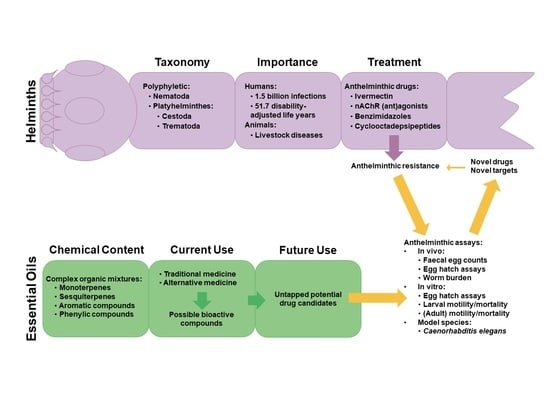
Essential Oils as Novel Anthelmintic Drug Candidates
Abstract
1. Introduction
1.1. Epidemiology of Human Helminthosis
1.2. Common Human Helminths
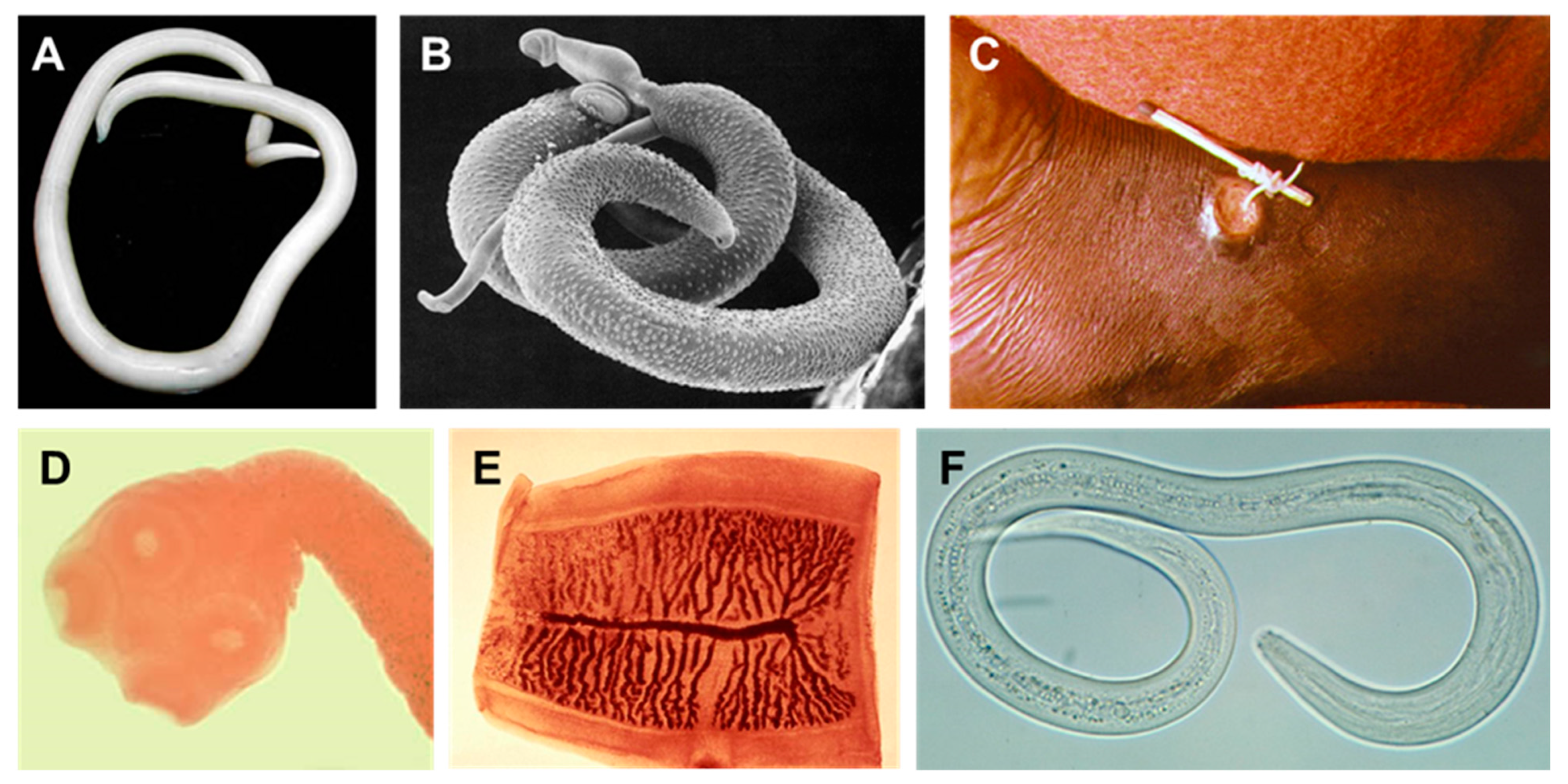
1.2.1. Nematoda
1.2.2. Platyhelminthes
Trematoda
Cestoda
1.3. Animal Parasites
2. Current Anthelmintics
2.1. Ivermectin
2.2. Nicotinic Acetylcholine Receptor (ant)Agonists
2.3. Benzimidazoles
2.4. Cyclooctadepsipeptides
3. Future Strategies
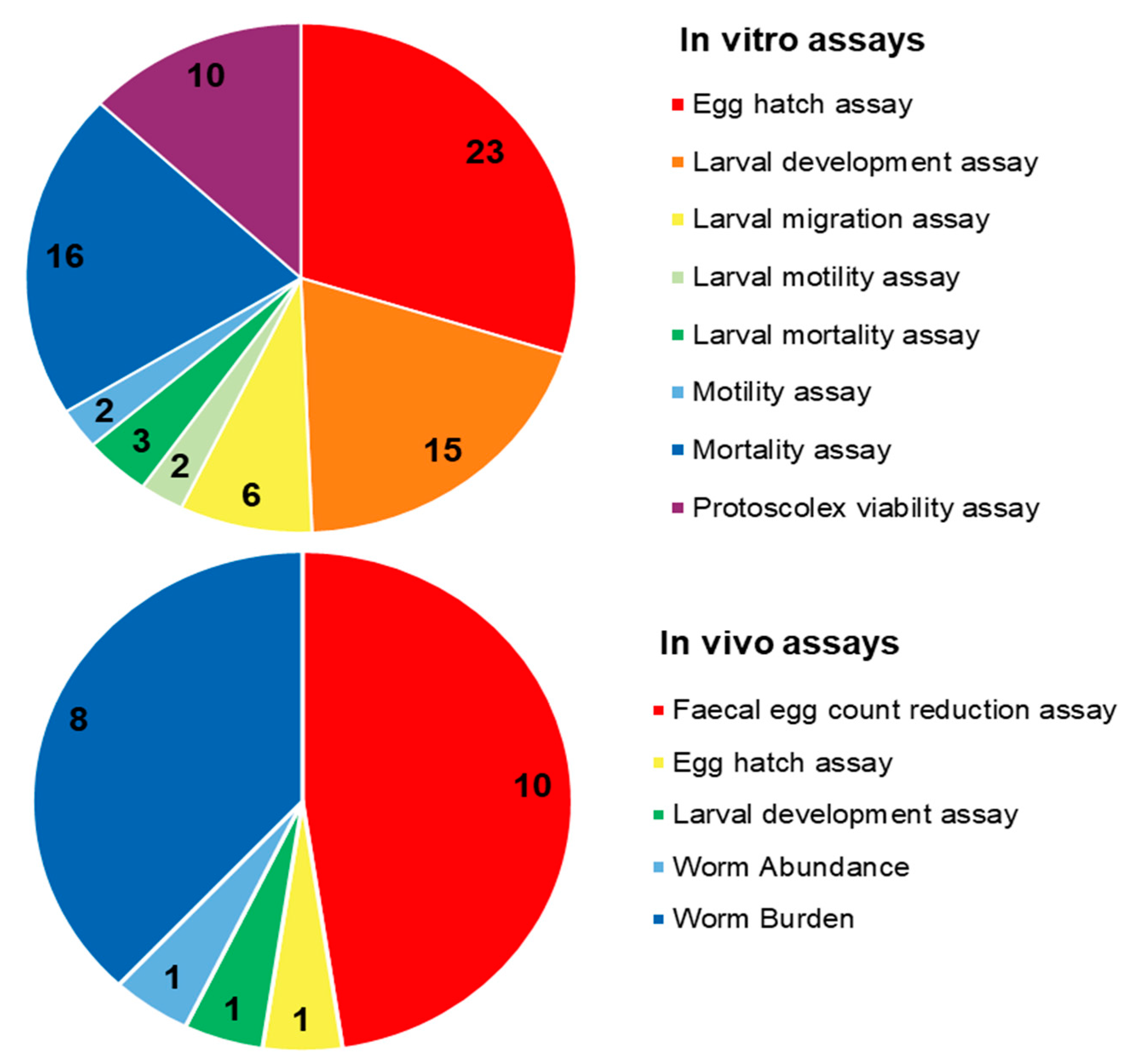
4. Anthelmintic Assays
5. Essential Oils as Anthelmintic Candidates
6. Chemical Classes of EO Components with Reported Anthelmintic Activity
6.1. Monoterpenes
6.2. Sesquiterpenes
6.3. Alcohols
6.4. EO Phenols
6.5. Phenyl Methyl Ethers
6.6. Aldehydes
6.7. Esters
6.8. Ketones
6.9. EO Quinones
6.10. Miscellaneous EO Compounds
7. Traditional Knowledge: The Key to Novel Anthelmintic Drug Candidates
8. Clinical Studies with Essential Oils as Anthelmintic Candidates
9. Patent Literature with Essential Oils as Anthelmintic Candidates
10. Future Prospects and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NTDs | Neglected tropical diseases |
| WHO | World Health Organization |
| DALYs | Disability-adjusted life years |
| GluCl | Glutamate-gated chloride channels |
| nAChRs | Nicotinic acetylcholine receptors |
| AAD | Amino-acetonitrile derivatives |
| EOs | Essential oils |
| EOC | Essential oil component |
| Hp | Heligosomoides polygyrus polygyrus |
| SOAT | Syphacia obvelata and Aspiculuris tetraptera |
| GIN | Gastro-intestinal nematodes |
| Hc | Haemonchus contortus |
| Hs | Haemonchus spp. |
| Ts | Trichostrongylus spp. |
| FECRA | Fecal egg count reduction assay |
| WB | Worm burden |
| Sm | Schistosoma mansoni |
| HC | Hydatid cyst |
| ES | Echinococcus spp. protoscolices |
| GINC | GI nematodes, cattle |
| Ts | Trichostrongylus spp. |
| Pp | Pheritima posthuman |
| Sr | Strongyloides ratti |
| As | Anisakis simplex |
| GINS | Gastro-intestinal nematodes, sheep |
| MA | Mortality assay |
| LDA | Larval development assay |
| EHA | Egg hatch assay |
| LMA | Larval mortality assay |
| LMgA | Larval migration assay |
| PVA | Protoscolex viability assay |
References
- Hotez, P.J.; Pearce, E.J.; Jacobson, J.; Hotez, P.J.; Brindley, P.J.; Bethony, J.M.; King, C.H.; Pearce, E.J.; Jacobson, J. Helminth infections: The great neglected tropical diseases. J. Clin. Investig. 2008, 118, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.L.; Liwanag, H.J.; Snyder, J.S.; Akogun, O.; Belizario, V.; Freeman, M.C.; Gyorkos, T.W.; Imtiaz, R.; Keiser, J.; Krolewiecki, A.; et al. Toward the 2020 goal of soil-transmitted helminthiasis control and elimination. PLoS Negl. Trop. Dis. 2018, 12, e0006606. [Google Scholar] [CrossRef]
- Lustigman, S.; Prichard, R.K.; Gazzinelli, A.; Grant, W.N.; Boatin, B.A.; McCarthy, J.S.; Basáñez, M.G. A research agenda for helminth diseases of humans: The problem of helminthiases. PLoS Negl. Trop. Dis. 2012, 6, e1582. [Google Scholar] [CrossRef]
- Weatherhead, J.E.; Hotez, P.J.; Mejia, R. The Global State of Helminth Control and Elimination in Children. Pediatr. Clin. N. Am. 2017, 64, 867–877. [Google Scholar] [CrossRef]
- Idris, O.A.; Wintola, O.A.; Afolayan, A.J. Helminthiases; prevalence, transmission, host-parasite interactions, resistance to common synthetic drugs and treatment. Heliyon 2019, 5, e01161. [Google Scholar] [CrossRef] [PubMed]
- Weaver, H.J.; Hawdon, J.M.; Hoberg, E.P. Soil-transmitted helminthiases: Implications of climate change and human behavior. Trends Parasitol. 2010, 26, 574–581. [Google Scholar] [CrossRef]
- Tinsley, R.C.; York, J.E.; Everard, A.L.E.; Stott, L.C.; Chapple, S.J.; Tinsley, M.C. Environmental constraints influencing survival of an African parasite in a north temperate habitat: Effects of temperature on egg development. Parasitology 2011, 138, 1029–1038. [Google Scholar] [CrossRef]
- Hotez, P.J.; Bundy, D.A.P.; Beegle, K.; Brooker, S.; Drake, L.; de Silva, N.; Montresor, A.; Engels, D.; Jukes, M.; Chitsulo, L.; et al. Helminth Infections: Soil-transmitted Helminth Infections and Schistosomiasis. In Disease Control Priorities in Developing Countries; Jamison, D.T., Breman, J.G., Measham, A.R., Alleyne, G., Claeson, M., Evans, D.B., Jha, P., Mills, A., Musgrove, P., Eds.; Oxford University Press: New York, NY, USA, 2006; pp. 467–482. ISBN 0-8213-6179-1. [Google Scholar]
- Wright, J.E.; Werkman, M.; Dunn, J.C.; Anderson, R.M. Current epidemiological evidence for predisposition to high or low intensity human helminth infection: A systematic review. Parasites Vectors 2018, 11, 1–12. [Google Scholar] [CrossRef]
- Ekundayo, O.J.; Aliyu, M.H.; Jolly, P.E. A review of intestinal helminthiasis in Nigeria and the need for school-based intervention. J. Rural Trop. Public Health 2007, 6, 33–39. [Google Scholar]
- Crompton, D.W.T.; Nesheim, M.C. Nutritional Impact of Intestinal Helminthiasis During the Human life Cycle. Annu. Rev. Nutr. 2002, 22, 35–59. [Google Scholar] [CrossRef]
- Cox, F.E.G. History of Human Parasitology. Clin. Microbiol. Rev. 2002, 15, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Marti, H. The Discovery of Helminth Life Cycles. In Highlighting Operational and Implementation Research for Control of Helminthiasis; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; Volume 103, ISBN 9780081027509. [Google Scholar]
- Molyneux, D.H.; Savioli, L.; Engels, D. Neglected tropical diseases: Progress towards addressing the chronic pandemic. Lancet 2017, 389, 312–325. [Google Scholar] [CrossRef] [PubMed]
- Hotez, P.J.; Kamath, A. Neglected tropical diseases in sub-Saharan Africa: Review of their prevalence, distribution, and disease burden. PLoS Negl. Trop. Dis. 2009, 3, e412. [Google Scholar] [CrossRef]
- WHO. Neglected Tropical Diseases. [WWW Document]. World Health Organization. 2020. Available online: https://www.who.int/neglected_diseases/diseases/en/ (accessed on 23 May 2022).
- Hotez, P.J.; Fenwick, A.; Savioli, L.; Molyneux, D.H. Rescuing the bottom billion through control of neglected tropical diseases. Lancet 2009, 373, 1570–1575. [Google Scholar] [CrossRef]
- Gabrielli, A.-F.; Montresor, A.; Chitsulo, L.; Engels, D.; Savioli, L. Preventive chemotherapy in human helminthiasis: Theoretical and operational aspects. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 683–693. [Google Scholar] [CrossRef] [PubMed]
