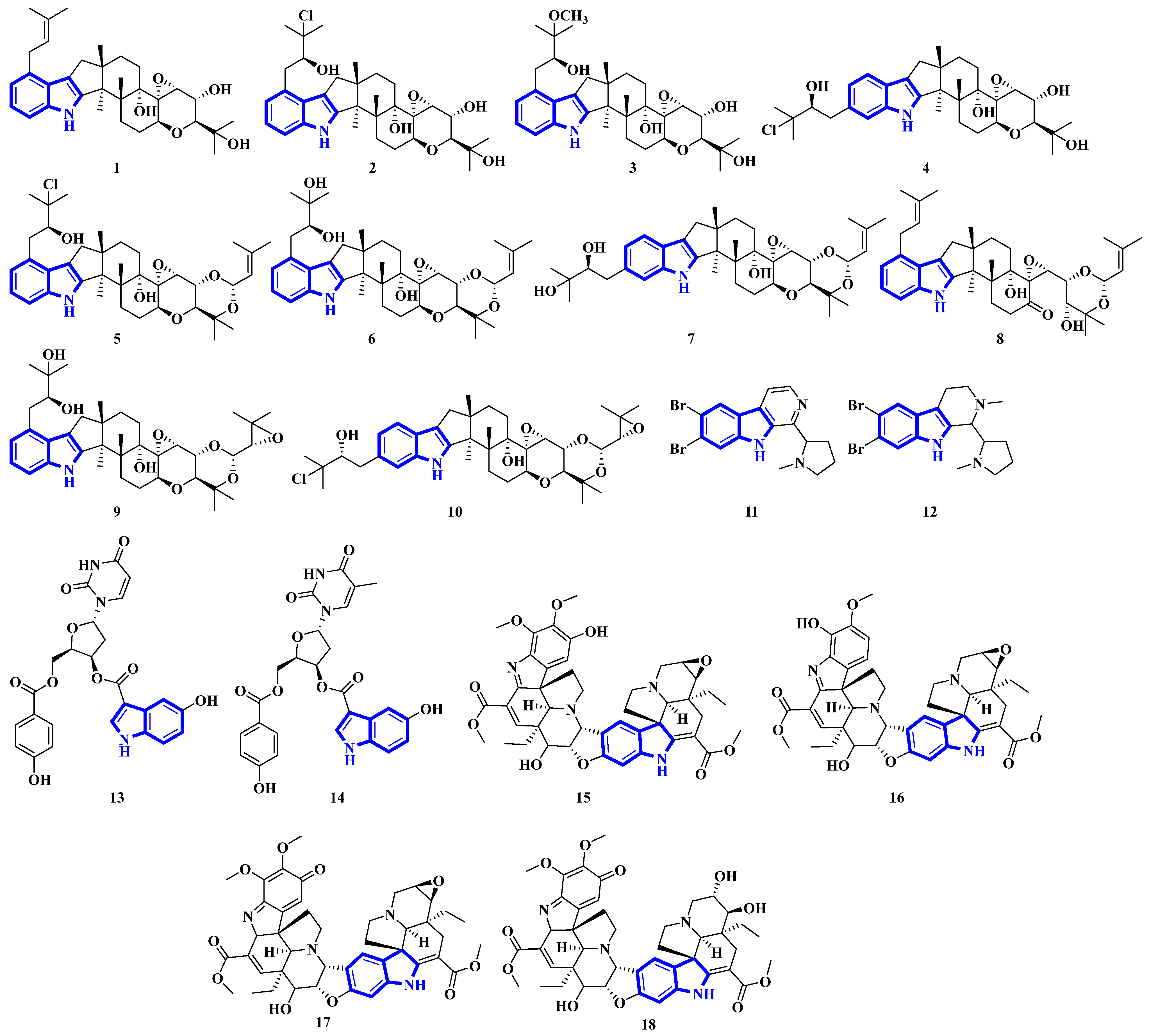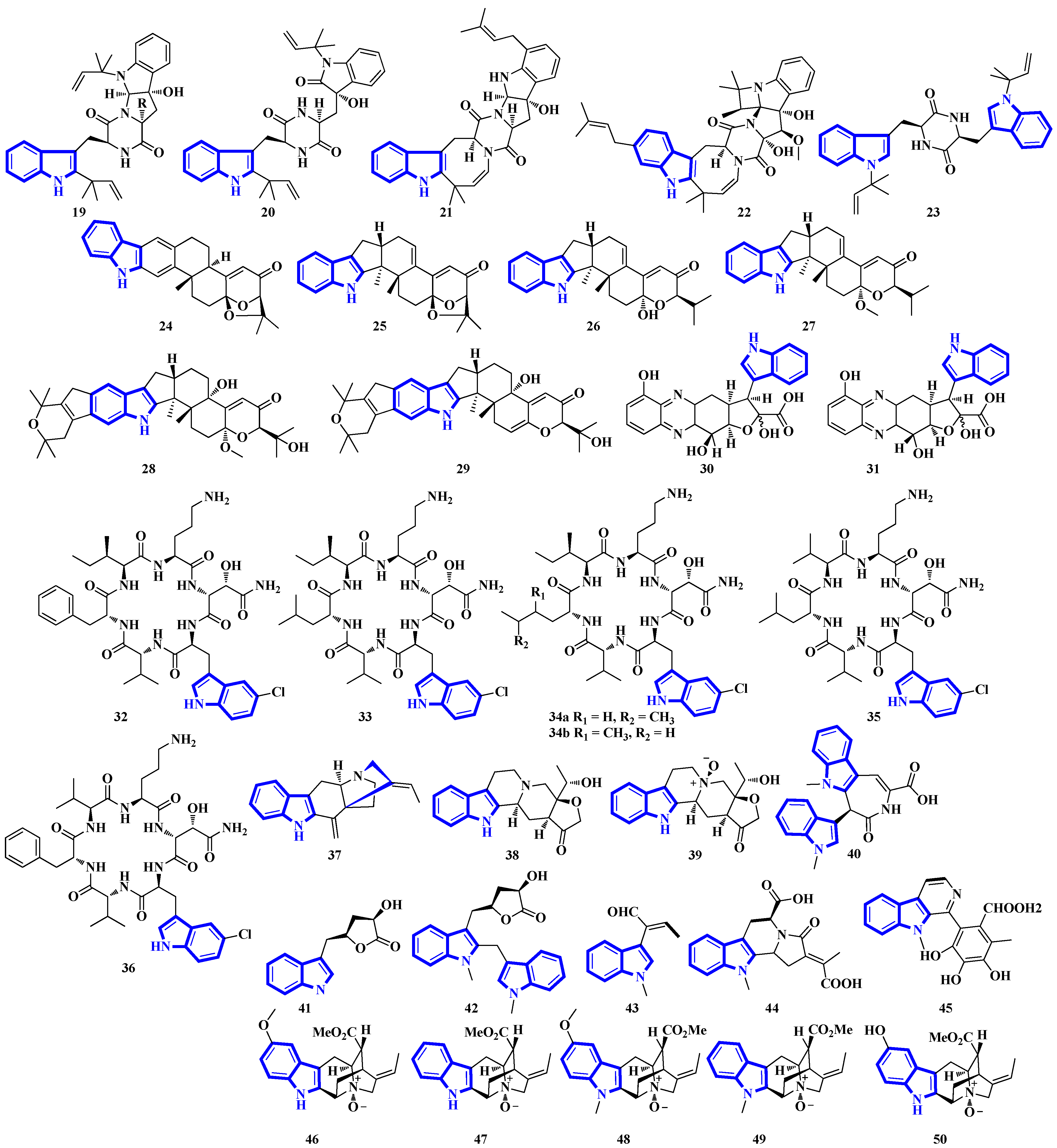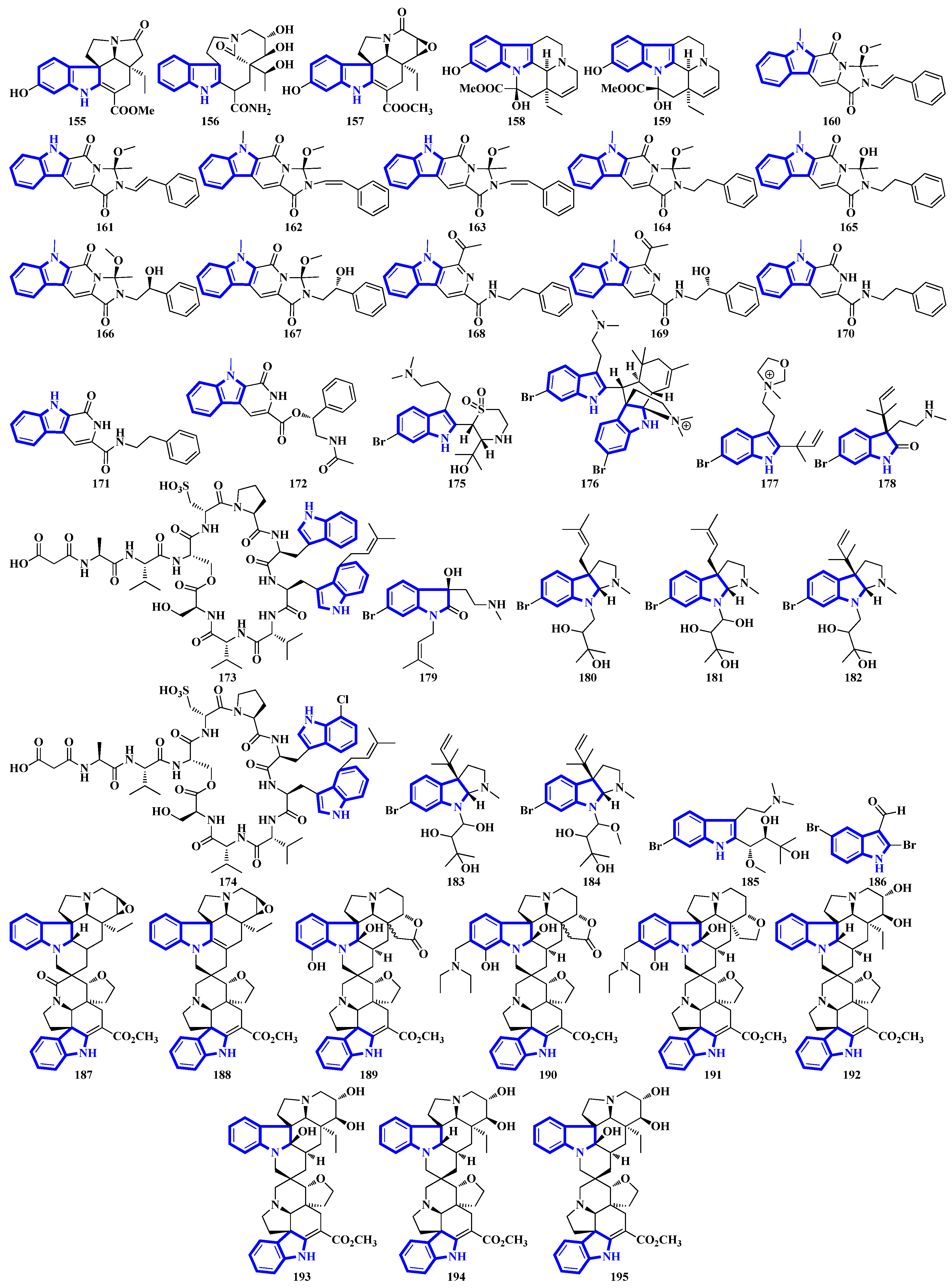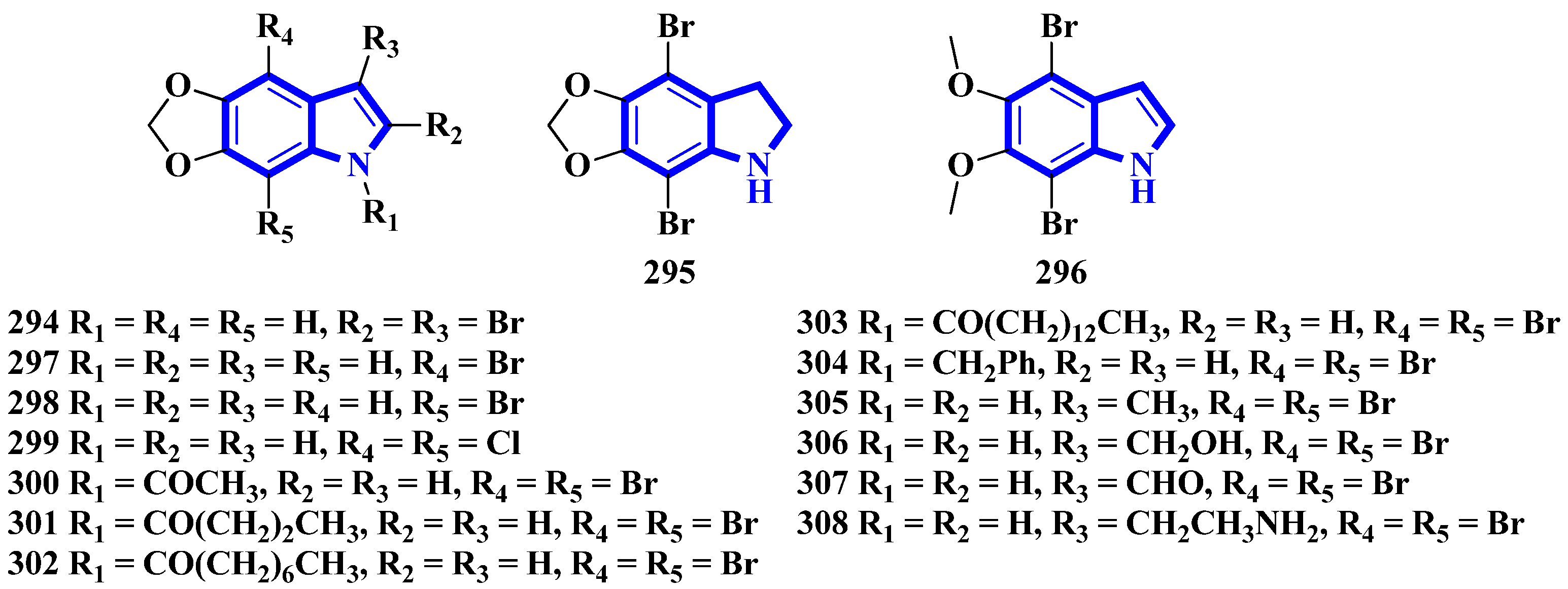Abstract
Indole alkaloids represent a large subset of natural products, with more than 4100 known compounds. The majority of these alkaloids are biologically active, with some exhibiting excellent antitumor, antibacterial, antiviral, antifungal, and antiplasmodial activities. Consequently, the natural products of this class have attracted considerable attention as potential leads for novel therapeutics and are routinely isolated, characterized, and profiled to gauge their biological potential. However, data on indole alkaloids, their various structures, and bioactivities are complex due to their diverse sources, such as plants, fungi, bacteria, sponges, tunicates, and bryozoans; thus, isolation methods produce an incredible trove of information. The situation is exacerbated when synthetic derivatives, as well as their structures, bioactivities, and synthetic schemes, are considered. Thus, to make such data comprehensive and inform researchers about the current field’s state, this review summarizes recent reports on novel indole alkaloids. It deals with the isolation and characterization of 250 novel indole alkaloids, a reappraisal of previously reported compounds, and total syntheses of indole alkaloids. In addition, several syntheses and semi-syntheses of indole-containing derivatives and their bioactivities are reported between January 2019 and July 2022.
Keywords:
indole; natural products; cytotoxic; anticancer; antimicrobial; antiviral; other bioactivities 1. Introduction
Despite the decreasing interest of modern pharmaceuticals in pursuing natural products as leads for new medicine [1], natural products and their derivatives still represent a significant fraction of approved drugs, with more than 22 compounds in the WHO list of essential medicine being sourced exclusively from flowering plants [2]. Furthermore, approximately 40% of all available medicine is either a natural product or a semi-synthetic derivative [3,4]. This ubiquity of natural products in medicine can be explained by their inherent nature as secondary metabolites. These compounds have been fine-tuned over millennia, acquiring biological functions to boost their host’s survivability [5]. Thus, these compounds can regulate endogenous defense mechanisms and interactions with other organisms, explaining their potency as antiviral, antibacterial, and antitumor agents [6]. Natural products also represent desired targets in synthesis due to their structural complexity or potent bioactivity.
Additionally, the pursuit of synthesizing natural products has led to novel reaction methodologies, catalyst design, and natural product-inspired synthetic analogs [7,8]. However, so far, only 5–15% of terrestrial plant species have been investigated as potential sources of therapeutic agents. Similarly, despite accounting for 90% of all-natural diversity, less than 1% of the microbial domain has been explored [1]. This minimal investigation of natural products’ sources (plants, insects, and microorganisms) represents a significant opportunity for chemists to scrutinize these sources and further discover nature’s offerings [9].
Due to their immense structural diversity, natural products are generally grouped into four broad categories: phenolics (phenylpropanoids), polyketides, terpenoids, and alkaloids [10,11]. Alkaloids are nitrogen-containing secondary metabolites, including important therapeutic agents such as hyoscyamine, quinine, or emetine [12,13]. Additionally, alkaloids are further divided into classes based on which nitrogen-containing moiety they possess; tropane, isoquinoline, imidazole, piperidine, and pyrrolizidine alkaloids are some examples [14].
Alkaloids containing indoles—indole alkaloids—are among the most significant alkaloid subsets, with more than 4100 different compounds [15]. Commercially available drugs having the indole moiety are ajmaline (antiarrhythmic agent), physostigmine (used to treat anticholinergic poisoning), and vincristine (antitumor agent) [16,17,18]. Similarly, many controlled substances such as ibogaine, psilocybin, and LSD (all powerful psychedelics) feature an indole moiety, demonstrating the group’s ubiquity in bioactive molecules [19].
This review aims to describe and summarize recent discoveries in indole-containing natural products and hopes to aid researchers by providing all relevant information (isolation method, synthesis, and bioactivity) in one place. For data collection, PubMed was used as the primary database (using “indole” and “natural product” as the key search words). Reports containing natural products with the indole or indoline rings were selected from about the last three and a half years (January 2019 to July 2022), while alkaloids containing structurally similar motifs such as sidelines, hydroindoles, and quinolines are not included. These articles described the isolations of novel indole-containing natural products, reappraisals of the structures of previously reported natural products, the total and semi-synthesis of indole alkaloids, and the syntheses of indole-containing derivatives. Bioactivities of all novel compounds and structure–activity relationship analyses (wherever available) are also included.
The articles reported 354 indole alkaloids, 250 isolated from 24 families, across the groups of living organisms. These alkaloids displayed immense biological activities, with cytotoxicity being the most common, followed by antibacterial, antifungal, antiplasmodial, and antiviral activities. Other bioactivities, including the promotion of glucose uptake, immunomodulatory effects (influencing the behavior of B-cells and DCs), analgesic effects, HDAC inhibition, and RANKL-induced multinuclear osteoclasts inhibition, were also reported. This variety in biological activities is matched by the diversity of structural classes present, with novel indole alkaloids possessing alstonine, aspidosperma, chetomin, corynanthean, epicoccin, eudistomin, fumiquinazoline, ibogaine, izumiphenazine A, macroline, notoamide, okaramine, paxilline, pleiocarpamine, sarpagine, serpentine, ursane, vincamine, and vobasine types of structures. Simultaneously, the article presents unique motifs and new classes, such as the psammocindoles.
2. Isolation of Novel Indole Alkaloids
2.1. Source Analysis
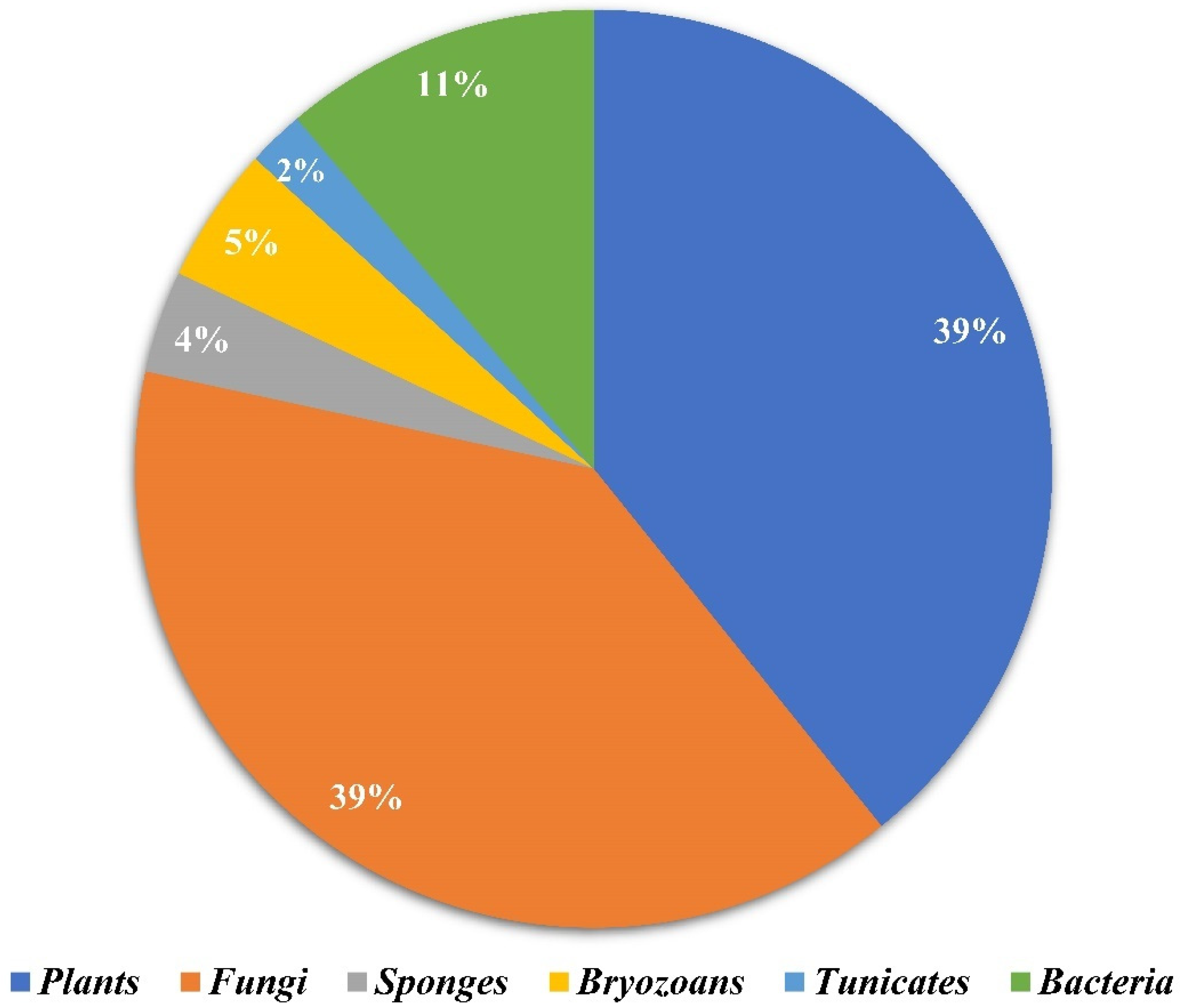
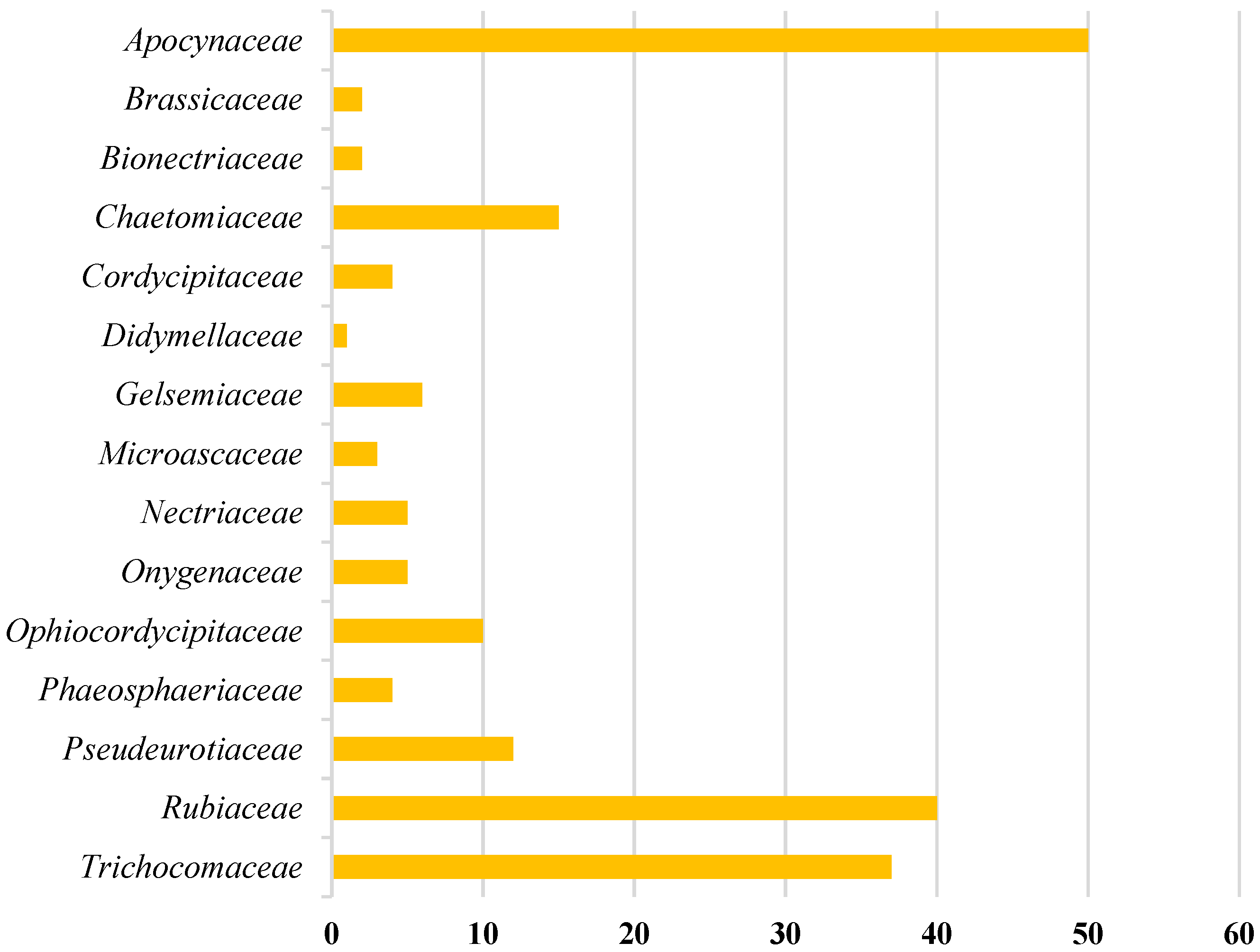
Amongst 250, 98 of the compounds isolated (representing 39% of the total, Figure 1) were sourced exclusively from plants and trees (Apocynaceae, Rubiaceae, Gelsemiaceae, and Brassicaceae families; Figure 2). Similarly, 98 indole alkaloids were isolated from various fungi, with 37 of them being sourced from the Trichocomaceae family alone. Other fungal families such as Chaetomiaceae, Pseudeurotiaceae, and Ophiocordycipitaceae were sources of 15, 12, and 10 novel indole alkaloids, respectively (Figure 2). On the other hand, only 28 alkaloids were isolated from those in the Pseudomonadaceae and Streptomycetaceae families, producing 13 and nine, respectively. Other sources include bryozoans (Flustridae and Vesiculariidae), marine sponges (Coelosphaeridae, Irciniidae, and Petrosiidae), and tunicates (Polyclinidae, Polycitoridae, Didemnidae, and Pseudodistoma) from which 12, nine, and five indole alkaloids, respectively, were isolated (Figure 1).
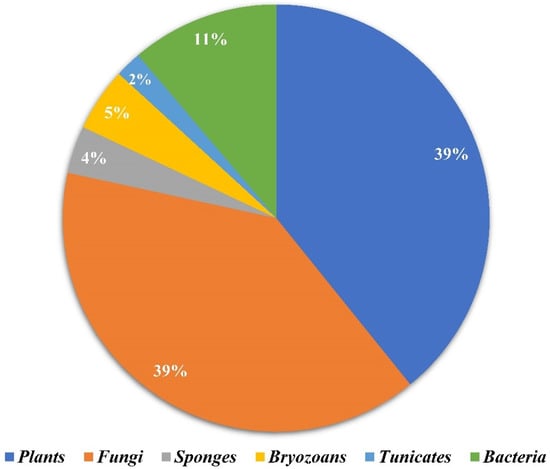
Figure 1.
Distribution of the sources of novel indole alkaloids.
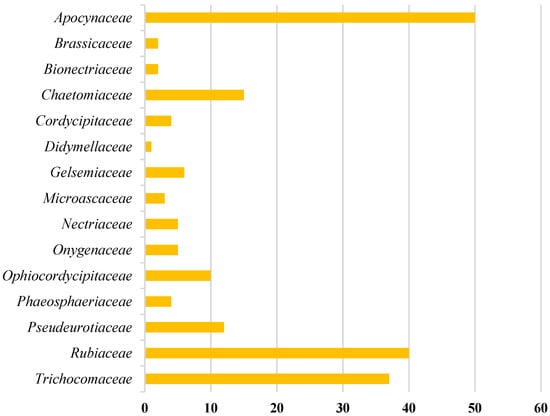
Figure 2.
