Light and Potassium Improve the Quality of Dendrobium officinale through Optimizing Transcriptomic and Metabolomic Alteration
Abstract
:1. Introduction
2. Results
2.1. Phenotypic Changes in Light- or K-Treated D. officinale Pseudobulb
2.2. Metabolomic Changes in of Light- or K-Treated D. officinale Pseudobulb
2.3. Transcriptomic Changes in Light- or K-Treated D. officinale Pseudobulb
2.4. Identification of Key Genes Correlated to Flavonoid Biosynthesis via Coexpression Analysis
2.5. Gene-Metabolite Correlations Reveal Significant Interactions between Flavonoid-Related DEGs and DAMs
2.6. The Effect of JA on the Synthesis of Anthocyanins in D. officinale Pseudobulbs
3. Discussion
3.1. Light and K Regulate the Biosynthesis of Flavonoids in D. officinale
3.2. JA Promoted the Synthesis of Anthocyanin Which Contributed for the Purple Stem of D. officinale
4. Materials and Methods
4.1. Plant Materials and Treatments
4.2. Metabolome Analyses
4.3. RNA Extraction, Library Construction, and Sequencing
4.4. Transcriptome Analyses
4.5. qRT-PCR Analysis
4.6. KEGG Enrichment Analysis of DEGs and Gene Coexpression Analysis
4.7. Transcriptome and Metabolome Joint Analysis
4.8. Determination of the Total Anthocyanin Content and Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Hou, B.; Tian, M.; Luo, J.; Ji, Y.; Xue, Q.; Ding, X. Genetic diversity assessment and ex situ conservation strategy of the endangered Dendrobium officinale (Orchidaceae) using new trinucleotide microsatellite markers. Plant Syst. Evol. 2012, 298, 1483–1491. [Google Scholar] [CrossRef]
- Hou, B.; Luo, J.; Zhang, Y.; Niu, Z.; Xue, Q.; Ding, X. Iteration expansion and regional evolution: Phylogeography of Dendrobium officinale and four related taxa in southern China. Sci. Rep. 2017, 7, 43525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, J.; Dang, P.; Zhao, Z.; Yuan, L.; Zhou, Z.; Wolf, D.; Luo, Y. An assessment of the Chinese medicinal Dendrobium industry: Supply, demand and sustainability. J. Ethnopharmacol. 2019, 229, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Xu, Q.; Bian, C.; Tsai, W.; Yeh, C.; Liu, K.; Yoshida, K.; Zhang, L. The Dendrobium catenatum Lindl. genome sequence provides insights into polysaccharide synthase, floral development and adaptive evolution. Sci. Rep. 2016, 6, 19029. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; He, T.; Cuan, X.; Wang, X.; Hu, J.; Sheng, J. 1,4-beta-d-glucomannan from Dendrobium officinale activates NF-small ka, CyrillicB via TLR4 to regulate the immune response. Molecules 2018, 23, 2685. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Yue, H.; Wang, Y.; Guo, C.; Du, Z.; Jin, C.; Kan, D. Intestinal microbes derived butyrate is related to the immunomodulatory activities of Dendrobium officinale polysaccharide. Int. J. Biol. Macromol. 2020, 149, 717–723. [Google Scholar] [CrossRef]
- Liang, C.; Liang, Y.; Liu, H.; Zhu, D.; Hou, S.; Wu, Y.; Huang, S.; Lai, X. Effect of Dendrobium officinale on D-galactose-induced aging mice. Chin. J. Integr. Med. 2017, 1–9. [Google Scholar] [CrossRef]
- Zhao, M.; Sun, Y.; Gao, Z.; Cui, H.; Chen, J.; Wang, M.; Wang, Z. Gigantol attenuates the metastasis of human bladder cancer cells, possibly through Wnt/EMT signaling. OncoTargets Ther. 2020, 13, 11337–11346. [Google Scholar] [CrossRef]
