Non-Destructive Analysis of Chlorpheniramine Maleate Tablets and Granules by Chemometrics-Assisted Attenuated Total Reflectance Infrared Spectroscopy
Abstract
1. Introduction
2. Results
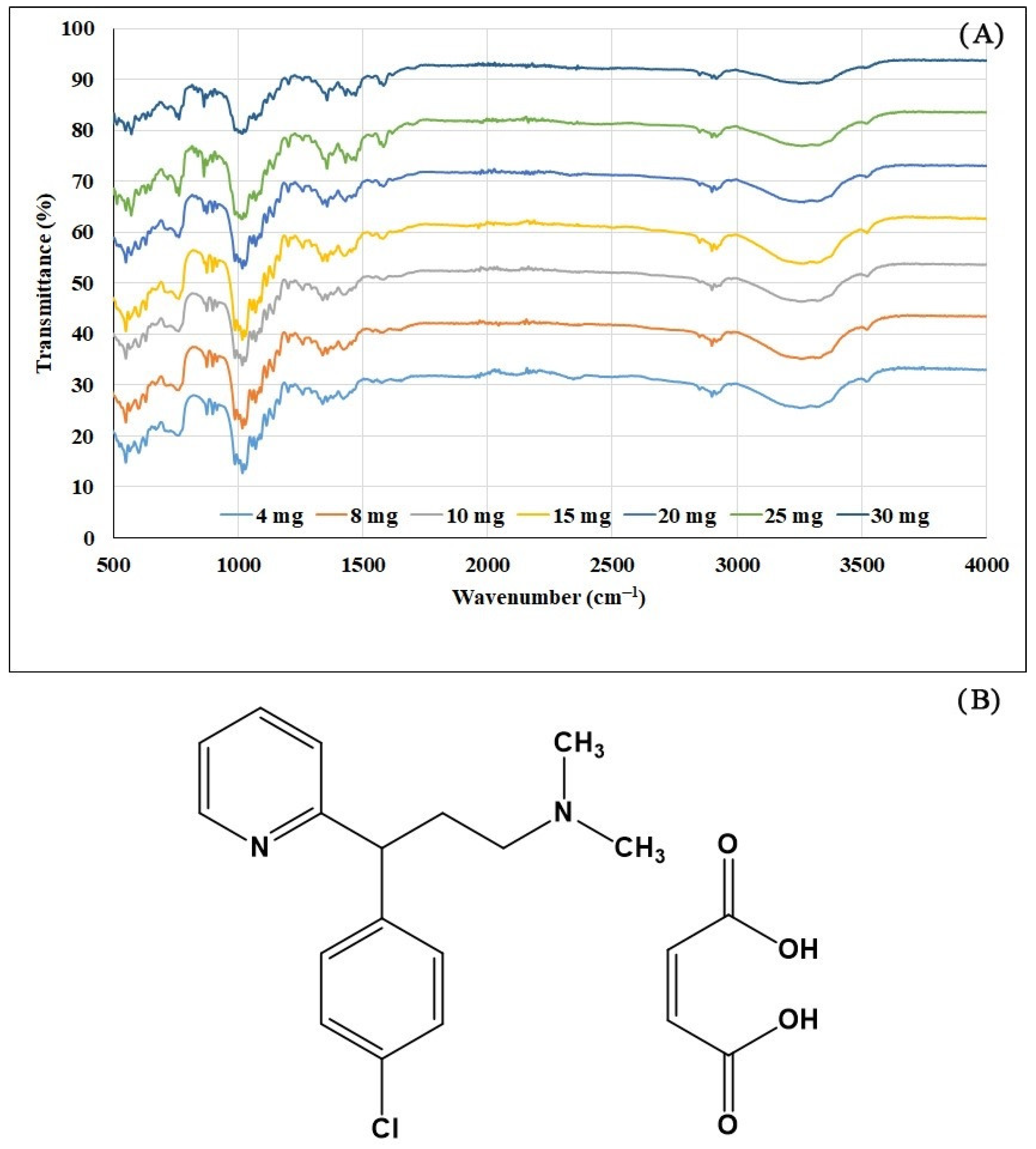
2.1. ATR-IR Measurement
2.2. HPLC Method and Method Validation
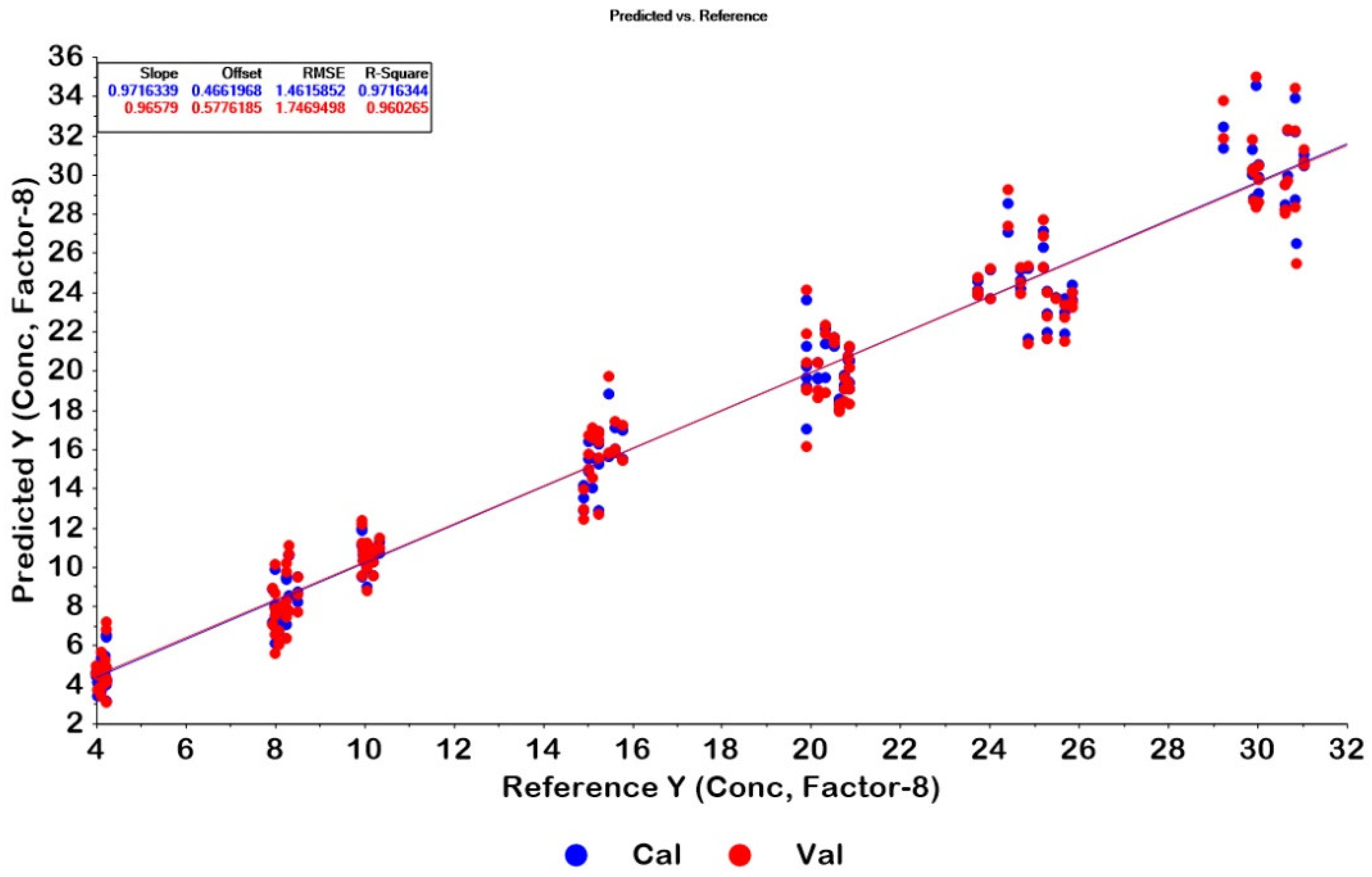
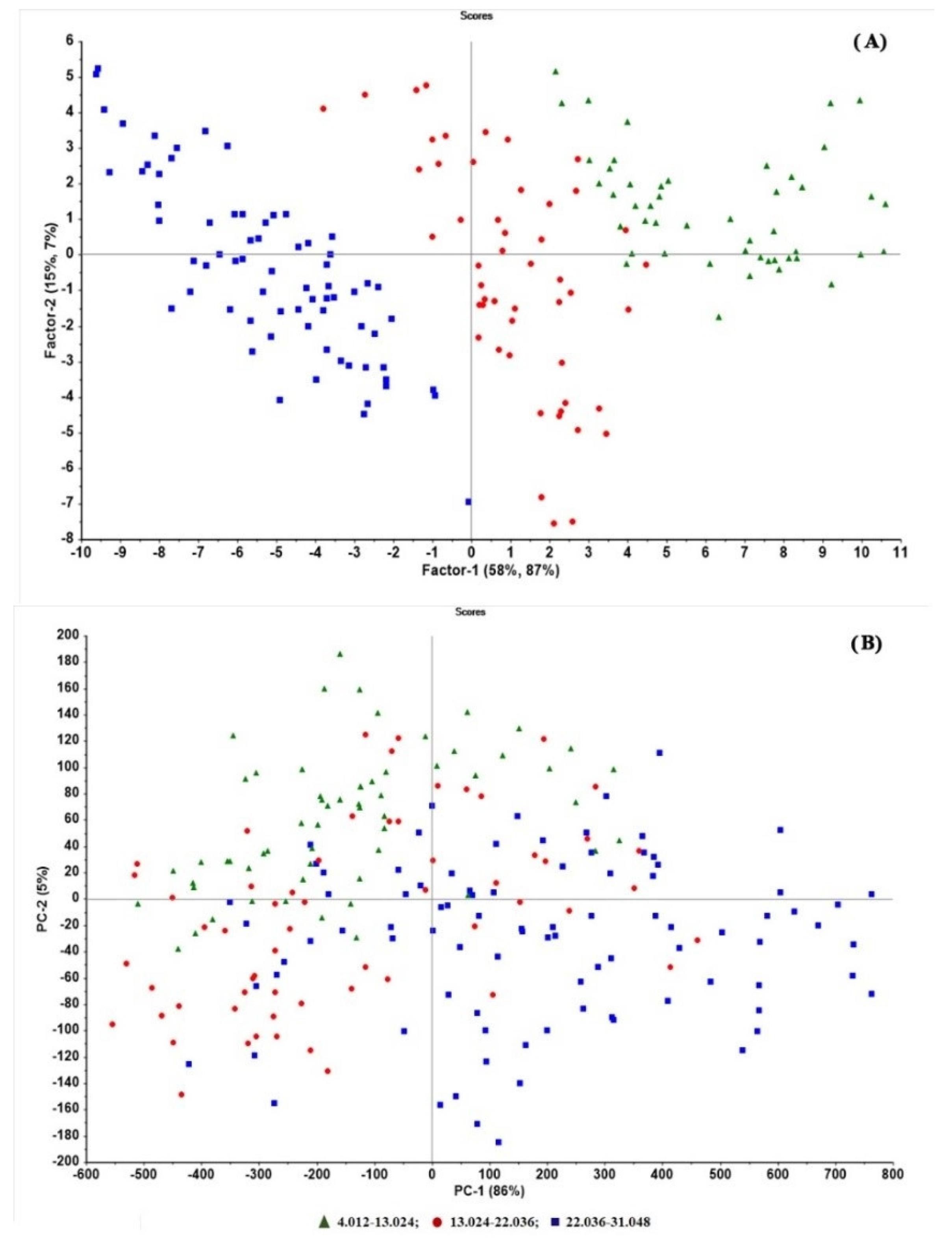
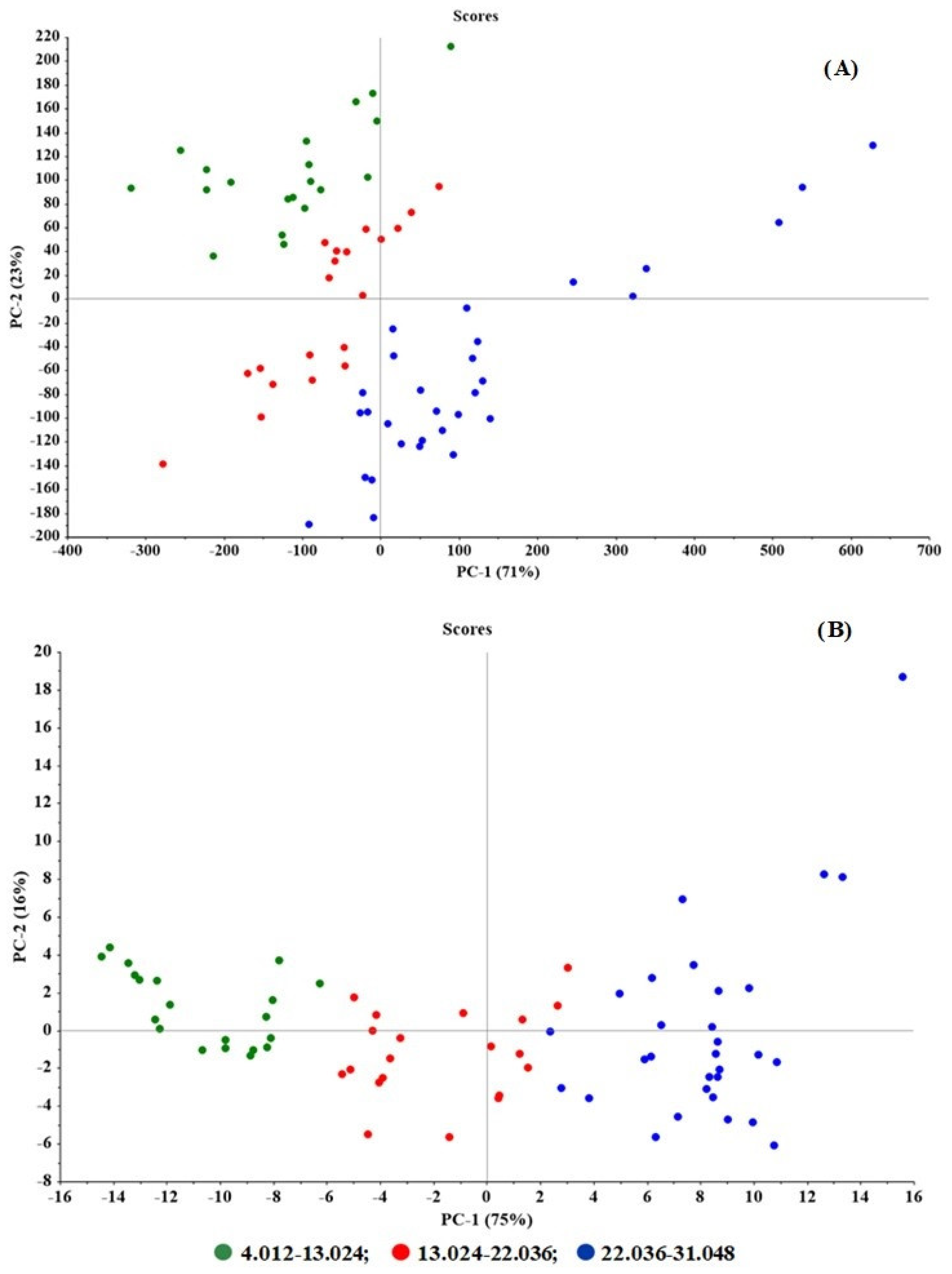
2.3. PLSR Modelling
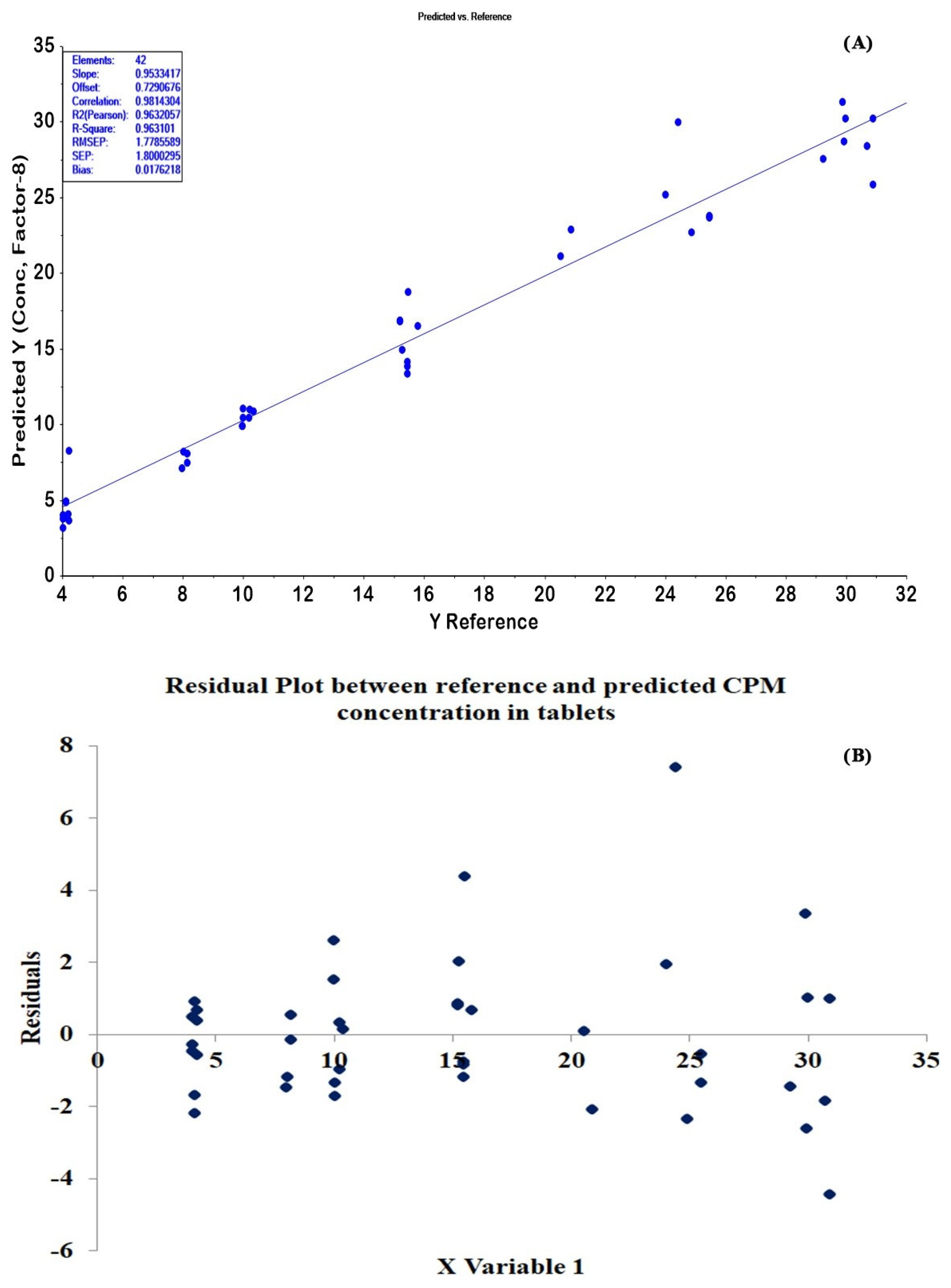
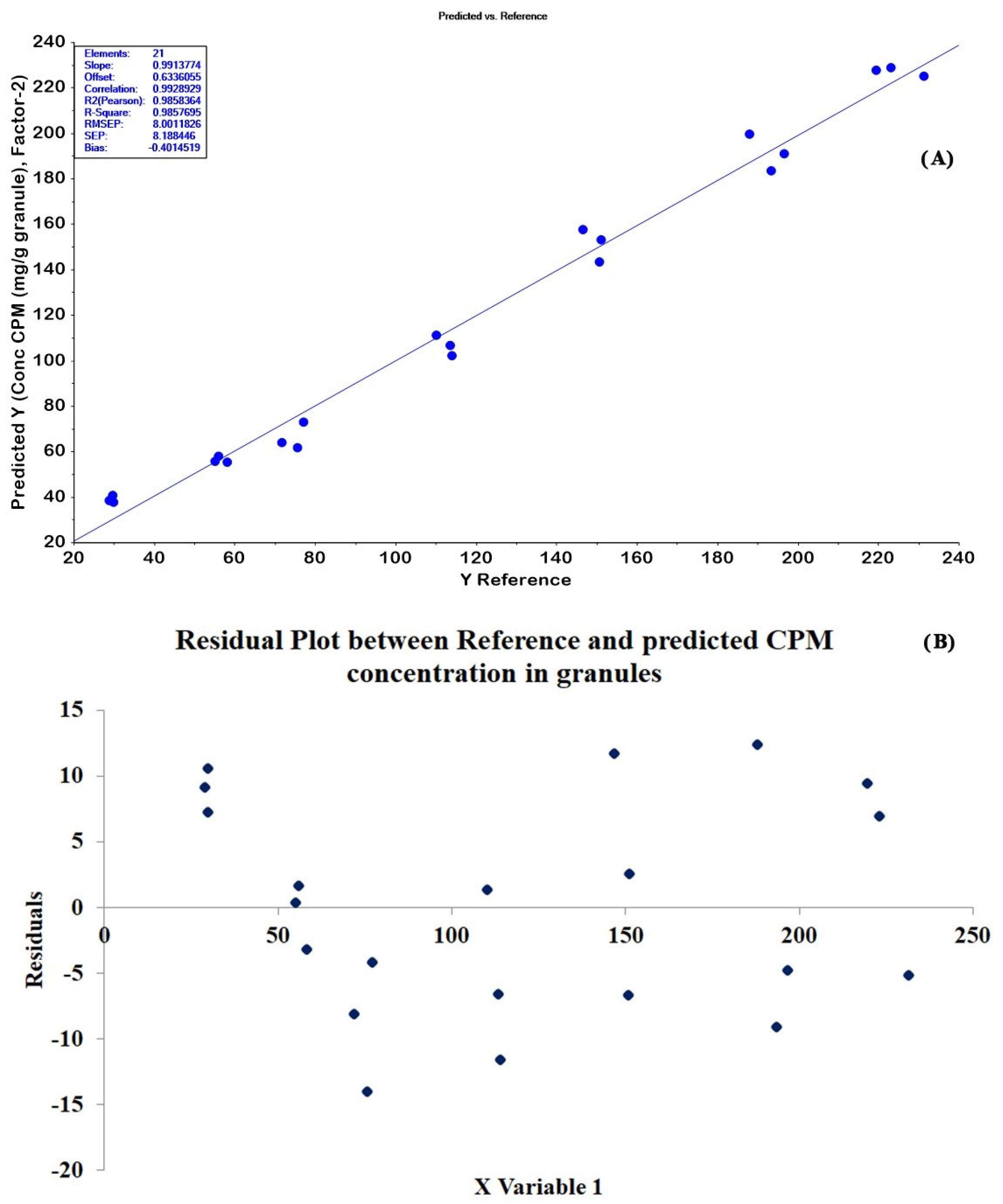
2.4. Quantitative Determination of CPM Tablets and Granules by PLSR and HPLC Methods
3. Discussion
