Qualitative Analysis Related to Palynological Characterization and Biological Evaluation of Propolis from Prespa National Park (Greece)
Abstract
1. Introduction
2. Results
2.1. Propolis Composition
2.1.1. GC-MS Analysis
2.1.2. Isolation of Compounds
2.2. Pollen Grain Analysis
2.3. Antimicrobial Activity
2.4. Total Phenolic Content and Antioxidant Activity
3. Discussion
4. Materials and Methods
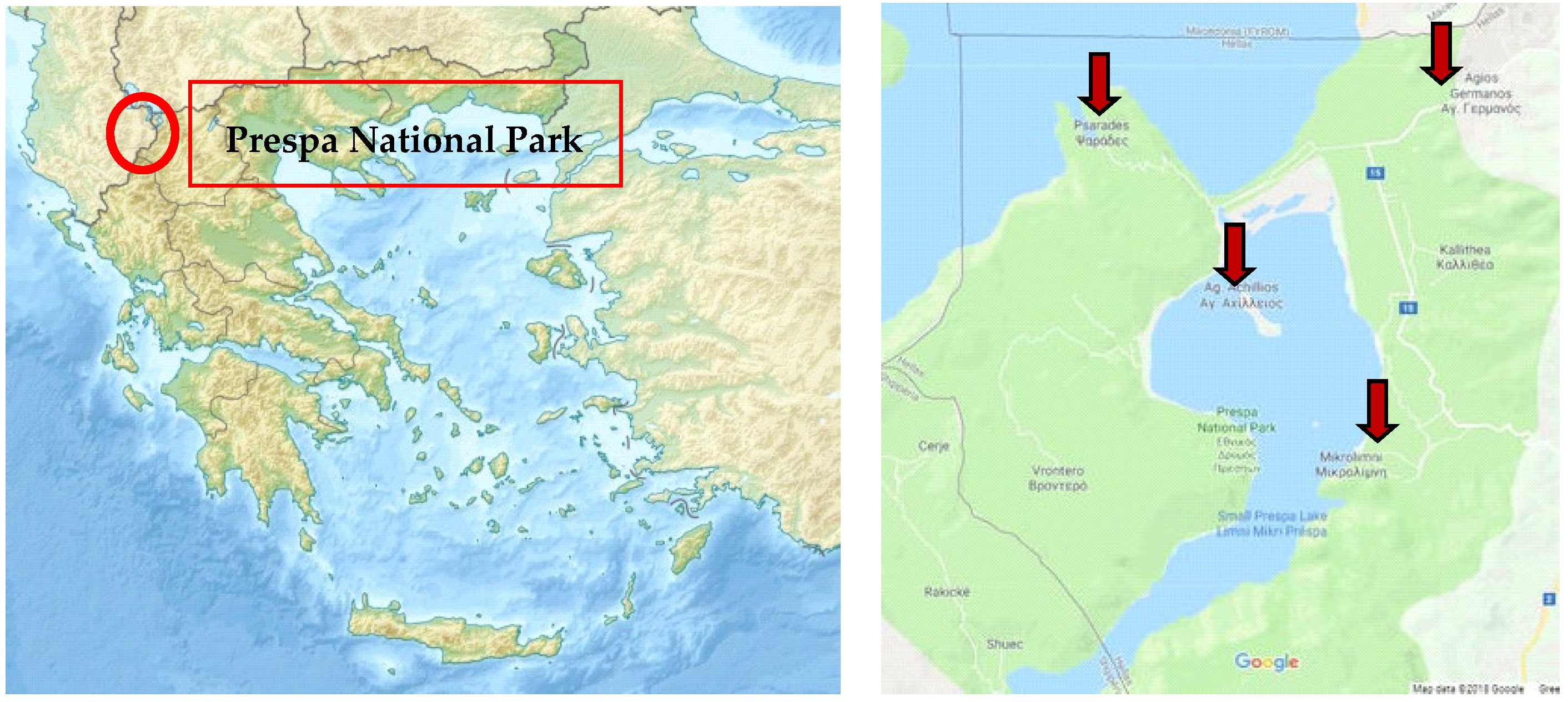
4.1. Samples
4.2. Pollen Analysis
4.3. Extraction and Sample Derivatization
4.4. GC-MS Analysis
4.5. Isolation of Compounds
4.6. Antimicrobial Bioassay
4.7. Total Phenolic Content
4.8. DPPH Assay
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Graikou, K.; Popova, M.; Gortzi, O.; Bankova, V.; Chinou, I. Characterization and Biological Evaluation of Selected Mediterranean Propolis Samples. Is It a New Type? LWT-Food Sci. Technol. 2016, 65, 261–267. [Google Scholar] [CrossRef]
- Santos, L.M.; Fonseca, M.S.; Sokolonski, A.R.; Deegan, K.R.; Araújo, R.P.; Umsza-Guez, M.A.; Barbosa, J.D.; Portela, R.D.; Machado, B.A. Propolis: Types, Composition, Biological Activities, and Veterinary Product Patent Prospecting. J. Sci. Food Agric. 2020, 100, 1369–1382. [Google Scholar] [CrossRef] [PubMed]
- Popova, M.P.; Graikou, K.; Chinou, I.; Bankova, V.S. GC-MS Profiling of Diterpene Compounds in Mediterranean Propolis from Greece. J. Agric. Food Chem. 2010, 58, 3167–3176. [Google Scholar] [CrossRef] [PubMed]
- Anjum, S.I.; Ullah, A.; Khan, K.A.; Attaullah, M.; Khan, H.; Ali, H.; Bashir, M.A.; Tahir, M.; Ansari, M.J.; Ghramh, H.A.; et al. Composition and Functional Properties of Propolis (Bee Glue): A Review. Saudi J. Biol. Sci. 2019, 26, 1695–1703. [Google Scholar] [CrossRef]
- Popova, M.; Giannopoulou, E.; Skalicka-Woźniak, K.; Graikou, K.; Widelski, J.; Bankova, V.; Kalofonos, H.; Sivolapenko, G.; Gaweł-Bęben, K.; Antosiewicz, B.; et al. Characterization and Biological Evaluation of Propolis from Poland. Molecules 2017, 22, 1159. [Google Scholar] [CrossRef]
- El-Guendouz, S.; Lyoussi, B.; Miguel, M.G. Insight on Propolis from Mediterranean Countries: Chemical Composition, Biological Activities and Application Fields. Chem. Biodivers. 2019, 16, e1900094. [Google Scholar] [CrossRef]
- Pasupuleti, V.R.; Sammugam, L.; Ramesh, N.; Gan, S.H. Honey, Propolis, and Royal Jelly: A Comprehensive Review of Their Biological Actions and Health Benefits. Oxid. Med. Cell. Longev. 2017, 2017, 1259510. [Google Scholar] [CrossRef]
