Essential Roles of Peroxiredoxin IV in Inflammation and Cancer
Abstract
1. Introduction
2. Prx4 Structure and Function
2.1. Structure
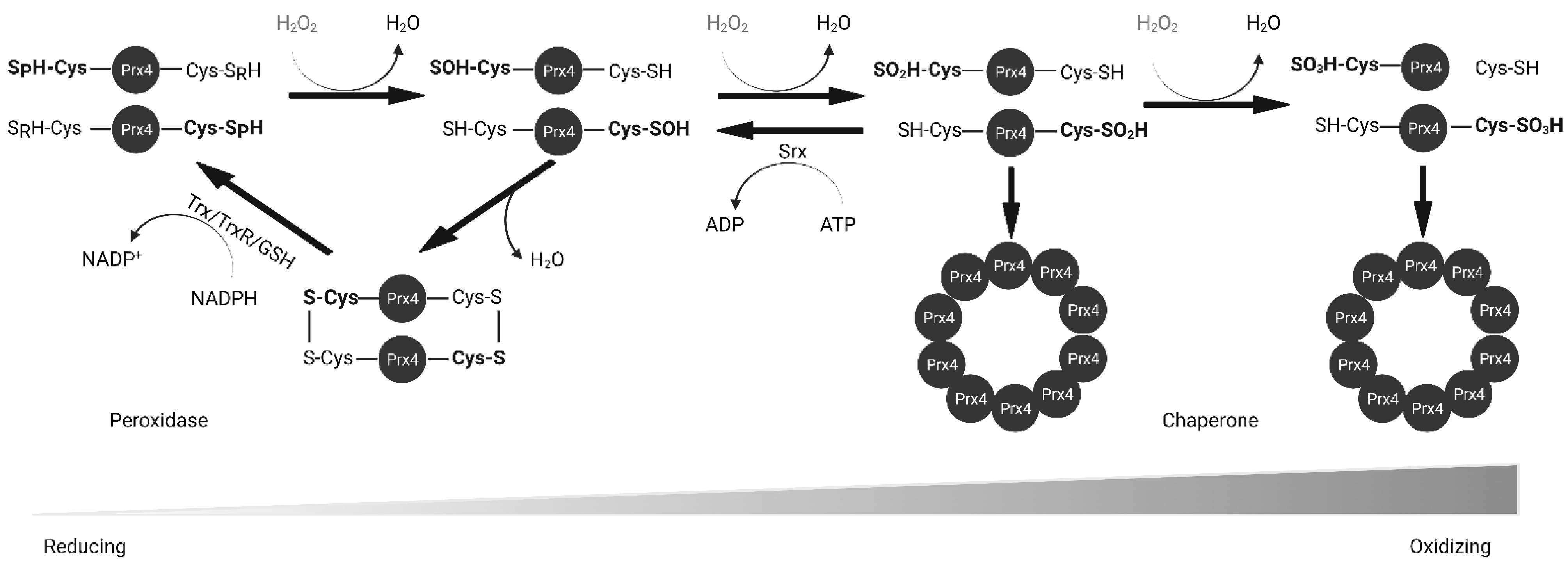
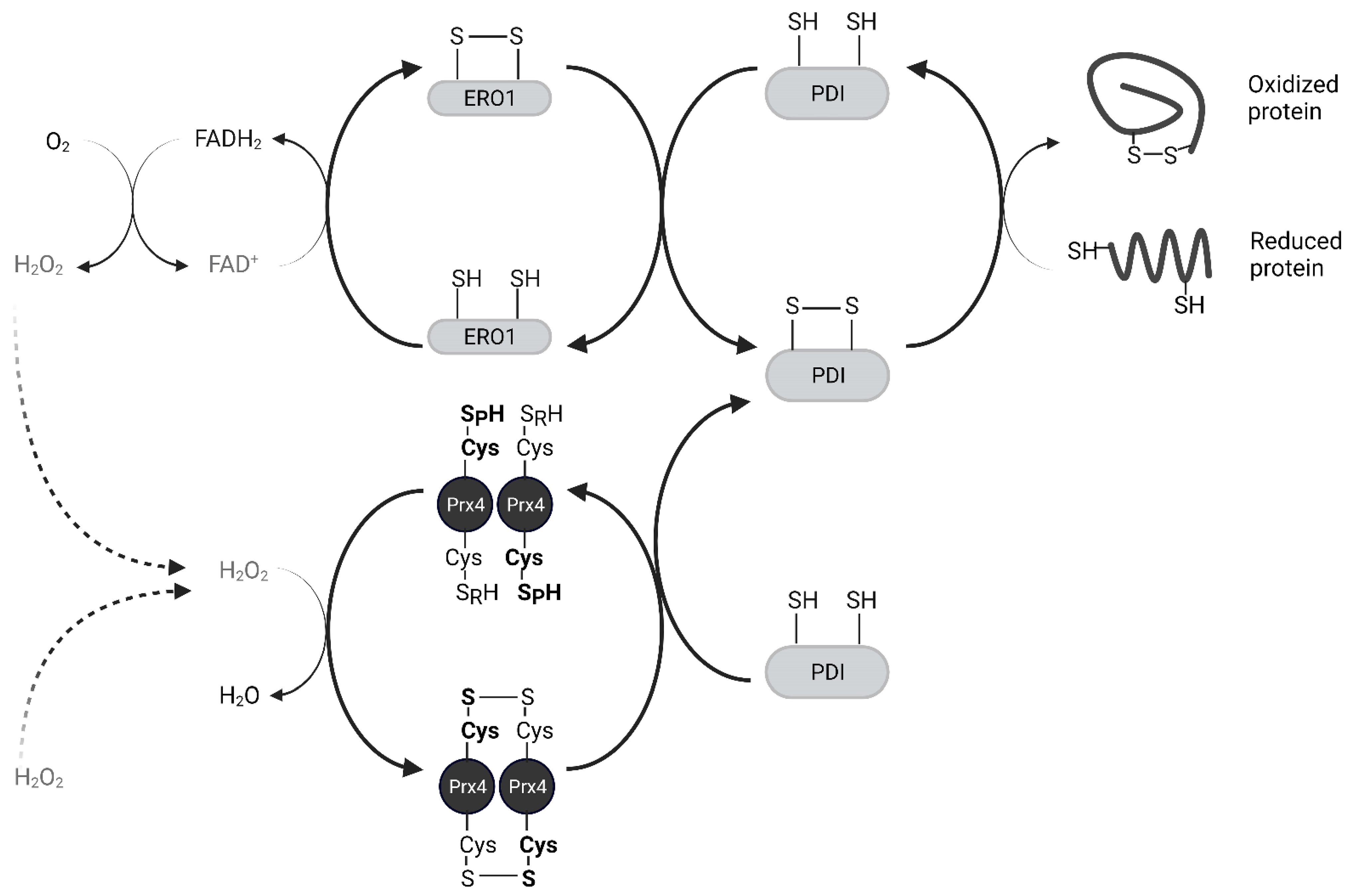
2.2. Biochemical Function
3. Prx4 and Inflammation
3.1. Roles of Prx4 in Inflammatory Diseases
3.1.1. Diabetes and Nonalcoholic Fatty Liver Disease (NAFLD)
3.1.2. Cardiovascular Diseases
3.1.3. Cerebral Ischemia and Alzheimer’s Disease
3.1.4. Colitis
3.1.5. Rheumatism and Other Inflammatory Conditions
3.2. Signaling Pathways Regulated by Prx4 in Inflammation
3.2.1. NF-κB
3.2.2. Inflammasome
3.2.3. Others
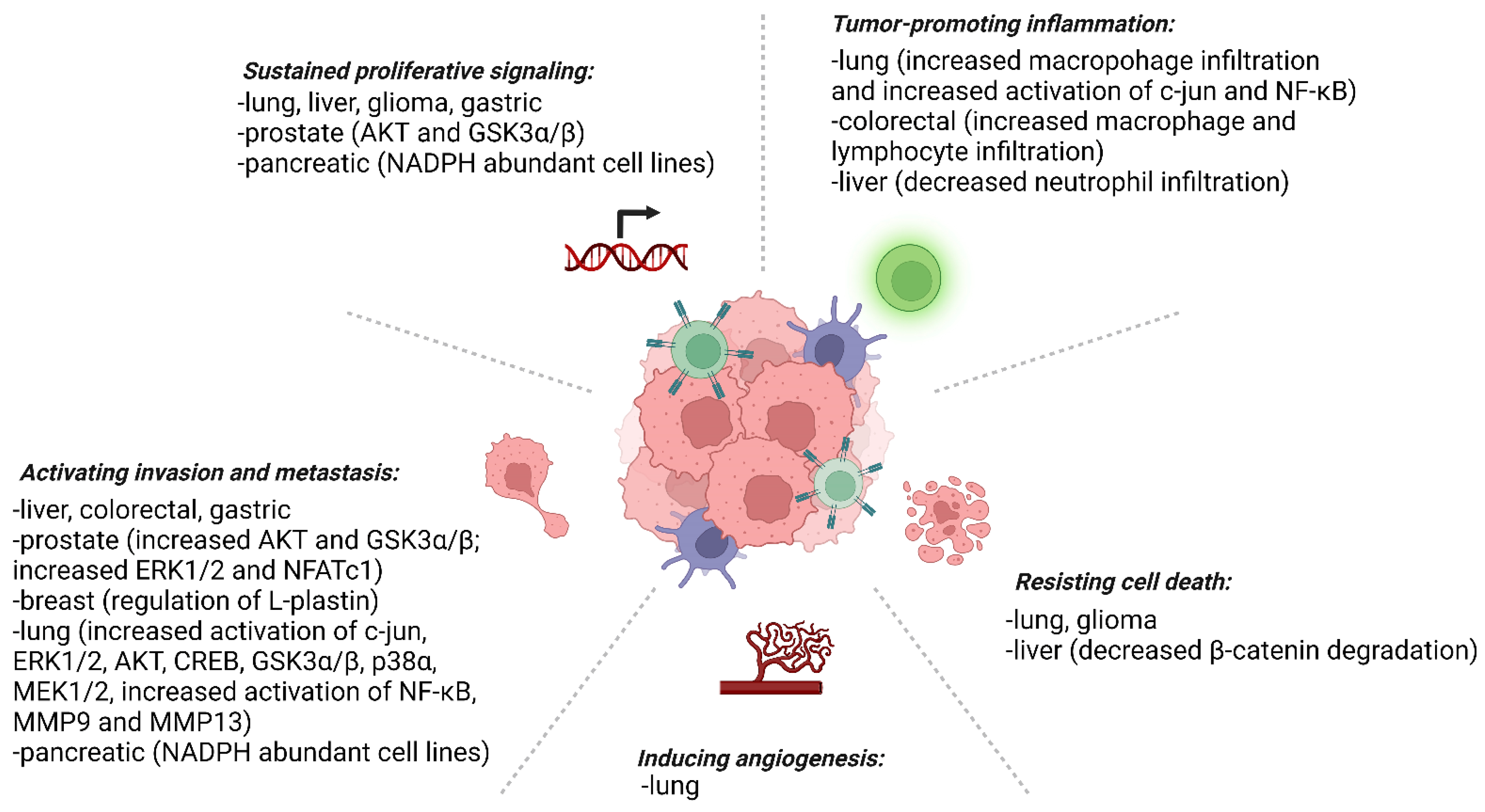
4. Prx4 and Cancer
4.1. Prostate Cancer
4.2. Breast Cancer
4.3. Lung Cancer
4.4. Colorectal Cancer
4.5. Esophageal Carcinoma and Gastric Cancer
4.6. Liver Cancer
4.7. Glioma
4.8. Melanoma of Skin
4.9. Non-Hodgkin Lymphoma and Leukemia
4.10. Oral Squamous Cell Carcinoma (OSCC)
4.11. Pancreatic Cancer
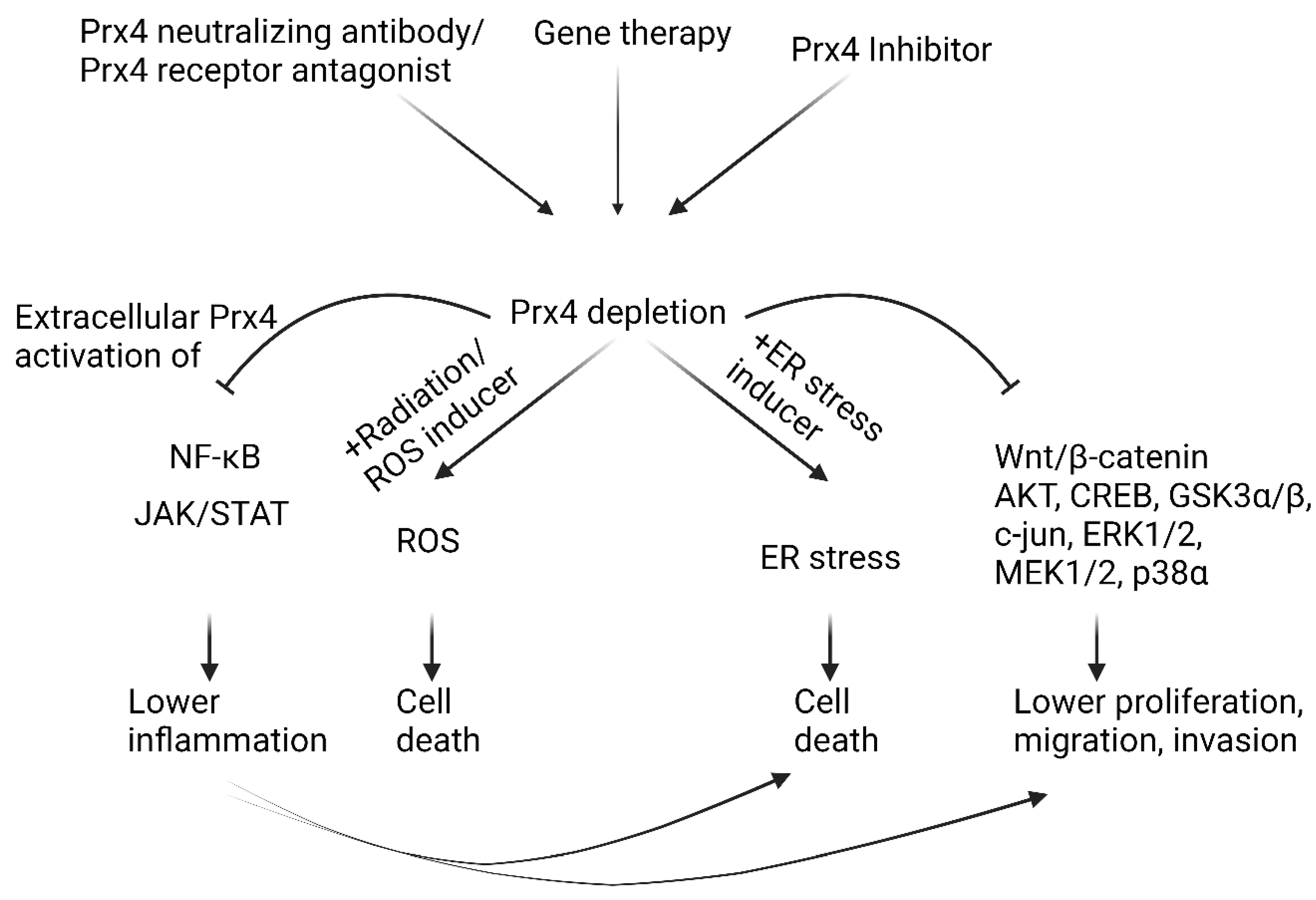
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | alzheimer’s disease |
| APL | acute promyelocytic leukemia |
| APP | amyloid precursor protein |
| COX-2 | Cyclooxygenase-2 |
| Cp | peroxidatic cysteine |
| Cr | resolving cysteine |
| CRC | colorectal cancer |
| Cys-SOH | sulfenic acid |
| DEN | diethylnitrosamone |
| DM | diabetes mellitus |
| DSS | dextran sulfate sodium |
| ELISA | enzyme-linked immunosorbent assay |
| ER | endoplasmic reticulum |
| Ero1 | ER oxidoreductin 1 |
| ERp44 | endoplasmic reticulum protein 44 |
| FAD | flavin adenine dinucleotide |
| GBM | glioblastoma multiforme |
| GPx | glutathione peroxidase |
| GSH | glutathione |
| H2O2 | hydrogen peroxide |
| HCC | hepatocellular carcinoma |
| HGG | high grade-glioma |
| JAK | janus kinase |
| LUAD | lung adenocarcinoma |
| MDP | microbial product muramyl dipeptide |
| MSCs | mesenchymal stromal cells |
| NAC | N-acetyl cysteine |
| NAFLD | nonalcoholic fatty liver disease |
| NF-κB | nuclear factor kappa b |
| NSAIDs | non-steroidal anti-inflammatory drugs |
| NSCLC | non-small cell lung cancer |
| OGD | oxygen glucose deprivation |
| OSCC | oral Squamous Cell Carcinoma |
| PDI | protein disulfide isomerase |
| Prx | peroxiredoxin |
| QSOX1 | quiescin sulfhydryl oxidase 1 |
| RA | rheumatoid arthritis |
| ROS | reactive oxygen species |
| SCC | squamous cell carcinoma |
| SNPs | single-nucleotide polymorphisms |
| SOD | superoxide dismutase |
| Srx | sulfiredoxin |
| STAT | signal transducer and activator of transcription |
| TBARS | thiobarbituric acid reactive substances |
| Trx | thioredoxin |
References
- Pizzorno, J. Glutathione! Integr. Med. 2014, 13, 8–12. [Google Scholar]
- Lu, J.; Holmgren, A. The thioredoxin antioxidant system. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Duan, D.; Osama, A.; Fang, J. Natural Molecules Targeting Thioredoxin System and Their Therapeutic Potential. Antioxid. Redox Signal. 2021, 34, 1083–1107. [Google Scholar] [CrossRef] [PubMed]
- Fridovich, I. Superoxide radical and superoxide dismutases. Annu. Rev. Biochem. 1995, 64, 97–112. [Google Scholar] [CrossRef]
- Glorieux, C.; Calderon, P.B. Catalase, a remarkable enzyme: Targeting the oldest antioxidant enzyme to find a new cancer treatment approach. Biol. Chem. 2017, 398, 1095–1108. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R.; Maiorino, M. Glutathione peroxidases. Biochim. Biophys. Acta 2013, 1830, 3289–3303. [Google Scholar] [CrossRef]
- Kim, K.; Kim, I.H.; Lee, K.Y.; Rhee, S.G.; Stadtman, E.R. The isolation and purification of a specific “protector” protein which inhibits enzyme inactivation by a thiol/Fe(III)/O2 mixed-function oxidation system. J. Biol. Chem. 1988, 263, 4704–4711. [Google Scholar] [CrossRef]
- Nogoceke, E.; Gommel, D.U.; Kiess, M.; Kalisz, H.M.; Flohé, L. A unique cascade of oxidoreductases catalyses trypanothione-mediated peroxide metabolism in Crithidia fasciculata. Biol. Chem. 1997, 378, 827–836. [Google Scholar] [CrossRef]
- Bryk, R.; Griffin, P.; Nathan, C. Peroxynitrite reductase activity of bacterial peroxiredoxins. Nature 2000, 407, 211–215. [Google Scholar] [CrossRef]
- Hillas, P.J.; del Alba, F.S.; Oyarzabal, J.; Wilks, A.; Ortiz De Montellano, P.R. The AhpC and AhpD antioxidant defense system of Mycobacterium tuberculosis. J. Biol. Chem. 2000, 275, 18801–18809. [Google Scholar] [CrossRef]
- Rhee, S.G.; Kang, S.W.; Chang, T.S.; Jeong, W.; Kim, K. Peroxiredoxin, a novel family of peroxidases. IUBMB Life 2001, 52, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.S.; Kang, S.W.; Kim, K.; Baines, I.C.; Lee, T.H.; Rhee, S.G. Identification of a new type of mammalian peroxiredoxin that forms an intramolecular disulfide as a reaction intermediate. J. Biol. Chem. 2000, 275, 20346–20354. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A.B. Peroxiredoxin 6: A bifunctional enzyme with glutathione peroxidase and phospholipase A₂ activities. Antioxid. Redox Signal. 2011, 15, 831–844. [Google Scholar] [CrossRef] [PubMed]
- NCBI Resource Coordinators. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2018, 46, D8–D13. [Google Scholar] [CrossRef]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J.; et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7, 539. [Google Scholar] [CrossRef]
- Goujon, M.; McWilliam, H.; Li, W.; Valentin, F.; Squizzato, S.; Paern, J.; Lopez, R. A new bioinformatics analysis tools framework at EMBL-EBI. Nucleic Acids Res. 2010, 38, W695–W699. [Google Scholar] [CrossRef]
