Biotransformation of Benzoate to 2,4,6-Trihydroxybenzophenone by Engineered Escherichia coli
Abstract
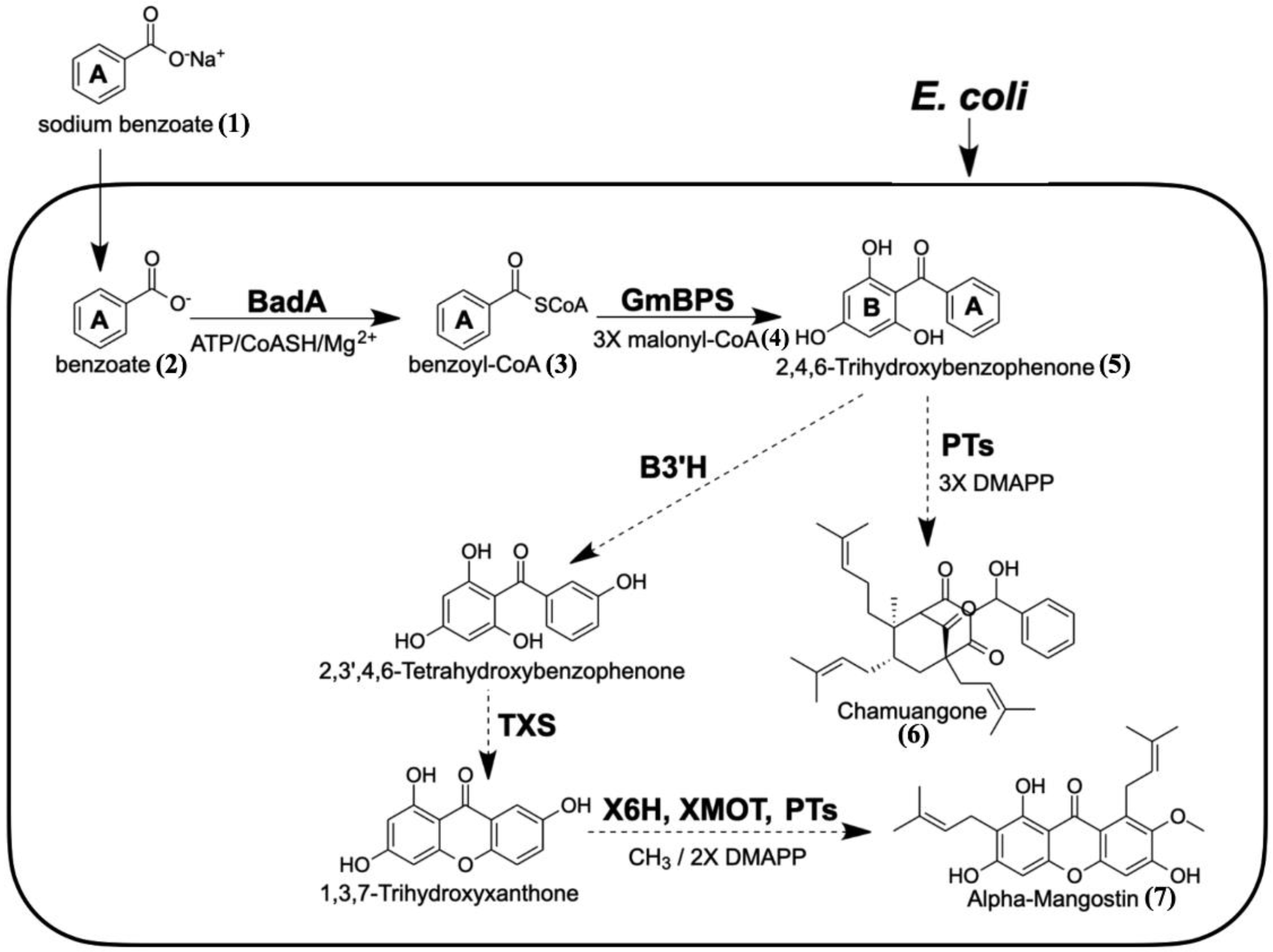
1. Introduction
2. Results
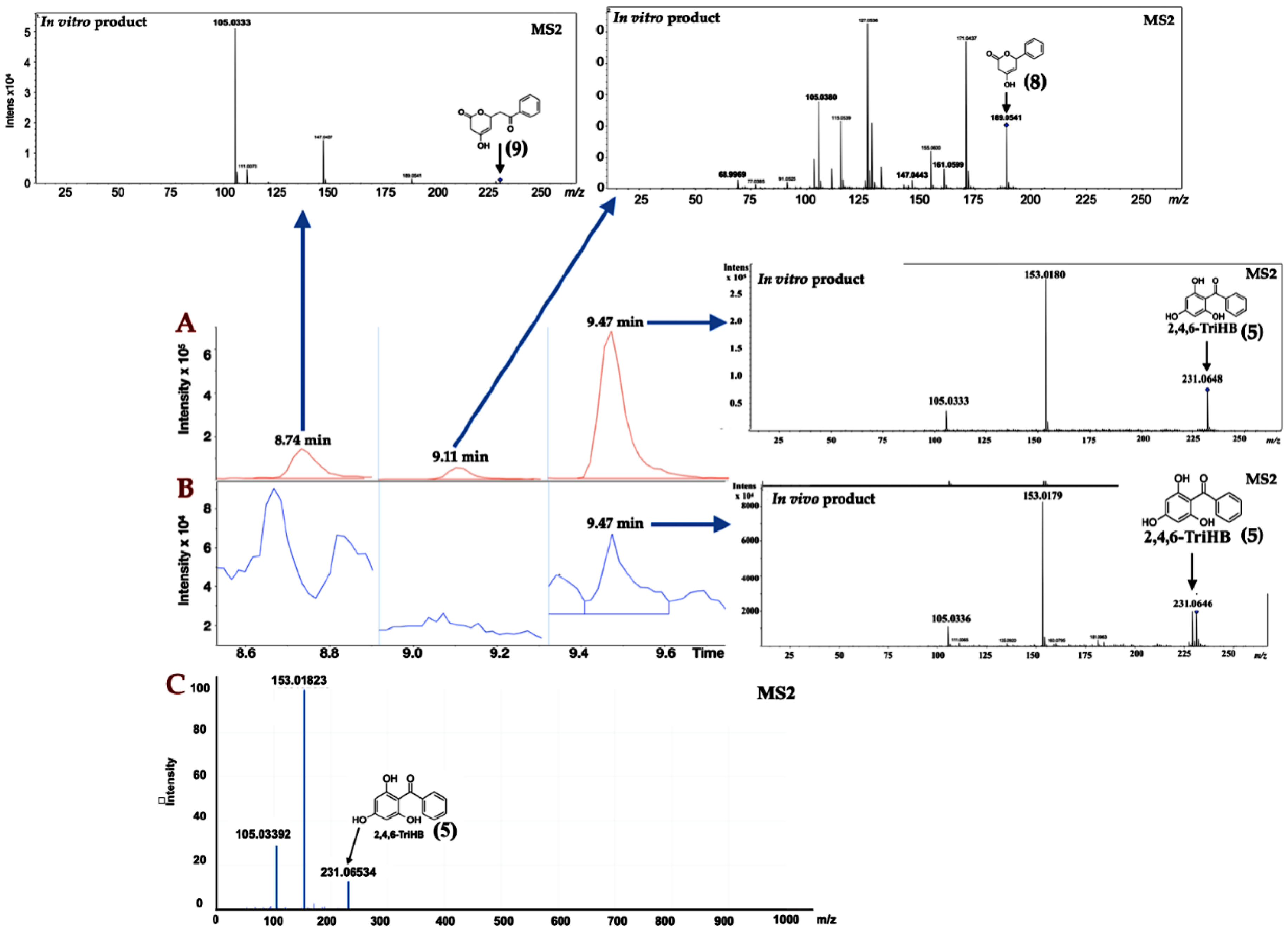
2.1. Identification of 2,4,6-TriHB (5) by Using the High-Resolution LC-ESI-QTOF-MS/MS
2.2. Localisation of 2,4,6-TriHB (5)
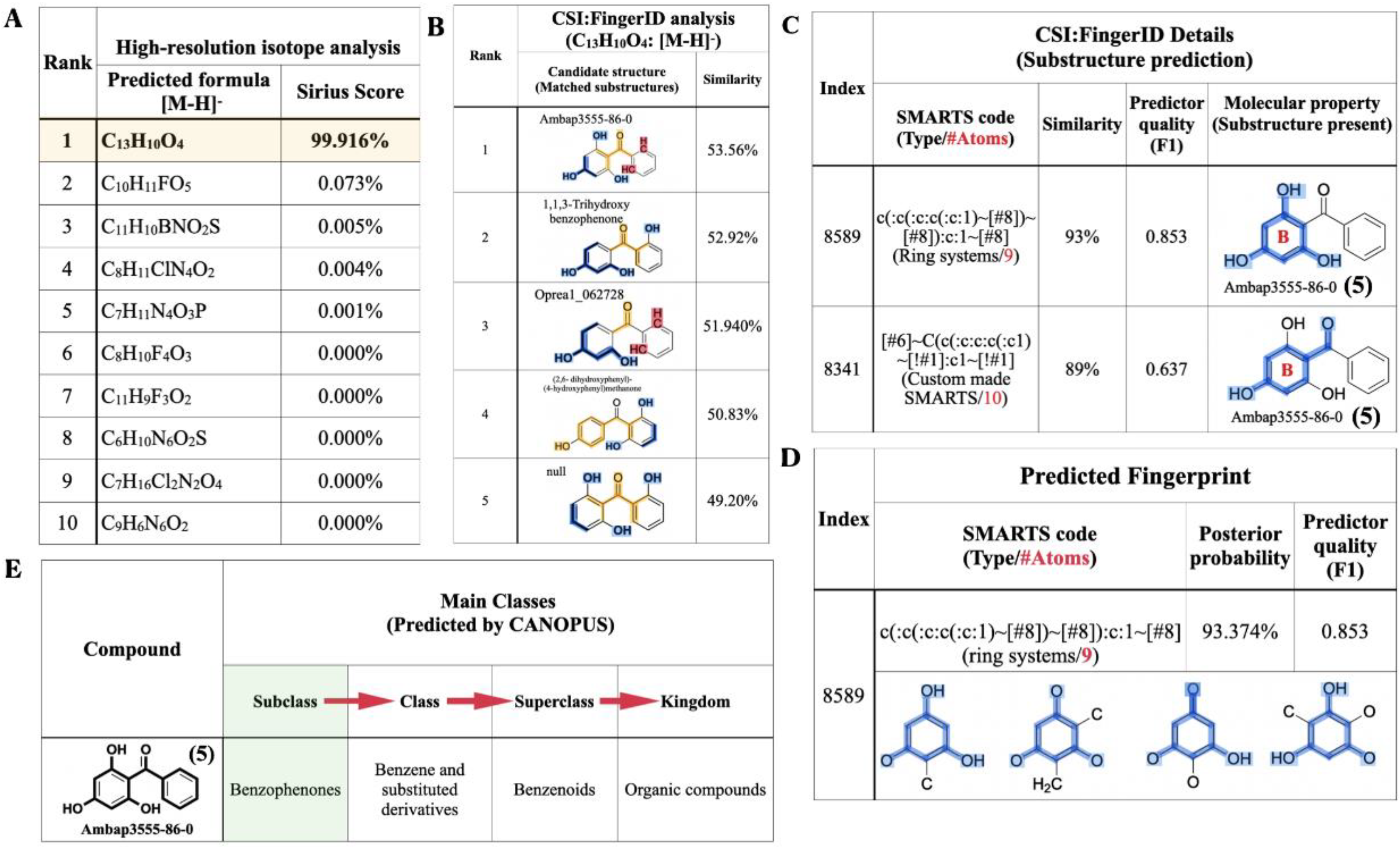
2.3. Structural Confirmation of 2,4,6-TriHB (5) by Using SIRIUS
Molecular Formula Annotation from the Positive Mode Mass Data
2.4. Molecular Formula and Structural Annotation from the Negative Mode Mass Data
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Enzymatic Preparation of 2,4,6-TriHB (5)
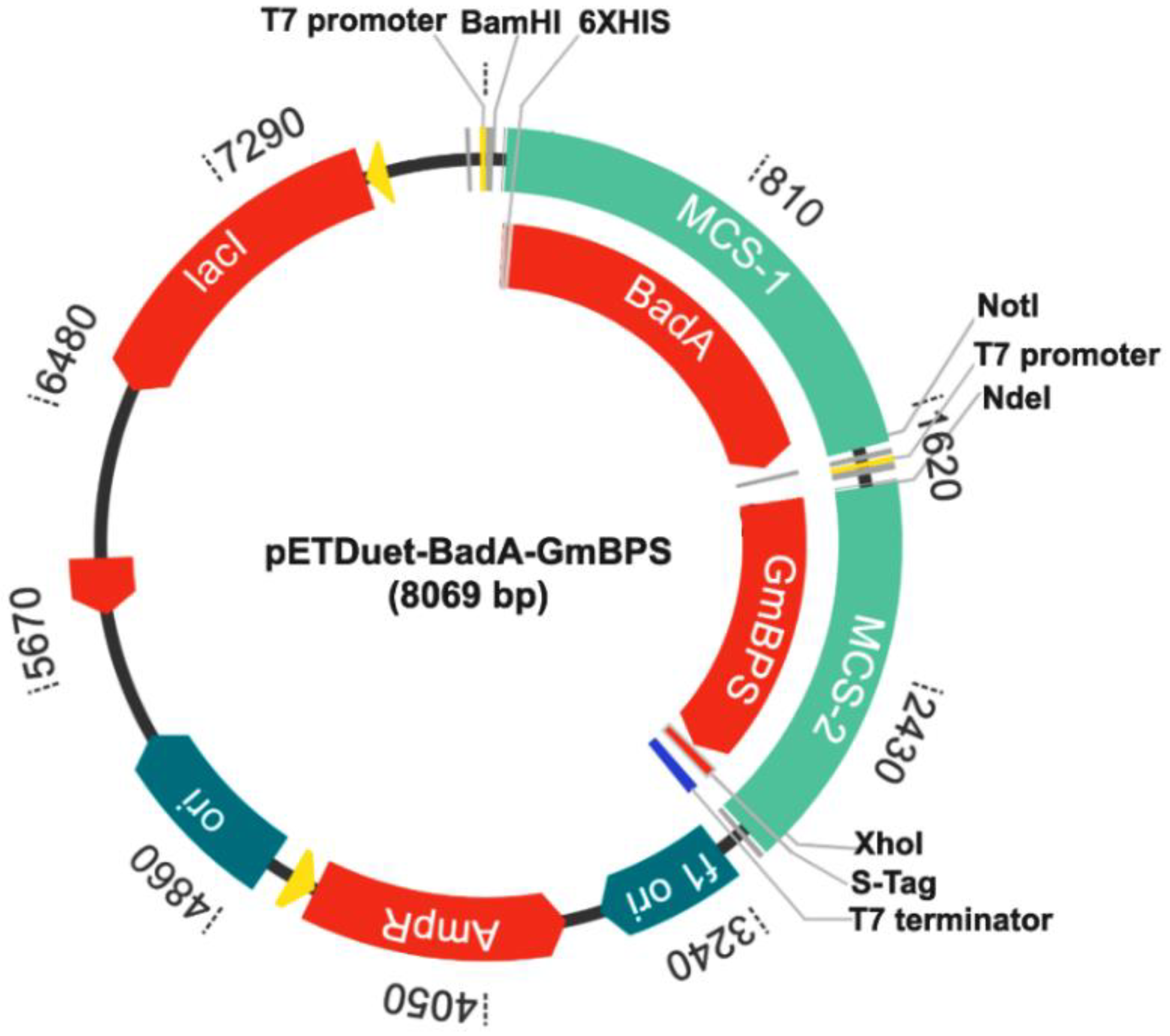
4.3. Construction of Plasmid pETDuet-BadA-GmBPS
4.4. Bioconversion of Benzoate (1) to 2,4,6-TriHB (5)
4.5. Metabolite Extraction from the Cell Pellets
4.6. Extraction of the Secreted Metabolites from the Culture Medium
4.7. Metabolite Detection by Using the High-Resolution LC-ESI-QTOF-MS/MS
4.8. Identification of 2,4,6-TriHB (5) Using an in Silico Mass Prediction Tool