- Guo, Z.; Zhou, Y.; Yang, J.; Shao, X. Dendrobium candidum extract inhibits proliferation and induces apoptosis of liver cancer cells by inactivating Wnt/beta-catenin signaling pathway. Biomed. Pharmacother. 2019, 110, 371–379. [Google Scholar] [CrossRef]
- Zeng, J.; Li, D.; Li, Z.; Zhang, J.; Zhao, X. Dendrobium officinale attenuates myocardial fibrosis via inhibiting EMT signaling pathway in HFD/STZ-induced diabetic mice. Biol. Pharm. Bull. 2020, 43, 864–872. [Google Scholar] [CrossRef]
- Zheng, H.; Pan, L.; Xu, P.; Zhu, J.; Wang, R.; Zhu, W.; Hu, Y.; Gao, H. An NMR-based metabolomic approach to unravel the preventive effect of water-soluble extract from Dendrobium officinale Kimura et Migo on streptozotocin-induced diabetes in mice. Molecules 2017, 22, 1543. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Liang, J.; Zhong, Y.; Xiao, G.; Efferth, T.; Georgiev, M.I.; Vargas-De-La-Cruz, C.; Bajpai, V.K.; Caprioli, G.; Liu, J.; et al. Dendrobium officinale Polysaccharide Alleviates Intestinal Inflammation by Promoting Small Extracellular Vesicle Packaging of miR-433–3p. J. Agric. Food Chem. 2021, 69, 13510–13523. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Z.; Liu, J.; Zheng, Z.; Li, Q.; Wang, H.; Chen, Z.; Wang, K. Identification of the core active structure of a Dendrobium officinale polysaccharide and its protective effect against dextran sulfate sodium-induced colitis via alleviating gut microbiota dysbiosis. Food Res. Int. 2020, 137, 109641. [Google Scholar] [CrossRef]
- Yue, H.; Liu, Y.; Qu, H.; Ding, K. Structure analysis of a novel heteroxylan from the stem of Dendrobium officinale and anti-angiogenesis activities of its sulfated derivative. Int. J. Bioll. Macromol. 2017, 103, 533–542. [Google Scholar] [CrossRef]
- Chen, W.; Lu, J.; Zhang, J.; Wu, J.; Yu, L.; Qin, L.; Zhu, B. Traditional Uses, Phytochemistry, Pharmacology, and Quality Control of Dendrobium officinale Kimura et. Migo. Front. Pharmacol. 2021, 12, 726528. [Google Scholar] [CrossRef]
- Hu, J.; Huang, W.; Zhang, F.; Luo, X.; Chen, Y.; Xie, J. Variability of volatile compounds in the medicinal plant Dendrobium officinale from different regions. Molecules 2020, 25, 5046. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, W.; Liu, Y.; Meng, X.; Su, X.; Cao, M.; Wu, L.; Yu, N.; Xing, S.; Peng, D. Putative genes in alkaloid biosynthesis identified in Dendrobium officinale by correlating the contents of major bioactive metabolites with genes expression between protocorm-like bodies and leaves. BMC Genom. 2021, 22, 579. [Google Scholar] [CrossRef]
- Ma, X.; Meng, Y.; Wang, P.; Tang, Z.; Wang, H.; Xie, T. Bioinformatics-assisted, integrated omics studies on medicinal plants. Brief. Bioinform. 2019, 21, 1857–1874. [Google Scholar] [CrossRef]
- Scossa, F.; Benina, M.; Alseekh, S.; Zhang, Y.; Fernie, A. The integration of metabolomics and Next-Generation Sequencing Data to elucidate the pathways of natural product metabolism in medicinal plants. Planta Med. 2018, 84, 855–873. [Google Scholar] [CrossRef] [Green Version]
- Yan, L.; Wang, X.; Liu, H.; Tian, Y.; Lian, J.; Yang, R.; Hao, S.; Wang, X.; Yang, S.; Li, Q.; et al. The genome of Dendrobium officinale illuminates the biology of the important traditional Chinese orchid herb. Mol. Plant. 2015, 8, 922–934. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; He, C.; Wu, K. Transcriptome analysis of Dendrobium officinale and its application to the identification of genes associated with polysaccharide synthesis. Front. Plant. Sci 2016, 7, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wan, R.; Sun, J.; He, T.; Hu, Y.; Zhao, Y.; Wu, Y.; Chun, Z. Cloning cDNA and functional characterization of UDP-glucose pyrophosphorylase in Dendrobium officinale. Biol. Plant. 2017, 61, 147–154. [Google Scholar] [CrossRef]