- Madeira, F.; Park, Y.M.; Lee, J.; Buso, N.; Gur, T.; Madhusoodanan, N.; Basutkar, P.; Tivey, A.R.N.; Potter, S.C.; Finn, R.D.; et al. The EMBL-EBI search and sequence analysis tools APIs in 2019. Nucleic Acids Res. 2019, 47, W636–W641. [Google Scholar] [CrossRef]
- Fujii, J.; Ikeda, Y.; Kurahashi, T.; Homma, T. Physiological and pathological views of peroxiredoxin 4. Free Radic. Biol. Med. 2015, 83, 373–379. [Google Scholar] [CrossRef]
- Yim, S.H.; Kim, Y.J.; Oh, S.Y.; Fujii, J.; Zhang, Y.; Gladyshev, V.N.; Rhee, S.G. Identification and characterization of alternatively transcribed form of peroxiredoxin IV gene that is specifically expressed in spermatids of postpubertal mouse testis. J. Biol. Chem. 2011, 286, 39002–39012. [Google Scholar] [CrossRef]
- Matsumoto, A.; Okado, A.; Fujii, T.; Fujii, J.; Egashira, M.; Niikawa, N.; Taniguchi, N. Cloning of the peroxiredoxin gene family in rats and characterization of the fourth member. FEBS Lett. 1999, 443, 246–250. [Google Scholar] [CrossRef]
- Tavender, T.J.; Sheppard, A.M.; Bulleid, N.J. Peroxiredoxin IV is an endoplasmic reticulum-localized enzyme forming oligomeric complexes in human cells. Biochem. J. 2008, 411, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Haridas, V.; Ni, J.; Meager, A.; Su, J.; Yu, G.L.; Zhai, Y.; Kyaw, H.; Akama, K.T.; Hu, J.; Van Eldik, L.J.; et al. TRANK, a novel cytokine that activates NF-kappa B and c-Jun N-terminal kinase. J. Immunol. 1998, 161, 1–6. [Google Scholar] [PubMed]
- Okado-Matsumoto, A.; Matsumoto, A.; Fujii, J.; Taniguchi, N. Peroxiredoxin IV is a secretable protein with heparin-binding properties under reduced conditions. J. Biochem. 2000, 127, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Kakihana, T.; Araki, K.; Vavassori, S.; Iemura, S.; Cortini, M.; Fagioli, C.; Natsume, T.; Sitia, R.; Nagata, K. Dynamic regulation of Ero1α and peroxiredoxin 4 localization in the secretory pathway. J. Biol. Chem. 2013, 288, 29586–29594. [Google Scholar] [CrossRef] [PubMed]
- Tempio, T.; Orsi, A.; Sicari, D.; Valetti, C.; Yoboue, E.D.; Anelli, T.; Sitia, R. A virtuous cycle operated by ERp44 and ERGIC-53 guarantees proteostasis in the early secretory compartment. iScience 2021, 24, 102244. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, L.; Wang, X.; Sun, F.; Wang, C.C. Structural insights into the peroxidase activity and inactivation of human peroxiredoxin 4. Biochem. J. 2012, 441, 113–118. [Google Scholar] [CrossRef]
- Cao, Z.; Tavender, T.J.; Roszak, A.W.; Cogdell, R.J.; Bulleid, N.J. Crystal structure of reduced and of oxidized peroxiredoxin IV enzyme reveals a stable oxidized decamer and a non-disulfide-bonded intermediate in the catalytic cycle. J. Biol. Chem. 2011, 286, 42257–42266. [Google Scholar] [CrossRef]
- Zeida, A.; Reyes, A.M.; Lebrero, M.C.; Radi, R.; Trujillo, M.; Estrin, D.A. The extraordinary catalytic ability of peroxiredoxins: A combined experimental and QM/MM study on the fast thiol oxidation step. Chem. Commun. 2014, 50, 10070–10073. [Google Scholar] [CrossRef]
- Tavender, T.J.; Springate, J.J.; Bulleid, N.J. Recycling of peroxiredoxin IV provides a novel pathway for disulphide formation in the endoplasmic reticulum. EMBO J. 2010, 29, 4185–4197. [Google Scholar] [CrossRef]
- Rabilloud, T.; Heller, M.; Gasnier, F.; Luche, S.; Rey, C.; Aebersold, R.; Benahmed, M.; Louisot, P.; Lunardi, J. Proteomics analysis of cellular response to oxidative stress. Evidence for in vivo overoxidation of peroxiredoxins at their active site. J. Biol. Chem. 2002, 277, 19396–19401. [Google Scholar] [CrossRef]
- Roussel, X.; Béchade, G.; Kriznik, A.; Van Dorsselaer, A.; Sanglier-Cianferani, S.; Branlant, G.; Rahuel-Clermont, S. Evidence for the formation of a covalent thiosulfinate intermediate with peroxiredoxin in the catalytic mechanism of sulfiredoxin. J. Biol. Chem. 2008, 283, 22371–22382. [Google Scholar] [CrossRef]
- Mishra, M.; Jiang, H.; Wu, L.; Chawsheen, H.A.; Wei, Q. The sulfiredoxin-peroxiredoxin (Srx-Prx) axis in cell signal transduction and cancer development. Cancer Lett. 2015, 366, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Elko, E.A.; Manuel, A.M.; White, S.; Zito, E.; van der Vliet, A.; Anathy, V.; Janssen-Heininger, Y.M.W. Oxidation of peroxiredoxin-4 induces oligomerization and promotes interaction with proteins governing protein folding and endoplasmic reticulum stress. J. Biol. Chem. 2021, 296, 100665. [Google Scholar] [CrossRef] [PubMed]
- Bonnichsen, R.K.; Chance, B.; Theorell, H. Catalase Activity. Acta Chem. Scand. 1947, 1, 685–709. [Google Scholar] [CrossRef]
- Winterbourn, C.C. The biological chemistry of hydrogen peroxide. Methods Enzymol. 2013, 528, 3–25. [Google Scholar] [CrossRef]
- Winterbourn, C.C.; Metodiewa, D. Reactivity of biologically important thiol compounds with superoxide and hydrogen peroxide. Free Radic. Biol. Med. 1999, 27, 322–328. [Google Scholar] [CrossRef]
- Nguyen, V.D.; Saaranen, M.J.; Karala, A.R.; Lappi, A.K.; Wang, L.; Raykhel, I.B.; Alanen, H.I.; Salo, K.E.; Wang, C.C.; Ruddock, L.W. Two endoplasmic reticulum PDI peroxidases increase the efficiency of the use of peroxide during disulfide bond formation. J. Mol. Biol. 2011, 406, 503–515. [Google Scholar] [CrossRef]
