In Silico Evaluation of the Antimicrobial Activity of Thymol—Major Compounds in the Essential Oil of Lippia thymoides Mart. & Schauer (Verbenaceae)
Abstract
:1. Introduction
2. Results and Discussions
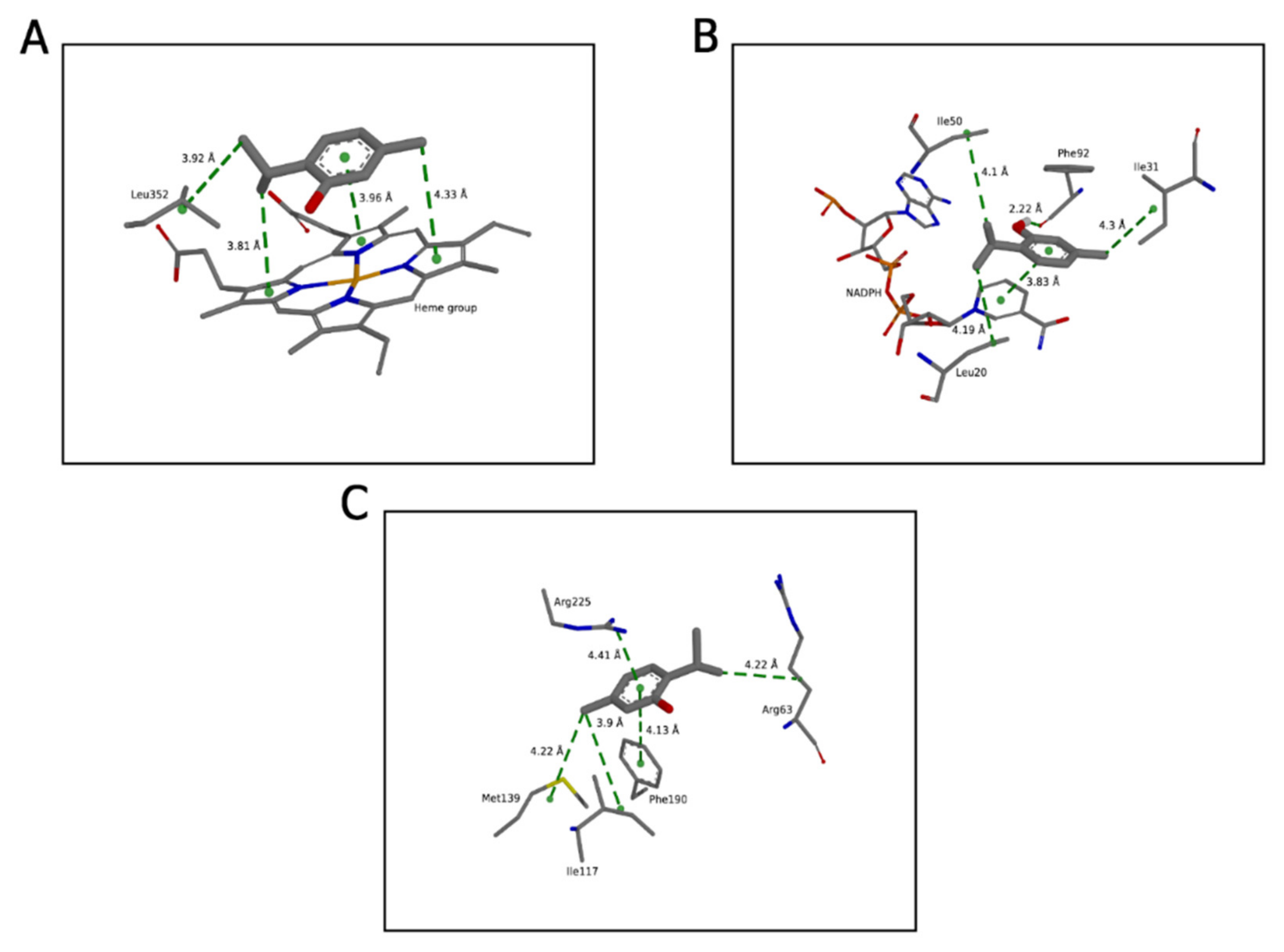
2.1. Molecular Binding Mode
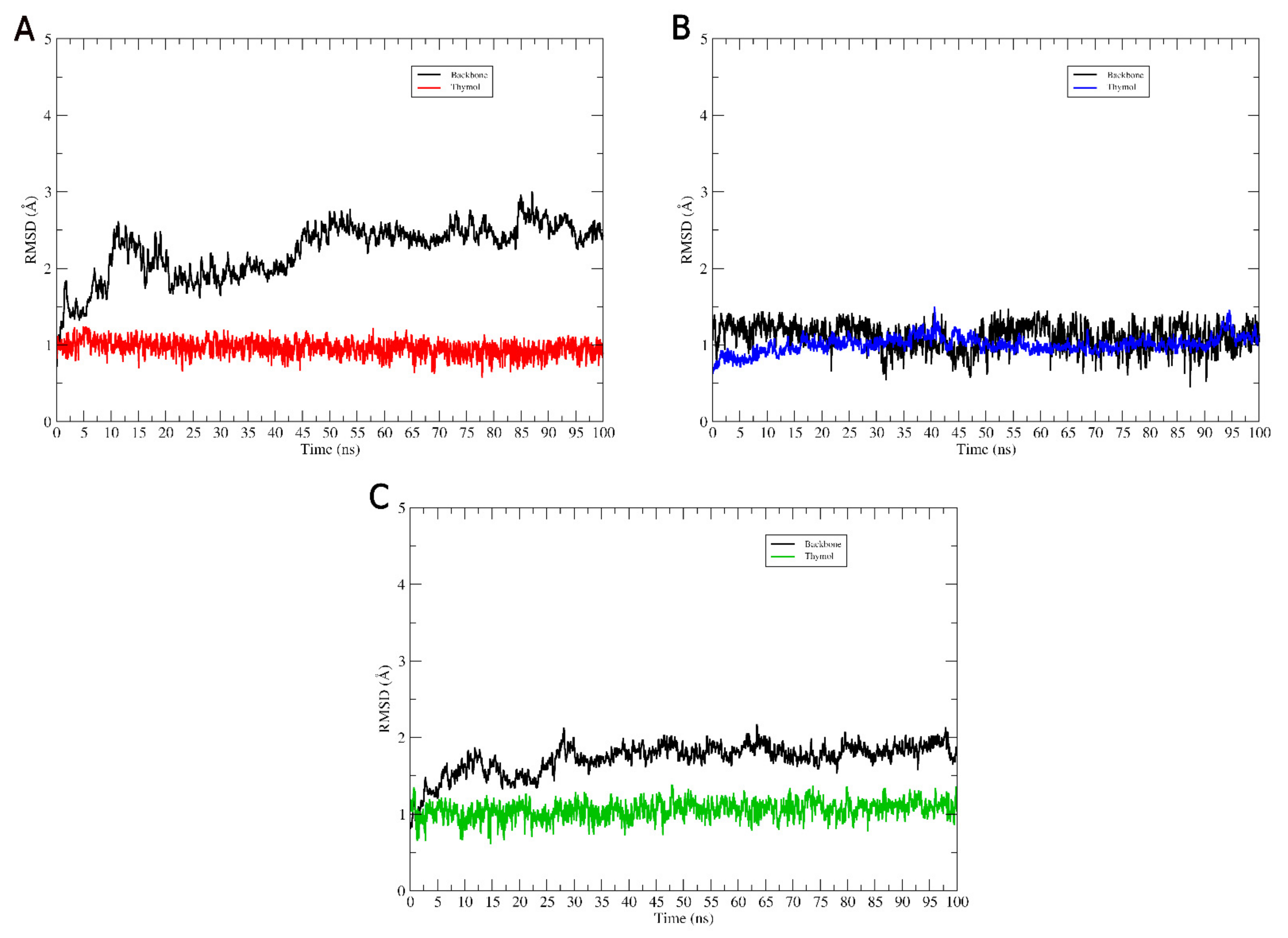
2.2. DM Trajectory Stability
2.3. Free Energy Calculation of the Thymol-Receptor Complexes
3. Materials and Methods
3.1. Molecular Docking
3.2. MD Simulations
3.3. Free Energy Calculations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Ferreira, O.O.; Neves da Cruz, J.; de Jesus Pereira Franco, C.; Silva, S.G.; da Costa, W.A.; de Oliveira, M.S.; de Aguiar Andrade, E.H. First Report on Yield and Chemical Composition of Essential Oil Extracted from Myrcia Eximia DC (Myrtaceae) from the Brazilian Amazon. Molecules 2020, 25, 783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartmann, T. From Waste Products to Ecochemicals: Fifty Years Research of Plant Secondary Metabolism. Phytochemistry 2007, 68, 2831–2846. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G.; Scheffer, J.J.C. Factors Affecting Secondary Metabolite Production in Plants: Volatile Components and Essential Oils. Flavour Fragr. J. 2008, 23, 213–226. [Google Scholar] [CrossRef]
- Ferreira, O.O.; Cruz, J.N.; de Moraes, Â.A.; de Jesus Pereira Franco, C.; Lima, R.R.; Anjos, T.O.; Siqueira, G.M.; Nascimento, L.D.; Cascaes, M.M.; de Oliveira, M.S.; et al. Essential Oil of the Plants Growing in the Brazilian Amazon: Chemical Composition, Antioxidants, and Biological Applications. Molecules 2022, 27, 4373. [Google Scholar] [CrossRef]
- Do Nascimento, L.D.; de Moraes, A.A.B.; da Costa, K.S.; Galúcio, J.M.P.; Taube, P.S.; Costa, C.M.L.; Cruz, J.N.; Andrade, E.H.d.A.; de Faria, L.J.G. Bioactive Natural Compounds and Antioxidant Activity of Essential Oils from Spice Plants: New Findings and Potential Applications. Biomolecules 2020, 10, 988. [Google Scholar] [CrossRef]
- Sodeifian, G.; Sajadian, S.A.; Saadati Ardestani, N. Experimental Optimization and Mathematical Modeling of the Supercritical Fluid Extraction of Essential Oil from Eryngium Billardieri: Application of Simulated Annealing (SA) Algorithm. J. Supercrit. Fluids 2017, 127, 146–157. [Google Scholar] [CrossRef]
