In Silico Analysis of Metabolites from Peruvian Native Plants as Potential Therapeutics against Alzheimer’s Disease
Abstract
:1. Introduction
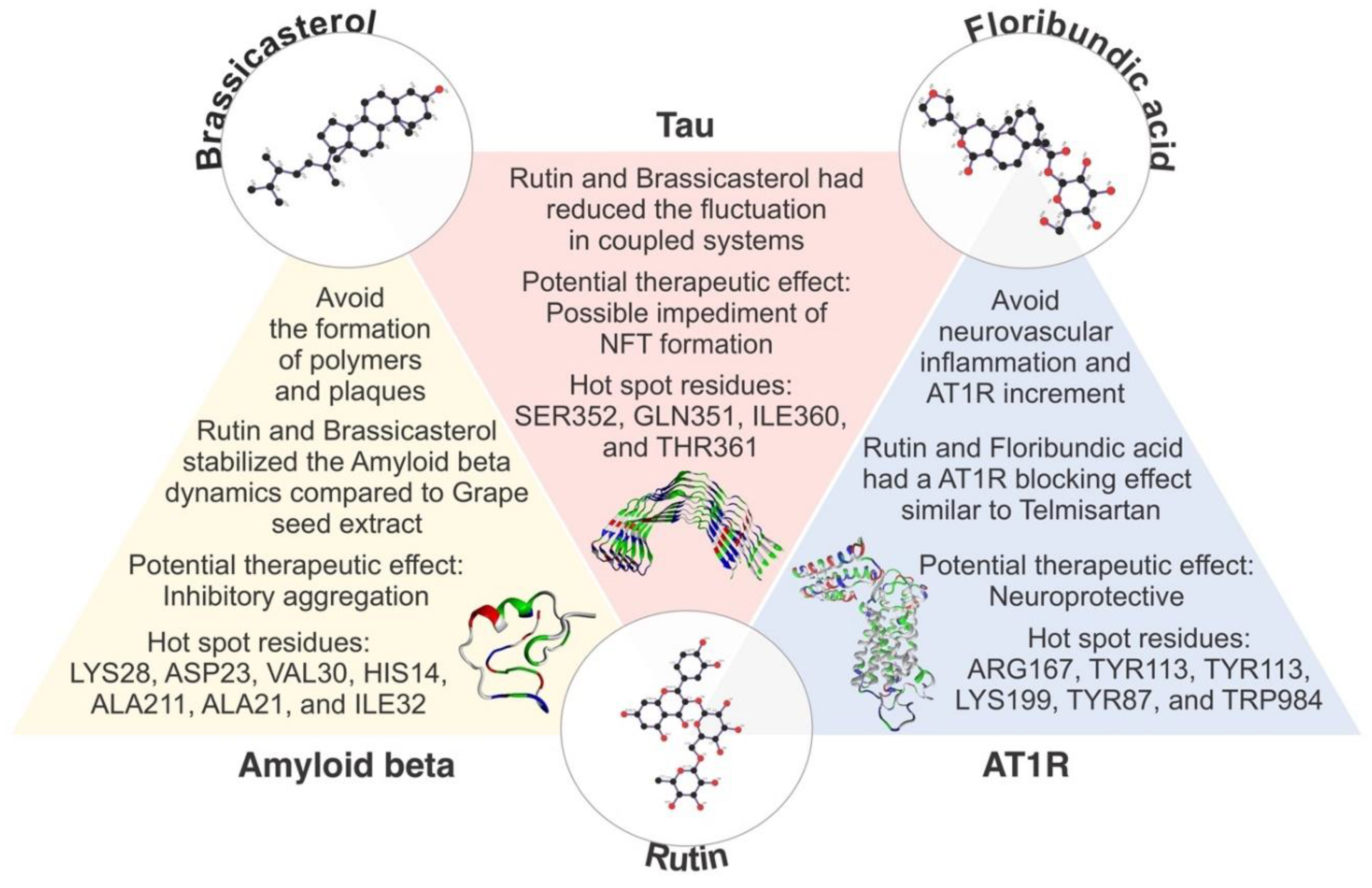
2. Results
2.1. Literature Research
2.2. Docking Procedure and Virtual Screening
2.3. ADME/TOX Analysis
2.4. Molecular Dynamics Simulations and Molecular Mechanics Generalized Born Surface Area Calculations
3. Discussion
4. Materials and Methods
4.1. Literature Search Strategy and Data Collection
4.2. Docking Procedure and Virtual Screening
4.3. ADME/TOX
4.4. Ligand Preparation
4.5. Molecular Dynamics Simulations and Molecular Mechanics Generalized Born Surface Area Calculations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amin, F.; Shamsi, A.; Asghar, M.N.; Khaki, P.S.S.; Khan, M.S.; Tabrez, S.; Zaidi, S.K.; Khan, W.; Bano, B. Alzheimer’s: A Progressive Brain Disease: Causes, Symptoms, and Prevention. In Biological, Diagnostic and Therapeutic Advances in Alzheimer’s Disease; Springer: Berlin/Heidelberg, Germany, 2019; pp. 31–51. [Google Scholar]
- Graham, W.V.; Bonito-Oliva, A.; Sakmar, T.P. Update on Alzheimer’s disease therapy and prevention strategies. Annu. Rev. Med. 2017, 68, 413–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabinovici, G.D. Late-onset Alzheimer disease. Contin. Lifelong Learn. Neurol. 2019, 25, 14. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T. Neuropathological alterations in Alzheimer disease. Cold Spring Harb. Perspect. Med. 2011, 1, a006189. [Google Scholar] [CrossRef] [PubMed]
- Taipa, R.; Ferreira, V.; Brochado, P.; Robinson, A.; Reis, I.; Marques, F.; Mann, D.M.; Melo-Pires, M.; Sousa, N. Inflammatory pathology markers (activated microglia and reactive astrocytes) in early and late onset Alzheimer disease: A post mortem study. Neuropathol. Appl. Neurobiol. 2018, 44, 298–313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aguilar-Pineda, J.A.; Vera-Lopez, K.J.; Shrivastava, P.; Chávez-Fumagalli, M.A.; Nieto-Montesinos, R.; Alvarez-Fernandez, K.L.; Mamani, L.D.G.; Del-Carpio, G.D.; Gomez-Valdez, B.; Miller, C.L.; et al. Vascular smooth muscle cell dysfunction contribute to neuroinflammation and Tau hyperphosphorylation in Alzheimer disease. Iscience 2021, 24, 102993. [Google Scholar] [CrossRef]
- Uemura, M.T.; Maki, T.; Ihara, M.; Lee, V.M.Y.; Trojanowski, J.Q. Brain microvascular pericytes in vascular cognitive impairment and dementia. Front. Aging Neurosci. 2020, 12, 80. [Google Scholar] [CrossRef] [Green Version]
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B. V Blood–brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 2018, 14, 133–150. [Google Scholar] [CrossRef]
- Levit, A.; Hachinski, V.; Whitehead, S.N. Neurovascular unit dysregulation, white matter disease, and executive dysfunction: The shared triad of vascular cognitive impairment and Alzheimer disease. Geroscience 2020, 42, 445–465. [Google Scholar] [CrossRef]
- Iturria-Medina, Y.; Sotero, R.C.; Toussaint, P.J.; Mateos-Pérez, J.M.; Evans, A.C. Early role of vascular dysregulation on late-onset Alzheimer’s disease based on multifactorial data-driven analysis. Nat. Commun. 2016, 7, 11934. [Google Scholar] [CrossRef]
- Iadecola, C. The pathobiology of vascular dementia. Neuron 2013, 80, 844–866. [Google Scholar] [CrossRef] [Green Version]
- Oscanoa, T.J.; Amado, J.; Vidal, X.; Romero-Ortuno, R. Angiotensin-receptor blockers (ARBs) and risk of Alzheimer’s disease: A systematic review and metaanalysis. J. Neurol. Neurophysiol. 2019, 10, 50–51. [Google Scholar]
- Cosarderelioglu, C.; Nidadavolu, L.S.; George, C.J.; Oh, E.S.; Bennett, D.A.; Walston, J.D.; Abadir, P.M. Brain Renin–Angiotensin System at the Intersect of Physical and Cognitive Frailty. Front. Neurosci. 2020, 14, 586314. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.A.; Ishrat, T.; Pillai, B.; Bunting, K.M.; Vazdarjanova, A.; Waller, J.L.; Ergul, A.; Fagan, S.C. Angiotensin receptor (AT2R) agonist C21 prevents cognitive decline after permanent stroke in aged animals—A randomized double-blind pre-clinical study. Behav. Brain Res. 2019, 359, 560–569. [Google Scholar] [CrossRef] [PubMed]