Distribution of indole alkaloids across plant, tree, and fungal sources.
2.2. Isolation Methods
The isolation methods generally included using organic solvents (dichloromethane, ethanol, ethyl acetate, or methanol) to extract a crude mixture of alkaloids directly from the source (bacteria, fungi, dried plant material, etc.). This mixture was then concentrated and resuspended in acidic media (acidified using HCl, H2SO4, or tartaric acid). Next, the organic layer containing fats and other dissolved material was removed, while the aqueous layer was isolated and basified using NH3, NH4OH, or NaOH. Subsequently, the organic layer was concentrated and purified by column chromatography, affording pure alkaloids.
2.3. Structural Novelties
Of the 250 novel indole alkaloids isolated and analyzed (Table 1), 207 of them were monoindole alkaloids (MIAs), 41 were bisindoles, and only two were trisindoles (238 and 239). Similarly, 40 compounds were monoterpenoids, and 19 were diterpenoids. Some of the alkaloids discovered had structures like or derived from alstonine, aspidosperma, chetomin, corynanthean, epicoccin, eudistomin, fumiquinazoline, ibogaine, izumiphenazine A, macroline, notoamide, okaramine, paxillin, pleiocarpamine, sarpagine, serpentine, ursane, vincamine, and vobasine. In addition, novel compounds such as a long polypeptide bearing a C-terminus tryptamine (63), two oxindole alkaloid glycosides (92 and 93), an oxazolidinium-containing indole found in nature (177), and a methylenedioxy dibromoindole alkaloid with antiosteoporosis activity (233) were isolated.

Table 1.
Sources and isolation procedures of novel indole alkaloids (1–250).
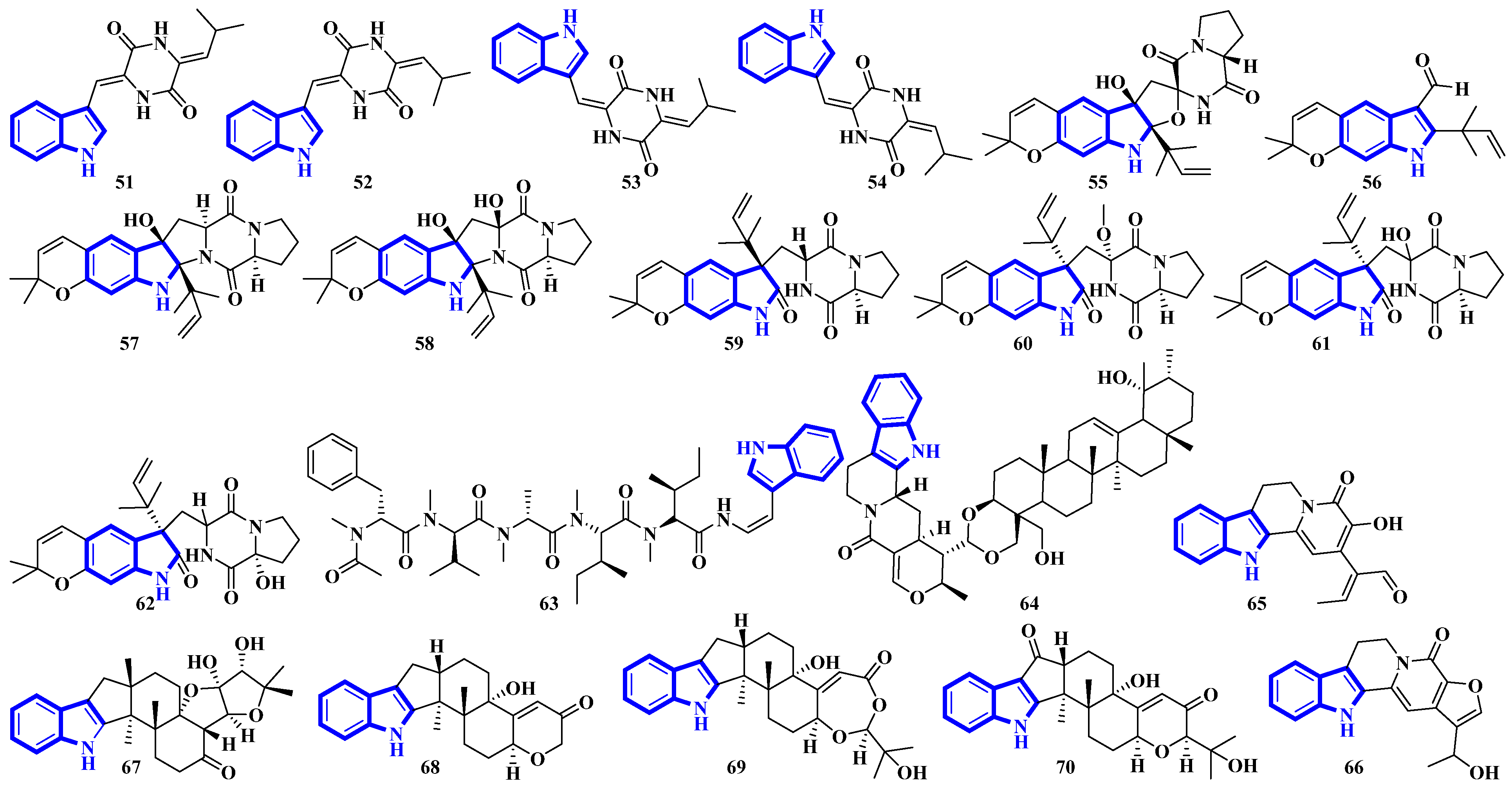
Compounds 1 to 10 are prenylated indole diterpenoids, with chlorinated analogs (2, 4, 5, and 10) being the first indole diterpenes isolated from fungi. These alkaloids resemble terpendole I and terpendole C, themselves biosynthesized from an alkaloid, paspaline [20]. Compounds 11 and 12 are eudistomin-type alkaloids isolated from the tunicate families Eudistoma and Pseudodistoma, whereas compounds 13 and 14 are novel nucleosides isolated from Didemnidae, also a tunicate [21]. Lastly, compounds 15 and 16 are bisindole alkaloids, while compounds 17 and 18 are MIAs, with all four being isolated from the leaves of Tabernaemontana corymbosa (Figure 3) [22].
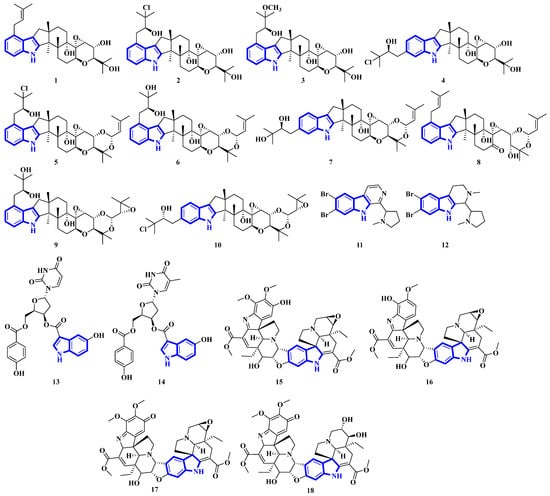
Figure 3.
Novel indole alkaloids 1 to 18.
Compounds 19 to 23 were isolated from Aphanoascus fulvescens, obtained from goose dung, and represent okaramine-type bisindoles (Figure 4) [23]. Compounds 24 to 29 are indole diterpenoids in which compound 24 is a new scaffold with a rare carbazole unit forming a 6/5/6/6/6/6/5 heterocyclic system bearing an aromatic ring C [24]. Compounds 30 and 31 are izumiphenazine A-type alkaloids containing a novel 5-hydroxyquinoxaline and alpha-keto acid motif [25]; however, compounds 32 to 36 are chlorinated cyclic hexapeptides. Unfortunately, the structure of compound 34 could not be completely resolved since minute amounts of it were isolated (34a and 34b) [26]. Compounds 37 to 39 were isolated from the stem bark of Kopsia arborea. Compound 37 is an unusual pentacyclic monoterpenoid alkaloid with a new carbon–nitrogen skeleton. Furthermore, compounds 38 and 39 are pentacyclic corynanthean-type alkaloids that contain a hydroxyethyl-substituted tetrahydrofuranone ring [27]. Compounds 40 and 42 are bisindoles, with the former having a β-carboline motif. Compounds 44 and 45 are demethylated derivatives of chaetogline A and F (demethylation on 19-O and 20-O, respectively) [28]. Compounds 46 to 50 were isolated using the Computer-Assisted Natural Products Anticipation (CANPA) workflow. It is the first nonpeptidic molecular networking-based natural product chemistry workflow. It generated the targeted structures of the alkaloids even before their isolations. Additionally, compounds 46 to 50 are sarpagine-type N-oxide alkaloids [29].
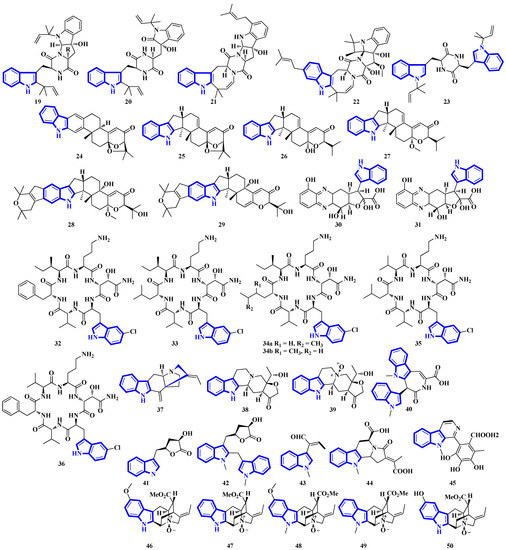
Figure 4.
Novel indole alkaloids 19 to 50.
Compounds 51 to 54 are unsaturated diketopiperazine derivatives [30], and 55 to 62 are linearly fused prenylated indole alkaloids (Figure 5). Compound 55 is the first diketopiperazine derived from d-proline and l-tryptophan and possesses an unprecedented C-11-spiro-fused 6/6/5/5/6/5 ring system. Similarly, compound 56 is the first linearly fused 6/6/5 tricyclic prenylated indole alkaloid discovered [31]. Compound 63 is a highly modified linear hexapeptide with an unusual per methylation of the amino-acid backbone. It is the most extended polypeptide with a tryptamine C-terminus and displays acetylation on its N-terminus, a rare feature among natural and fungal products [32]. Alkaloids 64 to 66 were isolated from the heartwood of Nauclea latifolia. Compound 64 contains an unusual monoterpene indole alkaloid unit condensed with an ursane-type pentacyclic triterpenoid motif that is further bonded to a six-membered-ring acetal; furthermore, monoterpenoid 66 is a constitutional isomer of compound 65 [33]. Compounds 67 to 70 were isolated from a Penicillium fungus extracted from Meretrix lusoria, a bivalve mollusk (Figure 5). Compound 67 is an indolediterpenoid with a unique 6/5/5/6/6/5/5 heptacyclic ring system. Furthermore, compounds 68 and 69 are paxilline-type alkaloids with some modifications; compound 68 contains three fewer carbons (C-23/24/25), while compound 69 contains an additional oxygen atom between C-21 and C-22 than paxilline. Compound 69 also has an unusual 6/5/5/6/6/7 hexacyclic ring system with a 1,3-dioxepane ring, a rarity among natural products [34].
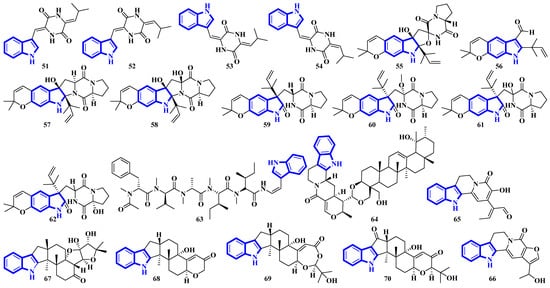
Figure 5.
Novel indole alkaloids 51 to 70.
Bisindoles 71 to 77 were isolated from the stem bark of Alstonia penangiana (Figure 6). Compound 77 is a macroline-pleiocarpamine-type alkaloid, and compound 74 is a constitutional isomer of compound 73. Compounds 75 and 76 exist as an equilibrium mixture of their ring-opened and hemiketal (75a and 76a) states. The hemiketal versions are more prevalent in their solid forms, while dissolving the alkaloids in CDCl3 yields ring-opened forms [35]. Compounds 78 and 79 were isolated from the leaves of Uncaria longiflora; nonetheless, compounds 80 to 88 and 89 to 98 were isolated from the hooks and hook-bearing stems of Uncaria rhynchophylla, respectively. Interestingly, all 21 alkaloids were monoterpenoids, with compounds 89 to 98 containing gluco-conjugated motifs [36,37]. Furthermore, compound 89 is a new subtype of oxindole alkaloids with a seven-membered D-ring; compound 98 contains a rare glucosyl moiety at C-9. Compounds 92 and 93 represent the first two oxindole alkaloid diglycosides reported [38].
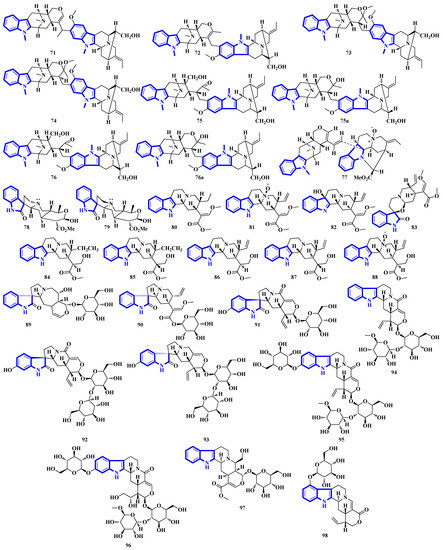
Figure 6.
Novel indole alkaloids 71 to 98.
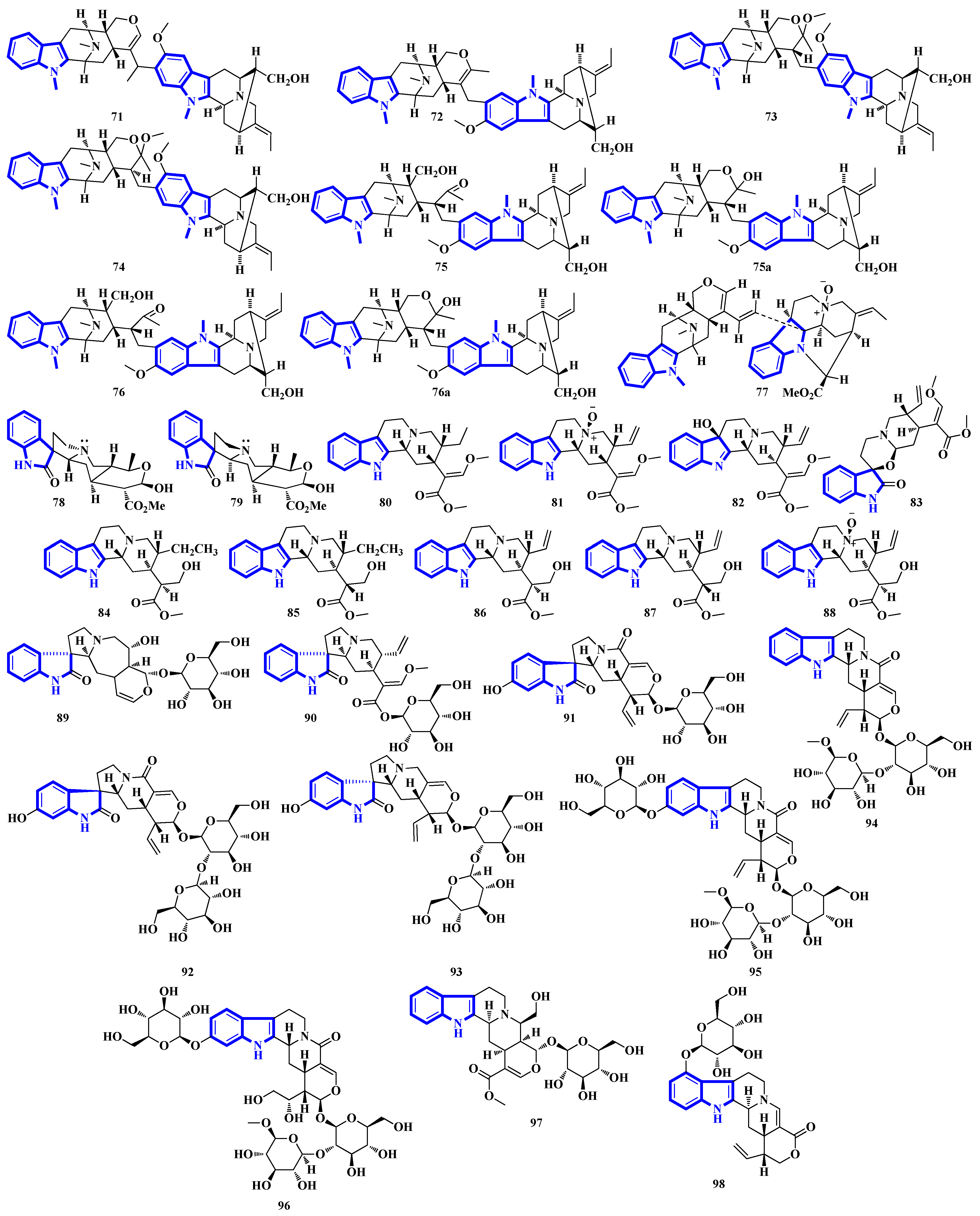
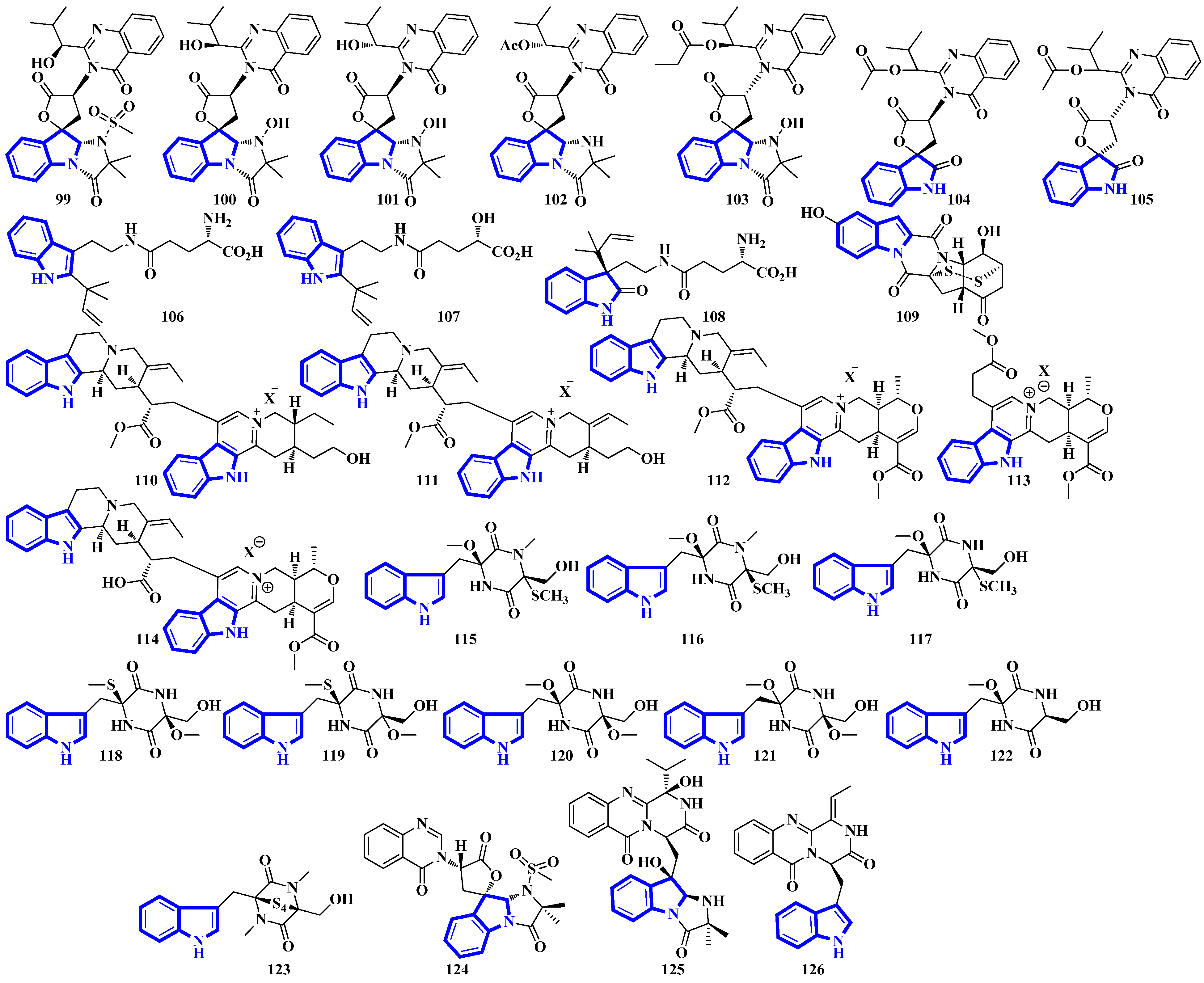
Quinazoline-containing compounds 99 to 105 were isolated from a fungus in the Aspergillus genus extracted from Sanguinolaria chinensis, a bivalve mollusk. Additionally, compounds 99 to 103 all contain a 5/5/6/5/6/6 hexacyclic ring system and 1-hydroxy-2-methyl propyl groups at C-26. Some have an acylated hydroxy, while compound 99 possesses a rare amino sulfonyl group. Removing the C-14/C-15/N-16 fragment, and the two methyl groups bonded to C-15 in compounds 99 to 103 yields the structural framework for compounds 104 and 105 [39]. Compounds 106 to 108 are water-soluble glutamic acid derivatives [40]. Compound 109 was isolated from Epicoccum nigrum, extracted from the gills of Amphilophus. It contains an aromatic indole motif, a rarity among the epicoccin type of epipolythiodioxopiperazines (ETPs) [41]. Alkaloids 110 to 114 were isolated from the aerial parts of Picralima nitida. Compounds 110 to 113 are serpentine-type bisindoles, while compound 114 is analstonine-type MIA [42]. Similarly, compounds 115 to 123 are also ETPs. These were isolated from Chaetomium cochliodes, which were extracted from animal manure. Compounds 115 to 123 are chetomin-type MIAs, with compound 123 possessing a unique tetra-sulfur bridge [43]. Compounds 124 to 126 are fumiquinazoline-type alkaloids isolated from Scedosporium apiospermum, extracted from the inner tissue of Lobophytum crissum, a coral (Figure 7) [44].
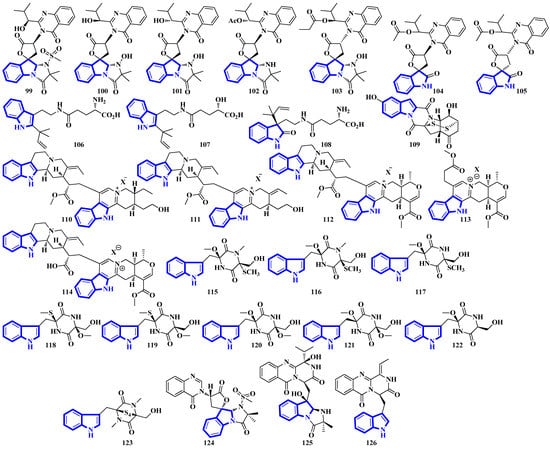
Figure 7.
Novel indole alkaloids 99 to 126.
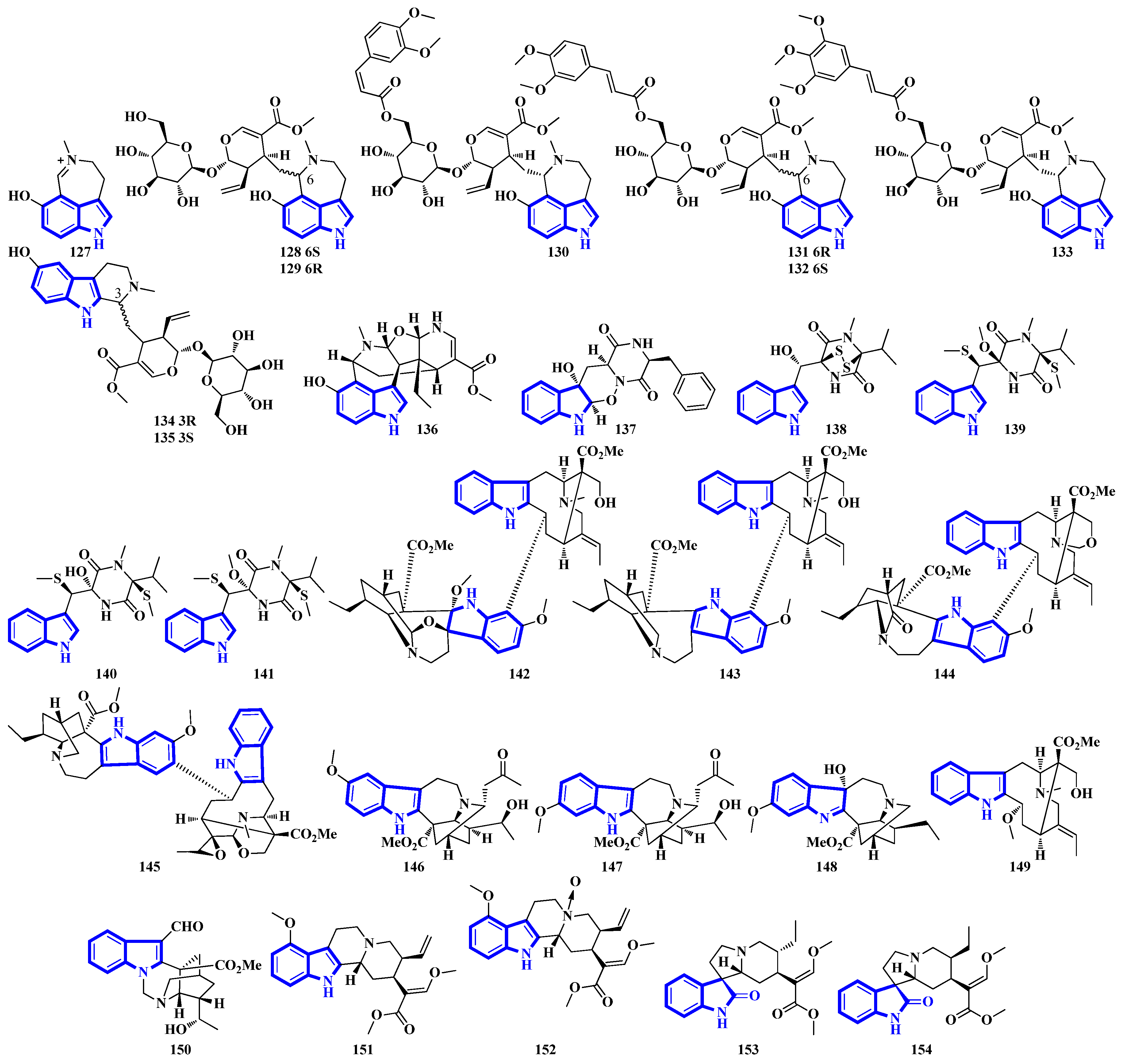
Alkaloids 127 to 136 were isolated from the leaves of Psychotria nemorosa (Figure 8). Compounds 127 to 133 and 136 are azepine-type indole alkaloids, while compounds 134 and 135 are β-carboline derivatives [45]. Compound 137 is an indole diketopiperazine alkaloid with an unusual pyrazino [1′,2′:2,3][1,2]oxazino [6,5-b]indole tetraheterocyclic ring system [46]. Compounds 138 to 141 are thiodiketopiperazine alkaloids isolated from Phaeosphaeria fuckelii, extracted from Phlomis umbrosa. They all contain an unusual β-(oxy)thiotryptophan motif [47]. Compounds 142 to 150 were isolated from the twigs and leaves of Tabernaemontana corymbose. Compounds 142 to 145 are vobasinyl-ibogaine-type alkaloids, with compound 142 additionally containing an unusual 1,3-oxazinane motif [48]. Compounds 151 and 152 were isolated from Mitragyna speciosa, while compounds 153 and 154 were isolated from Havil. Rubiaceae. Compound 151 is a diastereomer of paynantheine, while compound 152 is a paynantheine-type N-4 oxide. Furthermore, compounds 153 and 154 are spirocyclic oxindoles, and are C-3 epimers of rhynchophylline and corynoxine B, respectively [49].
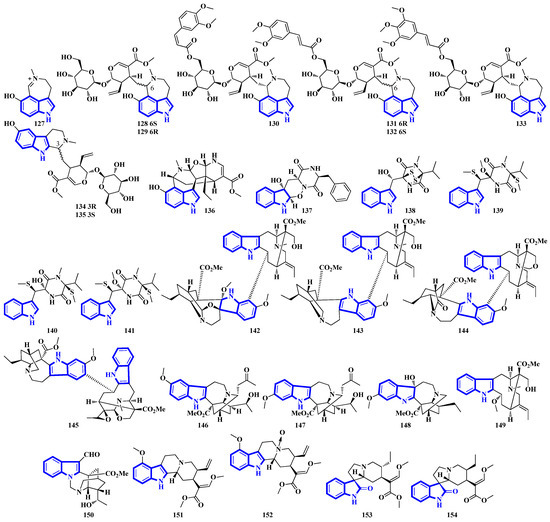
Figure 8.
