monophosphate-Activated Protein Kinase. Metabolism 2013, 62, 1159–1167. [Google Scholar] [CrossRef]
- Hu, Y.; Davies, G.E. Berberine Inhibits Adipogenesis in High-Fat Diet-Induced Obesity Mice. Fitoterapia 2010, 81, 358–366. [Google Scholar] [CrossRef]
- Yu, Y.; Liu, L.; Wang, X.; Liu, X.; Liu, X.; Xie, L.; Wang, G. Modulation of Glucagon-like Peptide-1 Release by Berberine: In Vivo and in Vitro Studies. Biochem. Pharmacol. 2010, 79, 1000–1006. [Google Scholar] [CrossRef]
- Pan, G.-Y.; Huang, Z.-J.; Wang, G.-J.; Fawcett, J.P.; Liu, X.-D.; Zhao, X.-C.; Sun, J.-G.; Xie, Y.-Y. The Antihyperglycaemic Activity of Berberine Arises from a Decrease of Glucose Absorption. Planta Med. 2003, 69, 632–636. [Google Scholar] [CrossRef]
- Ye, M.; Fu, S.; Pi, R.; He, F. Neuropharmacological and Pharmacokinetic Properties of Berberine: A Review of Recent Research. J. Pharm. Pharmacol. 2009, 61, 831–837. [Google Scholar] [CrossRef]
- Wei, X.; Zhu, L.; Wang, C. Efficacy and Safety of Berberine in Patients with Type 2 Diabetes Mellitus: A Meta-Analysis. Chin. Herb. Med. 2015, 7, 344–353. [Google Scholar] [CrossRef]
- Li, C.; Guan, X.-M.; Wang, R.-Y.; Xie, Y.-S.; Zhou, H.; Ni, W.-J.; Tang, L.-Q. Berberine Mitigates High Glucose-Induced Podocyte Apoptosis by Modulating Autophagy via the MTOR/P70S6K/4EBP1 Pathway. Life Sci. 2020, 243, 117277. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, J.; Zhang, Y.-J.; Chai, Q.-Y.; Zhang, K.; Ma, H.-L.; Wu, X.-K.; Liu, J.-P. The Effect of Berberine on Insulin Resistance in Women with Polycystic Ovary Syndrome: Detailed Statistical Analysis Plan (SAP) for a Multicenter Randomized Controlled Trial. Trials 2016, 17, 512. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, Y.; Ma, H.; Zhang, Y.; Kuang, H.; Ng, E.H.Y.; Hou, L.; Wu, X. Effect of Berberine on Insulin Resistance in Women with Polycystic Ovary Syndrome: Study Protocol for a Randomized Multicenter Controlled Trial. Trials 2013, 14, 226. [Google Scholar] [CrossRef] [PubMed]
- Dahlberg, C.J.; Ou, J.J.; Babish, J.G.; Lamb, J.J.; Eliason, S.; Brabazon, H.; Gao, W.; Kaadige, M.R.; Tripp, M.L. A 13-Week Low Glycemic Load Diet and Lifestyle Modification Program Combining Low Glycemic Load Protein Shakes and Targeted Nutraceuticals Improved Weight Loss and Cardio-Metabolic Risk Factors. Can. J. Physiol. Pharmacol. 2017, 95, 1414–1425. [Google Scholar] [CrossRef]
- Kumar, R.; Awasthi, M.; Sharma, A.; Padwad, Y.; Sharma, R. Berberine Induces Dose-Dependent Quiescence and Apoptosis in A549 Cancer Cells by Modulating Cell Cyclins and Inflammation Independent of MTOR Pathway. Life Sci. 2020, 244, 117346. [Google Scholar] [CrossRef]
- Bogoyevitch, M.A.; Boehm, I.; Oakley, A.; Ketterman, A.J.; Barr, R.K. Targeting the JNK MAPK Cascade for Inhibition: Basic Science and Therapeutic Potential. Biochim. Biophys. Acta 2004, 1697, 89–101. [Google Scholar] [CrossRef]
- Zheng, F.; Tang, Q.; Wu, J.; Zhao, S.; Liang, Z.; Li, L.; Wu, W.; Hann, S. P38α MAPK-Mediated Induction and Interaction of FOXO3a and P53 Contribute to the Inhibited-Growth and Induced-Apoptosis of Human Lung Adenocarcinoma Cells by Berberine. J. Exp. Clin. Cancer Res. 2014, 33, 36. [Google Scholar] [CrossRef]
- Chen, Q.; Shi, J.; Ding, Z.; Xia, Q.; Zheng, T.; Ren, Y.; Li, M.; Fan, L. Berberine Induces Apoptosis in Non-Small-Cell Lung Cancer Cells by Upregulating MiR-19a Targeting Tissue Factor. Cancer Manag. Res. 2019, 11, 9005–9015. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, M.; Shang, D. Berberine Inhibits Human Gastric Cancer Cell Growth via Deactivation of P38/JNK Pathway, Induction of Mitochondrial-Mediated Apoptosis, Caspase Activation and NF-ΚB Inhibition. J. Balk. Union Oncol. 2020, 25, 314–318. [Google Scholar]
- Li, H.-L.; Wu, H.; Zhang, B.-B.; Shi, H.-L.; Wu, X.-J. MAPK Pathways Are Involved in the Inhibitory Effect of Berberine Hydrochloride on Gastric Cancer MGC 803 Cell Proliferation and IL-8 Secretion in Vitro and in Vivo. Mol. Med. Rep. 2016, 14, 1430–1438. [Google Scholar] [CrossRef]
- Wu, X.-K.; Wang, Y.-Y.; Liu, J.-P.; Hou, L.-H.; Gao, Y.-Q.; Du, S.-M.; Yan, Y.; Zhang, J.-F.; Xue, H.-Y.; Li, W.-L.; et al. Letrozole, Berberine, or a Combination for Infertility in Chinese Women with Polycystic Ovary Syndrome: A Multicentre, Randomised, Double-Blind, Placebo-Controlled Trial. Lancet 2015, 386, S70. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Reggi, A.; Parini, A.; Morbini, M.; Rosticci, M.; Grandi, E.; Borghi, C. Berberine and Monacolin Effects on the Cardiovascular Risk Profile of Women with Oestroprogestin-Induced Hypercholesterolemia. High Blood Press. Cardiovasc. Prev. 2014, 21, 221–226. [Google Scholar] [CrossRef]
- Zhang, S.; Zhou, J.; Gober, H.-J.; Leung, W.T.; Wang, L. Effect and Mechanism of Berberine against Polycystic Ovary Syndrome. Biomed. Pharmacother. 2021, 138, 111468. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.-K.; Wang, Y.-Y.; Liu, J.-P.; Liang, R.-N.; Xue, H.-Y.; Ma, H.-X.; Shao, X.-G.; Ng, E.H.Y.; Hou, L.-H.; Wang, Y.-Y.; et al. Randomized Controlled Trial of Letrozole, Berberine, or a Combination for Infertility in the Polycystic Ovary Syndrome. Fertil. Steril. 2016, 106, 757–765.e1. [Google Scholar] [CrossRef] [PubMed]
- Imanshahidi, M.; Hosseinzadeh, H. Pharmacological and Therapeutic Effects of Berberis Vulgaris and Its Active Constituent, Berberine. Phytother. Res. 2008, 22, 999–1012. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; D’Angelo, A.; Vanelli, A.; Maffioli, P. An Evaluation of a Nutraceutical with Berberine, Curcumin, Inositol, Banaba and Chromium Picolinate in Patients with Fasting Dysglycemia. Diabetes Metab. Syndr. Obes. 2020, 13, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Dandona, P.; Aljada, A.; Bandyopadhyay, A. Inflammation: The Link between Insulin Resistance, Obesity and Diabetes. Trends Immunol. 2004, 25, 4–7. [Google Scholar] [CrossRef]