- McKinley, M.J.; Albiston, A.L.; Allen, A.M.; Mathai, M.L.; May, C.N.; McAllen, R.M.; Oldfield, B.J.; Mendelsohn, F.A.O.; Chai, S.Y. The brain renin–angiotensin system: Location and physiological roles. Int. J. Biochem. Cell Biol. 2003, 35, 901–918. [Google Scholar] [CrossRef]
- Amouyel, P.; Richard, F.; Berr, C.; David-Fromentin, I.; Helbecque, N. The renin angiotensin system and Alzheimer’s disease. Ann. N. Y. Acad. Sci. 2000, 903, 437–441. [Google Scholar] [CrossRef]
- Wright, J.W.; Harding, J.W. Brain renin-angiotensin—a new look at an old system. Prog. Neurobiol. 2011, 95, 49–67. [Google Scholar] [CrossRef]
- Lange-Jacobs, D.; Shaikh-Kader, A.; Thomas, B.; Nyakudya, T.T. An Overview of the Potential Use of Ethno-Medicinal Plants Targeting the Renin–Angiotensin System in the Treatment of Hypertension. Molecules 2020, 25, 2114. [Google Scholar] [CrossRef]
- Verma, R.; Hanif, K.; Sasmal, D.; Raghubir, R. Resurgence of herbal antihypertensives in management of hypertension. Curr. Hypertens. Rev. 2010, 6, 190–198. [Google Scholar] [CrossRef]
- Caballero-George, C.; Vanderheyden, P.M.L.; De Bruyne, T.; Shahat, A.-A.; Van den Heuvel, H.; Solis, P.N.; Gupta, M.P.; Claeys, M.; Pieters, L.; Vauquelin, G. In vitro inhibition of [3H]-angiotensin II binding on the human AT1 receptor by proanthocyanidins from Guazuma ulmifolia bark. Planta Med. 2002, 68, 1066–1071. [Google Scholar] [CrossRef]
- Wang, Y.; Li, C.; Ouyang, Y.; Yu, J.; Guo, S.; Liu, Z.; Li, D.; Han, J.; Wang, W. Cardioprotective effects of Qishenyiqi mediated by angiotensin II type 1 receptor blockade and enhancing angiotensin-converting enzyme 2. Evid. Based Complement. Altern. Med. 2012, 2012, 978127. [Google Scholar] [CrossRef]
- Ren, S.; Zhang, H.; Mu, Y.; Sun, M.; Liu, P. Pharmacological effects of Astragaloside IV: A literature review. J. Tradit. Chinese Med. 2013, 33, 413–416. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, Y.M.; Yu, G.Y. Effects of gastrodin injection on blood pressure and vasoactive substances in treatment of old patients with refractory hypertension: A randomized controlled trial. J. Chin. Integr. Med. 2008, 6, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, L.; Yu, J.; Asare, P.F.; Zhao, Y.-Q. Gastrodin reduces blood pressure by intervening with RAAS and PPARγ in SHRs. Evid. Based Complement. Altern. Med. 2015, 2015, 828427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, B.-R.; Seo, H.-S.; Ku, J.-M.; Kim, G.-J.; Jeon, C.Y.; Park, J.H.; Jang, B.-H.; Park, S.-J.; Shin, Y.-C.; Ko, S.-G. Silibinin inhibits the production of pro-inflammatory cytokines through inhibition of NF-κB signaling pathway in HMC-1 human mast cells. Inflamm. Res. 2013, 62, 941–950. [Google Scholar] [CrossRef] [Green Version]
- Sant’Anna, L.S.; Merlugo, L.; Ehle, C.S.; Limberger, J.; Fernandes, M.B.; Santos, M.C.; Mendez, A.S.L.; Paula, F.R.; Moreira, C.M. Chemical Composition and Hypotensive Effect of Campomanesia xanthocarpa. Evid. Based Complement. Altern. Med. 2017, 2017, 1591762. [Google Scholar] [CrossRef]
- Li, L.; Su, C.; Chen, X.; Wang, Q.; Jiao, W.; Luo, H.; Tang, J.; Wang, W.; Li, S.; Guo, S. Chlorogenic acids in cardiovascular disease: A review of dietary consumption, pharmacology, and pharmacokinetics. J. Agric. Food Chem. 2020, 68, 6464–6484. [Google Scholar] [CrossRef]
- Ministerio de Educación Guía de Aplicación de Arquitectura Bioclimática en Locales Educativos. Available online: https://www2.congreso.gob.pe/sicr/cendocbib/con4_uibd.nsf/9A45F1BED1AB7C6705257CCA00550ABD/$FILE/GuiaBioclim%C3%A1tica2008.pdf (accessed on 5 March 2021).