4. Conclusions
5. Experimental
5.1. Chemicals and Reagents
5.2. Preparation of Chlorpheniramine Maleate Tablets
5.3. ATR-IR Measurement
5.4. HPLC Analysis
5.5. PLSR Modeling
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- General Chapter: USP.Uniformity of dosage units <905>. In USP–NF; USP: Rockville, MD, USA, 2022. [CrossRef]
- Pawar, P.; Talwar, S.; Reddy, D.; Bandi, C.K.; Wu, H.; Sowrirajan, K.; Friedman, R.; Drazer, G.; Drennen, J.K., III; Muzzio, F.J. A “Large-N” content uniformity process analytical technology (PAT) method for phenytoin sodium tablets. J. Pharm. Sci. 2019, 108, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Doctor, J.; Thakkar, P.; Prajapati, M.; Patel, N.; Mehta, P.J. Non-destructive Raman spectroscopic method for estimation of montelukast from tablet dosage form. Int. J. Pharm. Pharm. Sci. 2017, 9, 161–165. [Google Scholar] [CrossRef][Green Version]
- Kandpal, L.M.; Park, E.; Tewari, J.; Cho, B.K. Spectroscopic techniques for nondestructive quality inspection of pharmaceutical products: A Review. J. Biosyst. Eng. 2015, 40, 394–408. [Google Scholar] [CrossRef]
- Takeuchi, I.; Shimakura, K.; Ohtake, H.; Takayanagi, J.; Tomoda, K.; Nakajima, T.; Terada, H.; Makino, K. Nondestructive