Novel indole alkaloids 127 to 154.
Compounds 155 to 159 were isolated from the twigs and leaves of Melodinus hemsleyanus (Figure 9). Compound 155 is an unusual aspidosperma-type alkaloid possessing a 6/5/6/5/5 pentacyclic architecture with a smaller E ring (loss of CH2), while compounds 158 and 159 are vincamine-type alkaloids [50]. Compounds 160 to 172 are β-carboline alkaloids that were isolated from Actinoalloteichus that was obtained from sea mud. Compounds 160 to 167 share a novel indole–pyridone–imidazole tetracyclic framework. Additionally, compounds 164 to 167 are analogs of 160 but lack Δ15 and have different substitutions at C-13 and C-16 [51]. Compounds 173 and 174 are krisynomycins isolated from Streptomyces canus and obtained from sand in the Kalahari Desert. They are non-ribosomal cyclic depsipeptides bisindoles with prenylated and chlorinated tryptophan monomers [52]. Compounds 175 to 185 are bromotryptamine alkaloids, whereas 175 possesses a rare 1,4-thiazine-1,1-dioxide group. Furthermore, compound 177 is the first oxazolidinium-containing indole from nature [53]. Compound 186 was isolated from Amathialamourouxi, a bryozoan found in rock pools. It is a simpler molecule; its name is 2,5-dibromo-1-methyl-1H-indole-3-carbaldehyde [54]. Compounds 187 to 195 are monoterpenoid indole alkaloids that were extracted from the leaves of Tabernaemontana pachysiphon. They are all aspidosperma–aspidosperma-type alkaloids and possess a rare spiro heterocycle between their two constituent units. Compounds 190 and 191 contain an indole ring fused with an (N,N-diethyl)methyl amino group [55].
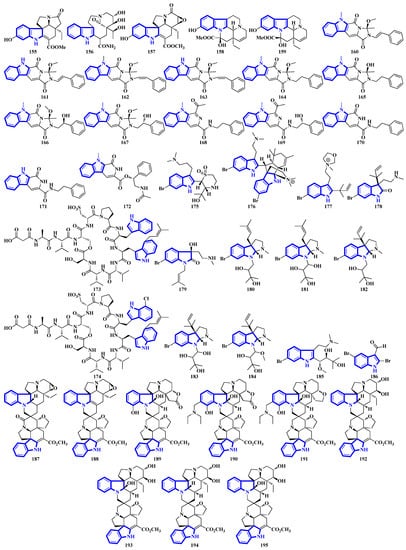
Figure 9.
Novel indole alkaloids 155 to 195.
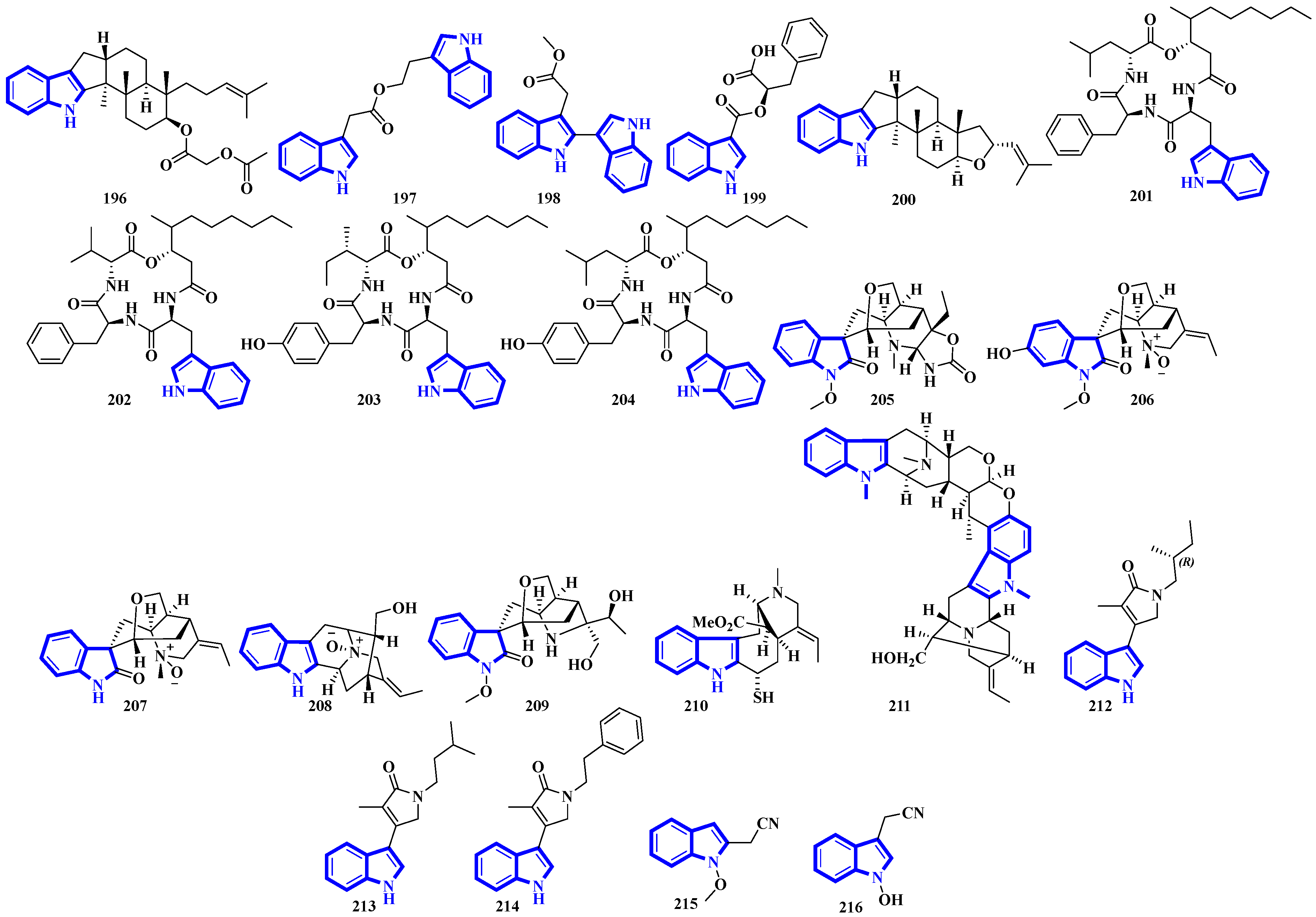
Alkaloids 196 to 200 were isolated from Fusarium obtained from the inner tissue of Acanthaster planci, and compounds 197 and 198 are bisindoles [56]. Compounds 201 to 204 are cyclotetradepsipeptides isolated from Beauveria fungi and obtained from the fresh stems of Gypsoplaca macrophylla. These alkaloids contain a 3-hydroxy-4-methyldecanoic acid (HMDA) group [57]. Compounds 205 to 209 are monoterpenoid indole alkaloids that were isolated from the stems of Gelsemium elegans. Furthermore, compound 205 is a novel triamino alkaloid with a novel 6/5/7/6/6/5 hetero hexacyclic ring system bearing a unique hexahydrooxazolo [4,5-b]pyridin-2(3H)-one motif. In contrast, compound 208 is the first N-oxide of a sarpagine-type alkaloid isolated from a plant in the Gelsemium genus [58]. Compound 210 was isolated from the stems of Mostuea brunonis and is a novel sulfur-containing vobasine-type indole alkaloid. It is among the rare monomeric vobasines lacking oxygen at C-3 [59]. Compound 211 was isolated from the stem bark of Alstonia penangiana and is a linearly fused macroline-sarpagine-type bisindole [60]. Compounds 212 to 214 are indole-γ-lactams and represent psammocindoles, a new class of indole alkaloids derived from three amino acids (isoleucine, leucine, and phenylalanine) [61]. Compounds 215 and 216 were isolated from the roots of Isatis indigotica, and they contain an indole acetonitrile motif (Figure 10) [62].
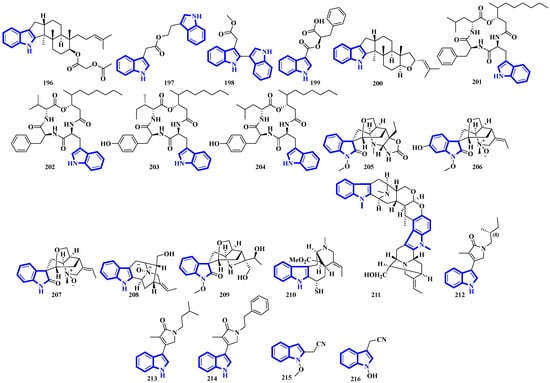
Figure 10.
Novel indole alkaloids 196 to 216.
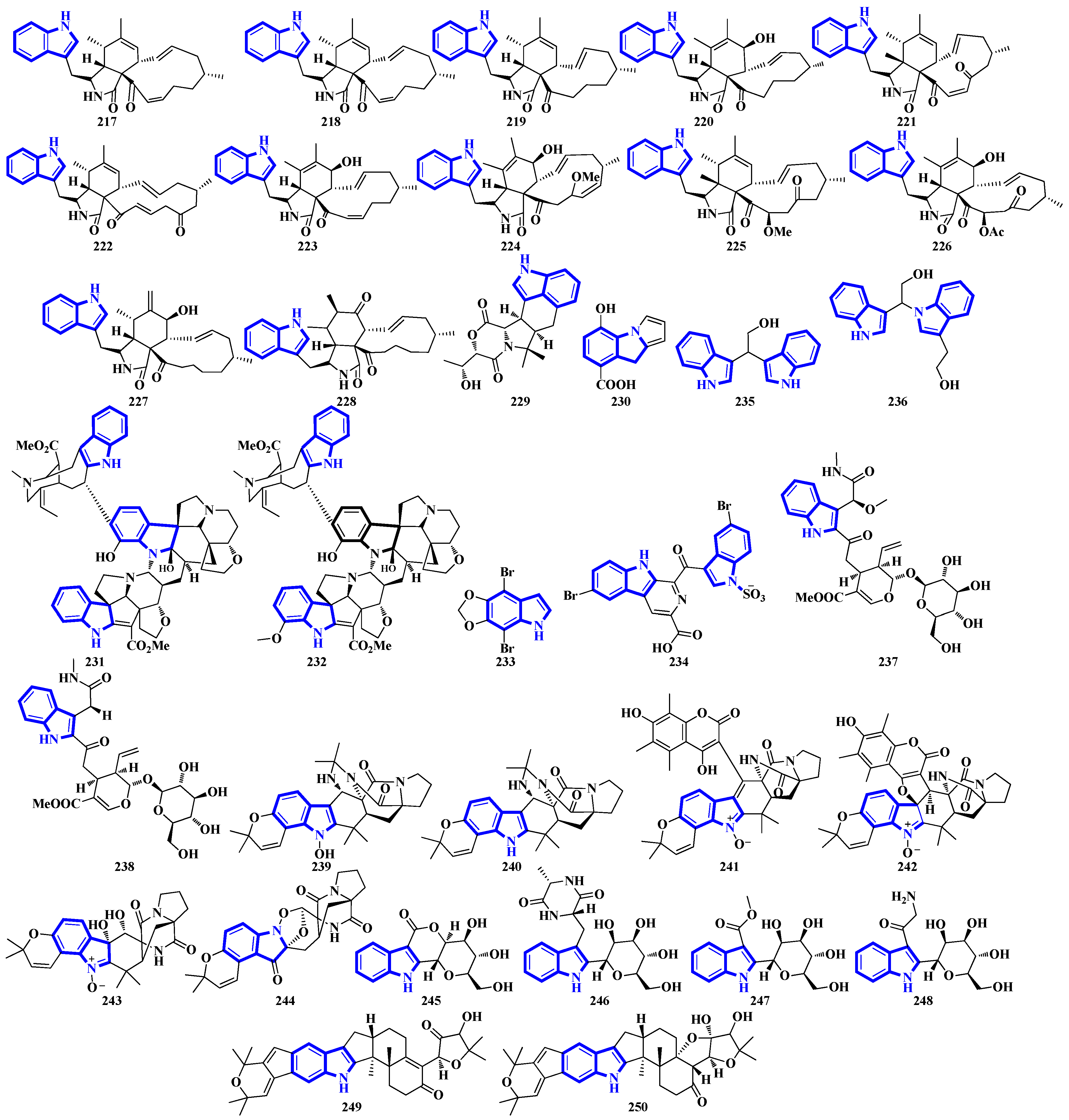
Compounds 217 to 228 are novel [11]-chaetoglobosins isolated from Pseudeurotium bakeri, obtained from macrocoma moss (family: Orthotrichaceae). The alkaloids, containing an 11-membered macrocyclic ring, represent a rare motif as only seven naturally occurring [11]-chaetoglobosins have been reported [63]. Compound 229 is a novel indole-linked 2,5-diketomorpholine alkaloid with an unprecedented 6/5/6/6/5 core, while compound 230 contains an indole fused with pyrrole and possesses a 6/5/5 core [64]. Compounds 231 and 232 were isolated from the bark of Voacanga africana, and they are the first monoterpene trisindole alkaloids containing a vobasine–aspidosperma–aspidosperma skeleton [65]. Similarly, compound 233 is the first methylenedioxy dibromoindole alkaloid exhibiting antiosteoporosis activity [66]. Additionally, compound 234 is a dibrominated β-carboline sulfamate, one of the only 11 indole sulfamate-containing marine natural products reported in the Marin Lit. database [67]. Compounds 235 and 236 were products of the heterologous expression of the EDB gene cluster (found in P. fluorescens NZI7) in E. coli. The metabolites are isoindoles containing an ethanol group [68]. Compounds 237 and 238 were isolated by processing the whole Ophiorrhiza japonica plant representing two novel glycosides [69]. Compounds 239 to 244 are notoamide-type alkaloids isolated from Aspergillus sclerotiorum, extracted from a gorgonian coral. Compounds 239 and 240 possess a unique 2,2-diaminopropane group, while compounds 241 and 242 are novel notoamide hybrids having a coumarin moiety. Compound 244 is a new highly oxidized notoamide scaffold [70]. Compounds 245 to 248 are indole-C-mannopyranoside alkaloids: rare indole C-glycosides in which an α-D-mannose residue is connected to the indole ring at C-2 [71]. Compounds 249 and 250 are diterpenoid indole alkaloids isolated from Penicillium oxalicum obtained from the mantis shrimp, Oratosquilla oratoria. Compound 249 is the first indole-diterpenoid derivative with a 4-hydroxy-5,5-dimethyldihydrofuran-3-one as a side-chain (6/5/6/5/5/6/6-5 system). Compound 250 is a hemiketal containing a unique 6/5/6/5/5/6/6/5/5 ring system (Figure 11) [72].
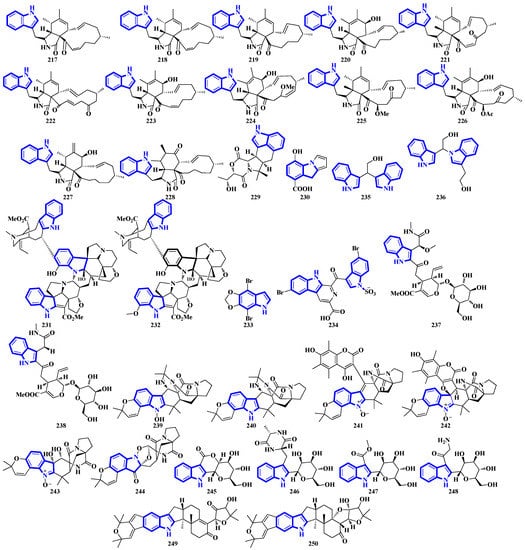
Figure 11.
Novel indole alkaloids 217 to 250.
2.4. Structure Reappraisals
Due to the stereochemical richness of natural products and structural complexity, extensive spectroscopic analyses are required to elucidate their complete structures. Occasionally, some fragments within the compounds are misidentified, possibly due to a lack of spectroscopic data resulting from insufficient quantities of the compound to analyze. Later, due to contradictory results (often emerging from the synthesis of homologs or the analysis of their decomposition products), the postulated structures of compounds are reevaluated, and the errors in the initially proposed structures are rectified.
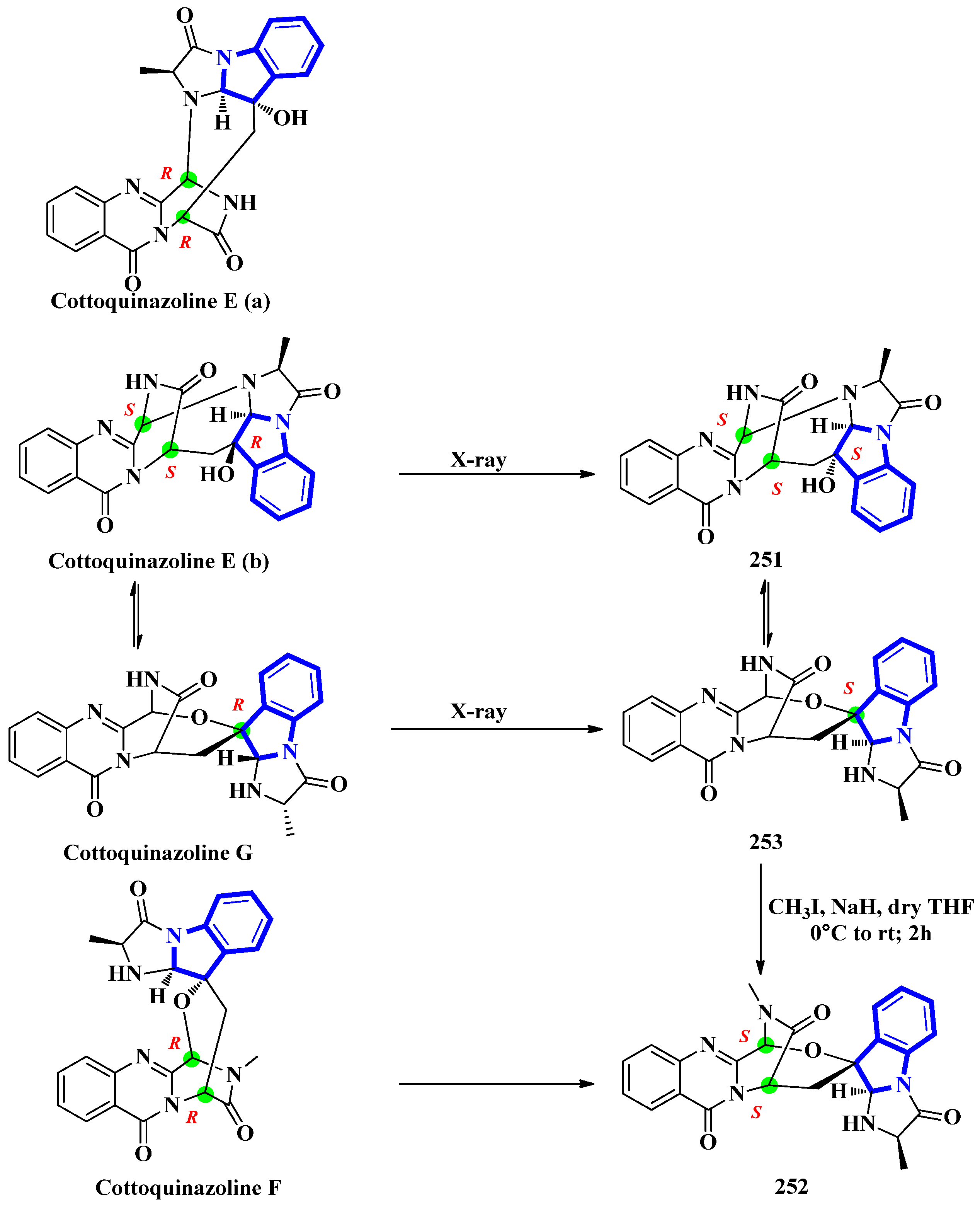
Cottoquinazolines E-G (251 to 253) were isolated from the fungus Neosartorya fischeri, found in the medicinal arthropod Cryptotympana atrata [73]. The fumiquinazoline-type alkaloids were reinvestigated due to unresolved absolute configuration of compounds 251 and 252. Extensive spectroscopic analysis, electronic circular dichroism data, and X-ray crystallography confirmed the absolute configurations of all three indole alkaloids (Figure 12). Cottoquinazoline E had two posited steric conformations: cottoquinazoline E (a) with C-3R, C-14R, and C-16S configurations or cottoquinazoline E (b) with C-3S, C-14S, and C-16R configurations. In addition, spectroscopic studies confirmed the structure of 251 and its configurations (C-3S, C-14S, and C-16S). Similarly, the absolute configurations of 252 were changed from C-3R and C-14R to C-3S and C-14S, and 253 was modified from C-16R to C-16S [74].
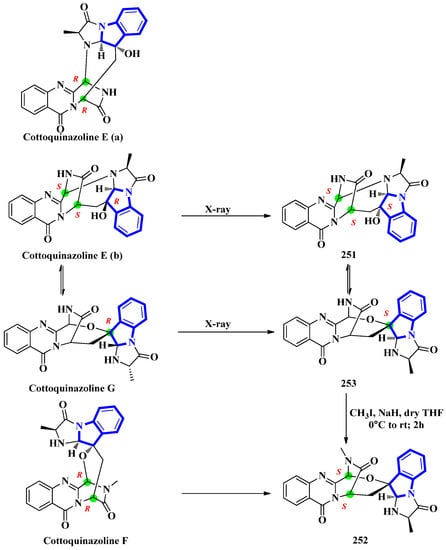
Figure 12.
Reappraised structures of cottoquinazolines (E–G) 251–253.
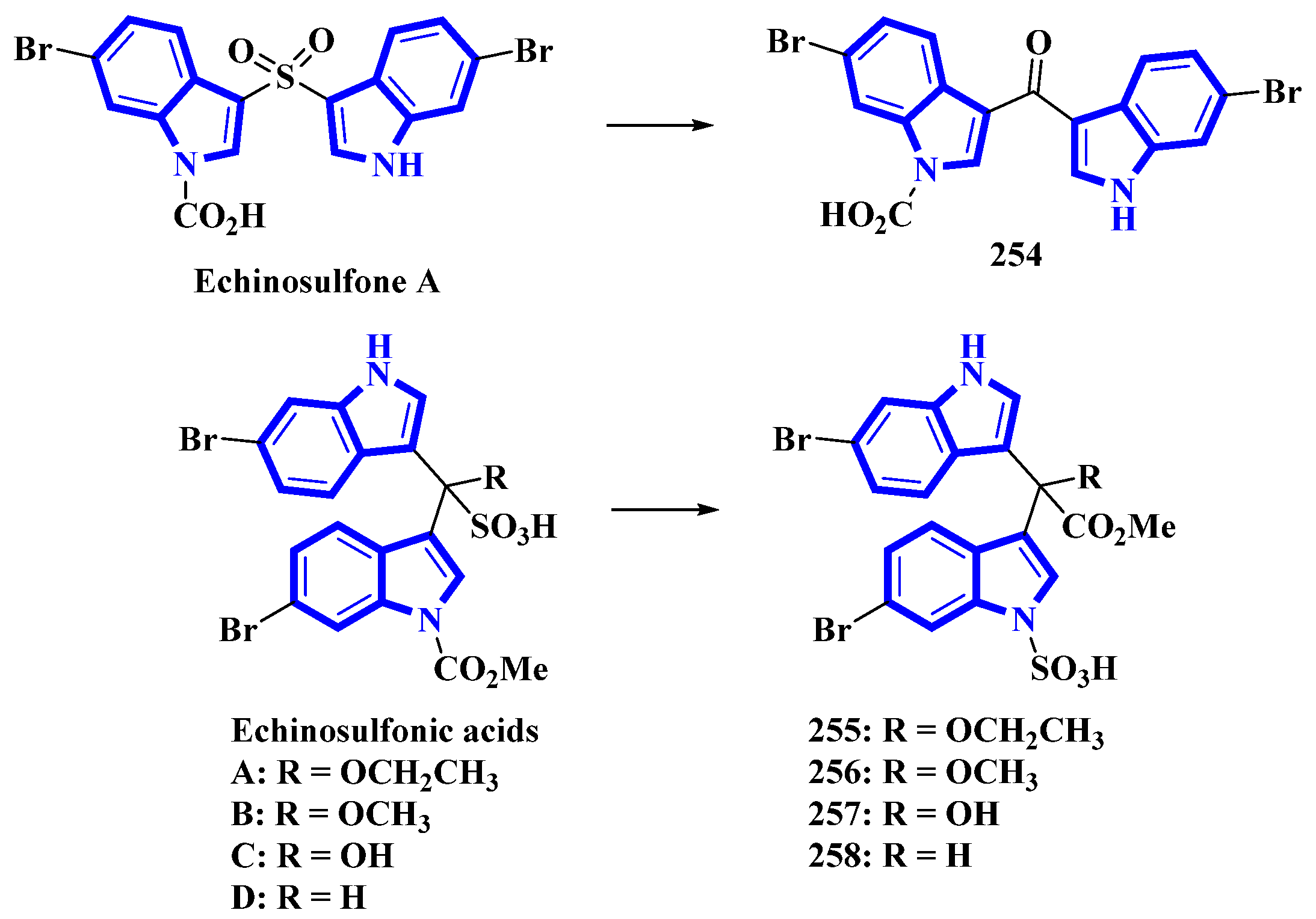
Echinosulfone A (254) and echinosulfonic acids A–C (255–257) were initially isolated from Echinodictyum, a marine sponge [75]. Similarly, echinosulfonic acid D (258) was isolated from a Psammoclema sponge, and its structure was assigned on the basis of the previous report [76]. However, discrepancies arose when decomposition products of 254 were analyzed using X-ray diffraction. Thus, the original alkaloids were reisolated from sponges in the Crella genus. Their structures were re-evaluated using 1D and 2D NMR data in conjunction with MS fragmentation, X-ray crystallography, and DFT predictions of 13C-NMR shifts. For compound 254, the original sulfonyl bridge between the two indole monomers was modified to a carbonyl bridge, while the N-1-bonded carboxylic acid was replaced with an N-1-bonded sulfonic acid (Figure 11). Similar changes were made in compounds 255 to 258, where the C1ʹʹ-bonded sulfonic acid was changed with the C1ʹʹ-bonded methoxycarbonyl group. The N-1-bonded methoxycarbonyl was replaced with an N-1-bonded sulfonic acid moiety (Figure 13) [77].
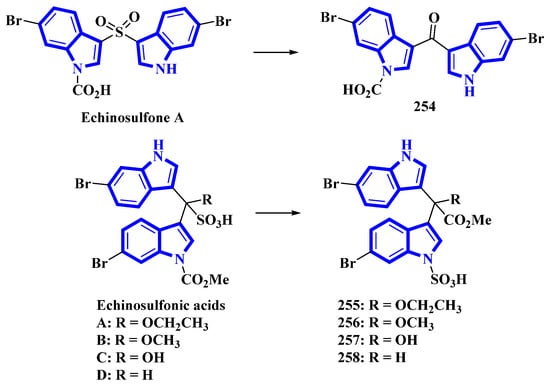
Figure 13.
Reappraised structures of echinosulfone A and echinosulfonic acids (A–D) 254–258.
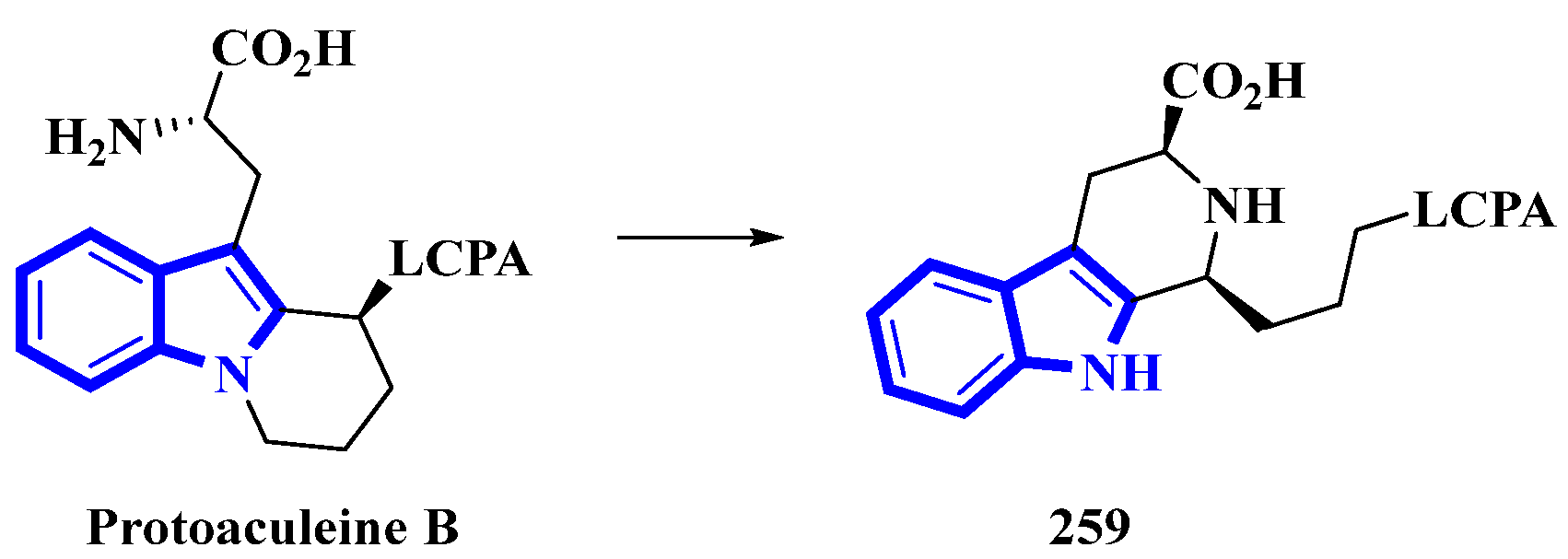
Protoaculeine B (259) is the N-terminal residue of aculeine B, a peptide toxin that is a potent membrane disruptor [78]. Aculeines are found in the aqueous extracts of Axinyssa aculeata, a marine sponge. Aculeine B consists of a 44 amino-acid long sequence (containing three disulfide bonds), connected to a long-chain polyamine (LCPA; consisting of a 1,3-propanediamine oligomer) by a tryptophan-derived amino acid (259). A homolog of 259 was synthesized, but its spectroscopic and chemical properties differed significantly from its natural counterpart. Thus, the structure of compound 259 was re-evaluated, and its C-3-bonded amino-acid chain was modified into a fused pyridine-2-carboxylic acid ring bonded to the LCPA via a propane group (Figure 14) [79].
