Synthesis and Anticancer Properties of New 3-Methylidene-1-sulfonyl-2,3-dihydroquinolin-4(1H)-ones
Abstract
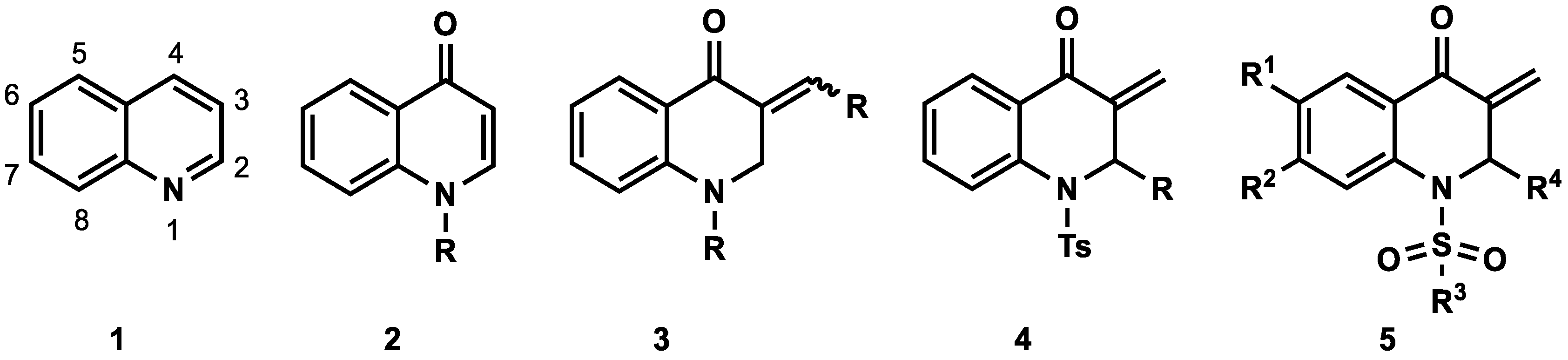
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Biology
2.2.1. In Vitro Cytotoxic Activity
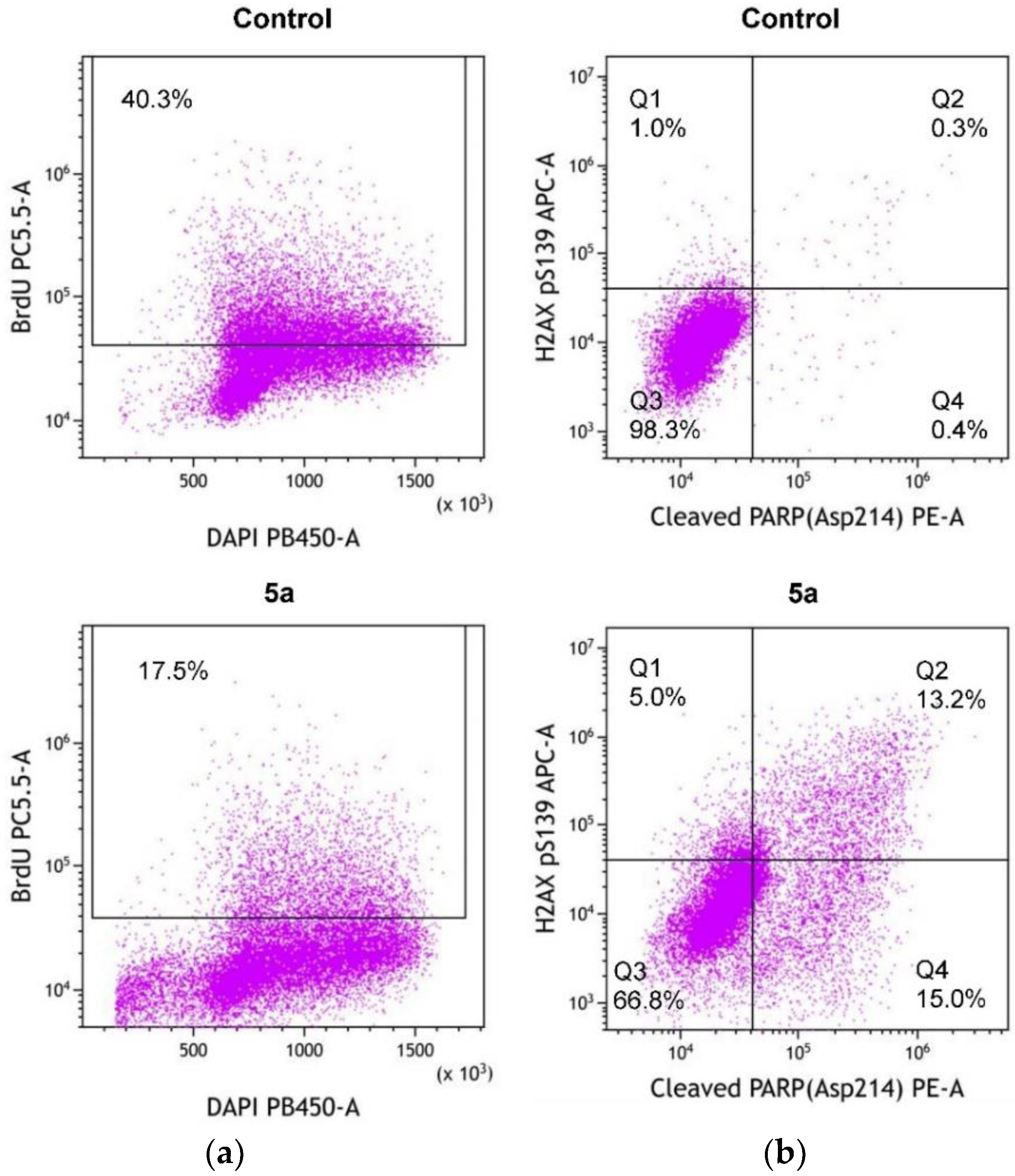
2.2.2. Inhibition of Cell Proliferation, Generation of DNA Damage, and Induction of Apoptosis by 5a
2.2.3. Influence of 5a on ABCB1 Gene Expression
3. Materials and Methods
3.1. Chemistry
3.1.1. General
3.1.2. Experimental Procedure for the Synthesis of 3-Methylidene-1-sulfonyl-2,3-dihydroquinolin-4(1H)-ones 5a-t
3.2. Biology
3.2.1. Cell Lines and Cell Culture
3.2.2. In Vitro Cytotoxicity Assay (MTT)
3.2.3. Analysis of Apoptosis by Flow Cytometry Using FITC-Annexin V and Propidium Iodide (PI) Double-Staining
3.2.4. Analysis of Cell Proliferation, Apoptosis, and DNA Damage by Flow Cytometry
3.2.5. Real-Time PCR
3.2.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Shiro, T.; Fukaya, T.; Tobe, M. The chemistry and biological activity of heterocycle-fused quinolinone derivatives: A review. Eur. J. Med. Chem. 2015, 97, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Beniddir, M.A.; Le Borgne, E.; Iorga, B.I.; Loaec, N.; Lozach, O.; Meijer, L.; Awang, K.; Litaudon, M. Acridone alkaloids from Glycosmis chlorosperma as DYRK1A inhibitors. J. Nat. Prod. 2014, 77, 1117–1122. [Google Scholar] [CrossRef] [PubMed]
- Cretton, S.; Dorsaz, S.; Azzollini, A.; Favre-Godal, Q.; Marcourt, L.; Ebrahimi, S.N.; Voinesco, F.; Michellod, E.; Sanglard, D.; Gindro, K.; et al. Antifungal Quinoline Alkaloids from Waltheria indica. J. Nat. Prod. 2016, 79, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Emmerson, A.M.; Jones, A.M. The quinolones: Decades of development and use. J. Antimicrob. Chemother. 