- Bankova, V.; Bertelli, D.; Borba, R.; Conti, B.J.; da Silva Cunha, I.B.; Danert, C.; Eberlin, M.N.; Falcão, S.I.; Isla, M.I.; Moreno, M.I.N.; et al. Standard Methods for Apis Mellifera Propolis Research. J. Apic. Res. 2019, 58, 1–49. [Google Scholar] [CrossRef]
- Al-Hariri, M. Immune’s-Boosting Agent: Immunomodulation Potentials of Propolis. J. Fam. Community Med. 2019, 26, 57–60. [Google Scholar] [CrossRef]
- Guler, H.I.; Tatar, G.; Yildiz, O.; Belduz, A.O.; Kolayli, S. Investigation of Potential Inhibitor Properties of Ethanolic Propolis Extracts against ACE-II Receptors for COVID-19 Treatment by Molecular Docking Study. Arch. Microbiol. 2021, 203, 3557–3564. [Google Scholar] [CrossRef]
- Refaat, H.; Mady, F.M.; Sarhan, H.A.; Rateb, H.S.; Alaaeldin, E. Optimization and Evaluation of Propolis Liposomes as a Promising Therapeutic Approach for COVID-19. Int. J. Pharm. 2021, 592, 120028. [Google Scholar] [CrossRef]
- Shaldam, M.A.; Yahya, G.; Mohamed, N.H.; Abdel-Daim, M.M.; Al Naggar, Y. In Silico Screening of Potent Bioactive Compounds from Honeybee Products against COVID-19 Target Enzymes. Environ. Sci. Pollut. Res. 2021, 28, 40507–40514. [Google Scholar] [CrossRef]
- Silveira, M.A.D.; De Jong, D.; Berretta, A.A.; dos Santos Galvão, E.B.; Ribeiro, J.C.; Cerqueira-Silva, T.; Amorim, T.C.; da Conceição, L.F.M.R.; Gomes, M.M.D.; Teixeira, M.B.; et al. Efficacy of Brazilian Green Propolis (EPP-AF®) as an Adjunct Treatment for Hospitalized COVID-19 Patients: A Randomized, Controlled Clinical Trial. Biomed. Pharmacother. 2021, 138, 111526. [Google Scholar] [CrossRef]
- Bachevski, D.; Damevska, K.; Simeonovski, V.; Dimova, M. Back to the Basics: Propolis and COVID-19. Dermatol. Ther. 2020, 33, e13780. [Google Scholar] [CrossRef]
- Berretta, A.A.; Silveira, M.A.D.; Cóndor Capcha, J.M.; De Jong, D. Propolis and Its Potential against SARS-CoV-2 Infection Mechanisms and COVID-19 Disease. Biomed. Pharmacother. 2020, 131, 110622. [Google Scholar] [CrossRef]
- UNESCO. Available online: https://whc.unesco.org/en/tentativelists/5864/ (accessed on 9 September 2022).
- Strid, A.; Bergmeier, E.; Fotiadis, G. Flora and Vegetation of the Prespa National Park, Greece; Society for the Protection of Prespa: Athens, Greece, 2020; ISBN 978-960-302-285-5.