- Shen, C.; Guo, H.; Chen, H.; Shi, Y.; Meng, Y.; Lu, J.; Feng, S.; Wang, H. Identification and analysis of genes associated with the synthesis of bioactive constituents in Dendrobium officinale using RNA-Seq. Sci. Rep. 2017, 7, 187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, X.; Li, Y.; Li, C.; Luo, H.; Wang, L.; Qian, J.; Luo, X.; Xiang, L.; Song, J.; Sun, C.; et al. Analysis of the Dendrobium officinale transcriptome reveals putative alkaloid biosynthetic genes and genetic markers. Gene 2013, 527, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, Y.; Lyu, P.; Chen, L.; Shen, C.; Sun, C. Comparative transcriptomic analysis reveal the regulation mechanism underlying MeJA-induced accumulation of alkaloids in Dendrobium officinale. J. Plant Res. 2019, 132, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, S.; Kohler, A.; Yan, B.; Luo, H.; Chen, X.; Guo, S. iTRAQ and RNA-Seq analyses provide new insights into regulation mechanism of symbiotic germination of Dendrobium officinale Seeds (Orchidaceae). J. Proteome Res. 2017, 16, 2174–2187. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Liu, X.; Teixeira, D.; Liu, N.; Zhang, M.; Duan, J. Transcriptome sequencing and metabolite profiling analyses provide comprehensive insight into molecular mechanisms of flower development in Dendrobium officinale (Orchidaceae). Plant Mol. Biol. 2020, 104, 529–548. [Google Scholar] [CrossRef]
- Chen, Y.; Shen, Q.; Lin, R.; Zhao, Z.; Shen, C.; Sun, C. De novo transcriptome analysis in Dendrobium and identification of critical genes associated with flowering. Plant Physiol. Biochem. 2017, 119, 319–327. [Google Scholar] [CrossRef]
- Wu, Z.; Jiang, W.; Chen, S.; Mantri, N.; Tao, Z.; Jiang, C. Insights from the cold transcriptome and metabolome of Dendrobium officinale: Global reprogramming of metabolic and gene regulation networks during cold acclimation. Front. Plant Sci. 2016, 7, 1653. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Yu, Z.; Zeng, D.; Si, C.; Zhao, C.; Wang, H.; Li, C.; He, C.; Duan, J. Transcriptome and metabolome reveal salt-stress responses of leaf tissues from Dendrobium officinale. Biomolecules 2021, 11, 736. [Google Scholar] [CrossRef]
- Jiang, W.; Wu, Z.; Wang, T.; Mantri, N.; Huang, H.; Li, H.; Tao, Z.; Guo, Q. Physiological and transcriptomic analyses of cadmium stress response in Dendrobium officinale seedling. Plant Physiol. Biochem. 2020, 148, 152–165. [Google Scholar] [CrossRef]
- Cao, H.; Ji, Y.; Li, S.; Lu, L.; Tian, M.; Yang, W.; Li, H. Extensive metabolic profiles of leaves and stems from the medicinal plant Dendrobium officinale Kimura et Migo. Metabolites 2019, 9, 215. [Google Scholar] [CrossRef] [Green Version]
- Meng, Y.; Yu, D.; Xue, J.; Lu, J.; Feng, S.; Shen, C.; Wang, H. A transcriptome-wide, organ-specific regulatory map of Dendrobium officinale, an important traditional Chinese orchid herb. Sci. Rep. 2016, 6, 18864. [Google Scholar] [CrossRef] [Green Version]