- Cascaes, M.M.; Dos, O.; Carneiro, S.; Diniz Do Nascimento, L.; Antônio Barbosa De Moraes, Â.; Santana De Oliveira, M.; Neves Cruz, J.; Skelding, G.M.; Guilhon, P.; Helena De Aguiar Andrade, E.; et al. Essential Oils from Annonaceae Species from Brazil: A Systematic Review of Their Phytochemistry, and Biological Activities. Int. J. Mol. Sci. 2021, 22, 12140. [Google Scholar] [CrossRef]
- Santana de Oliveira, M.; Pereira da Silva, V.M.; Cantão Freitas, L.; Gomes Silva, S.; Nevez Cruz, J.; de Aguiar Andrade, E.H. Extraction Yield, Chemical Composition, Preliminary Toxicity of Bignonia Nocturna (Bignoniaceae) Essential Oil and in Silico Evaluation of the Interaction. Chem. Biodivers. 2021, 18, cbdv.202000982. [Google Scholar] [CrossRef]
- Benavides, S.; Villalobos-Carvajal, R.; Reyes, J.E. Physical, Mechanical and Antibacterial Properties of Alginate Film: Effect of the Crosslinking Degree and Oregano Essential Oil Concentration. J. Food Eng. 2012, 110, 232–239. [Google Scholar] [CrossRef]
- Leal, S.M.; Pino, N.; Stashenko, E.E.; Martínez, J.R.; Escobar, P. Antiprotozoal Activity of Essential Oils Derived from Piper Spp. Grown in Colombia. J. Essent. Oil Res. 2013, 25, 512–519. [Google Scholar] [CrossRef]
- Silva, F.V.; Guimarães, A.G.; Silva, E.R.S.; Sousa-Neto, B.P.; MacHado, F.D.F.; Quintans-Júnior, L.J.; Arcanjo, D.D.R.; Oliveira, F.A.; Oliveira, R.C.M. Anti-Inflammatory and Anti-Ulcer Activities of Carvacrol, a Monoterpene Present in the Essential Oil of Oregano. J. Med. Food 2012, 15, 984–991. [Google Scholar] [CrossRef]
- Da Silva Júnior, O.S.; Franco, C.d.J.P.; de Moraes, A.A.B.; Cruz, J.N.; da Costa, K.S.; do Nascimento, L.D.; Andrade, E.H.d.A. In Silico Analyses of Toxicity of the Major Constituents of Essential Oils from Two Ipomoea L. Species. Toxicon 2021, 195, 111–118. [Google Scholar] [CrossRef]
- Tanrıkulu, G.İ.; Ertürk, Ö.; Yavuz, C.; Can, Z.; Çakır, H.E. Chemical Compositions, Antioxidant and Antimicrobial Activities of the Essential Oil and Extracts of Lamiaceae Family (Ocimum Basilicum and Thymbra Spicata) from Turkey. Int. J. Second. Metab. 2017, 4, 340–348. [Google Scholar] [CrossRef]
- Calo, J.R.; Crandall, P.G.; O’Bryan, C.A.; Ricke, S.C. Essential Oils as Antimicrobials in Food Systems—A Review. Food Control 2015, 54, 111–119. [Google Scholar] [CrossRef]
- Akthar, M.S.; Degaga, B.; Azam, T. Antimicrobial Activity of Essential Oils Extracted from Medicinal Plants against the Pathogenic Microorganisms: A Review. Issues Biol. Sci. Pharm. Res. 2014, 2, 1–7. [Google Scholar]