- Ruiz, H. Florae Peruvianae, et Chilensis: Sive, Descriptiones, et Icones Plantarum Peruvianarum, et Chilensium, Secundum Systema Linnaeanum Digestae, cum Characteribus Plurium Generum Evulgatorum Reformatis; Typis Gabrielis de Sancha: New York, NY, USA, 1798; Volume 1. [Google Scholar]
- Tewari, D.; Stankiewicz, A.M.; Mocan, A.; Sah, A.N.; Tzvetkov, N.T.; Huminiecki, L.; Horbańczuk, J.O.; Atanasov, A.G. Ethnopharmacological approaches for dementia therapy and significance of natural products and herbal drugs. Front. Aging Neurosci. 2018, 10, 3. [Google Scholar] [CrossRef] [Green Version]
- Rubio, J.; Caldas, M.; Dávila, S.; Gasco, M.; Gonzales, G.F. Effect of three different cultivars of Lepidium meyenii (Maca) on learning and depression in ovariectomized mice. BMC Complement. Altern. Med. 2006, 6, 23. [Google Scholar] [CrossRef] [Green Version]
- Cui, B.; Zheng, B.L.; He, K.; Zheng, Q.Y. Imidazole alkaloids from lepidium m eyenii. J. Nat. Prod. 2003, 66, 1101–1103. [Google Scholar] [CrossRef]
- Muhammad, I.; Zhao, J.; Dunbar, D.C.; Khan, I.A. Constituents of Lepidium meyenii ‘maca’. Phytochemistry 2002, 59, 105–110. [Google Scholar] [CrossRef]
- Zhou, J.-Y.; Zhou, S.-W. Isorhynchophylline: A plant alkaloid with therapeutic potential for cardiovascular and central nervous system diseases. Fitoterapia 2012, 83, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Ip, S.-P.; Liu, L.; Xian, Y.-F.; Lin, Z.-X. Uncaria rhynchophylla and its major constituents on central nervous system: A review on their pharmacological actions. Curr. Vasc. Pharmacol. 2020, 18, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Tettey, C.O.; Yang, I.-J.; Shin, H.-M. Vasodilatory effect of kaempferol-7-O-α-L-rhamnopyranoside via NO-cGMP-PKG signaling. Arch. Biochem. Biophys. 2019, 667, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Lock, O.; Perez, E.; Villar, M.; Flores, D.; Rojas, R. Bioactive compounds from plants used in Peruvian traditional medicine. Nat. Prod. Commun. 2016, 11, 315–337. [Google Scholar] [PubMed]
- Rubio, J.; Dang, H.; Gong, M.; Liu, X.; Chen, S.; Gonzales, G.F. Aqueous and hydroalcoholic extracts of Black Maca (Lepidium meyenii) improve scopolamine-induced memory impairment in mice. Food Chem. Toxicol. 2007, 45, 1882–1890. [Google Scholar] [CrossRef] [PubMed]
- Koeberle, A.; Werz, O. Multi-target approach for natural products in inflammation. Drug Discov. Today 2014, 19, 1871–1882. [Google Scholar] [CrossRef]
- Rastelli, G.; Pinzi, L. Computational polypharmacology comes of age. Front. Pharmacol. 2015, 6, 157. [Google Scholar] [CrossRef] [Green Version]
- Chaudhari, R.; Fong, L.W.; Tan, Z.; Huang, B.; Zhang, S. An up-to-date overview of computational polypharmacology in modern drug discovery. Expert Opin. Drug Discov. 2020, 15, 1025–1044. [Google Scholar] [CrossRef]
- Baek, S.; Choi, N.H.; Lee, K.-P.; Jhun, H.; Kim, J. Smallanthus sonchifolius leaf attenuates neuroinflammation. J. Exerc. Nutr. Biochem. 2018, 22, 31–35. [Google Scholar] [CrossRef]
- Martinez-Oliveira, P.; de Oliveira, M.F.; Alves, N.; Coelho, R.P.; Pilar, B.C.; Güllich, A.A.; Ströher, D.J.; Boligon, A.; Piccoli, J.d.C.E.; Mello-Carpes, P.B. Yacon leaf extract supplementation demonstrates neuroprotective effect against memory deficit related to β-amyloid-induced neurotoxicity. J. Funct. Foods 2018, 48, 665–675. [Google Scholar] [CrossRef]
- Olofinnade, A.T.; Alawode, A.; Onaolapo, A.Y.; Onaolapo, O.J. Lepidium meyenii Supplemented Diet Modulates Neurobehavioral and Biochemical Parameters in Mice Fed High-Fat High-Sugar Diet. Endocr. Metab. Immune Disord. Drug Targets 2021, 21, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Leuci, R.; Brunetti, L.; Poliseno, V.; Laghezza, A.; Loiodice, F.; Tortorella, P.; Piemontese, L. Natural compounds for the prevention and treatment of cardiovascular and neurodegenerative diseases. Foods 2021, 10, 29. [Google Scholar] [CrossRef] [PubMed]
- Gugnani, K.S.; Vu, N.; Rondón-Ortiz, A.N.; Böhlke, M.; Maher, T.J.; Pino-Figueroa, A.J. Neuroprotective activity of macamides on manganese-induced mitochondrial disruption in U-87 MG glioblastoma cells. Toxicol. Appl. Pharmacol. 2018, 340, 67–76. [Google Scholar] [CrossRef]