- Hatahet, F.; Ruddock, L.W. Protein disulfide isomerase: A critical evaluation of its function in disulfide bond formation. Antioxid. Redox Signal. 2009, 11, 2807–2850. [Google Scholar] [CrossRef]
- Tu, B.P.; Weissman, J.S. The FAD- and O(2)-dependent reaction cycle of Ero1-mediated oxidative protein folding in the endoplasmic reticulum. Mol. Cell 2002, 10, 983–994. [Google Scholar] [CrossRef]
- Gross, E.; Sevier, C.S.; Heldman, N.; Vitu, E.; Bentzur, M.; Kaiser, C.A.; Thorpe, C.; Fass, D. Generating disulfides enzymatically: Reaction products and electron acceptors of the endoplasmic reticulum thiol oxidase Ero1p. Proc. Natl. Acad. Sci. USA 2006, 103, 299–304. [Google Scholar] [CrossRef]
- Sato, Y.; Kojima, R.; Okumura, M.; Hagiwara, M.; Masui, S.; Maegawa, K.; Saiki, M.; Horibe, T.; Suzuki, M.; Inaba, K. Synergistic cooperation of PDI family members in peroxiredoxin 4-driven oxidative protein folding. Sci. Rep. 2013, 3, 2456. [Google Scholar] [CrossRef] [PubMed]
- Zito, E.; Hansen, H.G.; Yeo, G.S.; Fujii, J.; Ron, D. Endoplasmic reticulum thiol oxidase deficiency leads to ascorbic acid depletion and noncanonical scurvy in mice. Mol. Cell 2012, 48, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.C.; Kim, G.M.; Kim, E.K.; Lee, H.N.; Ha, B.; Lee, S.Y.; Jang, H.H. Reversal of 2-Cys peroxiredoxin oligomerization by sulfiredoxin. Biochem. Biophys. Res. Commun. 2013, 432, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Pattwell, D.; Ashton, T.; McArdle, A.; Griffiths, R.D.; Jackson, M.J. Ischemia and reperfusion of skeletal muscle lead to the appearance of a stable lipid free radical in the circulation. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H2400–H2404. [Google Scholar] [CrossRef][Green Version]
- Pattwell, D.M.; McArdle, A.; Morgan, J.E.; Patridge, T.A.; Jackson, M.J. Release of reactive oxygen and nitrogen species from contracting skeletal muscle cells. Free Radic. Biol. Med. 2004, 37, 1064–1072. [Google Scholar] [CrossRef]
- Salzano, S.; Checconi, P.; Hanschmann, E.M.; Lillig, C.H.; Bowler, L.D.; Chan, P.; Vaudry, D.; Mengozzi, M.; Coppo, L.; Sacre, S.; et al. Linkage of inflammation and oxidative stress via release of glutathionylated peroxiredoxin-2, which acts as a danger signal. Proc. Natl. Acad. Sci. USA 2014, 111, 12157–12162. [Google Scholar] [CrossRef]
- Manabe, Y.; Takagi, M.; Nakamura-Yamada, M.; Goto-Inoue, N.; Taoka, M.; Isobe, T.; Fujii, N.L. Redox proteins are constitutively secreted by skeletal muscle. J. Physiol. Sci. 2014, 64, 401–409. [Google Scholar] [CrossRef]
- Wadley, A.J.; Keane, G.; Cullen, T.; James, L.; Vautrinot, J.; Davies, M.; Hussey, B.; Hunter, D.J.; Mastana, S.; Holliday, A.; et al. Characterization of extracellular redox enzyme concentrations in response to exercise in humans. J. Appl. Physiol. 2019, 127, 858–866. [Google Scholar] [CrossRef]
- Yamada, S.; Guo, X. Peroxiredoxin 4 (PRDX4): Its critical in vivo roles in animal models of metabolic syndrome ranging from atherosclerosis to nonalcoholic fatty liver disease. Pathol. Int. 2018, 68, 91–101. [Google Scholar] [CrossRef]
- Gateva, A.; Assyov, Y.; Velikova, T.; Kamenov, Z. Increased peroxiredoxin 4 levels in patients with prediabetes compared to normal glucose tolerance subjects. Clin. Endocrinol. 2016, 85, 551–555. [Google Scholar] [CrossRef]
- Ding, Y.; Yamada, S.; Wang, K.Y.; Shimajiri, S.; Guo, X.; Tanimoto, A.; Murata, Y.; Kitajima, S.; Watanabe, T.; Izumi, H.; et al. Overexpression of peroxiredoxin 4 protects against high-dose streptozotocin-induced diabetes by suppressing oxidative stress and cytokines in transgenic mice. Antioxid. Redox Signal. 2010, 13, 1477–1490. [Google Scholar] [CrossRef]
- Tran, D.T.; Pottekat, A.; Mir, S.A.; Loguercio, S.; Jang, I.; Campos, A.R.; Scully, K.M.; Lahmy, R.; Liu, M.; Arvan, P.; et al. Unbiased Profiling of the Human Proinsulin Biosynthetic Interaction Network Reveals a Role for Peroxiredoxin 4 in Proinsulin Folding. Diabetes 2020, 69, 1723–1734. [Google Scholar] [CrossRef] [PubMed]
- Homma, T.; Kurahashi, T.; Lee, J.; Nabeshima, A.; Yamada, S.; Fujii, J. Double Knockout of Peroxiredoxin 4 (Prdx4) and Superoxide Dismutase 1 (Sod1) in Mice Results in Severe Liver Failure. Oxidative Med. Cell. Longev. 2018, 2018, 2812904. [Google Scholar] [CrossRef] [PubMed]
- Luo, P.; Liu, D.; Zhang, Q.; Yang, F.; Wong, Y.K.; Xia, F.; Zhang, J.; Chen, J.; Tian, Y.; Yang, C.; et al. Celastrol induces ferroptosis in activated HSCs to ameliorate hepatic fibrosis via targeting peroxiredoxins and HO-1. Acta Pharm. Sin. B 2022, 12, 2300–2314. [Google Scholar] [CrossRef] [PubMed]
- Caillard, A.; Sadoune, M.; Cescau, A.; Meddour, M.; Gandon, M.; Polidano, E.; Delcayre, C.; Da Silva, K.; Manivet, P.; Gomez, A.M.; et al. QSOX1, a novel actor of cardiac protection upon acute stress in mice. J. Mol. Cell. Cardiol. 2018, 119, 75–86. [Google Scholar] [CrossRef]
- Ibarrola, J.; Arrieta, V.; Sadaba, R.; Martinez-Martinez, E.; Garcia-Pena, A.; Alvarez, V.; Fernandez-Celis, A.; Gainza, A.; Santamaria, E.; Fernandez-Irigoyen, J.; et al. Galectin-3 down-regulates antioxidant peroxiredoxin-4 in human cardiac fibroblasts: A new pathway to induce cardiac damage. Clin. Sci. 2018, 132, 1471–1485. [Google Scholar] [CrossRef]
- Tian, Y.; Lv, W.; Lu, C.; Jiang, Y.; Yang, X.; Song, M. Galectin-3 inhibition attenuates doxorubicin-induced cardiac dysfunction by upregulating the expression of peroxiredoxin-4. Can. J. Physiol. Pharmacol. 2020, 98, 700–707. [Google Scholar] [CrossRef]
