The Remediation in Enzyme’s Activities in Plants: Tea Waste as a Modifier to Improve the Efficiency of Growth of Helianthus annuus in Contaminated Soil
Abstract
1. Introduction
2. Results and Discussion
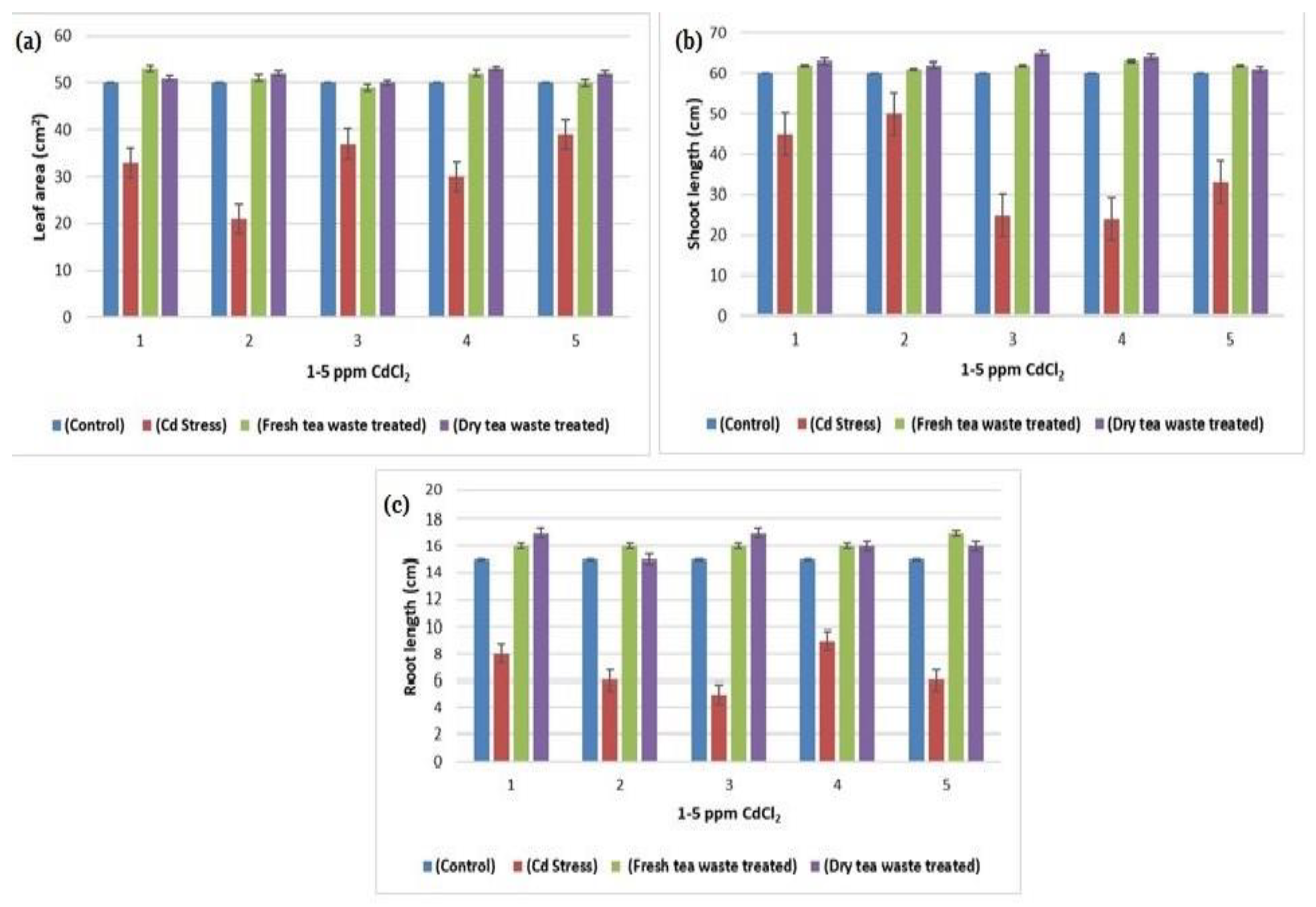
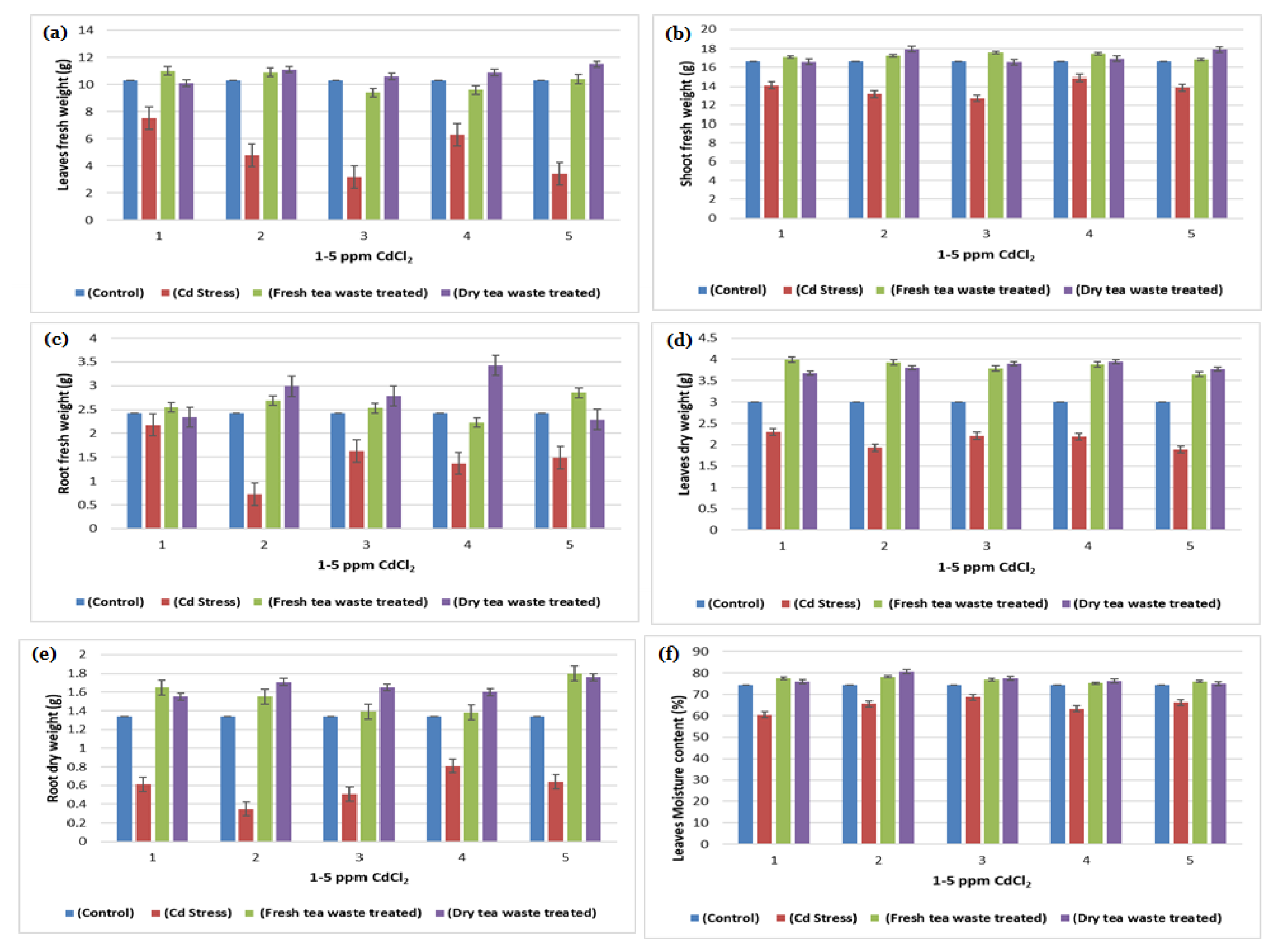
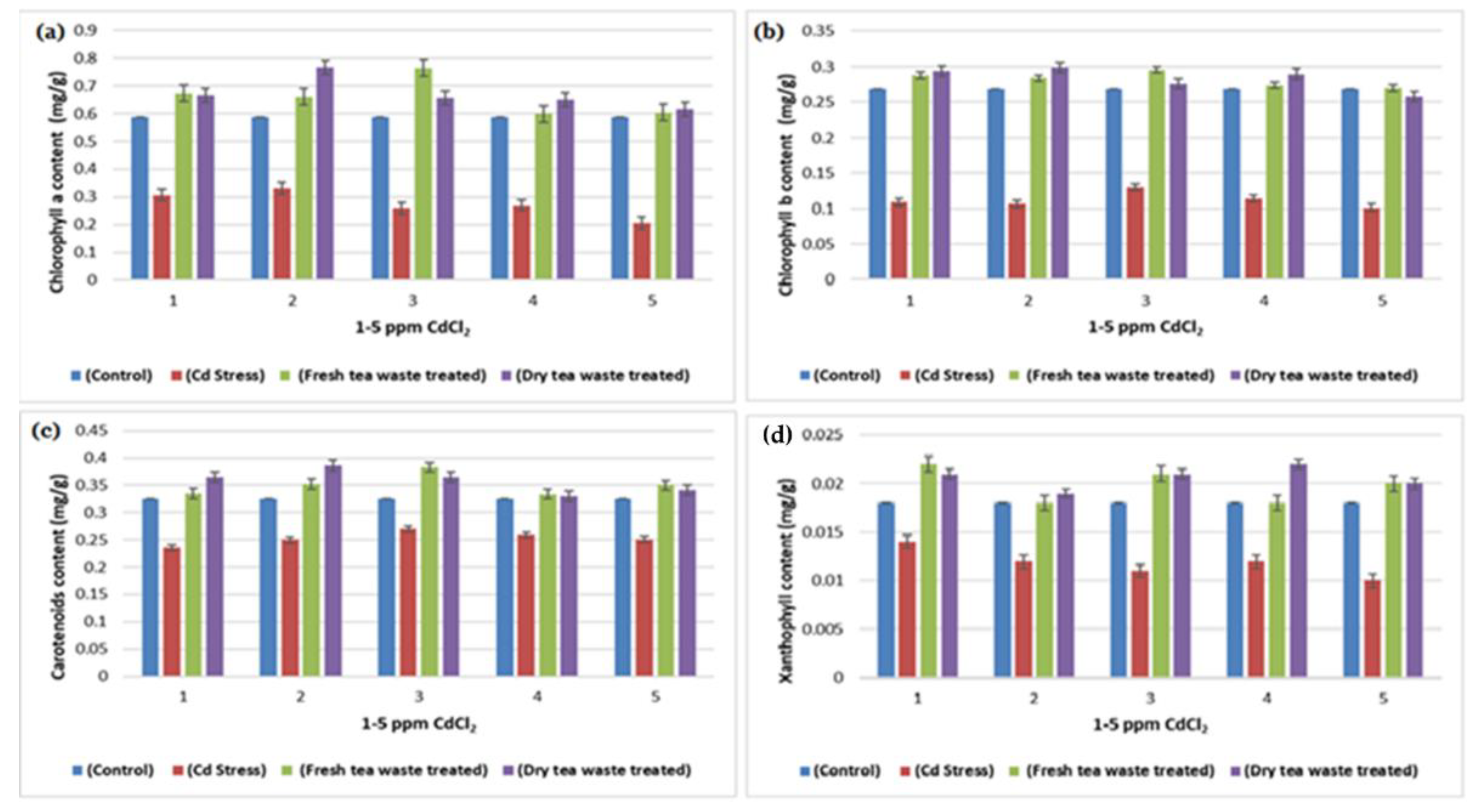
2.1. Biomass Determination and Analysis of Photosynthesis
2.2. Scan Electron Microscopy of the Helianthus annuus Plants
2.3. The Remediation of Enzymes as a Biomarker
2.4. Mechanism of Remediation in Enzymes
3. Materials and Methods
3.1. Cultivation of the Plants and Experimental Design
3.2. Physical Measurements of the Soil and Plant
3.3. Growth Measurement
3.4. Determination of Pigments Content
3.5. Analysis of Carbohydrate
3.6. Analysis of Reducing Sugar
3.7. Estimation of Amylase Activity
3.8. Estimation of Peroxidase
3.9. Determination of Nitrite Reductase
3.10. Determination of Nitrate Reductase
3.11. Scanning Electron Microscopy of Surface of Leaves
3.12. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rao, M.A.; Scelza, R.; Scotti, R.; Gianfreda, L. Role of enzymes in the remediation of polluted environments. J. Soil Sci. Plant Nutr. 2010, 10, 333–353. [Google Scholar] [CrossRef]
- Ojuederie, O.B.; Babalola, O.O. Microbial and plant-assisted bioremediation of heavy metal polluted environments: A review. Int. J. Environ. Res. Public Health 2017, 14, 1504. [Google Scholar] [CrossRef] [PubMed]
- Hanumanth Kumar, G.; Pramoda Kumari, J. Heavy Metal Lead Influative Toxicity and Its Assessment in Phytoremediating Plants—A Review. Water Air Soil Pollut. 2015, 226, 1–11. [Google Scholar] [CrossRef]
- Łukowski, A.; Wiater, J. The content and solubility of lead in arable soils of the Podlasie Province (eastern Poland). Soil Sci. Annu. J. 2016, 67, 190–196. [Google Scholar] [CrossRef]