- King, C.H. Helminthiasis Epidemiology and Control: Scoring Successes and Meeting the Remaining Challenges. In Highlighting Operational and Implementation Research for Control of Helminthiasis; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; Volume 103, ISBN 9780081027509. [Google Scholar]
- Yap, P.; Fürst, T.; Müller, I.; Kriemler, S.; Utzinger, J.; Steinmann, P. Determining soil-transmitted helminth infection status and physical fitness of school-aged children. J. Vis. Exp. 2012, 66, e3966. [Google Scholar] [CrossRef]
- WHO. WHO Roadmap Inspires Unprecedented Support to Defeat Neglected Tropical Diseases; [WWW Document]; World Health Organization: Geneva, Switzerland, 2012.
- Lo, N.C.; Addiss, D.G.; Hotez, P.J.; King, C.H.; Stothard, J.R.; Evans, D.S.; Colley, D.G.; Lin, W.; Coulibaly, J.T.; Bustinduy, A.L.; et al. A call to strengthen the global strategy against schistosomiasis and soil-transmitted helminthiasis: The time is now. Lancet Infect. Dis. 2017, 17, e64–e69. [Google Scholar] [CrossRef]
- Dobson, A.; Lafferty, K.D.; Kuris, A.M.; Hechinger, R.F.; Jetz, W. Homage to linnaeus: How many parasites? How many hosts? Light Evol. 2009, 2, 63–82. [Google Scholar] [CrossRef]
- De León, G.P.P.; Poulin, R. An updated look at the uneven distribution of cryptic diversity among parasitic helminths. J. Helminthol. 2018, 92, 197–202. [Google Scholar] [CrossRef]
- Wells, K.; Gibson, D.I.; Clark, N.J. Global patterns in helminth host specificity: Phylogenetic and functional diversity of regional host species pools matter. Ecography 2019, 42, 416–427. [Google Scholar] [CrossRef]
- Wells, K.; Gibson, D.I.; Clark, N.J.; Ribas, A.; Morand, S.; McCallum, H.I. Global spread of helminth parasites at the human–domestic animal–wildlife interface. Glob. Chang. Biol. 2018, 24, 3254–3265. [Google Scholar] [CrossRef]
- Baron, S. (Ed.) Medical Microbiology, 4th ed.; University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996; ISBN 0-9631172-1-1. [Google Scholar]
- Coghlan, A.; Tyagi, R.; Cotton, J.A.; Holroyd, N.; Rosa, B.A.; Tsai, I.J.; Laetsch, D.R.; Beech, R.N.; Day, T.A.; Hallsworth-Pepin, K.; et al. Comparative genomics of the major parasitic worms. Nat. Genet. 2019, 51, 163–174. [Google Scholar] [CrossRef]
- Sayers, E.W.; Barrett, T.; Benson, D.A.; Bryant, S.H.; Canese, K.; Chetvernin, V.; Church, D.M.; Dicuccio, M.; Edgar, R.; Federhen, S.; et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2009, 37, 5–15. [Google Scholar] [CrossRef]
- Batiha, G.E.; Olatunde, A.; El-Mleeh, A.; Hetta, H.F.; Al-Rejaie, S.; Alghamdi, S.; Zahoor, M.; Magdy Beshbishy, A.; Murata, T.; Zaragoza-Bastida, A.; et al. Bioactive Compounds, Pharmacological Actions, and Pharmacokinetics of Wormwood (Artemisia absinthium). Antibiotics 2020, 9, 353. [Google Scholar] [CrossRef]
- Strzemski, M.; Wójciak-Kosior, M.; Sowa, I.; Załuski, D.; Verpoorte, R. Historical and traditional medical applications of Carlina acaulis L.—A critical ethnopharmacological review. J. Ethnopharmacol. 2019, 239, 111842. [Google Scholar] [CrossRef] [PubMed]
- Yadav, V.; Krishnan, A.; Vohora, D. A systematic review on Piper longum L.: Bridging traditional knowledge and pharmacological evidence for future translational research. J. Ethnopharmacol. 2020, 247, 112255. [Google Scholar] [CrossRef]
- Luna, E.C.; Luna, I.S.; Scotti, L.; Monteiro, A.F.M.; Scotti, M.T.; De Moura, R.O.; De Araújo, R.S.A.; Monteiro, K.L.C.; De Aquino, T.M.; Ribeiro, F.F.; et al. Active essential oils and their components in use against neglected diseases and arboviruses. Oxid. Med. Cell. Longev. 2019, 2019, 6587150. [Google Scholar] [CrossRef]
- Islam, M.T.; Martorell, M.; Salehi, B.; Setzer, W.N.; Sharifi-Rad, J. Anti-Schistosoma mansoni effects of essential oils and their components. Phyther. Res. 2020, 34, 1761–1769. [Google Scholar] [CrossRef]
- Page, A. The Cuticle. WormBook 2007, 1–15. [Google Scholar] [CrossRef]
- WHO. Soil-Transmitted Helminth Infections. [WWW Document]. World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/soil-transmitted-helminth-infections (accessed on 17 April 2022).
- CDC Parasites-Enterobiasis (also Known as Pinworm Infection). [WWW Document]. Centers for Disease Control and Prevention. 2013. Available online: https://www.cdc.gov/parasites/pinworm/ (accessed on 17 April 2022).
- Melrose, W.D. Lymphatic filariasis: New insights into an old disease. Int. J. Parasitol. 2002, 32, 947–960. [Google Scholar] [CrossRef]
- WHO. Onchocerciasis. [WWW Document]. World Health Organization. 2019. Available online: https://www.who.int/news-room/fact-sheets/detail/onchocerciasis (accessed on 17 April 2022).
- WHO. Lymphatic Filariasis. World Health Organization. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/lymphatic-filariasis (accessed on 17 April 2022).
- WHO. Dracunculiasis (Guinea-Worm Disease). [WWW Document]. World Health Organization. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/dracunculiasis-(guinea-worm-disease) (accessed on 17 April 2022).
- WHO. Foodborne Trematodiases. [WWW Document]. World Health Organization. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/foodborne-trematodiases (accessed on 18 April 2022).
- WHO. Schistosomiasis. World Health Organization. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/schistosomiasis (accessed on 18 April 2022).
- Fürst, T.; Sayasone, S.; Odermatt, P.; Keiser, J.; Utzinger, J. Manifestation, diagnosis, and management of foodborne trematodiasis. BMJ 2012, 344, e4093. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; Utzinger, J. Emerging foodborne trematodiasis. Emerg. Infect. Dis. 2005, 11, 1507–1514. [Google Scholar] [CrossRef]
- Gryseels, B.; Polman, K.; Clerinx, J.; Kestens, L. Human schistosomiasis. Lancet 2006, 368, 1106–1118. [Google Scholar] [CrossRef]
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 2014, 383, 2253–2264. [Google Scholar] [CrossRef]
- Inobaya, M.T.; Olveda, R.M.; Chau, T.N.; Olveda, D.U.; Ross, A.G. Prevention and control of schistosomiasis: A current perspective. Res. Rep. Trop. Med. 2014, 5, 65–75. [Google Scholar] [CrossRef]
- WHO. Taeniasis/Cysticercosis Fact Sheet. [WWW Document]. World Health Organization. 2019. Available online: https://www.who.int/news-room/fact-sheets/detail/taeniasis-cysticercosis (accessed on 18 April 2022).
- WHO. Echinococcosis Fact Sheet. [WWW Document]. World alth Organization. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/echinococcosis (accessed on 18 April 2022).