- Li, X.-Y.; Zhao, Z.-X.; Huang, M.; Feng, R.; He, C.-Y.; Ma, C.; Luo, S.-H.; Fu, J.; Wen, B.-Y.; Ren, L.; et al. Effect of Berberine on Promoting the Excretion of Cholesterol in High-Fat Diet-Induced Hyperlipidemic Hamsters. J. Transl. Med. 2015, 13, 278. [Google Scholar] [CrossRef]
- Wang, Y.; Zidichouski, J.A. Update on the Benefits and Mechanisms of Action of the Bioactive Vegetal Alkaloid Berberine on Lipid Metabolism and Homeostasis. Cholesterol 2018, 2018, 7173920. [Google Scholar] [CrossRef]
- Barrios, V.; Escobar, C.; Cicero, A.F.G.; Burke, D.; Fasching, P.; Banach, M.; Bruckert, E. A Nutraceutical Approach (Armolipid Plus) to Reduce Total and LDL Cholesterol in Individuals with Mild to Moderate Dyslipidemia: Review of the Clinical Evidence. Atheroscler. Suppl. 2017, 24, 1–15. [Google Scholar] [CrossRef]
- Song, B.; Tang, X.; Wang, X.; Huang, X.; Ye, Y.; Lu, X.; Wei, X.; Zeng, Y. Bererine Induces Peripheral Lymphocytes Immune Regulations to Realize Its Neuroprotective Effects in the Cerebral Ischemia/Reperfusion Mice. Cell. Immunol. 2012, 276, 91–100. [Google Scholar] [CrossRef]
- Wang, L.; Peng, L.; Wei, G.; Ge, H. Therapeutic Effects of Berberine Capsule on Patients with Mild Hyperlipidemia. Zhongguo Zhong Xi Yi Jie He Za Zhi 2016, 36, 681–684. [Google Scholar] [PubMed]
- Cereda, E.; Cappello, S.; Caraccia, M.; Turri, A.; Masi, S.; Nappi, R.; Caccialanza, R. SUN-PO004: Nutraceutical Intervention with Berberine, Chlorogenic Acid and Tocotrienols for Menopause-Associated Dyslipidemia: A Pilot, Single-ARM Trial. Clin. Nutr. 2019, 38, S60. [Google Scholar] [CrossRef]
- Yan, H.-M.; Xia, M.-F.; Wang, Y.; Chang, X.-X.; Yao, X.-Z.; Rao, S.-X.; Zeng, M.-S.; Tu, Y.-F.; Feng, R.; Jia, W.-P.; et al. Efficacy of Berberine in Patients with Non-Alcoholic Fatty Liver Disease. PLoS ONE 2015, 10, e0134172. [Google Scholar] [CrossRef]
- Kuno, T.; Hirayama-Kurogi, M.; Ito, S.; Ohtsuki, S. Effect of Intestinal Flora on Protein Expression of Drug-Metabolizing Enzymes and Transporters in the Liver and Kidney of Germ-Free and Antibiotics-Treated Mice. Mol. Pharm. 2016, 13, 2691–2701. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Lin, H.; Huang, W. Modulating Gut Microbiota as an Anti-Diabetic Mechanism of Berberine. Med. Sci. Monit. 2011, 17, RA164–RA167. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Van Hul, M.; Lefort, C.; Depommier, C.; Rastelli, M.; Everard, A. Microbial Regulation of Organismal Energy Homeostasis. Nat. Metab. 2019, 1, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Ning, G. Decade in Review-Type 2 Diabetes Mellitus: At the Centre of Things. Nat. Rev. Endocrinol. 2015, 11, 636–638. [Google Scholar] [CrossRef]
- Manichanh, C.; Rigottier-Gois, L.; Bonnaud, E.; Gloux, K.; Pelletier, E.; Frangeul, L.; Nalin, R.; Jarrin, C.; Chardon, P.; Marteau, P.; et al. Reduced Diversity of Faecal Microbiota in Crohn’s Disease Revealed by a Metagenomic Approach. Gut 2006, 55, 205–211. [Google Scholar] [CrossRef]
- Louis, P.; Hold, G.L.; Flint, H.J. The Gut Microbiota, Bacterial Metabolites and Colorectal Cancer. Nat. Rev. Microbiol. 2014, 12, 661–672. [Google Scholar] [CrossRef]
- Bloomgarden, Z. Diabetes and Branched-Chain Amino Acids: What Is the Link? J. Diabetes. 2018, 10, 350–352. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshini, M.; Wicksteed, B.; Schiltz, G.E.; Gilchrist, A.; Layden, B.T. SCFA Receptors in Pancreatic β Cells: Novel Diabetes Targets? Trends Endocrinol. Metab. 2016, 27, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, E.D.; Sonnenburg, J.L. Starving Our Microbial Self: The Deleterious Consequences of a Diet Deficient in Microbiota-Accessible Carbohydrates. Cell Metab. 2014, 20, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A Core Gut Microbiome in Obese and Lean Twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Manchester, J.K.; Semenkovich, C.F.; Gordon, J.I. Mechanisms Underlying the Resistance to Diet-Induced Obesity in Germ-Free Mice. Proc. Natl. Acad. Sci. USA 2007, 104, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Larsen, N.; Vogensen, F.K.; Van Den Berg, F.W.J.; Nielsen, D.S.; Andreasen, A.S.; Pedersen, B.K.; Al-Soud, W.A.; Sørensen, S.J.; Hansen, L.H.; Jakobsen, M. Gut Microbiota in Human Adults with Type 2 Diabetes Differs from Non-Diabetic Adults. PLoS ONE 2010, 5, e9085. [Google Scholar] [CrossRef]
- Sun, R.; Yang, N.; Kong, B.; Cao, B.; Feng, D.; Yu, X.; Ge, C.; Huang, J.; Shen, J.; Wang, P.; et al. Orally Administered Berberine Modulates Hepatic Lipid Metabolism by Altering Microbial Bile Acid Metabolism and the Intestinal FXR Signaling Pathway. Mol. Pharmacol. 2017, 91, 110–122. [Google Scholar] [CrossRef]
- Xie, W.; Gu, D.; Li, J.; Cui, K.; Zhang, Y. Effects and Action Mechanisms of Berberine and Rhizoma Coptidis on Gut Microbes and Obesity in High-Fat Diet-Fed C57BL/6J Mice. PLoS ONE 2011, 6, e24520. [Google Scholar] [CrossRef]
- Gao, Z.; Li, Q.; Wu, X.; Zhao, X.; Zhao, L.; Tong, X. New Insights into the Mechanisms of Chinese Herbal Products on Diabetes: A Focus on the “Bacteria-Mucosal Immunity-Inflammation-Diabetes” Axis. J. Immunol. Res. 2017, 2017, 1813086. [Google Scholar] [CrossRef]
- Cui, H.-X.; Hu, Y.-N.; Li, J.-W.; Yuan, K.; Guo, Y. Preparation and Evaluation of Antidiabetic Agents of Berberine Organic Acid Salts for Enhancing the Bioavailability. Molecules 2019, 24, 103. [Google Scholar] [CrossRef]