- Diedrich, C.; da Silva, L.D.; Sari, R.; de Cristo Borges, G.C.; Muniz, H.S.; de Lima, V.A.; Oldoni, T.L.C.; Carpes, S.T. Bioactive compounds extraction of Croton lechleri barks from Amazon forest using chemometrics tools. J. King Saud Univ. 2021, 33, 101416. [Google Scholar] [CrossRef]
- Chen, Z.; Bertin, R.; Marin, R.; Medjiofack Djeujo, F.; Froldi, G. Effects of Croton lechleri sap (Sangre de Drago) on AGEs formation, LDL oxidation and oxidative stress related to vascular diseases. Nat. Prod. Res. 2021, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.-Q.; Shaw, P.C.; Hu, Z.; Yang, W.; Ip, S.-P.; Xian, Y.-F.; Lin, Z.-X. Comparison of the chemical constituents and anti-Alzheimer’s disease effects of Uncaria rhynchophylla and Uncaria tomentosa. Chin. Med. 2021, 16, 110. [Google Scholar] [CrossRef]
- Shi, Z.; Lu, Z.; Zhao, Y.; Wang, Y.; Zhao-Wilson, X.; Guan, P.; Duan, X.; Chang, Y.-Z.; Zhao, B. Neuroprotective effects of aqueous extracts of Uncaria tomentosa: Insights from 6-OHDA induced cell damage and transgenic Caenorhabditis elegans model. Neurochem. Int. 2013, 62, 940–947. [Google Scholar] [CrossRef]
- Castilhos, L.G.; Oliveira, J.S.; Adefegha, S.A.; Manzoni, A.G.; Passos, D.F.; Assmann, C.E.; Silveira, L.L.; Trelles, K.B.; Kronbauer, M.; Doleski, P.H.; et al. Uncaria tomentosa improves cognition, memory and learning in middle-aged rats. Exp. Gerontol. 2020, 138, 111016. [Google Scholar] [CrossRef]
- Franco, L.A.; Matiz, G.E.; Calle, J.; Pinzón, R.; Ospina, L.F. Actividad antinflamatoria de extractos y fracciones obtenidas de cálices de Physalis peruviana L. Biomédica 2007, 27, 110–115. [Google Scholar] [CrossRef] [Green Version]
- Areiza-Mazo, N.; Robles, J.; Zamudio-Rodriguez, J.A.; Giraldez, L.; Echeverria, V.; Barrera-Bailon, B.; Aliev, G.; Sahebkar, A.; Ashraf, G.M.; Barreto, G.E. Extracts of Physalis peruviana Protect Astrocytic Cells Under Oxidative Stress With Rotenone. Front. Chem. 2018, 6, 276. [Google Scholar] [CrossRef] [Green Version]
- Benites, J.; Guerrero-Castilla, A.; Salas, F.; Martinez, J.L.; Jara-Aguilar, R.; Venegas-Casanova, E.A.; Suarez-Rebaza, L.; Guerrero-Hurtado, J.; Calderon, P.B. Chemical composition, in vitro cytotoxic and antioxidant activities of the essential oil of Peruvian Minthostachys mollis Griseb. Boletín Latinoam. Caribe Plantas Med. Aromáticas 2018, 17, 566–574. [Google Scholar]
- Espinoza Medrano, D.A. Efecto Antiinflamatorio de un Gel Elaborado a Base de Extracto Seco de Hojas de Minthostachys Mollis (Muña) en Rattus Rattus. Bachelor’s Thesis, Universidad Católica los Ángeles de Chimbote, Chimbote, Peru, 2018. [Google Scholar]
- Sabbagh, M.N.; Hendrix, S.; Harrison, J.E. FDA position statement “Early Alzheimer’s disease: Developing drugs for treatment, Guidance for Industry”. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2019, 5, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Fleck, L.M. Alzheimer’s and Aducanumab: Unjust Profits and False Hopes. Hastings Cent. Rep. 2021, 51, 9–11. [Google Scholar] [CrossRef] [PubMed]
- Lamar, M.; Boots, E.A.; Arfanakis, K.; Barnes, L.L.; Schneider, J.A. Common brain structural alterations associated with cardiovascular disease risk factors and Alzheimer’s dementia: Future directions and implications. Neuropsychol. Rev. 2020, 30, 546–557. [Google Scholar] [CrossRef]
- Lin, Y.-T.; Wu, Y.-C.; Sun, G.-C.; Ho, C.-Y.; Wong, T.-Y.; Lin, C.-H.; Chen, H.-H.; Yeh, T.-C.; Li, C.-J.; Tseng, C.-J. Effect of resveratrol on reactive oxygen species-induced cognitive impairment in rats with angiotensin II-induced early Alzheimer’s disease. J. Clin. Med. 2018, 7, 329. [Google Scholar] [CrossRef] [Green Version]
- Trofimiuk, E.; Wielgat, P.; Braszko, J.J. Candesartan, angiotensin II type 1 receptor blocker is able to relieve age-related cognitive impairment. Pharmacol. Rep. 2018, 70, 87–92. [Google Scholar] [CrossRef]
- Royea, J.; Hamel, E. Brain angiotensin II and angiotensin IV receptors as potential Alzheimer’s disease therapeutic targets. Geroscience 2020, 42, 1237–1256. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.; Lee, G.; Ritter, A.; Sabbagh, M.; Zhong, K. Alzheimer’s disease drug development pipeline: 2020. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2020, 6, e12050. [Google Scholar] [CrossRef]