- Nansen, P. Production losses and control of helminths in ruminants of temperate regions. Int. J. Parasitol. 1987, 17, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Charlier, J.; van der Voort, M.; Kenyon, F.; Skuce, P.; Vercruysse, J. Chasing helminths and their economic impact on farmed ruminants. Trends Parasitol. 2014, 30, 361–367. [Google Scholar] [CrossRef]
- Fitzpatrick, J.L. Global food security: The impact of veterinary parasites and parasitologists. Vet. Parasitol. 2013, 195, 233–248. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.A.; Coop, R.L.; Wall, R.L. (Eds.) Veterinary Parasitology, 4th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; ISBN 9781119073680. [Google Scholar]
- Leles, D.; Gardner, S.L.; Reinhard, K.; Ĩiguez, A.; Araujo, A. Are Ascaris lumbricoides and Ascaris suum a single species? Parasites Vectors 2012, 5, 42. [Google Scholar] [CrossRef]
- Verschave, S.H.; Charlier, J.; Rose, H.; Claerebout, E.; Morgan, E.R. Cattle and Nematodes Under Global Change: Transmission Models as an Ally. Trends Parasitol. 2016, 32, 724–738. [Google Scholar] [CrossRef]
- Fox, N.J.; Marion, G.; Davidson, R.S.; White, P.C.L.; Hutchings, M.R. Livestock helminths in a changing climate: Approaches and restrictions to meaningful predic. Animals 2012, 2, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Fiel, C.A.; Fernández, A.S.; Rodríguez, E.M.; Fusé, L.A.; Steffan, P.E. Observations on the free-living stages of cattle gastrointestinal nematodes. Vet. Parasitol. 2012, 187, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Winter, J.; Rehbein, S.; Joachim, A. Transmission of helminths between species of ruminants in Austria appears more likely to occur than generally assumed. Front. Vet. Sci. 2018, 5, 30. [Google Scholar] [CrossRef] [PubMed]
- Waller, P.J. Sustainable helminth control of ruminants in developing countries. Vet. Parasitol. 1997, 71, 195–207. [Google Scholar] [CrossRef]
- Hoste, H.; Torres-Acosta, J.F.J. Non chemical control of helminths in ruminants: Adapting solutions for changing worms in a changing world. Vet. Parasitol. 2011, 180, 144–154. [Google Scholar] [CrossRef]
- Cabaret, J.; Bouilhol, M.; Mage, C. Managing helminths of ruminants in organic farming. Vet. Res. 2002, 33, 625–640. [Google Scholar] [CrossRef]
- Hewitson, J.P.; Maizels, R.M. Vaccination against helminth parasite infections. Expert Rev. Vaccines 2014, 13, 473–487. [Google Scholar] [CrossRef]
- Holden-Dye, L.; Walker, R.J. Anthelmintic drugs and nematicides: Studies in Caenorhabditis elegans. WormBook 2014, 1–29. [Google Scholar] [CrossRef]
- Hu, Y.; Ellis, B.L.; Yiu, Y.Y.; Miller, M.M.; Urban, J.F.; Shi, L.Z.; Aroian, R.V. An Extensive Comparison of the Effect of Anthelmintic Classes on Diverse Nematodes. PLoS ONE 2013, 8, e70702. [Google Scholar] [CrossRef]
- Geary, T.G. Ivermectin 20 years on: Maturation of a wonder drug. Trends Parasitol. 2005, 21, 530–532. [Google Scholar] [CrossRef] [PubMed]
- Laing, R.; Gillan, V.; Devaney, E. Ivermectin–Old Drug, New Tricks? Trends Parasitol. 2017, 33, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Geerts, S.; Gryseels, B. Drug resistance in human helminths: Current situation and lessons from livestock. Clin. Microbiol. Rev. 2000, 13, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.P.; Buxton, S.K.; Puttachary, S.; Williamson, S.M.; Wolstenholme, A.J.; Neveu, C.; Cabaret, J.; Charvet, C.L.; Martin, R.J. Antinematodal Drugs-Modes of Action and Resistance: And Worms Will Not Come to Thee (Shakespeare: Cymbeline: IV, ii). In Parasitic Helminths: Targets, Screens, Drugs and Vaccines; Wiley: Hoboken, NJ, USA, 2012; pp. 233–249. [Google Scholar] [CrossRef]
- Waghorn, T.S.; Miller, C.M.; Leathwick, D.M. Confirmation of ivermectin resistance in Ostertagia ostertagi in cattle in New Zealand. Vet. Parasitol. 2016, 229, 139–143. [Google Scholar] [CrossRef]
- El-Abdellati, A.; Geldhof, P.; Claerebout, E.; Vercruysse, J.; Charlier, J. Monitoring macrocyclic lactone resistance in Cooperia oncophora on a Belgian cattle farm during four consecutive years. Vet. Parasitol. 2010, 171, 167–171. [Google Scholar] [CrossRef]
- Ploeger, H.W.; Everts, R.R. Alarming levels of anthelmintic resistance against gastrointestinal nematodes in sheep in the Netherlands. Vet. Parasitol. 2018, 262, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Dent, J.A.; Smith, M.M.; Vassilatis, D.K.; Avery, L. The genetics of ivermectin resistance in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 2000, 97, 2674–2679. [Google Scholar] [CrossRef]
- Bygarski, E.E.; Prichard, R.K.; Ardelli, B.F. Resistance to the macrocyclic lactone moxidectin is mediated in part by membrane transporter P-glycoproteins: Implications for control of drug resistant parasitic nematodes. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 143–151. [Google Scholar] [CrossRef][Green Version]
- Strong, L. Overview: The impact of avermectins on pastureland ecology. Vet. Parasitol. 1993, 48, 3–17. [Google Scholar] [CrossRef]
- Wolstenholme, A.J.; Fairweather, I.; Prichard, R.; Von Samson-Himmelstjerna, G.; Sangster, N.C. Drug resistance in veterinary helminths. Trends Parasitol. 2004, 20, 469–476. [Google Scholar] [CrossRef]
- dos Santos, J.M.L.; Vasconcelos, J.F.; Frota, G.A.; de Freitas, E.P.; Teixeira, M.; da Silva Vieira, L.; Bevilaqua, C.M.L.; Monteiro, J.P. Quantitative molecular diagnosis of levamisole resistance in populations of Haemonchus contortus. Exp. Parasitol. 2019, 205, 107734. [Google Scholar] [CrossRef] [PubMed]
- Sales, N.; Love, S. Resistance of Haemonchus sp. to monepantel and reduced efficacy of a derquantel / abamectin combination confirmed in sheep in NSW, Australia. Vet. Parasitol. 2016, 228, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Kaminsky, R.; Ducray, P.; Jung, M.; Clover, R.; Rufener, L.; Bouvier, J.; Weber, S.S.; Wenger, A.; Wieland-Berghausen, S.; Goebel, T.; et al. A new class of anthelmintics effective against drug-resistant nematodes. Nature 2008, 452, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Fru, M.F.; Puoti, A. Acquired resistance to monepantel in C. elegans—What about parasitic nematodes? Worm 2014, 3, e29738. [Google Scholar] [CrossRef]
- Hamer, K.; Bartley, D.; Jennings, A.; Morrison, A.; Sargison, N. Lack of efficacy of monepantel against trichostrongyle nematodes in a UK sheep floc. Vet. Parasitol. 2018, 257, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Niciura, S.C.M.; Cruvinel, G.G.; Moraes, C.V.; Chagas, A.C.S.; Esteves, S.N.; Benavides, M.V.; Amarante, A.F.T. In vivo selection for Haemonchus contortus resistance to monepantel. J. Helminthol. 2020, 94, e46. [Google Scholar] [CrossRef] [PubMed]
- Lacey, E. The role of the cytoskeletal protein, tubulin, in the mode of action and mechanism of drug resistance to benzimidazoles. Int. J. Parasitol. 1988, 18, 885–936. [Google Scholar] [CrossRef]
- Silvestre, A.; Humbert, J.F. Diversity of benzimidazole-resistance alleles in populations of small ruminant parasites. Int. J. Parasitol. 2002, 32, 921–928. [Google Scholar] [CrossRef]
- Alvarez-Sanchez, M.A.; Mainar-Jaime, R.C.; Perez-Garcia, J.; Rojo-Vazquez, F.A. Resistance of Fasciola hepatica to triclabendazole and albendazole in sheep in Spain. Vet. Rec. 2006, 159, 424–425. [Google Scholar] [CrossRef] [PubMed]
- Borgsteede, F.H.M.; Dercksen, D.D.; Huijbers, R. Doramectin and albendazole resistance in sheep in The Netherlands. Vet. Parasitol. 2007, 144, 180–183. [Google Scholar] [CrossRef]
- Furtado, L.F.V.; de Paiva Bello, A.C.P.; Rabelo, É.M.L. Benzimidazole resistance in helminths: From problem to diagnosis. Acta Trop. 2016, 162, 95–102. [Google Scholar] [CrossRef]
- Kotze, A.C.; Cowling, K.; Bagnall, N.H.; Hines, B.M.; Ruffell, A.P.; Hunt, P.W.; Coleman, G.T. Relative level of thiabendazole resistance associated with the E198A and F200Y SNPs in larvae of a multi-drug resistant isolate of Haemonchus contortus. Int. J. Parasitol. Drugs Drug Resist. 2012, 2, 92–97. [Google Scholar] [CrossRef]
- Martin, R.J.; Buxton, S.K.; Neveu, C.; Charvet, C.L.; Robertson, A.P. Emodepside and SL0-1 potassium channels: A review. Exp. Parasitol. 2012, 132, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Mehlhorn, H.; Schmahl, G.; Frese, M.; Mevissen, I.; Harder, A.; Krieger, K. Effects of a combination of emodepside and praziquantel on parasites of reptiles and rodents. Parasitol. Res. 2005, 97, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Von Samson-Himmelstjerna, G.; Harder, A.; Sangster, N.C.; Coles, G.C. Efficacy of two cyclooctadepsipeptides, PF1022A and emodepside, against anthelmintic-resistant nematodes in sheep and cattle. Parasitology 2005, 130, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.M.; Vidyashankar, A.N. An inconvenient truth: Global worming and anthelmintic resistance. Vet. Parasitol. 2012, 186, 70–78. [Google Scholar] [CrossRef]
