Dry Powder Inhalers for Proteins Using Cryo-Milled Electrospun Polyvinyl Alcohol Nanofiber Mats
Abstract
:1. Introduction
2. Results
2.1. Physicochemical Properties of Electrospun Nanofiber Mats and Milled Nanofiber Mats
2.2. In Vitro Aerosol Performance of the Milled Nanofiber Mats
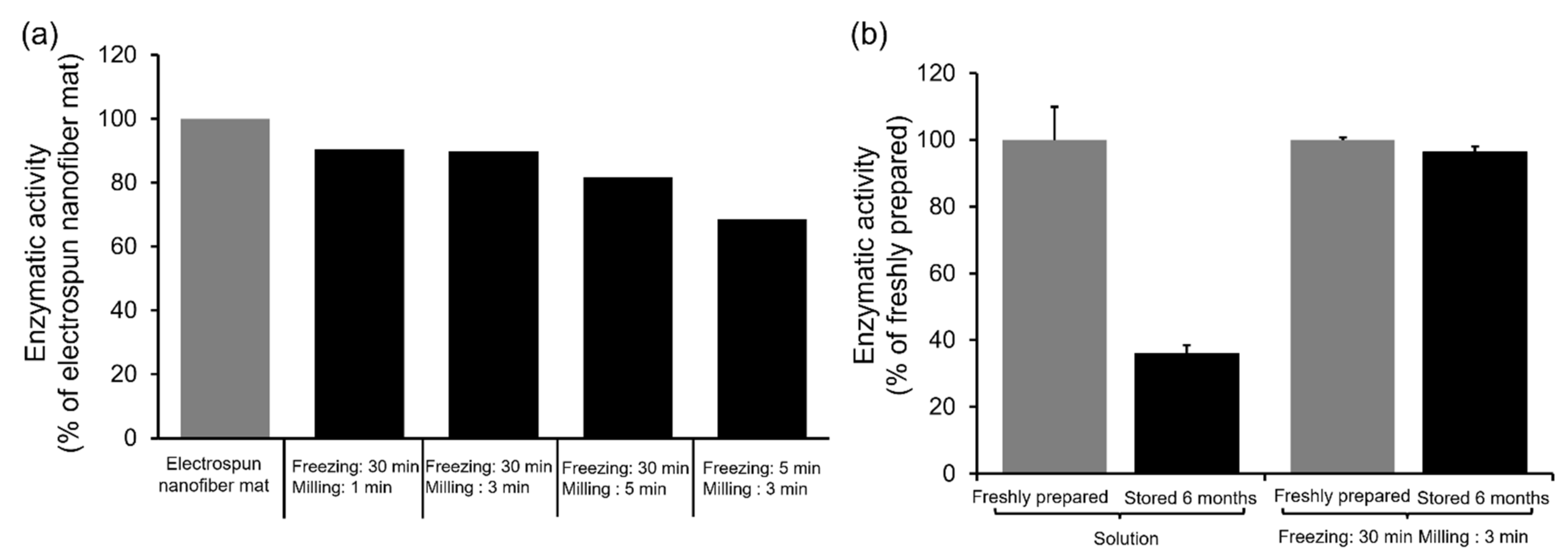
2.3. Enzyme Activities of Milled Nanofiber Mats Containing α-Chymotrypsin
3. Discussion
4. Materials and Methods
4.1. Preparation of α-Chy-Loaded Electrospun PVA Nanofiber Mats
4.2. Electrospinning/Cryo-Milling Processes of Electrospun Nanofiber Mats
4.3. Physicochemical Properties of Electrospun Nanofiber Mats and Milled Nanofiber Mats
4.4. In Vitro Aerosol Performance
4.5. Enzymatic Activities of Milled Nanofiber Mats Containing α-Chymotrypsin
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Abbreviations
| α-Chy | α-chymotrypsin |
| ACI | Andersen cascade impactor |
| DD | delivered dose |
| DPI | dry powder inhaler |
| FPF | fine particle fraction |
| MMAD | mass median aerodynamic diameter |
| PVA | polyvinyl alcohol |
References
- EvaluatePharma, 2021. Evaluate Pharma World Preview 2021, Outlook to 2026. Available online: https://www.evaluate.com/jp/node/17096 (accessed on 26 April 2022).