- Yu, Z.; Liao, Y.; Teixeira, D.; Yang, Z.; Duan, J. Differential accumulation of anthocyanins in Dendrobium officinale stems with red and green peels. Int. J. Mol. Sci. 2018, 19, 2857. [Google Scholar] [CrossRef] [Green Version]
- Zhan, X.; Qi, J.; Zhou, B.; Mao, B. Metabolomic and transcriptomic analyses reveal the regulation of pigmentation in the purple variety of Dendrobium officinale. Sci. Rep. 2020, 10, 17700. [Google Scholar] [CrossRef]
- Wink, M. Modes of action of herbal medicines and plant secondary metabolites. Medicines 2015, 2, 251–286. [Google Scholar] [CrossRef]
- Li, Y.; Kong, D.; Fu, Y.; Sussman, M.R.; Wu, H. The effect of developmental and environmental factors on secondary metabolites in medicinal plants. Plant Physiol. Biochem. 2020, 148, 80–89. [Google Scholar] [CrossRef]
- Suh, D.; Kim, Y.; Jung, E.; Lee, S.; Park, J.; Lee, C.; Sung, J. Characterization of metabolic changes under low mineral supply (N, K, or Mg) and supplemental LED lighting (red, blue, or red-blue combination) in Perilla frutescens using a metabolomics approach. Molecules 2020, 25, 4714. [Google Scholar] [CrossRef]
- Pettigrew, W. Potassium influences on yield and quality production for maize, wheat, soybean and cotton. Physiol. Plant. 2008, 133, 670–681. [Google Scholar] [CrossRef]
- Ajewole, E.; Santamaria-Kisiel, L.; Pajak, A.; Jaskolski, M.; Marsolais, F. Structural basis of potassium activation in plant asparaginases. Febs. J. 2018, 285, 1528–1539. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, M.; Jaafar, H.; Karimi, E.; Ghasemzadeh, A. Primary, secondary metabolites, photosynthetic capacity and antioxidant activity of the Malaysian Herb Kacip Fatimah (Labisia Pumila Benth) exposed to potassium fertilization under greenhouse conditions. Int. J. Mol. Sci. 2012, 13, 15321–15342. [Google Scholar] [CrossRef]
- Landi, M.; Zivcak, M.; Sytar, O.; Brestic, M.; Allakhverdiev, S. Plasticity of photosynthetic processes and the accumulation of secondary metabolites in plants in response to monochromatic light environments: A review. Biochim. Biophys. Acta Bioenerg. 2020, 1861, 148131. [Google Scholar] [CrossRef]
- Lobiuc, A.; Vasilache, V.; Pintilie, O.; Stoleru, T.; Burducea, M.; Oroian, M.; Zamfirache, M. Blue and red LED illumination improves growth and bioactive compounds contents in acyanic and cyanic Ocimum basilicum L. microgreens. Molecules 2017, 22, 2111. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Liu, C.; Shi, Q.; Yang, F.; Wei, M. Mixed red and blue light promotes ripening and improves quality of tomato fruit by influencing melatonin content. Environ. Exp. Bot. 2021, 185, 104407. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Song, S.; Su, W.; Hao, Y.; Liu, H. Supplementary red light results in the earlier ripening of tomato fruit depending on ethylene production. Environ. Exp. Bot. 2020, 175, 104044. [Google Scholar] [CrossRef]
- Zou, L.; Zhong, G.; Wu, B.; Yang, Y.; Li, S.; Liang, Z. Effects of sunlight on anthocyanin accumulation and associated co-expression gene networks in developing grape berries. Environ. Exp. Bot. 2019, 166, 103811. [Google Scholar] [CrossRef]
- Zhong, Y.; Wang, L.; Ma, Z.; Du, X. Physiological responses and transcriptome analysis of Spirodela polyrhiza under red, blue, and white light. Planta 2021, 255, 11. [Google Scholar] [CrossRef] [PubMed]