- Buckingham, S.D.; Partridge, F.A.; Sattelle, D.B. Automated, high-throughput, motility analysis in Caenorhabditis elegans and parasitic nematodes: Applications in the search for new anthelmintics. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Fazzio, L.E.; Sánchez, R.O.; Streitenberger, N.; Galvan, W.R.; Giudici, C.J.; Gimeno, E.J. The effect of anthelmintic resistance on the productivity in feedlot cattle. Vet. Parasitol. 2014, 206, 240–245. [Google Scholar] [CrossRef]
- Bloemhoff, Y.; Danaher, M.; Forbes, A.; Morgan, E.; Mulcahy, G.; Power, C.; Sayers, R. Parasite control practices on pasture-based dairy farms in the Republic of Ireland. Vet. Parasitol. 2014, 204, 352–363. [Google Scholar] [CrossRef]
- O’Shaughnessy, J.; Earley, B.; Mee, J.F.; Doherty, M.L.; Crosson, P.; Barrett, D.; de Waal, T. Controlling nematodes in dairy calves using targeted selective treatments. Vet. Parasitol. 2015, 209, 221–228. [Google Scholar] [CrossRef]
- Moser, W.; Schindler, C.; Keiser, J. Drug Combinations against Soil-Transmitted Helminth Infections. In Highlighting Operational and Implementation Research for Control of Helminthiasis; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; Volume 103, ISBN 9780081027509. [Google Scholar]
- Salinas, G.; Risi, G. Caenorhabditis elegans: Nature and nurture gift to nematode parasitologists. Parasitology 2018, 145, 979–987. [Google Scholar] [CrossRef]
- Holden-Dye, L.; Walker, R.J. How Relevant is Caenorhabditis elegans as a Model for the Analysis of Parasitic Nematode Biology? In Parasitic Helminths; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2012; pp. 23–41. ISBN 9783527330591. [Google Scholar]
- Keiser, J.; Utzinger, J. Efficacy of Current Drugs Against Soil-Transmitted Helminth Infections. JAMA 2008, 299, 1937–1948. [Google Scholar] [CrossRef] [PubMed]
- Doyle, S.R.; Cotton, J.A. Genome-wide Approaches to Investigate Anthelmintic Resistance. Trends Parasitol. 2019, 35, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Wolstenholme, A.J.; Martin, R.J. Anthelmintics—From Discovery to Resistance. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 218–219. [Google Scholar] [CrossRef][Green Version]
- Coles, G.C.; Bauer, C.; Borgsteede, F.H.M.; Geerts, S.; Klei, T.R.; Taylor, M.A.; Waller, P.J. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) methods for the detection of anthelmintic resistance in nematodes of veterinary importance. Vet. Parasitol. 1992, 44, 35–44. [Google Scholar] [CrossRef]
- Katiki, L.M.; Ferreira, J.F.S.; Zajac, A.M.; Masler, C.; Lindsay, D.S.; Chagas, A.C.S.; Amarante, A.F.T. Caenorhabditis elegans as a model to screen plant extracts and compounds as natural anthelmintics for veterinary use. Vet. Parasitol. 2011, 182, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Sant’anna, V.; Vommaro, R.C.; de Souza, W. Caenorhabditis elegans as a model for the screening of anthelminthic compounds: Ultrastructural study of the effects of albendazole. Exp. Parasitol. 2013, 135, 1–8. [Google Scholar] [CrossRef]
- Saha, S.; Lachance, S. Effect of essential oils on cattle gastrointestinal nematodes assessed by egg hatch, larval migration and mortality testing. J. Helminthol. 2019, 94, e111. [Google Scholar] [CrossRef]
- Jabbar, A.; Iqbal, Z.; Khan, M.N. In vitro anthelmintic activity of Trachyspermum ammi seeds. Pharmacogn. Mag. 2006, 2, 126–129. [Google Scholar]
- Lorimer, S.D.; Perry, N.B.; Foster, L.M.; Burgess, E.J.; Douch, P.G.C.; Hamilton, M.C.; Donaghy, M.J.; McGregor, R.A. A Nematode Larval Motility Inhibition Assay for Screening Plant Extracts and Natural Products. J. Agric. Food Chem. 1996, 44, 2842–2845. [Google Scholar] [CrossRef]
- Kotze, A.C.; Clifford, S.; O’Grady, J.; Behnke, J.M.; Mccarthy, J.S. An in vitro larval motility assay to determine anthelmintic sensitivity for human hookworm and Strongyloides species. Am. J. Trop. Med. Hyg. 2004, 71, 608–616. [Google Scholar] [CrossRef]
- Mathew, M.D.; Mathew, N.D.; Miller, A.; Simpson, M.; Au, V.; Garland, S.; Gestin, M.; Edgley, M.L.; Flibotte, S.; Balgi, A.; et al. Using C. elegans Forward and Reverse Genetics to Identify New Compounds with Anthelmintic Activity. PLoS Negl. Trop. Dis. 2016, 10, e0005058. [Google Scholar] [CrossRef] [PubMed]
- Mathew, M.D.; Mathew, N.D.; Ebert, P.R. WormScan: A technique for high-throughput phenotypic analysis of Caenorhabditis elegans. PLoS ONE 2012, 7, 3–8. [Google Scholar] [CrossRef]
- Puckering, T.; Thompson, J.; Sathyamurthy, S.; Sukumar, S.; Shapira, T.; Ebert, P. Automated Wormscan. F1000Research 2019, 6, 192. [Google Scholar] [CrossRef]
- Risi, G.; Aguilera, E.; Ladós, E.; Suárez, G.; Carrera, I.; Álvarez, G.; Salinas, G. Caenorhabditis elegans infrared-based motility assay identified new hits for nematicide drug development. Vet. Sci. 2019, 6, 29. [Google Scholar] [CrossRef]
- Simonetta, S.H.; Golombek, D.A. An automated tracking system for Caenorhabditis elegans locomotor behavior and circadian studies application. J. Neurosci. Methods 2007, 161, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Burns, A.R.; Luciani, G.M.; Musso, G.; Bagg, R.; Yeo, M.; Zhang, Y.; Rajendran, L.; Glavin, J.; Hunter, R.; Redman, E.; et al. Caenorhabditis elegans is a useful model for anthelmintic discovery. Nat. Commun. 2015, 6, 8485. [Google Scholar] [CrossRef]
- Liu, M.; Kipanga, P.; Mai, A.H.; Dhondt, I.; Braeckman, B.P.; De Borggraeve, W.; Luyten, W. Bioassay-guided isolation of three anthelmintic compounds from Warburgia ugandensis Sprague subspecies ugandensis, and the mechanism of action of polygodial. Int. J. Parasitol. 2018, 48, 833–844. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Benelli, G. Plant-borne ovicides in the fight against mosquito vectors of medical and veterinary importance: A systematic review. Parasitol. Res. 2015, 114, 3201–3212. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R. Beyond mosquitoes—Essential oil toxicity and repellency against bloodsucking insects. Ind. Crops Prod. 2018, 117, 382–392. [Google Scholar] [CrossRef]
- Solórzano-Santos, F.; Miranda-Novales, M.G. Essential oils from aromatic herbs as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, R.; Ayres, V.F.S.; Carvalho, C.E.; Souza, M.G.M.; Guimarães, A.C.; Corrêa, G.M.; Martins, C.H.G.; Takeara, R.; Silva, E.O.; Crotti, A.E.M. Chemical Composition and Antibacterial Activity of the Essential Oil of Vitex agnus-castus L. (Lamiaceae). An. Acad. Bras. Cienc. 2017, 89, 2825–2832. [Google Scholar] [CrossRef] [PubMed]
- Laura de Sene Amâncio Zara, A.; Maria dos Santos, S.; Synthia Fernandes-Oliveira, E.; Gomes Carvalho, R.; Evelim Coelho, G. Estratégias de controle do Aedes aegypti: Uma revisão. Epidemiol. Serviços Saúde 2016, 25, 1–2. [Google Scholar] [CrossRef]
- Silvério, M.R.S.; Espindola, L.S.; Lopes, N.P.; Vieira, P.C. Plant Natural Products for the Control of Aedes aegypti: The Main Vector of Important Arboviruses. Molecules 2020, 25, 3484. [Google Scholar] [CrossRef]
- Cai, J.; Yan, R.; Shi, J.; Chen, J.; Long, M.; Wu, W.; Kuca, K. Antifungal and mycotoxin detoxification ability of essential oils: A review. Phyther. Res. 2022, 36, 62–72. [Google Scholar] [CrossRef]
- Maes, C.; Meersmans, J.; Lins, L.; Bouquillon, S.; Fauconnier, M.-L. Essential Oil-Based Bioherbicides: Human Health Risks Analysis. Int. J. Mol. Sci. 2021, 22, 9396. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Lao, Y.; Pan, Y.; Chen, Y.; Zhao, H.; Gong, L.; Xie, N.; Mo, C.-H. Synergistic Antimicrobial Effectiveness of Plant Essential Oil and Its Application in Seafood Preservation: A Review. Molecules 2021, 26, 307. [Google Scholar] [CrossRef]
- Ellse, L.; Wall, R. The use of essential oils in veterinary ectoparasite control: A review. Med. Vet. Entomol. 2014, 28, 233–243. [Google Scholar] [CrossRef]
- Lam, N.S.; Long, X.; Su, X.; Lu, F. Melaleuca alternifolia (tea tree) oil and its monoterpene constituents in treating protozoan and helminthic infections. Biomed. Pharmacother. 2020, 130, 110624. [Google Scholar] [CrossRef]
- Abidi, A.; Sebai, E.; Dhibi, M.; Alimi, D.; Rekik, M.; B’chir, F.; Maizels, R.M.; Akkari, H. Chemical analyses and anthelmintic effects of Artemisia campestris essential oil. Vet. Parasitol. 2018, 263, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Camurça-Vasconcelos, A.L.F.; Bevilaqua, C.M.L.; Morais, S.M.; Maciel, M.V.; Costa, C.T.C.; Macedo, I.T.F.; Oliveira, L.M.B.; Braga, R.R.; Silva, R.A.; Vieira, L.S. Anthelmintic activity of Croton zehntneri and Lippia sidoides essential oils. Vet. Parasitol. 2007, 148, 288–294. [Google Scholar] [CrossRef]
- Macedo, I.T.F.; de Oliveira, L.M.B.; André, W.P.P.; de Araújo Filho, J.V.; dos Santos, J.M.L.; Rondon, F.C.M.; Ribeiro, W.L.C.; Camurça-Vasconcelos, A.L.F.; de Oliveira, E.F.; de Paula, H.C.B.; et al. Anthelmintic effect of Cymbopogon citratus essential oil and its nanoemulsion on sheep gastrointestinal nematodes. Rev. Bras. Parasitol. Vet. 2019, 28, 522–527. [Google Scholar] [CrossRef]