- Bergmeier, E.; Sakellarakis, F.N.; Strid, A.; Swinkels, C. New additions to the flora of Prespa, Greece. Phytol. Balc. 2020, 26, 103–130. [Google Scholar]
- Pavlides, G. The Flora of Prespa National Park with Emphasis on Species of Conservation Interest. Hydrobiologia 1997, 351, 35–40. [Google Scholar] [CrossRef]
- Bankova, V.; Popova, M.; Bogdanov, S.; Sabatini, A.-G. Chemical Composition of European Propolis: Expected and Unexpected Results. Z. Nat. C 2002, 57, 530–533. [Google Scholar] [CrossRef]
- Graikou, K.; Andreou, A.; Chinou, I. Chemical profile οf Greek Arbutusunedo honey: Biological properties. J. Apic. Res. 2022, 61, 100–106. [Google Scholar] [CrossRef]
- Sharonova, N.; Nikitin, E.; Terenzhev, D.; Lyubina, A.; Amerhanova, S.; Bushmeleva, K.; Rakhmaeva, A.; Fitsev, I.; Sinyashin, K. Comparative Assessment of the Phytochemical Composition and Biological Activity of Extracts of Flowering Plants of Centaurea cyanus L., Centaurea jacea L. and Centaurea scabiosa L. Plants 2021, 10, 1279. [Google Scholar] [CrossRef]
- Ahmad, S.; Zeb, A. Phytochemical Profile and Pharmacological Properties of Trifolium Repens. J. Basic Clin. Physiol. Pharmacol. 2020, 32, 20200015. [Google Scholar] [CrossRef]
- Georges, P.; Legault, J.; Lavoie, S.; Grenon, C.; Pichette, A. Diterpenoids from the Buds of Pinus banksiana Lamb. Molecules 2012, 17, 9716–9727. [Google Scholar] [CrossRef]
- Seca, A.M.L.; Silva, A.M.S.; Bazzocchi, I.L.; Jimenez, I.A. Diterpene constituents of leaves from Juniperus brevifolia. Phytochemistry 2008, 69, 498–505. [Google Scholar] [CrossRef]
- Bertelli, D.; Papotti, G.; Bortolotti, L.; Marcazzan, G.L.; Plessi, M. 1H-NMR Simultaneous Identification of Health-Relevant Compounds in Propolis Extracts: NMR Identification of Health-Relevant Compounds in Propolis. Phytochem. Anal. 2012, 23, 260–266. [Google Scholar] [CrossRef]
- Popova, M.P.; Chinou, I.B.; Marekov, I.N.; Bankova, V.S. Terpenes with Antimicrobial Activity from Cretan Propolis. Phytochemistry 2009, 70, 1262–1271. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Okuyama, T.; Ishii, T.; Okumura, T.; Ikeya, Y.; Nishizawa, M. Sakuranetin downregulates inducible nitric oxide synthase expression by affecting interleukin-1 receptor and CCAAT/enhancer-binding protein β. J. Nat. Med. 2019, 73, 353–368. [Google Scholar] [CrossRef]
- Patel, N.K.; Jaiswal, G.; Bhutani, K.K. A review on biological sources, chemistry and pharmacological activities of pinostrobin. Nat. Prod. Res. 2016, 30, 2017–2027. [Google Scholar] [CrossRef]