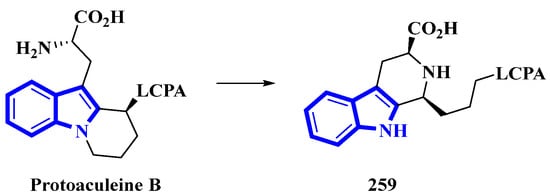
Figure 14.
Reappraised structure of protoaculeine B 259.
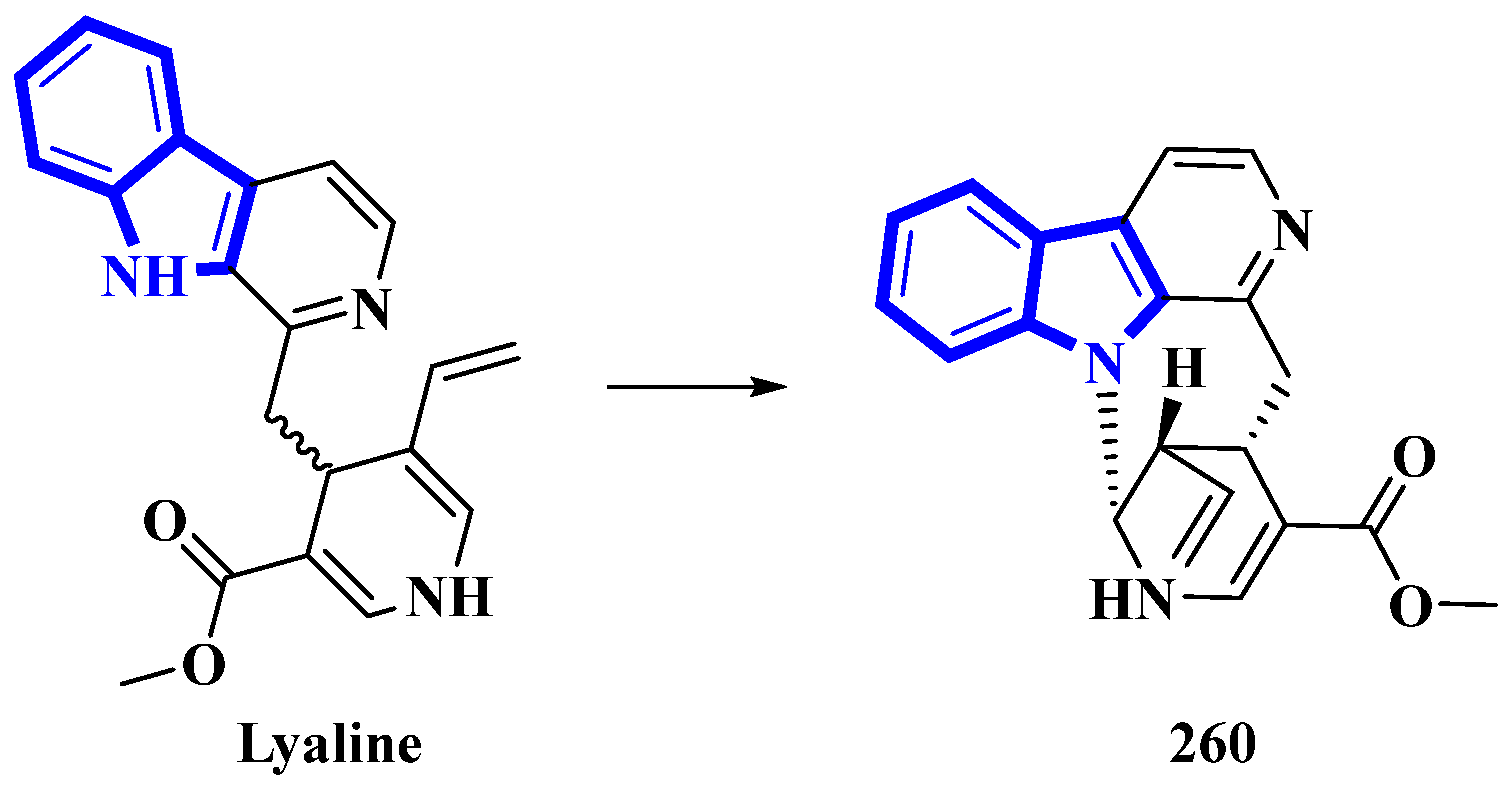
Lyaline (260) was isolated from Pauridiantha paucinervis, and was reported as an unusual harman-1,4-dihydropyridine derivative [80]. However, efforts to synthesize the monoterpene indole alkaloid resulted in discrepancies (the synthetic product was extremely unstable; nevertheless, the natural analog did not show any degradation) [81]. Thus, the absolute configuration of C-15 was elucidated as S, and a single bond between C-21 and N-1 was added. As Δ20 from the original structure was reduced, it was revised as S (Figure 15). Lastly, considering the revised structure of 260, it was established as the first naturally occurring nacycline analog [82].
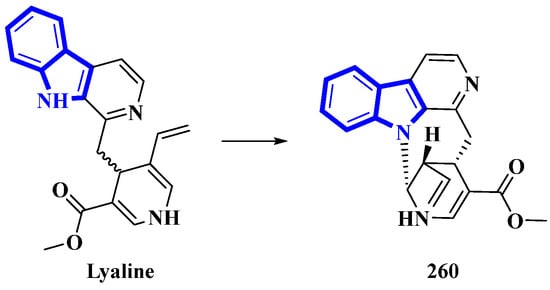
Figure 15.
Revised structure of lyaline 260.
3. Synthesis of Indole Alkaloids
3.1. Total Synthesis of Indole Alkaloids
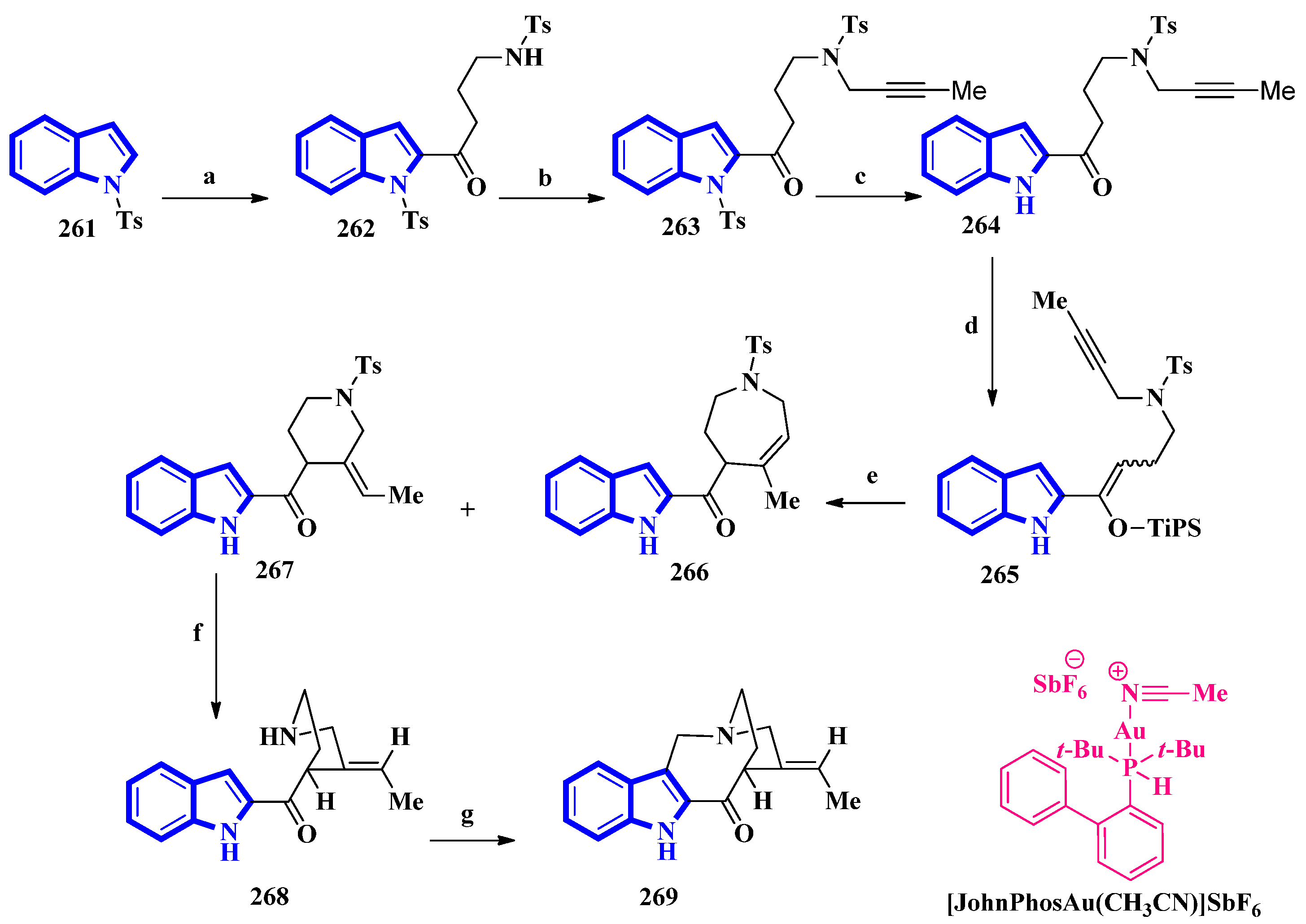
(±)-Conolidine (269), a potent nonopioid analgesic, was synthesized in six steps without any nonstrategic redox manipulations. It was isolated from the stem bark of Tabernaemontana divaricate in 2004 [83], and its first total synthesis was carried out in 2011 with a nine-step synthetic route in an 18% overall yield [84]. The latest total synthesis attempts primarily utilized gold(I)-catalyzed Conia-ene and Pictet–Spengler reactions, producing 269 in six steps in overall 19% yield (Figure 16). They also used DFT calculations to develop a successful scheme [85].
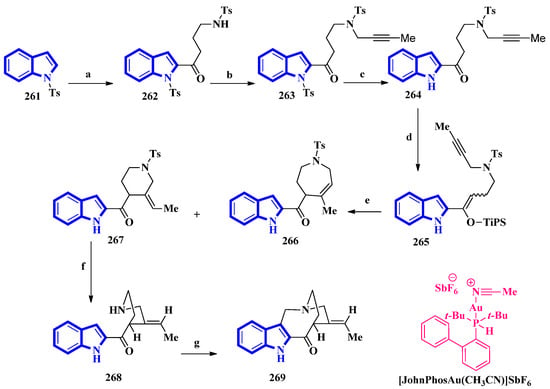
Figure 16.
The total synthesis of (±)-conolidine 269. (a) n-BuLi, THF, −78 °C, 5 min, then rt, 1 h, then N-tosylpyrrolidonein THF, −30 °C to −15 °C, 4 h, 48%; (b) K2CO3, 1-bromo-2-butyne, CH3CN, 80 °C, 94%; (c) TBAF, CH3CN 35 °C, 15 h, 93%; (d) TiPSOTf, 2,6-lutidine, 35 °C, 5 h, 97%, E:Z = 8:92; (e) [JohnPhosAu(CH3CN)]SbF6, H2O, toluene, 60 °C, 2 h, 15% for 266, 73% for 267; (f) Sodium naphthalenide, THF, −78 °C, 86%; (g) (CH2O)n, TFA, CH3CN, reflux, 2 h, 82%.
For the total synthesis of compound 269, compound 261 was treated with n-butyllithium and dropwise addition to N-tosylpyrrolidone affording 262 that underwent a nucleophilic reaction with 1-bromo-2-butyne to give compound 263. Next, compound 263 was treated with TBAF to remove the N-1-tosyl group producing compound 264. Afterward, 264 was added to TiPSOTf in 2,6-lutidine, affording a pair of stereoisomers of 265 (E:Z = 8:92). Its gold-catalyzed reaction using [JohnPhosAu(CH3CN)]SbF6 produced compounds 266 and 267. The latter was exposed to sodium naphthalenide to deprotect its N-2 tosyl group affording 268. Lastly, a Pictet–Spengler reaction using TFA and paraformaldehyde in acetonitrile was used to convert 268 into 269 [85].
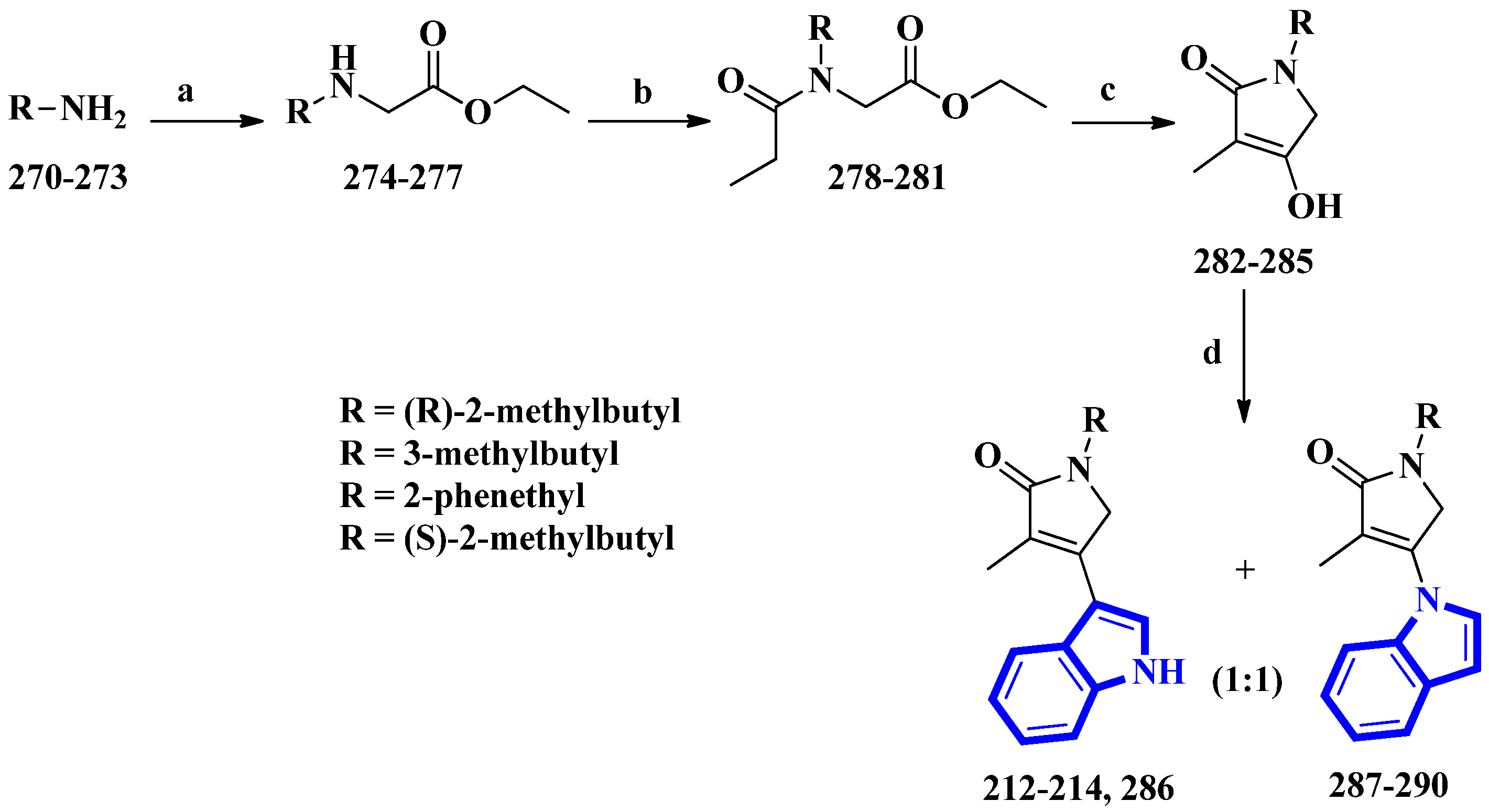
Psammocindoles A and B (212 and 213) significantly increased adiponectin production. Thus, to further investigate their pharmacology and assign the absolute configuration of compound 212 at C-2′, the two alkaloids and their congener, psammocindole C (214), were synthesized. In addition, enantiomers of psammocindole D (286), and four N-lactam analogs, isopsammocindoles A–D (287 to 290), were produced. The synthetic scheme was four steps long, and the overall yields were 23%, 21%, and 21% for compounds 212, 213, and 214, respectively (Figure 17). As a result, the R-configuration of compound 212 at C-2′ was assigned [61].
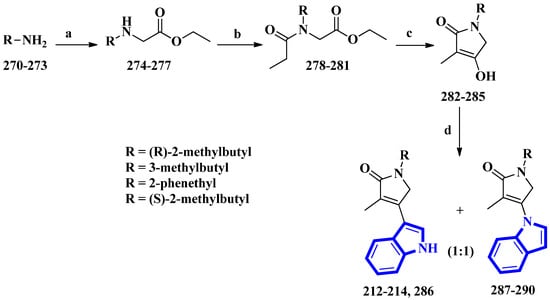
Figure 17.
The total synthesis of psammocindoles (A–D) 212–214, 286, and their isomers 287–290. (a) Ethyl bromoacetate, DCM, Et3N, rt, 2 h, 90–93%; (b) Propionyl chloride, Et3N, 0 °C to rt, 1.5 h, 94–95%; (c) NaH, THF, reflux, 12 h, 83–86%; (d) Indole, BF3-Et2O, 4 Å MS, PhCl, 100 °C, 1.5 h, 27–31%.
(R)-2-Methylbutan-1-amine was prepared in an 88% enantiomeric excess using a five-step synthetic scheme, while the other amines were obtained commercially. The amines (270 to 273) were reacted with ethyl bromoacetate, affording compounds 274 to 277. Subsequently, further condensation with propionyl chloride produced compounds 278 to 281, which were used to prepare the N-alkyl-α,β-unsaturated γ-lactams (282 to 285) by an intramolecular Claisen condensation. Lastly, condensation between the lactams (at C-3) and the indole’s nitrogen afforded compounds 212 to 214 and 286 to 290, with approximately 30% yields for all analogs [61].
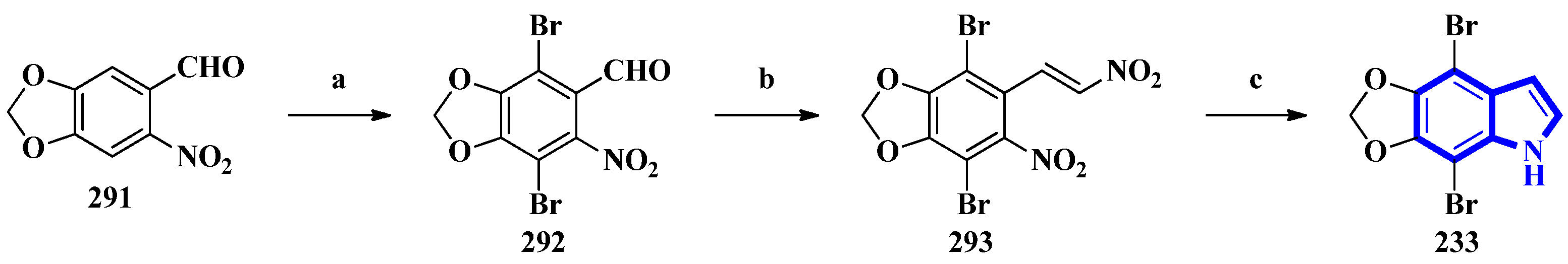
To confirm the structure of amakusamine (233), it was synthesized by the same research group that first isolated it. Upon initial failure of a direct debromination of 5,6-methylenedioxyindole, they used an alternative reaction scheme containing three steps and obtained an overall yield of 56% (Figure 18). The synthetic method also enabled the synthesis of 15 synthetic derivatives of compound 233 (294 to 308) for an SAR analysis (Figure 19) [66]. The synthesis involved the reaction of 6-nitropiperonal (291) with NBS in concentrated H2SO4, producing 292. A Henry reaction (using Al2O3 as a base and nitromethane) followed by dehydration using acetic anhydride transformed 292 into 293. However, since the latter was crystalline and hard to dissolve in many solvents, its crude mixture was reduced directly by an excess of iron in acetic acid, affording compound 233 [66].
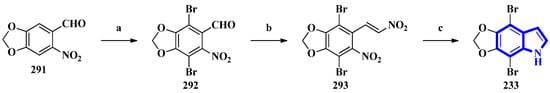
Figure 18.
The total synthesis of amakusamine 233. (a) NBS, conc. H2SO4, 63%; (b) (1) Al2O3, CH3NO2 and (2) Ac2O; (c) Iron in AcOH, (last two steps) 89%.
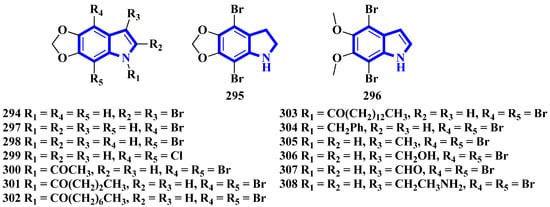
Figure 19.
Amakusamine derivatives synthesized 294–308.
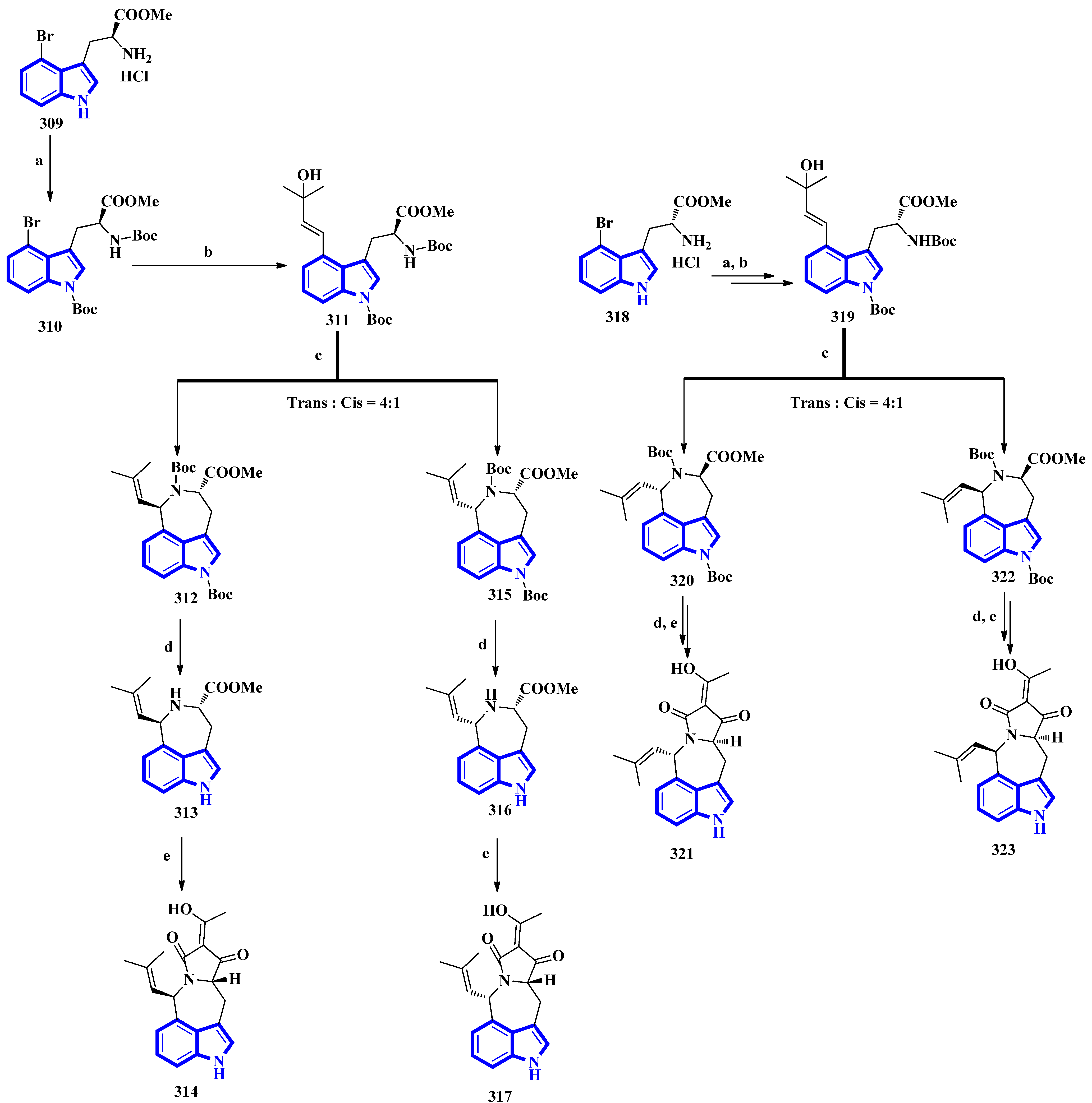
The first total synthesis of griseofamine B (314) and its three stereoisomers, 16-epi-griseofamine B, ent-griseofamine B, and 11-epi-griseofamine B (317, 321, and 323), was carried out in 2022. Compound 314 is an indole-tetramic acid alkaloid isolated from the Penicillium griseofulvum fungus in 2018 [86]. In the total synthesis, 4-bromo tryptophan methyl ester hydrochloride was used as the starting reagent, with its l-enantiomer being used to synthesize compounds 314 and 317. d-Enantiomer 4-bromo tryptophan methyl ester hydrochloride was used to synthesize compounds 321 and 323. All four compounds were obtained in five steps with yields of 18%, 5%, 19%, and 5%, respectively (Figure 20) [87]. Next, compound 309 was reacted with Boc2O, Et3N, and DMAP under reflux conditions in DCM to afford 310 in 90% yield. A Heck–Mizoroki reaction of 310 with 2-methyl-3-buten-2-ol in the presence of PdCl2(PPh3)4, Ag2CO3, and Et3N in 1,4-dioxane produced compound 311 in 67% yield. Afterward, refluxing 311 with PdCl2(CH3CN)2 in CH3CN generated a pair of diastereomers 312 and 315. TMSOTf and 2,6-lutidine in DCM were used to remove the Boc groups and gave compounds 313 and 316. A tandem acylation/Lacey–Dieckmann cyclization of 313 and 316 with diketene was performed to afford 314 and 317, respectively. Additionally, using the same synthetic scheme with compound 318 as the starting reagent afforded compounds 321 and 323 [87].
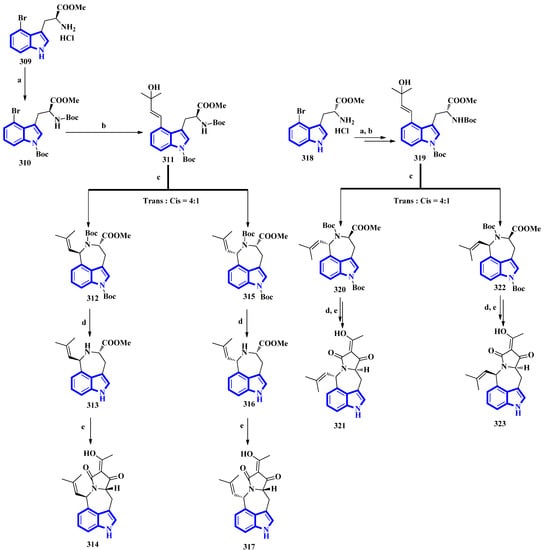
Figure 20.
The total synthesis of griseofamine B (314) and its isomers 317, 321, and 323. (a) Boc2O, Et3N, DMAP, DCM, reflux, 2 h, 90%; (b) 2-methyl-3-buten-2-ol, PdCl2(PPh3)4, Ag2CO3, Et3N, 1,4-dioxane, 100 °C, 6 h; 67% (c) PdCl2(CH3CN)2, CH3CN, reflux, 2 h; E:Z = 56–58%:15–16% (d) TMSOTf, 2,6-lutidine, DCM, rt, overnight, 87–88%; (e) diketene, Et3N, DCM, rt, overnight, 62–63%.
3.2. Synthesis of Indole Derivatives
As β-carboline derivatives generally display only moderate cytotoxicity, new synthetic derivatives were designed to contain a hydroxycinnamic acid moiety inferring HDAC-inhibitory properties that provide synergy and improve their antiproliferative effects [88]. These derivatives (347 to 352) differed only in the number of carbons connecting the β-carboline with the hydroxycinnamic acid motif and in the substituents on the phenyl ring in the hydroxycinnamic acid moiety (Figure 21).