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hemshekhar, M.; Sunitha, K.; Santhosh, M.S.; Devaraja, S.; Kemparaju, K.; Vishwanath, B.S.; Niranjana, S.R.; Girish, K.S. An Overview on Genus Garcinia: Phytochemical and Therapeutical Aspects. Phytochem. Rev. 2011, 10, 325–351. [Google Scholar] [CrossRef]
- Wu, S.B.; Long, C.; Kennelly, E.J. Structural Diversity and Bioactivities of Natural Benzophenones. Nat. Prod. Rep. 2014, 31, 1158–1174. [Google Scholar] [CrossRef]
- Baggett, S.; Mazzola, E.P.; Kennelly, E.J. The Benzophenones: Isolation, Structural Elucidation and Biological Activities. Stud. Nat. Prod. Chem. 2005, 32, 721–771. [Google Scholar] [CrossRef]
- Beerhues, L.; Liu, B. Biosynthesis of Biphenyls and Benzophenones—Evolution of Benzoic Acid-Specific Type III Polyketide Synthases in Plants. Phytochemistry 2009, 70, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Ciochina, R.; Grossman, R.B. Polycyclic Polyprenylated Acylphloroglucinols. Chem. Rev. 2006, 106, 3963–3986. [Google Scholar] [CrossRef] [PubMed]