- Jones, G.M.M.; Sahakian, B.J.; Levy, R.; Warburton, D.M.; Gray, J.A. Effects of acute subcutaneous nicotine on attention, information processing and short-term memory in Alzheimer’s disease. Psychopharmacology 1992, 108, 485–494. [Google Scholar] [CrossRef]
- Baum, L.; Lam, C.W.K.; Cheung, S.K.-K.; Kwok, T.; Lui, V.; Tsoh, J.; Lam, L.; Leung, V.; Hui, E.; Ng, C. Six-month randomized, placebo-controlled, double-blind, pilot clinical trial of curcumin in patients with Alzheimer disease. J. Clin. Psychopharmacol. 2008, 28, 110–113. [Google Scholar] [CrossRef] [Green Version]
- Szaniszlo, P.; German, P.; Hajas, G.; Saenz, D.N.; Kruzel, M.; Boldogh, I. New insights into clinical trial for colostrininTM in Alzheimer’s disease. J. Nutr. Health Aging 2009, 13, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, R.R.; Popovic-Nikolic, M.R.; Nikolic, K.; Uliassi, E.; Bolognesi, M.L. A perspective on multi-target drug discovery and design for complex diseases. Clin. Transl. Med. 2018, 7, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Pei, J.; Lai, L. Computational multitarget drug design. J. Chem. Inf. Model. 2017, 57, 403–412. [Google Scholar] [CrossRef]
- Astiti, M.A.; Jittmittraphap, A.; Leaungwutiwong, P.; Chutiwitoonchai, N.; Pripdeevech, P.; Mahidol, C.; Ruchirawat, S.; Kittakoop, P. LC-QTOF-MS/MS Based Molecular Networking Approach for the Isolation of α-Glucosidase Inhibitors and Virucidal Agents from Coccinia grandis (L.) Voigt. Foods 2021, 10, 3041. [Google Scholar] [CrossRef]
- Goyzueta-Mamani, L.D.; Barazorda-Ccahuana, H.L.; Mena-Ulecia, K.; Chávez-Fumagalli, M.A. Antiviral Activity of Metabolites from Peruvian Plants against SARS-CoV-2: An In Silico Approach. Molecules 2021, 26, 3882. [Google Scholar] [CrossRef]
- Chen, S.-Y.; Chu, C.-C.; Chyau, C.-C.; Yang, J.-W.; Duh, P.-D. Djulis (Chenopodium formosanum) and its bioactive compounds affect vasodilation, angiotensin converting enzyme activity, and hypertension. Food Biosci. 2019, 32, 100469. [Google Scholar] [CrossRef]
- Satari, A.; Ghasemi, S.; Habtemariam, S.; Asgharian, S.; Lorigooini, Z. Rutin: A Flavonoid as an Effective Sensitizer for Anticancer Therapy; Insights into Multifaceted Mechanisms and Applicability for Combination Therapy. Evid. Based Complement. Altern. Med. 2021, 2021, 9913179. [Google Scholar] [CrossRef]
- Hassan, S.T.S. Brassicasterol with Dual Anti-Infective Properties against HSV-1 and Mycobacterium tuberculosis, and Cardiovascular Protective Effect: Nonclinical In Vitro and In Silico Assessments. Biomedicines 2020, 8, 132. [Google Scholar] [CrossRef]
- Yu, P.; Lou, D.; Qi, L.; Chen, Z. The novel antiviral properties of brassicasterol against human adenovirus. Future Virol. 2021, 16, 591–600. [Google Scholar] [CrossRef]
- Ikarashi, N.; Ono, T.; Hoshino, M.; Toda, T.; Yazawa, Y.; Sugiyama, K. Mechanistic analysis of bladder cancer suppression by brassicasterol in a rat model of N-butyl-N-(4-hydroxybutyl) nitrosamine-induced bladder cancer. Tradit. Kampo Med. 2021, 8, 163–169. [Google Scholar] [CrossRef]
- Vanmierlo, T.; Popp, J.; Kölsch, H.; Friedrichs, S.; Jessen, F.; Stoffel-Wagner, B.; Bertsch, T.; Hartmann, T.; Maier, W.; von Bergmann, K. The plant sterol brassicasterol as additional CSF biomarker in Alzheimer’s disease. Acta Psychiatr. Scand. 2011, 124, 184–192. [Google Scholar] [CrossRef] [PubMed]
- De Marino, S.; Gala, F.; Zollo, F.; Vitalini, S.; Fico, G.; Visioli, F.; Iorizzi, M. Identification of minor secondary metabolites from the latex of Croton lechleri (Muell-Arg) and evaluation of their antioxidant activity. Molecules 2008, 13, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Kellici, T.F.; Ntountaniotis, D.; Liapakis, G.; Tzakos, A.G.; Mavromoustakos, T. The dynamic properties of angiotensin II type 1 receptor inverse agonists in solution and in the receptor site. Arab. J. Chem. 2019, 12, 5062–5078. [Google Scholar] [CrossRef]
- Zhang, H.; Unal, H.; Desnoyer, R.; Han, G.W.; Patel, N.; Katritch, V.; Karnik, S.S.; Cherezov, V.; Stevens, R.C. Structural Basis for Ligand Recognition and Functional Selectivity at Angiotensin Receptor. J. Biol. Chem. 2015, 290, 29127–29139. [Google Scholar] [CrossRef] [Green Version]