2003, 51, 13–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kahriman, N.; Iskender, N.Y.; Yucel, M.; Yayli, N.; Demir, E.; Demirbag, Z. Microwave-assisted synthesis of 1,3′-diaza-flavanone/flavone and their alkyl derivatives with antimicrobial activity. J. Heterocycl. Chem. 2012, 49, 71–79. [Google Scholar] [CrossRef]
- Naeem, A.; Badshah, S.L.; Muska, M.; Ahmad, N.; Khan, K. The current case of quinolones: Synthetic approaches and antibacterial activity. Molecules 2016, 21, 268. [Google Scholar] [CrossRef] [Green Version]
- Batalha, P.N.; Vieira de Souza, M.C.; Peña-Cabrera, E.; Cruz, D.C.; da Costa Santos Boechat, F. Quinolones in the search for new anticancer agents. Curr. Pharm. Des. 2016, 22, 6009–6020. [Google Scholar] [CrossRef]
- Rajput, S.; Gardner, C.R.; Failes, T.W.; Arndt, G.M.; Black, D.S.; Kumar, N. Synthesis and anticancer evaluation of 3-substituted quinolin-4-ones and 2,3-dihydroquinolin-4-ones. Bioorg. Med. Chem. 2014, 22, 105–115. [Google Scholar] [CrossRef]
- Gong, Y.; Kato, K. New synthesis of 2-trifluoromethyl-2,3-dihydro-1H-quinolin-4-ones. J. Fluor. Chem. 2004, 125, 767–773. [Google Scholar] [CrossRef]
- Xiao, X.; Liu, X.; Dong, S.; Cai, Y.; Lin, L.; Feng, X. Asymmetric synthesis of 2,3-dihydroquinolin-4-one derivatives catalyzed by a chiral bisguanidium salt. Chem. Eur. J. 2012, 18, 15922–15926. [Google Scholar] [CrossRef]
- Abdel-Aal, M.A.A.; Abdel-Aziz, S.A.; Shaykoon, M.S.A.; Abuo-Rahma, G.E.A. Toward anticancer fluoroquinolones: A rewiew article. Arch. Pharm. 2019, 352, 1800376. [Google Scholar] [CrossRef] [PubMed]