- Cheng, J.; Fu, S.; Qin, Z.; Han, Y.; Yang, X. Self-assembled natural small molecule diterpene acids with favorable anticancer activity and biosafety for synergistically enhanced antitumor chemotherapy. J. Mater. Chem. B 2021, 9, 2674–2687. [Google Scholar] [CrossRef] [PubMed]
- Muto, N.; Tomokuni, T.; Haramoto, M.; Tatemoto, H.; Nakanishi, T.; Inatomi, Y.; Murata, H.; Inada, A. Isolation of Apoptosis- and Differentiation-Inducing Substances toward Human Promyelocytic Leukemia HL-60 Cells from Leaves of Juniperus taxifolia. Biosci. Biotechnol. Biochem. 2008, 72, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Silva-Carvalho, R.; Baltazar, F.; Almeida-Aguiar, C. Propolis: A Complex Natural Product with a Plethora of Biological Activities That Can Be Explored for Drug Development. Evid. Based Complement. Alternat. Med. 2015, 2015, 206439. [Google Scholar] [CrossRef]
- Urbanowicz, C.; Muñiz, P.A.; McArt, S.H. Honey bees and wild pollinators differ in their preference for and use of introduced floral resources. Ecol. Evol. 2020, 10, 6741–6751. [Google Scholar] [CrossRef]
- Tsigouri, A.; Passaloglou-Katrali, M.; Sabatakou, O. Palynological characteristics of different unifloral honeys from Greece. Grana 2004, 43, 122–128. [Google Scholar] [CrossRef]
- Panagiotopoulos, K.; Aufgebauer, A.; Schäbitz, F.; Wagner, B. Vegetation and climate history of the Lake Prespa region since the Lateglacial. Quat. Int. 2013, 293, 157–169. [Google Scholar] [CrossRef]
- Maggi, M.; Tourn, E.; Negri, P.; Szawarski, N.; Marconi, A.; Gallez, L.; Medici, S.; Ruffinengo, S.; Brasesco, C.; De Feudis, L.; et al. A New Formulation of Oxalic Acid for Varroa Destructor Control Applied in Apis Mellifera Colonies in the Presence of Brood. Apidologie 2016, 47, 596–605. [Google Scholar] [CrossRef]
- Ricciardelli D’ Albore, G. Textbook of Melissopalynology; Apimondia: Bucharest, Romania, 1997. [Google Scholar]
- Von Der Ohe, W.; Persano Oddo, L.; Piana, M.L.; Morlot, M.; Martin, P. Harmonized methods of melissopalynology. Apidologie 2004, 35, 18–23. [Google Scholar] [CrossRef]
- Stavropoulou, M.-I.; Stathopoulou, K.; Cheilari, A.; Benaki, D.; Gardikis, K.; Chinou, I.; Aligiannis, N. NMR Metabolic Profiling of Greek Propolis Samples: Comparative Evaluation of Their Phytochemical Compositions and Investigation of Their Anti-Ageing and Antioxidant Properties. J. Pharm. Biomed. Anal. 2021, 194, 113814. [Google Scholar] [CrossRef]
