Evidence of the Beneficial Effects of Ursolic Acid against Lung Cancer
Abstract
:1. Introduction
2. Effects of Ursolic Acid against Lung Cancer
2.1. Effects of Ursolic Acid against Lung Cancer: In Vitro Studies
| Cell Type | Dose/Duration | Findings | Mechanism | Reference |
|---|---|---|---|---|
| A549 | UA 2–40 µM 0–72 h | ↓ Proliferation ↑ Apoptosis G1 phase cell cycle arrest | ↑ p53 protein ↓ Cyclin D1, D2 and E ↑ Fas/APO-1 receptor ↑ FasL ↑ Bax protein ↓ NF-kB/p65 activity ↓ Bcl-2 protein ↓ Bcl-Xl protein | [65] |
| H460 | UA 3, 10 and 30 µM 24 h | ↑ Apoptosis ↓ Proliferation ↓ Migration | ↑ Cleaved caspase-3 ↑ MMP 1, 2, 3, 9 and 10 gene expression ↑ Cytosolic glucocorticoid receptor | [66] |
| A549 H3255 Calu-6 | UA 2, 4, 8 and 16 µM | ↑ Apoptosis ↓ Cell viability ↓ Cell migration | ↓ NA+-K+-ATPase activity ↓ PKC activity ↓ VEGF protein ↓ ICAM-1 mRNA ↓ Fibronectin mRNA ↓ MMP 2 and 9 mRNA | [67] |
| A549 | UA 5–20 µM 24 h | ↓ Proliferation ↓ Cell adhesion ↓ Wound healing ↓ Cell migration | ↑ E-cadherin ↓ N-cadherin ↓ vimentin ↓ AEG-1 ↓ NF-kB | [69] |
| A549/H460 | UA 30 µM 12, 24 and 48 h | ↓ Cell viability ↓ Proliferation ↑ Apoptosis ↑ Chromatin condensation | ↑ Cleaved caspase 3/9 ↓ Bcl-2 ↑ Bax ↑ p-AMPK ↓ p-mTOR ↓ ACC activity ↓ FASN l activity | [70] |
| A549 | UA 25, 50, 100, 250 and 500 µM | ↓ Cell viability | ↓ VRK1 autophosphorylation ↓ VRK1 activity ↓ p-CREB ↓ p-His-H3 ↓ Cyclin D1 mRNA | [71] |
| PC9, H1299, A549, H1650, H358 and H1975 | UA 5, 10, 20, 30, 40, 50 and 80 µM 24, 48 and 72 h | ↓ Cell growth ↑ Apoptosis | ↑ pSAPK/JNK ↓ SP1 protein ↓ DNMT1 protein ↓ EZH2 protein | [72] |
| H28, H2452 and MSTO-211H | UA 0–80µM 24–72 h | ↑ Cytotoxicity ↓ Proliferation ↑ Sub-G1 population ↓ EMT | ↑ Cleaved caspase-3 ↑ Cleaved PARP ↑ E-cadherin ↓ N-cadherin ↓ β-catenin ↓ p-GSK2α/β ↓ cyclin D1 ↓ p-AKT ↓ NF-kB | [73] |
| A549 | UA 10–100 µM 24 h | ↓ Proliferation ↑ Apoptosis ↑ S-phase cell cycle arrest ↑ Autophagy | ↓ Bcl-2 protein ↑ Cleaved PARP ↑ LC3-II/LC3-I ratio ↑ p62 | [75] |
| A549 | UA 11, 22, 44 and 88 µM 24 and 48 h | ↓ Cell viability ↑ Autophagy ↑ Mitophagy | ↑ LC3-II/LC3-I ratio ↑ p62 protein ↑ PINK1 protein ↓ p-AKT ↓ p-mTOR ↑ Nrf2 protein ↑ ROS | [76] |
| A549 | UA 5, 10 and 20 µM 24, 48 and 72 h | ↓ Stemness ↓ Chemoresistance | ↓ CD133 ↓ Oct-4 ↓ Notch3 ↓ Nanog ↓ Sox2 | [78] |
| H1975 NSCLC with EGFR T790M mutation | UA 1, 5, 25, 50 and 100 µM | ↓ Cell growth ↑ Apoptosis ↓ Cell motility | ↓ CT45A2 mRNA ↓ TCF4 ↓ p-β-catenin @ Ser33/37/Thr41 ↑ p-GSK-3b @ Ser9 | [77] |
| H1975 | UA 0.001–0.1 µM | ↓ EMT | ↓ N-cadherin ↑ E-cadherin ↓ MMP-2 and -9 ↓ TGF-β1 | [79] |
| NCI-H292 | UA 3, 6, 9, 12 and 15 µM 24 and 48 h | ↓ Cell viability ↑ Apoptosis ↑ Ca2+ production ↓ Mitochondrial membrane potential | ↑ Cleaved caspase-7 ↑ Cleaved PARP ↑ Chromatin condensation ↑ Cytochrome c ↑ endo G ↑ AIF protein ↓ Bcl-2 protein ↓ BID protein | [80] |
| A549, H460, H1975, H1299 and H520 H82 and H446 LLC | UA 5–40 µM/ 48 h | ↓ Proliferation ↑ Apoptosis ↑ Autophagy ↓ Cell Viability | ↑ Cleaved PARP ↓ Bcl-2 protein ↑ LC3-II protein ↓ p-S6K @ T389 ↓ p-S6 @ S240-244 ↓ p-4E-BPI @ S65 ↓ p-AKT | [81] |
| A549 and H460 | UA 10 and 20 µM | ↓ Proliferation ↑ Apoptosis ↑ G0/G1 cell cycle arrest ↓ Angiogenesis ↓ Migration ↓ Invasion ↓ Tumorsphere formation | ↓ p-EGFR ↓ VEGF ↓ MMP-2 ↓ PD-L1 ↓ CDK4 mRNA and protein ↓ CCND1 mRNA ↓ CCNE1 mRNA ↑ CDKN1A mRNA ↑ CDKN1B mRNA | [82] |
| Human bronchial epithelial cells exposed to cigarette smoke extract | UA 3, 6, 12 and 25 µM | ↓ CSE-induced cytotoxicity | ↑ Nrf2 activity | [68] |
| H1299 | UA 50 and 80 µM 24 h | ↓ Cell survival ↑ Radiosensitivity | ↓ GSH (intracellular) ↓ HIF-1α protein | [74] |
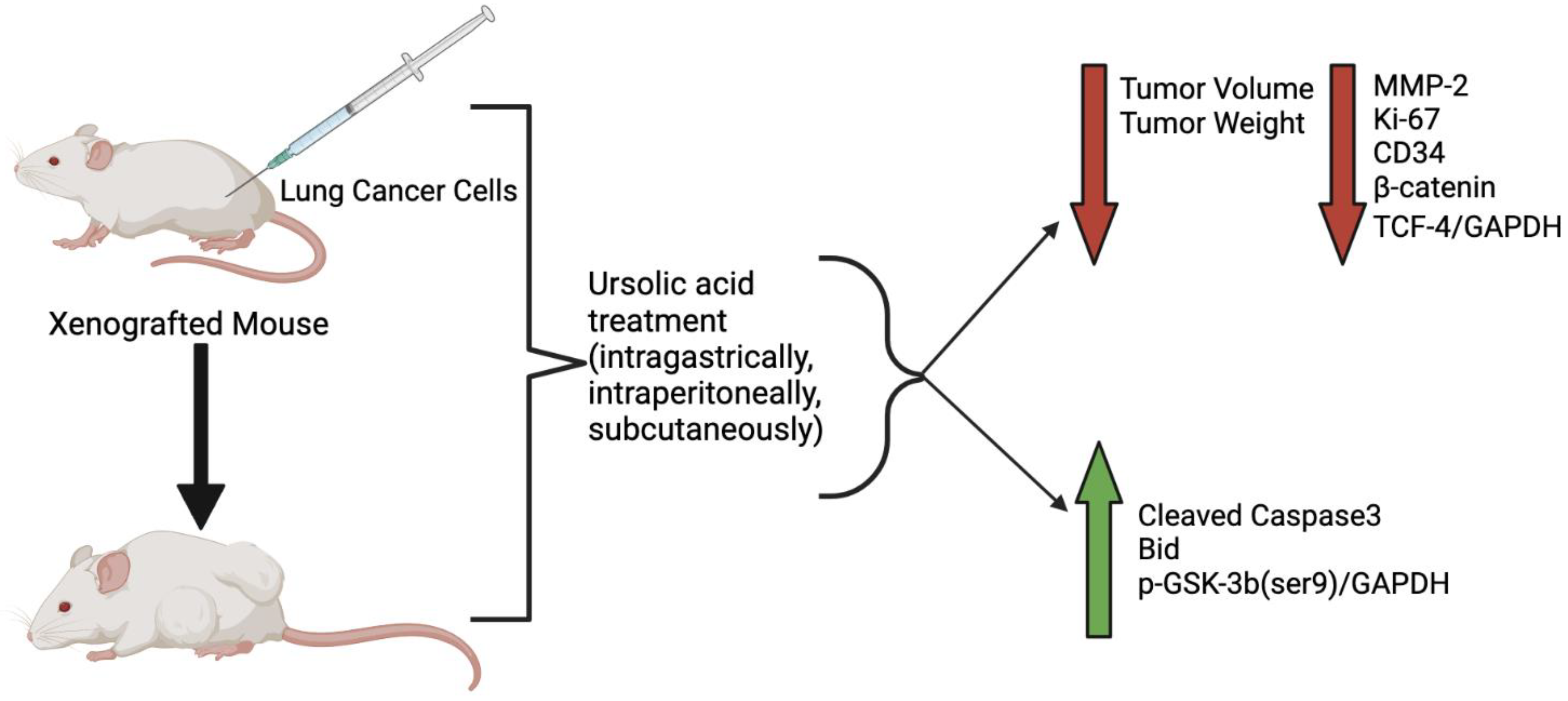
2.2. Effects of Ursolic Acid against Lung Cancer: In Vivo Studies