- Shakour, N.; Bianconi, V.; Pirro, M.; Barreto, G.E.; Hadizadeh, F.; Sahebkar, A. In silico evidence of direct interaction between statins and β-amyloid. J. Cell. Biochem. 2019, 120, 4710–4715. [Google Scholar] [CrossRef] [PubMed]
- Castro-Silva, E.S.; Bello, M.; Rosales-Hernández, M.C.; Correa-Basurto, J.; Hernández-Rodríguez, M.; Villalobos-Acosta, D.; Méndez-Méndez, J.V.; Estrada-Pérez, A.; Murillo-Álvarez, J.; Muñoz-Ochoa, M. Fucosterol from Sargassum horridum as an amyloid-beta (Aβ1-42) aggregation inhibitor: In vitro and in silico studies. J. Biomol. Struct. Dyn. 2021, 39, 1271–1283. [Google Scholar] [CrossRef]
- Shahid, S.M.A.; Kuddus, M.; Ahmed, M.Q.; Saleem, M.; Kausar, M.A.; Khalid, M.A.; Alghassab, T.A.; Acar, T.; Alenazi, F.S.H. In silico approach to discover the role of metals for the treatment of Alzheimer’s disease amyloid-beta (Aβ) peptide. Biochem. Cell. Arch. 2018, 18, 629–635. [Google Scholar]
- Fardanesh, A.; Zibaie, S.; Shariati, B.; Attar, F.; Rouhollah, F.; Akhtari, K.; Shahpasand, K.; Saboury, A.A.; Falahati, M. Amorphous aggregation of tau in the presence of titanium dioxide nanoparticles: Biophysical, computational, and cellular studies. Int. J. Nanomed. 2019, 14, 901. [Google Scholar] [CrossRef] [Green Version]
- Murugan, N.A.; Nordberg, A.; Ågren, H. Different positron emission tomography tau tracers bind to multiple binding sites on the tau fibril: Insight from computational modeling. ACS Chem. Neurosci. 2018, 9, 1757–1767. [Google Scholar] [CrossRef] [Green Version]
- Kuang, G.; Murugan, N.A.; Zhou, Y.; Nordberg, A.; Ågren, H. Computational Insight into the Binding Profile of the Second-Generation PET Tracer PI2620 with Tau Fibrils. ACS Chem. Neurosci. 2020, 11, 900–908. [Google Scholar] [CrossRef]
- Franzoni, L.; Nicastro, G.; Pertinhez, T.A.; Oliveira, E.; Nakaie, C.R.; Paiva, A.C.M.; Schreier, S.; Spisni, A. Structure of two fragments of the third cytoplasmic loop of the rat angiotensin II AT1A receptor: Implications with respect to receptor activation and G-protein selection and coupling. J. Biol. Chem. 1999, 274, 227–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rezaei-Ghaleh, N.; Andreetto, E.; Yan, L.-M.; Kapurniotu, A.; Zweckstetter, M. Interaction between amyloid beta peptide and an aggregation blocker peptide mimicking islet amyloid polypeptide. PLoS ONE 2011, 6, e20289. [Google Scholar] [CrossRef] [PubMed]
- Mudedla, S.K.; Murugan, N.A.; Agren, H. Free energy landscape for alpha-helix to beta-sheet interconversion in small amyloid forming peptide under nanoconfinement. J. Phys. Chem. B 2018, 122, 9654–9664. [Google Scholar] [CrossRef] [PubMed]
- Hernandes-Alejandro, M.; Montaño, S.; Harrington, C.R.; Wischik, C.M.; Salas-Casas, A.; Cortes-Reynosa, P.; Pérez Salazar, E.; Cazares-Apatiga, J.; Apatiga-Perez, R.; Ontiveros Torres, M.Á. Analysis of the relationship between metalloprotease-9 and tau protein in Alzheimer’s disease. J. Alzheimer’s Dis. 2020, 76, 553–569. [Google Scholar] [CrossRef] [PubMed]
- Lippens, G.; Gigant, B. Elucidating Tau function and dysfunction in the era of cryo-EM. J. Biol. Chem. 2019, 294, 9316–9325. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.; Dwivedi, D.H.; Maji, S.; Kishor, S. Screening of phytometabolites in oil from cape gooseberry (Physalis peruviana L.) fruit pomace. Acta Hortic. 2020, 1292, 371–376. [Google Scholar] [CrossRef]
- Ramadan, M.F.; Mörsel, J.-T. Oil goldenberry (Physalis peruviana L.). J. Agric. Food Chem. 2003, 51, 969–974. [Google Scholar] [CrossRef]
- Ramadan, M.F. Bioactive phytochemicals of cape gooseberry (Physalis peruviana L.). In Bioactive Compounds in Underutilized Fruits and Nuts; Springer: Cham, Switzerland, 2020; pp. 75–90. [Google Scholar]
- Moreno, W.F.Q.; Torres, W.D.Q.; Ana, A.; Ariasc, G.; Cevallosc, E.; Zambranoc, Z.; Britoa, H.; Salazara, K. Essential oil of Minthostachys mollis: Extraction and chemical composition of fresh and stored samples. Arab. J. Med. Aromat. Plants 2019, 5, 59–71. [Google Scholar]
- Flores-Sanchez, I.J.; Ramos-Valdivia, A.C. A review from patents inspired by two plant genera: Uncaria and Hamelia. Phytochem. Rev. 2017, 16, 693–723. [Google Scholar] [CrossRef]