- Macedo, I.T.F.; de Oliveira, L.M.B.; Ribeiro, W.L.C.; dos Santos, J.M.L.; Silva, K.; das Chagas Silva, K.; de Araújo Filho, J.V.; Camurça-Vasconcelos, A.L.F.; Bevilaqua, C.M.L. Anthelmintic activity of Cymbopogon citratus against Haemonchus contortus. Rev. Bras. Parasitol. Veterinária 2015, 24, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Katiki, L.M.; Chagas, A.C.S.; Takahira, R.K.; Juliani, H.R.; Ferreira, J.F.S.; Amarante, A.F.T. Evaluation of Cymbopogon schoenanthus essential oil in lambs experimentally infected with Haemonchus contortus. Vet. Parasitol. 2012, 186, 312–318. [Google Scholar] [CrossRef] [PubMed]
- De Araújo-Filho, J.V.; Ribeiro, W.L.C.; André, W.P.P.; Cavalcante, G.S.; Rios, T.T.; Schwinden, G.M.; Da Rocha, L.O.; Macedo, I.T.F.; de Morais, S.M.; Bevilaqua, C.M.L.; et al. Anthelmintic activity of Eucalyptus citriodora essential oil and its major component, citronellal, on sheep gastrointestinal nematodes. Rev. Bras. Parasitol. Vet. 2019, 28, 644–651. [Google Scholar] [CrossRef]
- Ribeiro, J.C.; Ribeiro, W.L.C.; Camurça-Vasconcelos, A.L.F.; Macedo, I.T.F.; Santos, J.M.L.; Paula, H.C.B.; Araújo Filho, J.V.; Magalhães, R.D.; Bevilaqua, C.M.L. Efficacy of free and nanoencapsulated Eucalyptus citriodora essential oils on sheep gastrointestinal nematodes and toxicity for mice. Vet. Parasitol. 2014, 204, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Macedo, I.T.F.; Bevilaqua, C.M.L.; de Oliveira, L.M.B.; Camurça-Vasconcelos, A.L.F.; da Silva Vieira, L.; Dos Santos Albano Amóra, S. Evaluation of Eucalyptus citriodora essential oil on goat gastrointestinal nematodes. Rev. Bras. Parasitol. Veterinária 2011, 20, 223–227. [Google Scholar] [CrossRef] [PubMed]
- De Aquino Mesquita, M.; Silva Júnior, J.B.E.; Panassol, A.M.; De Oliveira, E.F.; Vasconcelos, A.L.C.F.; De Paula, H.C.B.; Bevilaqua, C.M.L. Anthelmintic activity of Eucalyptus staigeriana encapsulated oil on sheep gastrointestinal nematodes. Parasitol. Res. 2013, 112, 3161–3165. [Google Scholar] [CrossRef] [PubMed]
- Macedo, I.T.F.; Bevilaqua, C.M.L.; de Oliveira, L.M.B.; Camurça-Vasconcelos, A.L.F.; Vieira, L.; Oliveira, F.R.; Queiroz-Junior, E.M.; Tomé, A.R.; Nascimento, N.R.F. Anthelmintic effect of Eucalyptus staigeriana essential oil against goat gastrointestinal nematodes. Vet. Parasitol. 2010, 173, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Camurça-Vasconcelos, A.L.F.; Bevilaqua, C.M.L.; Morais, S.M.; Maciel, M.V.; Costa, C.T.C.; Macedo, I.T.F.; Oliveira, L.M.B.; Braga, R.R.; Silva, R.A.; Vieira, L.S.; et al. Anthelmintic activity of Lippia sidoides essential oil on sheep gastrointestinal nematodes. Vet. Parasitol. 2008, 154, 167–170. [Google Scholar] [CrossRef]
- Grando, T.H.; Baldissera, M.D.; Gressler, L.T.; de Sá, M.F.; Bortoluzzi, B.N.; Schafer, A.S.; Ebling, R.C.; Raffin, R.P.; Santos, R.C.V.; Stefani, L.M.; et al. Melaleuca alternifolia anthelmintic activity in gerbils experimentally infected by Haemonchus contortus. Exp. Parasitol. 2016, 170, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.E.; Benincasa, B.I.; Fachin, A.L.; França, S.C.; Contini, S.S.H.T.; Chagas, A.C.S.; Beleboni, R.O. Thymus vulgaris L. essential oil and its main component thymol: Anthelmintic effects against Haemonchus contortus from sheep. Vet. Parasitol. 2016, 228, 70–76. [Google Scholar] [CrossRef] [PubMed]
- de Melo, N.I.; Magalhaes, L.G.; de Carvalho, C.E.; Wakabayashi, K.A.L.; de P. Aguiar, G.; Ramos, R.C.; Mantovani, A.L.L.; Turatti, I.C.C.; Rodrigues, V.; Groppo, M.; et al. Schistosomicidal activity of the essential oil of Ageratum conyzoides L. (Asteraceae) against adult Schistosoma mansoni worms. Molecules 2011, 16, 762–773. [Google Scholar] [CrossRef]
- Macedo, I.T.F.; de Oliveira, L.M.B.; Camurça-Vasconcelos, A.L.F.; Ribeiro, W.L.C.; dos Santos, J.M.L.; De Morais, S.M.; dePaula, H.C.B.; Bevilaqua, C.M.L. In vitro effects of Coriandrum sativum, Tagetes minuta, Alpinia zerumbet and Lantana camara essential oils on Haemonchus contortus. Rev. Bras. Parasitol. Veterinária 2013, 22, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Castro, L.M.; Pinto, N.B.; Moura, M.Q.; Villela, M.M.; Capella, G.A.; Freitag, R.A.; Berne, M.E.A. Antihelminthic action of the Anethum graveolens essential oil on Haemonchus contortus eggs and larvae. Braz. J. Biol. 2020, 81, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.E.; Benincasa, B.I.; Fachin, A.L.; Contini, S.H.T.; França, S.C.; Chagas, A.C.S.; Beleboni, R.O. Essential oils of Citrus aurantifolia, Anthemis nobile and Lavandula officinalis: In vitro anthelmintic activities against Haemonchus contortus. Parasit. Vectors 2018, 11, 269. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Dai, J.; Yang, L.; Qiu, J. Anthelmintic activity of Arisaema franchetianum and Arisaema lobatum essential oils against Haemonchus contortus. J. Ethnopharmacol. 2013, 148, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Dai, J.; Yang, L.; Qiu, J. In vitro ovicidal and larvicidal activity of the essential oil of Artemisia lancea against Haemonchus contortus (Strongylida). Vet. Parasitol. 2013, 195, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; de Mesquita, L.S.S.; Silva, C.R.; De Mesquita, J.W.C.; De Sá Rocha, E.; Bose, J.; Abiri, R.; Figueiredo, P.D.M.S.; Costa, L.M. Chemical profile and biological activities of essential oil from artemisia vulgaris L. Cultivated in Brazil. Pharmaceuticals 2019, 12, 49. [Google Scholar] [CrossRef] [PubMed]
- Parreira, N.A.; Magalhães, L.G.; Morais, D.R.; Caixeta, S.C.; De Sousa, J.P.B.; Bastos, J.K.; Cunha, W.R.; Silva, M.L.A.; Nanayakkara, N.P.D.; Rodrigues, V.; et al. Antiprotozoal, schistosomicidal, and antimicrobial activities of the essential oil from the leaves of baccharis dracunculifolia. Chem. Biodivers. 2010, 7, 993–1001. [Google Scholar] [CrossRef]
- de Oliveira, R.N.; Rehder, V.L.G.; Santos Oliveira, A.S.; Júnior, Í.M.; de Carvalho, J.E.; de Ruiz, A.L.T.G.; de Lourdes Sierpe Jeraldo, V.; Linhares, A.X.; Allegretti, S.M. Schistosoma mansoni: In vitro schistosomicidal activity of essential oil of Baccharis trimera (less) DC. Exp. Parasitol. 2012, 132, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudvand, H.; Tavakoli Oliaei, R.; Mirbadie, S.R.; Kheirandish, F.; Tavakoli Kareshk, A.; Ezatpour, B.; Mahmoudvand, H. Efficacy and Safety of Bunium Persicum (Boiss) to Inactivate Protoscoleces during Hydatid Cyst Operations. Surg. Infect. (Larchmt). 2016, 17, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.H.G.; Fracarolli, L.; Vieira, T.M.; Dias, H.J.; Cruz, M.G.; Deus, C.C.H.; Nicolella, H.D.; Stefani, R.; Rodrigues, V.; Tavares, D.C.; et al. Schistosomicidal Effects of the Essential Oils of Citrus limonia and Citrus reticulata Against Schistosoma mansoni. Chem. Biodivers. 2017, 14, e1600194. [Google Scholar] [CrossRef]
- Aguiar, G.P.; Melo, N.I.; Wakabayashi, K.A.L.; Lopes, M.H.S.; Mantovani, A.L.L.; Dias, H.J.; Fukui, M.J.; Keles, L.C.; Rodrigues, V.; Groppo, M.; et al. Chemical composition and in vitro schistosomicidal activity of the essential oil from the flowers of Bidens sulphurea (Asteraceae). Nat. Prod. Res. 2013, 27, 920–924. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudvand, H.; Pakravanan, M.; Aflatoonian, M.R.; Khalaf, A.K.; Niazi, M.; Mirbadie, S.R.; Tavakoli Kareshk, A.; Khatami, M. Efficacy and safety of Curcuma longa essential oil to inactivate hydatid cyst protoscoleces. BMC Complement. Altern. Med. 2019, 19, 187. [Google Scholar] [CrossRef] [PubMed]
- Katiki, L.M.; Chagas, A.C.S.; Bizzo, H.R.; Ferreira, J.F.S.; Amarante, A.F.T. Anthelmintic activity of Cymbopogon martinii, Cymbopogon schoenanthus and Mentha piperita essential oils evaluated in four different in vitro tests. Vet. Parasitol. 2011, 183, 103–108. [Google Scholar] [CrossRef]
- Nirmal, S.A.; Girme, A.S.; Bhalke, R.D. Major constituents and anthelmintic activity of volatile oils from leaves and flowers of Cymbopogon martini Roxb. Nat. Prod. Res. 2007, 21, 1217–1220. [Google Scholar] [CrossRef]
- Soares, M.H.; Dias, H.J.; Vieira, T.M.; de Souza, M.G.M.; Cruz, A.F.F.; Badoco, F.R.; Nicolella, H.D.; Cunha, W.R.; Groppo, M.; Martins, C.H.G.; et al. Chemical Composition, Antibacterial, Schistosomicidal, and Cytotoxic Activities of the Essential Oil of Dysphania ambrosioides (L.) Mosyakin & Clemants (Chenopodiaceae). Chem. Biodivers. 2017, 14, e1700149. [Google Scholar] [CrossRef]