2.3. Ursolic Acid Derivatives and Their Effect against Lung Cancer
3. Patent Applications and Clinical Trials Related to Ursolic Acid Use
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez-Canales, J.; Parra-Cuentas, E.; Wistuba, I.I. Diagnosis and Molecular Classification of Lung Cancer. Cancer Treat Res. 2016, 170, 25–46. [Google Scholar] [CrossRef] [PubMed]
- Bade, B.C.; Dela Cruz, C.S. Lung Cancer 2020: Epidemiology, Etiology, and Prevention. Clin. Chest Med. 2020, 41, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Nooreldeen, R.; Bach, H. Current and Future Development in Lung Cancer Diagnosis. Int. J. Mol. Sci 2021, 22, 8661. [Google Scholar] [CrossRef] [PubMed]
- Gazdar, A.F.; Bunn, P.A.; Minna, J.D. Small-Cell Lung Cancer: What We Know, What We Need to Know and the Path Forward. Nat. Rev. Cancer 2017, 17, 725–737. [Google Scholar] [CrossRef]
- Arbour, K.C.; Riely, G.J. Systemic Therapy for Locally Advanced and Metastatic Non–Small Cell Lung Cancer: A Review. JAMA 2019, 322, 764–774. [Google Scholar] [CrossRef]
- Wee, P.; Wang, Z. Epidermal Growth Factor Receptor Cell Proliferation Signaling Pathways. Cancers 2017, 9, 52. [Google Scholar] [CrossRef] [Green Version]
- Drosten, M.; Barbacid, M. Targeting the MAPK Pathway in KRAS-Driven Tumors. Cancer Cell 2020, 37, 543–550. [Google Scholar] [CrossRef]
- Shaw, R.J.; Cantley, L.C. Ras, PI(3)K and MTOR Signalling Controls Tumour Cell Growth. Nature 2006, 441, 424–430. [Google Scholar] [CrossRef]
- Mossmann, D.; Park, S.; Hall, M.N. MTOR Signalling and Cellular Metabolism Are Mutual Determinants in Cancer. Nat. Rev. Cancer 2018, 18, 744–757. [Google Scholar] [CrossRef]
- Kim, J.; Guan, K.-L. MTOR as a Central Hub of Nutrient Signalling and Cell Growth. Nat. Cell. Biol. 2019, 21, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Guertin, D.A.; Sabatini, D.M. Defining the Role of MTOR in Cancer. Cancer Cell 2007, 12, 9–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, H.; Luo, Y.; Huang, S. Updates of MTOR Inhibitors. Anticancer Agents Med. Chem. 2010, 10, 571–581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuurbiers, O.C.J.; Kaanders, J.H.A.M.; van der Heijden, H.F.M.; Dekhuijzen, R.P.N.; Oyen, W.J.G.; Bussink, J. The PI3-K/AKT-Pathway and Radiation Resistance Mechanisms in Non-Small Cell Lung Cancer. J. Thorac. Oncol. 2009, 4, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Serna-Blasco, R.; Sanz-Álvarez, M.; Aguilera, Ó.; García-Foncillas, J. Targeting the RAS-Dependent Chemoresistance: The Warburg Connection. Semin. Cancer Biol. 2019, 54, 80–90. [Google Scholar] [CrossRef]
- Liu, R.; Chen, Y.; Liu, G.; Li, C.; Song, Y.; Cao, Z.; Li, W.; Hu, J.; Lu, C.; Liu, Y. PI3K/AKT Pathway as a Key Link Modulates the Multidrug Resistance of Cancers. Cell Death Dis. 2020, 11, 797. [Google Scholar] [CrossRef]
- Yang, J.; Nie, J.; Ma, X.; Wei, Y.; Peng, Y.; Wei, X. Targeting PI3K in Cancer: Mechanisms and Advances in Clinical Trials. Mol. Cancer 2019, 18, 26. [Google Scholar] [CrossRef] [Green Version]
- Mijit, M.; Caracciolo, V.; Melillo, A.; Amicarelli, F.; Giordano, A. Role of P53 in the Regulation of Cellular Senescence. Biomolecules 2020, 10, 420. [Google Scholar] [CrossRef] [Green Version]
- Bykov, V.J.N.; Eriksson, S.E.; Bianchi, J.; Wiman, K.G. Targeting Mutant P53 for Efficient Cancer Therapy. Nat. Rev. Cancer 2018, 18, 89–102. [Google Scholar] [CrossRef]
- Kontomanolis, E.N.; Koutras, A.; Syllaios, A.; Schizas, D.; Mastoraki, A.; Garmpis, N.; Diakosavvas, M.; Angelou, K.; Tsatsaris, G.; Pagkalos, A.; et al. Role of Oncogenes and Tumor-Suppressor Genes in Carcinogenesis: A Review. Anticancer Res. 2020, 40, 6009–6015. [Google Scholar] [CrossRef]