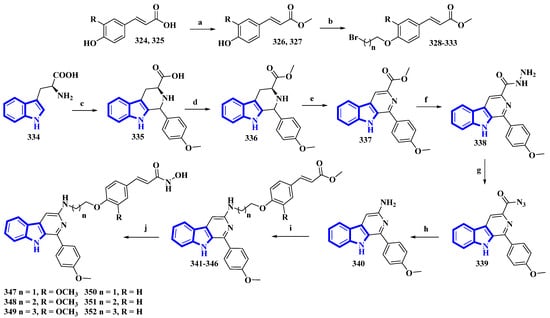
Figure 21.
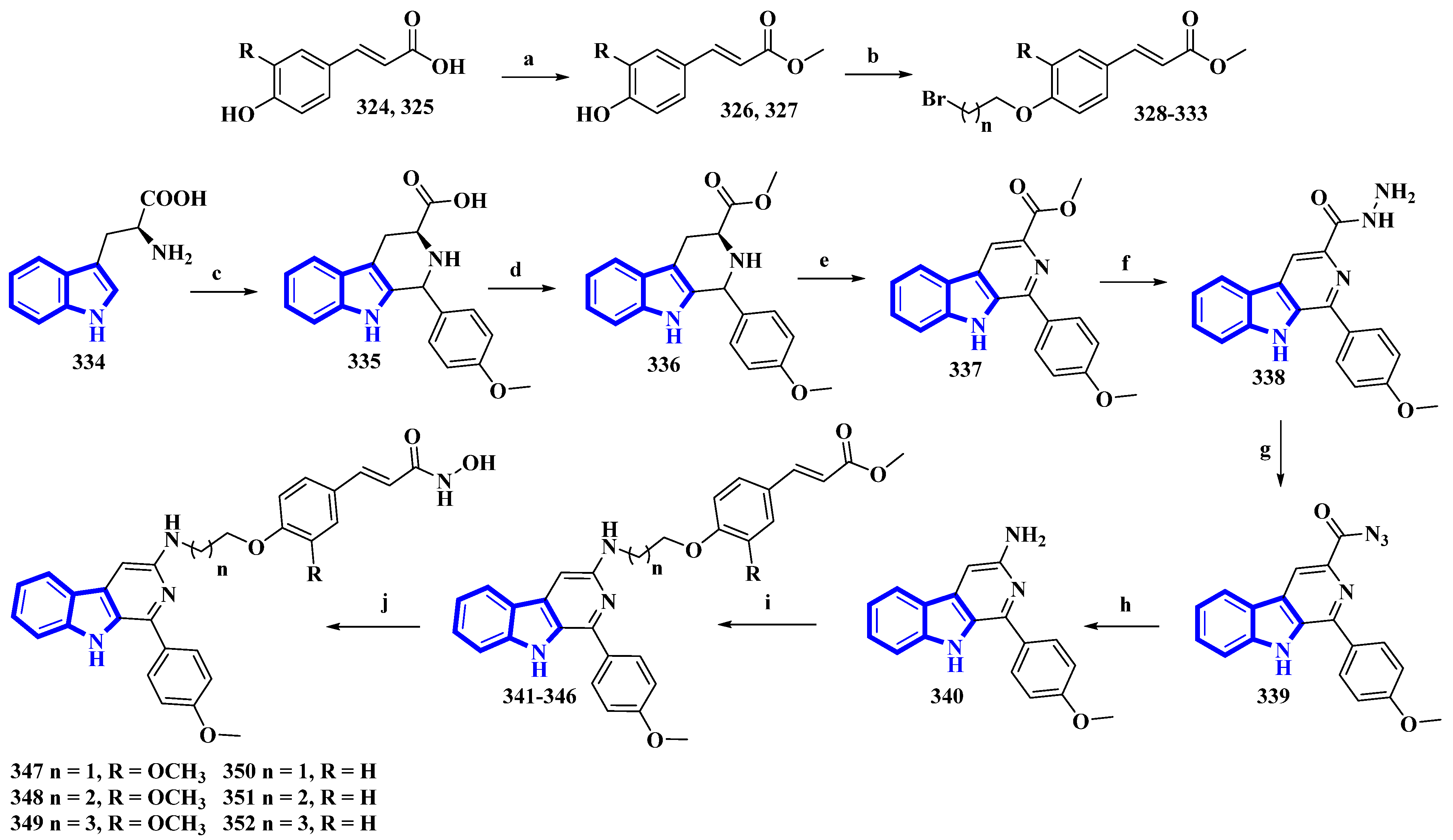
Synthesis of hydroxamic derivatives 347–352. (a) SOCl2, CH3OH, 0 °C, 1 h, and then 65 °C, 4 h; (b) ω-Dibromoalkane, K2CO3, CH3CN, reflux, 4 h, 66–75%; (c) 4-methoxybenzaldehyde, AcOH, reflux, 4 h; (d) Thionyl chloride, CH3OH, 0 °C, 1 h, and then 65 °C, 6 h; (e) KMnO4, DMF, rt, 5 h; (f) Hydrazine monohydrate, CH3OH, 50 °C, 6 h; (g) NaNO2, HCl; (h) AcOH, H2O, 50 °C, 6 h; (i) 328–333, K2CO3, CH3CN, reflux, 12 h; (j) NH2OK, CH3OH, rt, 8–12 h, 46–56%.
(E)-Ferulic acid (324) and p-coumaric acid (325) were esterified using SOCl2 in CH3OH, yielding compounds 326 and 327. These compounds were then treated with ω-dibromoalkanes (1,2-dibromoethane, 1,2-dibromopropane, and 1,2-dibromobutane) in the presence of K2CO3 to produce compounds 328 to 333. Using a Pictet–Spengler reaction, l-tryptophan (334) was transformed into 335 with 4-methoxybenzaldehyde. Compound 335 was then esterified using SOCl2 in CH3OH, giving compound 336, which was oxidized using KMnO4 in DMF, affording compound 337. It was then reacted with hydrazine monohydrate to give compound 338 and was converted into 339 using NaNO2. Next, compound 339 underwent a Curtis rearrangement to produce compound 340. Lastly, compounds 328 to 333 were reacted with 340, yielding residues 341 to 346, which were then treated with NH2OK to produce derivatives 347 to 352.
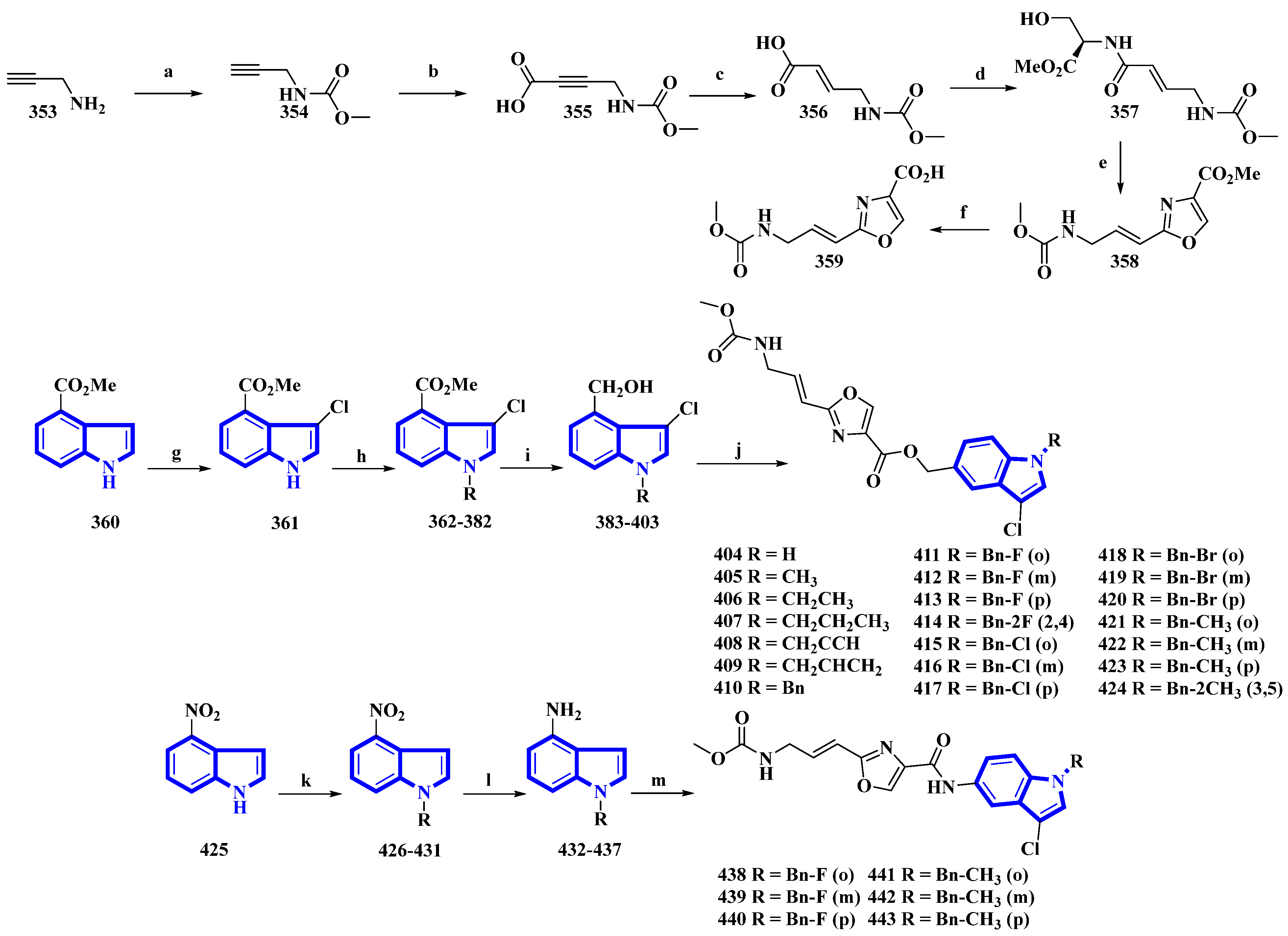
Neopeltolide, a marine natural product, was isolated from a sponge in the neopeltidae family. It is a highly potent antitumor agent (IC50 < 1 nm) and strongly inhibits cytochrome bc1, an essential component of the mitochondrial respiratory chain [89]. Compounds (404 to 424 and 438 to 443) were synthesized, replacing its macrolactone ring with an indole having potential as a fungicide. The derivatives were synthesized in two series. The first series (404 to 424) contained an ester linkage between the oxazole and indole heterocycles (Figure 22). The second series (313 to 318), guided through the bioactivities of the compounds in the first series (Table 2) and the docking studies of leads, replaced the linkage with an amide group and contained only fluorine or methoxy substituents (Figure 22) [90]. The reaction of prop-2-yn-1-amine (353) with NaHCO3 in 1,4-dioxane affords 354. Carboxylation of 354 using n-butyllithium in THF and a CO2 atmosphere produced 355 that was reduced to 356 using Lindlar’s catalyst. Compound 356 reacted with l-serine methyl ester hydrochloride, N-methylmorpholine, and isobutyl chloroformate to produce 357. Compound 357 was reacted with DAST, DBU, and BrCCl3 at a low temperature to yield 358, and LiOH hydrolyzed it to give 359. Separately, methyl indole-4-carboxylate (360) was treated with N-chlorosuccinimide in acidic conditions, producing 361. It was then reacted with substituted benzyl chlorides and NaH in THF, yielding 362 to 382, which were reduced by DIBAL-H, giving 383 to 403. Lastly, 359 was reacted with each of the 21 intermediates to give derivatives 404 to 424. The second derivatives were synthesized by reacting 4-nitroindole (425) with substituted benzyl bromides and NaH in anhydrous DMF, yielding 426 to 431. They were reduced using iron and NH4Cl in EtOH and water, affording 432 to 437. Another time, 359 reacted with each of the six derivatives, producing derivatives of 438 to 443 [90].
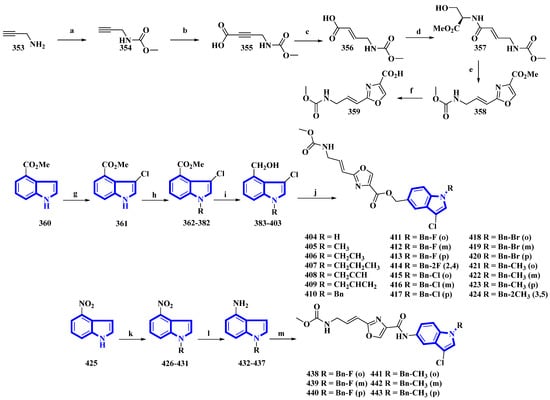
Figure 22.
Synthesis of neopeltolide derivatives 404 to 424 and 438 to 443. (a) NaHCO3, 1,4-dioxane, rt, 95%; (b) n-BuLi, CO2, THF, −78 °C, 84%; (c) H2, Lindlar’s catalyst, EtOAc, 91%; (d) l-Serine methyl ester hydrochloride, i-BuOCOCl, N-CH3-morpholine, THF, 75%; (e) (1) DAST, DCM, −78 °C and (2) BrCCl3, DBU, −20 °C, 62%; (f) LiOH, THF/H2O, 88%; (g) NCS, HCl, THF, 82%; (h) Substituted benzyl chlorides, NaH, DMF, rt, quantitative; (i) DIBAL-H, THF, −78 °C, 57–75%; (j) 359, EDCI, HOBt, Et3N, DMF, 43–71%; (k) substituted benzyl bromides, NaH, DMF, rt, quantitative; (l) Fe, NH4Cl, H2O, EtOH, reflux, 53–67%; (m) 359, EDCI, HOBt, DMF, 49–70%.
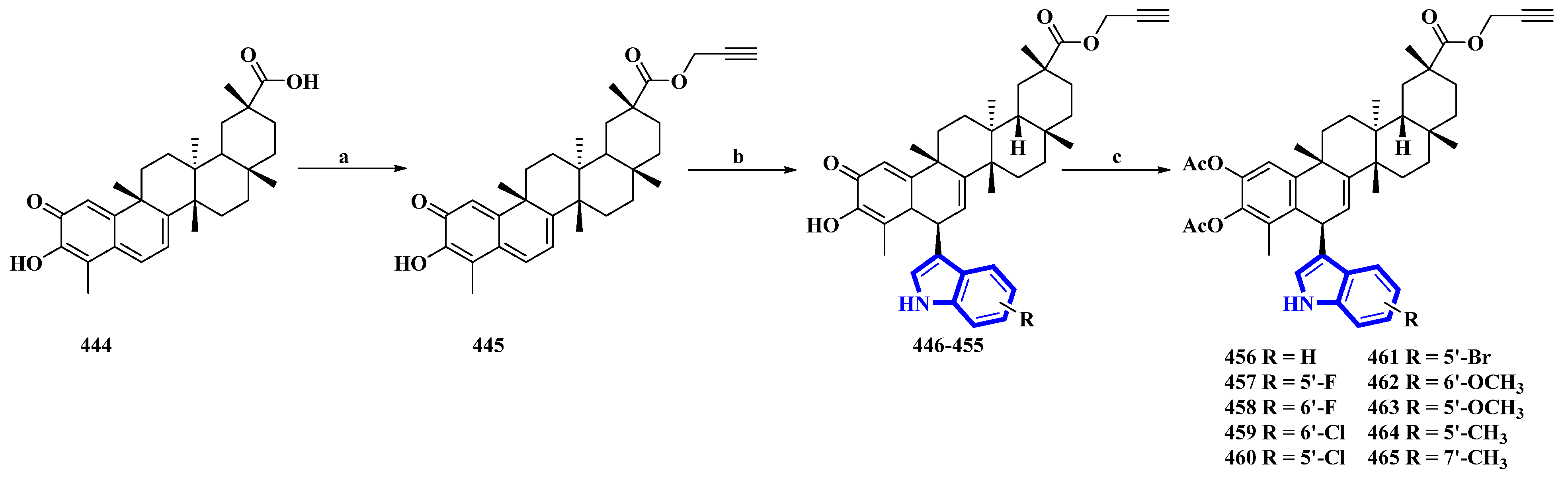
Celastrol (444) is a friedelane-type triterpenoid isolated from Tripterygium wilfordii and possesses immunosuppressive properties [91]. However, it has toxic properties. Therefore, its synthetic derivatives were prepared to obtain lower cytotoxicity. Ten celastrol derivatives (456 to 465) with indole substituents were synthesized that differed only in the substituents attached to the indole group (Figure 23) [92]. In the synthesis, compound 444 was converted into 445 via a nucleophilic reaction using propargyl bromide and NaHCO3 in DMF at room temperature. Compound 445 was transformed using a Friedel–Crafts reaction using substituted indoles and FeCl3·6H2O in DCM, giving compounds 446 to 455. These compounds were acetylated using DMAP and Ac2O immediately without purification, yielding derivatives 456 to 465.
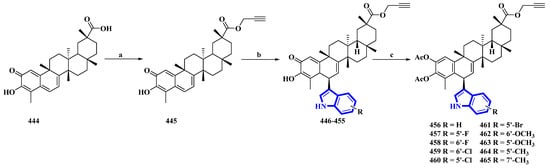
Figure 23.
Synthesis of celastrol derivatives 456 to 465. (a) NaHCO3, propargyl bromide, DMF, rt, 78%; (b) substituted indoles, FeCl3.6H2O, DCM; (c) Ac2O, DMAP, DCM, 37–54%.
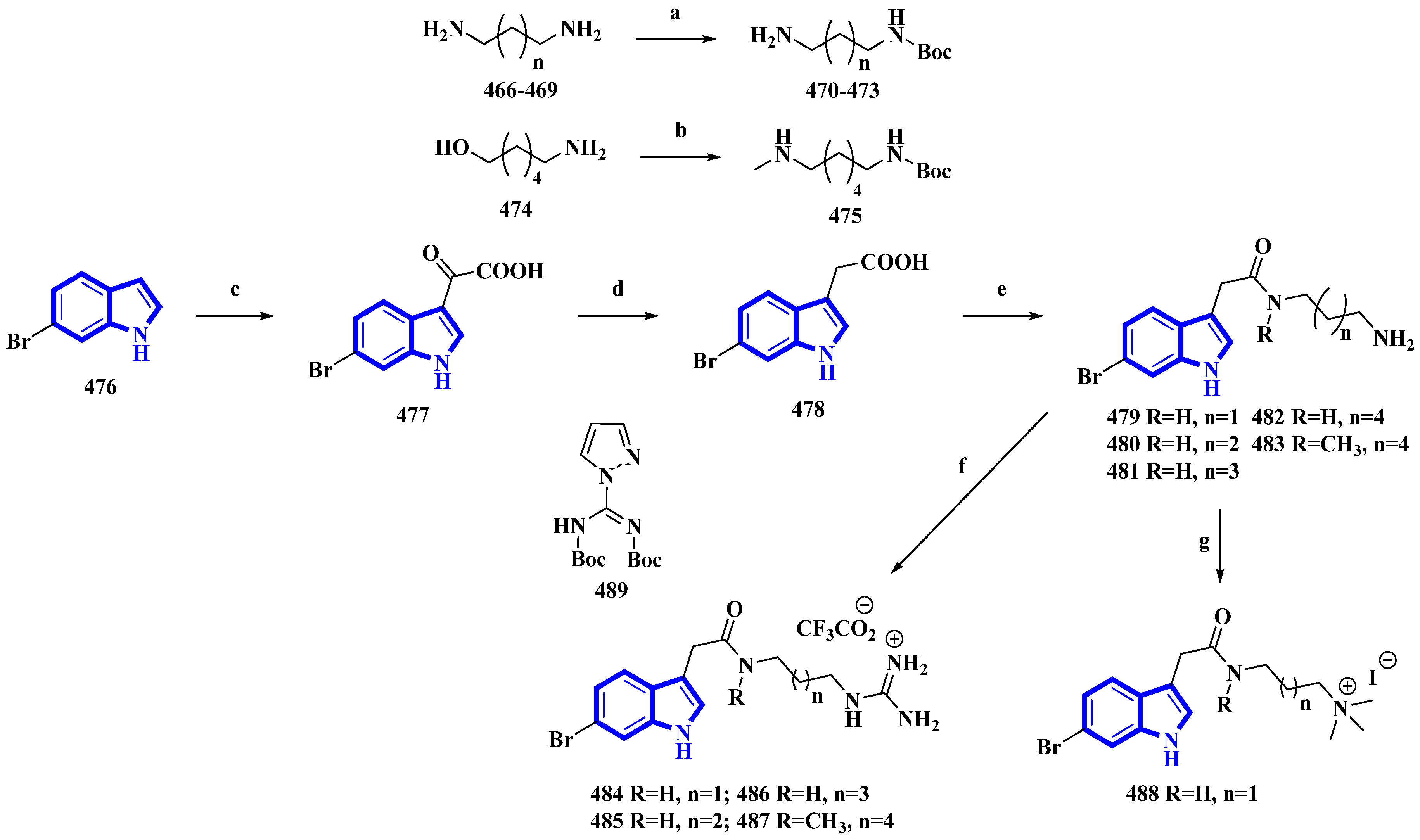
Phidianidine A is a marine natural product isolated from Phidiana militaris, a mollusk. The natural product and its synthetic analogs possess both cytotoxic and immunosuppressive activities. However, the antifouling properties of its derivatives were not previously explored despite its resemblance with other potent antifouling MNPs. Therefore, 10 synthetic derivatives (479 to 488) having primary amines, guanidines, and a quaternary ammonium compound were produced (Figure 24) [93]. Initially, the diamines 466 to 469 (propan-1,3-diamine, butan-1,4-diamine, and pentan-1,5-diamine) and 6-aminohexanol (474) were protected using Boc2O. Then, compound 474 was converted into compound 475 using methane sulfonyl chloride and methylamine. Separately, 6-bromoindole (476) was converted into derivative 477 using oxalyl chloride. It was then reduced to compound 478 using hydrazine and sodium methoxide. The derivative 478 was then reacted with the diamines 470 to 473 and 475 and deprotected using TFA, affording the primary amines 479 to 483. Additionally, compounds 479 to 481 and 483 were reacted with DIPEA and 489 to yield guanidine derivatives 484 to 487. Furthermore, compound 479 was reacted with NaBH3CN in formaldehyde and acetic acid, producing quaternary ammonium derivative 488 [93].
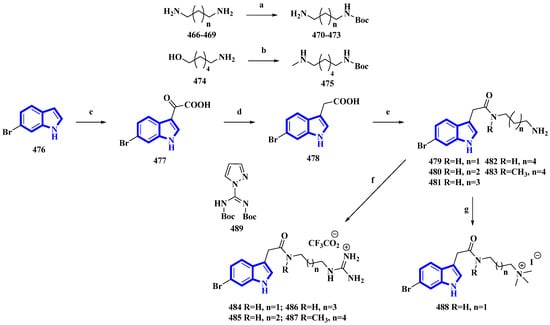
Figure 24.
Synthesis of phidianidine A derivatives 479 to 488. (a) Boc2O, rt, overnight, 93–95%; (b) (1) Boc2O, Et3N, rt, 2 h, (2) MsCl, Et3N, 0 °C, 5 h and (3) CH3NH2, 60 °C, 1 h, 58%; (c) oxalyl chloride, 30 min, 77%; (d) (1) Hydrazine, 80 °C, MW, 15 min and (2) NaOCH3, 80 °C, MW, 15 min, 80%; (e) (1) 470–473, 475, DIPEA, HATU, 1.5 h and (2) TFA, DCM, 4 h, 8–73%; (f) (1) 479–481, 483, 489, DIPEA, 3 h and (2) TFA, DCM, 2 h, 24–83%; (g) 479, CH2O, NaBH3CN, AcOH, 21 h and (2) CH3I, 0 °C, 53%.
3.3. Semi-Synthesis of Indole Alkaloids
Fradcarbazole A, a novel staurosporine-type indole alkaloid containing a thiazole group, was isolated from a mutant strain of Streptomyces fradiae and semi-synthesized from staurosporine by the same group [94,95]. To enhance its efficacy as an antitumor agent, 14 derivatives (535 to 548) of fradcarbazole A were synthesized by variation only in the substituents on two indole units (Figure 25) [96]. In the synthesis, 5-fluoro/chloro/bromo/methoxy-tryptamines (489 to 493) were protected using Boc2O to give derivatives 494 to 498 and oxidized using DDQ, producing compounds 499 to 503. TFA-mediated Boc deprotection resulted in molecules 504 to 508. Staurosporine (509) was protected using Boc2O, giving 510, which was halogenated using NCS and NBS (compounds 511 and 512), and deprotected by TFA (compounds 513 and 514). These derivatives and compound 509 were converted to compounds 515 to 517 using TCDI. These compounds were alkylated by CH3I in CH3CN, affording compounds 518 to 520. They were then converted into compounds 521 to 534 via a nucleophilic reaction with 504 to 508. Lastly, intramolecular cyclization reactions of compounds 521 to 534 resulted in 535 to 548 [96].
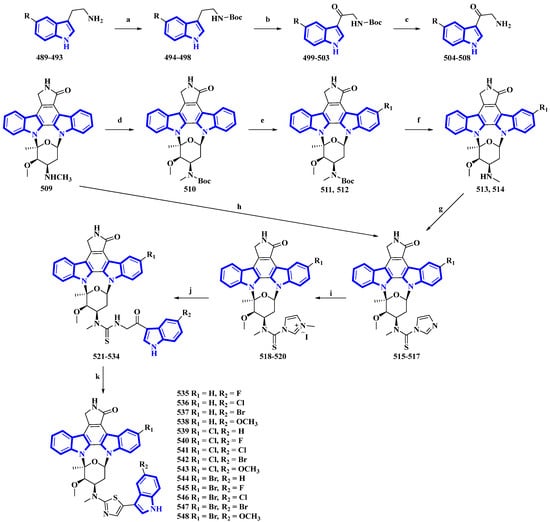
Figure 25.
Synthesis of fradcarbazole A derivatives 535 to 548. (a) Boc2O, Et3N, THF, 10 °C, 83–99%; (b) DDQ, THF/H2O 0 °C, 58–81%; (c) TFA, 10 °C, 72–94%; (d) Boc2O, Et3N, THF, 0 °C, 76%; (e) NBS, CH3OH, DCM, 0 °C, 94%, or NCS, CH3OH, DCM, rt, 48%; (f) TFA, DCM, 0 °C, 50–93%; (g) TCDI, Et3N, DCM, rt, 71–88%; (h) TCDI, Et3N, DCM, rt, 82%; (i) CH3I, CH3CN, rt, 68–74%; (j) 504–508, Et3N, DMF, rt, 38–56%; (k) (CF3CO)2O, DCM, EtOH, 0 °C, 47–81%.
(−)-Melodinine K (556), a complex bisindole alkaloid, was semi-synthesized, using (−)-tabersonine as the starting reagent. The aspidosperma–aspidosperma-type alkaloid was isolated from the Melodinus tenuicaudatus plant in 2010. Its cytotoxicity was tested by the same group and was reported to be more potent than cisplatin and vinorelbine in four of the five cancer cell lines [97]. The synthetic route adopted had six steps in its most extended linear sequence (eight steps overall) and afforded compound 556 with a 4% yield (Figure 26) [98]. While planning its synthesis, 556 was divided into two fragments, both of which could be synthesized from (−)-tabersonine (549). The total synthesis of 549 was accomplished by the same group in 2013 [99]. However, this precursor was mainly isolated from the seeds of Voacanga africana (a small tree). It was bio-transformed into compound 550 using T16H yeast and allylated by allyl bromide and K2CO3 in DMF, resulting in 551, the northern fragment of compound 556. Separately, compound 549 was protected using TrocCl, affording 552, which was further converted to 553 by TFA and m-CPBA. Compound 553 was reacted with m-CPBA in DCM, producing its N-oxide analog 554. Compound 554 was treated with TFAA in DCM and coupled with 551 in a Polonovski–Potier reaction, which resulted in 555. Compound 555 was deprotected by treating with Pd(PPh3)4, affording 556 [98].
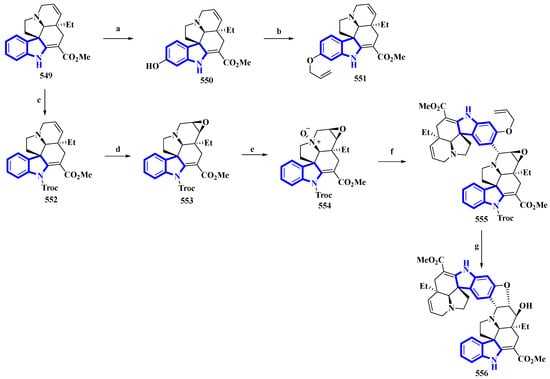
Figure 26.
The semi-synthesis of (−)-melodinine K 556. (a) T16H yeast, 64%; (b) Allyl bromide, K2CO3, DMF, 68%; (c) NaH, TrocCl, THF/DMF 0 °C, 92%; (d) TFA then m-CPBA, DCM, −10 °C then rt, 38%; (e) m-CPBA, DCM, 0 °C, 36%; (f) (1) TFAA, DCM, 0 °C to rt and (2) 551, DCM, 78%; (g) (1) Pd(PPh3)4, pyrrolidine, DCM, rt and (2) Zn, KH2PO4, THF, 60 °C, 40%.