- Cai, Y.; Evans, F.J.; Roberts, M.F.; Phillipson, J.D.; Zenk, M.H.; Gleba, Y.Y. Polyphenolic compounds from Croton lechleri. Phytochemistry 1991, 30, 2033–2040. [Google Scholar] [CrossRef]
- Ubillas, R.; Jolad, S.D.; Bruening, R.C.; Kernan, M.R.; King, S.R.; Sesin, D.F.; Barrett, M.; Stoddart, C.A.; Flaster, T.; Kuo, J. SP-303, an antiviral oligomeric proanthocyanidin from the latex of Croton lechleri (Sangre de Drago). Phytomedicine 1994, 1, 77–106. [Google Scholar] [CrossRef]
- Caetano, B.F.R.; de Moura, N.A.; Almeida, A.P.S.; Dias, M.C.; Sivieri, K.; Barbisan, L.F. Yacon (Smallanthus sonchifolius) as a Food Supplement: Health-Promoting Benefits of Fructooligosaccharides. Nutrients 2016, 8, 436. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.B.; Chagas-Paula, D.A.; Secatto, A.; Gasparoto, T.H.; Faccioli, L.H.; Campanelli, A.P.; Da Costa, F.B. Topical anti-inflammatory activity of yacon leaf extracts. Rev. Bras. Farmacogn. 2013, 23, 497–505. [Google Scholar] [CrossRef] [Green Version]
- Khajehei, F.; Merkt, N.; Claupein, W.; Graeff-Hoenninger, S. Yacon (Smallanthus sonchifolius Poepp. & Endl.) as a novel source of health promoting compounds: Antioxidant activity, phytochemicals and sugar content in flesh, peel, and whole tubers of seven cultivars. Molecules 2018, 23, 278. [Google Scholar]
- Contreras-Puentes, N.; Alvíz-Amador, A. Hypoglycaemic Property of Yacon (Smallanthus sonchifolius (Poepp. and Hendl.) H. Robinson): A Review. Pharmacogn. Rev. 2020, 14, 37–44. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Villaorduña, L.; Gasco, M.; Rubio, J.; Gonzales, C. Maca (Lepidium meyenii Walp), una revisión sobre sus propiedades biológicas. Rev. Peru. Med. Exp. Salud Publica 2014, 31, 100–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peres, N.d.S.L.; Bortoluzzi, L.C.P.; Marques, L.L.M.; Formigoni, M.; Fuchs, R.H.B.; Droval, A.A.; Cardoso, F.A.R. Medicinal effects of Peruvian maca (Lepidium meyenii): A review. Food Funct. 2020, 11, 83–92. [Google Scholar] [CrossRef]
- Sander, T.; Freyss, J.; von Korff, M.; Rufener, C. DataWarrior: An open-source program for chemistry aware data visualization and analysis. J. Chem. Inf. Model. 2015, 55, 460–473. [Google Scholar] [CrossRef]
- Dallakyan, S.; Olson, A.J. Small-molecule library screening by docking with PyRx. Methods Mol. Biol. 2015, 1263, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, P.; Eckert, A.O.; Schrey, A.K.; Preissner, R. ProTox-II: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 2018, 46, W257–W263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Da Silva, A.W.S.; Vranken, W.F. ACPYPE-Antechamber python parser interface. BMC Res. Notes 2012, 5, 367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Wang, W.; Kollman, P.A.; Case, D.A. Automatic atom type and bond type perception in molecular mechanical calculations. J. Mol. Graph. Model. 2006, 25, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Van Der Spoel, D.; Lindahl, E.; Hess, B.; Groenhof, G.; Mark, A.E.; Berendsen, H.J.C. GROMACS: Fast, flexible, and free. J. Comput. Chem. 2005, 26, 1701–1718. [Google Scholar] [CrossRef]
- Racine, J. gnuplot 4.0: A portable interactive plotting utility. J. Appl. Econom. 2006, 21, 133–141. [Google Scholar] [CrossRef]
- Valdés-Tresanco, M.S.; Valdés-Tresanco, M.E.; Valiente, P.A.; Moreno, E. gmx_MMPBSA: A new tool to perform end-state free energy calculations with GROMACS. J. Chem. Theory Comput. 2021, 17, 6281–6291. [Google Scholar] [CrossRef]
- Miller III, B.R.; McGee, T.D., Jr.; Swails, J.M.; Homeyer, N.; Gohlke, H.; Roitberg, A.E. MMPBSA. py: An efficient program for end-state free energy calculations. J. Chem. Theory Comput. 2012, 8, 3314–3321. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Laskowski, R.A.; Swindells, M.B. LigPlot+: Multiple ligand–protein interaction diagrams for drug discovery. J. Chem. Inf. Model. 2011, 51, 2778–2786. [Google Scholar] [CrossRef]
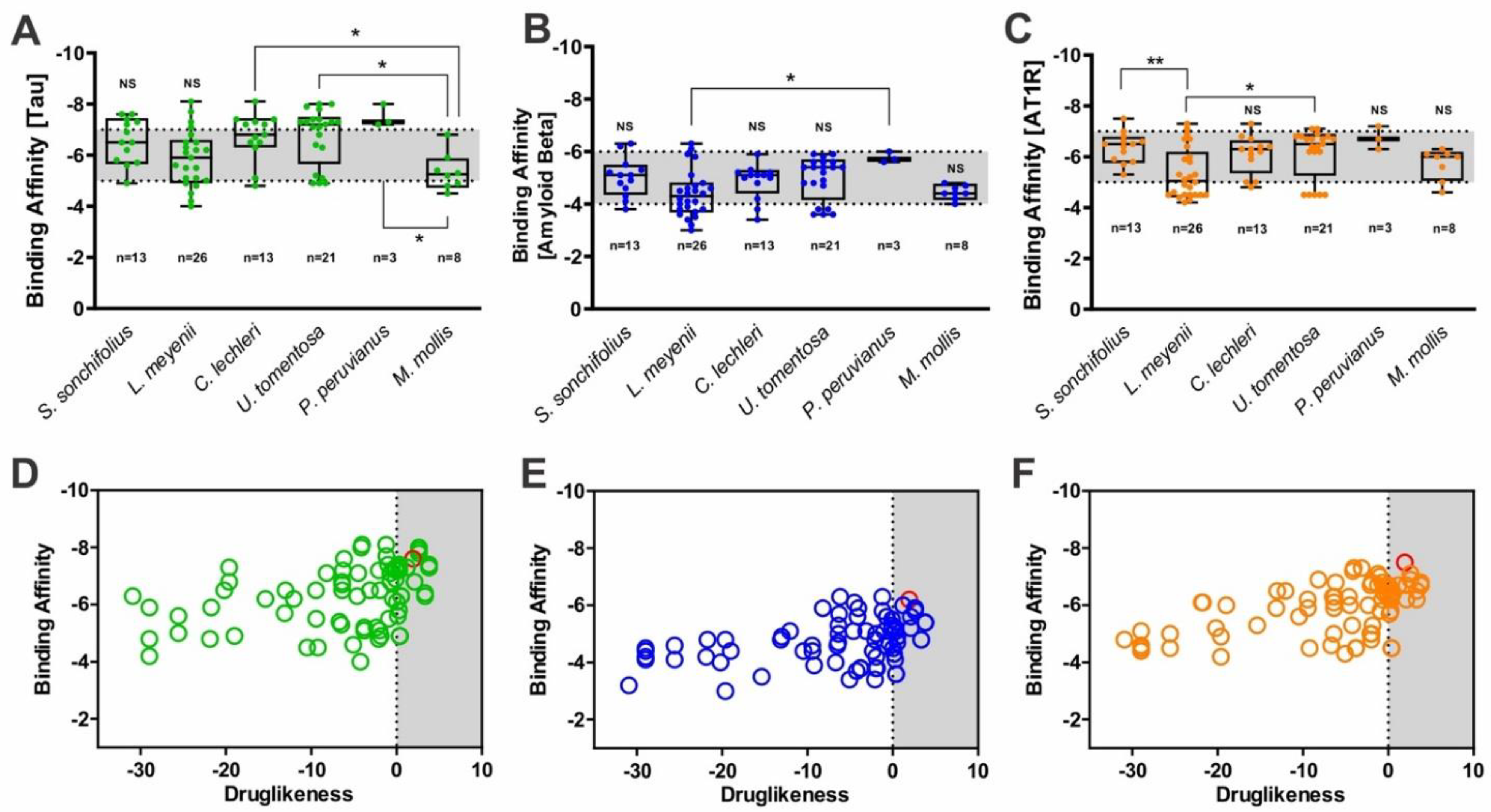
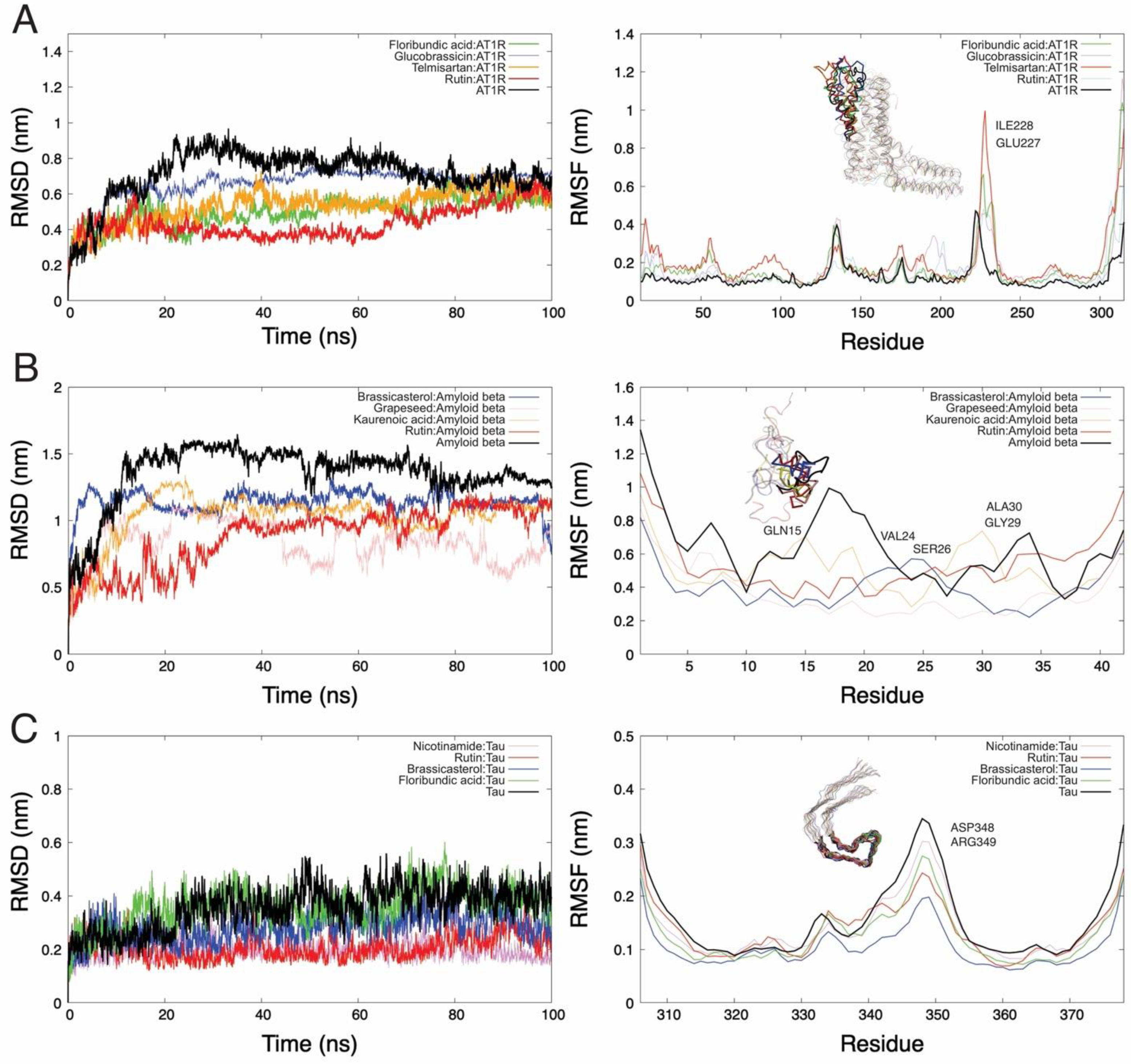
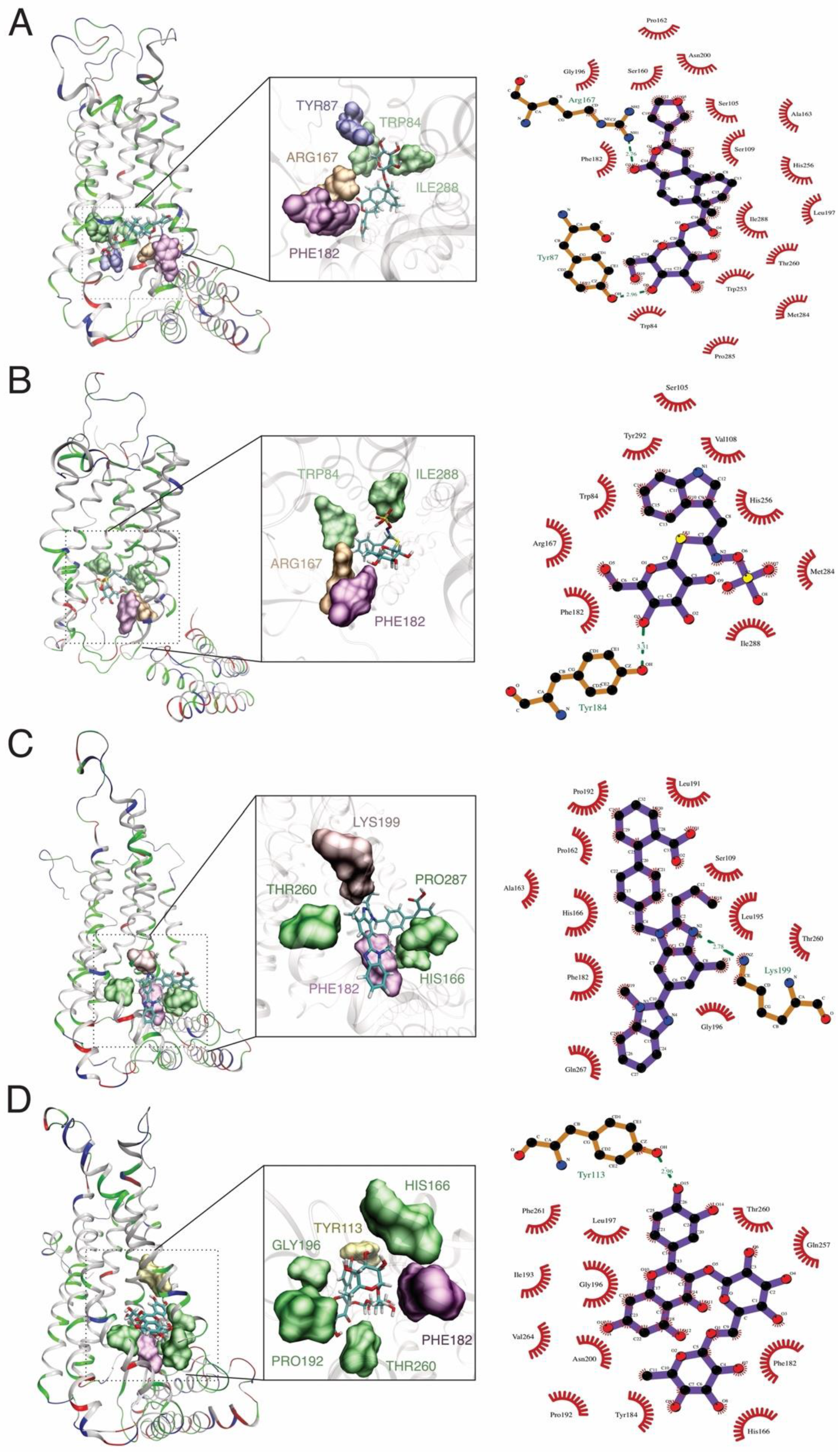

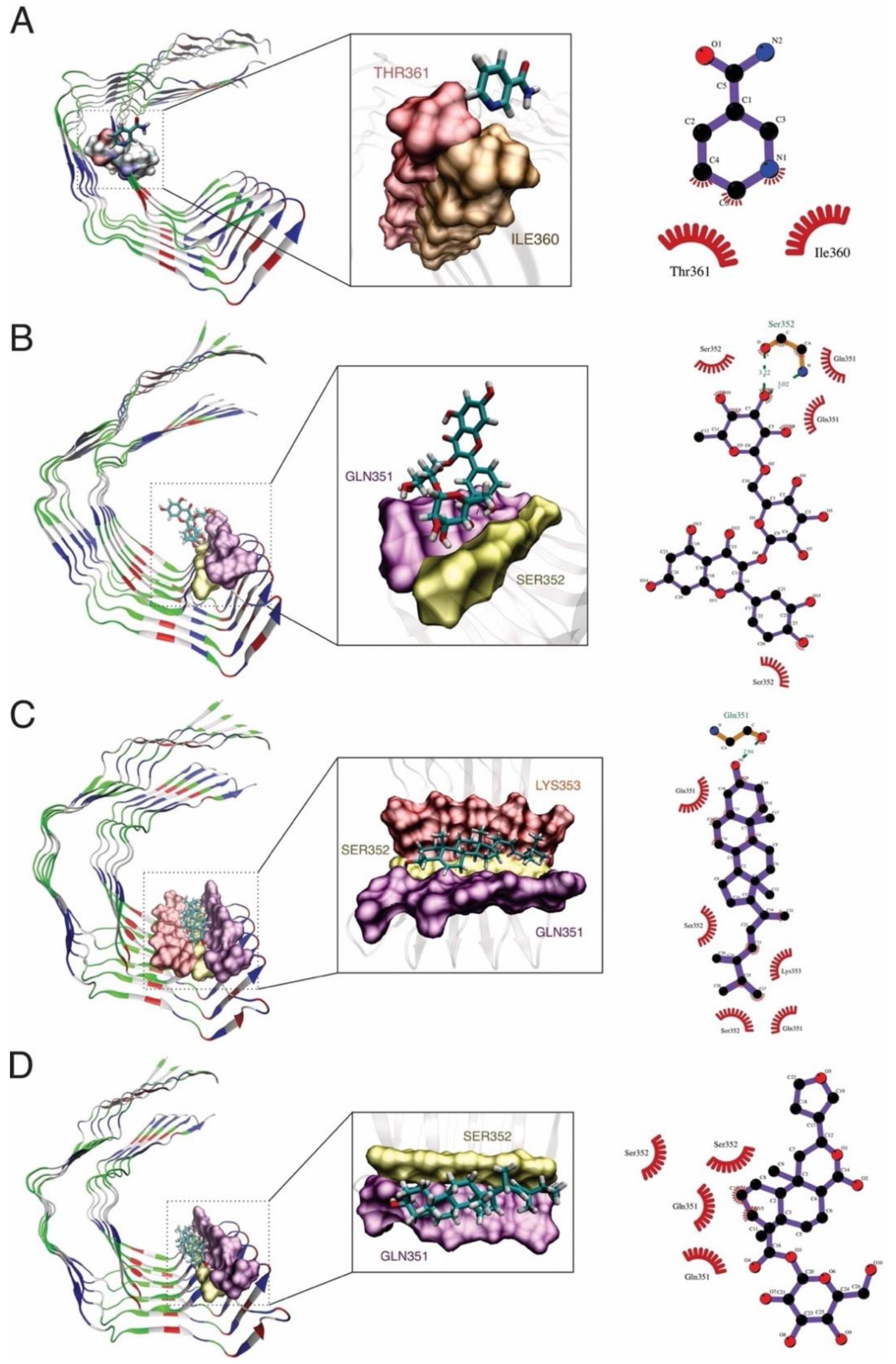
| Control | PubChem CID | Compound | Binding Affinity |
|---|---|---|---|
| Tau | 936 | Nicotinamide | −4.2 |
| 60150609 | TRx0237 | −3.1 | |
| Amyloid beta | 91973920 | Grapeseed extract | −6.7 |