- Xia, L.; Tan, S.; Zhou, Y.; Lin, J.; Wang, H.; Oyang, L.; Tian, Y.; Liu, L.; Su, M.; Wang, H.; et al. Role of the NFκB-Signaling Pathway in Cancer. Onco. Targets. 2018, 11, 2063–2073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuchs, Y.; Steller, H. Programmed Cell Death in Animal Development and Disease. Cell 2011, 147, 742–758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kashyap, D.; Garg, V.K.; Goel, N. Intrinsic and Extrinsic Pathways of Apoptosis: Role in Cancer Development and Prognosis. Adv. Protein. Chem. Struct. Biol. 2021, 125, 73–120. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, B.A.; El-Deiry, W.S. Targeting Apoptosis in Cancer Therapy. Nat. Rev. Clin. Oncol. 2020, 17, 395–417. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, F.R.; Scagliotti, G.V.; Mulshine, J.L.; Kwon, R.; Curran, W.J.; Wu, Y.-L.; Paz-Ares, L. Lung Cancer: Current Therapies and New Targeted Treatments. Lancet 2017, 389, 299–311. [Google Scholar] [CrossRef]
- Yip, H.Y.K.; Papa, A. Signaling Pathways in Cancer: Therapeutic Targets, Combinatorial Treatments, and New Developments. Cells 2021, 10, 659. [Google Scholar] [CrossRef]
- Murugesan, S.; Murugesan, J.; Palaniappan, S.; Palaniappan, S.; Murugan, T.; Siddiqui, S.S.; Loganathan, S. Tyrosine Kinase Inhibitors (TKIs) in Lung Cancer Treatment: A Comprehensive Analysis. Curr. Cancer Drug Targets 2021, 21, 55–69. [Google Scholar] [CrossRef]
- Weaver, B.A. How Taxol/Paclitaxel Kills Cancer Cells. Mol. Biol. Cell 2014, 25, 2677–2681. [Google Scholar] [CrossRef]
- Van Oosterom, A.T.; Schrijvers, D.; Schriivers D [corrected to Schrijvers, D. Docetaxel (Taxotere), a Review of Preclinical and Clinical Experience. Part II: Clinical Experience. Anticancer Drugs 1995, 6, 356–368. [Google Scholar] [CrossRef]
- Wu, S.-G.; Shih, J.-Y. Management of Acquired Resistance to EGFR TKI-Targeted Therapy in Advanced Non-Small Cell Lung Cancer. Mol. Cancer 2018, 17, 38. [Google Scholar] [CrossRef] [Green Version]
- Passaro, A.; Brahmer, J.; Antonia, S.; Mok, T.; Peters, S. Managing Resistance to Immune Checkpoint Inhibitors in Lung Cancer: Treatment and Novel Strategies. J. Clin. Oncol. 2022, 40, 598–610. [Google Scholar] [CrossRef] [PubMed]
- Demain, A.L.; Vaishnav, P. Natural Products for Cancer Chemotherapy. Microb. Biotechnol. 2011, 4, 687–699. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, E.S.; Akter, S.; Ramproshad, S.; Mondal, B.; Riaz, T.A.; Islam, M.T.; Khan, I.N.; Docea, A.O.; Calina, D.; Sharifi-Rad, J.; et al. Targeting Ras-ERK Cascade by Bioactive Natural Products for Potential Treatment of Cancer: An Updated Overview. Cancer Cell Int. 2022, 22, 246. [Google Scholar] [CrossRef] [PubMed]
- Mlala, S.; Oyedeji, A.O.; Gondwe, M.; Oyedeji, O.O. Ursolic Acid and Its Derivatives as Bioactive Agents. Molecules 2019, 24, 2751. [Google Scholar] [CrossRef] [Green Version]
- Price, K.R.; Johnson, I.T.; Fenwick, G.R. The Chemistry and Biological Significance of Saponins in Foods and Feedingstuffs. Crit. Rev. Food Sci. Nutr. 1987, 26, 27–135. [Google Scholar] [CrossRef]
- Ivanov, I.; Petkova, N.; Tumbarski, Y.; Vrancheva, R.; Stoyanova, M. Lavender Waste – Promising Source of Triterpenoids and Polyphenols with Antioxidant and Antimicrobial Activity. Ind. Technol. 2018, 5, 26–32. [Google Scholar]
- Woźniak, Ł.; Skąpska, S.; Marszałek, K. Ursolic Acid—A Pentacyclic Triterpenoid with a Wide Spectrum of Pharmacological Activities. Molecules 2015, 20, 20614–20641. [Google Scholar] [CrossRef] [Green Version]
- Kowalski, R. Studies of Selected Plant Raw Materials as Alternative Sources of Triterpenes of Oleanolic and Ursolic Acid Types. J. Agric. Food Chem. 2007, 55, 656–662. [Google Scholar] [CrossRef]