analysis of structure and components of tablet coated with film by the usage of Terahertz time-domain reflection spectroscopy. J. Pharm. Sci. 2014, 103, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Woo, Y.A. Content uniformity in granules for aceclonac controlled release (CR) tablets determined using near-infrared spectroscopy and wide area illumination (WAI) Raman spectroscopy. Arch. Pharm. Res. 2012, 35, 351–358. [Google Scholar] [CrossRef]
- Moros, J.; Galipienso, N.; Vilches, R.; Garrigues, S.; de la Guardia, M. Nondestructive direct determination of heroin in Seized lllicit street drugs by diffuse reflectance near-infrared spectroscopy. Anal. Chem. 2008, 80, 7257–7265. [Google Scholar] [CrossRef]
- Wikstrom, H.; RoMero-Torres, S.; Wongweragiat, S.; Williams, J.A.S.; Grant, E.R.; Taylor, L.S. On-line content uniformity determination of tablets using low-resolution Raman scpectroscopy. Appl. Spectrosc. 2006, 60, 672–681. [Google Scholar] [CrossRef]
- International Conference on Harmonisation, ICH Q2(R1) Revision: Guideline on Validation of Analytical Procedures. Retrieved 11 March 2022. Available online: https://database.ich.org/sites/default/files/Q2R2Q14_EWG_Concept_Paper.pdf (accessed on 31 March 2022).