- Habtemariam, S. Berberine Pharmacology and the Gut Microbiota: A Hidden Therapeutic Link. Pharmacol. Res. 2020, 155, 104722. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.-H.; Jeong, I.-H.; Choi, D.-H.; Oh, J.-W.; Ahn, Y.-J. Growth-Inhibiting Effects of Coptis Japonica Root-Derived Isoquinoline Alkaloids on Human Intestinal Bacteria. J. Agric. Food Chem. 1999, 47, 934–938. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, Y.; Huang, W.; Selwyn, F.P.; Klaassen, C.D. Dose-Response Effect of Berberine on Bile Acid Profile and Gut Microbiota in Mice. BMC Complement Altern. Med. 2016, 16, 394. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Cai, J.; Gui, W.; Nichols, R.G.; Koo, I.; Zhang, J.; Anitha, M.; Patterson, A.D. Berberine Directly Affects the Gut Microbiota to Promote Intestinal Farnesoid X Receptor Activation. Drug Metab. Dispos. 2019, 47, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Lynch, C.J.; Adams, S.H. Branched-Chain Amino Acids in Metabolic Signalling and Insulin Resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Yue, S.-J.; Liu, J.; Wang, A.-T.; Meng, X.-T.; Yang, Z.-R.; Peng, C.; Guan, H.-S.; Wang, C.-Y.; Yan, D. Berberine Alleviates Insulin Resistance by Reducing Peripheral Branched-Chain Amino Acids. Am. J. Physiol.-Endocrinol. Metab. 2018, 316, E73–E85. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y.; Zhang, M.; Pang, X.; Xu, J.; Kang, C.; Li, M.; Zhang, C.; Zhang, Z.; Zhang, Y.; et al. Structural Changes of Gut Microbiota during Berberine-Mediated Prevention of Obesity and Insulin Resistance in High-Fat Diet-Fed Rats. PLoS ONE 2012, 7, e42529. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shou, J.-W.; Li, X.-Y.; Zhao, Z.-X.; Fu, J.; He, C.-Y.; Feng, R.; Ma, C.; Wen, B.-Y.; Guo, F.; et al. Berberine-Induced Bioactive Metabolites of the Gut Microbiota Improve Energy Metabolism. Metabolism 2017, 70, 72–84. [Google Scholar] [CrossRef]
- Gu, Y.; Wang, X.; Li, J.; Zhang, Y.; Zhong, H.; Liu, R.; Zhang, D.; Feng, Q.; Xie, X.; Hong, J.; et al. Analyses of Gut Microbiota and Plasma Bile Acids Enable Stratification of Patients for Antidiabetic Treatment. Nat. Commun. 2017, 8, 1785. [Google Scholar] [CrossRef]
- Sun, L.; Xie, C.; Wang, G.; Wu, Y.; Wu, Q.; Wang, X.; Liu, J.; Deng, Y.; Xia, J.; Chen, B.; et al. Gut Microbiota and Intestinal FXR Mediate the Clinical Benefits of Metformin. Nat. Med. 2018, 24, 1919–1929. [Google Scholar] [CrossRef]
- Zhang, Y.; Gu, Y.; Ren, H.; Wang, S.; Zhong, H.; Zhao, X.; Ma, J.; Gu, X.; Xue, Y.; Huang, S.; et al. Gut Microbiome-Related Effects of Berberine and Probiotics on Type 2 Diabetes (the PREMOTE Study). Nat. Commun. 2020, 11, 5015. [Google Scholar] [CrossRef] [PubMed]
- Chiang, J.Y.L. Bile Acids: Regulation of Synthesis. J. Lipid Res. 2009, 50, 1955–1966. [Google Scholar] [CrossRef] [PubMed]
- Maslowski, K.M.; Vieira, A.T.; Ng, A.; Kranich, J.; Sierro, F.; Yu, D.; Schilter, H.C.; Rolph, M.S.; Mackay, F.; Artis, D.; et al. Regulation of Inflammatory Responses by Gut Microbiota and Chemoattractant Receptor GPR43. Nature 2009, 461, 1282–1286. [Google Scholar] [CrossRef]
- Sun, Y.; Jin, C.; Zhang, X.; Jia, W.; Le, J.; Ye, J. Restoration of GLP-1 Secretion by Berberine Is Associated with Protection of Colon Enterocytes from Mitochondrial Overheating in Diet-Induced Obese Mice. Nutr. Diabetes 2018, 8, 53. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhao, Y.; Xu, J.; Xue, Z.; Zhang, M.; Pang, X.; Zhang, X.; Zhao, L. Modulation of Gut Microbiota by Berberine and Metformin during the Treatment of High-Fat Diet-Induced Obesity in Rats. Sci. Rep. 2015, 5, 14405. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, J.-H.; Yu, T.; Chen, Q.-K. Effects of Berberine and Metformin on Intestinal Inflammation and Gut Microbiome Composition in Db/Db Mice. Biomed. Pharmacother. 2019, 118, 109131. [Google Scholar] [CrossRef]
- Vidrine, K.; Ye, J.; Martin, R.J.; McCutcheon, K.L.; Raggio, A.M.; Pelkman, C.; Durham, H.A.; Zhou, J.; Senevirathne, R.N.; Williams, C.; et al. Resistant Starch from High Amylose Maize (HAM-RS2) and Dietary Butyrate Reduce Abdominal Fat by a Different Apparent Mechanism. Obesity 2014, 22, 344–348. [Google Scholar] [CrossRef]
- Qin, J.; Li, Y.; Cai, Z.; Li, S.; Zhu, J.; Zhang, F.; Liang, S.; Zhang, W.; Guan, Y.; Shen, D.; et al. A Metagenome-Wide Association Study of Gut Microbiota in Type 2 Diabetes. Nature 2012, 490, 55–60. [Google Scholar] [CrossRef]
- Reddy, P.; Lent-Schochet, D.; Ramakrishnan, N.; McLaughlin, M.; Jialal, I. Metabolic Syndrome Is an Inflammatory Disorder: A Conspiracy between Adipose Tissue and Phagocytes. Clin. Chim. Acta 2019, 496, 35–44. [Google Scholar] [CrossRef]
- Gurung, P.; Moussa, K.; Adams-Huet, B.; Devaraj, S.; Jialal, I. Increased Mast Cell Abundance in Adipose Tissue of Metabolic Syndrome: Relevance to the Proinflammatory State and Increased Adipose Tissue Fibrosis. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E504–E509. [Google Scholar] [CrossRef]
- Moussa, K.; Gurung, P.; Adams-Huet, B.; Devaraj, S.; Jialal, I. Increased Eosinophils in Adipose Tissue of Metabolic Syndrome. J. Diabetes Complicat. 2019, 33, 535–538. [Google Scholar] [CrossRef] [PubMed]
- Jialal, I.; Devaraj, S. Subcutaneous Adipose Tissue Biology in Metabolic Syndrome. Horm. Mol. Biol. Clin. Investig. 2018, 33. [Google Scholar] [CrossRef] [PubMed]
- Shim, K.; Gulhar, R.; Jialal, I. Exploratory Metabolomics of Nascent Metabolic Syndrome. J. Diabetes Complicat. 2019, 33, 212–216. [Google Scholar] [CrossRef]
- Bremer, A.A.; Jialal, I. Adipose Tissue Dysfunction in Nascent Metabolic Syndrome. J. Obes. 2013, 2013, 393192. [Google Scholar] [CrossRef]
- Ayati, S.H.; Fazeli, B.; Momtazi-borojeni, A.A.; Cicero, A.F.G.; Pirro, M.; Sahebkar, A. Regulatory Effects of Berberine on MicroRNome in Cancer and Other Conditions. Crit. Rev. Oncol. Hematol. 2017, 116, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.-H.; Tang, W.-C.; Cheng, Y.-W.; Sia, P.; Huang, C.-C.; Lee, Y.-C.; Jiang, H.-Y.; Wu, M.-H.; Lai, I.-L.; Lee, J.-W.; et al. Targeting of Multiple Oncogenic Signaling Pathways by Hsp90 Inhibitor Alone or in Combination with Berberine for Treatment of Colorectal Cancer. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2015, 1853, 2261–2272. [Google Scholar] [CrossRef] [PubMed]