- Silva, M.G.V.; Vieira, I.G.P.; Mendes, F.N.P.; Albuquerque, I.L.; dos Santos, R.N.; Silva, F.O.; Morais, S.M. Variation of Ursolic Acid Content in Eight Ocimum Species from Northeastern Brazil. Molecules 2008, 13, 2482–2487. [Google Scholar] [CrossRef]
- Jäger, S.; Trojan, H.; Kopp, T.; Laszczyk, M.; Scheffler, A. Pentacyclic Triterpene Distribution in Various Plants – Rich Sources for a New Group of Multi-Potent Plant Extracts. Molecules 2009, 14, 2016–2031. [Google Scholar] [CrossRef] [Green Version]
- Guinda, Á.; Rada, M.; Delgado, T.; Gutiérrez-Adánez, P.; Castellano, J.M. Pentacyclic Triterpenoids from Olive Fruit and Leaf. J. Agric. Food Chem. 2010, 58, 9685–9691. [Google Scholar] [CrossRef] [PubMed]
- Radojković, M.; Vujanović, M.; Majkić, T.; Zengin, G.; Beara, I.; Catauro, M.; Montesano, D. Evaluation of Sambucus Nigra, L. Biopotential as an Unused Natural Resource. Appl. Sci. 2021, 11, 11207. [Google Scholar] [CrossRef]
- Butkeviciute, A.; Viskelis, J.; Liaudanskas, M.; Viskelis, P.; Bobinas, C.; Janulis, V. Variation of Triterpenes in Apples Stored in a Controlled Atmosphere. Molecules 2021, 26, 3639. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Liu, R.H. Triterpenoids Isolated from Apple Peels Have Potent Antiproliferative Activity and May Be Partially Responsible for Apple’s Anticancer Activity. J. Agric. Food. Chem. 2007, 55, 4366–4370. [Google Scholar] [CrossRef]
- Kondo, M.; MacKinnon, S.L.; Craft, C.C.; Matchett, M.D.; Hurta, R.A.R.; Neto, C.C. Ursolic Acid and Its Esters: Occurrence in Cranberries and Other Vaccinium Fruit and Effects on Matrix Metalloproteinase Activity in DU145 Prostate Tumor Cells: Anti-Tumor Activity and Content of Ursolic Acid from Vaccinium Fruit. J. Sci. Food Agric. 2011, 91, 789–796. [Google Scholar] [CrossRef]
- Sun, L.; Tao, S.; Zhang, S. Characterization and Quantification of Polyphenols and Triterpenoids in Thinned Young Fruits of Ten Pear Varieties by UPLC-Q TRAP-MS/MS. Molecules 2019, 24, 159. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Wang, T.; Zhou, B.; Gao, W.; Cao, J.; Huang, L. Chemical Composition and Antioxidant and Anti-Inflammatory Potential of Peels and Flesh from 10 Different Pear Varieties (Pyrus Spp.). Food Chem. 2014, 152, 531–538. [Google Scholar] [CrossRef]
- Allouche, Y.; Jiménez, A.; Uceda, M.; Aguilera, M.P.; Gaforio, J.J.; Beltrán, G. Triterpenic Content and Chemometric Analysis of Virgin Olive Oils from Forty Olive Cultivars. J. Agric. Food Chem. 2009, 57, 3604–3610. [Google Scholar] [CrossRef]
- Wójciak-Kosior, M.; Sowa, I.; Kocjan, R.; Nowak, R. Effect of Different Extraction Techniques on Quantification of Oleanolic and Ursolic Acid in Lamii Albi Flos. Ind. Crops Prod. 2013, 44, 373–377. [Google Scholar] [CrossRef]
- Yin, M.-C.; Lin, M.-C.; Mong, M.-C.; Lin, C.-Y. Bioavailability, Distribution, and Antioxidative Effects of Selected Triterpenes in Mice. J. Agric. Food Chem. 2012, 60, 7697–7701. [Google Scholar] [CrossRef]
- Shanmugam, M.K.; Dai, X.; Kumar, A.P.; Tan, B.K.H.; Sethi, G.; Bishayee, A. Ursolic Acid in Cancer Prevention and Treatment: Molecular Targets, Pharmacokinetics and Clinical Studies. Biochemical. Pharmacol. 2013, 85, 1579–1587. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.Y.; Lee, S.R.; Heo, J.-W.; No, M.-H.; Rhee, B.D.; Ko, K.S.; Kwak, H.-B.; Han, J. Ursolic Acid in Health and Disease. Korean J. Physiol. Pharm. 2018, 22, 235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kashyap, D.; Sharma, A.; Tuli, H.S.; Punia, S.; Sharma, A.K. Ursolic Acid and Oleanolic Acid: Pentacyclic Terpenoids with Promising Anti-Inflammatory Activities. IAD 2016, 10, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zheng, H.; Sui, Z.; Jing, F.; Quan, X.; Zhao, W.; Liu, G. Ursolic Acid Exhibits Anti-Inflammatory Effects through Blocking TLR4-MyD88 Pathway Mediated by Autophagy. Cytokine 2019, 123, 154726. [Google Scholar] [CrossRef] [PubMed]