- Palou, A.; Cruz, J.; Blanco, M.; Tomas, J.; de los Rios, J.; Alcala, M. Determination of drug, excipients and coating distribution in pharmaceutical tablets using NIR-CI. J. Pharm. Anal. 2012, 2, 90–97. [Google Scholar] [CrossRef]
- Bansal, A.; Chhabra, V.; Rawal, R.K.; Sharma, S. Chemometrics: A new scenario in herbal drug standardization. J. Pharm. Anal. 2014, 4, 223–233. [Google Scholar] [CrossRef]
- Eid, S.M.; Soliman, S.S.; Elghobashy, M.R.; Abdalla, O.M. ATR-FTIR coupled with chemometrics for quantification of vildagliptin and metformin in pharmaceutical combinations having diverged concentration ranges. Vib. Spectrosc. 2020, 106, 102995. [Google Scholar] [CrossRef]
- Lawson, G.; Ogwu, J.; Tanna, S. Quantitative screening of the pharmaceutical ingredient for the rapid identification of substandard and falsified medicines using reflectance infrared spectroscopy. PLoS ONE 2018, 13, e0202059. [Google Scholar] [CrossRef] [PubMed]
- Hassib, S.T.; Hassan, G.S.; El-Zaher, A.A.; Fouad, M.A.; Taha, E.A. Quantitative analysis of anti-inflammatory drugs using FTIR-ATR spectrometry. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2017, 186, 59–65. [Google Scholar] [CrossRef]
- Sruthi, K.; Prasanth, S.; Kamarunnisa, K.; Drisya, N.; Ashyam, K. FTIR-spectrophotometric analysis of levosulpiride and its pharmaceutical formulations. Int. J. Pharm. Chem. Anal. 2018, 5, 151–155. [Google Scholar]
- Riyanto, R.; Nas, S.W. Validation of analytical methods for determination of methamphetamine using Fourier transform infrared (FTIR) spectroscopy. IOSR J. Pharm. Biol. Sci. 2016, 11, 51–59. [Google Scholar] [CrossRef]
- Acheampong, A.; Gyasi, W.O.; Darko, G.; Apau, J.; Addai-Arhin, S. Validated RP-HPLC method for simultaneous determination and quantification of chlorpheniramine maleate, paracetamol and caffeine in tablet formulation. Springerplus 2016, 5, 625. [Google Scholar] [CrossRef] [PubMed]
- Sanchaniya, P.M.; Mehta, F.A.; Uchadadiya, N.B. Development and validation of an RP-HPLC method for estimation of chlorpheniramine maleate, ibuprofen, and phenylephrine hydrochloride in combined pharmaceutical dosage form. Chromatogr. Res. Int. 2013, 2013, 424865. [Google Scholar] [CrossRef]
- Moyano, M.A.; Rosasco, M.A.; Pizzorno, M.T.; Segall, A.I. Simultaneous determination of chlorpheniramine maleate and dexamethasone in a tablet dosage form by liquid chromatography. J. AOAC Int. 2005, 88, 1677–1683. [Google Scholar] [CrossRef]
- Pojanagaroon, T.; Liawruangrath, S.; Liawruangrath, S. A Direct current polarographic method for the determination of chlorpheniramine maleate in pharmaceutical preparations. Chiang Mai J. Sci. 2007, 34, 135–142. [Google Scholar]
- Xu, M.; Zhou, L.; Zhang, Q.; Wu, Z.; Shi, X.; Qiao, Y. Near-infrared chemical imaging for quantitative analysis of chlorpheniramine maleate and distribution homogeneity assessment in pharmaceutical formulations. J. Innov. Opt. Health Sci. 2016, 9, 1650002. [Google Scholar] [CrossRef]
- Mahmoud, A.R.; Al-Healy, F.M. UV-Spectral studies on chlorpheniramine maleate in pure form and pharmaceutical preparations. Egypt. J. Chem. 2021, 64, 4151–4156. [Google Scholar]
- Bratty, M.A. Visible Spectrophotometric determination of chlorpheniramine maleate and diphenhydramine hydrochloride in raw and dosage form using potassium permanganate. Orient. J. Chem. 2016, 32, 885–894. [Google Scholar] [CrossRef]
- Monograph: USP. Chlorpheniramine Maleate Tablets. In USP–NF; USP: Rockville, MD, USA, 2022. [CrossRef]
- Joshi, R.; Sathasivam, R.; Park, S.U.; Lee, H.; Kim, M.S.; Baek, I.; Cho, B.K. Application of fourier transform infrared spectroscopy and multivariate analysis methods for the non-destructive evaluation of phenolics compounds in moringa powder. Agriculture 2022, 12, 10. [Google Scholar] [CrossRef]