- Hamsa, T.P.; Kuttan, G. Antiangiogenic Activity of Berberine Is Mediated through the Downregulation of Hypoxia-Inducible Factor-1, VEGF, and Proinflammatory Mediators. Drug Chem. Toxicol. 2012, 35, 57–70. [Google Scholar] [CrossRef]
- Fukuda, K.; Hibiya, Y.; Mutoh, M.; Koshiji, M.; Akao, S.; Fujiwara, H. Inhibition by Berberine of Cyclooxygenase-2 Transcriptional Activity in Human Colon Cancer Cells. J. Ethnopharmacol. 1999, 66, 227–233. [Google Scholar] [CrossRef]
- Och, A.; Zalewski, D.; Komsta, Ł.; Kołodziej, P.; Kocki, J.; Bogucka-Kocka, A. Cytotoxic and Proapoptotic Activity of Sanguinarine, Berberine, and Extracts of Chelidonium majus L. and Berberis Thunbergii DC. toward Hematopoietic Cancer Cell Lines. Toxins 2019, 11, 485. [Google Scholar] [CrossRef]
- Palmieri, A.; Scapoli, L.; Iapichino, A.; Mercolini, L.; Mandrone, M.; Poli, F.; Giannì, A.B.; Baserga, C.; Martinelli, M. Berberine and Tinospora cordifolia Exert a Potential Anticancer Effect on Colon Cancer Cells by Acting on Specific Pathways. Int. J. Immunopathol. Pharmacol. 2019, 33. [Google Scholar] [CrossRef]
- Liu, D.; Meng, X.; Wu, D.; Qiu, Z.; Luo, H. A Natural Isoquinoline Alkaloid With Antitumor Activity: Studies of the Biological Activities of Berberine. Front. Pharmacol. 2019, 10, 9. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Yang, Y.; Cui, H.; Guan, Z.; Yang, Y.; Zhang, H.; Chen, X.; Zhu, H.; Yang, X.; Cai, J.; et al. The Natural Compound Gambogic Acid Radiosensitizes Nasopharyngeal Carcinoma Cells under Hypoxic Conditions. Tumori 2016, 102, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, X.; Sharvan, R.; Gao, J.; Qu, S. Berberine Could Inhibit Thyroid Carcinoma Cells by Inducing Mitochondrial Apoptosis, G0/G1 Cell Cycle Arrest and Suppressing Migration via PI3K-AKT and MAPK Signaling Pathways. Biomed. Pharmacother. 2017, 95, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Jantová, S.; Čipák, L.; Čerňáková, M.; Košt‘álová, D. Effect of Berberine on Proliferation, Cell Cycle and Apoptosis in HeLa and L1210 Cells. J. Pharm. Pharmacol. 2003, 55, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Zhang, C.; Feng, J.; Hou, L.; Yan, L.; Zhou, Z.; Liu, Z.; Liu, C.; Fan, Y.; Zheng, B.; et al. Induction of G1 Cell Cycle Arrest and Apoptosis by Berberine in Bladder Cancer Cells. Eur. J. Pharmacol. 2011, 661, 1–7. [Google Scholar] [CrossRef]
- Gong, C.; Hu, X.; Xu, Y.; Yang, J.; Zong, L.; Wang, C.; Zhu, J.; Li, Z.; Lu, D. Berberine Inhibits Proliferation and Migration of Colorectal Cancer Cells by Downregulation of GRP78. Anti-Cancer Drugs 2020, 31, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Mu, L.; Cui, Y.; Li, Y.; Chen, P.; Xie, H.; Wang, X. Long Non-coding RNA CASC2 Enhances Berberine-induced Cytotoxicity in Colorectal Cancer Cells by Silencing BCL2. Mol. Med. Rep. 2019, 20, 995–1006. [Google Scholar] [CrossRef]
- Liu, Y.; Hua, W.; Li, Y.; Xian, X.; Zhao, Z.; Liu, C.; Zou, J.; Li, J.; Fang, X.; Zhu, Y. Berberine Suppresses Colon Cancer Cell Proliferation by Inhibiting the SCAP/SREBP-1 Signaling Pathway-Mediated Lipogenesis. Biochem. Pharmacol. 2020, 174, 113776. [Google Scholar] [CrossRef]
- Farooqi, A.A.; Qureshi, M.Z.; Khalid, S.; Attar, R.; Martinelli, C.; Sabitaliyevich, U.Y.; Nurmurzayevich, S.B.; Taverna, S.; Poltronieri, P.; Xu, B. Regulation of Cell Signaling Pathways by Berberine in Different Cancers: Searching for Missing Pieces of an Incomplete Jig-Saw Puzzle for an Effective Cancer Therapy. Cancers 2019, 11, 478. [Google Scholar] [CrossRef]
- Hu, X.; Wu, X.; Huang, Y.; Tong, Q.; Takeda, S.; Qing, Y. Berberine Induces Double-Strand DNA Breaks in Rev3 Deficient Cells. Mol. Med. Rep. 2014, 9, 1883–1888. [Google Scholar] [CrossRef]
- Chidambara Murthy, K.N.; Jayaprakasha, G.K.; Patil, B.S. The Natural Alkaloid Berberine Targets Multiple Pathways to Induce Cell Death in Cultured Human Colon Cancer Cells. Eur. J. Pharmacol. 2012, 688, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Kalaiarasi, A.; Anusha, C.; Sankar, R.; Rajasekaran, S.; John Marshal, J.; Muthusamy, K.; Ravikumar, V. Plant Isoquinoline Alkaloid Berberine Exhibits Chromatin Remodeling by Modulation of Histone Deacetylase To Induce Growth Arrest and Apoptosis in the A549 Cell Line. J. Agric. Food Chem. 2016, 64, 9542–9550. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Kao, S.-T.; Chen, G.-W.; Ho, H.-C.; Chung, J.-G. Apoptosis of Human Leukemia HL-60 Cells and Murine Leukemia WEHI-3 Cells Induced by Berberine through the Activation of Caspase-3. Anticancer Res. 2006, 26, 227–242. [Google Scholar] [PubMed]
- Letašiová, S.; Jantová, S.; Čipák, L.; Múčková, M. Berberine—Antiproliferative Activity in Vitro and Induction of Apoptosis/Necrosis of the U937 and B16 Cells. Cancer Lett. 2006, 239, 254–262. [Google Scholar] [CrossRef]
- Sanna, L.; Marchesi, I.; Melone, M.A.B.; Bagella, L. The Role of Enhancer of Zeste Homolog 2: From Viral Epigenetics to the Carcinogenesis of Hepatocellular Carcinoma. J. Cell. Physiol. 2018, 233, 6508–6517. [Google Scholar] [CrossRef] [PubMed]
- Refaat, A.; Abdelhamed, S.; Yagita, H.; Inoue, H.; Yokoyama, S.; Hayakawa, Y.; Saiki, I. Berberine Enhances Tumor Necrosis Factor-related Apoptosis-inducing Ligand-mediated Apoptosis in Breast Cancer. Oncol. Lett. 2013, 6, 840–844. [Google Scholar] [CrossRef]
- Hsu, W.-H.; Hsieh, Y.-S.; Kuo, H.-C.; Teng, C.-Y.; Huang, H.-I.; Wang, C.-J.; Yang, S.-F.; Liou, Y.-S.; Kuo, W.-H. Berberine Induces Apoptosis in SW620 Human Colonic Carcinoma Cells through Generation of Reactive Oxygen Species and Activation of JNK/P38 MAPK and FasL. Arch. Toxicol. 2007, 81, 719–728. [Google Scholar] [CrossRef]