- Habtemariam, S. Antioxidant and Anti-Inflammatory Mechanisms of Neuroprotection by Ursolic Acid: Addressing Brain Injury, Cerebral Ischemia, Cognition Deficit, Anxiety, and Depression. Oxidative Med. Cell. Longev. 2019, 2019, 8512048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gudoityte, E.; Arandarcikaite, O.; Mazeikiene, I.; Bendokas, V.; Liobikas, J. Ursolic and Oleanolic Acids: Plant Metabolites with Neuroprotective Potential. IJMS 2021, 22, 4599. [Google Scholar] [CrossRef] [PubMed]
- Camer, D.; Yu, Y.; Szabo, A.; Huang, X.-F. The Molecular Mechanisms Underpinning the Therapeutic Properties of Oleanolic Acid, Its Isomer and Derivatives for Type 2 Diabetes and Associated Complications. Mol. Nutr. Food Res. 2014, 58, 1750–1759. [Google Scholar] [CrossRef] [Green Version]
- Chan, E.W.C.; Soon, C.Y.; Tan, J.B.L.; Wong, S.K.; Hui, Y.W. Ursolic Acid: An Overview on Its Cytotoxic Activities against Breast and Colorectal Cancer Cells. J. Integr. Med. 2019, 17, 155–160. [Google Scholar] [CrossRef]
- Iqbal, J.; Abbasi, B.A.; Ahmad, R.; Mahmood, T.; Kanwal, S.; Ali, B.; Khalil, A.T.; Shah, S.A.; Alam, M.M.; Badshah, H. Ursolic Acid a Promising Candidate in the Therapeutics of Breast Cancer: Current Status and Future Implications. Biomed. Pharmacother. 2018, 108, 752–756. [Google Scholar] [CrossRef]
- Zou, J.; Lin, J.; Li, C.; Zhao, R.; Fan, L.; Yu, J.; Shao, J. Ursolic Acid in Cancer Treatment and Metastatic Chemoprevention: From Synthesized Derivatives to Nanoformulations in Preclinical Studies. CCDT 2019, 19, 245–256. [Google Scholar] [CrossRef]
- Wang, L.; Yin, Q.; Liu, C.; Tang, Y.; Sun, C.; Zhuang, J. Nanoformulations of Ursolic Acid: A Modern Natural Anticancer Molecule. Front. Pharmacol. 2021, 12, 706121. [Google Scholar] [CrossRef] [PubMed]
- Yin, R.; Li, T.; Tian, J.X.; Xi, P.; Liu, R.H. Ursolic Acid, a Potential Anticancer Compound for Breast Cancer Therapy. Crit. Rev. Food Sci. Nutr. 2018, 58, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.; Ali, S.; Ahmed, S.; Elasbali, A.M.; Adnan, M.; Islam, A.; Hassan, M.I.; Yadav, D.K. Therapeutic Potential of Ursolic Acid in Cancer and Diabetic Neuropathy Diseases. Int. J. Mol. Sci. 2021, 22, 12162. [Google Scholar] [CrossRef] [PubMed]
- Khwaza, V.; Oyedeji, O.O.; Aderibigbe, B.A. Ursolic Acid-Based Derivatives as Potential Anti-Cancer Agents: An Update. Int. J. Mol. Sci. 2020, 21, 5920. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.-L.; Kuo, P.-L.; Lin, C.-C. Proliferative Inhibition, Cell-Cycle Dysregulation, and Induction of Apoptosis by Ursolic Acid in Human Non-Small Cell Lung Cancer A549 Cells. Life Sci. 2004, 75, 2303–2316. [Google Scholar] [CrossRef]
- Lai, M.Y.; Leung, H.W.C.; Yang, W.H.; Chen, W.H.; Lee, H.Z. Up-Regulation of Matrix Metalloproteinase Family Gene Involvement in Ursolic Acid-Induced Human Lung Non-Small Carcinoma Cell Apoptosis. Anticancer Res. 2007, 27, 145–153. [Google Scholar]
- Huang, C.-Y.; Lin, C.-Y.; Tsai, C.-W.; Yin, M.-C. Inhibition of Cell Proliferation, Invasion and Migration by Ursolic Acid in Human Lung Cancer Cell Lines. Toxicol. Vitr. 2011, 25, 1274–1280. [Google Scholar] [CrossRef]
- Liu, W.; Tan, X.; Shu, L.; Sun, H.; Song, J.; Jin, P.; Yu, S.; Sun, M.; Jia, X. Ursolic Acid Inhibits Cigarette Smoke Extract-Induced Human Bronchial Epithelial Cell Injury and Prevents Development of Lung Cancer. Molecules 2012, 17, 9104–9115. [Google Scholar] [CrossRef]
- Liu, K.; Guo, L.; Miao, L.; Bao, W.; Yang, J.; Li, X.; Xi, T.; Zhao, W. Ursolic Acid Inhibits Epithelial–Mesenchymal Transition by Suppressing the Expression of Astrocyte-Elevated Gene-1 in Human Nonsmall Cell Lung Cancer A549 Cells. Anti-Cancer Drugs 2013, 24, 494–503. [Google Scholar] [CrossRef]