- Rohman, A.; Setyaningrum, D.L.; Riyanto, S. FTIR spectroscopy combined with partial least square for analysis of red fruit oil in ternary mixture system. Int. J. Spectrosc. 2014, 2014, 785914. [Google Scholar] [CrossRef][Green Version]
- Mishra, P.; Nordon, A.; Roger, J.M. Improved prediction of tablet properties with near-infrared spectroscopy by a fusion of scatter correction techniques. J. Pharm. Biomed. Anal. 2021, 192, 113684. [Google Scholar] [CrossRef]
- Kim, G.; Hong, S.J.; Lee, A.Y.; Lee, Y.E.; Im, S. Moisture content measurement of broadleaf litters using near-infrared spectroscopy technique. Remote Sens. 2017, 9, 1212. [Google Scholar] [CrossRef]
- Sirigiri, B.; Chengalva, P.; Parameswari, S.A.; Aruna, G. A novel HPLC method for the simultaneous determination of chlorpheniramine maleate and dextromethorphan in bulk and pharmaceutical formulation. Int. J. Pharm. Sci. Res. 2018, 9, 1147–1151. [Google Scholar]
- General Chapter: USP. Chromatography <621>. In USP–NF; USP: Rockville, MD, USA, 2022. [CrossRef]
- Rao, T.P.; Biju, V.M. Spectrophotometry|Organic Compounds. In Encyclopedia of Analytical Science, 2nd ed.; Worsfold, P., Townshend, A., Poole, C., Eds.; Elsevier: New York, NY, USA, 2005; pp. 358–366. [Google Scholar]
- Khajehsharifi, H.; Pourbasheer, E.; Tavallali, H.; Sarvi, S.; Sadeghi, M. The comparison of partial least squares and principal component regression in simultaneous spectrophotometric determination of ascorbic acid, dopamine and uric acid in real samples. Arab. J. Chem. 2017, 10, S3451–S3458. [Google Scholar] [CrossRef]
- Planinsek, O.; Planinsek, D.; Zega, A.; Breznik, M.; Srcic, S. Surface analysis of powder binary mixtures with ATR FTIR spectroscopy. Int. J. Pharm. 2006, 319, 13–19. [Google Scholar] [CrossRef]
- Kennard, R.W.; Stone, L.A. Computer-aided design of experiments. Technometrics 1969, 11, 137–148. [Google Scholar] [CrossRef]
- Khajehsharifi, H.; Eskandari, Z.; Sareban, N. Using partial least squares and principal component regression in simultaneous spectrophotometric analysis of pyrimidine base. Arab. J. Chem. 2017, 10, S141–S147. [Google Scholar] [CrossRef]


| Method Validation Characteristics | Results |
|---|---|
| Range | 10–30 µg/mL |
| Linearity | |
| Equation | y = 14,603x − 2363 |
| R2 (n = 3) | 0.9999 |
| Accuracy (% Recovery) | 100.0–102.9% |
| Precision | |
| Repeatability (n = 9) | 1.26 |
| Intermediate precision (n = 18) | 1.13 |
| Specificity | |
| Peak purity index (standard) | 1.0000 |
| Peak purity index (standard spiked placebo) | 0.9999 |
| Model Number | Spectral Range (cm−1) | Spectral Data * | Latent Factors | R2 (Model) | R2 (Pearson) | RMSEC | RMSEP | Bias | Derivative Polynomial Order |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 400–4000 | original | 5 | 0.9142 | 0.9328 | 2.5419 | 2.4797 | 0.1986 | - |
| 2 | 400–4000 | D2 | 9 | 0.9545 | 0.9309 | 1.8506 | 2.4850 | −0.0359 | - |
| 3 | 400–4000 | area-normalized | 4 | 0.9083 | 0.9287 | 2.6284 | 2.5648 | 0.1862 | - |
| 4 | 400–4000 | area-normalized + SNV | 3 | 0.9355 | 0.9438 | 2.2040 | 2.2418 | 0.2695 | - |
| 5 | 2700–4000 | area-normalized + SNV | 5 | 0.9605 | 0.9591 | 1.7251 | 2.0673 | −0.1105 | - |
| 6 | 400–4000 | D1 | 5 | 0.9264 | 0.9312 | 2.3546 | 2.478 | −0.0123 | 2 order 11 pt. |
| 7 | 400–4000 | D1 + SNV | 9 | 0.9394 | 0.941 | 2.1359 | 2.3093 | −0.2371 | 2 order 11 pt. |
| 8 | 500–1700 | D2 | 7 | 0.9716 | 0.9417 | 1.4625 | 2.2801 | −0.3258 | 2 order 11 pt. |
| 9 | 2500–4000 | D2 | 5 | 0.9823 | 0.9579 | 1.1555 | 2.0986 | −0.1973 | 2 order 11 pt. |