- Solórzano-Santos, F.; Miranda-Novales, M.G. Essential Oils from Aromatic Herbs as Antimicrobial Agents. Curr. Opin. Biotechnol. 2012, 23, 136–141. [Google Scholar] [CrossRef]
- Bilia, A.R.; Santomauro, F.; Sacco, C.; Bergonzi, M.C.; Donato, R. Essential Oil of Artemisia Annua L.: An Extraordinary Component with Numerous Antimicrobial Properties. Evid. Based Complement. Altern. Med. 2014, 2014, 159819. [Google Scholar] [CrossRef] [Green Version]
- Silva, S.G.; de Oliveira, M.S.; Cruz, J.N.; da Costa, W.A.; da Silva, S.H.M.; Barreto Maia, A.A.; de Sousa, R.L.; Carvalho Junior, R.N.; de Aguiar Andrade, E.H. Supercritical CO2 Extraction to Obtain Lippia Thymoides Mart. & Schauer (Verbenaceae) Essential Oil Rich in Thymol and Evaluation of Its Antimicrobial Activity. J. Supercrit. Fluids 2021, 168, 105064. [Google Scholar] [CrossRef]
- Costa, E.B.B.; Silva, R.C.C.; Espejo-Román, J.M.M.; Neto, M.F.d.A.F.d.A.; Cruz, J.N.N.; Leite, F.H.A.H.A.; Silva, C.H.T.P.H.T.P.; Pinheiro, J.C.C.; Macêdo, W.J.C.J.C.; Santos, C.B.R.B.R. Chemometric Methods in Antimalarial Drug Design from 1,2,4,5-Tetraoxanes Analogues. SAR QSAR Environ. Res. 2020, 31, 677–695. [Google Scholar] [CrossRef]
- Castro, A.L.G.; Cruz, J.N.; Sodré, D.F.; Correa-Barbosa, J.; Azonsivo, R.; de Oliveira, M.S.; de Sousa Siqueira, J.E.; da Rocha Galucio, N.C.; de Oliveira Bahia, M.; Burbano, R.M.R.; et al. Evaluation of the Genotoxicity and Mutagenicity of Isoeleutherin and Eleutherin Isolated from Eleutherine Plicata Herb. Using Bioassays and in Silico Approaches. Arab. J. Chem. 2021, 14, 103084. [Google Scholar] [CrossRef]
- Galucio, N.C.d.R.; Moysés, D.d.A.; Pina, J.R.S.; Marinho, P.S.B.; Gomes Júnior, P.C.; Cruz, J.N.; Vale, V.V.; Khayat, A.S.; Marinho, A.M.d.R. Antiproliferative, Genotoxic Activities and Quantification of Extracts and Cucurbitacin B Obtained from Luffa Operculata (L.) Cogn. Arab. J. Chem. 2022, 15, 103589. [Google Scholar] [CrossRef]
- Rego, C.M.A.; Francisco, A.F.; Boeno, C.N.; Paloschi, M.V.; Lopes, J.A.; Silva, M.D.S.; Santana, H.M.; Serrath, S.N.; Rodrigues, J.E.; Lemos, C.T.L.; et al. Inflammasome NLRP3 Activation Induced by Convulxin, a C-Type Lectin-like Isolated from Crotalus Durissus Terrificus Snake Venom. Sci. Rep. 2022, 12, 4706. [Google Scholar] [CrossRef]