- Way, T.-D.; Tsai, S.-J.; Wang, C.-M.; Ho, C.-T.; Chou, C.-H. Chemical Constituents of Rhododendron Formosanum Show Pronounced Growth Inhibitory Effect on Non-Small-Cell Lung Carcinoma Cells. J. Agric. Food Chem. 2014, 62, 875–884. [Google Scholar] [CrossRef]
- Kim, S.-H.; Ryu, H.G.; Lee, J.; Shin, J.; Harikishore, A.; Jung, H.-Y.; Kim, Y.S.; Lyu, H.-N.; Oh, E.; Baek, N.-I.; et al. Ursolic Acid Exerts Anti-Cancer Activity by Suppressing Vaccinia-Related Kinase 1-Mediated Damage Repair in Lung Cancer Cells. Sci. Rep. 2015, 5, 14570. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Zhao, S.; Tang, Q.; Zheng, F.; Chen, Y.; Yang, L.; Yang, X.; Li, L.; Wu, W.; Hann, S.S. Activation of SAPK/JNK Mediated the Inhibition and Reciprocal Interaction of DNA Methyltransferase 1 and EZH2 by Ursolic Acid in Human Lung Cancer Cells. J. Exp. Clin. Cancer Res. 2015, 34, 99. [Google Scholar] [CrossRef] [PubMed]
- Sohn, E.J.; Won, G.; Lee, J.; Yoon, S.W.; Lee, I.; Kim, H.J.; Kim, S.-H. Blockage of Epithelial to Mesenchymal Transition and Upregulation of Let 7b Are Critically Involved in Ursolic Acid Induced Apoptosis in Malignant Mesothelioma Cell. Int. J. Biol. Sci. 2016, 12, 1279–1288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, B.; Zhang, Q.; Yu, M.; Qi, X.; Wang, G.; Xiao, L.; Yi, Q.; Jin, W. Ursolic Acid Sensitizes Radioresistant NSCLC Cells Expressing HIF-1α through Reducing Endogenous GSH and Inhibiting HIF-1α. Oncol. Lett. 2017, 13, 754–762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.-J.; Liang, W.-M.; Chen, C.-J.; Tsang, H.; Chiou, J.-S.; Liu, X.; Cheng, C.-F.; Lin, T.-H.; Liao, C.-C.; Huang, S.-M.; et al. Network Analysis and Mechanisms of Action of Chinese Herb-Related Natural Compounds in Lung Cancer Cells. Phytomedicine 2019, 58, 152893. [Google Scholar] [CrossRef] [PubMed]
- Castrejón-Jiménez, N.S.; Leyva-Paredes, K.; Baltierra-Uribe, S.L.; Castillo-Cruz, J.; Campillo-Navarro, M.; Hernández-Pérez, A.D.; Luna-Angulo, A.B.; Chacón-Salinas, R.; Coral-Vázquez, R.M.; Estrada-García, I.; et al. Ursolic and Oleanolic Acids Induce Mitophagy in A549 Human Lung Cancer Cells. Molecules 2019, 24, 3444. [Google Scholar] [CrossRef] [Green Version]
- Yang, K.; Chen, Y.; Zhou, J.; Ma, L.; Shan, Y.; Cheng, X.; Wang, Y.; Zhang, Z.; Ji, X.; Chen, L.; et al. Ursolic Acid Promotes Apoptosis and Mediates Transcriptional Suppression of CT45A2 Gene Expression in Non-small-cell Lung Carcinoma Harbouring EGFR T790M Mutations. Br. J. Pharm. 2019, 176, 4609–4624. [Google Scholar] [CrossRef]
- Chen, Q.; Luo, J.; Wu, C.; Lu, H.; Cai, S.; Bao, C.; Liu, D.; Kong, J. The MiRNA-149-5p/MyD88 Axis Is Responsible for Ursolic Acid-mediated Attenuation of the Stemness and Chemoresistance of Non-small Cell Lung Cancer Cells. Environ. Toxicol. 2020, 35, 561–569. [Google Scholar] [CrossRef]
- Ruan, J.S.; Zhou, H.; Yang, L.; Wang, L.; Jiang, Z.S.; Sun, H.; Wang, S.M. Ursolic Acid Attenuates TGF-Β1-Induced Epithelial-Mesenchymal Transition in NSCLC by Targeting Integrin AVβ5/MMPs Signaling. Oncol. Res. 2019, 27, 593–600. [Google Scholar] [CrossRef]
- Chen, C.-J.; Shih, Y.-L.; Yeh, M.-Y.; Liao, N.-C.; Chung, H.-Y.; Liu, K.-L.; Lee, M.-H.; Chou, P.-Y.; Hou, H.-Y.; Chou, J.-S.; et al. Ursolic Acid Induces Apoptotic Cell Death Through AIF and Endo G Release Through a Mitochondria-Dependent Pathway in NCI-H292 Human Lung Cancer Cells In Vitro. Vivo 2019, 33, 383–391. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Yu, H.; Wu, R.; Chen, Z.; Hu, Q.; Zhang, Y.; Gao, S.; Zhou, G. Autophagy Inhibition Enhances the Inhibitory Effects of Ursolic Acid on Lung Cancer Cells. Int. J. Mol. Med. 2020, 46, 1816–1826. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.Y.; Sp, N.; Lee, J.-M.; Jang, K.-J. Antitumor Effects of Ursolic Acid through Mediating the Inhibition of STAT3/PD-L1 Signaling in Non-Small Cell Lung Cancer Cells. Biomedicines 2021, 9, 297. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.S.; Yuan, Y.; Song, G.; Lin, S.Q. Inhibitory Effect of Ursolic Acid and Oleanolic Acid from Eriobotrya Fragrans on A549 Cell Viability in Vivo. Genet. Mol. Res. 2016, 15, 1–8. [Google Scholar] [CrossRef]