- Rowe, D.D.; Leonardo, C.C.; Hall, A.A.; Shahaduzzaman, M.D.; Collier, L.A.; Willing, A.E.; Pennypacker, K.R. Cord blood administration induces oligodendrocyte survival through alterations in gene expression. Brain Res. 2010, 1366, 172–188. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, J.; Cai, J.; Qiu, Y.; Zheng, H.; Lai, X.; Sui, X.; Wang, Y.; Lu, Q.; Zhang, Y.; et al. Targeted homing of CCR2-overexpressing mesenchymal stromal cells to ischemic brain enhances post-stroke recovery partially through PRDX4-mediated blood-brain barrier preservation. Theranostics 2018, 8, 5929–5944. [Google Scholar] [CrossRef]
- Goemaere, J.; Knoops, B. Peroxiredoxin distribution in the mouse brain with emphasis on neuronal populations affected in neurodegenerative disorders. J. Comp. Neurol. 2012, 520, 258–280. [Google Scholar] [CrossRef]
- Majd, S.; Power, J.H.T. Oxidative Stress and Decreased Mitochondrial Superoxide Dismutase 2 and Peroxiredoxins 1 and 4 Based Mechanism of Concurrent Activation of AMPK and mTOR in Alzheimer’s Disease. Curr. Alzheimer Res. 2018, 15, 764–776. [Google Scholar] [CrossRef] [PubMed]
- Kam, M.K.; Lee, D.G.; Kim, B.; Lee, H.S.; Lee, S.R.; Bae, Y.C.; Lee, D.S. Peroxiredoxin 4 ameliorates amyloid beta oligomer-mediated apoptosis by inhibiting ER-stress in HT-22 hippocampal neuron cells. Cell Biol. Toxicol. 2019, 35, 573–588. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Kim, M.H.; Lee, H.J.; Huh, J.W.; Lee, H.S.; Lee, D.S. Peroxiredoxin 4 attenuates glutamate-induced neuronal cell death through inhibition of endoplasmic reticulum stress. Free Radic. Res. 2020, 54, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Schrotter, A.; Pfeiffer, K.; El Magraoui, F.; Platta, H.W.; Erdmann, R.; Meyer, H.E.; Egensperger, R.; Marcus, K.; Muller, T. The amyloid precursor protein (APP) family members are key players in S-adenosylmethionine formation by MAT2A and modify BACE1 and PSEN1 gene expression-relevance for Alzheimer’s disease. Mol. Cell. Proteom. 2012, 11, 1274–1288. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, N.A.; Andersen, V.; Møller, J.C.; Møller, H.S.; Jessen, F.; Purup, S.; Larsen, L.B. Comparative analysis of inflamed and non-inflamed colon biopsies reveals strong proteomic inflammation profile in patients with ulcerative colitis. BMC Gastroenterol. 2012, 12, 76. [Google Scholar] [CrossRef]
- Takagi, T.; Homma, T.; Fujii, J.; Shirasawa, N.; Yoriki, H.; Hotta, Y.; Higashimura, Y.; Mizushima, K.; Hirai, Y.; Katada, K.; et al. Elevated ER stress exacerbates dextran sulfate sodium-induced colitis in PRDX4-knockout mice. Free Radic. Biol. Med. 2019, 134, 153–164. [Google Scholar] [CrossRef]
- Horie, K.; Mikami, T.; Yoshida, T.; Sato, Y.; Okayasu, I. Peroxiredoxin 1 expression in active ulcerative colitis mucosa identified by proteome analysis and involvement of thioredoxin based on immunohistochemistry. Oncol. Lett. 2018, 15, 2364–2372. [Google Scholar] [CrossRef]
- Rao, Z.; Wang, S.; Wang, J. Peroxiredoxin 4 inhibits IL-1β-induced chondrocyte apoptosis via PI3K/AKT signaling. Biomed. Pharm. 2017, 90, 414–420. [Google Scholar] [CrossRef]
- Chang, X.; Cui, Y.; Zong, M.; Zhao, Y.; Yan, X.; Chen, Y.; Han, J. Identification of proteins with increased expression in rheumatoid arthritis synovial tissues. J. Rheumatol. 2009, 36, 872–880. [Google Scholar] [CrossRef]
- Aihaiti, Y.; Tuerhong, X.; Zheng, H.; Cai, Y.; Yang, M.; Xu, P. Peroxiredoxin 4 regulates tumor-cell-like characteristics of fibroblast-like synoviocytes in rheumatoid arthritis through PI3k/Akt signaling pathway. Clin. Immunol. 2022, 237, 108964. [Google Scholar] [CrossRef]
- Han, K.; Du, S.S.; Wang, H.; Qiao, J.J.; Zhang, X.; Wang, P.; Shen, F.H. Differential expression of PRDX4 in alveolar macrophages of patients with silicosis. Chin. J. Ind. Hyg. Occup. Dis. 2021, 39, 17–19. [Google Scholar] [CrossRef]
- Bertolotti, M.; Yim, S.H.; Garcia-Manteiga, J.M.; Masciarelli, S.; Kim, Y.J.; Kang, M.H.; Iuchi, Y.; Fujii, J.; Vene, R.; Rubartelli, A.; et al. B- to plasma-cell terminal differentiation entails oxidative stress and profound reshaping of the antioxidant responses. Antioxid. Redox Signal. 2010, 13, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, R.; Guo, X.; Zheng, J.; Zhang, J.; Han, J.; Shioya, A.; Uramoto, H.; Mochizuki, T.; Yamada, S. Peroxiredoxin 4 improved aging-related delayed wound healing in mice. J. Investig. Dermatol. 2021, 141, 2720–2729. [Google Scholar] [CrossRef]
- Greten, F.R.; Eckmann, L.; Greten, T.F.; Park, J.M.; Li, Z.W.; Egan, L.J.; Kagnoff, M.F.; Karin, M. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 2004, 118, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Popivanova, B.K.; Kitamura, K.; Wu, Y.; Kondo, T.; Kagaya, T.; Kaneko, S.; Oshima, M.; Fujii, C.; Mukaida, N. Blocking TNF-alpha in mice reduces colorectal carcinogenesis associated with chronic colitis. J. Clin. Investig. 2008, 118, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Dolcet, X.; Llobet, D.; Pallares, J.; Matias-Guiu, X. NF-kB in development and progression of human cancer. Virchows Arch. 2005, 446, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.Y.; Chae, H.Z.; Rhee, S.G.; Jeang, K.T. Regulatory role for a novel human thioredoxin peroxidase in NF-kappaB activation. J. Biol. Chem. 1997, 272, 30952–30961. [Google Scholar] [CrossRef]