- Tabari, M.A.; Youssefi, M.R.; Nasiri, M.; Hamidi, M.; Kiani, K.; Alian Samakkhah, S.; Maggi, F. Towards green drugs against cestodes: Effectiveness of Pelargonium roseum and Ferula gummosa essential oils and their main component on Echinococcus granulosus protoscoleces. Vet. Parasitol. 2019, 266, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, K.A.L.; De Melo, N.I.; Aguiar, D.P.; De Oliveira, P.F.; Groppo, M.; Da Silva Filho, A.A.; Rodrigues, V.; Cunha, W.R.; Tavares, D.C.; Magalhães, L.G.; et al. Anthelmintic Effects of the Essential Oil of Fennel (Foeniculum vulgare Mill., Apiaceae) against Schistosoma mansoni. Chem. Biodivers. 2015, 12, 1105–1114. [Google Scholar] [CrossRef]
- Matos-Rocha, T.J.; Dos Santos Cavalcanti, M.G.; Barbosa-Filho, J.M.; Lúcio, A.S.S.C.; Veras, D.L.; Feitosa, A.P.S.; De Siqueira Júnior, J.P.; De Almeida, R.N.; Marques, M.O.M.; Alves, L.C.; et al. In vitro evaluation of schistosomicidal activity of essential oil of mentha x villosa and some of its chemical constituents in adult worms of schistosoma mansoni. Planta Med. 2013, 79, 1307–1312. [Google Scholar] [CrossRef]
- Mahmoudvand, H.; Fallahi, S.; Mahmoudvand, H.; Shakibaie, M.; Harandi, M.F.; Dezaki, E.S. Efficacy of Myrtus communis L. to Inactivate the Hydatid Cyst Protoscoleces. J. Investig. Surg. 2016, 29, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Olounladé, P.A.; Azando, E.V.B.; Hounzangbé-Adoté, M.S.; Ha, T.B.T.; Leroy, E.; Moulis, C.; Fabre, N.; Magnaval, J.F.; Hoste, H.; Valentin, A. In vitro anthelmintic activity of the essential oils of Zanthoxylum zanthoxyloides and Newbouldia laevis against Strongyloides ratti. Parasitol. Res. 2012, 110, 1427–1433. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudvand, H.; Dezaki, E.S.; Kheirandish, F.; Ezatpour, B.; Jahanbakhsh, S.; Harandi, M.F. Scolicidal effects of black cumin seed (nigella sativa) essential oil on hydatid cysts. Korean J. Parasitol. 2014, 52, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Pessoa, L.; Morais, S.; Bevilaqua, C.M.; Luciano, J.H. Anthelmintic activity of essential oil of Ocimum gratissimum Linn. and eugenol against Haemonchus contortus. Vet. Parasitol. 2002, 109, 59–63. [Google Scholar] [CrossRef] [PubMed]
- López, V.; Cascella, M.; Benelli, G.; Maggi, F.; Gómez-Rincón, C. Green drugs in the fight against Anisakis simplex—Larvicidal activity and acetylcholinesterase inhibition of Origanum compactum essential oil. Parasitol. Res. 2018, 117, 861–867. [Google Scholar] [CrossRef]
- Lahlou, M. Potential of Origanum compactum as a cercaricide in Morocco. Ann. Trop. Med. Parasitol. 2002, 96, 587–593. [Google Scholar] [CrossRef]
- López, V.; Pavela, R.; Gómez-Rincón, C.; Les, F.; Bartolucci, F.; Galiffa, V.; Petrelli, R.; Cappellacci, L.; Maggi, F.; Canale, A.; et al. Efficacy of origanum syriacum essential oil against the mosquito vector culex quinquefasciatus and the gastrointestinal parasite anisakis simplex, with insights on acetylcholinesterase inhibition. Molecules 2019, 24, 2563. [Google Scholar] [CrossRef]
- Kozan, E.; Ilhan, M.; Tümen, I.; Küpeli Akkol, E. The scolicidal activity of the essential oil obtained from the needles of Pinus nigra Arn. subsp. pallasiana (Lamb.) Holmboe on hydatid cyst. J. Ethnopharmacol. 2019, 235, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Gaínza, Y.A.; Chagas, A.C.; Fantatto, R.R.; Chaves, F.C.M.; Bizzo, H.R.; Esteves, S.N. Piper aduncum against Haemonchus contortus isolates: Cross resistance and the research of natural bioactive compounds. Rev. Bras. Parasitol. Vet. 2016, 25, 383–393. [Google Scholar] [CrossRef]
- Caixeta, S.C.; Magalhães, L.G.; Demelo, N.I.; Wakabayashi, K.A.L.; de P. Aguiar, G.; de P. Aguiar, D.; Mantovani, A.L.L.; Alves, J.M.; Oliveira, P.F.; Tavares, D.C.; et al. Chemical composition and in vitro schistosomicidal activity of the essential oil of plectranthus neochilus grown in Southeast Brazil. Chem. Biodivers. 2011, 8, 2149–2157. [Google Scholar] [CrossRef]
- Pinto, N.B.; De Castro, L.M.; Azambuja, R.H.M.; Capella, G.D.A.; De Moura, M.Q.; Terto, W.D.; Freitag, R.A.; Jeske, S.T.; Villela, M.M.; Cleff, M.B.; et al. Ovicidal and larvicidal potential of rosmarinus officinalis to control gastrointestinal nematodes of sheep. Rev. Bras. Parasitol. Vet. 2019, 28, 807–811. [Google Scholar] [CrossRef]
- Ortu, E.; Sanna, G.; Scala, A.; Pulina, G.; Caboni, P.; Battacone, G. In vitro anthelmintic activity of active compounds of the fringed rue Ruta chalepensis against dairy ewe gastrointestinal nematodes. J. Helminthol. 2017, 91, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Moazeni, M.; Saharkhiz, M.J.; Hoseini, A.A.; Alavi, A.M. In vitro scolicidal effect of Satureja khuzistanica (Jamzad) essential oil. Asian Pac. J. Trop. Biomed. 2012, 2, 616–620. [Google Scholar] [CrossRef]
- Politi, F.A.S.; Souza Júnior, A.A.; Fantatto, R.R.; Pietro, R.C.L.R.; Barioni Júnior, W.; Rabelo, M.D.; Bizzo, H.R.; de Souza Chagas, A.C.; Furlan, M. Chemical Composition and In vitro Anthelmintic Activity of Extracts of Tagetes patula against a Multidrug-Resistant Isolate of Haemonchus contortus. Chem. Biodivers. 2018, 15, e1700507. [Google Scholar] [CrossRef]
- Godinho, L.S.; Aleixo De Carvalho, L.S.; Barbosa De Castro, C.C.; Dias, M.M.; Pinto, P.D.F.; Crotti, A.E.M.; Pinto, P.L.S.; De Moraes, J.; Da Silva Filho, A.A. Anthelmintic activity of crude extract and essential oil of tanacetum vulgare (Asteraceae) against adult worms of Schistosoma mansoni. Sci. World J. 2014, 2014, 460342. [Google Scholar] [CrossRef]
- de Melo, N.I.; Mantovani, A.L.L.; de Oliveira, P.F.; Groppo, M.; da Silva Filho, A.A.; Rodrigues, V.; Cunha, W.R.; Tavares, D.C.; Magalhães, L.G.; Crottii, A.E.M. Antischistosomal and Cytotoxic Effects of the Essential Oil of Tetradenia riparia (Lamiaceae). Nat. Prod. Commun. 2015, 10, 1627–1630. [Google Scholar] [CrossRef] [PubMed]
- Moazeni, M.; Saharkhiz, M.J.; Hosseini, A.A. In vitro lethal effect of ajowan (Trachyspermum ammi L.) essential oil on hydatid cyst protoscoleces. Vet. Parasitol. 2012, 187, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Wang, W.X.; Dai, J.L.; Zhu, L. In vitro anthelmintic activity of Zanthoxylum simulans essential oil against Haemonchus contortus. Vet. Parasitol. 2015, 211, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Grando, T.H.; De Sá, M.F.; Baldissera, M.D.; Oliveira, C.B.; De Souza, M.E.; Raffin, R.P.; Santos, R.C.V.; Domingues, R.; Minho, A.P.; Leal, M.L.R.; et al. In vitro activity of essential oils of free and nanostructured Melaleuca alternifolia and of terpinen-4-ol on eggs and larvae of Haemonchus contortus. J. Helminthol. 2016, 90, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Balahbib, A.; El Omari, N.; Hachlafi, N.E.; Lakhdar, F.; El Menyiy, N.; Salhi, N.; Mrabti, H.N.; Bakrim, S.; Zengin, G.; Bouyahya, A. Health beneficial and pharmacological properties of p-cymene. Food Chem. Toxicol. 2021, 153, 112259. [Google Scholar] [CrossRef]
- Panda, S.K.; Luyten, W. Antiparasitic activity in Asteraceae with special attention to ethnobotanical use by the tribes of Odisha, India. Parasite 2018, 25, 10. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Rincón, C.; Langa, E.; Murillo, P.; Valero, M.S.; Berzosa, C.; López, V. Activity of tea tree (Melaleuca alternifolia) essential oil against L3 larvae of Anisakis simplex. Biomed Res. Int. 2014, 2014, 549510. [Google Scholar] [CrossRef]
- Rosenau, M.J. Preventive Medicine and Hygiene; D. Appleton: New York, NY, USA, 1913. [Google Scholar]
- Ferrell, J.A. The Rural School and Hookworm Disease; Bulletin (United States. Bureau of Education); U.S. Government Printing Office: Washington, DC, USA, 1914.
- El-kady, A.M.; Ahmad, A.A.; Hassan, T.M.; El-Deek, H.E.M.; Fouad, S.S.; Al-Thaqfan, S.S. Eugenol, a potential schistosomicidal agent with anti-inflammatory and antifibrotic effects against Schistosoma mansoni, induced liver pathology. Infect. Drug Resist. 2019, 12, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.C.; Noguera, M.A.; Romero, M.C.; Montilla, M.P.; González de Selgas, J.M.; Valero, A. Anisakis simplex s.l.: Larvicidal activity of various monoterpenic derivatives of natural origin against L3 larvae in vitro and in vivo. Exp. Parasitol. 2008, 120, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Arı, S.; Kargıoğlu, M.; Temel, M.; Konuk, M. Traditional Tar Production from the Anatolian Black Pine [Pinus nigra Arn. subsp. pallasiana (Lamb.) Holmboe var. pallasiana] and its usages in Afyonkarahisar, Central Western Turkey. J. Ethnobiol. Ethnomed. 2014, 10, 29. [Google Scholar] [CrossRef]
- Hmamouchi, M.; Lahlou, M.; Agoumi, A. Molluscicidal activity of some Moroccan medicinal plants. Fitoterapia 2000, 71, 308–314. [Google Scholar] [CrossRef]
- Kliks, M.M. Studies on the traditional herbal anthelmintic chenopodium ambrosioides L.: Ethnopharmacological evaluation and clinical field trials. Soc. Sci. Med. 1985, 21, 879–886. [Google Scholar] [CrossRef]
- Ketzis, J.; Taylor, A.; Bowman, D.; Brown, D.; Warnick, L.; Erb, H. Chenopodium ambrosioides and its essential oil as treatments for Haemonchus contortus and mixed adult-nematode infections in goats. Small Rumin. Res. 2002, 44, 193–200. [Google Scholar] [CrossRef]