- Janecka, A.; Wyrębska, A.; Gach, K.; Fichna, J.; Janecki, T. Natural and synthetic α-methylenelactones and α-methylenelactams with anticancer potential. Drug Discov. Today 2012, 17, 561. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Wu, K.; Xu, A.; Jiao, P.; Li, H.; Xing, L. The sesquiterpene lactone eupatolide induces apoptosis in non-small cell lung cancer cells by suppressing STAT3 signaling. Environ. Toxicol. Pharmacol. 2021, 81, 103513. [Google Scholar] [CrossRef] [PubMed]
- De Cicco, P.; Busà, R.; Ercolano, G.; Formisano, C.; Allegra, M.; Taglialatela-Scafati, O.; Ianaro, A. Inhibitory effects of cynaropicrin on human melanoma progression by targeting MAPK, NF-κB, and Nrf-2 signaling pathways in vitro. Phytother Res. 2020, 35, 1432–1442. [Google Scholar] [CrossRef]
- Pati, H.N.; Das, U.; Sharma, R.K.; Dimmock, J.R. Cytotoxic thiol alkylators. Mini Rev. Med. Chem. 2007, 7, 131–139. [Google Scholar] [CrossRef]
- Koszuk, J.; Bartosik, T.; Wojciechowki, J.; Wolf, W.M.; Janecka, A.; Drogosz, J.; Długosz, A.; Krajewska, U.; Mirowski, M.; Janecki, T. Synthesis of 3-methylidene-1-tosyl-2,3-dihydroquinolin-4(1H)-ones as potent cytotoxic agents. Chem. Biodivers. 2018, 15, e1800242. [Google Scholar] [CrossRef]
- Degterev, A.; Boyce, M.; Yuan, J. A decade of caspases. Oncogene 2003, 22, 8543–8567. [Google Scholar] [CrossRef] [Green Version]
- Roos, W.P.; Kaina, B. DNA damage-induced cell death by apoptosis. Trends Mol. Med. 2006, 12, 440–450. [Google Scholar] [CrossRef]
- Dlugosz, A.; Janecka, A. ABC Transporters in the Development of Multidrug Resistance in Cancer Therapy. Curr. Pharm. Des. 2016, 22, 4705–4716. [Google Scholar] [CrossRef]