- Ferrati, S.; Wu, T.; Kanapuram, S.R.; Smyth, H.D.C. Dosing considerations for inhaled biologics. Int. J. Pharm. 2018, 549, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.A.; Hannavy, K.; Davies, N.; Pirrie, A.; Coffee, R.A.; Hyde, S.C.; Gill, D.R. Electrohydrodynamic comminution: A novel technique for the aerosolisation of plasmid DNA. Pharm. Res. 2005, 22, 1294–1304. [Google Scholar] [CrossRef] [PubMed]
- Tahara, K.; Hashimoto, W.; Takeuchi, H. Inhalation properties and stability of nebulized naked siRNA solution for pulmonary therapy. Chem. Pharm. Bull. 2016, 64, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Luu, Y.K.; Kim, K.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Development of a nanostructured DNA delivery scaffold via electrospinning of PLGA and PLA-PEG block copolymers. J. Control. Release 2003, 89, 341–353. [Google Scholar] [CrossRef]
- Shibata, T.; Yoshimura, N.; Kobayashi, A.; Ito, T.; Hara, K.; Tahara, K. Emulsion-electrospun polyvinyl alcohol nanofibers as a solid dispersion system to improve solubility and control the release of probucol, a poorly water-soluble drug. J. Drug Deliv. Sci. Technol. 2022, 67, 102953. [Google Scholar] [CrossRef]
- Newman, A.P.; Hollingworth, A.; Clark, A.R. Effect of different modes of inhalation on drug delivery from a dry powder inhaler. Int. J. Pharm. 1994, 102, 127–132. [Google Scholar] [CrossRef]
- Dolovich, M.B.; Dhand, R. Aerosol drug delivery: Developments in device design and clinical use. Lancet 2011, 377, 1032–1045. [Google Scholar] [CrossRef]
- Chaurasiya, B.; Zhao, Y.Y. Dry powder for pulmonary delivery: A comprehensive review. Pharmaceutics 2021, 13, 31. [Google Scholar] [CrossRef]
- Le, V.N.P.; Robins, E.; Flament, M.P. Agglomerate behaviour of fluticasone propionate within dry powder inhaler formulations. Eur. J. Pharm. Biopharm. 2012, 80, 596–603. [Google Scholar] [CrossRef]
- Leung, S.S.Y.; Tang, P.; Zhou, Q.T.; Tong, Z.; Leung, C.; Decharaksa, J.; Yang, R.; Chan, H.K. De-agglomeration effect of the US pharmacopeia and Alberta throats on carrier-based powders in commercial inhalation products. AAPS J. 2015, 17, 1407–1416. [Google Scholar] [CrossRef]
- Guenette, E.; Barrett, A.; Kraus, D.; Brody, R.; Harding, L.; Magee, G. Understanding the effect of lactose particle size on the properties of DPI formulations using experimental design. Int. J. Pharm. 2009, 380, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Seville, P.C.; Kellaway, I.W.; Birchall, L.C. Preparation of dry powder dispersions for non-viral gene delivery by freeze-drying and spray-drying. J. Gene Med. 2002, 4, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Tanikawa, Y.; Ido, Y.; Ando, R.; Obata, A.; Nagata, K.; Kasuga, T.; Mizuno, T. Coaxial electrospun fibermat of poly(AM/DAAM)/ADH and PCL: Versatile platform for functioning active enzymes. Bull. Chem. Soc. Jpn. 2020, 93, 1155–1163. [Google Scholar] [CrossRef]
- Lozano, P.; Diego, T.D.; Iborra, J.L. Dynamic Structure/Function Relationships in the α-Chymotrypsin Deactivation Process by Heat and pH. Eur. J. Biochem. 1997, 248, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Otake, H.; Okuda, T.; Hira, D.; Kojima, H.; Shimada, Y.; Okamoto, H. Inhalable spray-freezedried powder with L-leucine that delivers particles independent of inspiratory flow pattern and inhalation device. Pharm. Res. 2016, 33, 922–931. [Google Scholar] [CrossRef]