- Xu, J.; Zhou, F.; Ji, B.P.; Pei, R.S.; Xu, N. The Antibacterial Mechanism of Carvacrol and Thymol against Escherichia Coli. Lett. Appl. Microbiol. 2008, 47, 174–179. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; De Martino, L.; Coppola, R.; De Feo, V. Effect of Essential Oils on Pathogenic Bacteria. Pharmaceuticals 2013, 6, 1451–1474. [Google Scholar] [CrossRef]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial Activity of Some Essential Oils—Present Status and Future Perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef] [Green Version]
- Dutta, S.; Kundu, A.; Saha, S.; Prabhakaran, P.; Mandal, A. Characterization, Antifungal Properties and in Silico Modelling Perspectives of Trachyspermum Ammi Essential Oil. LWT 2020, 131, 109786. [Google Scholar] [CrossRef]
- Arokiyaraj, S.; Choi, S.H.; Lee, Y.; Bharanidharan, R.; Hairul-Islam, V.I.; Vijayakumar, B.; Oh, Y.K.; Dinesh-Kumar, V.; Vincent, S.; Kim, K.H. Characterization of Ambrette Seed Oil and Its Mode of Action in Bacteria. Molecules 2015, 20, 384–395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bishoyi, A.K.; Mahapatra, M.; Paidesetty, S.K.; Padhy, R.N. Design, Molecular Docking, and Antimicrobial Assessment of Newly Synthesized Phytochemical Thymol Mannich Base Derivatives. J. Mol. Struct. 2021, 1244, 130908. [Google Scholar] [CrossRef]
- Dos Santos Barbosa, C.R.; Scherf, J.R.; de Freitas, T.S.; de Menezes, I.R.A.; Pereira, R.L.S.; dos Santos, J.F.S.; de Jesus, S.S.P.; Lopes, T.P.; de Sousa Silveira, Z.; de Morais Oliveira-Tintino, C.D.; et al. Effect of Carvacrol and Thymol on NorA Efflux Pump Inhibition in Multidrug-Resistant (MDR) Staphylococcus Aureus Strains. J. Bioenerg. Biomembr. 2021, 53, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Nagle, P.; Pawar, Y.; Sonawane, A.; Bhosale, S.; More, D. Docking Simulation, Synthesis and Biological Evaluation of Novel Pyridazinone Containing Thymol as Potential Antimicrobial Agents. Med. Chem. Res. 2014, 23, 918–926. [Google Scholar] [CrossRef]
- Dos Santos, K.L.B.; Cruz, J.N.; Silva, L.B.; Ramos, R.S.; Neto, M.F.A.; Lobato, C.C.; Ota, S.S.B.; Leite, F.H.A.; Borges, R.S.; da Silva, C.H.T.P.; et al. Identification of Novel Chemical Entities for Adenosine Receptor Type 2a Using Molecular Modeling Approaches. Molecules 2020, 25, 1245. [Google Scholar] [CrossRef] [Green Version]