- de Figueiredo-Pontes, L.L.; Pintão, M.C.; Oliveira, L.C.; Dalmazzo, L.F.; Jácomo, R.H.; Garcia, A.B.; Falcão, R.P.; Rego, E.M. Determination of P-glycoprotein, MDR-related protein 1, breast cancer resistance protein, and lung-resistance protein expression in leukemic stem cells of acute myeloid leukemia. Cytom. B Clin. Cytom. 2008, 74, 163–168. [Google Scholar] [CrossRef]
- Robey, R.W.; Pluchino, K.M.; Hall, M.D.; Fojo, A.T.; Bates, S.E. Gottesman MM. Revisiting the role of ABC transporters in multidrug-resistant cancer. Nat. Rev. Cancer 2018, 18, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Winer, J.; Jung, C.K.; Shackel, I.; Williams, P.M. Development and Validation of Real-Time Quantitative Reverse Transcriptase–Polymerase Chain Reaction for Monitoring Gene Expression in Cardiac Myocytes. Anal. Biochem. 1999, 15, 41–49. [Google Scholar] [CrossRef] [PubMed]
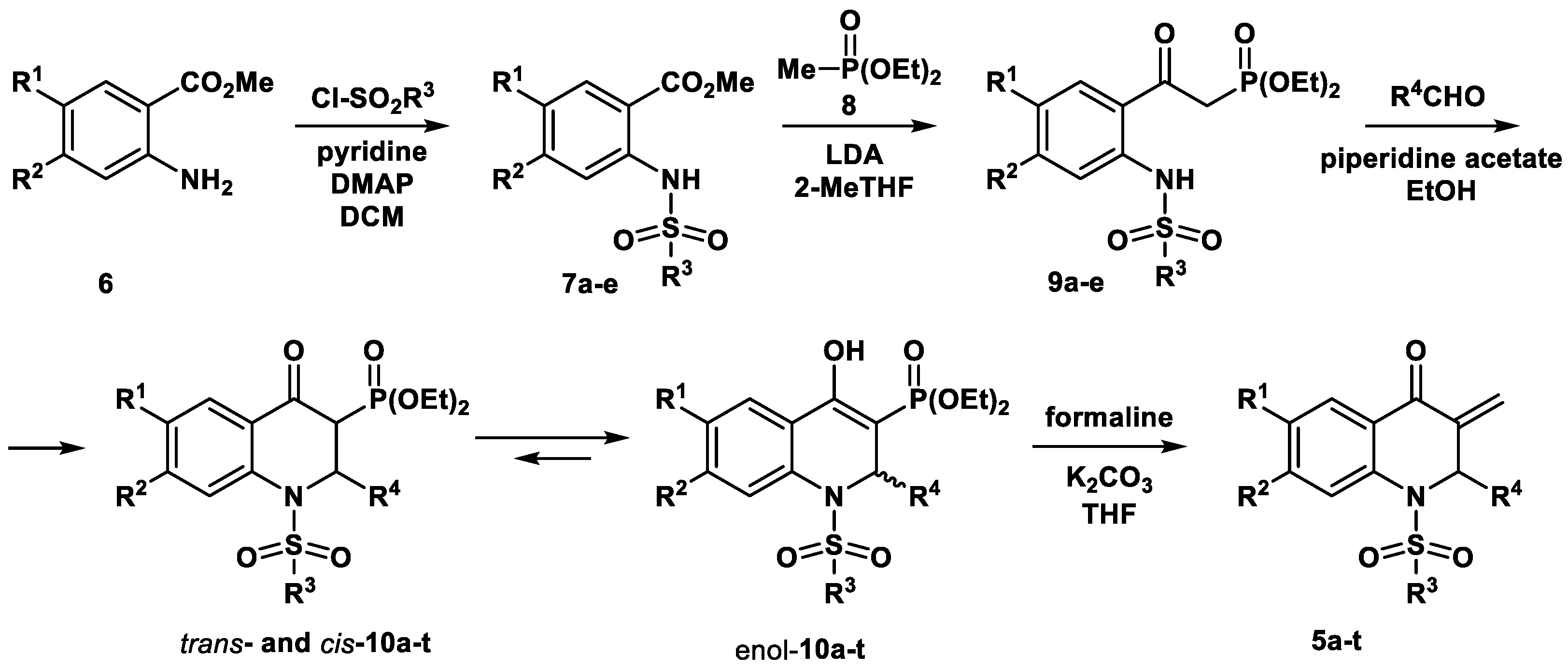




| Compound | R1 | R2 | R3 | R4 | 10 | 5 | |
|---|---|---|---|---|---|---|---|
| trans-10/cis-10/ enol-10 1 | Yield [%] 2 | Yield [%] 2 | |||||
| a | H | H | Phenyl | Et | 2.0/4.0/94.0 | 46 | 64 |
| b | i-Pr | 2.0/1.0/97.0 | 72 | 29 | |||
| c | Phenyl | 1.0/–/99.0 | 71 | 51 | |||
| d | 4-Methoxyphenyl | 1.0/–/99.0 | 87 | 52 | |||
| e | H | H | 4-Chlorophenyl | Et | 1.5/2.5/96.0 | 80 | 94 |
| f | i-Pr | 2.5/0.5/97.0 | 54 | 80 | |||
| g | Phenyl | 1.0/–/99.0 | 73 | 56 | |||
| h | 4-Methoxyphenyl | 1.0/–/99.0 | 77 | 95 | |||
| i | OMe | OMe | 4-Methylphenyl | Et | 5.0/25.0/70.0 | 65 | 42 |
| j | i-Pr | 8.0/3.0/89.0 | 51 | 85 | |||
| k | Phenyl | 3.0/2.0/95.0 | 69 | 76 | |||