- Brokbartold, M.; Wischermann, M.; Marschner, B. Plant availability and uptake of lead, zinc, and cadmium in soils contaminated with anti-corrosion paint from pylons in comparison to heavy metal contaminated Urban soils. Water Air Soil Pollut. 2012, 223, 199–213. [Google Scholar] [CrossRef]
- Azmat, R.; Akhter, H. Changes in some biophysical and biochemical parameters of mungbean [Vigna radiata (L.) Wilczek] grown on chromium-contaminated soils treated with solid tea wastage. Pak. J. Bot. 2010, 42, 3065–3071. [Google Scholar]
- Chugh, L.K.; Sawhney, S.K. Effect of cadmium on germination, amylases and rate of respiration of germinating pea seeds. Environ. Pollut. 1996, 92, 1–5. [Google Scholar] [CrossRef]
- Asada, K. The water-water cycle in chloroplasts: Scavenging of active oxygens and dissipation of excess photons. Annu. Rev. Plant Biol. 1999, 50, 601–639. [Google Scholar] [CrossRef]
- Gouia, H.; Habib Ghorbal, M.; Meyer, C. Effects of cadmium on activity of nitrate reductase and on other enzymes of the nitrate assimilation pathway in bean. Plant Physiol. Biochem. 2000, 38, 629–638. [Google Scholar] [CrossRef]
- Chaffei, C.; Pageau, K.; Suzuki, A.; Gouia, H.; Ghorbel, M.H.; Masclaux-Daubresse, C. Cadmium toxicity induced changes in nitrogen management in Lycopersicon esculentum leading to a metabolic safeguard through an amino acid storage strategy. Plant Cell Physiol. 2004, 45, 1681–1693. [Google Scholar] [CrossRef]
- Romero-Puertas, M.C.; Palma, J.M.; Gómez, M.; Del Río, L.A.; Sandalio, L.M. Cadmium causes the oxidative modification of proteins in pea plants. Plant Cell Environ. 2002, 25, 677–686. [Google Scholar] [CrossRef]
- Hussain, S.; Anjali, K.P.; Hassan, S.T.; Dwivedi, P.B. Waste tea as a novel adsorbent: A review. Appl. Water Sci. 2018, 8, 165. [Google Scholar] [CrossRef]
- Milinković, M.; Lalević, B.; Jovičić-Petrović, J.; Golubović-Ćurguz, V.; Kljujev, I.; Raičević, V. Biopotential of compost and compost products derived from horticultural waste—Effect on plant growth and plant pathogens’ suppression. Process Saf. Environ. Prot. 2019, 121, 299–306. [Google Scholar] [CrossRef]