5-Methylpsilocybin (561), a novel analog of the psychedelic psilocybin, was produced by enzymatically phosphorylating its synthetic precursor, 5-methylpsilocin (560). The 4-hydroxytryptamine kinase (PsiK) enzyme was purified from the Psilocybe cubensis fungus. On the basis of the amount of 561 isolated, the overall yield was 28% (Figure 27) [100]. 5-Methyl-1H-indol-4-ol (557) was acetylated with acetic anhydride and NaHCO3 in toluene into compound 558. It was then treated with oxalyl chloride, followed by dimethylamine in THF, to result in 559, which, on reduction by LiAlH4, resulted in 560. Compound 560 was incubated with PsiK and ATP for 16 h; the analysis of the mixture by HPLC indicated that 90% of it was successfully converted into 5-methylpsilocybin (561) with an overall isolated yield of only 40% [100].
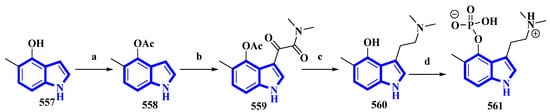
Figure 27.
The semi-synthesis of 5-methylpsilocybin 561. (a) Ac2O, NaHCO3, toluene, 97%; (b) (1) oxalyl chloride and (2) (CH3)2NH, THF, 77%; (c) LiAlH4, THF, 84%; (d) PsiK, ATP, MgCl2, H2O, 16 h, 44%.

Table 2.
Bioactivities of all novel indole alkaloids discussed in the article. (a) Tested as a mixture; (b) KB/VJ300 cells grown with vincristine (0.1 μM), which did not affect their growth; (c) methicillin-resistant strain; (d) IC90; (e) IC40.
Table 2.
Bioactivities of all novel indole alkaloids discussed in the article. (a) Tested as a mixture; (b) KB/VJ300 cells grown with vincristine (0.1 μM), which did not affect their growth; (c) methicillin-resistant strain; (d) IC90; (e) IC40.
| Compound | Cytotoxicity (GI50) µM [Cell Line] | Ref. |
|---|---|---|
| 1 | 38 [Huh7], 32 [LN229], 19 [HCT116], 16 [MGC803], 16 [A549], 21 [MDA231] | [20] |
| 15 | 1.1 [KB/S], 4.1 [KB/VJ300 b], 4.7 [PC-3], 4.8 [MCF7], 3.6 [MDA-MB-231], 4.3 [HCT 116], 2.2 [HT-29h] | [22] |
| 16 | 0.1 [KB/S], 1.3 [KB/VJ300 b], 7.9 [PC-3], 9.6 LNCaP, 0.6 [MCF7], 1.4 [MDA-MB-231], 1.1 [HCT 116], 0.3 [HT-29h] | [22] |
| 17 | 0.1 [KB/S], 8.6 [KB/VJ300], 1.5 [KB/VJ300 b], 0.7 [PC-3], 0.3 [MCF7], 0.4 [MDA-MB-231], 0.2 [HCT 116], 0.2 [HT-29h] | [22] |
| 18 | 0.2 [KB/S], 2.6 [LNCaP], 3.5 [MCF7], 5.4 [MDA-MB-231], 3.1 [HCT 116], 2.3 [HT-29h] | [22] |
| 24 | 3.6 [L5178Y], 8.7 [A2780b], 40 [J82], 29 [HEK-293] | [24] |
| 25 | 5.3 [L5178Y], 12 [A2780b], 42 [J82], 22 [HEK-293] | [24] |
| 26 | 5.3 [L5178Y], 28 [HEK-293] | [24] |
| 27 | 12 [A2780b], 55 [J82] | [24] |
| 28 | 32 [A2780b], 100 [J82] | [24] |
| 29 | 8.1 [L5178Y], 7.8 [A2780b], 32 [J82], 37 [HEK-293] | [24] |
| 37 | 6.2 [HT-29] | [27] |
| 51–54a | 4.1 × 10−4 [U87], 7.5 × 10−4 [SKOV3], 5.4 [MDA-MB-231 and HCT116] | [30] |
| 55 | 7.3 [HeLa] | [31] |
| 56 | 6.4 [HeLa] | [31] |
| 71 | 0.6 [KB/S], 6.7 [KB/VJ300], 0.2 [KB/VJ300 b], 8.2 [PC-3], 6.2 [LNCaP], 4.5 [MCF7], 4.5 [MDA-MB-231], 0.3 [HT-29], 7 [A549], 7.9 [MRC-5] | [35] |
| 72 | 2.8 [KB/S], 5.9 [KB/VJ300], 6.1 [PC-3], 2 [MCF7], 4.8 [MDA-MB-231], 0.07 [HT-29], 4.2 [HCT 116], 9 [A549], 9.7 [CCD-18Co] | [35] |
| 73 | 1.1 [KB/S], 3.9 [KB/VJ300B], 3.7 [LNCaP], 5.3 [MCF7], 4.8 [MDA-MB-231], 0.07 [HT-29] | [35] |
| 74 | 2.4 [KB/S], 2.9 [KB/VJ300], 3.1 [PC-3], 1.3 [MCF7], 1.4 [MDA-MB-231], 0.03 [HT-29], 2.9 [HCT 116], 6.8 [A549], 4.3 [CCD-18Co] | [35] |
| 75 | 2.5 [KB/S], 5.5 [KB/VJ300], 8.8 [KB/VJ300 b], 7.7 [MCF7], 4.1 [MDA-MB-231], 0.03 [HT-29], 1.5 [HCT 116], 3.9 [A549], 4.1 [CCD-18Co] | [35] |
| 76 | 1.7 [KB/S], 3.6 [KB/VJ300], 4.2 [KB/VJ300 b], 8.5 [MCF7], 0.02 [HT-29], 2.1 [HCT 116], 3 [A549], 5.1 [CCD-18Co] | [35] |
| 117 | 81 [HeLa] | [43] |
| 118 | 42 [HeLa] | [43] |
| 119 | 84 [Huh7], 92 [HeLa] | [43] |
| 123 | 2.45 [A549], 0.96 [Huh7], 0.93 [HeLa] | [43] |
| 145 | 7.5 [SW480] | [48] |
| 157 | 18.7 [HepG2], 28.7 [A-549] | [50] |
| 160 | 17 [U251], 4.5 [U87MG] | [51] |
| 161 | 36 [U251], 43 [U87MG] | [51] |
| 162 | 12 [U251], 2.3 [U87MG] | [51] |
| 164 | 27 [U251], 8.9 [U87MG] | [51] |
| 166 | 36 [U251], 29 [U87MG] | [51] |
| 167 | 19 [U251], 17 [U87MG] | [51] |
| 168 | 12 [U251], 7.4 [U87MG] | [51] |
| 170 | 13 [U251], 11 [U87MG] | [51] |
| 172 | 15 [U251], 19 [U87MG] | [51] |
| 187 | 6.8 [SK-MEL-28], 9.8 [SW480], 6.3 [HepG2], 9.8 [T47D] | [55] |
| 191 | 8.3 [SK-MEL-28], 7.4 [HepG2] | [55] |
| 192 | 6.7 [SK-MEL-28], 7.8 [SW480], 2.5 [HepG2], 8.7 [T47D] | [55] |
| 193 | 9.5 [T47D] | [55] |
| 211 | 5 [KB/S], 7.6 [KB/VJ300], 6.1 [PC-3], 5.9 [MDA-MB-231], 0.2 [HT-29], 4.7 [HCT 116], 2.1 [A549] | [60] |
| 217 | 44.0 [A549], 40.4 [HT-29], 37.3 [HepG2] | [63] |
| 218 | 8.2 [A549], 4.7 [A427], 8.0 [HCT116], 6.7 [HT-29], 6.2 MCF-7, 7.1 [HeLa], 9.1 [HepG2], 44.1 [LO2] | [63] |
| 219 | 13.8 [A427], 38.5 [HCT116], 20.3 [HeLa] | [63] |
| 220 | 18.9 [HeLa], 46.3 [LO2] | [63] |
| 221 | 7.9 [A549], 6.6 [A427], 10.6 [HCT116], 8.2 [HT-29], 7.2 MCF-7, 8.4 [HeLa], 9.6 [HepG2], 35.5 [LO2] | [63] |
| 222 | 9.5 [A549], 4.8 [A427], 12.2 [HCT116], 8.9 [HT-29], 6.5 MCF-7, 8.6 [HeLa], 9.1 [HepG2], 36.3 [LO2] | [63] |
| 223 | 6.9 [A549], 7.5 [A427], 11.2 [HCT116], 8.8 [HT-29], 6.8 MCF-7, 10.1 [HeLa], 10.7 [HepG2], 41.8 [LO2] | [63] |
| 224 | 21.5 [A549], 26.1 [HCT116], 41.5 [HeLa] | [63] |
| 225 | 23.3 [A549], 30.9 [HCT116], 43.8 [HT-29], 44.4 [HepG2] | [63] |
| 226 | 15.9 [A549], 21.2 [HCT116] | [63] |
| 227 | 40.4 [A549], 45.3 [HCT116] | [63] |
| 239 | 1.7 [HeLa], 1.6 [A549], 1.8 [HepG2], 1.5 [SMMC7721] | [70] |
| 242 | 7.9 [HeLa], 7.8 [A549], 8.1 [HepG2], 6.7 [SMMC7721] | [70] |
| 347 | 4 [SMMC-7721], 4.8 [HepG2], 1.8 [Bel7402], 2.8 [Huh7] | [88] |
| 348 | 2.9 [SMMC-7721], 1.4 [HepG2], 1.0 [Bel7402], 1.1 [Huh7] | [88] |
| 349 | 7.1 [SMMC-7721], 5.3 [HepG2], 4.6 [Bel7402], 6.2 [Huh7] | [88] |
| 350 | 6.3 [SMMC-7721], 5.8 [HepG2], 5.2 [Bel7402] | [88] |
| 351 | 4.1 [SMMC-7721], 3.4 [HepG2], 4.0 [Bel7402] | [88] |
| 352 | 9.5 [HepG2], 8.9 [Bel7402] | [88] |
| 535 | 0.51 [MV4-11] | [96] |
| 536 | 0.42 [MV4-11] | [96] |
| 537 | 0.41 [MV4-11] | [96] |
| 538 | 0.36 [MV4-11] | [96] |
| 539 | 0.56 [MV4-11] | [96] |
| 540 | 0.32 [MV4-11] | [96] |
| 541 | 0.59 [MV4-11] | [96] |
| 542 | 0.43 [MV4-11] | [96] |
| 543 | 0.44 [MV4-11] | [96] |
| 544 | 0.36 [MV4-11] | [96] |
| 545 | 0.96 [MV4-11] | [96] |
| 546 | 0.70 [MV4-11] | [96] |
| 547 | 0.39 [MV4-11] | [96] |
| 548 | 0.51 [MV4-11] | [96] |
| Antibacterial (MIC; µg/mL) | ||
| 1 | 25 [B. cereus], 12.5 [S. aureus c] | [20] |
| 2 | 50 [S. aureus c] | [20] |
| 8 | 12.5 [B. cereus], 25 [S. aureusc], 25 [M. lysodeikticus], 25 [B. paratyphosum], 25 [B. subtilis], 25 [E. aerogenes], 25 [S. typhi], 25 [P. vulgaris] | [20] |
| 32 | 4 [B. subtilis], 16 [S. typhimurium], 8 [M. luteus], 8 [M.phlei] | [26] |
| 33 | 32 [B. subtilis], 32 [S. typhimurium], 32 [M. luteus], 32 [M. phlei] | [26] |
| 40 | 8 [X. o. pv. oryzae], 32 [R. solanacearum], 32 [X. o. pv. oryzicola], 128 [P. s. pv. lachrymans] | [28] |
| 42 | 32 [X. o. pv. oryzae] | [28] |
| 44 | 32 [X. o. pv. oryzae], 128 [R. solanacearum], 64 [X. o. pv. oryzicola] | [28] |
| 45 | 64 [X. o. pv. oryzae] | [28] |
| 64 | 25 [H. influenzae ATCC 4] | [33] |
| 65 | 50 [H. influenzae ATCC 4] | [33] |
| 66 | 25 [H. influenzae ATCC 4] | [33] |
| 231 | 25 [M. smegmatis], 25 [M. abscessus], 25 [M. bovis] | [65] |
| Antifungal (MIC; µg/mL) | ||
| 1 | 6.25 [A. fragariae], 25 [C. cassiicola], 25 [A. alternata], 6.25 [B. cinereal Pers], 25 [C. personata], 6.25 [V. dahliaekleb], 25 [S. sclerotiorum] | [20] |
| 2 | 25 [A. fragariae] | [20] |
| 3 | 25 [A. fragariae] | [20] |
| 4 | 50 [A. fragariae] | [20] |
| 5 | 6.25 [A. fragariae] | [20] |
| 6 | 25 [A. fragariae] | [20] |
| 7 | 6.25 [A. fragariae] | [20] |
| 9 | 50 [A. fragariae] | [20] |
| 10 | 25 [A. fragariae] | [20] |
| 229 | 50 [F. oxysporum] | [64] |
| Antiplasmodial (IC50 µM) | ||
| 63 | 6.1 d [P. falciparum] | [32] |
| 110 | 8.7 [P. falciparum FcB1] | [42] |
| 111 | 9.5 [P. falciparum FcB1] | [42] |
| 112 | 2.6 [P. falciparum FcB1] | [42] |
| 113 | 5.2 [P. falciparum FcB1] | [42] |
| 114 | 3.0 [P. falciparum FcB1] | [42] |
| 210 | 1.05 [P. falciparum FcB1] | [59] |
| 234 | 15.1 [P. falciparum 3D7] | [67] |
| Antiviral (EC50 µM) | ||
| 109 | 70 [HSV-2] | [41] |
| 137 | 5.7 [HCV] | [46] |
| 196 | 7.5 [ZIKV] | [56] |
| 197 | 38 [ZIKV] | [56] |
| 198 | 50 [ZIKV] | [56] |
| 249 | 9.4 [H1N1], 6.7 [RSV] | [72] |
| 250 | 3.7 [H1N1], 2.8 [RSV] | [72] |
| Antifouling activity (EC50 µM) | ||
| 486 | 2.2 [A. improvisus] | [93] |
| 487 | 0.7 [A. improvisus] | [93] |
| Inhibition of protein phosphatases (IC50 µM) | ||
| 67 | 14 [PTP1B], 38 [PTPsigma] | [34] |
| 68 | 27 [PTP1B] | [34] |
| 70 | 23 [PTP1B], 35 [TCPTP] | [34] |
| Inhibition of DC secretion of IL-12p40 (% inhibition at 10 µg/mL) | ||
| 177 | 38% | [53] |
| 185 | 36% | [53] |
| Promotion of DC secretion of IL-10 (% promotion at 10 µg/mL) | ||
| 176 | 19% | [53] |
| Promotion of hBM-MSC secretion of adiponectin (EC50 µM) | ||
| 212 | 9.86 | [61] |
| 213 | 6.20 | [61] |
| Inhibition of LPS-induced B-cell proliferation (IC50 µM) | ||
| 237 | 0.38 | [69] |
| 238 | 47.37 | [69] |
| Analgesic activity | ||
| 205 | 64.7% (1 mg/kg) | [58] |
| 206 | 50% (5 mg/kg) | [58] |
| 207 | 67.6% (0.04 mg/kg), 76.1% (0.2 mg/kg) | [58] |
| 208 | 55% (5 mg/kg) | [58] |
| 209 | 53% (5 mg/kg) | [58] |
| 5-HT1A receptor agonist (EC50 µM) | ||
| 83 | 10 | [37] |
| 84 | 2.2 | [37] |
| 86 | 0.1 | [37] |
| 87 | 54 | [37] |
| Inhibition of HDACs (EC50 nM) | ||
| 347 | 32 [HDAC1], 125 [HDAC3], 17 [HDAC6] | [88] |
| 348 | 27 [HDAC1], 148 [HDAC3], 13 [HDAC6] | [88] |
| 349 | 61 [HDAC1] | [88] |
| 350 | 75 [HDAC1] | [88] |
| 351 | 57 [HDAC1] | [88] |
| 352 | 125 [HDAC1] | [88] |
| Inhibition of porcine SCR (EC50 µM) | ||
| 411 | 10 | [90] |
| 438 | 1.34 | [90] |
| 439 | 1.75 | [90] |
| 440 | 1.46 | [90] |
| 442 | 0.70 | [90] |
| Inhibition of acetylcholinesterase (IC50 µM) | ||
| 98 | 10.5 | [38] |
| Inhibition of quorum sensing (MIC µg/well) | ||
| 104 | 32 [C. violaceum CV026] | [39] |
| 105 | 32 [C. violaceum CV026] | [39] |
| Glucose uptake activity in L6 myoblasts at 50 µM (mmol/L) | ||
| 203 | 5 | [57] |
| 204 | 5 | [57] |
| Triglyceride accumulation promotion in 3T3-L1 cells (EC50 µM) | ||
| 126 | 1.03 | [44] |
| Inhibition of MAO-A (IC50 µM) | ||
| 127 | 0.9 [MAO-A] | [45] |
| Inhibition of misc. enzymes (inhibition % at 10 µM) | ||
| 127 | 31 [MAO-B], 21 [BChE] | [45] |
| Inhibition of mushroom tyrosinase (IC50 µM) | ||
| 141 | 33.2 | [47] |
| Inhibition of autophagic flux (EC50 µM) | ||
| 145 | 20.2 | [48] |
| Vasorelaxant activity (EC50 µM) | ||
| 158 | 2.8 | [50] |
| 159 | 2.4 | [50] |
| Potentiation of imipenem activity (4 ug/mL of imipenem; MIC µg/mL) | ||
| 173 | 8–16 [S. aureus c] | [52] |
| 174 | 2–4 [S. aureus c] | [52] |
| Inhibition of RANKL-induced multinuclear osteoclasts (IC50 µM) | ||
| 233 | 10.5 [RAW264] | [66] |
| 294 | 50 e [RAW264] | [66] |
| 295 | 25.6 [RAW264] | [66] |
| 296 | 6.3 [RAW264] | [66] |
| 297 | 16.8 [RAW264] | [66] |
| 298 | 35.4 [RAW264] | [66] |
| 299 | 40.0 [RAW264] | [66] |
| 300 | 11.7 [RAW264] | [66] |
| 301 | 7.9 [RAW264] | [66] |
| 302 | 11.1 [RAW264] | [66] |
| 305 | 8.1 [RAW264] | [66] |
| 306 | 10.0 [RAW264] | [66] |
| 307 | 13.9 [RAW264] | [66] |
| 308 | 5.9 [RAW264] | [66] |
4. Bioactivities of Novel Indole Alkaloids
Table 2 only includes the bioactivities of novel indole alkaloids that were isolated, synthesized, and tested from 2019–2022 to enable an extensive structure–activity relationship analysis; the activities of previously reported natural products (which were tested alongside the novel indole alkaloids) are also included. These compounds are referred to by their trivial names. Furthermore, since most articles presented a range of biological activities, we only summarize these activities in this section.
The diterpenoids 1–10 only possessed mild bioactivities. The most potent compound, 1, showed moderate antifungal and antibacterial activities and weak cytotoxicity. Alkaloids 2–7 and 9–10 showed weak antifungal activities, while 8 displayed moderate antibacterial activity. None of the compounds exhibited inhibitory activity against three pathogenic fungi (F. oxysporum Schlecht, A. solani Sorauer, and R. solani) [20]. Compounds 15–18 displayed potent (sub-micromolar) cytotoxicities against many cell lines, especially against KB/S cells [22]. Compounds 19 to 23 were tested for cytotoxicity against L5178Y cells using the MTT assay. However, all of these were inactive. Previously reported okaramines were also tested, with okaramine H, being the most potent, followed by okaramine G, okaramine A, and okaramine C (IC50 = 4.0, 12.8, 13.8, and 14.7 μM, respectively). Additionally, okaramine J was inactive. Hydroxylation at C-2 (19 vs. okaramine C) or cleavage between 1-N and C-8a (20 vs. okaramine C) caused a complete loss of cytotoxicity. When an α,α–dimethylallyl group was attached to N-8, the cleavage between N-3′ and C-4′ (okaramine G vs. okaramine A) or reduction of Δ1′ (okaramine A vs. okaramine C) resulted in weak suppression of cytotoxicity. However, when an isoprenyl group was attached at C-7, reduction of Δ1′ (3 or okaramine J vs. okaramine H) completely suppressed cytotoxicity [23]. Compounds 24–29 displayed moderate cytotoxicities against L5187Y cells and weak cytotoxicities against others. Other alkaloids, including 7-hydroxy-13-dehydroxypaxilline, pyrapaxilline, shearinine P, 7-methoxyshearinine P, shearinine Q, and paspalicine were also tested (IC50 = 6.2, 10.9, 7.6 μM; latter three inactive). Compound 24’s aromatic ring C was the major determinant in producing its cytotoxicity (compound 24 vs. paspalicine). Additionally, the presence of Δ13 increased the activity of paspalinine derivatives (compound 25 vs. paspalicine) but did not affect the activity of paxillin-type alkaloids. Thus, compound 26 received no benefit from such a motif (compound 26 vs. 7-hydroxy-13-dehydroxypaxilline). Furthermore, the presence of Δ6 slightly enhanced the cytotoxicity of janthitremane derivatives (compound 29 vs. pyrapaxilline). The cleavage of the keto-amide ring also decreased activity (shearinine Q vs. shearinine P). Similarly, replacing a proton at C-7 with a CH3 group suppressed activity (pyrapaxilline vs. compound 28 and shearinine P vs. 7-methoxyshearinine P) [24].
Compounds 30 to 31 showed no antifungal, antibacterial, and cytotoxicity activities. A congener, baraphenazine E, exhibited some cytotoxicity and antibacterial activity, indicating that its C-11′-amide may be responsible for its moderately potent activities, as the other alkaloids contained a carboxylic acid group instead. All compounds showed no activity or toxicity in an axolotl embryo tail regeneration assay [25]. The chlorinated cyclic hexapeptides, 32–36, exhibited no cytotoxicity (tested on HeLa cells). However, compounds 32 and 33 displayed moderate and weak antibacterial activities, respectively, as replacing the phenylalanine residue (32) with a leucine residue (33) could decrease antibacterial activity. Similarly, replacing the isoleucine residue (32 vs. 33) with a valine residue (35 vs. 36) completely suppressed activity. Compounds 38 and 39 showed no cytotoxicity, while compound 37 demonstrated moderate cytotoxicity on HT-29 cells [26]. Compounds 41 and 43 showed no antibacterial activity, while compounds 40, 42, 44, and 45 displayed moderate to weak inhibitory activity against Xanthomonas oryzaepv. Oryzae (rice bacterial leaf blight). Treatment with compound 40 at 100 µg/mL offered 61% protection, while treatment with 200 µg/mL offered 82% protection from the same pathogen. Furthermore, chaetogline A displayed antifungal properties against S. sclerotiorum (EC50 = 10.3 μg/mL), while 40 to 45 were inactive. The results indicated that the C-19 carboxylic acid in compound 44 is necessary for its antibacterial activity, while the C-19 ester in chaetogline A is essential for its antifungal activity. Similarly, a second N-methylated indole increased antibacterial activity (41 vs. 42) [28].
A mixture of compounds 51–54 was evaluated. It was found to be extremely potent and selective inhibitors of the U87 and SKOV3 cell lines with an EC50 of 0.41 nM and 0.75 nM, respectively (SI > 7000; cytotoxicity was also tested against MDA-MB-221 and HCT116 cell lines). The mechanism of action is not proven similar to that of plinabulin (a similar diketopiperazine) due to the nonexistence of vascular disruption and tubulin binding studies [30]. Among compounds 55–62, only 55 and 56 displayed moderate cytotoxicity, while all other tested indole alkaloids were inactive. Comparing compound 60 with asperversiamide H (both inactive) demonstrated that replacing the C-11 proton with a methoxy group did not significantly affect activity [31]. Compound 63 demonstrated potent antiplasmodial activity, inhibiting more than 90% of liver-stage growth of P. falciparum at 6.1 μM. It showed similar antimalarial activity to primaquine [32]. Compounds 64–66 exhibited weak antibacterial activity against the ATCC-4 strain of H. influenzae, and compounds 64 and 66 showed slightly more activity than compound 65. The results indicated that replacing the C-19α proton with a hydroxy group did not alter the bioactivities of (3β,6β,23-trihydroxyolean-12-en-28-oic acid vs. 3β,6β,19α,23-tetrahydroxyolean-12-en-28-oic acid; MIC = 18.8 μg/mL for both), suggesting that it is not a determinant for activities. Additionally, naucleidinal most potently inhibited H. influenzae strain ATCC-4 (MIC = 3.1 μg/mL) [33].
Compounds 67–70 are weak inhibitors of PTP1B, while compound 67 is also weakly inhibiting PTP sigma and 70 inhibiting TCPTP. Compound 69 presented no PTP inhibition, and all compounds did not inhibit VHR. Similarly, all four alkaloids did not display any cytotoxicities [34]. Compounds 71–76 showed strong cytotoxicities against multiple cell lines, especially HT-29 (76 IC50: 20 nM) and KB/S (71 IC50: 0.6 μM). Alkaloid 77 was not tested [35].
Compounds 80, 81, 82, 85, and 88 displayed no activities against the 5-HT1A receptor, a therapeutic target in CNS diseases. Of the remaining four compounds, 83 and 87 displayed weak to moderate activity, while compounds 84 and 86 showed potent agonism. The results demonstrate the importance of stereochemistry in biological systems; 84 and 86 contain a C-16S, whereas their diastereomers 85 and 87 were inactive, having a C-16R configuration. Additionally, a vinyl group at C-20 instead of an ethyl group (86 vs. 84) increased bioactivity. Furthermore, docking studies showed that 84 interacted with the 5-HT1A receptor primarily through its two hydrogen bonds between the indole-nitrogen (NH-1) and Asp116, Tyr390. Alternatively, the interaction of compound 86 comprised only one hydrogen bond between the indole-nitrogen (NH-1) and Asn386 [37].
Compounds 89–98 were assessed for cytotoxicity and acetylcholinesterase (AChE) inhibition; none of the alkaloids showed cytotoxicity. Compound 98 moderately inhibited AChE, and docking studies were used to investigate its interactions with the enzyme’s active site. The C-22 carbonyl, N-1, N-4, and the hydroxy group at C-5′ and C-6′ formed hydrogen bonds with Tyr124, Tyr341, Arg296, and His287, respectively, indicating that both the carbonyl group at C-22 and the indole ring’s N-1 were important for its activity [38]. Compounds 99–105 were tested for quorum-sensing inhibition, cytotoxicity, and antibacterial activity. The only bioactive alkaloids were 104 and 105, which displayed only weak quorum-sensing inhibition in C. violaceum strain CV026. Despite stereochemistry being essential in determining bioactivities, enantiomers 103 and 104 displayed the same activities. Additionally, scequinadoline G inhibited quorum-sensing most potently (MIC = 16 μg/well) [39].
Compounds 106–108 showed no cytotoxicity and antibacterial activity, possibly due to their self-fluorescence. Slight modifications such as reducing Δ2 (solitumine B vs. solitumine A; both inactive) or replacing the C-19-bonded NH2 with an –OH (106 vs. 107) showed no detectable differences, suggesting that they are not significant determinants of bioactivity [40]. Compound 109 displayed weak antiviral activity against the herpes simplex virus 2 (HSV-2). At the same time, other amphiepicoccins, such as amphiepicoccin B, C, D, and H, were also tested for their antiviral and antibacterial (against B. subtilis) activities. Amphiepicoccin C and H displayed weak antiviral activities (IC50 = 64 and 29 μM), while amphiepicoccin D and H showed moderate to weak antibacterial activities (MIC = 13 and 25 μM). The rest were inactive. The carbonyl at C-5 is a critical determinant of bioactivity if replacing it with a hydroxy group significantly decreased activity (amphiepicoccin C vs. B). Interestingly, reducing the number of sulfur atoms in the C-2′ to C-7′ sulfide bridge from two to one (amphiepicoccin D vs. H) meaningfully increased antiviral activity but moderately decreased antibacterial activity [41].
Compounds 110–114 displayed moderate to strong antiparasitic activities against the P. falciparum strain FcB1. No cytotoxicity was detected against MRC-5 cells. The results showed that the reduction of Δ19′ slightly boosted activity (111 vs. 110), but replacing C-22 with a proton significantly suppressed bioactivity (112 vs. 113) [42]. Compounds 115–123 were evaluated for cytotoxicity, but only 123 showed potent activity, while 117–119 showed only weak cytotoxicity. Considering compound 123 had a sulfide bridge, compounds 118 and 119 contained an SCH3-group at C-6, and compounds 120 to 122 were sulfur-free analogs, especially the sulfide bridge greatly influenced the activity of diketopiperazine [43].
Compounds 124–126 were tested for their ability to promote triglyceride accumulation in 3T3-L1 cells. While compounds 124 and 125 showed no significant activity, compound 126 was a moderate promoter. Additionally, no compound exhibited toxicity against 3T3-L1 cells at 50 µM. Analyzing the structure–activity relationships yielded two important moieties responsible for the bioactivities of compounds: the pyrazino [2,1-b]quinazoline-3,6-dione core and the unsaturated isopropyl group at C-3. Both these motifs are present in scequinadoline D, scequinadoline E (most potent among the compounds tested; EC50 = 0.27 and 0.36 µM), while compound 126 only contains the tricyclic core. Suppose the core was replaced with another tricyclic system (quinazoline–pyridine–tetrahydrofuran) or a quinazolinone ring suppressed activity (124, scedapin C, and scedapin A; all inactive). Replacing the unsaturated isopropyl group with its saturated analog also reduced activity (125, fiscalin C, epi-fiscalin C, scequinadoline A, scequinadoline B, and fiscalin B; all inactive). Furthermore, substituents at C-22 also modulated activity, with ethyl and methyl groups resulting in the most potent analogs (scequinadoline D, scequinadoline E) [44].