| RT (min) | Compound | PP1 | PP2 | PP3 | PP4 | PP5 |
|---|---|---|---|---|---|---|
| 16.03 | succinic acid | 1.77 | 0.54 | |||
| 20.13 | decanoic acid/capric acid | 0.46 | ||||
| 21.30 | malic acid | 0.82 | 0.22 | |||
| 22.04 | unidentified sugar | 1.16 | ||||
| 22.09 | cinnamic acid | 0.93 | 0.86 | 1.17 | ||
| 24.04 | unidentified sugar | 1.08 | 0.33 | |||
| 24.14 | p-hydroxybenzoic acid | 0.26 | 0.24 | |||
| 24.33 | unidentified sugar | 1.07 | 0.17 | |||
| 24.60 | dodecanoic acid/lauric acid | 0.48 | ||||
| 24.00–26.00 | unidentified sugars | 2.01 | 2.98 | 0.21 | ||
| 26.35 | pentanedioic acid | 0.37 | 0.38 | |||
| 27.65 | p-coumaric acid | 0.52 | ||||
| 27.90 | nonanedioic acid/azelaic acid | 1.13 | ||||
| 27.50–30.50 | unidentified sugars | 46.8 | 79.91 | 69.75 | 69.88 | 65.10 |
| 29.86 | vanillylpropionic acid | 0.38 | ||||
| 30.1–30.6 | unidentified sugars | 6.97 | ||||
| 30.60 | 4-hydroxycinnamic acid/p-coumaric acid | 3.26 | 0.61 | 0.91 | 0.85 | |
| 31.06–32.24 | unidentified sugars | 8.86 | 2.90 | 1.05 | 12.93 | |
| 31.50 | epi-manoyloxide | 0.43 | ||||
| 32.09 | palmitoleic acid | 0.98 | ||||
| 32.20 | 3,4-dimethoxy cinnamic acid | 1.29 | ||||
| 32.25 | unidentified sugar | 3.61 | ||||
| 32.49 | 3-hydroxymyristic acid | 0.37 | ||||
| 32.51 | gluconic acid | 1.63 | 0.41 | |||
| 32.55 | unidentified sugar | 0.37 | 0.32 | |||
| 32.62 | palmitic acid | 0.75 | 0.98 | 2.12 | 2.74 | 0.80 |
| 32.8 | unidentified sugar | 0.27 | ||||
| 33.30 | ferulic acid | 2.86 | 0.68 | 0.53 | 0.68 | |
| 33.57 | isoferulic acid | 2.70 | ||||
| 34.17 | unidentified sugar | 0.38 | 0.23 | |||
| 34.48 | caffeic acid | 3.53 | 1.15 | 0.69 | 1.06 | |
| 35.40 | pentenyl p-coumarate | 0.25 | ||||
| 35.47 | unidentified sugar | 0.29 | ||||
| 35.50 | linoleic acid | 0.78 | <0.4 | |||
| 35.62 | oleic acid | 1.30 | 0.68 | 2.36 | 1.66 | 0.48 |
| 35.81 | unidentified sugar | 0.92 | 0.40 | |||
| 35.90 | diterpenic acid | <0.09 | ||||
| 36.24 | ferruginol | 0.41 | ||||
| 37.01 | pimaric acid | 1.41 | ||||
| 37.11 | abietic acid | 0.36 | ||||
| 37.18 | pentenyl ferulate | 0.30 | ||||
| 37.43 | unidentified sugar | 0.28 | ||||
| 37.35 | isopimaric acid | 2.08 | ||||
| 37.45 | communic acid | 1.85 | ||||
| 37.55 | totarol | 0.55 | ||||
| 37.93 | pentenyl ester of isoferulic acid | 0.31 | ||||