- Giedrė, S.; Akvilė, V.; Aušra, B.; Julė, J.; Sandra, S.; Viktorija, V.; Algirdas, N.; Alina, V.; Audrius, S.; Pavelas, D. Blue light dosage affects carotenoids and tocopherols in microgreens. Food Chem. 2017, 228, 50–56. [Google Scholar]
- Naveed, A.; Abdur, R.; Nisar, A. Light-induced biochemical variations in secondary metabolite production and antioxidant activity in callus cultures of Stevia rebaudiana (Bert). J. Photochem. Photobiol. B 2016, 154, 51–56. [Google Scholar]
- Wu, Q.; Zhu, X.; Zhao, X.; Shen, R. Potassium affects cadmium resistance in Arabidopsis through facilitating root cell wall Cd retention in a nitric oxide dependent manner. Environ. Exp. Bot. 2020, 178, 104175. [Google Scholar] [CrossRef]
- Ozlem, Y.; Kadriye, K.; Rengin, O.; Baris, U.; Ismail, T.; Levent, O. Growth performance and antioxidative response in bread and durum wheat plants grown with varied potassium treatments under ambient and elevated carbon dioxide. Environ. Exp. Bot. 2017, 137, 26–35. [Google Scholar]
- Rizwan, Z.; Haoran, D.; Muhammad, A.; Zhao, W.; Wang, Y.; Zhou, Z. Potassium fertilizer improves drought stress alleviation potential in cotton by enhancing photosynthesis and carbohydrate metabolism. Environ. Exp. Bot. 2017, 137, 73–83. [Google Scholar]
- Elisa, P.; Enrico, B.; Marco, Z.; Carlo, P.; Alberto, B.; Sonia, P.; Angelo, V. Plant flavonoids—Biosynthesis, transport and involvement in stress responses. Int. J. Mol. Sci. 2013, 14, 14950–14973. [Google Scholar]
- Zhang, A.; Zheng, J.; Chen, X.; Shi, X.; Wang, H.; Fu, Q. Comprehensive analysis of transcriptome and metabolome reveals the flavonoid metabolic pathway is associated with fruit peel coloration of melon. Molecules 2021, 26, 2830. [Google Scholar] [CrossRef]
- Zakaryan, H.; Arabyan, E.; Oo, A.; Zandi, K. Flavonoids: Promising natural compounds against viral infections. Arch. Virol. 2017, 162, 2539–2551. [Google Scholar] [CrossRef]
- Wen, K.; Fang, X.; Yang, J.; Yao, Y.; Nandakumar, K.; Salem, M.; Cheng, K. Recent research on flavonoids and their biomedical applications. Curr. Med. Chem. 2021, 28, 1042–1066. [Google Scholar] [CrossRef]
- Loreti, E.; Povero, G.; Novi, G.; Solfanelli, C.; Alpi, A.; Perata, P. Gibberellins, jasmonate and abscisic acid modulate the sucrose-induced expression of anthocyanin biosynthetic genes in Arabidopsis. New Phytol. 2008, 179, 1004–1016. [Google Scholar] [CrossRef]
- Jiang, Y.; Daryl, C. ABA effects on ethylene production, PAL activity, anthocyanin and phenolic contents of strawberry fruit. J. Plant. Growth Regul. 2003, 39, 171–174. [Google Scholar] [CrossRef]
- Perez, A.G.; Sanz, C.; Olias, R.; Olias, J.M. Effect of methyl jasmonate on in vitro strawberry ripening. J. Agric. Food Chem. 1997, 45, 3733–3737. [Google Scholar] [CrossRef]
- Zhong, C.; Tang, Y.; Pang, B.; Li, X.; Yang, Y.; Deng, J.; Feng, C.; Li, L.; Ren, G.; Wang, Y.; et al. The R2R3-MYB transcription factor GhMYB1a regulates flavonol and anthocyanin accumulation in Gerbera hybrida. Hortic. Res. 2020, 7, 78. [Google Scholar] [CrossRef]
- Li, H.; Li, Y.; Yu, J.; Wu, T.; Zhang, J.; Tian, J.; Yao, Y. MdMYB8 is associated with flavonol biosynthesis via the activation of the MdFLS promoter in the fruits of Malus crabapple. Hortic. Res. 2020, 7, 19. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Yang, Z.; Zeng, Q.; Wang, S.; Luo, Y.; Huang, Y.; Xin, Y.; He, N. Abnormal expression of bHLH3 disrupts a flavonoid homeostasis network, causing differences in pigment composition among mulberry fruits. Hortic. Res. 2020, 7, 83. [Google Scholar] [CrossRef]