- Surana, K.; Chaudhary, B.; Diwaker, M.; Sharma, S. Benzophenone: A Ubiquitous Scaffold in Medicinal Chemistry. Medchemcomm 2018, 9, 1803–1817. [Google Scholar] [CrossRef] [PubMed]
- Pardo-Andreu, G.L.; Nuñez-Figueredo, Y.; Tudella, V.G.; Cuesta-Rubio, O.; Rodrigues, F.P.; Pestana, C.R.; Uyemura, S.A.; Leopoldino, A.M.; Alberici, L.C.; Curti, C. The Anticancer Agent Guttiferone—A Permeabilizes Mitochondrial Membrane: Ensuing Energetic and Oxidative Stress Implications. Toxicol. Appl. Pharmacol. 2011, 253, 282–289. [Google Scholar] [CrossRef]
- Seo, E.K.; Wani, M.C.; Wall, M.E.; Navarro, H.; Mukherjee, R.; Farnsworth, N.R.; Kinghorn, A.D. New Bioactive Aromatic Compounds from Vismia guianensis. Phytochemistry 2000, 55, 35–42. [Google Scholar] [CrossRef]
- Iinuma, M.; Tosa, H.; Tanaka, T.; Kanamaru, S.; Asai, F.; Kobayashi, Y.; Miyauchi, K.I.; Shimano, R. Antibacterial Activity of some Garcinia Benzophenone Derivatives against Methicillin-Resistant Staphylococcus aureus. Biol. Pharm. Bull. 1996, 19, 311–314. [Google Scholar] [CrossRef]