| 38.10 | 3-methyl-3-butenyl ester of caffeic acid | 3.23 | 1.06 | 1.39 | 0.57 | |
| 38.24 | 3-methyl-6-butenyl of ferulic acid | 0.24 | ||||
| 38.33 | pinostrobin | 0.34 | ||||
| 38.36 | dehydroabietic acid | 0.43 | ||||
| 38.75 | 2-methyl-2-butenyl ester of caffeic acid | 0.91 | 0.33 | <0.4 | <0.2 | |
| 38.97 | 3-methyl-2-butenyl ester of caffeic acid | 2.95 | 1.02 | 0.76 | 0.63 | |
| 39.12 | isoagatholal | <0.09 | ||||
| 39.45 | caffeic acid ester | 0.27 | ||||
| 40.16 | pinostrobin chalcone | 0.28 | 0.99 | |||
| 40.71 | pinocembrin chalcone | 0.95 | 0.25 | |||
| 40.82 | pinocembrin | 1.06 | ||||
| 40.96 | agathadiol + imbricataloic acid | 2.37 | ||||
| 41.33 | pinobanksin chalcone | 0.46 | ||||
| 41.58 | isocupressic acid | 0.42 | ||||
| 41.75 | pinobanksin | 0.80 | ||||
| 42.06 | 3-acetyl alpinon | 1.09 | ||||
| 42.64 | sakuranetin | 0.85 | ||||
| 42.92 | pinobanksin 3-o-acetate | 0.50 | ||||
| 43.38 | benzyl ester of caffeic acid | 0.35 | 1.02 | 3.37 | 0.07 | |
| 43.71 | chrysin | 0.52 | 1.96 | 0.60 | ||
| 44.03 | galangin | 0.68 | ||||
| 44.53 | phenylethyl ester of caffeic acid | 0.39 | 0.37 | 0.73 | 0.32 | |
| 47.64 | cinnamyl ester of caffeic acid | 0.30 | 0.23 | 0.53 | ||
| 51.02 | triterpene | 1.58 |
| PP1 | PP2 | PP3 | PP4 | PP5 | |
|---|---|---|---|---|---|
| Aliphatic acids (%) | 6.14 | 1.66 | 9.18 | 4.40 | 3.21 |
| Phenolic acids (%) | 13.54 | 2.96 | - | 4.28 | 4.00 |
| Phenolic acid esters (%) | 9.26 | 4.03 | - | 6.25 | 2.36 |
| Diterpens (%) | - | - | 10.31 | - | - |
| Triterpens (%) | - | - | - | 1.58 | - |
| Flavonoids and chalcones (%) | 5.25 | 1.33 | - | 3.90 | 0.85 |
| Unidentified sugars (%) | 60.70 | 86.75 | 76.12 | 70.93 | 85.73 |
| Other unknown compounds (%) | 4.16 | 2.96 | 3.27 | 8.67 | 1.60 |
| PP1 | PP2 | PP3 | PP4 | PP5 | |
|---|---|---|---|---|---|
| Secondary pollen (16–45%) | Centaurea sp. Asteraceae 27% Liliaceae 25% Pyrus/Prunus Rosaceae 24% | Trifolium sp. Fabaceae 25% Pyrus/Prunus Rosaceae 25% Centaurea sp. Asteraceae, 16% | Liliaceae 26% Trifolium sp. Fabaceae 24% Boraginaceae 22% | Liliaceae 28% Trifolium sp. Fabaceae 25% | Asteraceae 28% Trifolium sp. Fabaceae 27% |
| Important minor pollen (3–15%) | Asteraceae, 8% Apiaceae 5% | Asteraceae 14% Apiaceae 11% | Centaurea sp. Asteraceae 15% Centaurea cyanus Asteraceae 8% | Pyrus/Prunus Rosaceae 13% Asteraceae 12% Rosaceae 9% Centaurea cyanus Asteraceae 7% | Pyrus/Prunus Rosaceae 14% Rosaceae 14% Liliaceae 12% |