- Behnia, M.; Haghighi, A.; Komeylizadeh, H.; Tabaei, S.-J.S.; Abadi, A. Inhibitory Effects of Iranian Thymus vulgaris Extracts on in Vitro Growth of Entamoeba histolytica. Korean J. Parasitol. 2008, 46, 153. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Huang, B. A review of chemistry and bioactivities of a medicinal spice: Foeniculum vulgare. J. Med. Plants Res. 2011, 5, 3595–3600. [Google Scholar]
- Wang, J.-F.; Deng, Y.-H.; Yang, S.-H.; Liu, Y.-Q.; Wang, Y.-H.; Pan, W.-W.; Zhou, X.-J. Characterization and biological evaluation of six new dimeric lignans with an unusual α,β-unsaturated ketone motif from Zanthoxylum simulans. Bioorg. Med. Chem. Lett. 2014, 24, 4667–4671. [Google Scholar] [CrossRef] [PubMed]
- Ritter, R.A.; Monteiro, M.V.B.; Monteiro, F.O.B.; Rodrigues, S.T.; Soares, M.L.; Silva, J.C.R.; das Dores Correia Palha, M.; Biondi, G.F.; Rahal, S.C.; Tourinho, M.M. Ethnoveterinary knowledge and practices at Colares island, Pará state, eastern Amazon, Brazil. J. Ethnopharmacol. 2012, 144, 346–352. [Google Scholar] [CrossRef]
- Arhoghro, E.M.; Kpomah, D.E.; Uwakwe, A.A. Curative Potential of Aqueous Extract of Lemon Grass (Cymbopogon Citratus) on Cisplatin Induced Hepatotoxicity in Albino Wistar Rats. J. Physiol. Pharmacol. Adv. 2012, 2, 282–294. [Google Scholar]
- Metuge, J.A.; Babiaka, S.B.; Mbah, J.A.; Ntie-Kang, F.; Ayimele, G.A.; Cho-Ngwa, F. Anti-onchocerca Metabolites from Cyperus articulatus: Isolation, In Vitro Activity and In Silico ‘Drug-Likeness’. Nat. Prod. Bioprospect. 2014, 4, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Rosselli, S.; Bruno, M.; Raimondo, F.M.; Spadaro, V.; Varol, M.; Koparal, A.T.; Maggio, A. Cytotoxic Effect of Eudesmanolides Isolated from Flowers of Tanacetum vulgare ssp. siculum. Molecules 2012, 17, 8186–8195. [Google Scholar] [CrossRef] [PubMed]
- Botsaris, A.S. Plants used traditionally to treat malaria in Brazil: The archives of Flora Medicinal. J. Ethnobiol. Ethnomed. 2007, 3, 18. [Google Scholar] [CrossRef] [PubMed]
- Craveiro, A.A.; Fernandes, A.G.; Andrade, C.H.S.; Matos, F.J.A.; Alencar, J.W. Óleos essenciais de canelas silvestres regionais. Cienc. Cult. 1977, 29, 445. [Google Scholar]
- Massoud, A.; El Sisi, S.; Salama, O.; Massoud, A. Preliminary study of therapeutic efficacy of a new fasciolicidal drug derived from Commiphora molmol (MYRRh). Am. J. Trop. Med. Hyg. 2001, 65, 96–99. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Elmorshedy, H.; Tanner, M.; Bergquist, R.N.; Sharaf, S.; Barakat, R. Prophylactic effect of artemether on human schistosomiasis mansoni among Egyptian children: A randomized controlled trial. Acta Trop. 2016, 158, 52–58. [Google Scholar] [CrossRef]
- Mbanzulu, K.M.; Zanga, J.K.; Mukendi, J.P.K.; Mbaya Ntita, F.; Matangila, J.R.; Muhindo, H.M.; Mpoyi Wa Mpoyi, S.; Aloni, M.N.; Wumba, R. The Impact of Artesunate-Amodiaquine on Schistosoma mansoni Infection among Children Infected by Plasmodium in Rural Area of Lemfu, Kongo Central, Democratic Republic of the Congo. Biomed Res. Int. 2018, 2018, 3487183. [Google Scholar] [CrossRef]
- Tallmadge, E.H. Patenting Natural Products after Myriad. Harv. J. Law Technol. 2017, 30, 569–600. [Google Scholar]
- Hood, J.J.D. Essential Oil of Kunzea ambigua and Methods of Use. Australia Patent AU2008241370B2, 26 June 2014. [Google Scholar]
- Hood, J.J.D. Essential Oil of Kunzea ambigua and Methods of Use. European Patent EP2192912B1, 1 June 2016. [Google Scholar]
- Park, C.G.; Jang, M.; Shin, E.; Kim, J. Myrtaceae plant essential oils and their β-triketone components as insecticides against drosophila suzukii. Molecules 2017, 22, 1050. [Google Scholar] [CrossRef]
- Thomas, J.; Webb, C.E.; Narkowicz, C.; Jacobson, G.A.; Peterson, G.M.; Davies, N.W.; Russell, R.C. Evaluation of repellent properties of volatile extracts from the australian native plant kunzea ambigua against aedes aegypti (diptera: Culcidae). J. Med. Entomol. 2009, 46, 1387–1391. [Google Scholar] [CrossRef] [PubMed]
- Bon, K.K.; Chul, L.H. Nematicidal Composition Contaning Geraniol. KR101862202B1, 29 May 2018. [Google Scholar]
- Kumaran, A.M.; D’Souza, P.; Agarwal, A.; Bokkolla, R.M.; Balasubramaniam, M. Geraniol, the putative anthelmintic principle ofCymbopogon martinii. Phyther. Res. 2003, 17, 957. [Google Scholar] [CrossRef]
- Gwon, P., II; Sang-cheol, S.; Sang-gil, L. Composition of Nematocides Comprising Plant Essential Oils. KR20090093604A, 3 June 2010. [Google Scholar]
- Fabbri, J.; Maggiore, M.A.; Pensel, P.E.; Denegri, G.M.; Gende, L.B.; Elissondo, M.C. In vitro and in vivo efficacy of carvacrol against Echinococcus granulosus. Acta Trop. 2016, 164, 272–279. [Google Scholar] [CrossRef]
- Xiaohui, W.; Xiao, H.; Gangzhong, S.; Huamei, L.; Yonghua, G.; Rui, H.; Bo, Z.; Haiyan, J. Pesticide for Controlling Nematodes. CN107372644B, 22 November 2019. [Google Scholar]
- Albright, R.B.; Lawrence, M.C.; Sabnis, S.S.; Ranjan, S. Benzimidazole Anthelmintic Compositions. Australia Patent AU2009245834B2, 14 November 2013. [Google Scholar]
- Albright, R.B.; Lawrence, M.C.; Sabnis, S.S.; Ranjan, S. Benzimidazole Anthelmintic Compositions. Canada Patent CA2737102C, 18 February 2014. [Google Scholar]
- Albright, R.B.; Lawrence, M.C.; Sabnis, S.S.; Ranjan, S. Benzimidazole Anthelmintic Compositions. European Patent EP2331068B1, 4 January 2017. [Google Scholar]
- Albright, R.B.; Lawrence, M.C.; Sabnis, S.S.; Ranjan, S. Benzimidazole Anthelmintic Compositions. U.S. Patent US9283176B, 15 March 2016. [Google Scholar]
- Green, W.A.; Strobel, G.A. Compositions of Volatile Organic Compounds and Methods of Use Thereof. Canada Patent CA2757537C, 8 January 2019. [Google Scholar]
- Green, W.A.; Strobel, G.A. Compositions of Volatile Organic Compounds and Methods of Use Thereof. European Patent EP2413692B1, 29 August 2018. [Google Scholar]
- Green, W.A.; Strobel, G.A. Compositions of Volatile Organic Compounds and Methods of Use Thereof. U.S. Patent US8968798B2, 3 March 2015. [Google Scholar]
- Nixon, S.A.; Welz, C.; Woods, D.J.; Costa-Junior, L.; Zamanian, M.; Martin, R.J. Where are all the anthelmintics? Challenges and opportunities on the path to new anthelmintics. Int. J. Parasitol. Drugs Drug Res. 2020, 14, 8–16. [Google Scholar] [CrossRef] [PubMed]
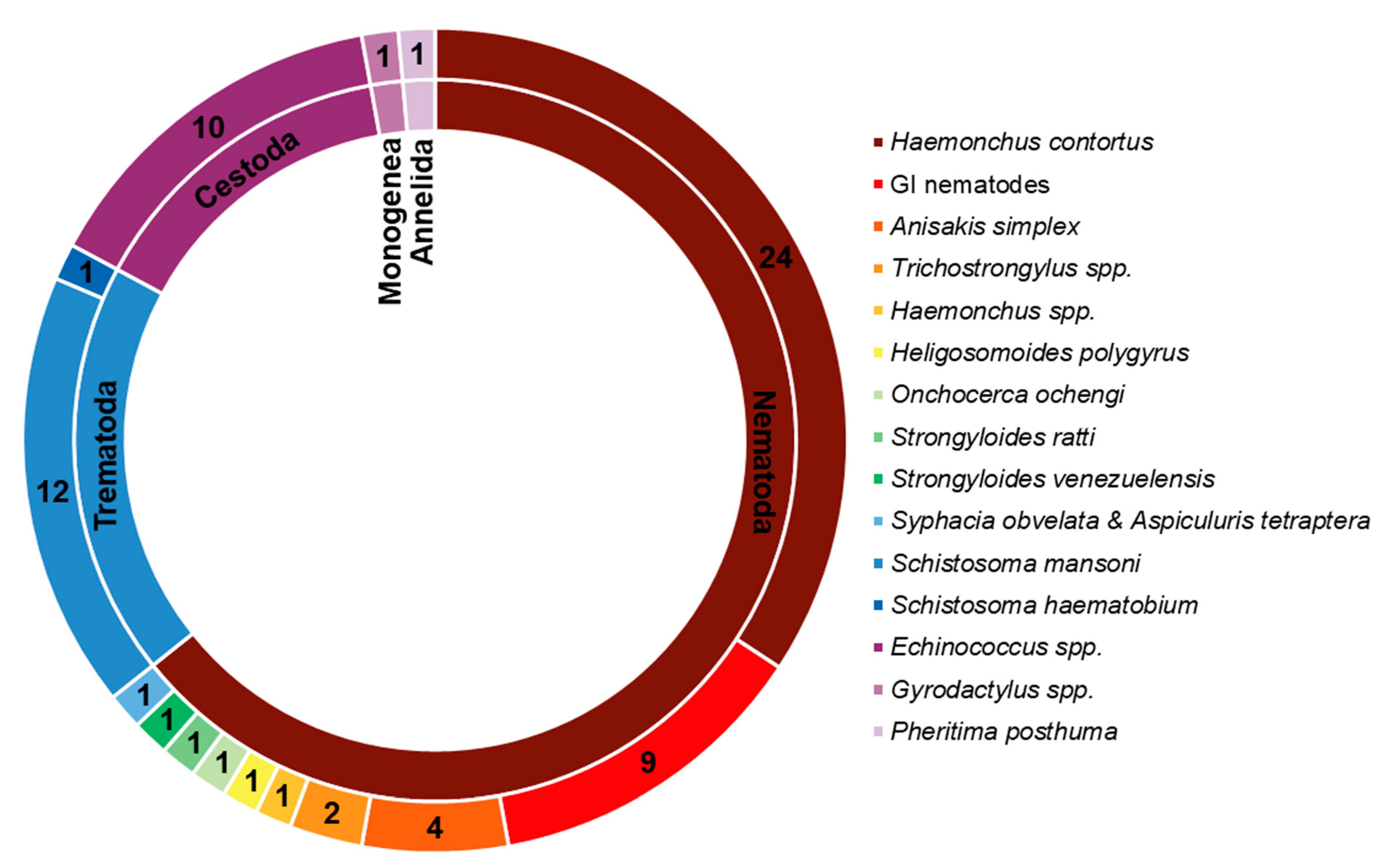
| Name of the Essential Oil | Major Active Component(s) | Parasite Model | Host | In Vivo | Effect/ Reduction/ Mortality | Assay | References |
|---|---|---|---|---|---|---|---|
| Artemisia campestris L. | β-Pinene (36.4%), 2-Undecanone (14.7%) and Limonene (10.57%) | HP | Mice | 5000 mg/kg | 72% | FECRA | [129] |
| Artemisia campestris | β-Pinene (36.4%), 2-Undecanone (14.7%) and Limonene (10.57%) | HP | Mice | 5000 mg/kg | 72% | WB | [129] |
| * Croton zehntneri Pax and K.Hoffm. | Anethole (63.88%) and Estragole (21.84%) | SOAT | Albino Swiss mice | 800 mg/kg | 11.64 ± 35.0% | WB | [130] |
| * Cymbopogon citratus (DC.) Stapf | 3,7-Nonadien-2-one, 4,8-dimethyl (24.86%), Geranial (18.98%) and Neral (17.77%) | GIN | Sheep | 500 mg/kg | 46.90% | WB | [131] |
| Cymbopogon citratus | Geranial (57.3%) and Neral (40.4%) | Hc | Gerbil | 800 mg/kg | 38.60% | WB | [132] |
| Cymbopogon schoenanthus (L.) Spreng. | Geraniol (59.42%) and Geranial (13.49%) | Hc | Sheep | 180 mg/kg | 97.50 ± 0.66% | EHA | [133] |
| Cymbopogon schoenanthus | Geraniol (59.42%) and Geranial (13.49%) | Hc | Sheep | 180 mg/kg | 93.33 ± 1.99% | LDA | [133] |
| Corymbia citriodora (Hook.) K.D.Hill and L.A.S.Johnson (Synonym: Eucalyptus citriodora Hook.) | Citronellal (63.94%) | GIN | Sheep | 500 mg/kg | 41.8%/69.5% | FECRA | [134] |
| Eucalyptus citriodora | Citronellal (67.5%) | GIN | Sheep | 500 mg/kg | 55.9%/34.5% | FECRA | [135] |
| Eucalyptus citriodora | β-Citronellal (71.77%) | GIN | Goat | 500 mg/kg | 66.25/60.34/58.45% | FECRA | [136] |
| Eucalyptus staigeriana F.Muell. ex F.M.Bailey | Limonene (72.91%) | GIN | Sheep | 365 mg/kg | 60.79% | WB | [137] |
| Eucalyptus staigeriana | Limonene (28.82%), E-Citral (14.16%) and Z-Citral (10.77%) | Hc | Goat | 500 mg/kg | 61.4/76.57/73.66% | FECRA | [138] |
| * Lippia sidoides Cham. | Thymol (59.65%) and (E)-Caryophyllene (10.60%) | SOAT | Albino Swiss mice | 1600 mg/kg | 68.94 ± 15.1% | WB | [130] |
| Lippia sidoides | Thymol (59.65%) and (E)-Caryophyllene (10.60%) | Ts | Sheep | 200 mg/kg | 63.6 ± 10.2% | WB | [139] |
| Lippia sidoides | Thymol (59.65%) and (E)-Caryophyllene (10.60%) | Hs | Sheep | 283 mg/kg | 56.9 ± 10.7% | WB | [139] |
| Melaleuca alternifolia (Maiden and Betche) Cheel | Terpinen-4-ol (41.98%), γ-Terpinene (20.15%) and α-terpinene (9.85%) | Hc | Gerbil | 0.75 mL/kg | 46.36% | WB | [140] |
| * Thymus vulgaris L. | Thymol (50.22%) and para-Cymene (23.76%) | Hc | Sheep | 300 mg/kg | / | FECRA | [141] |