| l | 4-Methoxyphenyl | 2.0/2.0/96.0 | 57 | 75 | |||
| m | Cl | H | 4-Methylphenyl | Et | 1.0/2.0/97.0 | 88 | 13 |
| n | i-Pr | 1.5/0.5/98.0 | 85 | 33 | |||
| o | Phenyl | 1.0/–/99.0 | 75 | 52 | |||
| p | 4-Methoxyphenyl | 1.0/–/99.0 | 63 | 75 | |||
| q | Br | H | 4-Methylphenyl | Et | 1.0/3.0/96.0 | 60 | 64 |
| r | i-Pr | 1.5/0.5/98.0 | 77 | 48 | |||
| s | Phenyl | 1.0/–/99.0 | 76 | 51 | |||
| t | 4-Methoxyphenyl | 1.0/–/99.0 | 73 | 38 | |||
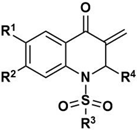 5a-t | ||||||||
|---|---|---|---|---|---|---|---|---|
| Compound | R1 | R2 | R3 | R4 | IC50 [µM] 1 | |||
| MCF-7 | HL-60 | HUVEC | HUVEC/HL-60 IC50 Ratio | |||||
| 5a | H | H | Phenyl | Et | 2.74 ± 0.07 | 0.34 ± 0.03 | 1.87 ± 0.02 | 5.50 |
| 5b | i-Pr | 0.83 ± 0.02 | 0.29 ± 0.02 | 0.78 ± 0.21 | 2.67 | |||
| 5c | Phenyl | 2.25 ± 0.20 | 1.36 ± 0.13 | |||||
| 5d | 4-Methoxyphenyl | 1.21 ± 0.24 | 1.12 ± 0.37 | |||||
| 5e | H | H | 4-Chlorophenyl | Et | 1.61 ± 0.07 | 0.37 ± 0.05 | 0.75 ± 0.00 | 2.03 |
| 5f | i-Pr | 0.86 ± 0.02 | 0.23 ± 0.01 | 0.99 ± 0.07 | 4.30 | |||
| 5g | Phenyl | 1.89 ± 0.03 | 0.78 ± 0.07 | |||||
| 5h | 4-Methoxyphenyl | 2.10 ± 0.16 | 0.92 ± 0.01 | |||||
| 5i | OMe | OMe | 4-Methylphenyl | Et | 1.01 ± 0.05 | 0.37 ± 0.02 | 0.42 ± 0.08 | 1.14 |
| 5j | i-Pr | 1.44 ± 0.07 | 0.43 ± 0.02 | 0.99 ± 0.07 | 2.30 | |||
| 5k | Phenyl | 1.46 ± 0.09 | 0.58 ± 0.02 | |||||
| 5l | 4-Methoxyphenyl | 1.15 ± 0.04 | 1.48 ± 0.06 | |||||
| 5m | Cl | H | 4-Methylphenyl | Et | 3.48 ± 0.42 | 1.75 ± 0.03 | ||
| 5n | i-Pr | 1.65 ± 0.05 | 0.67 ± 0.01 | 0.68 ± 0.03 | 1.01 | |||
| 5o | Phenyl | 3.67 ± 0.41 | 2.04 ± 0.05 | |||||
| 5p | 4-Methoxyphenyl | 3.60 ± 0.19 | 2.07 ± 0.02 | |||||