- Yang, M.; Yang, J.; Su, L.; Sun, K.; Li, D.; Liu, Y.; Wang, H.; Chen, Z.; Guo, T. Metabolic profile analysis and identification of key metabolites during rice seed germination under low-temperature stress. Plant Sci. 2019, 289, 110282. [Google Scholar] [CrossRef]
- Sun, T.; Zhang, J.; Zhang, Q.; Li, X.; Li, M.; Yang, Y.; Zhou, J.; Wei, Q.; Zhou, B. Integrative physiological, transcriptome, and metabolome analysis reveals the effects of nitrogen sufficiency and deficiency conditions in apple leaves and roots. Environ. Exp. Bot. 2021, 192, 104633. [Google Scholar] [CrossRef]
- Guang, Y.; Kunnan, L.; Zaizhi, Z.; Xiyang, W.; Guihua, H. UPLC-ESI-MS/MS-based widely targeted metabolomics analysis of wood metabolites in teak (Tectona grandis). Molecules 2020, 25, 2189. [Google Scholar]
- Guo, X.; Shakeel, M.; Wang, D.; Qu, C.; Yang, S.; Ahmad, S.; Song, Z. Metabolome and transcriptome profiling unveil the mechanisms of light-induced anthocyanin synthesis in rabbiteye blueberry (vaccinium ashei: Reade). BMC Plant Biol. 2022, 22, 223. [Google Scholar] [CrossRef]
- Li, M.; Liu, J.; Zhou, Y.; Zhou, S.; Zhang, S.; Tong, H.; Zhao, A. Transcriptome and metabolome profiling unveiled mechanisms of tea (Camellia sinensis) quality improvement by moderate drought on pre-harvest shoots. Phytochemistry 2020, 180, 112515. [Google Scholar] [CrossRef]
- Zheng, T.; Han, J.; Su, K.; Sun, B.; Liu, S. Regulation mechanisms of flavonoids biosynthesis of Hancheng Dahongpao peels (Zanthoxylum bungeanum Maxim) at different development stages by integrated metabolomics and transcriptomics analysis. BMC Plant Biol. 2022, 22, 251. [Google Scholar] [CrossRef]
- Jiang, T.; Zhang, M.; Wen, C.; Xie, X.; Tian, W.; Wen, S.; Lu, R.; Liu, L. Integrated metabolomic and transcriptomic analysis of the anthocyanin regulatory networks in Salvia miltiorrhiza Bge. Flowers. BMC Plant Biol. 2020, 20, 349. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Zhang, M.; Su, H.; Li, M.; Wang, Y.; Jin, L.; Li, M. Integrated metabolomic and transcriptomic analysis reveals differential mechanism of flavonoid biosynthesis in two cultivars of Angelica sinensis. Molecules 2022, 27, 306. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Shen, G.; Yang, Y.; Li, C.; Chen, X.; Yang, X.; Guo, X.; Miao, J.; Li, L.; Lei, M. Metabolic and transcriptomic analyses reveal the effects of ethephon on Taraxacum kok-saghyz Rodin. Molecules 2022, 27, 3548. [Google Scholar] [CrossRef] [PubMed]
- Trapnell, C.; Roberts, A.; Goff, L.; Pertea, G.; Kim, D.; Kelley, D.; Pimentel, H.; Salzberg, S.; Rinn, J.; Pachter, L. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 2012, 7, 562–578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, X.; Cai, T.; Olyarchuk, J.; Wei, L. Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 2005, 21, 3787–3793. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Dong, X.; Li, Q.; Zhou, X.; Gao, S.; Chen, R.; Sun, L.; Zhang, L.; Chen, W. Biosynthesis of the active compounds of Isatis indigotica based on transcriptome sequencing and metabolites profiling. BMC Genom. 2013, 14, 857. [Google Scholar] [CrossRef] [Green Version]