- Kalani, K.; Yadav, D.K.; Khan, F.; Srivastava, S.K.; Suri, N. Pharmacophore, QSAR, and ADME Based Semisynthesis and in Vitro Evaluation of Ursolic Acid Analogs for Anticancer Activity. J. Mol. Model 2012, 18, 3389–3413. [Google Scholar] [CrossRef] [PubMed]
- Rashid, S.; Dar, B.A.; Majeed, R.; Hamid, A.; Bhat, B.A. Synthesis and Biological Evaluation of Ursolic Acid-Triazolyl Derivatives as Potential Anti-Cancer Agents. Eur. J. Med. Chem. 2013, 66, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Dar, B.A.; Lone, A.M.; Shah, W.A.; Qurishi, M.A. Synthesis and Screening of Ursolic Acid-Benzylidine Derivatives as Potential Anti-Cancer Agents. Eur. J. Med. Chem. 2016, 111, 26–32. [Google Scholar] [CrossRef]
- Mendes, V.I.S.; Bartholomeusz, G.A.; Ayres, M.; Gandhi, V.; Salvador, J.A.R. Synthesis and Cytotoxic Activity of Novel A-Ring Cleaved Ursolic Acid Derivatives in Human Non-Small Cell Lung Cancer Cells. Eur. J. Med. Chem. 2016, 123, 317–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gou, W.; Luo, N.; Wei, H.; Wu, H.; Yu, X.; Duan, Y.; Bi, C.; Ning, H.; Hou, W.; Li, Y. Ursolic Acid Derivative UA232 Evokes Apoptosis of Lung Cancer Cells Induced by Endoplasmic Reticulum Stress. Pharm. Biol. 2020, 58, 707–715. [Google Scholar] [CrossRef]
- Huang, R.-Z.; Hua, S.-X.; Liao, Z.-X.; Huang, X.-C.; Wang, H.-S. Side Chain-Functionalized Aniline-Derived Ursolic Acid Derivatives as Multidrug Resistance Reversers That Block the Nuclear Factor-Kappa B (NF-ΚB) Pathway and Cell Proliferation. Med. Chem. Commun. 2017, 8, 1421–1434. [Google Scholar] [CrossRef]
- Jiang, W.; Huang, R.-Z.; Zhang, J.; Guo, T.; Zhang, M.-T.; Huang, X.-C.; Zhang, B.; Liao, Z.-X.; Sun, J.; Wang, H.-S. Discovery of Antitumor Ursolic Acid Long-Chain Diamine Derivatives as Potent Inhibitors of NF-ΚB. Bioorg. Chem. 2018, 79, 265–276. [Google Scholar] [CrossRef]
- Yang, L.; Sun, Z.; Zu, Y.; Zhao, C.; Sun, X.; Zhang, Z.; Zhang, L. Physicochemical Properties and Oral Bioavailability of Ursolic Acid Nanoparticles Using Supercritical Anti-Solvent (SAS) Process. Food Chem. 2012, 132, 319–325. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, W.; Jiang, Y.; Wang, X.; Zhang, X.; Liu, H.; Zhang, T. Preparation of Ursolic Acid–Phospholipid Complex by Solvent-Assisted Grinding Method to Improve Dissolution and Oral Bioavailability. Pharm. Dev. Technol. 2020, 25, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Wei, G.; Si, D.; Liu, C. Quantitation of Ursolic Acid in Human Plasma by Ultra Performance Liquid Chromatography Tandem Mass Spectrometry and Its Pharmacokinetic Study. J. Chromatogr. B 2011, 879, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Rodríguez, A.M.; González-Ortiz, M.; Martínez-Abundis, E.; Acuña Ortega, N. Effect of Ursolic Acid on Metabolic Syndrome, Insulin Sensitivity, and Inflammation. J. Med. Food 2017, 20, 882–886. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-H.; Zhou, S.-Y.; Qian, Z.-Z.; Zhang, H.-L.; Qiu, L.-H.; Song, Z.; Zhao, J.; Wang, P.; Hao, X.-S.; Wang, H.-Q. Evaluation of Toxicity and Single-Dose Pharmacokinetics of Intravenous Ursolic Acid Liposomes in Healthy Adult Volunteers and Patients with Advanced Solid Tumors. Expert Opin. Drug Metab. Toxicol. 2013, 9, 117–125. [Google Scholar] [CrossRef]
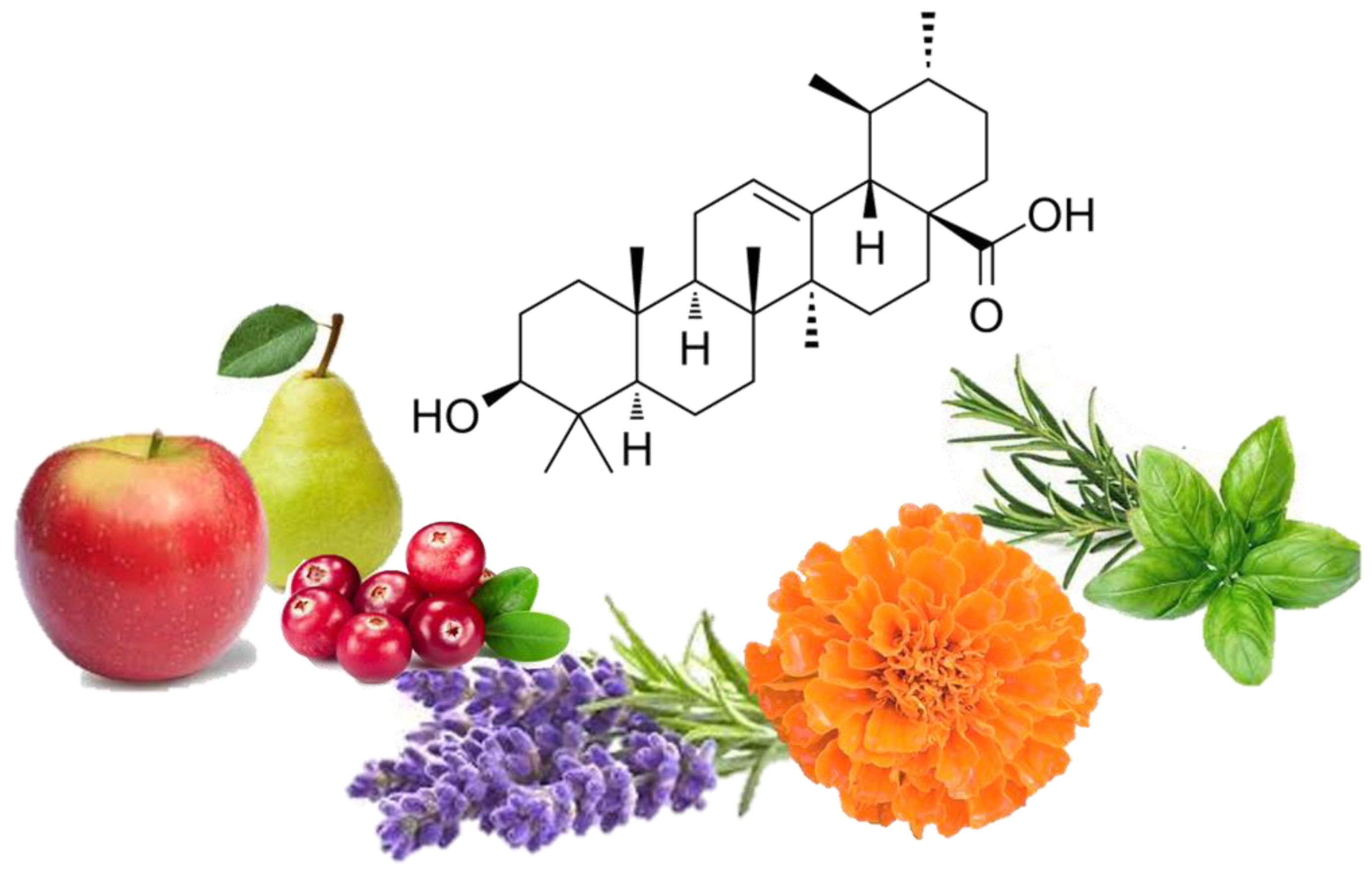

| Source | Concentration of UA | Reference | ||
|---|---|---|---|---|