- Tantapakul, C.; Phakhodee, W.; Ritthiwigrom, T.; Cheenpracha, S.; Prawat, U.; Deachathai, S.; Laphookhieo, S. Rearranged Benzophenones and Prenylated Xanthones from Garcinia propinqua Twigs. J. Nat. Prod. 2012, 75, 1660–1664. [Google Scholar] [CrossRef]
- Li, E.; Jiang, L.; Guo, L.; Zhang, H.; Che, Y. Pestalachlorides A-C, Antifungal Metabolites from the Plant Endophytic Fungus Pestalotiopsis adusta. Bioorg. Med. Chem. 2008, 16, 7894–7899. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.H.; Baltrusaitis, J.; Gloer, J.B.; Wicklow, D.T. Phomalevones A-C: Dimeric and Pseudodimeric Polyketides from a Fungicolous Hawaiian Isolate of Phoma sp. (Cucurbitariaceae). J. Nat. Prod. 2011, 74, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Baggett, S.; Protiva, P.; Mazzola, E.P.; Yang, H.; Ressler, E.T.; Basile, M.J.; Weinstein, I.B.; Kennelly, E.J. Bioactive Benzophenones from Garcinia xanthochymus Fruits. J. Nat. Prod. 2005, 68, 354–360. [Google Scholar] [CrossRef]