| Name of the Essential Oil | Major Active Constituents | Parasite Model | In Vitro | Effect/Reduction/Mortality | Assay | References |
|---|---|---|---|---|---|---|
| Ageratum conyzoides (L.) L. | Precocene I (74.30%) and (E)-Caryophyllene (14.23%) | Sm | 100 µg/mL | 25%/0% | MA | [142] |
| Alpinia zerumbet (Pers.) B.L.Burtt and R.M.Sm. | 1,8-Cineole (24.69%), p-Cymene (22.56%) and 4-Terpineol (17.43%) | Hc | 3880 (2940–5090) µg/mL | EC50 | LDA | [143] |
| Alpinia zerumbet | 1,8-Cineole (24.69%), p-Cymene (22.56%) and 4-Terpineol (17.43%) | Hc | 940 (670–1280) µg/mL | EC50 | EHA | [143] |
| Anethum graveolens L. | Dihydrocarvone (39.1%), Carvone (22.24%) and D-Limonene (16.84%) | Hc | 6 µg/mL | IC50 | EHA | [144] |
| * Anthemis aaronsohnii Eig (Synonym: Anthemis nobilis L.) | Isobutyl angelate (29.26%) and Isoamyl angelate (15.27%) | Hc | 117 µg/mL | IC50 | LDA | [145] |
| Arisaema franchetianum Engl. | Linalool (8.89%) | Hc | 1100 (940–1270) µg/mL | CE50 | LDA | [146] |
| Arisaema lobatum | Carvacrol (7.05%) | Hc | 480 (390–570) µg/mL | CE50 | LDA | [146] |
| Artemisia lancea Vaniot | 1,8-Cineole (34.56%) and Camphor (16.65%) | Hc | 1430 (1040–1840) µg/mL | LC50 | LMGA | [147] |
| Artemisia vulgaris L. | Caryophyllene (37.45%), Germacrene D (16.17%) and Humulene (13.66%) | Hc | 1200 µg/mL | LC50 | LMGA | [148] |
| Baccharis dracunculifolia DC. | (E)-Nerolidol (33.51%) and Spathulenol (16.24%) | Sm | 10 µg/mL | 100% | MA | [149] |
| Baccharis trimera (Less.) DC. | Germacrene D (15.31), trans-Caryophyllene (14.77%) and Bicyclogermacrene (14.67%) | Sm | 130 µg/mL | 80% | MA | [150] |
| Bunium persicum (Boiss.) B.Fedtsch. | γ-Terpinene (46.1%) and Cuminaldehyde (15.5%) | Es | 25 µL/mL | 100/100% | PVA | [151] |
| * Citrus aurantifolia (Christm.) Swingle | Limonene (56.37%), β-Pinene (11.86%) and γ-Terpinene (11.42%) | Hc | 44 µg/mL | IC50 | LDA | [145] |
| Citrus limon (L.) Osbeck | Limonene (29.9%) and β-Pinene (12.0%) | Sm | 81.7 µg/mL | LC50 | MA | [152] |
| Coriandrum sativum L. | β-Linalool (73.21%) | Hc | 2890 (2600–3200) µg/mL | EC50 | LDA | [143] |
| * Cosmos sulphureus Cav. | 2,6-di-tert-butyl-4-methylphenol (44.98%), Germacrene D (33.70%) and β-Caryophyllene (10.23%) | Sm | 100 µg/mL | 75%/50% | MA | [153] |
| Curcuma longa L. | β-Turmerone (21.8%), Ar-Turmerone (14.7%) and α-Turmerone (12.4%) | Es | 200 µL/mL | 100% | PVA | [154] |
| Cymbopogon martini (Roxb.) W.Watson | Geraniol (81.4%) and Isomenthyl isomenthyl acetate (10.1%) | Ts | 130 (110–140) µg/mL | CL50 | EHA | [155] |
| Cymbopogon martinii | Geraniol (69.63%) | Pp | 20,000 µg/mL | 3.21 ± 0.31 min | MA | [156] |
| * Dysphania ambrosioides (L.) Mosyakin and Clemants | cis-Piperitone oxide (35.2%), para-Cymene (14.5%) and trans-Isoascaridole (14.1%) | Sm | 6.50 ± 0.38 µg/mL | LC50 | MA | [157] |
| Ferula gummosa Boiss. | β-Pinene (57.0%) and β-Acorenone (11.4%) | Es | 17.18 µg/mL | LC50 | PVA | [158] |
| * Foeniculum vulgare Mill. | (E)-Anethole (69.8%) and Limonene (22.5%) | Sm | 100 µg/mL | 50 ± 25%/50 ± 25% | MA | [159] |
| * Lavandula angustifolia Mill. (Synonym: Lavandula officinalis Chaix) | Linalool acetate (35.97%) and trans-Sabinene hydrate (29.17%) | Hc | 280 µg/mL | IC50 | LDA | [145] |
| Mentha piperita | Menthol (42.5%) and Menthone (27.4%) | Ts | 260 (230–300) µg/mL | CL50 | LDA | [155] |
| * Mentha x villosa Huds. | Rotundifolone (70.96%) | Sm | 100 µg/mL | 100% | MA | [160] |
| Myrtus communis L. | α-Pinene (24.7%), 1,8-Cineole (19.6%) and Linalool (12.6%) | Es | 100 µL/mL | 100% | PVA | [161] |
| * Newbouldia laevis (P.Beauv.) Seem. | β-Caryophyllene (36.08%) | Sr | 51.7 ± 7.7 µg/mL | IC50 | LMGA | [162] |
| Nigella sativa L. | Thymoquinone (42.4%), para-Cymene (14.1%) and Carvacrol (10.3%) | Es | 100 µg/mL | 21.6%/76.6% | PVA | [163] |
| Ocimum gratissimum L. | Eugenol (43.7%) and 1,8-Cineole (32.71%) | Hc | 0.5vol% | 100.0 ± 6.13% | EHA | [164] |
| Origanum compactum Benth. | Carvacrol (50.32%), Thymol (14.8%) and γ-terpinene (13.6%) | As | 429 µg/mL | LD50 | MA | [165] |
| Origanum compactum | Carvacrol (59.1%) and para-Cymene (11.7%) | Sh | 1 µg/mL | 100% | MA | [166] |
| * Origanum syriacum L. | Carvacrol (82.6%) | As | 87 µg/mL | LC50 | LMA | [167] |
| Pelargonium radens H.E.Moore | Citronellol (37.7%), Geraniol (17.6%) and Citronellyl formate (11.0%) | Es | 8.52 µg/mL | LC50 | PVA | [158] |
| * Pinus nigra subsp. pallasiana (Lamb.) Holmboe | α-Pinene (27.46%) and β-Caryophyllene (11.03%) | Es | 10,000 µg/mL | 61.69%/81.76% | PVA | [168] |
| Piper aduncum L. | Dillapiole (76.2%) | Hc | 100 (90–110) µg/mL | IC50 | LDA | [169] |
| Plectranthus neochilus Schltr. | β-Caryophyllene (28.23%), α-Pinene (12.63%) and α-Thujene (12.22%) | Sm | 100 µg/mL | 100%/100% | MA | [170] |
| Rosmarinus officinalis L. | 1,8-Cineole (42.11%), 2-Bornanone (16.37%) and α-Pinene (14.76%) | GINS | 7100 µg/mL | 97.40% | EHA | [171] |
| Ruta chalepensis L. | 2-Nonanone (25.31%), 2-Undecanone (24.01%) and Limonene (12.82%) | GINS | 1290 ± 1100 µg/mL | EC50 | LMGA | [172] |
| * Satureja khuzistanica Jamzad | Carvacrol (94.97%) | Es | 10,000 µg/mL | 100.00% | PVA | [173] |
| Tagetes minuta L. | Piperitone (86.27%) and Limonene (13.73%) | Hc | 1670 (1020–2530) µg/mL | EC50 | LDA | [143] |
| Tagetes patula L. | Piperitenone (23.5%) and Piperitone (20.1%) | Hc | 40 (35–44) µg/mL | LC50 | LDA | [174] |
| * Tanacetum vulgare L. | β-Thujone (84.13%) | Sm | 200 µg/mL | 100% | MA | [175] |
| Tetradenia riparia (Hochst.) Codd | Fenchone (18.9%), (E,E)-Farnesol (17.7%) and Aromadendrene oxide (17.3%) | Sm | 100 µg/mL | 100% | MA | [176] |
| Trachyspermum ammi (L.) Sprague | Thymol (50.07%), γ-Terpinene (23.92%) and para-Cymene (22.9%) | Es | 10,000 µg/mL | 100.00% | PVA | [177] |
| * Zanthoxylum bungeanum Maxim. | Borneol (18.61%) and β-Elemene (10.87%) | Hc | 3980 (2890–5310) µg/mL | LC50 | EHA | [178] |
| * Zanthoxylum zanthoxyloides (Lam.) Zepern. and Timler | γ-Terpinene (18.0%) and Undecane (14.84%) | Sr | 18.2 ± 0.5 µg/mL | IC50 | EHA | [162] |
| EO/EO Component | Role | Other Components | Publication Number | Application Year | Title | Institution/ Applicant |
|---|---|---|---|---|---|---|
| Cinnamon EO | Synergist | Abamectin B2 | CN 107372644 B | 2017 | Pesticide for controlling nematodes | Wuhan Kernel Biotech Co., Ltd. |
| Geraniol | Main component | Ganghal extract | KR 101862202 B1 | 2016 | Nematicidal composition containing geraniol | Ecowin Co., Ltd.; Kyung-Bon, Koo |
| Basil EO, black pepper EO, (+/−) camphor, carvacrol, trans-cinnamaldehyde, cinnamon leaf EO, cinnamon bark EO, citronellol, citral, (+/−) citronella, clove bud EO, eucalyptol, eucalyptus EO, eugenol, fennel EO, geraniol, ginger EO, jojoba EO, lemongrass EO, limonene, linalool, patchouli EO, peppermint EO, α-terpinene, rosemary EO, tea-tree EO and thyme EO | Additional component | isobutyric acid, isobutyric anhydride, napthalene and/or caryophyllene | CA 2757537 C; EP 2413692 B1; US 8968798 B2 | 2010; 2010; 2010; 2013 | Compositions of volatile organic compounds and methods of use thereof | Synthetic Genomics Inc. |
| 1,8-cineole, 1,4-cineole, eugenol, limonene, tea-tree EO, citronellol | Solvent system | Benzimidazole (triclabendazole) and a lactone solvent (γ-hexalactone, moxidectin) | AU 2009245834 B2; CA 2737102 C; CN 102176899 B; EP 2331068 B1; JP 5547738 B2; KR 101318603 B1; RU 2493825 C2; US 9283176 B2; ZA 201103282 B | 2009; 2009; 2009; 2009; 2009; 2009; 2009; 2009; 2011 | Benzimidazole anthelmintic compositions | Zoetis Services LLC (AU, EU, JP, US) Wyeth LLC (CA, CN, SK, RU) Pah W LLC (ZA) |
| Ajowan EO, styrax EO or ballerina EO; thymol, carvacrol, trans- cinnamyl alcohol and cis-asaron | Main component | KR 100960871 B1 | 2008 | Composition of nematocides comprising plant essential oils | The Republic of Korea (Forestry Administration Forestry Research Institute) | |
| Kunzea ambigua EO | Main component | AU 2008241370 B2; EP 2192912 B1 | 2008; 2008 | Essential oil of Kunzea ambigua and methods of use | Hood, John James David |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panda, S.K.; Daemen, M.; Sahoo, G.; Luyten, W. Essential Oils as Novel Anthelmintic Drug Candidates. Molecules 2022, 27, 8327. https://doi.org/10.3390/molecules27238327
Panda SK, Daemen M, Sahoo G, Luyten W. Essential Oils as Novel Anthelmintic Drug Candidates. Molecules. 2022; 27(23):8327. https://doi.org/10.3390/molecules27238327
Chicago/Turabian StylePanda, Sujogya Kumar, Marijn Daemen, Gunanidhi Sahoo, and Walter Luyten. 2022. "Essential Oils as Novel Anthelmintic Drug Candidates" Molecules 27, no. 23: 8327. https://doi.org/10.3390/molecules27238327
APA StylePanda, S. K., Daemen, M., Sahoo, G., & Luyten, W. (2022). Essential Oils as Novel Anthelmintic Drug Candidates. Molecules, 27(23), 8327. https://doi.org/10.3390/molecules27238327