- Weichart, D.; Gobom, J.; Klopfleisch, S.; Häsler, R.; Gustavsson, N.; Billmann, S.; Lehrach, H.; Seegert, D.; Schreiber, S.; Rosenstiel, P. Analysis of NOD2-mediated proteome response to muramyl dipeptide in HEK293 cells. J. Biol. Chem. 2006, 281, 2380–2389. [Google Scholar] [CrossRef]
- Yu, S.; Mu, Y.; Ao, J.; Chen, X. Peroxiredoxin IV regulates pro-inflammatory responses in large yellow croaker (Pseudosciaena crocea) and protects against bacterial challenge. J. Proteome Res. 2010, 9, 1424–1436. [Google Scholar] [CrossRef]
- Mu, Y.; Lian, F.M.; Teng, Y.B.; Ao, J.; Jiang, Y.L.; He, Y.X.; Chen, Y.; Zhou, C.Z.; Chen, X. The N-terminal beta-sheet of peroxiredoxin 4 in the large yellow croaker Pseudosciaena crocea is involved in its biological functions. PLoS ONE 2013, 8, e57061. [Google Scholar] [CrossRef]
- Radyuk, S.N.; Klichko, V.I.; Michalak, K.; Orr, W.C. The effect of peroxiredoxin 4 on fly physiology is a complex interplay of antioxidant and signaling functions. FASEB J. 2013, 27, 1426–1438. [Google Scholar] [CrossRef]
- Zhao, L.X.; Du, J.R.; Zhou, H.J.; Liu, D.L.; Gu, M.X.; Long, F.Y. Differences in Proinflammatory Property of Six Subtypes of Peroxiredoxins and Anti-Inflammatory Effect of Ligustilide in Macrophages. PLoS ONE 2016, 11, e0164586. [Google Scholar] [CrossRef] [PubMed]
- Rathinam, V.A.; Fitzgerald, K.A. Inflammasome Complexes: Emerging Mechanisms and Effector Functions. Cell 2016, 165, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Lee, H.S.; Kwak, M.S.; Shin, J.S. Inflammasome-Dependent Peroxiredoxin 2 Secretion Induces the Classical Complement Pathway Activation. Immune Netw. 2021, 21, e36. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Wang, C.; Bai, W.; Li, J.; Pan, Y.; Huang, X.; Yang, H.; Feng, Z.; Xiang, Q.; Fei, L.; et al. CD1d1 intrinsic signaling in macrophages controls NLRP3 inflammasome expression during inflammation. Sci. Adv. 2020, 6, eaaz7290. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Li, S.; Tang, D.; Peng, Y.; Meng, J.; Peng, S.; Deng, Z.; Qiu, S.; Liao, X.; Chen, H.; et al. Circulating Peroxiredoxin-1 is a novel damage-associated molecular pattern and aggravates acute liver injury via promoting inflammation. Free Radic. Biol. Med. 2019, 137, 24–36. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, Y.; Wang, Z.; Sun, R.; Zou, B.; Li, R.; Liu, D.; Lin, M.; Zhou, J.; Ning, S.; et al. Peroxiredoxin 3 Inhibits Acetaminophen-Induced Liver Pyroptosis Through the Regulation of Mitochondrial ROS. Front. Immunol. 2021, 12, 652782. [Google Scholar] [CrossRef]
- Liu, W.; Guo, W.; Zhu, Y.; Peng, S.; Zheng, W.; Zhang, C.; Shao, F.; Zhu, Y.; Hang, N.; Kong, L.; et al. Targeting Peroxiredoxin 1 by a Curcumin Analogue, AI-44, Inhibits NLRP3 Inflammasome Activation and Attenuates Lipopolysaccharide-Induced Sepsis in Mice. J. Immunol. 2018, 201, 2403–2413. [Google Scholar] [CrossRef]
- Vázquez-Medina, J.P.; Tao, J.Q.; Patel, P.; Bannitz-Fernandes, R.; Dodia, C.; Sorokina, E.M.; Feinstein, S.I.; Chatterjee, S.; Fisher, A.B. Genetic inactivation of the phospholipase A(2) activity of peroxiredoxin 6 in mice protects against LPS-induced acute lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 316, L656–L668. [Google Scholar] [CrossRef]
- Lipinski, S.; Pfeuffer, S.; Arnold, P.; Treitz, C.; Aden, K.; Ebsen, H.; Falk-Paulsen, M.; Gisch, N.; Fazio, A.; Kuiper, J.; et al. Prdx4 limits caspase-1 activation and restricts inflammasome-mediated signaling by extracellular vesicles. EMBO J. 2019, 38, e101266. [Google Scholar] [CrossRef]
- Minghetti, L. Cyclooxygenase-2 (COX-2) in inflammatory and degenerative brain diseases. J. Neuropathol. Exp. Neurol. 2004, 63, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ruzhi, D.; Hua, X.; Zhang, L.; Lu, F.; Coursey, T.G.; Pflugfelder, S.C.; Li, D.Q. Blueberry Component Pterostilbene Protects Corneal Epithelial Cells from Inflammation via Anti-oxidative Pathway. Sci. Rep. 2016, 6, 19408. [Google Scholar] [CrossRef] [PubMed]
- Hua, X.; Deng, R.; Li, J.; Chi, W.; Su, Z.; Lin, J.; Pflugfelder, S.C.; Li, D.Q. Protective Effects of L-Carnitine Against Oxidative Injury by Hyperosmolarity in Human Corneal Epithelial Cells. Investig. Ophthalmol. Vis. Sci. 2015, 56, 5503–5511. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, J.; Qi, Q.; Yang, L.; Sun, P.; Yuan, X. Modulatory effect of fructooligosaccharide against triphenyltin-induced oxidative stress and immune suppression in goldfish (Carassius auratus). Ecotoxicol. Environ. Saf. 2021, 212, 111966. [Google Scholar] [CrossRef] [PubMed]
- Melhem, H.; Spalinger, M.R.; Cosin-Roger, J.; Atrott, K.; Lang, S.; Wojtal, K.A.; Vavricka, S.R.; Rogler, G.; Frey-Wagner, I. Prdx6 Deficiency Ameliorates DSS Colitis: Relevance of Compensatory Antioxidant Mechanisms. J. Crohn’s Colitis 2017, 11, 871–884. [Google Scholar] [CrossRef]
- Ran, X.Q.; Gao, L.; Yan, M.; Kang, C.J. Peroxiredoxin 4 Interacts With Domeless and Participates in Antibacterial Immune Response Through the JAK/STAT Pathway. Front. Immunol. 2022, 13, 907183. [Google Scholar] [CrossRef] [PubMed]
- Ummanni, R.; Barreto, F.; Venz, S.; Scharf, C.; Barett, C.; Mannsperger, H.A.; Brase, J.C.; Kuner, R.; Schlomm, T.; Sauter, G.; et al. Peroxiredoxins 3 and 4 are overexpressed in prostate cancer tissue and affect the proliferation of prostate cancer cells in vitro. J. Proteome Res. 2012, 11, 2452–2466. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Banerjee, H.; Rojas, H.; Martinez, S.R.; Roy, S.; Jia, Z.; Lilly, M.B.; De Leon, M.; Casiano, C.A. Differential expression of peroxiredoxins in prostate cancer: Consistent upregulation of PRDX3 and PRDX4. Prostate 2011, 71, 755–765. [Google Scholar] [CrossRef]