Among compounds 127–136, only compounds 127–129 and 132 were tested for their activities based on chemometric results. Among those three, only compound 127 was active, potently inhibiting MAO-A, while weakly inhibiting MAO-B and BChE. Cimitrypazepine and fargesine were also weak to moderate inhibitors of BChE (18% and 41% inhibition at 10 µM) and potent inhibitors of MAO-A (IC50 = 1.4 μM for both). Interestingly, replacing the tertiary amine in cimitrypazepine with an N-oxide (as in fargesine) significantly increased BChE inhibition while not affecting MAO-A inhibition. Furthermore, replacing the tertiary amine with a quaternary ammonium ion, as in compound 127, slightly boosted both BChE and MAO-A inhibition. Conversely, the glucosylated alkaloids 129 and 132 were inactive due to the bulkiness of the glucose group, which hampered the interaction of the alkaloids with the enzyme’s active site [45].
Compound 137 displayed no antibacterial or cytotoxic activities but moderately and selectively inhibited the Hepatitis C virus (SI > 35) [46]. Compounds 138–141 were tested for antibacterial, antifungal activities, and α-glucosidase inhibition but were inactive. Compound 141 was weak against mushroom tyrosinases inhibitor, and it is the first example of inhibiting tyrosinases by a thiodiketopiperazine. Changing the steric configuration of C-2 from R to S increased bioactivity (139 vs. 141). Additionally, the presence of an OCH3/SCH3 group at C-2′ and an N-4′-CH3 group is necessary for the bioactivities of compounds. Furthermore, a second indole group significantly boosted bioactivity (Leptosin D, IC50 = 28.4 μM) [47].
Among compounds 142–150, only 145 showed moderate cytotoxicity against SW480 while being inactive against other cell lines. The cytotoxicity of tabernaricatine C was also tested, and it potently inhibited HL-60 and SW480 cells (EC50 = 3.2 and 3.5 μM). Previously, other alkaloids such as taburnaemine D, tabernaemine I, tabercorine B, tabernaelegantine B, conodurine, and tabernaricatine C (among others), along with compounds 142–150, were tested for their inhibitory activity against autophagic flux. However, only bisindoles were active, with the most potent inhibitor being taburnaemine D (EC50 = 12.9 μM). The linkage pattern of the two indole units might be a crucial factor in determining the activities of the compound [48].
Among compounds 155–159, only compound 157 showed weak cytotoxicity. The vincamine-type alkaloids 158 and 159 demonstrated vasorelaxant activity. Replacement of the C-11-bonded –OH group in compound 158 with a proton (Δ14-vincamine; EC50 = 1.9 μM) or a methoxy group (Δ14-vincine; EC50 = 2.2 μM) exhibited slightly increased activity. Interestingly, replacing the same group in its diastereomer 159 with a proton decreased activity (16-epi-Δ14-vincamine; EC50 = 3.8 μM) while replacing it with a methoxy group increased activity (16-epi-Δ14-vincine; EC50 = 1.9μM). Furthermore, oxidizing C-3 greatly improved vasorelaxant activity (tabersonine vs. 3-oxotabersonine; EC50 >10 and 0.8 μM) [50].
Compounds 160–172 displayed weak to moderate cytotoxicities when tested against cell lines U251 and U87MG, and none of them exhibited antimicrobial activities. Nonetheless, results indicated that the methyl substituent on N-1 (the nitrogen of the indole ring) was an important determinant of activity as replacing it with a proton drastically lowered activity (160, 162, or 170 vs. 161, 163, or 171, respectively). Similarly, an OH group at C-13 completely suppressed the cytotoxicity (164 vs. 165), and an α-OH group at C-16 deactivated 169 compared to 168. Furthermore, replacing the C-9 carbonyl with an acetyl group (resulting in the oxidation of the C-9-N-2 bond) meaningfully boosted the activity, which is evident by comparing compound 171 with marine carboline C (EC50 = 12, 13 μM against U251, U87MG cells) and to a lower extent 170 with 168. Furthermore, the steric configurations of the substituents also modulated the activity by replacing the β-OH group on C-16 with an α-OH group, causing a noteworthy increase in cytotoxicity (166 vs. 167). Interestingly, changing the geometric configuration of Δ15 from E- to Z-configuration slightly boosted the potency of methylated-indole analogs (160 vs. 162). Nonetheless, non-methylated indole analogs (161 vs. 163) were inactive [51].
Compounds 173 and 174 are inactive in vitro antibacterial assays. Nevertheless, they enhanced the activity if co-administered with imipenem (imipenem; MIC = 16–32 μg/mL). Considering that krisynomycin (MIC = 0.25–0.5 μg/mL with 4 μg/mL of imipenem) was more chlorinated than 174, which was more chlorinated than 173, the extent of chlorination was thought to be a significant cause of the imipenem-potentiating effects of the compound. Furthermore, chlorination in the alkaloid resulted in increased potentiating effects of imipenem at lower doses [52].
Among compounds 175–185, only 177 and 185 inhibited the DC secretion of IL-12p40, while 176 increased the secretion of DC cytokine IL-10. Compounds 177 and 185 did not affect the viability of DCs, demonstrating that the decrease in secretion was not due to cell damage. The results indicated that the nature of the C-14 substituent did not affect bioactivity noticeably. Replacing a proton with a hydroxy (180 or 182 vs. 181 or 183) or methoxy (182 vs. 184) did not meaningfully affect activity. Furthermore, pyrrolo [2,3-b]indole (180 to 184) and oxindole (178 and 179) alkaloids were found to be inactive. On the other hand, 6-bromoindole alkaloids (177 and 185) were active, suggesting that modified indoles decreased the bioactivities of compounds [53]. Compound 186 demonstrated no antiplasmodial activity against the ring stage of the P. falciparum strain 3D7 nor the P. falciparum strain Dd2. It was non-cytotoxic against HEK293 cells, too [54].
The cytotoxicities of compounds 187–195 against various cell lines were determined. Molecules 187, 191, 192, and 193 exhibited moderate cytotoxicities. The only difference between 190 and 191 was the oxidation of C-18, suggesting that the absence of the carbonyl group significantly increases the compound’s activity. Furthermore, replacing a proton with a hydroxy group at C-2 causes a significantly suppressed activity (192 vs. 191). Stereochemistry (192 vs. 194 and 193 vs. 195) was also a foremost determinant of the activities of alkaloids; switching C-14′ from an S to R configuration significantly reduced their activities [55].
Compounds 196–200 were not cytotoxic against the A549 cell line. Similarly, only 196, 197, and 198 inhibited the Zika virus. Previously reported activities of natural products suggested that the F-ring was a significant factor of antiviral activity; an open F-ring afforded an inactive compound (emindole SB), while a furan F-ring yielded the most potent compound in the series, JBIR-03 (EC50 = 4.2 μM). However, compared to JBIR-03, the weaker activities of fusaindoterpene A (EC50 = 12 μM) and asporyzin A (EC50 = 18 μM) indicated that B- and C-rings also modulate bioactivity. Furthermore, adding an oxygen atom into the eight-membered ring of asporyzin A (as in fusaindoterpene A) slightly increased activity [56].
Compounds 201–204 were tested against the HEI-OC1 cell line to determine their protecting ability. Compound 201 displayed potent protecting effects (optical density = 0.7, compared to the blank control’s 0.5), while the other compounds were inactive. The alkaloids were also tested to determine if they stimulated glucose uptake in cultured rat L6 myoblasts. Only compounds 203 and 204 promoted glucose uptake in the myoblasts. Among the non-novel natural products tested, beauverolide Ka displayed both protective ability (optical density = 0.69) and promotion of glucose uptake (9 mmol/L). The results indicated that a hydroxy substituent on C-13 (para-substituent of the phenyl ring) instead of a proton decreased protective ability significantly (beauverolide Ka vs. 203 and 201 vs. 204). Interestingly, replacing the indole ring, the C-2 substituents, or the C-13 proton in beauverolide Ka drastically decreased its glucose uptake promotion activity, evident when comparing it to beauveamide E (inactive), 201, and 203 [57].
The analgesic effects of compounds 205–209 were studied, with compound 207 exhibiting more potent analgesic activities than morphine at 0.04 and 0.2 mg/kg doses. Since 205 was more active than humantenine N4-oxide and humantenine, its oxazolidinone ring may enhance bioactivity. Conversely, compound 207 and N-desmethoxyhumantenine were more potent than alkaloids with an N-1-OCH3 motif (206, humantenine N4-oxide, 11-hydroxyhumantenine, humantenine, and humantenirine), indicating that the group might cause a decline in activity. Moreover, compound 208 displayed lower activity than epi-koumidine and koumidine, inferring that the N4-oxide (not possessed by the latter two) decreased the activity of sarpagine-type alkaloids. Furthermore, compound 206, 11-hydroxyhumantenine, and humantenirine contained a hydroxy or methoxy substituent at C-11and showed less potent activities, suggesting that these groups decreased the activities of humantenine-type alkaloids [58]. Compound 210 demonstrated no cytotoxicity against the MRC-5 cell line; however, it potently inhibited P. falciparum strain FcB1. Comparing its antiplasmodial activity with vobasine (IC50 = 22.5 μM) revealed that the C-3 bonded –SH group is necessary for the activity of vobasane-type alkaloids; nevertheless, vobasine contains a carbonyl. Additionally, the discovery of compound 210 is of particular biosynthetic interest, as it was proposed to be a step in forming the ion brunonines A and B [59].
Compound 211 displayed moderate to robust cytotoxicities against various cell lines and selectivity (SI > 5) when tested against HT-29 and CCD-18Co cells. It induced mitochondrial apoptosis, and in vitro tubulin polymerization assays confirmed it as a microtubule-stabilizing agent. It was also predicted to bind at the taxol-binding site in β-tubulin. The docking models showed that compound 211 had several nonpolar interactions and hydrogen bonds with the amino-acid residues in the binding pocket, resulting in its calculated interaction strength being more significant than paclitaxel, which was consistent with the observed microtubule-stabilizing activity. Compound 211 also satisfied the criterion of drug-likeness, with a score of 3.28 (vincristine 4.03, Taxol 0.82), well within the proposed acceptable range of 2–7 [60,101].
The adiponectin secretion-promoting activities of compounds 212–214 and their synthetic isomers, 286–290, were investigated in hBM-MSCs. However, the EC50 of compounds 214 and 286–290 was greater than 10 μM. Compounds 212 and 213 were more potent than bezafibrate and increased lipid accumulation in differentiated adipocytes. The results suggested that they improved insulin sensitivity (like pioglitazone). Comparing the activities of the alkaloids demonstrated the importance of handedness in biological systems; enantiomers of the compound 212 and 286 displayed significantly less potency. A bulky phenyl group at C-2′ may suppress bioactivity by hindering the interactions of compounds with the target’s active sites (213 vs. 214) [61].
Docking studies showed the indole acetonitrile in compounds 215 and 216, and other compounds such as 3-indole acetonitrile and 4-methoxy-3-indole acetonitrile, which strongly bind with the HA and form hydrogen bonds with the THR59F and GLU90D residues. This hydrogen bonding possibly modulated the function of HA and produced the supposedly antiviral activities of compounds. Nevertheless, despite reporting compound 215 as an inhibitor of HA, no quantitative information is available [62].
Compounds 217–227 displayed weak to moderate cytotoxicities against various cell lines, while compound 228 was inactive. Western blot analysis showed that compound 218 induced apoptosis of MCF-7 cells via the Bcl-2/caspase-3/PARP pathway. Results revealed that reducing Δ19 caused a decrease in cytotoxicity (218 vs. 219 and 223 vs. 220), which infers that structural rigidity is required to exhibit cytotoxicity. Interestingly, the geometric conformation of Δ19 did not significantly influence activity, as evident when comparing the activities of the Z-isomer (271) and the E-isomer (272) [63]. No cytotoxicity or antibacterial activity was detected when compounds 229 and 230 were tested. Compound 229 displayed weak antifungal activity against F. oxysporum [64]. Compounds 231 and 232 showed no cytotoxicity, and only compound 231 showed weak antimycobacterial activity. Replacing the C-12 proton in compound 231 with a methoxy group as in 232 completely lost the activity [65].
Compound 233 and its novel synthetic derivatives (294–308) were tested for their inhibition of RANKL-induced multinuclear osteoclasts in RAW264 cells. The weaker activity of 295 compared to compound 233 demonstrated the importance of Δ2 for bioactivity. If the methylenedioxy ring was replaced with the two methoxy groups, as in 296, slightly enhanced activity was observed. The results also indicated that the presence and positions of bromine atoms are a significant determinant of bioactivity as 5,6-methylenedioxyindole. Shifting the position of bromines as in 294 or replacing them with chlorines as in 299 significantly suppressed the activities. While the bromine at C-4 was believed essential for activity, the bromine at C-7 slightly enhanced activity when C-4 was unsubstituted (298 compared to 5,6-methylenedioxyindole). Moreover, the presence of chlorines slightly improved potency when bromines were absent (299 versus 5,6-methylenedioxyindole). Similarly, N-acyl derivatives were used to study the effects of varying chain lengths on activity. Compounds 300–302 with C2–C8-alkyl chains showed activity, but compounds 303 (C14-alkyl chain) and 304 (benzoyl derivative) showed no activity, demonstrating that long or bulky groups cause a decline in activity. Furthermore, the effects of substituents at C-3 were studied by comparing the activities of compounds 305, 306, and 307, which contained methyl, hydroxymethyl, and formyl substituents, respectively. Although compounds 305 and 306 had similar activities as 233, compound 307 was less potent. Compound 308, a tryptamine derivative, was more potent but was found cytotoxic at 25 μM, while none of the other derivatives showed toxicity even at 50 μM [66].
Compound 234 displayed weak antiplasmodial activity against P. falciparum strain 3D7 and very weakly inhibited the growth of HEK293 cells (32.5% inhibition at 40 μM) [67].
While compounds 235 and 236 were not tested, other compounds—(1H-indol-3-yl)-oxoacetamide, and indole-3-aldehyde—were also produced by E. coli harboring the EDB cluster and were examined for their ability to repel C. elegans. The two molecules had a chemotaxis index of −0.18 and −0.08 at 0.2 mM, implying moderate repulsion and confirming that P. fluorescens NZI7′s EDB cluster enables it to convert external tryptophan into indole-containing molecules that repel their predators, C. elegans [68]. Compound 237 (and, to a much lesser extent, 238) inhibited LPS-induced B-cell proliferation, with the former displaying high selectivity (SI > 548). The results indicated that the C-6 methoxy group of compound 237 is essential for activity. Replacing it with a proton, as in 238, led to an increase in toxicity and a sharp decline in inhibitory activity. Compound 237 was also inactive against the four tyrosine kinases ibrutinib (the positive control) inhibited, suggesting that compound 237 exhibited its activity via a new mechanism. However, neither compound inhibited concanavalin-A-induced T-cell proliferation [69].
Among compounds, 239–244, 239 and 242 exhibited cytotoxic effects, indicating that the N-OH group was a key determinant of activity (239 compared to 240). Similarly, compound 242 had more potent activity than compound 241, suggesting that the more rigid cyclized form of compound 242 improved the cytotoxicity. Molecule 239 also caused mitochondrial dysfunction in HeLa cells, and compound 239-induced cell apoptosis was related to generating reactive oxygen species in HeLa cells. Furthermore, compound 239 increased the phosphorylation levels of JNK, ERK, and p38 in HeLa cells, suggesting that compound 239 activated the MAPK signaling pathway, which helps mediate cell proliferation and apoptosis [70,102].
The hepatoprotective activities of compounds 245–248 were assayed in transgenic fluorescent zebrafish with liver injury. Quantitative analyses showed that 20 μM of compounds 245, 246, and 248 exhibited moderate hepatoprotective activities equal to those of the positive control (50 μM S-adenosyl-l-methionine). In addition, C-3 substituents were also a significant determinant of bioactivity as replacing the 2-aminoethanal group in 248 with a methyl formate group (as in 247) or with a 2-aminopropanoic acid group (as in C-mannosyl tryptophan) resulted in inactivity [71].
Although compounds 249 and 250 displayed moderate to strong antiviral activities, they were toxic against MDCK and Hep-2 cells with CC50 values ranging from 13 to 31 μM, drastically lowering their therapeutic ratios. Interestingly, the hemiketal form (250) of compound 249 displayed more potent antiviral activities while also being more toxic [72].
The antibacterial activities of compounds 314, 317, 321, and 323 were evaluated but were considerably less potent than their congener, griseofamine A’s activity (MIC = 8–16 µg/mL). Since all compounds tested contained the 3-acyltetramic acid moiety, the putative notion that the motif was a major determinant of antibacterial activity was not supported [87]. Compounds 347–352 displayed moderate to strong cytotoxicities against various cell lines. The results indicated that increasing the length from four to five (counting the heteroatoms) of the linker chain increased cytotoxicity (347 vs. 348 and 350 vs. 351). However, a further increase (from five to six) suppressed bioactivity (348 vs. 349 and 351 vs. 352). Replacing the methoxy group in the trisubstituted phenyl ring with a proton weakened the activities of derivatives (347, 348, and 349 vs. 350, 351, and 352) [88]. Moreover, compound 348 displayed the most robust cytotoxicity in the series while exhibiting minimal toxicity on normal hepatic LO2 cells (showing high selectivity). It also inhibited HDAC1 and HDAC6 while enhancing the acetylation levels of histone H3 and α-tubulin. In addition, it induced more significant cancer cell apoptosis than SAHA by regulating the expression of apoptotic proteins Bax, Bcl-2, and caspase 3. Compound 348 also induced important autophagic flux activity in Bel7402 cells by increasing the expression of Beclin-1 and LC3-II proteins and suppressing LC3-I and p62 production. Furthermore, compound 348 also significantly inhibited PI3K/Akt/mTOR signaling, an essential cell-growth-promoting pathway aberrantly activated in many cancers [88,103].
Compounds 404–424 and 438–443 were evaluated for their inhibitory activity against porcine SCR (mixture of bc1 and complex II). The first series (404–424) showed weak activities, except 411, only inhibiting 10–49% of SCR at 10 µM. The binding of the lead compound 411 of the first series was analyzed with the complex. The carboxyl oxygen in carbamate formed an H-bond with Glu271. However, the indole and phenyl rings were almost perpendicular with Phe274, resulting in weak interactions with the residue. Previous studies by the same group showed that the π–π interactions between neopeltolide and Phe274 were essential for activity [104]. Furthermore, the carboxyl oxygen in the ester did not form an H-bond with His161, also an essential factor for bioactivity [104]. The second series (438–443) was synthesized with an amide linker to improve the interactions between the derivatives and key residues. Similarly, it contained only fluorine- and methyl-substituted benzyls as substituents on the indole ring (first series derivatives with such substituents were generally more potent). These modifications considerably increased the potency of the second series derivatives. Again, the binding mode of the most potent derivative 442 was analyzed. It formed π–π interactions with Phe274 and an H-bond with Glu271 and His161. Interestingly, the two positional isomers of compound 442 (which differed in the positions of substituents on the benzyl group), 441 (o-methyl), and 443 (p-methyl) were considerably less potent, demonstrating that m-methyl was the most bioactive configuration. In contrast, for the fluorine substituents, o-fluorine was the most bioactive configuration (438 vs. 439 vs. 440) [90].
None of the celastrol-indole derivatives (456–465) inhibited concanavalin A-induced T-cell proliferation or displayed cytotoxicities. [92] Phidianidine A and its synthetic structure variants (479–488) were screened against the settlement and metamorphosis of Amphibalanus improvisus cyprids, with compounds 486 and 487 exhibiting the most potent activities. It demonstrated that only the guanidine derivatives showed significant activity, while primary amine (479–483) or quaternary ammonium 488 derivatives were relatively inactive. Additionally, increasing chain length increased bioactivity among the guanidine derivatives (484–487). Furthermore, since both compounds 486 and 487 were more potent than phidianidine A (EC50 = 4.0 µg/mL), the original scaffold’s transformation into a simpler, more bioactive structure that did not contain the constrained 1,2,4-oxadiazole ring was deemed successful [93].
Compounds 535–548 displayed potent cytotoxicity against the MV4-11 cell line. In addition, compound 540 was the most powerful derivative in the series and induced apoptosis of MV4-11 cells. It also arrested cells in the G0/G1 phase and reduced the expression of FLT-3, CDK2, and c-kit. The preliminary structure–activity relationship study yielded interesting results: if the substituent on C-3 was a proton (535–538), the most activity-boosting substituent on C-5ʹʹ was a methoxy group (538). Additionally, if C-3 was bonded to chlorine (539–543), then fluorine on C-5ʹʹ produced 540 (the most potent derivative overall). Lastly, for bromine on C-3 (544–548), a proton on C-5ʹʹ afforded the most potent bromine-containing derivative 544 [96]. However, compound 561 was tested for psychedelic-like activity using the mouse head-twitch response assay, which showed that it was more potent than dimethyltryptamine but less potent than psilocybin [100].
5. Conclusions
The 354 novel indole alkaloids discussed—consisting of 250 isolated and 87 synthesized—represent only a tiny fraction of the known indole-alkaloids, according to both the number and the structural diversity of predicted indole-containing compounds yet to be discovered. This massive gap between known and predicted indole alkaloids and the group’s potent, selective, and variegated bioactivities represents a worthwhile opportunity for chemists, who can leverage modern spectroscopic and biological tools to determine the alkaloids’ structures and mechanisms of action efficiently. This article described recent advances in the field to draw further attention to unique alkaloids that either possess unprecedented motifs or display substantial activities against their targets and to provide a convenient, easily accessible list of information regarding the isolations and bioactivities of alkaloids. Similarly, reappraisals were included to demonstrate the efficacy of modern-day spectroscopic techniques. At the same time, syntheses (both total and semi) were discussed to highlight the second aspect of drug development: optimizing the lead.
Author Contributions
Conceptualization, R.S.Z.S. and K.M.K.; formal analysis, S.M.U. and R.S.Z.S.; data curation, S.M.U. and M.S.; writing—original draft preparation, S.M.U.; writing—review and editing, R.S.Z.S. and K.M.K.; visualization, M.S.; supervision, R.S.Z.S. and K.M.K.; funding acquisition, K.M.K. All authors have read and agreed to the published version of the manuscript.