| 10 | 500–1700, 2500–4000 | D2 | 7 | 0.9840 | 0.9562 | 1.0986 | 2.0155 | −0.415 | 2 order 11 pt. |
| 11 | 500–1700 | area-normalized + SNV | 8 | 0.9741 | 0.9639 | 1.3977 | 1.7611 | 0.0495 | - |
| 12 | 2500–4000 | area-normalized + SNV | 8 | 0.9481 | 0.9508 | 1.9759 | 2.1874 | −0.1130 | - |
| 13 | 500–1700, 2500–4000 | area-normalized + SNV | 8 | 0.9716 | 0.9632 | 1.4616 | 1.7786 | 0.0176 | - |
| Model Number | Spectral Range (cm−1) | Spectral Data * | Latent Factors | R2 (Model) | R2 (Pearson) | RMSEC | RMSEP | Bias | Derivative Polynomial Order |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 400–3700 | Original | 2 | 0.9498 | 0.9762 | 15.0802 | 10.6003 | −2.0409 | - |
| 2 | 400–1700, 2800–3700 | Original 2 | 2 | 0.9395 | 0.9782 | 16.5568 | 10.1016 | −1.8677 | - |
| 3 | 400–3700 | SNV | 1 | 0.9366 | 0.9835 | 16.9389 | 8.6349 | −0.3478 | - |
| 4 | 400–1700, 2800–3700 | SNV | 1 | 0.9355 | 0.9838 | 17.0872 | 8.5433 | −0.3631 | - |
| 5 | 400–3700 | Area normalization | 2 | 0.9295 | 0.9730 | 17.8691 | 11.2178 | −2.0792 | - |
| 6 | 400–3700 | SNV + D1 | 2 | 0.9577 | 0.9858 | 13.8447 | 8.0012 | −0.4014 | 2 order 11 pt. |
| 7 | 400–3700 | SNV + D1 | 2 | 0.9572 | 0.9851 | 13.9199 | 8.4352 | −0.6258 | 2 order 21 pt. |
| 8 | 400–3700 | SNV + D1 | 1 | 0.9362 | 0.9833 | 16.9942 | 8.6657 | −0.3323 | 3 order 11 pt. |
| 9 | 400–3700 | SNV + D1 | 1 | 0.9364 | 0.9834 | 16.9678 | 8.6601 | −0.3574 | 3 order 21 pt. |
| 10 | 400–3700 | SNV + D1 | 1 | 0.9368 | 0.9835 | 16.9125 | 8.6220 | −0.3550 | 4 order 11 pt. |
| 11 | 400–3700 | SNV + D1 | 1 | 0.9368 | 0.9835 | 16.9196 | 8.6263 | −0.3573 | 4 order 21 pt. |
| Active Content (%) | Tablets Model | Granules Model | ||
|---|---|---|---|---|
| Calibration | Validation | Calibration | Validation | |
| 4 | 24 | 6 | 7 | 3 |
| 8 | 24 | 6 | 7 | 3 |
| 12 | 24 | 6 | 7 | 3 |
| 15 | 24 | 6 | 7 | 3 |
| 20 | 24 | 6 | 7 | 3 |
| 25 | 24 | 6 | 7 | 3 |
| 30 | 24 | 6 | 7 | 3 |
| Total | 168 | 42 | 49 | 21 |
| Composition (mg/Tablet) | Formulation | ||||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | |
| CPM | 4.0 | 8.0 | 10.0 | 15.0 | 20.0 | 25.0 | 30.0 |
| Lactose monohydrate | 80.0 | 76.0 | 74.0 | 69.0 | 64.0 | 59.0 | 54.0 |
| Tapioca starch | 40.0 | 40.0 | 40.0 | 40.0 | 40.0 | 40.0 | 40.0 |
| Croscarmellose sodium | 3.7 | 3.7 | 3.7 | 3.7 | 3.7 | 3.7 | 3.7 |
| Corn starch | 3.6 | 3.6 | 3.6 | 3.6 | 3.6 | 3.6 | 3.6 |
| Magnesium stearate | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phechkrajang, C.; Khongkaew, P.; Limwikrant, W.; Jaturanpinyo, M. Non-Destructive Analysis of Chlorpheniramine Maleate Tablets and Granules by Chemometrics-Assisted Attenuated Total Reflectance Infrared Spectroscopy. Molecules 2022, 27, 3760. https://doi.org/10.3390/molecules27123760
Phechkrajang C, Khongkaew P, Limwikrant W, Jaturanpinyo M. Non-Destructive Analysis of Chlorpheniramine Maleate Tablets and Granules by Chemometrics-Assisted Attenuated Total Reflectance Infrared Spectroscopy. Molecules. 2022; 27(12):3760. https://doi.org/10.3390/molecules27123760
Chicago/Turabian StylePhechkrajang, Chutima, Putthiporn Khongkaew, Waree Limwikrant, and Montree Jaturanpinyo. 2022. "Non-Destructive Analysis of Chlorpheniramine Maleate Tablets and Granules by Chemometrics-Assisted Attenuated Total Reflectance Infrared Spectroscopy" Molecules 27, no. 12: 3760. https://doi.org/10.3390/molecules27123760
APA StylePhechkrajang, C., Khongkaew, P., Limwikrant, W., & Jaturanpinyo, M. (2022). Non-Destructive Analysis of Chlorpheniramine Maleate Tablets and Granules by Chemometrics-Assisted Attenuated Total Reflectance Infrared Spectroscopy. Molecules, 27(12), 3760. https://doi.org/10.3390/molecules27123760