- Lienhard, J.H., IV; Lienhard, J.H., V. A Heat Transfer Textbook, 5th ed.; Dover Publications: New York, NY, USA, 2020; ISBN 978-0486837352. [Google Scholar]
- Kadomae, Y.; Sugimoto, M.; Taniguchi, T.; Koyama, K. Relation between spinning conditions and jet profile in electrospinning. Seikei-Kakou 2010, 21, 627–632. [Google Scholar] [CrossRef]
- Nikolaou, M.; Krasia-Christoforou, T. Electrohydrodynamic methods for the development of pulmonary drug delivery systems. Eur. J. Pharm. Sci. 2018, 113, 29–40. [Google Scholar] [CrossRef]
- Hong, P.-Y.; Huang, Y.-H.; Lim, G.C.W.; Chen, Y.-P.; Hsiao, C.-J.; Chen, L.-H.; Ciou, J.-Y.; Hsieh, L.-S. Production of trans-cinnamic acid by immobilization of the Bambusa oldhamii BoPAL1 and BoPAL2 phenylalanine ammonia-lyases on electrospun nanofibers. Int. J. Mol. Sci. 2021, 22, 11184. [Google Scholar] [CrossRef]
- Maretschek, S.; Greiner, A.; Kissel, T. Electrospun biodegradable nanofiber nonwovens for controlled release of proteins. J. Control. Release 2008, 127, 180–187. [Google Scholar] [CrossRef]
- Katsogiannis, K.A.G.; Vladisavljević, G.T.; Georgiadou, S. Porous electrospun polycaprolactone (PCL) fibres by phase separation. Eur. Polym. J. 2015, 69, 284–295. [Google Scholar] [CrossRef]
- Thieme, M.; Agarwal, S.; Wendorff, J.H.; Greiner, A. Electrospinning and cutting of ultrafine bioerodible poly(lactide-co-ethylene oxide) tri- and multiblock copolymer fibers for inhalation applications. Polym. Adv. Technol. 2011, 22, 1335–1344. [Google Scholar] [CrossRef]
- Ito, T.; Okuda, T.; Takashima, Y.; Okamoto, H. Naked pDNA inhalation powder composed of hyaluronic acid exhibits high gene expression in the lungs. Mol. Pharm. 2019, 16, 489–497. [Google Scholar] [CrossRef] [PubMed]

| Formulation | D10 (µm) | D50 (µm) | D90 (µm) | Span |
|---|---|---|---|---|
| Freezing: 30 min, milling: 1 min | 8.73 ± 1.72 | 34.39 ± 1.22 | 54.99 ± 2.45 | 1.35 ± 0.10 |
| Freezing: 30 min, milling: 3 min | 6.91 ± 0.67 | 12.60 ± 0.67 | 24.25 ± 3.04 | 1.38 ± 0.26 |
| Freezing: 30 min, milling: 5 min | 3.90 ± 0.27 | 7.36 ± 0.80 | 15.11 ± 6.33 | 1.48 ± 0.68 |
| Freezing: 5 min, milling: 3 min | 4.89 ± 2.83 | 14.37 ± 1.71 | 28.83 ± 1.49 | 1.69 ± 0.33 |
| Formulation | DD (%) | FPF (%) **b | MMAD (µm) |
|---|---|---|---|
| Freezing: 30 min, milling: 1 min | 78.4 ± 3.6 *a | 16.3 ± 1.3 | 9.7 ± 1.0 |
| Freezing: 30 min, milling: 3 min | 81.8 ± 0.4 | 26.5 ± 0.8 | 5.9 ± 3.4 |
| Freezing: 30 min, milling: 5 min | 83.8 ± 2.8 | 10.7 ± 1.2 | 11.0 < |
| Freezing: 5 min, milling: 3 min | 88.9 ± 3.9 | 0.9 ± 0.3 | 11.0 < |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ito, T.; Yamazoe, E.; Tahara, K. Dry Powder Inhalers for Proteins Using Cryo-Milled Electrospun Polyvinyl Alcohol Nanofiber Mats. Molecules 2022, 27, 5158. https://doi.org/10.3390/molecules27165158
Ito T, Yamazoe E, Tahara K. Dry Powder Inhalers for Proteins Using Cryo-Milled Electrospun Polyvinyl Alcohol Nanofiber Mats. Molecules. 2022; 27(16):5158. https://doi.org/10.3390/molecules27165158
Chicago/Turabian StyleIto, Takaaki, Eriko Yamazoe, and Kohei Tahara. 2022. "Dry Powder Inhalers for Proteins Using Cryo-Milled Electrospun Polyvinyl Alcohol Nanofiber Mats" Molecules 27, no. 16: 5158. https://doi.org/10.3390/molecules27165158
APA StyleIto, T., Yamazoe, E., & Tahara, K. (2022). Dry Powder Inhalers for Proteins Using Cryo-Milled Electrospun Polyvinyl Alcohol Nanofiber Mats. Molecules, 27(16), 5158. https://doi.org/10.3390/molecules27165158