- Keniya, M.V.; Sabherwal, M.; Wilson, R.K.; Woods, M.A.; Sagatova, A.A.; Tyndall, J.D.A.; Monk, B.C. Crystal Structures of Full-Length Lanosterol 14α-Demethylases of Prominent Fungal Pathogens Candida Albicans and Candida Glabrata Provide Tools for Antifungal Discovery. Antimicrob. Agents Chemother. 2018, 62, e01134-18. [Google Scholar] [CrossRef] [Green Version]
- Heaslet, H.; Harris, M.; Fahnoe, K.; Sarver, R.; Putz, H.; Chang, J.; Subramanyam, C.; Barreiro, G.; Miller, J.R. Structural Comparison of Chromosomal and Exogenous Dihydrofolate Reductase from Staphylococcus Aureus in Complex with the Potent Inhibitor Trimethoprim. Proteins Struct. Funct. Bioinforma 2009, 76, 706–717. [Google Scholar] [CrossRef]
- Achari, A.; Somers, D.O.; Champness, J.N.; Bryant, P.K.; Rosemond, J.; Stammers, D.K. Crystal Structure of the Anti-Bacterial Sulfonamide Drug Target Dihydropteroate Synthase. Nat. Struct. Biol. 1997, 4, 490–497. [Google Scholar] [CrossRef]
- Escobar, A.; Pérez, M.; Romanelli, G.; Blustein, G. Thymol Bioactivity: A Review Focusing on Practical Applications. Arab. J. Chem. 2020, 13, 9243–9269. [Google Scholar] [CrossRef]
- Dennington, R.; Keith, T.; Millam, J. GaussView, Version 5; Semichem Inc.: Shawnee Mission, KS, USA, 2009. [Google Scholar]
- Becke, A.D. Density-Functional Thermochemistry. III. The Role of Exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef] [Green Version]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16 Revision 16.A.03; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Monk, B.C.; Keniya, M.V.; Sabherwal, M.; Wilson, R.K.; Graham, D.O.; Hassan, H.F.; Chen, D.; Tyndall, J.D.A. Azole Resistance Reduces Susceptibility to the Tetrazole Antifungal VT-1161. Antimicrob. Agents Chemother. 2019, 63, e02114-18. [Google Scholar] [CrossRef] [Green Version]
- Thomsen, R.; Christensen, M.H. MolDock: A New Technique for High-Accuracy Molecular Docking. J. Med. Chem. 2006, 49, 3315–3321. [Google Scholar] [CrossRef]
- Wang, J.; Cieplak, P.; Kollman, P.A. How Well Does a Restrained Electrostatic Potential (RESP) Model Perform in Calculating Conformational Energies of Organic and Biological Molecules? J. Comput. Chem. 2000, 21, 1049–1074. [Google Scholar] [CrossRef]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and Testing of a General Amber Force Field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef]