- De Corato, U. Agricultural waste recycling in horticultural intensive farming systems by on-farm composting and compost-based tea application improves soil quality and plant health: A review under the perspective of a circular economy. Sci. Total Environ. 2020, 738, 139840. [Google Scholar] [CrossRef]
- Samet, M.; Charfeddine, M.; Kamoun, L.; Nouri-Ellouze, O.; Gargouri-Bouzid, R. Effect of compost tea containing phosphogypsum on potato plant growth and protection against Fusarium solani infection. Environ. Sci. Pollut. Res. 2018, 25, 18921–18937. [Google Scholar] [CrossRef]
- Dahkaei, M.P.; Gholami, M.E.H.R.A.N. Effects of Different Media on Growth of Pot Plants Dracaena marginata Ait. and Beaucarnea recurvata Lem. Seed Plant Product. J. 2009, 25, 63–77. [Google Scholar]
- Arancon, N.Q.; Edwards, C.A.; Dick, R.; Dick, L. Vermicompost tea production and plant growth impacts. Biocycle 2007, 48, 51. [Google Scholar]
- Boothby, D.; Wright, S.T.C. Effects of kinetin and other plant growth regulators on starch degradation. Nature 1962, 196, 389–390. [Google Scholar] [CrossRef]
- Parmar, N.G.; Vithalani, S.D.; Chanda, S.V. Alteration in growth and peroxidase activity by heavy metals in Phaseolus seedlings. Acta Physiol. Plant. 2002, 24, 89–95. [Google Scholar] [CrossRef]
- MacFarlane, G.R.; Burchett, M.D. Photosynthetic pigments and peroxidase activity as indicators of heavy metal stress in the grey mangrove, Avicennia marina (Forsk.) Vierh. Mar. Pollut. Bull. 2001, 42, 233–240. [Google Scholar] [CrossRef]