Funding
The authors would like to acknowledge the financial support of the Sindh Higher Education Commission (SHEC), Sindh, Pakistan, vide letter No. NO.DD/SHEC/1-14/2014, Project code SHEC/SRSP/Med-3/15/2021-2.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| WHO | World Health Organization |
| LSD | Lysergic acid diethylamide |
| DCs | Dendritic cells |
| HDAC | Histone deacetylase |
| MIAs | Monoindole alkaloids |
| CANPA | Computer-Assisted Natural Products Anticipation |
| ETP | Epipolythiodioxopiperazine |
| HMDA | Hydroxy-4-methyldecanoic acid |
| EDB | Edible |
| LCPA | Long-chain polyamine |
| DFT | Density-functional theory |
| THF | Tetrahydrofuran |
| TBAF | Tetra-n-butylammonium fluoride |
| RANKL | Receptor activator of nuclear factor kappa-Β ligand |
| DCM | Dichloromethane |
| TiPSOTf | Triisopropylsilyl trifluoromethanesulfonate |
| HOBt | Hydroxybenzotriazole |
| DIPEA | N,N-Diisopropylethylamine |
| HATU | Hexafluorophosphate azabenzotriazoletetramethyl uronium |
| DDQ | 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone |
| TrocCl | 2,2,2-Trichloroethoxycarbonyl chloride |
| TFAA | Trifluoroacetic anhydride |
| MAO | Monoamine oxidase |
| SCR | Succinate-cytochrome c reductase |
| LPS | Lipopolysaccharide |
| IL | Interleukin |
| PTP | Protein tyrosine phosphatase |
| HSV-2 | Herpes simplex virus 2 |
| hBM-MSC | Human bone marrow mesenchymal stem cells |
| TFA | Trifluoroacetic acid |
| Et3N | Triethylamine |
| Et2O | Diethyl ether |
| NBS | N-Bromosuccinimide |
| AcOH | Acetic acid |
| Boc2O | Di-tert-butyl dicarbonate |
| Ac2O | Acetic anhydride |
| DMAP | 4-Dimethylaminopyridine |
| TMSOTf | Trimethylsilyl trifluoromethanesulfonate |
| DMF | Dimethylformamide |
| n-BuLi | n-Butyl lithium |
| EtOAc | Ethyl acetate |
| i-BuOCOCl | Isobutyl chloroformate |
| DAST | Diethylamino sulfur trifluoride |
| DBU | 1,8-Diazabicyclo [5.4.0]undec-7-ene |
| DIBAL-H | Diisobutylaluminium hydride |
| EDCl | 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide |
| MsCl | Methanesulfonyl chloride |
| NCS | N-Chlorosuccinimide |
| TCDI | Thiocarbonyldiimidazole |
| m-CPBA | Meta-chloroperoxybenzoic acid |
| AChE | Acetylcholinesterase |
| BChE | Butyrylcholinesterase |
| 5-HT1A | 5-hydroxytryptamine 1A |
| ZIKV | Zika virus |
| TCPTP | T-cell protein tyrosine phosphatase |
| RSV | Respiratory syncytial virus |
| ATP | Adenosine triphosphate |
References
- Lahlou, M. The success of natural products in drug discovery. Pharmacol. Pharm. 2013, 4, 17–31. [Google Scholar] [CrossRef]
- Veeresham, C. Natural products derived from plants as a source of drugs. J. Adv. Pharm. Technol. Res. 2012, 3, 200. [Google Scholar] [CrossRef] [PubMed]
- John, J.E. Natural products-based drug discovery: Some bottlenecks and considerations. Curr. Sci. 2009, 96, 753–754. [Google Scholar]
- Eddershaw, P.J.; Beresford, A.P.; Bayliss, M.K. ADME/PK as part of a rational approach to drug discovery. Drug Discov. Today 2000, 5, 409–414. [Google Scholar] [CrossRef]
- Appendino, G.; Fontana, G.; Pollastro, F. Natural Products Drug Discovery. In Comprehensive Natural Products II; Liu, H.-W., Mander, L., Eds.; Elsevier: Oxford, UK, 2010; pp. 205–236. [Google Scholar]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; International Natural Product Sciences Taskforce; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Mohr, J.T.; Krout, M.R.; Stoltz, B.M. Natural products as inspiration for the development of asymmetric catalysis. Nature 2008, 455, 323–332. [Google Scholar] [CrossRef]
- Grigalunas, M.; Burhop, A.; Christoforow, A.; Waldmann, H. Pseudo-natural products and natural product-inspired methods in chemical biology and drug discovery. Curr. Opin. Chem. Biol. 2020, 56, 111–118. [Google Scholar]
- Li, J.W.; Vederas, J.C. Drug discovery and natural products: End of an era or an endless frontier? Science 2009, 325, 161–165. [Google Scholar] [CrossRef]
- Hunter, P. Harnessing nature’s wisdom. Turning to nature for inspiration and avoiding her follies. EMBO Rep. 2008, 9, 838–840. [Google Scholar] [CrossRef]
- Hanson, J.R. Natural Products: The Secondary Metabolites; Royal Society of Chemistry: Cambridge, UK, 2003; Volume 17. [Google Scholar]
- Harden, R.N. Chronic neuropathic pain: Mechanisms, diagnosis, and treatment. Neurologist 2005, 11, 111–122. [Google Scholar]
- Esu, E.B.; Effa, E.E.; Opie, O.N.; Meremikwu, M.M. Artemether for severe malaria. Cochrane Database Syst. Rev. 2019, 6, CD010678. [Google Scholar] [PubMed]
- Kaur, R.; Arora, S. Alkaloids-important therapeutic secondary metabolites of plant origin. J. Crit. Rev. 2015, 2, 1–8. [Google Scholar]
- Seigler, D.S. Plant Secondary Metabolism; Springer: New York, NY, USA, 2001; p. 628. [Google Scholar]
- Kiesecker, C.; Zitron, E.; Luck, S.; Bloehs, R.; Scholz, E.P.; Kathofer, S.; Thomas, D.; Kreye, V.A.; Katus, H.A.; Schoels, W.; et al. Class Ia anti-arrhythmic drug ajmaline blocks HERG potassium channels: Mode of action. Naunyn Schmiedebergs Arch. Pharmacol. 2004, 370, 423–435. [Google Scholar] [CrossRef]
- Moore, P.W.; Rasimas, J.J.; Donovan, J.W. Physostigmine is the antidote for anticholinergic syndrome. J. Med. Toxicol. 2015, 11, 159–160. [Google Scholar] [CrossRef]
- Avendaño, C.; Menéndez, J.C. (Eds.) Chapter 8—Anticancer drugs targeting tubulin and microtubules. In Medicinal Chemistry of Anticancer Drugs; Elsevier: Amsterdam, The Netherlands, 2008; pp. 229–249. [Google Scholar]
- Koenig, X.; Hilber, K. The anti-addiction drug ibogaine and the heart: A delicate relation. Molecules 2015, 20, 2208–2228. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.L.; Hai, P.; Zhang, S.B.; Xiao, J.F.; Gao, Y.; Ma, B.J.; Fu, H.Y.; Chen, Y.M.; Yang, X.L. Prenylated indole diterpene alkaloids from a mine-soil-derived Tolypocladium sp. J. Nat. Prod. 2019, 82, 221–231. [Google Scholar] [CrossRef]
- Tabudravu, J.N.; Pellissier, L.; Smith, A.J.; Subko, K.; Autreau, C.; Feussner, K.; Hardy, D.; Butler, D.; Kidd, R.; Milton, E.J.; et al. LC-HRMS-database screening metrics for rapid prioritization of samples to accelerate the discovery of structurally new natural products. J. Nat. Prod. 2019, 82, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Sim, D.S.; Navanesan, S.; Sim, K.S.; Gurusamy, S.; Lim, S.H.; Low, Y.Y.; Kam, T.S. Conolodinines A-D, Aspidosperma-Aspidosperma bisindole alkaloids with antiproliferative activity from Tabernaemontana corymbosa. J. Nat. Prod. 2019, 82, 850–858. [Google Scholar] [CrossRef]
- Yu, X.; Müller, W.E.; Guo, Z.; Lin, W.; Zou, K.; Liu, Z.; Proksch, P. Indole alkaloids from the coprophilous fungus Aphanoascus fulvescens. Fitoterapia 2019, 136, 104168. [Google Scholar] [CrossRef]
- Ariantari, N.P.; Ancheeva, E.; Wang, C.; Mandi, A.; Knedel, T.O.; Kurtan, T.; Chaidir, C.; Muller, W.E.G.; Kassack, M.U.; Janiak, C.; et al. Indole diterpenoids from an endophytic Penicillium sp. J. Nat. Prod. 2019, 82, 1412–1423. [Google Scholar] [CrossRef]
- Wang, X.; Abbas, M.; Zhang, Y.; Elshahawi, S.I.; Ponomareva, L.V.; Cui, Z.; Van Lanen, S.G.; Sajid, I.; Voss, S.R.; Shaaban, K.A.; et al. Divergent fused phenazine-based metabolites from a Himalayan Streptomyces. J. Nat. Prod. 2019, 82, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- Mudalungu, C.M.; von Torne, W.J.; Voigt, K.; Ruckert, C.; Schmitz, S.; Sekurova, O.N.; Zotchev, S.B.; Sussmuth, R.D. Noursamycins, chlorinated cyclohexapeptides identified from molecular networking of Streptomyces noursei NTR-SR4. J. Nat. Prod. 2019, 82, 1478–1486. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.K.; Wong, S.P.; Sim, K.S.; Lim, S.H.; Low, Y.Y.; Kam, T.S. A cytotoxic indole characterized by incorporation of a unique carbon-nitrogen skeleton and two pentacyclic corynanthean alkaloids incorporating a substituted tetrahydrofuranone ring from Kopsia arborea. J. Nat. Prod. 2019, 82, 1902–1907. [Google Scholar] [CrossRef]
- Yan, W.; Zhao, S.S.; Ye, Y.H.; Zhang, Y.Y.; Zhang, Y.; Xu, J.Y.; Yin, S.M.; Tan, R.X. Generation of indoles with agrochemical significance through biotransformation by Chaetomium globosum. J. Nat. Prod. 2019, 82, 2132–2137. [Google Scholar] [CrossRef]
- Ramos, A.E.F.; Pavesi, C.; Litaudon, M.; Dumontet, V.; Poupon, E.; Champy, P.; Genta-Jouve, G.; Beniddir, M.A. CANPA: Computer-assisted natural products anticipation. Anal. Chem. 2019, 91, 11247–11252. [Google Scholar] [CrossRef]
- Kim, M.C.; Cullum, R.; Machado, H.; Smith, A.J.; Yang, I.; Rodvold, J.J.; Fenical, W. Photopiperazines A-D, photosensitive interconverting diketopiperazines with significant and selective activity against U87 glioblastoma cells, from a rare, marine-derived Actinomycete of the family Streptomycetaceae. J. Nat. Prod. 2019, 82, 2262–2267. [Google Scholar] [CrossRef]
- Li, H.; Xu, D.; Sun, W.; Yang, B.; Li, F.; Liu, M.; Wang, J.; Xue, Y.; Hu, Z.; Zhang, Y. HPLC-DAD-directed isolation of linearly fused prenylated indole alkaloids from a soil-derived Aspergillus versicolor. J. Nat. Prod. 2019, 82, 2181–2188. [Google Scholar] [CrossRef]
- Knestrick, M.A.; Wilson, N.G.; Roth, A.; Adams, J.H.; Baker, B.J. Friomaramide, a highly modified linear hexapeptide from an Antarctic sponge, inhibits Plasmodium falciparum liver-stage development. J. Nat. Prod. 2019, 82, 2354–2358. [Google Scholar] [CrossRef]
- Bankeu, J.J.K.; Kagho, D.U.K.; Fongang, Y.S.F.; Toghueo, R.M.K.; Mba’ning, B.M.; Feuya, G.R.T.; Fekam, F.B.; Tchouankeu, J.C.; Ngouela, S.A.; Sewald, N.; et al. Constituents from Nauclea latifolia with anti-haemophilus influenzae type b inhibitory activities. J. Nat. Prod. 2019, 82, 2580–2585. [Google Scholar] [CrossRef]
- Zhou, L.M.; Kong, F.D.; Fan, P.; Ma, Q.Y.; Xie, Q.Y.; Li, J.H.; Zheng, H.Z.; Zheng, Z.H.; Yuan, J.Z.; Dai, H.F.; et al. Indole-diterpenoids with protein tyrosine phosphatase inhibitory activities from the marine-derived fungus Penicillium sp. KFD28. J. Nat. Prod. 2019, 82, 2638–2644. [Google Scholar] [CrossRef]
- Yeap, J.S.; Saad, H.M.; Tan, C.H.; Sim, K.S.; Lim, S.H.; Low, Y.Y.; Kam, T.S. Macroline-sarpagine bisindole alkaloids with antiproliferative activity from Alstonia penangiana. J. Nat. Prod. 2019, 82, 3121–3132. [Google Scholar] [CrossRef]
- Salim, F.; Yunus, Y.M.; Anouar, E.H.; Awang, K.; Langat, M.; Cordell, G.A.; Ahmad, R. Absolute configuration of alkaloids from Uncaria longiflora through experimental and computational approaches. J. Nat. Prod. 2019, 82, 2933–2940. [Google Scholar] [CrossRef]
- Liang, J.H.; Luan, Z.L.; Tian, X.G.; Zhao, W.Y.; Wang, Y.L.; Sun, C.P.; Huo, X.K.; Deng, S.; Zhang, B.J.; Zhang, Z.J.; et al. Uncarialins A-I, monoterpenoid indole alkaloids from Uncaria rhynchophylla as natural agonists of the 5-HT1A receptor. J. Nat. Prod. 2019, 82, 3302–3310. [Google Scholar] [CrossRef]
- Guo, Q.; Si, X.; Shi, Y.; Yang, H.; Liu, X.; Liang, H.; Tu, P.; Zhang, Q. Glucoconjugated monoterpene indole alkaloids from Uncaria rhynchophylla. J. Nat. Prod. 2019, 82, 3288–3301. [Google Scholar] [CrossRef]
- Kong, F.D.; Zhang, S.L.; Zhou, S.Q.; Ma, Q.Y.; Xie, Q.Y.; Chen, J.P.; Li, J.H.; Zhou, L.M.; Yuan, J.Z.; Hu, Z.; et al. Quinazoline-containing indole alkaloids from the marine-derived fungus Aspergillus sp. HNMF114. J. Nat. Prod. 2019, 82, 3456–3463. [Google Scholar] [CrossRef]
- Rodriguez, J.P.G.; Bernardi, D.I.; Gubiani, J.R.; Magalhaes de Oliveira, J.; Morais-Urano, R.P.; Bertonha, A.F.; Bandeira, K.F.; Bulla, J.I.Q.; Sette, L.D.; Ferreira, A.G.; et al. Water-soluble glutamic acid derivatives produced in culture by Penicillium solitum IS1-A from King George Island, Maritime Antarctica. J. Nat. Prod. 2020, 83, 55–65. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, K.; Wang, W.; Zhang, G.; Zhu, T.; Che, Q.; Gu, Q.; Li, D. Amphiepicoccins A-J: Epipolythiodioxopiperazines from the fish-gill-derived Fungus Epicoccum nigrum HDN17-88. J. Nat. Prod. 2020, 83, 524–531. [Google Scholar] [CrossRef]
- Alcover, C.F.; Bernadat, G.; Kabran, F.A.; Le Pogam, P.; Leblanc, K.; Ramos, A.E.F.; Gallard, J.F.; Mouray, E.; Grellier, P.; Poupon, E.; et al. Molecular networking reveals serpentinine-related bisindole alkaloids from Picralima nitida, a previously well-investigated species. J. Nat. Prod. 2020, 83, 1207–1216. [Google Scholar] [CrossRef]
- Wang, M.H.; Zhang, X.Y.; Tan, X.M.; Niu, S.B.; Sun, B.D.; Yu, M.; Ding, G.; Zou, Z.M. Chetocochliodins A-I, epipoly(thiodioxopiperazines) from Chaetomium cochliodes. J. Nat. Prod. 2020, 83, 805–813. [Google Scholar] [CrossRef]
- Li, C.J.; Chen, P.N.; Li, H.J.; Mahmud, T.; Wu, D.L.; Xu, J.; Lan, W.J. Potential antidiabetic fumiquinazoline alkaloids from the marine-derived fungus Scedosporium apiospermum F41-1. J. Nat. Prod. 2020, 83, 1082–1091. [Google Scholar] [CrossRef]
- Klein-Junior, L.C.; Cretton, S.; Heyden, Y.V.; Gasper, A.L.; Nejad-Ebrahimi, S.; Christen, P.; Henriques, A.T. Bioactive azepine-indole alkaloids from Psychotria nemorosa. J. Nat. Prod. 2020, 83, 852–863. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hu, Y.; Hao, X.; Tan, J.; Li, F.; Qiao, X.; Chen, S.; Xiao, C.; Chen, M.; Peng, Z.; et al. Raistrickindole A, an anti-HCV oxazinoindole alkaloid from Penicillium raistrickii IMB17-034. J. Nat. Prod. 2019, 82, 1391–1395. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.J.; Huo, G.M.; Zhang, Q.; Li, D.; Wang, D.C.; Qi, J.Z.; Han, W.B.; Gao, J.M. Phaeosphaones: Tyrosinase inhibitory thiodiketopiperazines from an endophytic Phaeosphaeria fuckelii. J. Nat. Prod. 2020, 83, 1592–1597. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ding, X.; Yuan, Y.X.; Guo, L.L.; Hao, X.J. Cytotoxic monoterpenoid indole alkaloids from Tabernaemontana corymbosa as potent autophagy inhibitors by the attenuation of lysosomal acidification. J. Nat. Prod. 2020, 83, 1432–1439. [Google Scholar] [CrossRef]
- Flores-Bocanegra, L.; Raja, H.A.; Graf, T.N.; Augustinovic, M.; Wallace, E.D.; Hematian, S.; Kellogg, J.J.; Todd, D.A.; Cech, N.B.; Oberlies, N.H. The chemistry of Kratom [Mitragyna speciosa]: Updated characterization data and methods to elucidate indole and oxindole alkaloids. J. Nat. Prod. 2020, 83, 2165–2177. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Z.W.; Li, Y.; Wei, C.J.; Xie, J.; Yuan, M.F.; Zhang, D.M.; Ye, W.C.; Zhang, X.Q. Structurally diverse indole alkaloids with vasorelaxant activity from Melodinus hemsleyanus. J. Nat. Prod. 2020, 83, 2313–2319. [Google Scholar] [CrossRef]
- Qin, L.; Yi, W.; Lian, X.Y.; Zhang, Z. Bioactive alkaloids from the Actinomycete Actinoalloteichus sp. ZZ1866. J. Nat. Prod. 2020, 83, 2686–2695. [Google Scholar] [CrossRef]
- Perez-Bonilla, M.; Oves-Costales, D.; Gonzalez, I.; de la Cruz, M.; Martin, J.; Vicente, F.; Genilloud, O.; Reyes, F. Krisynomycins, imipenem potentiators against methicillin-resistant Staphylococcus aureus, Produced by Streptomyces canus. J. Nat. Prod. 2020, 83, 2597–2606. [Google Scholar] [CrossRef]
- Di, X.; Wang, S.; Oskarsson, J.T.; Rouger, C.; Tasdemir, D.; Hardardottir, I.; Freysdottir, J.; Wang, X.; Molinski, T.F.; Omarsdottir, S. Bromotryptamine and imidazole alkaloids with anti-inflammatory activity from the Bryozoan Flustra foliacea. J. Nat. Prod. 2020, 83, 2854–2866. [Google Scholar] [CrossRef]
- Kleks, G.; Holland, D.C.; Kennedy, E.K.; Avery, V.M.; Carroll, A.R. Antiplasmodial alkaloids from the Australian Bryozoan Amathia lamourouxi. J. Nat. Prod. 2020, 83, 3435–3444. [Google Scholar] [CrossRef]
- Yi, W.F.; Ding, X.; Chen, Y.Z.; Adelakun, T.A.; Zhang, Y.; Hao, X.J. Tabernaesines A-I, cytotoxic Aspidosperma-Aspidosperma-type bisindole alkaloids from Tabernaemontana pachysiphon. J. Nat. Prod. 2020, 83, 3215–3222. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.W.; Liu, X.J.; Yuan, J.; Li, H.J.; Mahmud, T.; Hong, M.J.; Yu, J.C.; Lan, W.J. L-Tryptophan induces a marine-derived Fusarium sp. to produce indole alkaloids with activity against the Zika virus. J. Nat. Prod. 2020, 83, 3372–3380. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.F.; Hu, K.; Wang, F.; Tang, J.W.; Zhang, L.; Sun, H.D.; Cai, X.H.; Puno, P.T. 3-Hydroxy-4-methyldecanoic acid-containing cyclotetradepsipeptides from an endolichenic Beauveria sp. J. Nat. Prod. 2021, 84, 1244–1253. [Google Scholar] [CrossRef]
- Jin, P.; Zhan, G.; Zheng, G.; Liu, J.; Peng, X.; Huang, L.; Gao, B.; Yuan, X.; Yao, G. Gelstriamine A a triamino monoterpene indole alkaloid with a caged 6/5/7/6/6/5 scaffold and analgesic alkaloids from Gelsemium elegans stems. J. Nat. Prod. 2021, 84, 1326–1334. [Google Scholar] [CrossRef]
- Otogo N’Nang, E.; Le Pogam, P.; Ndong Mba, T.; Sima Obiang, C.; Mouray, E.; Grellier, P.; Kumulungui, B.; Champy, P.; Beniddir, M.A. Targeted isolation of hemitheion from Mostuea brunonis, a proposed biosynthetic intermediate of theionbrunonines. J. Nat. Prod. 2021, 84, 1409–1413. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Yeap, J.S.; Lim, S.H.; Low, Y.Y.; Sim, K.S.; Kam, T.S. The bisindole alkaloids Angustilongines M and A from Alstonia penangiana induce mitochondrial apoptosis and G0/G1 cell cycle arrest in HT-29 cells through promotion of tubulin polymerization. J. Nat. Prod. 2021, 84, 1524–1533. [Google Scholar] [CrossRef]
- Kwon, O.; Ahn, S.; Jeon, J.; Park, I.G.; Won, T.H.; Sim, C.J.; Park, H.; Oh, D.; Oh, K.; Noh, M. Psammocindoles A–C: Isolation, synthesis, and bioactivity of indole-γ-lactams from the sponge Psammocinia vermis. Org. Lett. 2021, 23, 4667–4671. [Google Scholar] [CrossRef]
- Wang, F.; Bi, J.; He, L.; Chen, J.; Zhang, Q.; Hou, X.; Xu, H. The indole alkaloids from the roots of Isatidis Radix. Fitoterapia 2021, 153, 104950. [Google Scholar] [CrossRef]
- Duan, F.F.; Liu, L.; Gao, Y.; Peng, X.G.; Meng, X.G.; Ruan, H.L. [11]-Chaetoglobosins from Pseudeurotium bakeri induce G2/M cell cycle arrest and apoptosis in human cancer cells. J. Nat. Prod. 2021, 84, 1904–1914. [Google Scholar] [CrossRef]
- Jiang, C.X.; Yu, B.; Miao, Y.M.; Ren, H.; Xu, Q.; Zhao, C.; Tian, L.L.; Yu, Z.Q.; Zhou, P.P.; Wang, X.; et al. Indole alkaloids from a soil-derived Clonostachys rosea. J. Nat. Prod. 2021, 84, 2468–2474. [Google Scholar] [CrossRef]
- Fouotsa, H.; Le Pogam, P.; Mkounga, P.; Lannang, A.M.; Bernadat, G.; Vanheuverzwijn, J.; Zhou, Z.; Leblanc, K.; Rharrabti, S.; Nkengfack, A.E.; et al. Voatriafricanines A and B, trimeric Vobasine-Aspidosperma-Aspidosperma alkaloids from Voacanga africana. J. Nat. Prod. 2021, 84, 2755–2761. [Google Scholar] [CrossRef] [PubMed]
- Maeyama, Y.; Nakashima, Y.; Kato, H.; Hitora, Y.; Maki, K.; Inada, N.; Murakami, S.; Inazumi, T.; Ise, Y.; Sugimoto, Y.; et al. Amakusamine from a Psammocinia sp. Sponge: Isolation, synthesis, and SAR study on the inhibition of RANKL-induced formation of multinuclear osteoclasts. J. Nat. Prod. 2021, 84, 2738–2743. [Google Scholar] [CrossRef] [PubMed]
- Holland, D.C.; Prebble, D.W.; Er, S.; Hayton, J.B.; Robertson, L.P.; Avery, V.M.; Domanskyi, A.; Kiefel, M.J.; Hooper, J.N.A.; Carroll, A.R. α-Synuclein aggregation inhibitory prunolides and a dibrominated β-carboline sulfamate from the ascidian Synoicum prunum. J. Nat. Prod. 2022, 85, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Vergis, J.; Mahmud, T. EDB gene cluster-dependent indole production is responsible for the ability of Pseudomonas fluorescens NZI7 to repel grazing by Caenorhabditis elegans. J. Nat. Prod. 2022, 85, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Shi, B.B.; Ai, H.L.; Duan, K.T.; Feng, T.; Liu, J.K. Ophiorrhines F and G, key biogenetic intermediates of ophiorrhine alkaloids from Ophiorrhiza japonica and their immunosuppressant activities. J. Nat. Prod. 2022, 85, 453–457. [Google Scholar] [CrossRef]
- Guo, X.; Meng, Q.; Liu, J.; Wu, J.; Jia, H.; Liu, D.; Gu, Y.; Liu, J.; Huang, J.; Fan, A.; et al. Sclerotiamides C-H, notoamides from a marine gorgonian-derived fungus with cytotoxic activities. J. Nat. Prod. 2022, 85, 1067–1078. [Google Scholar] [CrossRef]
- Zhang, D.; Li, Y.; Li, X.; Han, X.; Wang, Z.; Zhang, W.; Dou, B.; Lu, Z.; Li, P.; Li, G. Neopetrosins A-D and haliclorensin D, indole-C-mannopyranosides and a diamine alkaloid isolated from the south china sea marine sponge Neopetrosia chaliniformis. J. Nat. Prod. 2022, 85, 1626–1633. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Li, L.; Li, Y.Q.; Luo, J.H.; Li, W.; Li, L.F.; Zheng, C.J.; Cao, F. Oxalierpenes A and B, unusual indole-diterpenoid derivatives with antiviral activity from a marine-derived strain of the fungus Penicillium oxalicum. J. Nat. Prod. 2022, 85, 1880–1885. [Google Scholar] [CrossRef]
- Shan, W.G.; Wang, S.L.; Lang, H.Y.; Chen, S.M.; Ying, Y.M.; Zhan, Z.J. Cottoquinazolines E and F from Neosartorya fischeri NRRL 181. Helv. Chim. Acta 2015, 98, 552–556. [Google Scholar] [CrossRef]
- Lin, S.; Yu, H.; Yang, B.; Li, F.; Chen, X.; Li, H.; Zhang, S.; Wang, J.; Hu, Y.; Hu, Z.; et al. Reisolation and configurational reinvestigation of cottoquinazolines E-G from an arthropod-derived strain of the fungus Neosartorya fischeri. J. Nat. Prod. 2020, 83, 169–173. [Google Scholar] [CrossRef]
- Ovenden, S.P.; Capon, R.J. Echinosulfonic acids A-C and echinosulfone A: Novel bromoindole sulfonic acids and a sulfone from a southern australian marine sponge, echinodictyum. J. Nat. Prod. 1999, 62, 1246–1249. [Google Scholar] [CrossRef] [PubMed]
- Rubnov, S.; Chevallier, C.; Thoison, O.; Debitus, C.; Laprevote, O.; Guénard, D.; Sévenet, T. Echinosulfonic acid D: An ESI MS n evaluation of a new cytotoxic alkaloid from the new-Caledonian sponge Psammoclemma sp. Nat. Prod. Res. 2005, 19, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Sala, S.; Nealon, G.L.; Sobolev, A.N.; Fromont, J.; Gomez, O.; Flematti, G.R. Structure reassignment of echinosulfone A and the echinosulfonic acids A-D supported by single-crystal X-ray diffraction and density functional theory analysis. J. Nat. Prod. 2020, 83, 105–110. [Google Scholar] [CrossRef]
- Matsunaga, S.; Jimbo, M.; Gill, M.B.; Lash-Van Wyhe, L.L.; Murata, M.; Nonomura, K.I.; Swanson, G.T.; Sakai, R. Isolation, amino acid sequence and biological activities of novel long-chain polyamine-associated peptide toxins from the sponge Axinyssa aculeata. ChemBioChem 2011, 12, 2191–2200. [Google Scholar] [CrossRef]
- Irie, R.; Miyako, K.; Matsunaga, S.; Sakai, R.; Oikawa, M. Structure revision of protoaculeine B, a post-translationally modified N-terminal residue in the peptide toxin aculeine B. J. Nat. Prod. 2021, 84, 1203–1209. [Google Scholar] [CrossRef] [PubMed]
- Levesque, J.; Pousset, J.; Cavé, A. Lyaline and lyadine-new alkaloids from Pauridiantha-lyalli brem(rubiaceae). Comptes Rendus Hebd. Seances L Acad. Des Sci. Ser. C 1974, 278, 959–961. [Google Scholar]
- Bennasar, M.L.; Roca, T.; Monerris, M. Total synthesis of the proposed structures of indole alkaloids lyaline and lyadine. J. Org. Chem. 2004, 69, 752–756. [Google Scholar] [CrossRef] [PubMed]
- Jagora, A.; Gallard, J.F.; Beniddir, M.A.; Le Pogam, P. A reappraisal of the structure of lyaline as the first naturally occurring nacycline monoterpene indole alkaloid. J. Nat. Prod. 2021, 84, 2617–2622. [Google Scholar] [CrossRef]
- Kam, T.S.; Pang, H.S.; Choo, Y.M.; Komiyama, K. Biologically active ibogan and vallesamine derivatives from Tabernaemontana divaricata. Chem. Biodivers. 2004, 1, 646–656. [Google Scholar] [CrossRef]
- Tarselli, M.A.; Raehal, K.M.; Brasher, A.K.; Streicher, J.M.; Groer, C.E.; Cameron, M.D.; Bohn, L.M.; Micalizio, G.C. Synthesis of conolidine, a potent non-opioid analgesic for tonic and persistent pain. Nat. Chem. 2011, 3, 449–453. [Google Scholar] [CrossRef]
- Chen, G.; Wang, C.; Zou, L.; Zhu, J.; Li, Y.; Qi, C. Six-step total synthesis of(+/-)-conolidine. J. Nat. Prod. 2019, 82, 2972–2978. [Google Scholar] [CrossRef] [PubMed]
- Zang, Y.; Genta-Jouve, G.; Zheng, Y.; Zhang, Q.; Chen, C.; Zhou, Q.; Wang, J.; Zhu, H.; Zhang, Y. Griseofamines A and B: Two indole-tetramic acid alkaloids with 6/5/6/5 and 6/5/7/5 ring systems from Penicillium griseofulvum. Org. Lett. 2018, 20, 2046–2050. [Google Scholar] [CrossRef] [PubMed]
- Sheng, T.; Ma, C.; Zhang, G.; Pan, X.; Liu, Z. Asymmetric total synthesis of griseofamine B and its three stereoisomers. J. Nat. Prod. 2022, 85, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.; Li, Y.; Zhu, R.; Qian, J.; Liu, J.; Gao, W.; Meng, C.; Miao, J.; Xiong, B.; Qiu, X.; et al. Hydroxamic acid derivatives of β-carboline/hydroxycinnamic acid hybrids inducing apoptosis and autophagy through the PI3K/Akt/mTOR pathways. J. Nat. Prod. 2019, 82, 1442–1450. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.E.; Botelho, J.C.; Guzman, E.; Harmody, D.; Linley, P.; McCarthy, P.J.; Pitts, T.P.; Pomponi, S.A.; Reed, J.K. Neopeltolide, a macrolide from a lithistid sponge of the family Neopeltidae. J. Nat. Prod. 2007, 70, 412–416. [Google Scholar] [CrossRef]
- Xiong, M.Q.; Chen, T.; Wang, Y.X.; Zhu, X.L.; Yang, G.F. Design and synthesis of potent inhibitors of bc1 complex based on natural product neopeltolide. Bioorg. Med. Chem. Lett. 2020, 30, 127324. [Google Scholar] [CrossRef]
- Venkatesha, S.H.; Moudgil, K.D. Celastrol suppresses experimental autoimmune encephalomyelitis via MAPK/SGK1-regulated mediators of autoimmune pathology. Inflamm. Res. 2019, 68, 285–296. [Google Scholar] [CrossRef]
- He, Q.W.; Feng, J.H.; Hu, X.L.; Long, H.; Huang, X.F.; Jiang, Z.Z.; Zhang, X.Q.; Ye, W.C.; Wang, H. Synthesis and biological evaluation of celastrol derivatives as potential immunosuppressive agents. J. Nat. Prod. 2020, 83, 2578–2586. [Google Scholar] [CrossRef]
- Labriere, C.; Elumalai, V.; Staffansson, J.; Cervin, G.; Le Norcy, T.; Denardou, H.; Rehel, K.; Moodie, L.W.K.; Hellio, C.; Pavia, H.; et al. Phidianidine A and synthetic analogues as naturally inspired marine antifoulants. J. Nat. Prod. 2020, 83, 3413–3423. [Google Scholar] [CrossRef]
- Wang, L.; Mei, X.; Wang, C.; Zhu, W. Biomimetic semi-synthesis of fradcarbazole A and its analogues. Tetrahedron 2015, 71, 7990–7997. [Google Scholar] [CrossRef]
- Fu, P.; Zhuang, Y.; Wang, Y.; Liu, P.; Qi, X.; Gu, K.; Zhang, D.; Zhu, W. New indolocarbazoles from a mutant strain of the marine-derived actinomycete Streptomyces fradiae 007M135. Org. Lett. 2012, 14, 6194–6197. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Xu, Y.; Zuo, M.; Liu, W.; Wang, L.; Zhu, W. Semisynthetic derivatives of fradcarbazole A and their cytotoxicity against acute myeloid leukemia cell lines. J. Nat. Prod. 2019, 82, 2279–2290. [Google Scholar] [CrossRef] [PubMed]
- Feng, T.; Li, Y.; Wang, Y.Y.; Cai, X.H.; Liu, Y.P.; Luo, X.D. Cytotoxic indole alkaloids from Melodinus tenuicaudatus. J. Nat. Prod. 2010, 73, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Walia, M.; Teijaro, C.N.; Gardner, A.; Tran, T.; Kang, J.; Zhao, S.; O’Connor, S.E.; Courdavault, V.; Andrade, R.B. Synthesis of(-)-melodinine K: A case study of efficiency in natural product synthesis. J. Nat. Prod. 2020, 83, 2425–2433. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Andrade, R.B. Domino Michael/Mannich/N-alkylation route to the tetrahydrocarbazole framework of aspidosperma alkaloids: Concise total syntheses of (-)-aspidospermidine, (-)-tabersonine, and (-)-vincadifformine. J. Am. Chem. Soc. 2013, 135, 13334–13337. [Google Scholar] [CrossRef]
- Fricke, J.; Sherwood, A.M.; Halberstadt, A.L.; Kargbo, R.B.; Hoffmeister, D. Chemoenzymatic synthesis of 5-methylpsilocybin: A tryptamine with potential psychedelic activity. J. Nat. Prod. 2021, 84, 1403–1408. [Google Scholar] [CrossRef]
- Walters, W.P.; Murcko, M.A. Prediction of ‘drug-likeness’. Adv. Drug Deliv. Rev. 2002, 54, 255–271. [Google Scholar] [CrossRef]
- Yue, J.; Lopez, J.M. Understanding MAPK signaling pathways in apoptosis. Int. J. Mol. Sci. 2020, 21, 2346. [Google Scholar] [CrossRef]
- Wang, F.; Ma, H.; Liu, Z.; Huang, W.; Xu, X.; Zhang, X. α-Mangostin inhibits DMBA/TPA-induced skin cancer through inhibiting inflammation and promoting autophagy and apoptosis by regulating PI3K/Akt/mTOR signaling pathway in mice. Biomed. Pharmacother. 2017, 92, 672–680. [Google Scholar] [CrossRef]
- Zhu, X.-L.; Zhang, R.; Wu, Q.-Y.; Song, Y.-J.; Wang, Y.-X.; Yang, J.-F.; Yang, G.-F. Natural product neopeltolide as a cytochrome bc 1 complex inhibitor: Mechanism of action and structural modification. J. Agric. Food Chem. 2019, 67, 2774–2781. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).