| 5q | Br | H | 4-Methylphenyl | Et | 1.83 ± 0.15 | 0.61 ± 0.04 | 1.00 ± 0.22 | 1.64 |
| 5r | i-Pr | 0.83 ± 0.02 | 0.19 ± 0.03 | 0.47 ± 0.03 | 2.47 | |||
| 5s | Phenyl | 2.67 ± 0.05 | 0.62 ± 0.01 | |||||
| 5t | 4-Methoxyphenyl | 2.81 ± 0.05 | 0.74 ± 0.06 | |||||
| Carboplatin | 2.90 ± 0.10 | 3.80 ± 0.45 | 5.35± 0.05 | 1.41 | ||||
| Gene | Primer Sequences | |
|---|---|---|
| Forward Primer | Reverse Primer | |
| GAPDH | 5′ GTCGCTGTTGAAGTCAGAGGAG 3′ | 5′ CGTGTCAGTGGTGGACCTGAC 3′ |
| ABCB1 | 5′ GTGGGGCAAGTCAGTTCATT 3′ | 5′ TCTTCACCTCCAGGCTCAGT 3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaskulska, A.; Gach-Janczak, K.; Drogosz-Stachowicz, J.; Janecki, T.; Janecka, A.E. Synthesis and Anticancer Properties of New 3-Methylidene-1-sulfonyl-2,3-dihydroquinolin-4(1H)-ones. Molecules 2022, 27, 3597. https://doi.org/10.3390/molecules27113597
Jaskulska A, Gach-Janczak K, Drogosz-Stachowicz J, Janecki T, Janecka AE. Synthesis and Anticancer Properties of New 3-Methylidene-1-sulfonyl-2,3-dihydroquinolin-4(1H)-ones. Molecules. 2022; 27(11):3597. https://doi.org/10.3390/molecules27113597
Chicago/Turabian StyleJaskulska, Agata, Katarzyna Gach-Janczak, Joanna Drogosz-Stachowicz, Tomasz Janecki, and Anna Ewa Janecka. 2022. "Synthesis and Anticancer Properties of New 3-Methylidene-1-sulfonyl-2,3-dihydroquinolin-4(1H)-ones" Molecules 27, no. 11: 3597. https://doi.org/10.3390/molecules27113597
APA StyleJaskulska, A., Gach-Janczak, K., Drogosz-Stachowicz, J., Janecki, T., & Janecka, A. E. (2022). Synthesis and Anticancer Properties of New 3-Methylidene-1-sulfonyl-2,3-dihydroquinolin-4(1H)-ones. Molecules, 27(11), 3597. https://doi.org/10.3390/molecules27113597