- Campbell, W.H. Nitrate reductase structure, function and regulation: Bridging the gap between biochemistry and physiology. Annu. Rev. Plant Biol. 1999, 50, 277–303. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, W.M.; Kandlbinder, A.; Stoimenova, M.; Glaab, J. Discrepancy between nitrate reduction rates in intact leaves and nitrate reductase activity in leaf extracts: What limits nitrate reduction in situ? Planta 2000, 210, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Barthes, L.; Deléens, E.; Bousser, A.; Hoarau, J.; Prioul, J.L. Xylem exudation is related to nitrate assimilation pathway in detopped maize seedlings: Use of nitrate reductase and glutamine synthetase inhibitors as tools. J. Exp. Bot. 1996, 47, 485–495. [Google Scholar] [CrossRef]
- Vicuna, D. The Role of Peroxidases in the Development of Plants and Their Responses to Abiotic Stresses. Ph.D. Thesis, Technological University Dublin, Dublin, Ireland, 2005; p. 146. [Google Scholar]
- Bavi, K.; Kholdebarin, B.; Moradshahi, A. Effect of cadmium on growth, protein content and peroxidase activity in pea plants. Pak. J. Bot. 2011, 43, 1467–1470. [Google Scholar]
- El-Shintinawy, F.; El-Ansary, A. Differential effect of Cd2+ and Ni2+ on amino acid metabolism in soybean seedlings. Biol. Plant. 2000, 43, 79–84. [Google Scholar] [CrossRef]
- Merlo, L.; Ferretti, M.; Ghisi, R.; Passera, C. Developmental changes of enzymes of malate metabolism in relation to respiration, photosynthesis and nitrate assimilation in peach leaves. Physiol. Plant. 1993, 89, 71–76. [Google Scholar] [CrossRef]
- Nussbaum, S.; Schmutz, D.; Brunold, C. Regulation of assimilatory sulfate reduction by cadmium in Zea mays L. Plant Physiol. 1988, 88, 1407–1410. [Google Scholar] [CrossRef]
- Hernández, L.E.; Carpena-Ruiz, R.; Gárate, A. Alterations in the mineral nutrition of pea seedlings exposed to cadmium. J. Plant Nutr. 1996, 19, 1581–1598. [Google Scholar] [CrossRef]
- Ouariti, O.; Gouia, H.; Ghorbal, M.H. Responses of bean and tomato plants to cadmium: Growth, mineral nutrition, and nitrate reduction. Plant Physiol. Biochem. 1997, 35, 347–354. [Google Scholar]