- Lu, B.; Hu, M.; Liu, K.; Peng, J. Cytotoxicity of Berberine on Human Cervical Carcinoma HeLa Cells through Mitochondria, Death Receptor and MAPK Pathways, and in-Silico Drug-Target Prediction. Toxicology In Vitro 2010, 24, 1482–1490. [Google Scholar] [CrossRef]
- Zarubin, T.; Han, J. Activation and Signaling of the P38 MAP Kinase Pathway. Cell Res. 2005, 15, 11–18. [Google Scholar] [CrossRef]
- Renault, V.M.; Thekkat, P.U.; Hoang, K.L.; White, J.L.; Brady, C.A.; Kenzelmann Broz, D.; Venturelli, O.S.; Johnson, T.M.; Oskoui, P.R.; Xuan, Z.; et al. The Pro-Longevity Gene FoxO3 Is a Direct Target of the P53 Tumor Suppressor. Oncogene 2011, 30, 3207–3221. [Google Scholar] [CrossRef]
- Macleod, K.F.; Sherry, N.; Hannon, G.; Beach, D.; Tokino, T.; Kinzler, K.; Vogelstein, B.; Jacks, T. P53-Dependent and Independent Expression of P21 during Cell Growth, Differentiation, and DNA Damage. Genes Dev. 1995, 9, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Hauck, L.; Harms, C.; Grothe, D.; An, J.; Gertz, K.; Kronenberg, G.; Dietz, R.; Endres, M.; von Harsdorf, R. Critical Role for FoxO3a-Dependent Regulation of P21CIP1/WAF1 in Response to Statin Signaling in Cardiac Myocytes. Circ. Res. 2007, 100, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Fernandez de Mattos, S.; van der Horst, A.; Klompmaker, R.; Kops, G.J.P.L.; Lam, E.W.-F.; Burgering, B.M.T.; Medema, R.H. Cell Cycle Inhibition by FoxO Forkhead Transcription Factors Involves Downregulation of Cyclin D. Mol. Cell. Biol. 2002, 22, 7842–7852. [Google Scholar] [CrossRef] [PubMed]
- Brunet, A.; Bonni, A.; Zigmond, M.J.; Lin, M.Z.; Juo, P.; Hu, L.S.; Anderson, M.J.; Arden, K.C.; Blenis, J.; Greenberg, M.E. Akt Promotes Cell Survival by Phosphorylating and Inhibiting a Forkhead Transcription Factor. Cell 1999, 96, 857–868. [Google Scholar] [CrossRef]
- Yang, J.-Y.; Zong, C.S.; Xia, W.; Yamaguchi, H.; Ding, Q.; Xie, X.; Lang, J.-Y.; Lai, C.-C.; Chang, C.-J.; Huang, W.-C.; et al. ERK Promotes Tumorigenesis by Inhibiting FOXO3a via MDM2-Mediated Degradation. Nat. Cell Biol. 2008, 10, 138–148. [Google Scholar] [CrossRef]
- Gazon, H.; Barbeau, B.; Mesnard, J.-M.; Peloponese, J.-M.J. Hijacking of the AP-1 Signaling Pathway during Development of ATL. Front. Microbiol. 2018, 8, 2686. [Google Scholar] [CrossRef]
- Abate, C.; Curran, T. Encounters with Fos and Jun on the Road to AP-1. Semin. Cancer Biol. 1990, 1, 19–26. [Google Scholar]
- Neyns, B.; Vermeij, J.; Bourgain, C.; Vandamme, B.; Amfo, K.; Lissens, W.; DeSutter, P.; Hooghe-Peters, E.; DeGrève, J. Expression of the Jun Family of Genes in Human Ovarian Cancer and Normal Ovarian Surface Epithelium. Oncogene 1996, 12, 1247–1257. [Google Scholar]
- Langer, S.; Singer, C.F.; Hudelist, G.; Dampier, B.; Kaserer, K.; Vinatzer, U.; Pehamberger, H.; Zielinski, C.; Kubista, E.; Schreibner, M. Jun and Fos Family Protein Expression in Human Breast Cancer: Correlation of Protein Expression and Clinicopathological Parameters. Eur. J. Gynaecol. Oncol. 2006, 27, 345–352. [Google Scholar]
- Kharman-Biz, A.; Gao, H.; Ghiasvand, R.; Zhao, C.; Zendehdel, K.; Dahlman-Wright, K. Expression of Activator Protein-1 (AP-1) Family Members in Breast Cancer. BMC Cancer 2013, 13, 441. [Google Scholar] [CrossRef]
- Drakos, E.; Leventaki, V.; Schlette, E.J.; Jones, D.; Lin, P.; Jeffrey Medeiros, L.; Rassidakis, G.Z. C-Jun Expression and Activation Are Restricted to CD30+ Lymphoproliferative Disorders. Am. J. Surg. Pathol. 2007, 31, 447–453. [Google Scholar] [CrossRef]
- Mao, X.; Orchard, G.; Russell-Jones, R.; Whittaker, S. Abnormal Activator Protein 1 Transcription Factor Expression in CD30-Positive Cutaneous Large-Cell Lymphomas. Br. J. Dermatol. 2007, 157, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Tulchinsky, E. Fos Family Members: Regulation, Structure and Role in Oncogenic Transformation. Histol. Histopathol. 2000. [Google Scholar] [CrossRef]
- Lin, J.G.; Chung, J.G.; Wu, L.T.; Chen, G.W.; Chang, H.L.; Wang, T.F. Effects of Berberine on Arylamine N-Acetyltransferase Activity in Human Colon Tumor Cells. Am. J. Chin. Med. 1999, 27, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Mitani, N.; Murakami, K.; Yamaura, T.; Ikeda, T.; Saiki, I. Inhibitory Effect of Berberine on the Mediastinal Lymph Node Metastasis Produced by Orthotopic Implantation of Lewis Lung Carcinoma. Cancer Lett. 2001, 165, 35–42. [Google Scholar] [CrossRef]
- Yu, F.-S.; Yang, J.-S.; Lin, H.-J.; Yu, C.-S.; Tan, T.-W.; Lin, Y.-T.; Lin, C.-C.; Lu, H.-F.; Chung, J.-G. Berberine Inhibits WEHI-3 Leukemia Cells In Vivo. In Vivo 2007, 21, 407–412. [Google Scholar]
- Li, L.; Peng, Z.; Hu, Q.; Xu, L.; Zou, X.; Yu, Y.; Huang, D.; Yi, P. Berberine Suppressed Tumor Growth through Regulating Fatty Acid Metabolism and Triggering Cell Apoptosis via Targeting FABPs. Evid.-Based Complementary Altern. Med. 2020, 2020, 6195050. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, W.; Tong, Y. Berberine Inhibits Proliferative Ability of Breast Cancer Cells by Reducing Metadherin. Med. Sci. Monit. 2019, 25, 9058–9066. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, C. Berberine Inhibits MDA-MB-231 Cells by Attenuating Their Inflammatory Responses. Biomed. Res. Int. 2020, 2020, 3617514. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, H.; Zhang, C.; Cheng, Y.; Hu, L.; Meng, X.; Zhao, Y. Protective Effects of Berberine on Radiation-Induced Lung Injury via Intercellular Adhesion Molecular-1 and Transforming Growth Factor-Beta-1 in Patients with Lung Cancer. Eur. J. Cancer 2008, 44, 2425–2432. [Google Scholar] [CrossRef]
- Yount, G.; Qian, Y.; Moore, D.; Basila, D.; West, J.; Aldape, K.; Arvold, N.; Shalev, N.; Haas-Kogan, D. Berberine Sensitizes Human Glioma Cells, but Not Normal Glial Cells, to Ionizing Radiation in Vitro. J. Exp. Ther. Oncol. 2004, 4, 137–143. [Google Scholar] [PubMed]