| Minor pollen (<3%) | Brassica sp. Brassicaceae 2% Trifolium sp. Fabaceae, 2% Boraginaceae, 2% Vicia sp. Fabaceae, 2% Castanea sativa Fagaceae 1% | Castanea sativa Fagaceae 2% Liliaceae 2% Ononis sp. Fabaceae 2% Brassica sp. Brassicaceae 1% | Asteraceae 2% Apiaceae 1% Pyrus/Prunus Rosaceae 1% | Centaurea sp. Asteraceae 2% Boraginaceae 2% | Apiaceae 3% |
| Pollen from nectarless plants/Isolated pollen | Quercus sp. Fagaceae, Chenopodium sp. Chenopodiaceae, Scabiosa sp. Dipsacaceae, Pinus sp. Pinaceae, Poaceae, Euphorbia sp. Euphorbiaceae, Campanula sp. Campanulaceae, Oxalis sp. Oxalidaceae Cistaceae Caryophyllaceae Geranium sp. Geraniaceae, Polygonum aviculare Polygonaceae Populus sp. Salicaceae | Salix sp. Salicaceae Quercus sp. Fagaceae, Cistaceae Campanula sp. Campanulaceae, Vicia sp. Fabaceae, Pinus sp. Pinaceae, Chenopodium sp. Chenopodiaceae, Scabiosa sp. Dipsacaceae, Geranium sp. Geraniaceae, Lamiaceae, Caryophyllaceae Carex sp. Cyperaceae Cupressaceae Poaceae, Cistaceae Tribulus sp. Zygophyllaceae Ephedra sp. Ephedraceae, | Cupressaceae Ephedra sp. Ephedraceae, Smilax sp. Smilacaceae Salix sp. Salicaceae, Quercus sp. Fagaceae, Papaver rhoeas Papaveraceae Campanula sp. Campanulaceae, Vicia sp. Fabaceae, Caryophyllaceae Cistaceae Buxus semprervirens Buxaceae Plantaginaceae, Fraxinus sp. Oleaceae, Onobrychis sp. Fabaceae Populus sp. Salicaceae, Rosaceae Poaceae, Brassica sp. Brassicaceae Euphorbia sp. Euphorbiaceae | Quercus sp. Fagaceae, Cistaceae Campanula sp. Campanulaceae, Pinus sp. Pinaceae, Poaceae, Cupressaceae Brassica sp. Brassicaceae Chenopodium sp. Chenopodiaceae, Malva sp. Malvaceae Geranium sp. Geraniaceae, Lamiaceae Verbascum sp. Scrophulariaceae Loranthus europaeus Loranthaceae, Euphorbia sp. Euphorbiaceae | Quercus sp. Fagaceae, Pinus sp. Pinaceae, Populus sp. Salicaceae Cupressaceae Fraxinus sp. Oleaceae, Campanula sp. Campanulaceae, Euphorbia sp. Euphorbiaceae Poaceae, Papaver rhoeas Papaveraceae, Scrophulariaceae Acer sp. Aceraceae Artemisia sp. Asteraceae, Rhamnus sp. Rhamnaceae |
| Samples | S. aureus | S. epidermidis | P. aeruginosa | K. pneumoniae | E. cloacae | E. coli | S. mutans | S. viridans | C. albicans | C. tropicalis | C. glabrata |
|---|---|---|---|---|---|---|---|---|---|---|---|
| PP1 | 15/0.96 | 15/0.88 | 12/1.87 | 13/1.52 | 12/1.36 | 12/1.42 | 13/1.23 | 13/1.25 | 10/2.38 | 12/1.53 | 12/1.30 |