| 51030870 | PQ912 | −5.7 | |
| 25008296 | ALZ 801 | −4.2 | |
| AT1R | 65999 | Telmisartan | −8.7 |
| 2541 | Candesartan | −7.7 | |
| 172198 | Angiotensin II | −7.6 |
| Control | PubChem CID | Compound | A | B | C | D | E | RO5 | Drug-Likeness |
|---|---|---|---|---|---|---|---|---|---|
| Tau | 5281327 | Brassicasterol | - | - | - | - | 890 | Yes | −1.2 |
| 15694360 | Floribundic acid | - | - | - | + | 274 | Yes | −4.0 | |
| 5280805 | Rutin | - | - | - | - | 500 | No | 1.93 | |
| 936 | Nicotinamide | - | - | - | - | 250 | Yes | −0.6 | |
| Amyloid beta | 73062 | Kaurenoic acid | + | - | - | - | 100 | Yes | −6.1 |
| 5281327 | Brassicasterol | - | - | - | - | 890 | Yes | −1.2 | |
| 5280805 | Rutin | - | - | - | - | 500 | No | 1.93 | |
| 91973920 | Grapeseed extract | - | - | - | - | 2500 | No | 1.83 | |
| AT1R | 5280805 | Rutin | - | - | - | - | 500 | No | 1.93 |
| 5317667 | Glucobrassicin | - | - | - | - | 200 | Yes | −3.1 | |
| 15694360 | Floribundic acid | - | - | - | + | 274 | Yes | −4.0 | |
| 65999 | Telmisartan | - | - | - | - | 500 | No | 0.95 |
| Energy Component | Substance | VDWAALS Kcal·mol−1 | EEL Kcal·mol−1 | EGB Kcal·mol−1 | ESURF Kcal·mol−1 | ΔGgas Kcal·mol−1 | ΔGsolv Kcal·.mol−1 | ΔTOTAL Kcal·mol−1 |
|---|---|---|---|---|---|---|---|---|
| Amyloid beta | Brassicasterol | −43.04 ± 3.4 | −4.86 ± 5.9 | 15.08 ± 5.0 | −5.27 ± 0.4 | −47.9 ± 6.7 | 9.81 ± 4.9 | −38.08 ± 3.7 |
| Kaurenoic acid | −28.75 ± 2.7 | −12.91 ± 3.9 | 18.48 ± 2.9 | −3.67 ± 0.3 | −50.34 ± 5.0 | 14.8 ± 2.9 | −35.54 ± 3.9 | |
| Rutin | −61.97 ± 4.9 | −32.69 ± 11.6 | 54.48 ± 8.7 | −7.86 ± 0.6 | −94.67 ± 14.4 | 46.62 ± 8.2 | −48.05 ± 7.0 | |
| Grapeseed extract | −37.72 ± 6.7 | −51.73 ± 13.6 | 51.15 ± 8.6 | −5.7 ± 0.6 | −89.46 ± 14.1 | 45.45 ± 8.2 | −44.00 ± 7.0 | |
| Tau | Brassicasterol | −42.49 ± 2.9 | −4.87 ± 3.9 | 14.00 ± 2.7 | −4.68 ± 0.3 | −47.37 ± 4.5 | 9.31 ± 2.7 | −38.05 ± 3.5 |
| Floribundic acid | −20.61 ± 6.2 | −12.8 ± 10.9 | 23.9 ± 12.9 | −2.70 ± 0.8 | −42.55 ± 16.0 | 21.16 ± 12.1 | −21.39 ± 5.2 | |
| Rutin | −33.86 ± 3.9 | −36.7 ± 15.8 | 48.20 ± 11.5 | −4.86 ± 0.4 | −70.60 ± 15.3 | 43.34 ± 11.4 | −27.25 ± 6.1 | |
| Nicotinamide | −4.04 ± 4.4 | −2.86 ± 4.8 | 5.77 ± 6.5 | −0.58 ± 0.6 | −6.90 ± 7.9 | 5.18 ± 6.0 | −1.72 ± 2.4 | |
| AT1R | Floribundic acid | −59.73 ± 3.0 | −27.18 ± 6.6 | 44.26 ± 3.9 | −7.6 ± 0.2 | −95.64 ± 6.1 | 36.66 ± 3.8 | −58.98 ± 4.4 |
| Glucobrassicin | −44.55 ± 2.5 | −7.11 ± 7.9 | 41.77 ± 6.6 | −5.64 ± 0.3 | −51.66 ± 8.3 | 36.13 ± 6.6 | −15.53 ± 4.5 | |
| Rutin | −61.99 ± 2.7 | −7.33 ± 5.3 | 37.06 ± 4.7 | −7.3 ± 0.3 | −69.32 ± 5.7 | 29.76 ± 4.7 | −39.56 ± 3.3 | |
| Telmisartan | −55.15 ± 3.0 | −10.27 ± 5.8 | 33.27 ± 5.2 | −6.71 ± 0.3 | −65.42 ± 6.3 | 26.56 ± 5.5 | −38.87 ± 3.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goyzueta-Mamani, L.D.; Barazorda-Ccahuana, H.L.; Chávez-Fumagalli, M.A.; F. Alvarez, K.L.; Aguilar-Pineda, J.A.; Vera-Lopez, K.J.; Lino Cardenas, C.L. In Silico Analysis of Metabolites from Peruvian Native Plants as Potential Therapeutics against Alzheimer’s Disease. Molecules 2022, 27, 918. https://doi.org/10.3390/molecules27030918
Goyzueta-Mamani LD, Barazorda-Ccahuana HL, Chávez-Fumagalli MA, F. Alvarez KL, Aguilar-Pineda JA, Vera-Lopez KJ, Lino Cardenas CL. In Silico Analysis of Metabolites from Peruvian Native Plants as Potential Therapeutics against Alzheimer’s Disease. Molecules. 2022; 27(3):918. https://doi.org/10.3390/molecules27030918
Chicago/Turabian StyleGoyzueta-Mamani, Luis Daniel, Haruna Luz Barazorda-Ccahuana, Miguel Angel Chávez-Fumagalli, Karla Lucia F. Alvarez, Jorge Alberto Aguilar-Pineda, Karin Jannet Vera-Lopez, and Christian Lacks Lino Cardenas. 2022. "In Silico Analysis of Metabolites from Peruvian Native Plants as Potential Therapeutics against Alzheimer’s Disease" Molecules 27, no. 3: 918. https://doi.org/10.3390/molecules27030918
APA StyleGoyzueta-Mamani, L. D., Barazorda-Ccahuana, H. L., Chávez-Fumagalli, M. A., F. Alvarez, K. L., Aguilar-Pineda, J. A., Vera-Lopez, K. J., & Lino Cardenas, C. L. (2022). In Silico Analysis of Metabolites from Peruvian Native Plants as Potential Therapeutics against Alzheimer’s Disease. Molecules, 27(3), 918. https://doi.org/10.3390/molecules27030918