- Dolinsky, T.J.; Nielsen, J.E.; McCammon, J.A.; Baker, N.A. PDB2PQR: An Automated Pipeline for the Setup of Poisson-Boltzmann Electrostatics Calculations. Nucleic Acids Res. 2004, 32, W665–W667. [Google Scholar] [CrossRef]
- Maier, J.A.; Martinez, C.; Kasavajhala, K.; Wickstrom, L.; Hauser, K.E.; Simmerling, C. Ff14SB: Improving the Accuracy of Protein Side Chain and Backbone Parameters from Ff99SB. J. Chem. Theory Comput. 2015, 11, 3696–3713. [Google Scholar] [CrossRef] [Green Version]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of Simple Potential Functions for Simulating Liquid Water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Santos, C.B.R.; Santos, K.L.B.; Cruz, J.N.; Leite, F.H.A.; Borges, R.S.; Taft, C.A.; Campos, J.M.; Silva, C.H.T.P. Molecular Modeling Approaches of Selective Adenosine Receptor Type 2A Agonists as Potential Anti-Inflammatory Drugs. J. Biomol. Struct. Dyn. 2020, 39, 3115–3127. [Google Scholar] [CrossRef]
- Araújo, P.H.F.; Ramos, R.S.; da Cruz, J.N.; Silva, S.G.; Ferreira, E.F.B.; de Lima, L.R.; Macêdo, W.J.C.; Espejo-Román, J.M.; Campos, J.M.; Santos, C.B.R. Identification of Potential COX-2 Inhibitors for the Treatment of Inflammatory Diseases Using Molecular Modeling Approaches. Molecules 2020, 25, 4183. [Google Scholar] [CrossRef]
- Darden, T.; York, D.; Pedersen, L. Particle Mesh Ewald: An N·log(N) Method for Ewald Sums in Large Systems. J. Chem. Phys. 1993, 98, 10089–10092. [Google Scholar] [CrossRef] [Green Version]
- Ryckaert, J.P.; Ciccotti, G.; Berendsen, H.J.C. Numerical Integration of the Cartesian Equations of Motion of a System with Constraints: Molecular Dynamics of n-Alkanes. J. Comput. Phys. 1977, 23, 327–341. [Google Scholar] [CrossRef] [Green Version]
- Lzaguirre, J.A.; Catarello, D.P.; Wozniak, J.M.; Skeel, R.D. Langevin Stabilization of Molecular Dynamics. J. Chem. Phys. 2001, 114, 2090–2098. [Google Scholar] [CrossRef]
- Hou, T.; Wang, J.; Li, Y.; Wang, W. Assessing the Performance of the MM/PBSA and MM/GBSA Methods. 1. The Accuracy of Binding Free Energy Calculations Based on Molecular Dynamics Simulations. J. Chem. Inf. Model. 2011, 51, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Leão, R.P.; Cruz, J.V.J.N.; da Costa, G.V.; Cruz, J.V.J.N.; Ferreira, E.F.B.; Silva, R.C.; de Lima, L.R.; Borges, R.S.; Dos Santos, G.B.; Santos, C.B.R. Identification of New Rofecoxib-Based Cyclooxygenase-2 Inhibitors: A Bioinformatics Approach. Pharmaceuticals 2020, 13, 209. [Google Scholar] [CrossRef] [PubMed]