- Ohaeri, J.U.; Akanji, A.O. Metabolic Syndrome in Severe Mental Disorders. Metab. Syndr. Relat. Disord. 2011, 9, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Lyra e Silva, N.D.M.; Lam, M.P.; Soares, C.N.; Munoz, D.P.; Milev, R.; De Felice, F.G. Insulin Resistance as a Shared Pathogenic Mechanism Between Depression and Type 2 Diabetes. Front. Psychiatry 2019, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Penninx, B.W.J.H.; Lange, S.M.M. Metabolic Syndrome in Psychiatric Patients: Overview, Mechanisms, and Implications. Dialogues Clin. Neurosci. 2018, 20, 63–73. [Google Scholar]
- Salvi, V.; Hajek, T. Editorial: Brain-Metabolic Crossroads in Severe Mental Disorders—Focus on Metabolic Syndrome. Front. Psychiatry 2019, 10, 492. [Google Scholar] [CrossRef] [PubMed]
- Bhuvaneswar, C.G.; Baldessarini, R.J.; Harsh, V.L.; Alpert, J.E. Adverse Endocrine and Metabolic Effects of Psychotropic Drugs. CNS Drugs 2009, 23, 1003–1021. [Google Scholar] [CrossRef]
- Kane, J.M. Pharmacologic Treatment of Schizophrenia. Biol. Psychiatry 1999, 46, 1396–1408. [Google Scholar] [CrossRef]
- Bressington, D.T.; Mui, J.; Cheung, E.F.C.; Petch, J.; Clark, A.B.; Gray, R. The Prevalence of Metabolic Syndrome amongst Patients with Severe Mental Illness in the Community in Hong Kong—A Cross Sectional Study. BMC Psychiatry 2013, 13, 87. [Google Scholar] [CrossRef]
- Pirillo, A.; Catapano, A.L. Berberine, a Plant Alkaloid with Lipid- and Glucose-Lowering Properties: From in Vitro Evidence to Clinical Studies. Atherosclerosis 2015, 243, 449–461. [Google Scholar] [CrossRef]
- Lan, J.; Zhao, Y.; Dong, F.; Yan, Z.; Zheng, W.; Fan, J.; Sun, G. Meta-Analysis of the Effect and Safety of Berberine in the Treatment of Type 2 Diabetes Mellitus, Hyperlipemia and Hypertension. J. Ethnopharmacol. 2015, 161, 69–81. [Google Scholar] [CrossRef]
- Hu, Y.; Young, A.J.; Ehli, E.A.; Nowotny, D.; Davies, P.S.; Droke, E.A.; Soundy, T.J.; Davies, G.E. Metformin and Berberine Prevent Olanzapine-Induced Weight Gain in Rats. PLoS ONE 2014, 9, e93310. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.-H.; Wu, C.-R.; Chen, C.-S.; Chen, C.-F.; Leu, Z.-C.; Hsieh, M.-T. Anxiolytic Effect of Berberine on Exploratory Activity of the Mouse in Two Experimental Anxiety Models: Interaction with Drugs Acting at 5-HT Receptors. Life Sci. 2004, 75, 2451–2462. [Google Scholar] [CrossRef] [PubMed]
- Kawano, M.; Takagi, R.; Kaneko, A.; Matsushita, S. Berberine Is a Dopamine D1- and D2-like Receptor Antagonist and Ameliorates Experimentally Induced Colitis by Suppressing Innate and Adaptive Immune Responses. J. Neuroimmunol. 2015, 289, 43–55. [Google Scholar] [CrossRef]
- Salehi, S.; Filtz, T.M. Berberine Possesses Muscarinic Agonist-like Properties in Cultured Rodent Cardiomyocytes. Pharmacol. Res. 2011, 63, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-H.; Cai, M.; Wang, H.-N.; Chen, Y.-C.; Zhang, R.-G.; Wang, Y.; McAlonan, G.M.; Bai, Y.-H.; Wu, W.-J.; Guo, L.; et al. An Assessor-Blinded, Randomized Comparison of Efficacy and Tolerability of Switching from Olanzapine to Ziprasidone and the Combination of Both in Schizophrenia Spectrum Disorders. J. Psychiatr. Res. 2017, 85, 59–65. [Google Scholar] [CrossRef]
- Haffner, S.M.; Miettinen, H.; Stern, M.P. The Homeostasis Model in the San Antonio Heart Study. Diabetes Care 1997, 20, 1087–1092. [Google Scholar] [CrossRef]
- Gharabawi, G.M.; Bossie, C.A.; Lasser, R.A.; Turkoz, I.; Rodriguez, S.; Chouinard, G. Abnormal Involuntary Movement Scale (AIMS) and Extrapyramidal Symptom Rating Scale (ESRS): Cross-Scale Comparison in Assessing Tardive Dyskinesia. Schizophr. Res. 2005, 77, 119–128. [Google Scholar] [CrossRef]
- Kysenius, K.; Brunello, C.A.; Huttunen, H.J. Mitochondria and NMDA Receptor-Dependent Toxicity of Berberine Sensitizes Neurons to Glutamate and Rotenone Injury. PLoS ONE 2014, 9, e107129. [Google Scholar] [CrossRef]
- Kulkarni, S.K.; Dhir, A. On the Mechanism of Antidepressant-like Action of Berberine Chloride. Eur. J. Pharmacol. 2008, 589, 163–172. [Google Scholar] [CrossRef]
- Lee, B.; Sur, B.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.-H. Effect of Berberine on Depression- and Anxiety-like Behaviors and Activation of the Noradrenergic System Induced by Development of Morphine Dependence in Rats. Korean J. Physiol. Pharmacol. 2012, 16, 379–386. [Google Scholar] [CrossRef]
- Kong, L.D.; Cheng, C.H.; Tan, R.X. Monoamine Oxidase Inhibitors from Rhizoma of Coptis Chinensis. Planta Med. 2001, 67, 74–76. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.-H.; Lo, K.-L.; Lee, Y.-H.; Hung, T.-H.; Lin, Y.-C. Berberine Produces Antidepressant-like Effects in the Forced Swim Test and in the Tail Suspension Test in Mice. Life Sci. 2007, 81, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Zhang, K.; Jin, Y.; Li, B.; Gao, S.; Zhu, J.; Cui, R. Pharmacological Effects of Berberine on Mood Disorders. J. Cell. Mol. Med. 2019, 23, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Zhang, N. Berberine: Pathways to Protect Neurons. Phytother. Res. 2018, 32, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Sharma, B. Toxicological Effects of Berberine and Sanguinarine. Front. Mol. Biosci. 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.; Gilani, A.-H.; Abdollahi, M.; Daglia, M.; Nabavi, S.F.; Nabavi, S.M. Berberine and Neurodegeneration: A Review of Literature. Pharmacol. Rep. 2015, 67, 970–979. [Google Scholar] [CrossRef]
- Kulkarni, S.K.; Dhir, A. Berberine: A Plant Alkaloid with Therapeutic Potential for Central Nervous System Disorders. Phytother. Res. 2010, 24, 317–324. [Google Scholar] [CrossRef]
- Imenshahidi, M.; Hosseinzadeh, H. Berberis Vulgaris and Berberine: An Update Review. Phytother. Res. 2016, 30, 1745–1764. [Google Scholar] [CrossRef]