- Acuña, U.M.; Figueroa, M.; Kavalier, A.; Jancovski, N.; Basile, M.J.; Kennelly, E.J. Benzophenones and Biflavonoids from Rheedia edulis. J. Nat. Prod. 2010, 73, 1775–1779. [Google Scholar] [CrossRef] [PubMed]
- Fuller, R.W.; Westergaard, C.K.; Collins, J.W.; Cardellina, J.H.; Boyd, M.R. Vismiaphenones D-G, New Prenylated Benzophenones from Vismia cayennensis. J. Nat. Prod. 1999, 62, 67–69. [Google Scholar] [CrossRef]
- Fuller, R.W.; Blunt, J.W.; Boswell, J.L.; Cardellina, J.H.; Boyd, M.R. Guttiferone F, the First Prenylated Benzophenone from Allanblackia stuhlmannii. J. Nat. Prod. 1999, 62, 130–132. [Google Scholar] [CrossRef]
- Casu, L.; Solinas, M.N.; Saba, A.R.; Cottiglia, F.; Caboni, P.; Floris, C.; Laconi, S.; Pompei, R.; Leonti, M. Benzophenones from the Roots of the Popoluca Amerindian Medicinal Plant Securidaca diversifolia (L.) S.F. Blake. Phytochem. Lett. 2010, 3, 226–229. [Google Scholar] [CrossRef]
- Abe, F.; Nagafuji, S.; Okabe, H.; Akahane, H.; Estrada-Muñiz, E.; Huerta-Reyes, M.; Reyes-Chilpa, R. Trypanocidal Constituents in Plants 3. Leaves of Garcinia intermedia and Heartwood of Calophyllum brasiliense. Biol. Pharm. Bull. 2004, 27, 141–143. [Google Scholar] [CrossRef]