- Yang, T.; Li, K.; Hao, S.; Zhang, J.; Song, T.; Tian, J.; Yao, Y. The Use of RNA Sequencing and Correlation Network Analysis to Study Potential Regulators of Crabapple Leaf Color Transformation. Plant Cell Physiol. 2018, 59, 1027–1042. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Tan, M.; Cheng, D.; Yang, Y.; Zhang, G.; Qin, M.; Chen, J.; Chen, Y.; Jiang, M. Co-expression network analysis of the transcriptomes of rice roots exposed to various cadmium stresses reveals universal cadmium-responsive genes. BMC Plant Biol. 2017, 17, 194. [Google Scholar] [CrossRef]
- Wei, R.; Tu, D.; Huang, X.; Luo, Z.; Huang, X.; Cui, N.; Xu, J.; Xiong, F.; Yan, H.; Ma, X. Genome-scale transcriptomic insights into the gene co-expression network of seed abortion in triploid Siraitia grosvenorii. BMC Plant Biol. 2022, 22, 173. [Google Scholar] [CrossRef]
- Zhou, C.; Mei, X.; Rothenberg, D.O.; Yang, Z.; Zhang, W.; Wan, S.; Yang, H.; Zhang, L. Metabolome and transcriptome analysis reveals putative genes involved in anthocyanin accumulation and coloration in white and pink tea (Camellia sinensis) flower. Molecules 2020, 25, 190. [Google Scholar] [CrossRef] [Green Version]
- Smoot, M.; Ono, K.; Ruscheinski, J.; Wang, P.; Ideker, T. Cytoscape 2.8: New features for data integration and network visualization. Bioinformatics 2011, 27, 431–432. [Google Scholar] [CrossRef] [Green Version]
- Premathilake, A.T.; Ni, J.; Shen, J.; Bai, S.; Teng, Y. Transcriptome analysis provides new insights into the transcriptional regulation of methyl jasmonate-induced flavonoid biosynthesis in pear calli. BMC Plant Biol. 2020, 20, 388. [Google Scholar] [CrossRef]
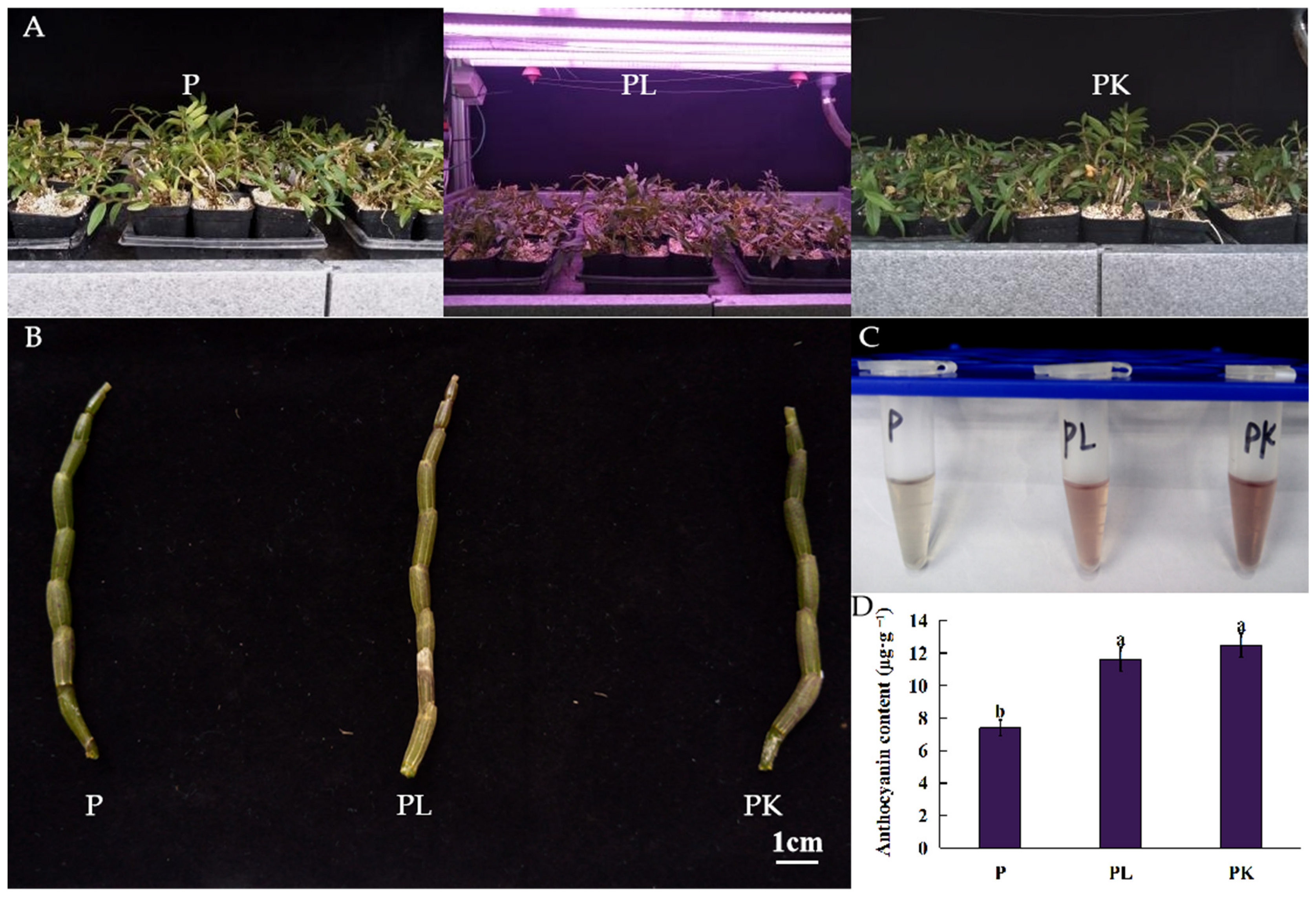
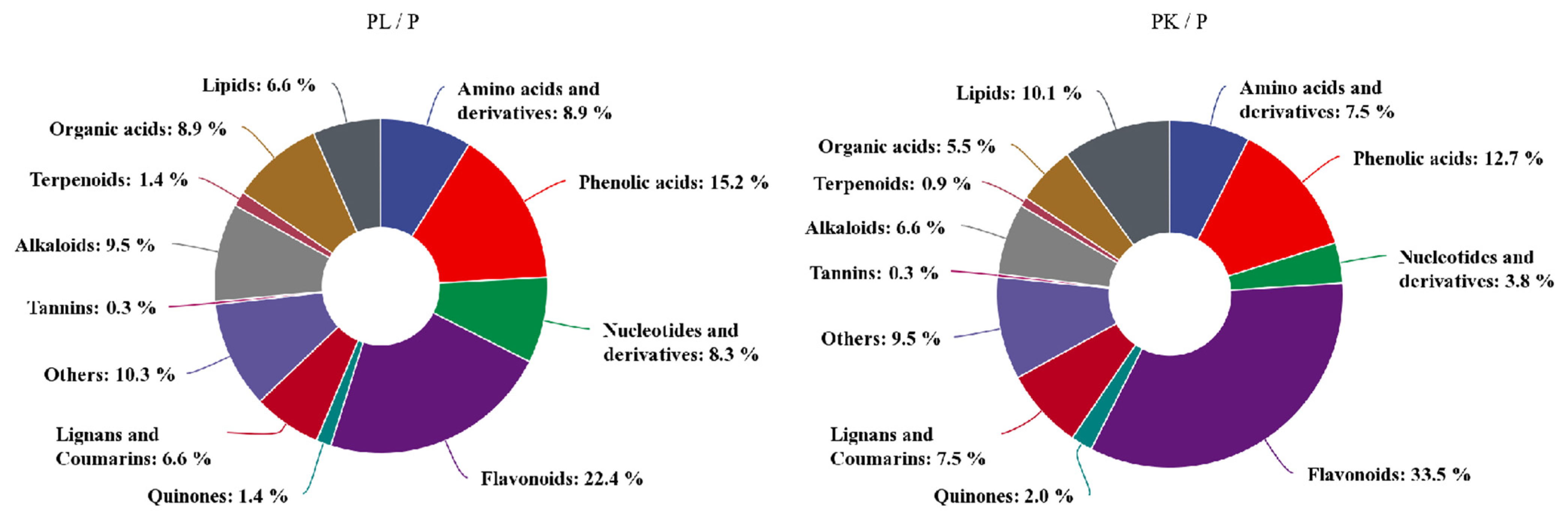

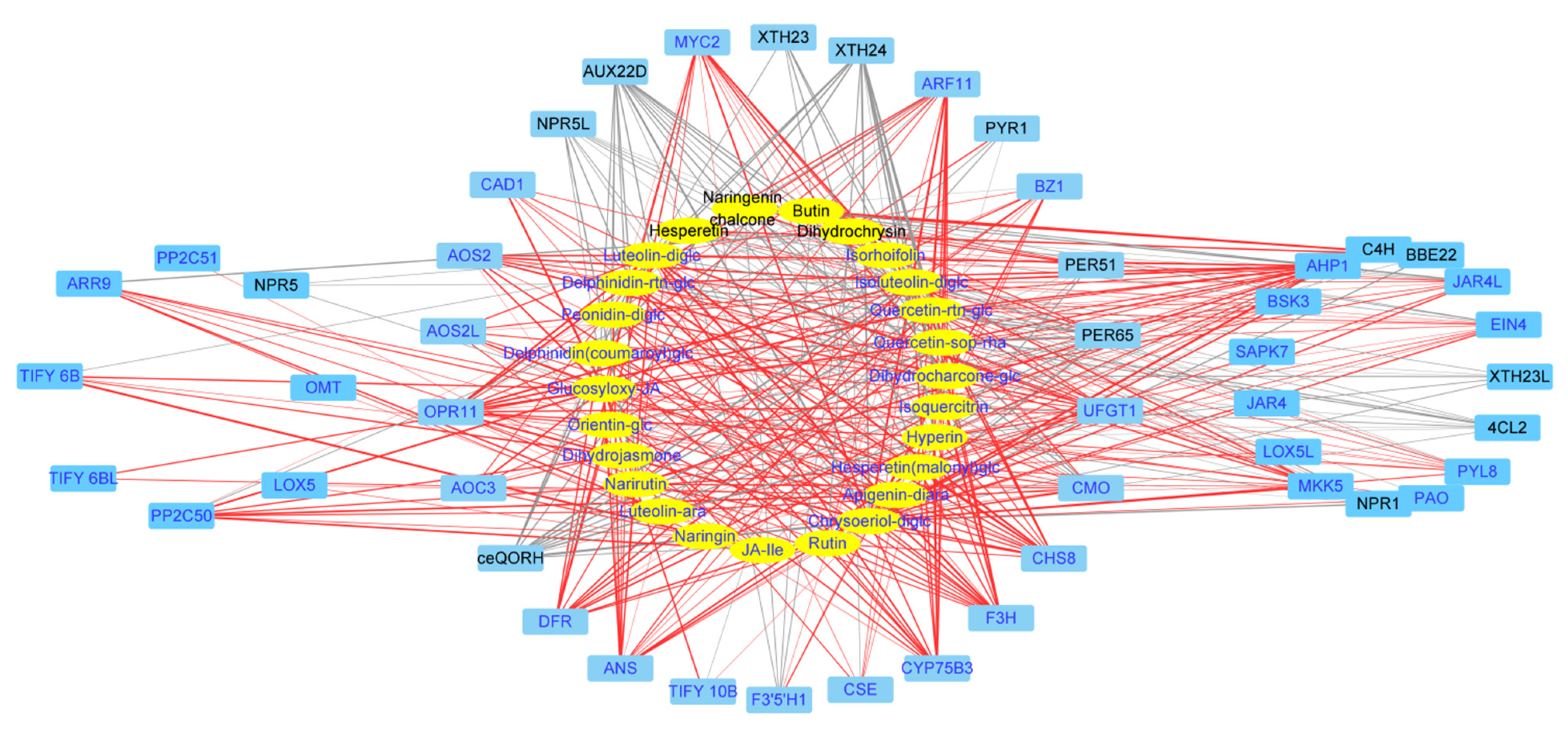

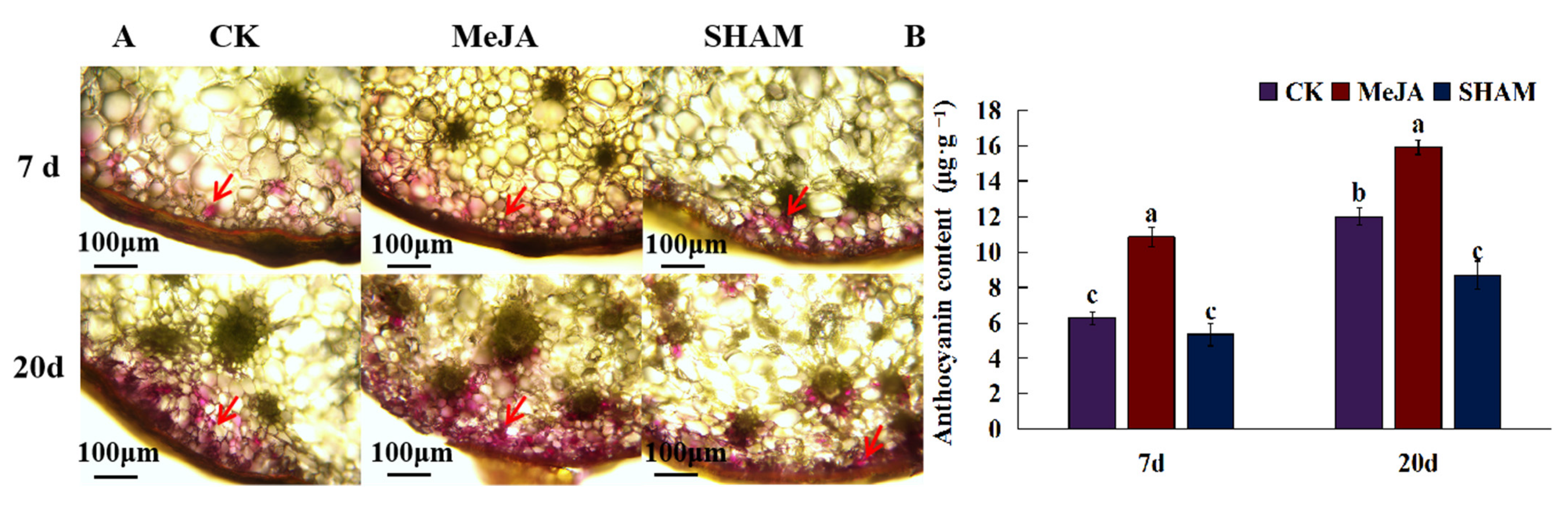
| Compounds | CAS | Log2FC | Compounds | CAS | Log2FC | ||
|---|---|---|---|---|---|---|---|
| PL/P | PK/P | PL/P | PK/P | ||||
| Flavonoids | |||||||
| Butin | 492-14-8 | −0.92 | −1.35 | Pinobanksin | 548-82-3 | −0.88 | −1.17 |
| Naringenin chalcone | 73,692-50-9 | −0.95 | −1.43 | Hesperetin | 520-33-2 | −0.62 | −1.52 |
| Genistein | 446-72-0 | −0.93 | −1.40 | Epigallocatechin-gallate | 989-51-5 | −1.13 | −1.66 |
| Dihydrochrysin | 480-39-7 | −1.86 | −1.11 | (-)-Gallocatechin gallate | 4233-96-9 | −1.12 | −2.03 |
| Fisetin | 528-48-3 | −0.70 | −0.67 | ||||
| Narirutin | 14,259-46-2 | 0.73 | 0.90 | Quercetin-glc-rha | - | 0.88 | 1.97 |
| Naringin | 10,236-47-2 | 0.75 | 0.68 | Isoquercitrin | 482-35-9 | 1.06 | 2.23 |
| Isoluteolin-diglc | - | 1.34 | 1.89 | Quercetin-sop-rha | - | 0.89 | 1.54 |
| Diosmetin-glc | - | 0.74 | 1.40 | Hyperin | 482-36-0 | 0.90 | 2.25 |
| Isorhoifolin | 552-57-8 | 0.65 | 0.89 | Quercetin-rtn | 147,714-62-3 | 0.92 | 2.04 |
| Apigenin-diara | - | 2.07 | 1.15 | Tricin (sinapoyl) glc | - | 0.73 | 0.63 |
| Orientin-glc | - | 1.15 | 2.12 | Dihydrocharcone-glc | - | 0.78 | 1.14 |
| Luteolin-ara | - | 0.78 | 0.66 | Hesperetin (malonyl) glc | - | 1.08 | 1.11 |
| Luteolin-diglc | 29,428-58-8 | 1.56 | 1.98 | HydroxyKaempferol-diglc | 142,674-16-6 | 0.98 | 1.16 |
| Astilbin | 29,838-67-3 | 0.77 | 0.68 | Hydroxykaempferol-rtn-glc | - | 0.93 | 1.66 |
| Chrysoeriol-rtn | - | 0.69 | 1.56 | Hydroxykaempferol-glc | - | 1.37 | 2.45 |
| Chrysoeriol-diglc | - | 0.61 | 1.02 | Malonylgenistin | 51,011-05-3 | 0.70 | 1.03 |
| Quercetin-rtn-glc | - | 1.12 | 1.77 | Peonidin-diglc | 47,851-83-2 | 0.72 | 0.88 |
| Rutin | 153-18-4 | 0.86 | 1.69 | Delphinidin-rtn-glc | - | 1.40 | 1.91 |