- Lee, B.; Yang, C.H.; Hahm, D.-H.; Choe, E.S.; Lee, H.-J.; Pyun, K.-H.; Shim, I. Inhibitory Effects of Coptidis Rhizoma and Berberine on Cocaine-Induced Sensitization. Evid.-Based Complementary Altern. Med. 2009, 6, 85–90. [Google Scholar] [CrossRef]
| Title | Conditions | Phase | Subjects | Duration | Interventions Given | Measures | Results in Berberine or Nutraceutical Combination Groups |
|---|---|---|---|---|---|---|---|
| A Mechanistic Randomized Controlled Trial on the Cardiovascular Effect of Berberine | Cardiovascular risk factor | Phase 2 Phase 3 | 84 men of Chinese ethnicity aged 20 to 65 years with hyperlipidemia, not currently receiving hormone replacement therapy such as testosterone replacement therapy in the past 12 months; not currently taking berberine or traditional Chinese medicine that contains berberine in the past 1 month; free of congenital diseases, infectious diseases, anemia, and glucose-6-phosphate dehydrogenase deficiency; and no history of any chronic diseases. including ischemic heart disease, myocardial infarction (heart attack), stroke, diabetes, cancer, liver/renal dysfunction, and gastrointestinal disorders | 12 weeks | Berberine verus placebo |
|
|
| Long-term efficacy and tolerability of a nutraceutical combination (red yeast rice, policosanols, and berberine (MBP-NC)) in patients with low-moderate risk of hypercholesterolemia: a double-blind, placebo-controlled Study of the | Hypercholesterolemia | Phase 4 | 60 adults between 18 and 60 years with newly diagnosed primary hypercholesterolemia, not previously treated, after a run-in period of 3 weeks on a stable hypolipidic diet, with a body mass index between 18,5 ad 29,9 Kg/m2, serum low-density lipoprotein cholesterol above 150 mg/dL, and an estimated 10-year cardiovascular risk of <20% according to the Framingham risk scoring | Assessement after 4, 12 and 24 weeks of treatment. | Nutraceutical combination of red yeast rice extract (monacolins), berberine, and policosanols after dinner, in addition to the hypolipidic diet versus placebo |
|
|
| effects of Armolipid Plus on cholesterol levels and endothelial function (mixture of berberine, policosanol, and red yeast) | Hyperlipidemia Endothelial dysfunction | Not Applicable | 50 adults aged between 18 and 70 with total cholesterol levels > 220 mg/dL; LDL-cholesterol > 130 mg/dL; and with concomitant pathology, such as diabetes, chronic heart failure, coronary artery disease, arterial hypertension, and dysthyroidism, if stable in the previous three months | 6 weeks | Mixture of berberine, policosanol, red yeast versus placebo |
|
|
| Nutraceutical combination in patients with low-grade systemic inflammation (berberine 200 mg, monacolin K 3 mg, chitosan 10 mg, and coenzyme Q 10 mg) | Atherosclerosis inflammation Hypercholesterolemia | Phase 4 | 100 adults aged 25 to 75 with suboptimal LDL cholesterol levels (LDL 100–160 mg/dL) and hsCRP levels of >2 mg/L, randomized after 30 days of a low-cholesterol diet | 3 months | Nutraceutical combination: red yeast rice extract (monacolins), policosanol, berberine, folic acid, coenzyme Q10, and astaxanthin with a low-cholesterol/low-saturated fat diet and a regular aerobic physical activity schedule versus a low-cholesterol/low-saturated fat diet and a regular aerobic physical activity schedule + placebo |
|
|
| The efficacy and tolerability of coleosoma nutraceutical formulations in dyslipidemic subjects (fermented red rice, berberine, and chitosan) | Dyslipidemias | Phase 2 | 39 adults aged 18 to 75 with non-HDL cholesterol ≥ 160 mg/dL | 12 weeks | Coleosoma-patented dietary supplement composed of berberin, fermented red rice from monascus purpureus (monacolin K), chitosan, and coenzyme Q10 |
|
|
| Combined effects of bioactive compounds on the lipid profile (red yeast rice and policosanol composed of berberine, folic acid, and coenzyme Q10 (Armolipid Plus ®, Rottapharm)) | Hyperlipidemia Low-density-lipoprotein-type Elevated triglycerides | Phase 2 Phase 3 | 118 adults with LDL-C plasma levels ≥ 130 mg/dL and ≤189 mg/dL that did not require lipid-lowering drug treatment according to the ATPIII guidelines, as well as adults that did not have cardiovascular disease, stroke, intermittent claudication, diabetes mellitus, renal issues, or effects/contraindications to lipid-lowering drug therapy | 12 weeks | Armolipid Plus (red yeast, astaxanthin, berberine, policosanol, coenzyme Q10, and folic acid) versus placebo |
|
|
| Effects of nutraceutical therapies on endothelial function, platelet accumulation, and coronary flow reserve | Hypercholesterolemia Endothelial dysfunction | Not Applicable | Adults aged between 18 and 70 years with hypercholesterolemia that did not require statins or were statin-intolerant | 8 weeks | Combination A (Armolipid Plus): policosanol, red yeast rice (monacolin K), berberine, astaxantine, folic acid and coenzyme Q10 or combination B: berberine, red yeast rice powder (monacolin K), and leaf extract of Morus alba |
|
|
| Study of berberine hydrochloride in the prevention of colorectal adenomas recurrence | Colorectal adenoma | Phase 2 | 1108 adults sged 18–75 who had at least one, and no more than 6, histologically confirmed colorectal adenomas that were removed within 6 months before recruitment, whose adenoma was not completely removed during a previous colonoscopy; a history of familial adenomatous polyposis or hereditary non-polyposis colorectal cancer (HNPCC, Lynch syndrome); or a history of subtotal/total gastrectomy or partial bowel resection | 3 years | Berberine hydrochloride versus placebo |
|
|