- Rai, V.; Khatoon, S.; Bisht, S.S.; Mehrotra, S. Effect of cadmium on growth, ultramorphology of leaf and secondary metabolites of Phyllanthus amarus Schum. and Thonn. Chemosphere 2005, 61, 1644–1650. [Google Scholar] [CrossRef]
- Barrs, H.; Weatherley, P. A Re-Examination of the Relative Turgidity Technique for Estimating Water Deficits in Leaves. Aust. J. Biol. Sci. 1962, 15, 413–428. [Google Scholar] [CrossRef]
- Maclachlan, S.; Zalik, S. Plastid structure, chlorophyll concentration, and free amino acid composition of a chlorophyll mutant of barley. Can. J. Bot. 1963, 41, 1053–1062. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 1954, 57, 508–514. [Google Scholar] [CrossRef]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Bernfeld, P. Amylases, alpha and beta. Methods Enzymol. 1955, 1, 149–158. [Google Scholar]
- Maehly, A.; Chance, B. Catalases and peroxidases. Methods Biochem. Anal. 1954, 1, 357–424. [Google Scholar] [PubMed]
- Ramarao, C.S.; Patil, V.H.; Dhak, B.D.; Kadrekar, S.B. A Simple in vivo Method for the Determination of Nitrite Reductase Activity in Rice Roots. J. Plant Physiol. 1983, 109, 81–85. [Google Scholar] [CrossRef]
- Sym, G.J. Optimisation of the in-vivo assay conditions for nitrate reductase in barley (Hordeum vulgare L. cv. Igri). J. Sci. Food Agric. 1984, 35, 725–730. [Google Scholar] [CrossRef]
- Pathan, A.K.; Bond, J.; Gaskin, R.E. Sample preparation for scanning electron microscopy of plant surfaces-Horses for courses. Micron 2008, 39, 1049–1061. [Google Scholar] [CrossRef]



| Plants Sample | Amylase Activity Content (µg/mL) | |
|---|---|---|
| Leaves | Roots | |
| 1 ppm CdCl2 | ||
| Pa (Control) | 37.46 ± 1.2 ** | 41.06 ± 0.1 ** |
| Pb1 (Cd Stress) | 21.80 ± 0.7 ** | 12.07 ± 0.8 ** |
| Pc1 (Fresh tea waste treated) | 38.32 ± 1.3 ** | 45.53 ± 0.4 ** |
| Pd1 (Dry tea waste treated) | 39.98 ± 0.4 ** | 45.33 ± 0.0 ** |
| 2 ppm CdCl2 | ||
| Pa (Control) | 37.46 ± 0.1 ** | 41.06 ± 0.0 ** |