| PP2 | 13/1.27 | 14/1.15 | 12/1.80 | 12/1.79 | 12/1.28 | 12/1.35 | 12/1.54 | 12/1.50 | 10/2.17 | 12/1.40 | 12/1.27 |
| PP3 | 15/0.85 | 17/0.56 | 13/1.00 | 13/1.12 | 13/0.98 | 14/0.85 | 15/0.77 | 14/0.90 | 11/1.20 | 12/1.13 | 12/1.15 |
| PP4 | 12/1.55 | 13/1.30 | 12/1.77 | 12/1.65 | 12/1.27 | 12/1.45 | 12/1.25 | 12/1.3 | 10/2.24 | 11/1.32 | 12/1.25 |
| PP5 | 13/1.78 | 13/1.85 | 12/1.95 | 12/1.70 | 12/1.60 | 11/1.90 | 12/1.48 | 12/1.59 | 10/2.40 | 11/1.41 | 11/1.37 |
| Samples | TPC mg/g Extract | DPPH (% Inhibition) | ||
|---|---|---|---|---|
| 200 μg/mL | 100 μg/mL | 50 μg/mL | ||
| PP1 | 203.77 ± 1.04 | 90.87 ± 0.23 | 83.25 ± 1.14 | 44.72 ± 0.46 |
| PP2 | 192.23 ± 3.39 | 91.31 ± 0.22 | 77.83 ± 1.79 | 42.32 ± 3.50 |
| PP3 | 39.23 ± 0.36 | 16.69 ± 0.97 | 8.97 ± 0.44 | 3.23 ± 0.33 |
| PP4 | 153.82 ± 1.45 | 86.74 ± 0.08 | 47.03 ± 0.83 | 27.12 ± 0.50 |
| PP5 | 173.37 ± 1.79 | 90.94 ± 0.07 | 74.54 ± 1.83 | 39.17 ± 2.53 |
| Sample | Collection Area | Collection Season |
|---|---|---|
| PP1 | Agios Germanos | Summer 2019 |
| PP2 | Agios Germanos | Summer 2020 |
| PP3 | Vrondero | Summer 2020 |
| PP4 | Latsista | Summer 2020 |
| PP5 | Agios Achillios | Summer 2020 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pyrgioti, E.; Graikou, K.; Aligiannis, N.; Karabournioti, S.; Chinou, I. Qualitative Analysis Related to Palynological Characterization and Biological Evaluation of Propolis from Prespa National Park (Greece). Molecules 2022, 27, 7018. https://doi.org/10.3390/molecules27207018
Pyrgioti E, Graikou K, Aligiannis N, Karabournioti S, Chinou I. Qualitative Analysis Related to Palynological Characterization and Biological Evaluation of Propolis from Prespa National Park (Greece). Molecules. 2022; 27(20):7018. https://doi.org/10.3390/molecules27207018
Chicago/Turabian StylePyrgioti, Elisavet, Konstantia Graikou, Nektarios Aligiannis, Sofia Karabournioti, and Ioanna Chinou. 2022. "Qualitative Analysis Related to Palynological Characterization and Biological Evaluation of Propolis from Prespa National Park (Greece)" Molecules 27, no. 20: 7018. https://doi.org/10.3390/molecules27207018
APA StylePyrgioti, E., Graikou, K., Aligiannis, N., Karabournioti, S., & Chinou, I. (2022). Qualitative Analysis Related to Palynological Characterization and Biological Evaluation of Propolis from Prespa National Park (Greece). Molecules, 27(20), 7018. https://doi.org/10.3390/molecules27207018