| Berberine effects on clinical symptoms and metabolic disturbance in patients with schizophrenia | Schizophrenia | Phase 4 | 65 adults aged 18 to 65 years who met the diagnosis of schizophrenia according to the DSM-IV, and have undergone monotherapy of atypical antipsychotics for 4 weeks or more, with at least 60 for positive and negative syndrome scale | 8 weeks | Berberine plus any atypical antipsychotic drug as the basic treatment |
|
|
| Berberine treat metabolic syndrome in schizophrenia | Metabolic syndrome Schizophrenia | Not Applicable | Adult females aged 18–60 with a diagnosis of schizophrenia, undergoing monotherapy of atypical antipsychotics for 2 weeks or more, including olanzapine, clozapine, risperidone, and perphenazine, with a diagnosed metabolic syndrome depending on the guidelines for the prevention and treatment of dyslipidemia in Chinese adults in 2007 | 8 weeks | Berberine in adjunctive group |
|
|
| Berberine hyperglycemic clamp | Diabetes mellitus | Phase 1 | 15 adult healthy males, aged 18–45 with BMI 18–25 kg/m2 and a normal oral glucose tolerance test prior to the study, with no family history of diabetes mellitus, and with no medication treatment within 4 weeks prior to the baseline visit, as well as during the study | 2 weeks | Berberine versus placebo |
|
|
| Efficacy and safety of berberine in the treatment of diabetes with dyslipidemia | Type 2 diabetes mellitus Metabolic syndrome | Phase 3 | 120 adults aged 25–70 with newly diagnosed type 2 diabetes, according to the 1999 World Health Organization criteria, with dyslipidemia with a TG of > 150 mg/dL (1.70 mmol/L), and/or TC > 200 mg/dL (5.16 mmol/L), and/or LDL-C > 100 mg/dL (2.58 mmol/L), according to the National Cholesterol Education Program’s Adult Treatment Panel III (NCEP: ATPIII) without previous treatment and with BMI 19–40 kg/m2 | 3 months | Berberine versus placebo |
|
|
| Therapeutic effects of berberine in patients with type 2 diabetes | Type 2 diabetes | Phase 1 Phase 2 | 70 adults aged 25 to 75 with a clinical diagnosis of type 2 diabetes with HbA1c > 7.0% or FBG > 7.0 mmol/L with stable or worsening glycemic control for at least 3 months | 13 weeks | Berberine versus metformin |
|
|
| Title | Conditions | Status | Phase | Measures |
|---|---|---|---|---|
| A Research of berberine hydrochloride to prevent colorectal adenomas in patients with previous colorectal cancer | Colorectal adenomas | Recruiting | Phase 2 Phase 3 |
|
| Comparison of berberine and metformin for the treatment of MS in schizophrenia patients | Schizophrenia Metabolic syndrome | Recruiting | Phase 4 |
|
| Effect of berberine versus metformin on glycemic control, insulin sensitivity, and insulin secretion in prediabetes | Prediabetes impaired Fasting glucose impaired Glucose tolerance | Active, not recruiting | Phase 4 |
|
| Effect of berberine for endothelial function and intestinal microflora in patients with coronary artery disease | Stable coronaryartery diseasePercutaneous coronary intervention | Active, not recruiting | Phase 1 Phase 2 |
|
| Berberine prevents contrast-induced nephropathy in patients with diabetes | Diabetes mellitus Chronic kidney disease | Recruiting | Phase 4 |
|
| Berberine as adjuvant treatment for schizophrenia patients | Schizophrenia Schizophrenia spectrum and other psychotic disorders Metabolic syndrome X | Recruiting | Phase 2 Phase 3 |
|
| Evaluating the tolerability and effects of berberine on major metabolic biomarkers: a pilot study | Metabolic syndrome | Recruiting | Not Applicable |
|
| Efficacy and safety of berberine in non-alcoholic steatohepatitis | Non-alcoholic steatohepatitis | Recruiting | Phase 4 |
|
| Study on the efficacy and gut microbiota of berberine and probiotics in patients with newly diagnosed type 2 diabetes | Type 2 diabetes | Active, not recruiting | Phase 3 |
|
| Effect of berberine hydrochloride on blood pressure and vascular endothelial function in patients With hypertension | Hypertension Endothelial dysfunction Blood pressure | Recruting | Phase 4 |
|
| Effect of berberine on metabolic syndrome, efficacy and safety in combination with antiretroviral therapy in PLWH | Metabolic syndrome HIV-1-infection Glucose intolerance | Not yet recruiting | Phase 3 |
|
| Berberine prevents contrast-induced nephropathy in patients with diabetes | Diabetes mellitus Chronic kidney disease | Recruiting | Phase 4 |
|
| Evaluating the tolerability and effects of berberine on major metabolic biomarkers: a pilot study | Metabolic syndrome | Recruiting | Not Applicable |
|
| Efficacy and safety of berberine in non-alcoholic steatohepatitis | Non-alcoholic steatohepatitis | Recruiting | Phase 4 |
|
| Study to determine the effect of synbiotics in patients with pre-diabetes | Pre-diabetes | Recruiting | Not Applicable |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Och, A.; Och, M.; Nowak, R.; Podgórska, D.; Podgórski, R. Berberine, a Herbal Metabolite in the Metabolic Syndrome: The Risk Factors, Course, and Consequences of the Disease. Molecules 2022, 27, 1351. https://doi.org/10.3390/molecules27041351
Och A, Och M, Nowak R, Podgórska D, Podgórski R. Berberine, a Herbal Metabolite in the Metabolic Syndrome: The Risk Factors, Course, and Consequences of the Disease. Molecules. 2022; 27(4):1351. https://doi.org/10.3390/molecules27041351
Chicago/Turabian StyleOch, Anna, Marek Och, Renata Nowak, Dominika Podgórska, and Rafał Podgórski. 2022. "Berberine, a Herbal Metabolite in the Metabolic Syndrome: The Risk Factors, Course, and Consequences of the Disease" Molecules 27, no. 4: 1351. https://doi.org/10.3390/molecules27041351
APA StyleOch, A., Och, M., Nowak, R., Podgórska, D., & Podgórski, R. (2022). Berberine, a Herbal Metabolite in the Metabolic Syndrome: The Risk Factors, Course, and Consequences of the Disease. Molecules, 27(4), 1351. https://doi.org/10.3390/molecules27041351