| Pb2 (Cd Stress) | 23.53 ± 0.2 ** | 13.26 ± 2.1 ** |
| Pc2 (Fresh tea waste treated) | 40.06 ± 0.8 ** | 39.86 ± 2.2 ** |
| Pd12 (Dry tea waste treated) | 41.65 ± 0.0 ** | 48.20 ± 0.8 ** |
| 3 ppm CdCl2 | ||
| Pa (Control) | 37.46 ± 0.3 ** | 41.06 ± 1.1 ** |
| Pb3 (Cd Stress) | 24.07 ± 3.1 ** | 18.73 ± 1.3 ** |
| Pc3 (Fresh tea waste treated) | 37.21 ± 2.0 ** | 37.86 ± 0.5 ** |
| Pd3 (Dry tea waste treated) | 38.67 ± 0.1 ** | 47.40 ± 0.2 ** |
| 4 ppm CdCl2 | ||
| Pa (Control) | 37.46 ± 0.2 ** | 41.06 ± 1.9 ** |
| Pb4 (Cd Stress) | 21.67 ± 0.0 ** | 19.80 ± 0.4 ** |
| Pc4 (Fresh tea waste treated) | 41.67 ± 1.7 ** | 44.53 ± 0.9 ** |
| Pd4 (Dry tea waste treated) | 42.06 ± 0.0 ** | 44.67 ± 0.4 ** |
| 5 ppm CdCl2 | ||
| Pa (Control) | 37.46 ± 0.1 ** | 41.06 ± 0.9 ** |
| Pb5 (Cd Stress) | 25.20 ± 0.8 ** | 13.47 ± 0.1 ** |
| Pc5 (Fresh tea waste treated) | 43.13 ± 1.7 ** | 46.88 ± 0.5 ** |
| Pd5 (Dry tea waste treated) | 44.47 ± 0.3 ** | 46.47 ± 1.6 ** |
| Plants Sample | Peroxidase Activity Content (µg/mL) | |
|---|---|---|
| Leaves | Roots | |
| 1 ppm CdCl2 | ||
| Pa (Control) | 83.53 ± 0.7 ** | 61.53 ± 0.1 ** |
| Pb1 (Cd Stress) | 92.69 ± 0.5 ** | 72.61 ± 0.7 ** |
| Pc1 (Fresh tea waste treated) | 78.46 ± 0.1 ** | 60.92 ± 0.6 ** |
| Pd1 (Dry tea waste treated) | 74.76 ± 1.5 ** | 61.46 ± 0.4 ** |
| 2 ppm CdCl2 | ||
| Pa (Control) | 83.53 ± 0.2 ** | 61.53 ± 0.1 ** |
| Pb2 (Cd Stress) | 90.15 ± 0.7 ** | 70.92 ± 0.5 ** |
| Pc2 (Fresh tea waste treated) | 80.46 ± 0.0 ** | 60.15 ± 1.2 ** |
| Pd2 (Dry tea waste treated) | 81.69 ± 1.7 ** | 61.23 ± 0.0 ** |
| 3 ppm CdCl2 | ||
| Pa (Control) | 83.53 ± 0.2 ** | 61.53 ± 0.2 ** |
| Pb3 (Cd Stress) | 93.23 ± 2.1 ** | 72.53 ± 0.0 ** |
| Pc3 (Fresh tea waste treated) | 82.46 ± 0.3 ** | 62.92 ± 1.2 ** |
| Pd3 (Dry tea waste treated) | 82.76 ± 0.1 ** | 61.61 ± 0.4 ** |
| 4 ppm CdCl2 | ||
| Pa (Control) | 83.53 ± 2.7 ** | 61.53 ± 0.4 ** |