- Lima, A.R.J.A.d.M.; Siqueira, A.S.; Möller, M.L.S.; Souza, R.C.D.; Cruz, J.N.; Lima, A.R.J.A.d.M.; da Silva, R.C.; Aguiar, D.C.F.; Junior, J.L.d.S.G.V.; Gonçalves, E.C. In Silico Improvement of the Cyanobacterial Lectin Microvirin and Mannose Interaction. J. Biomol. Struct. Dyn. 2020, 40, 1064–1073. [Google Scholar] [CrossRef]

| Drug Target | MolDock Score (Kcal/mol) |
|---|---|
| CYP51 (C. albicans) | −77.85 |
| Dihydrofolate reductase (S. aureus) | −67.53 |
| Dihydropteroate synthase (E. coli) | −60.88 |
| System | ΔEvdW | ΔEele | ΔGGB | ΔGNP | ΔGbind |
|---|---|---|---|---|---|
| CYP51(C. albicans) | −24.88 | −3.94 | 12.15 | −3.37 | −20.04 |
| dihydrofolate reductase (S. aureus) | −26.44 | −11.07 | 16.03 | −3.25 | −24.73 |
| dihydropteroate synthase (E. coli) | −21.71 | −12.43 | 19.23 | −2.93 | −17.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz, J.N.; Silva, S.G.; Pereira, D.S.; Souza Filho, A.P.d.S.; de Oliveira, M.S.; Lima, R.R.; Andrade, E.H.d.A. In Silico Evaluation of the Antimicrobial Activity of Thymol—Major Compounds in the Essential Oil of Lippia thymoides Mart. & Schauer (Verbenaceae). Molecules 2022, 27, 4768. https://doi.org/10.3390/molecules27154768
Cruz JN, Silva SG, Pereira DS, Souza Filho APdS, de Oliveira MS, Lima RR, Andrade EHdA. In Silico Evaluation of the Antimicrobial Activity of Thymol—Major Compounds in the Essential Oil of Lippia thymoides Mart. & Schauer (Verbenaceae). Molecules. 2022; 27(15):4768. https://doi.org/10.3390/molecules27154768
Chicago/Turabian StyleCruz, Jorddy Neves, Sebastião Gomes Silva, Daniel Santiago Pereira, Antônio Pedro da Silva Souza Filho, Mozaniel Santana de Oliveira, Rafael Rodrigues Lima, and Eloisa Helena de Aguiar Andrade. 2022. "In Silico Evaluation of the Antimicrobial Activity of Thymol—Major Compounds in the Essential Oil of Lippia thymoides Mart. & Schauer (Verbenaceae)" Molecules 27, no. 15: 4768. https://doi.org/10.3390/molecules27154768
APA StyleCruz, J. N., Silva, S. G., Pereira, D. S., Souza Filho, A. P. d. S., de Oliveira, M. S., Lima, R. R., & Andrade, E. H. d. A. (2022). In Silico Evaluation of the Antimicrobial Activity of Thymol—Major Compounds in the Essential Oil of Lippia thymoides Mart. & Schauer (Verbenaceae). Molecules, 27(15), 4768. https://doi.org/10.3390/molecules27154768