- Rafiei, S.; Tiedemann, K.; Tabaries, S.; Siegel, P.M.; Komarova, S.V. Peroxiredoxin 4: A novel secreted mediator of cancer induced osteoclastogenesis. Cancer Lett. 2015, 361, 262–270. [Google Scholar] [CrossRef]
- Tiedemann, K.; Sadvakassova, G.; Mikolajewicz, N.; Juhas, M.; Sabirova, Z.; Tabaries, S.; Gettemans, J.; Siegel, P.M.; Komarova, S.V. Exosomal Release of L-Plastin by Breast Cancer Cells Facilitates Metastatic Bone Osteolysis. Transl. Oncol. 2019, 12, 462–474. [Google Scholar] [CrossRef]
- Ding, N.; Jiang, H.; Thapa, P.; Hao, Y.; Alshahrani, A.; Allison, D.; Izumi, T.; Rangnekar, V.M.; Liu, X.; Wei, Q. Peroxiredoxin IV plays a critical role in cancer cell growth and radioresistance through the activation of the Akt/GSK3 signaling pathways. J. Biol. Chem. 2022, 298, 102123. [Google Scholar] [CrossRef] [PubMed]
- Riddell, J.R.; Maier, P.; Sass, S.N.; Moser, M.T.; Foster, B.A.; Gollnick, S.O. Peroxiredoxin 1 stimulates endothelial cell expression of VEGF via TLR4 dependent activation of HIF-1α. PLoS ONE 2012, 7, e50394. [Google Scholar] [CrossRef] [PubMed]
- Riddell, J.R.; Bshara, W.; Moser, M.T.; Spernyak, J.A.; Foster, B.A.; Gollnick, S.O. Peroxiredoxin 1 controls prostate cancer growth through Toll-like receptor 4-dependent regulation of tumor vasculature. Cancer Res. 2011, 71, 1637–1646. [Google Scholar] [CrossRef]
- Karihtala, P.; Mantyniemi, A.; Kang, S.W.; Kinnula, V.L.; Soini, Y. Peroxiredoxins in breast carcinoma. Clin. Cancer Res. 2003, 9, 3418–3424. [Google Scholar]
- Wang, G.; Zhong, W.C.; Bi, Y.H.; Tao, S.Y.; Zhu, H.; Zhu, H.X.; Xu, A.M. The Prognosis Of Peroxiredoxin Family in Breast Cancer. Cancer Manag. Res. 2019, 11, 9685–9699. [Google Scholar] [CrossRef]
- Karihtala, P.; Kauppila, S.; Soini, Y.; Arja Jukkola, V. Oxidative stress and counteracting mechanisms in hormone receptor positive, triple-negative and basal-like breast carcinomas. BMC Cancer 2011, 11, 262. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Hao, L.; Liu, X.; Sun, G.; Xu, R.; Wang, H.; Liu, C. Comprehensive analysis of peroxiredoxins expression profiles and prognostic values in breast cancer. Biomark. Res. 2019, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Edvardsen, H.; Brunsvig, P.F.; Solvang, H.; Tsalenko, A.; Andersen, A.; Syvanen, A.C.; Yakhini, Z.; Børresen-Dale, A.L.; Olsen, H.; Aamdal, S.; et al. SNPs in genes coding for ROS metabolism and signalling in association with docetaxel clearance. Pharm. J. 2010, 10, 513–523. [Google Scholar] [CrossRef]
- Hansen, S.N.; Ehlers, N.S.; Zhu, S.; Thomsen, M.B.; Nielsen, R.L.; Liu, D.; Wang, G.; Hou, Y.; Zhang, X.; Xu, X.; et al. The stepwise evolution of the exome during acquisition of docetaxel resistance in breast cancer cells. BMC Genom. 2016, 17, 442. [Google Scholar] [CrossRef]
- Wang, R.; Liu, Y.; Liu, L.; Chen, M.; Wang, X.; Yang, J.; Gong, Y.; Ding, B.S.; Wei, Y.; Wei, X. Tumor cells induce LAMP2a expression in tumor-associated macrophage for cancer progression. EBioMedicine 2019, 40, 118–134. [Google Scholar] [CrossRef]
- Jezierska-Drutel, A.; Attaran, S.; Hopkins, B.L.; Skoko, J.J.; Rosenzweig, S.A.; Neumann, C.A. The peroxidase PRDX1 inhibits the activated phenotype in mammary fibroblasts through regulating c-Jun N-terminal kinases. BMC Cancer 2019, 19, 812. [Google Scholar] [CrossRef] [PubMed]
- Bostanci, Z.; Mack, R.P., Jr.; Enomoto, L.M.; Alam, S.; Brown, A.; Neumann, C.; Soybel, D.I.; Kelleher, S.L. Marginal zinc intake reduces the protective effect of lactation on mammary gland carcinogenesis in a DMBA-induced tumor model in mice. Oncol. Rep. 2016, 35, 1409–1416. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gromov, P.; Gromova, I.; Bunkenborg, J.; Cabezon, T.; Moreira, J.M.; Timmermans-Wielenga, V.; Roepstorff, P.; Rank, F.; Celis, J.E. Up-regulated proteins in the fluid bathing the tumour cell microenvironment as potential serological markers for early detection of cancer of the breast. Mol. Oncol. 2010, 4, 65–89. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Jiang, H.; Xiao, Z.; Baker, A.; Young, M.R.; Veenstra, T.D.; Colburn, N.H. Sulfiredoxin-Peroxiredoxin IV axis promotes human lung cancer progression through modulation of specific phosphokinase signaling. Proc. Natl. Acad. Sci. USA 2011, 108, 7004–7009. [Google Scholar] [CrossRef]
- Jiang, H.; Wu, L.; Mishra, M.; Chawsheen, H.A.; Wei, Q. Expression of peroxiredoxin 1 and 4 promotes human lung cancer malignancy. Am. J. Cancer Res. 2014, 4, 445–460. [Google Scholar]
- Hwang, J.A.; Song, J.S.; Yu, D.Y.; Kim, H.R.; Park, H.J.; Park, Y.S.; Kim, W.S.; Choi, C.M. Peroxiredoxin 4 as an independent prognostic marker for survival in patients with early-stage lung squamous cell carcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 6627–6635. [Google Scholar]
- Zheng, J.; Guo, X.; Nakamura, Y.; Zhou, X.; Yamaguchi, R.; Zhang, J.; Ishigaki, Y.; Uramoto, H.; Yamada, S. Overexpression of PRDX4 Modulates Tumor Microenvironment and Promotes Urethane-Induced Lung Tumorigenesis. Oxidative Med. Cell. Longev. 2020, 2020, 8262730. [Google Scholar] [CrossRef]