| Pb4 (Cd Stress) | 95.69 ± 0.9 ** | 74.69 ± 0.2 ** |
| Pc4 (Fresh tea waste treated) | 82.30 ± 0.2 ** | 61.23 ± 0.6 ** |
| Pd4 (Dry tea waste treated) | 83.46 ± 1.7 ** | 61.38 ± 0.0 ** |
| 5 ppm CdCl2 | ||
| Pa (Control) | 83.53 ± 0.0 ** | 61.53 ± 0.3 ** |
| Pb5 (Cd Stress) | 89.92 ± 0.3 ** | 74.92 ± 0.8 ** |
| Pc5 (Fresh tea waste treated) | 82.92 ± 1.4 ** | 61.46 ± 0.1 ** |
| Pd5 (Dry tea waste treated) | 83.69 ± 2.7 ** | 62.15 ± 1.3 ** |
| Plants Sample | Nitrite Reductase Activity Content (µg/mL) | Nitrate Reductase Activity Content (µg/mL) | ||
|---|---|---|---|---|
| Leaves | Roots | Leaves | Roots | |
| 1 ppm CdCl2 | ||||
| Pa (Control) | 402.8 ± 0.9 ** | 263.4 ± 0.1 ** | 45.6 ± 0.0 ** | 38.3 ± 0.3 ** |
| Pb (Cd Stress) | 310.2 ± 1.6 ** | 186.8 ± 0.3 ** | 28.3 ± 0.1 ** | 22.6 ± 0.1 ** |
| Pc (Fresh tea waste treated) | 404.2 ± 0.1 ** | 265.6 ± 1.7 ** | 46.7 ± 0.3 ** | 39.3 ± 0.5 ** |
| Pd (Dry tea waste treated) | 403.2 ± 0.3 ** | 264.6 ± 0.1 ** | 45.9 ± 0.1 ** | 38.6 ± 1.5 ** |
| 2 ppm CdCl2 | ||||
| Pa (Control) | 402.8 ± 0.2 ** | 263.4 ± 0.0 ** | 45.6 ± 1.5 ** | 38.3 ± 0.6 ** |
| Pb2 (Cd Stress) | 308.8 ± 1.4 ** | 184.2 ± 0.1 ** | 24.1 ± 0.1 ** | 22.6 ± 0.2 ** |
| Pc2 (Fresh tea waste treated) | 402.9 ± 0.4 ** | 264.4 ± 0.6 ** | 45.9 ± 0.8 ** | 41.5 ± 1.7 ** |
| Pd2 (Dry tea waste treated) | 403.5 ± 0.6 ** | 265.7 ± 0.2 ** | 46.8 ± 0.2 ** | 42.9 ± 0.5 ** |
| 3 ppm CdCl2 | ||||
| Pa (Control) | 402.8 ± 0.2 ** | 263.45 ± 0.1 ** | 45.6 ± 1.6 ** | 38.3 ± 0.8 ** |
| Pb3 (Cd Stress) | 305.4 ± 0.1 ** | 181.36 ± 0.3 ** | 25.4 ± 1.1 ** | 23.8 ± 0.6 ** |
| Pc3 (Fresh tea waste treated) | 403.6 ± 0.4 ** | 263.81 ± 1.7 ** | 44.9 ± 0.2 ** | 39.6 ± 1.3 ** |
| Pd3 (Dry tea waste treated) | 404.2 ± 0.8 ** | 265.63 ± 0.3 ** | 46.2 ± 0.7 ** | 40.6 ± 0.3 ** |
| 4 ppm CdCl2 | ||||
| Pa (Control) | 402.8 ± 1.2 ** | 263.4 ± 0.2 ** | 45.6 ± 0.1 ** | 38.3 ± 0.1 ** |
| Pb4 (Cd Stress) | 307.4 ± 0.4 ** | 185.2 ± 0.1 ** | 29.3 ± 0.9 ** | 25.6 ± 0.9 ** |