| Quercetin-rob | 52,525-35-6 | 0.85 | 1.60 | Delphinidin (coumaroyl) glc | - | 0.98 | 1.96 |
| JA and derivatives | |||||||
| JA | 77,026-92-7 | 0.69 | - | 5′-Glucosyloxyjasmanic acid | 1.83 | 1.35 | |
| JA-Ile | 120,330-93-0 | 2.26 | 0.94 | Dihydrojasmone | 1128-08-1 | 1.44 | 0.73 |
| Cis-Jasmone | 488-10-8 | 0.72 | - | ||||
| GeneID | Gene | Annotation | Log2FC | p-Value | ||
|---|---|---|---|---|---|---|
| PL/P | PK/P | PL/P | PK/P | |||
| Phenylpropanoid biosynthesis | ||||||
| LOC110097226 | 4CL2 | 4-coumarate-CoA ligase 2 | - | −1.13 | - | 0.00 |
| LOC110093998 | BBE22 | berberine bridge enzyme-like 22 | - | −0.70 | - | 0.00 |
| LOC110113575 | C4H | trans-cinnamate 4-monooxygenase | - | −1.47 | - | 0.00 |
| LOC110102215 | CSE | caffeoylshikimate esterase | 1.57 | 0.99 | 0.00 | 0.03 |
| LOC110100929 | OMT | Tricetin 3′,4′,5′-O-trimethyltransferase | 0.62 | - | 0.00 | - |
| LOC110097445 | PER51 | peroxidase 51-like | −2.55 | −2.07 | 0.00 | 0.00 |
| LOC110095178 | PER65 | peroxidase 65 | −0.58 | −1.22 | 0.02 | 0.00 |
| LOC110095989 | UFGT1 | glucosyltransferase | 0.77 | 1.29 | 0.04 | 0.00 |
| Flavonoid biosynthesis | ||||||
| LOC110100597 | BZ1 | anthocyanidin 3-O-glucosyltransferase | 1.69 | 1.72 | 0.00 | 0.00 |
| LOC110103723 | ANS | anthocyanidin synthase | 2.40 | 1.91 | 0.00 | 0.00 |
| LOC110113809 | CHS8 | chalcone synthase 8 | 1.61 | 1.94 | 0.00 | 0.00 |
| LOC110101655 | DFR | dihydroflavonol-4-reductase | 0.88 | 1.79 | 0.10 | 0.00 |
| LOC110097388 | F3H | flavanone 3-hydroxylase | 1.54 | 1.83 | 0.00 | 0.00 |
| LOC110113268 | CYP75B3 | flavonoid 3′-monooxygenase CYP75B3-like | 1.05 | 1.87 | 0.01 | 0.00 |
| LOC110103762 | F3′5′H1 | flavonoid 3′,5′-hydroxylase 1 | 0.94 | 0.88 | 0.05 | 0.00 |
| JA synthesis | ||||||
| LOC110093045 | AOC3 | allene oxide cyclase, chloroplastic-like | 0.71 | 0.53 | 0.00 | 0.04 |
| LOC110103009 | AOS2 | allene oxide synthase2 | 0.87 | 0.91 | 0.00 | 0.00 |
| LOC110108695 | AOS2L | allene oxide synthase 2-like | 0.64 | 0.55 | 0.00 | 0.00 |
| LOC110096989 | OPR11 | Putative 12-oxophytodienoate reductase 11 | 0.86 | 1.34 | 0.00 | 0.00 |
| LOC110108000 | LOX5.1 | lipoxygenase 5.1 | 2.71 | - | 0.06 | - |
| LOC110108011 | LOX5.2 | lipoxygenase 5.2 | - | 0.69 | - | 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, Y.; Liu, J.; Xu, M.; Chen, G.; Tan, M.; Xiang, Z. Light and Potassium Improve the Quality of Dendrobium officinale through Optimizing Transcriptomic and Metabolomic Alteration. Molecules 2022, 27, 4866. https://doi.org/10.3390/molecules27154866
Jia Y, Liu J, Xu M, Chen G, Tan M, Xiang Z. Light and Potassium Improve the Quality of Dendrobium officinale through Optimizing Transcriptomic and Metabolomic Alteration. Molecules. 2022; 27(15):4866. https://doi.org/10.3390/molecules27154866
Chicago/Turabian StyleJia, Yue, Juan Liu, Mengyao Xu, Guihong Chen, Mingpu Tan, and Zengxu Xiang. 2022. "Light and Potassium Improve the Quality of Dendrobium officinale through Optimizing Transcriptomic and Metabolomic Alteration" Molecules 27, no. 15: 4866. https://doi.org/10.3390/molecules27154866
APA StyleJia, Y., Liu, J., Xu, M., Chen, G., Tan, M., & Xiang, Z. (2022). Light and Potassium Improve the Quality of Dendrobium officinale through Optimizing Transcriptomic and Metabolomic Alteration. Molecules, 27(15), 4866. https://doi.org/10.3390/molecules27154866