- Hao, C.C.; Luo, J.N.; Xu, C.Y.; Zhao, X.Y.; Zhong, Z.B.; Hu, X.N.; Jin, X.M.; Ge, X. TRIAP1 knockdown sensitizes non-small cell lung cancer to ionizing radiation by disrupting redox homeostasis. Thorac. Cancer 2020, 11, 1015–1025. [Google Scholar] [CrossRef]
- Shioya, A.; Guo, X.; Motono, N.; Mizuguchi, S.; Kurose, N.; Nakada, S.; Aikawa, A.; Ikeda, Y.; Uramoto, H.; Yamada, S. The Combination Of Weak Expression Of PRDX4 And Very High MIB-1 Labelling Index Independently Predicts Shorter Disease-free Survival In Stage I Lung Adenocarcinoma. Int. J. Med. Sci. 2018, 15, 1025–1034. [Google Scholar] [CrossRef]
- Mizutani, K.; Guo, X.; Shioya, A.; Zhang, J.; Zheng, J.; Kurose, N.; Ishibashi, H.; Motono, N.; Uramoto, H.; Yamada, S. The impact of PRDX4 and the EGFR mutation status on cellular proliferation in lung adenocarcinoma. Int. J. Med. Sci. 2019, 16, 1199–1206. [Google Scholar] [CrossRef]
- Kim, H.J.; Chae, H.Z.; Kim, Y.J.; Kim, Y.H.; Hwangs, T.S.; Park, E.M.; Park, Y.M. Preferential elevation of Prx I and Trx expression in lung cancer cells following hypoxia and in human lung cancer tissues. Cell Biol. Toxicol. 2003, 19, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Ahn, J.Y.; Liang, P.; Ip, C.; Zhang, Y.; Park, Y.M. Human prx1 gene is a target of Nrf2 and is up-regulated by hypoxia/reoxygenation: Implication to tumor biology. Cancer Res. 2007, 67, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Seong, J.B.; Kim, B.; Kim, S.; Kim, M.H.; Park, Y.H.; Lee, Y.; Lee, H.J.; Hong, C.W.; Lee, D.S. Macrophage peroxiredoxin 5 deficiency promotes lung cancer progression via ROS-dependent M2-like polarization. Free Radic. Biol. Med. 2021, 176, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Yi, N.; Xiao, M.B.; Ni, W.K.; Jiang, F.; Lu, C.H.; Ni, R.Z. High expression of peroxiredoxin 4 affects the survival time of colorectal cancer patients, but is not an independent unfavorable prognostic factor. Mol. Clin. Oncol. 2014, 2, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Lin, Y.M.; Hasegawa, S.; Shimokawa, T.; Murata, K.; Kameyama, M.; Ishikawa, O.; Katagiri, T.; Tsunoda, T.; Nakamura, Y.; et al. Genes associated with liver metastasis of colon cancer, identified by genome-wide cDNA microarray. Int. J. Oncol. 2004, 24, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Lee, K.C.; Tung, S.Y.; Huang, W.S.; Teng, C.C.; Lee, K.F.; Hsieh, M.C.; Kuo, H.C. 2D-DIGE-MS Proteomics Approaches for Identification of Gelsolin and Peroxiredoxin 4 with Lymph Node Metastasis in Colorectal Cancer. Cancers 2022, 14, 3189. [Google Scholar] [CrossRef]
- Ouyang, M.; Luo, Z.; Zhang, W.; Zhu, D.; Lu, Y.; Wu, J.; Yao, X. Protective effect of curcumin against irinotecaninduced intestinal mucosal injury via attenuation of NFkappaB activation, oxidative stress and endoplasmic reticulum stress. Int. J. Oncol. 2019, 54, 1376–1386. [Google Scholar] [CrossRef]
- Zhu, D.J.; Chen, X.W.; Wang, J.Z.; Ju, Y.L.; Ou Yang, M.Z.; Zhang, W.J. Proteomic analysis identifies proteins associated with curcumin-enhancing efficacy of irinotecan-induced apoptosis of colorectal cancer LOVO cell. Int. J. Clin. Exp. Pathol. 2014, 7, 1–15. [Google Scholar]
- Ribeiro, T.; Lemos, F.; Preto, M.; Azevedo, J.; Sousa, M.L.; Leao, P.N.; Campos, A.; Linder, S.; Vitorino, R.; Vasconcelos, V.; et al. Cytotoxicity of portoamides in human cancer cells and analysis of the molecular mechanisms of action. PLoS ONE 2017, 12, e0188817. [Google Scholar] [CrossRef]
- Song, I.S.; Jeong, Y.J.; Jeong, S.H.; Heo, H.J.; Kim, H.K.; Bae, K.B.; Park, Y.H.; Kim, S.U.; Kim, J.M.; Kim, N.; et al. FOXM1-Induced PRX3 Regulates Stemness and Survival of Colon Cancer Cells via Maintenance of Mitochondrial Function. Gastroenterology 2015, 149, 1006–1016.e9. [Google Scholar] [CrossRef]
- Kobayashi, S.; Hiwasa, T.; Arasawa, T.; Kagaya, A.; Ishii, S.; Shimada, H.; Ito, M.; Suzuki, M.; Kano, M.; Rahmutulla, B.; et al. Identification of specific and common diagnostic antibody markers for gastrointestinal cancers by SEREX screening using testis cDNA phage library. Oncotarget 2018, 9, 18559–18569. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Lee, Y.J.; Park, J.; Kim, T.H.; Hong, S.C.; Jung, E.J.; Ju, Y.T.; Jeong, C.Y.; Park, H.J.; Ko, G.H.; et al. PRDX4 overexpression is associated with poor prognosis in gastric cancer. Oncol. Lett. 2020, 19, 3522–3530. [Google Scholar] [CrossRef] [PubMed]
- Worfolk, J.C.; Bell, S.; Simpson, L.D.; Carne, N.A.; Francis, S.L.; Engelbertsen, V.; Brown, A.P.; Walker, J.; Viswanath, Y.K.; Benham, A.M. Elucidation of the AGR2 Interactome in Esophageal Adenocarcinoma Cells Identifies a Redox-Sensitive Chaperone Hub for the Quality Control of MUC-5AC. Antioxid. Redox Signal. 2019, 31, 1117–1132. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hao, Y.; Lowe, A.W. The adenocarcinoma-associated antigen, AGR2, promotes tumor growth, cell migration, and cellular transformation. Cancer Res. 2008, 68, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Noguchi, H.; Ishii, N.; Homma, T.; Hamada, T.; Hiraki, T.; Zhang, J.; Matsuo, K.; Yokoyama, S.; Ishibashi, H.; et al. The Association of Peroxiredoxin 4 with the Initiation and Progression of Hepatocellular Carcinoma. Antioxid. Redox Signal. 2019, 30, 1271–1284. [Google Scholar] [CrossRef]
- Wang, W.; Shen, X.B.; Huang, D.B.; Jia, W.; Liu, W.B.; He, Y.F. Peroxiredoxin 4 suppresses anoikis and augments growth and metastasis of hepatocellular carcinoma cells through the β-catenin/ID2 pathway. Cell. Oncol. 2019, 42, 769–781. [Google Scholar] [CrossRef]