| Pc4 (Fresh tea waste treated) | 404.2 ± 0.9 ** | 262.3 ± 1.2 ** | 45.5 ± 0.2 ** | 42.9 ± 0.0 ** |
| Pd4 (Dry tea waste treated) | 404.5 ± 1.5 ** | 264.0 ± 0.8 ** | 47.1 ± 1.3 ** | 43.1 ± 0.1 ** |
| 5 ppm CdCl2 | ||||
| Pa (Control) | 402.8 ± 0.3 ** | 263.4 ± 0.4 ** | 45.6 ± 0.4 ** | 38.3 ± 0.1 ** |
| Pb (Cd Stress) | 308.4 ± 2.1 ** | 183.8 ± 1.8 ** | 24.1 ± 0.2 ** | 23.1 ± 0.5 ** |
| Pc5 (Fresh tea waste treated) | 404.6 ± 1.6 ** | 264.8 ± 0.1 ** | 46.5 ± 1.7 ** | 39.9 ± 1.4 ** |
| Pd5 (Dry tea waste treated) | 405.7 ± 0.2 ** | 264.09 ± 1.2 ** | 46.9 ± 0.3 ** | 40.1 ± 1.5 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moin, S.; Azmat, R.; Ahmed, W.; Qayyum, A.; El-Serehy, H.A.; Hefft, D.I. The Remediation in Enzyme’s Activities in Plants: Tea Waste as a Modifier to Improve the Efficiency of Growth of Helianthus annuus in Contaminated Soil. Molecules 2022, 27, 6362. https://doi.org/10.3390/molecules27196362
Moin S, Azmat R, Ahmed W, Qayyum A, El-Serehy HA, Hefft DI. The Remediation in Enzyme’s Activities in Plants: Tea Waste as a Modifier to Improve the Efficiency of Growth of Helianthus annuus in Contaminated Soil. Molecules. 2022; 27(19):6362. https://doi.org/10.3390/molecules27196362
Chicago/Turabian StyleMoin, Sumeira, Rafia Azmat, Waseem Ahmed, Abdul Qayyum, Hamed A. El-Serehy, and Daniel Ingo Hefft. 2022. "The Remediation in Enzyme’s Activities in Plants: Tea Waste as a Modifier to Improve the Efficiency of Growth of Helianthus annuus in Contaminated Soil" Molecules 27, no. 19: 6362. https://doi.org/10.3390/molecules27196362
APA StyleMoin, S., Azmat, R., Ahmed, W., Qayyum, A., El-Serehy, H. A., & Hefft, D. I. (2022). The Remediation in Enzyme’s Activities in Plants: Tea Waste as a Modifier to Improve the Efficiency of Growth of Helianthus annuus in Contaminated Soil. Molecules, 27(19), 6362. https://doi.org/10.3390/molecules27196362