| Common Name | Botanical Name | |||
| Plants | Lavender | Lavandula | 106.7–153.1 mg/g 3.463–6.484 mg/g DW | [36] |
| White deadnettle | Lamii albi flos | 39.1–110.4 mg/g DW | [49] | |
| Marigold | Calendula officinalis | 20.53 mg/g D.W | [38] | |
| Basil | Ocimum tenuiflorum | 20.2 mg/g D.W | [39] | |
| Rosinweed, cup plant, compass plant | Silphium sp. flowers | 17.95–22.05 mg/g D.W | [38] | |
| Rosemary | Rosmarinus officinalis | 15.8–29.5 mg/g D.W | [40] | |
| Daylily | Hemerocallis sp | 0.19 ± 0.05 mg/g DW | [50] | |
| Fruits | Black elderberry extract | Sambucus nigra L | 6.62 ± 0.26–0.002 mg/g | [42] |
| Olive—adult olive tree leaves | Olea europaea L. | 2.23 ± 0.1 mg/g | [41] | |
| Apple—apple peel | Malus | 1.52 mg/g DW | [44] | |
| Apple—whole apple | Malus | 0.77 ± 0.1 mg/g to 1.85 ± 0.17 mg/g | [43] | |
| Cranberry | Vaccinium macrocarpon | 0.46–1.09 mg/g FW | [45] | |
| Pear—mature fruit peel | Pyrus | 0.3481 mg/g | [47] | |
| Pear—young fruit | Pyrus | 0.1293 mg/g FW | [46] | |
| Olive—virgin olive oil | Olea europaea L. | 0.00138 ± 0.00015 mg/g | [48] | |
| Xenograft Model | Dose/Duration | Findings | Mechanism | Reference |
|---|---|---|---|---|
| 6–8-week nude mice A549 cells injected subcutaneously | UA—10 mg/kg intragastrical administration/ 1 week | ↓ Tumor volume | Not investigated | [68] |
| C57 BL/6 mice injected with LLC-luciferase (1 × 107 cells/mouse) | UA—100 mg/kg intraperitoneally injected | ↓ Tumor volume ↓ Tumor weight | ↓ VRK1 activity | [71] |
| Female Balb/c nude mice A549 cells 2 × 106 cells/mouse | UA 50 or 100 mg/kg subcutaneous injection/every other day for 2 weeks | ↓ Tumor growth ↓ Tumor weight | ↓ MMP-2 ↓ Ki-67 ↓ CD34 ↑ Bid | [83] |
| Athymic nude mice H1975 cells subcutaneously injected 5 × 106 cells/mouse | UA—25 mg/kg−1 daily for 18 days | ↓ Tumor growth ↓ Tumor weight | Not investigated | [77] |
| Athymic Balb/c nude mice A549-PR cells | UA 20 µM 72hr pre-injection co-culture | ↓ Tumorigenesis | Not investigated | [78] |
| Cell Line | Derivative Name | Derivative Structure | Findings | Mechanism | Reference |
|---|---|---|---|---|---|
| A549 SF-295 (CNS) | UA-9 10 nM | 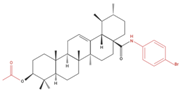 | ↓ Cell Density | Not investigated | [84] |
| A549 | UA-triazolyl derivative |  | ↓ Cell Density | Not investigated | [85] |
| A549 | Compound 3B 50 µM, 48h | 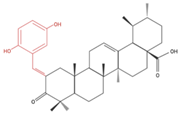 | ↓ Cell Growth | Not investigated | [86] |
| H460 H322 | Compound 17 | 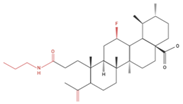 | ↑ Apoptosis ↑ Autophagy | ↑ Cleaved caspases 8 and 7 ↑ Cleaved PARP ↑ LC3A/B-II ratio ↓ Bcl-2 protein ↓ mTOR protein | [87] |
| A549 H460 | UA232 24, 48 and 72 h |  | ↑ Cell cycle arrest ↑ Apoptosis ↓ Proliferation | ↓ Cyclin D1 protein ↓ CDK4 protein ↑ CHOP protein ↑ Cleaved PARP | [88] |
| NCI-H460 | 5Y8 5 and 10 µM |  | ↑ Apoptosis G1 phase cell cycle arrest | ↓ p-NF-kB ↓ p-IKKα/β ↓ TAK1 ↓ TAB1 ↑ ROS | [89] |
| A549 | 8c 5, 10 and 20 µM 24 h | 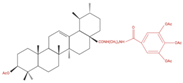 | ↑ Apoptosis G1 phase cell cycle arrest ↓ Cell migration | ↑ Caspase-3 cleavage ↓ p-NF-kB ↓ p-IKBα ↓ p-IKKα/β | [90] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kornel, A.; Nadile, M.; Tsiani, E. Evidence of the Beneficial Effects of Ursolic Acid against Lung Cancer. Molecules 2022, 27, 7466. https://doi.org/10.3390/molecules27217466
Kornel A, Nadile M, Tsiani E. Evidence of the Beneficial Effects of Ursolic Acid against Lung Cancer. Molecules. 2022; 27(21):7466. https://doi.org/10.3390/molecules27217466
Chicago/Turabian StyleKornel, Amanda, Matteo Nadile, and Evangelia Tsiani. 2022. "Evidence of the Beneficial Effects of Ursolic Acid against Lung Cancer" Molecules 27, no. 21: 7466. https://doi.org/10.3390/molecules27217466