- Chen, B.; Lan, J.; Xiao, Y.; Liu, P.; Guo, D.; Gu, Y.; Song, Y.; Zhong, Q.; Ma, D.; Lei, P.; et al. Long noncoding RNA TP53TG1 suppresses the growth and metastasis of hepatocellular carcinoma by regulating the PRDX4/β-catenin pathway. Cancer Lett. 2021, 513, 75–89. [Google Scholar] [CrossRef]
- Son, Y.W.; Cheon, M.G.; Kim, Y.; Jang, H.H. Prx2 links ROS homeostasis to stemness of cancer stem cells. Free Radic. Biol. Med. 2019, 134, 260–267. [Google Scholar] [CrossRef]
- Liu, Z.; Hu, Y.; Liang, H.; Sun, Z.; Feng, S.; Deng, H. Silencing PRDX3 Inhibits Growth and Promotes Invasion and Extracellular Matrix Degradation in Hepatocellular Carcinoma Cells. J. Proteome Res. 2016, 15, 1506–1514. [Google Scholar] [CrossRef]
- Kim, T.H.; Song, J.; Alcantara Llaguno, S.R.; Murnan, E.; Liyanarachchi, S.; Palanichamy, K.; Yi, J.Y.; Viapiano, M.S.; Nakano, I.; Yoon, S.O.; et al. Suppression of peroxiredoxin 4 in glioblastoma cells increases apoptosis and reduces tumor growth. PLoS ONE 2012, 7, e42818. [Google Scholar] [CrossRef]
- Kim, T.H.; Song, J.; Kim, S.H.; Parikh, A.K.; Mo, X.; Palanichamy, K.; Kaur, B.; Yu, J.; Yoon, S.O.; Nakano, I.; et al. Piperlongumine treatment inactivates peroxiredoxin 4, exacerbates endoplasmic reticulum stress, and preferentially kills high-grade glioma cells. Neuro-oncology 2014, 16, 1354–1364. [Google Scholar] [CrossRef] [PubMed]
- Hintsala, H.R.; Soini, Y.; Haapasaari, K.M.; Karihtala, P. Dysregulation of redox-state-regulating enzymes in melanocytic skin tumours and the surrounding microenvironment. Histopathology 2015, 67, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Tome, M.E.; Johnson, D.B.; Rimsza, L.M.; Roberts, R.A.; Grogan, T.M.; Miller, T.P.; Oberley, L.W.; Briehl, M.M. A redox signature score identifies diffuse large B-cell lymphoma patients with a poor prognosis. Blood 2005, 106, 3594–3601. [Google Scholar] [CrossRef] [PubMed]
- Palande, K.K.; Beekman, R.; van der Meeren, L.E.; Beverloo, H.B.; Valk, P.J.; Touw, I.P. The antioxidant protein peroxiredoxin 4 is epigenetically down regulated in acute promyelocytic leukemia. PLoS ONE 2011, 6, e16340. [Google Scholar] [CrossRef]
- Mimura, K.; Kua, L.F.; Shimasaki, N.; Shiraishi, K.; Nakajima, S.; Siang, L.K.; Shabbir, A.; So, J.; Yong, W.P.; Kono, K. Upregulation of thioredoxin-1 in activated human NK cells confers increased tolerance to oxidative stress. Cancer Immunol. Immunother. 2017, 66, 605–613. [Google Scholar] [CrossRef]
- Pedro, N.F.; Biselli, J.M.; Maniglia, J.V.; Santi-Neto, D.; Pavarino, E.C.; Goloni-Bertollo, E.M.; Biselli-Chicote, P.M. Candidate Biomarkers for Oral Squamous Cell Carcinoma: Differential Expression of Oxidative Stress-Related Genes. Asian Pac. J. Cancer Prev. 2018, 19, 1343–1349. [Google Scholar] [CrossRef]
- Zhang, M.; Hou, M.; Ge, L.; Miao, C.; Zhang, J.; Jing, X.; Shi, N.; Chen, T.; Tang, X. Induction of peroxiredoxin 1 by hypoxia regulates heme oxygenase-1 via NF-κB in oral cancer. PLoS ONE 2014, 9, e105994. [Google Scholar] [CrossRef]
- Jain, P.; Dvorkin-Gheva, A.; Mollen, E.; Malbeteau, L.; Xie, M.; Jessa, F.; Dhavarasa, P.; Chung, S.; Brown, K.R.; Jang, G.H.; et al. NOX4 links metabolic regulation in pancreatic cancer to endoplasmic reticulum redox vulnerability and dependence on PRDX4. Sci. Adv. 2021, 7, eabf7114. [Google Scholar] [CrossRef]
- Lee, T.H.; Jin, J.O.; Yu, K.J.; Kim, H.S.; Lee, P.C. Inhibition of peroxiredoxin 2 suppresses Wnt/β-catenin signaling in gastric cancer. Biochem. Biophys. Res. Commun. 2019, 512, 250–255. [Google Scholar] [CrossRef]
- Ye, Q.; Zhang, Y.; Cao, Y.; Wang, X.; Guo, Y.; Chen, J.; Horn, J.; Ponomareva, L.V.; Chaiswing, L.; Shaaban, K.A.; et al. Frenolicin B Targets Peroxiredoxin 1 and Glutaredoxin 3 to Trigger ROS/4E-BP1-Mediated Antitumor Effects. Cell Chem. Biol. 2019, 26, 366–377.e12. [Google Scholar] [CrossRef]


| Species | Gene Symbol |
% Sequence Similarity | |
|---|---|---|---|
| Protein | DNA | ||
| H. Sapiens | PRDX4 | ||
| M. mulatta (Rhesus macaque) | PRDX4 | 98.5 | 98.4 |
| C. lupus (Wolf) | PRDX4 | 93 | 89.2 |
| B. taurus (Cattle) | PRDX4 | 93.8 | 90.8 |
| M. musculus (House mouse) | Prdx4 | 95 | 89.1 |
| R. norvegicus (Brown rat) | Prdx4 | 94.5 | 90.3 |
| G. gallus (Red junglefowl) | PRDX4 | 91.9 | 81.6 |
| X. tropicalis (Western clawed frog) | prdx4 | 93.6 | 81.1 |
| D. rerio (Zebrafish) | prdx4 | 88.7 | 74.8 |
| D. melanogaster (Common fruit fly) | Jafrac2 | 71 | 64.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thapa, P.; Ding, N.; Hao, Y.; Alshahrani, A.; Jiang, H.; Wei, Q. Essential Roles of Peroxiredoxin IV in Inflammation and Cancer. Molecules 2022, 27, 6513. https://doi.org/10.3390/molecules27196513
Thapa P, Ding N, Hao Y, Alshahrani A, Jiang H, Wei Q. Essential Roles of Peroxiredoxin IV in Inflammation and Cancer. Molecules. 2022; 27(19):6513. https://doi.org/10.3390/molecules27196513
Chicago/Turabian StyleThapa, Pratik, Na Ding, Yanning Hao, Aziza Alshahrani, Hong Jiang, and Qiou Wei. 2022. "Essential Roles of Peroxiredoxin IV in Inflammation and Cancer" Molecules 27, no. 19: 6513. https://doi.org/10.3390/molecules27196513
APA StyleThapa, P., Ding, N., Hao, Y., Alshahrani, A., Jiang, H., & Wei, Q. (2022). Essential Roles of Peroxiredoxin IV in Inflammation and Cancer. Molecules, 27(19), 6513. https://doi.org/10.3390/molecules27196513