- Pereira, I.O.; Marques, M.J.; Pavan, A.L.R.; Codonho, B.S.; Barbiéri, C.L.; Beijo, L.A.; Doriguetto, A.C.; Costa, D.D.M.; dos Santos, M.H. Leishmanicidal Activity of Benzophenones and Extracts from Garcinia brasiliensis Mart. Fruits. Phytomedicine 2010, 17, 339–345. [Google Scholar] [CrossRef]
- Chen, J.J.; Lin, W.J.; Liao, C.H.; Shieh, P.C. Anti-Inflammatory Benzenoids from Antrodia camphorata. J. Nat. Prod. 2007, 70, 989–992. [Google Scholar] [CrossRef]
- Xin, W.B.; Man, X.H.; Zheng, C.J.; Jia, M.; Jiang, Y.P.; Zhao, X.X.; Jin, G.L.; Mao, Z.J.; Huang, H.Q.; Qin, L.P. Prenylated Phloroglucinol Derivatives from Hypericum sampsonii. Fitoterapia 2012, 83, 1540–1547. [Google Scholar] [CrossRef]
- Carroll, A.R.; Suraweera, L.; King, G.; Rali, T.; Quinn, R.J. Guttiferones O and P, Prenylated Benzophenone MAPKAPK-2 Inhibitors from Garcinia solomonensis. J. Nat. Prod. 2009, 72, 1699–1701. [Google Scholar] [CrossRef]
- Duan, Y.H.; Dai, Y.; Wang, G.H.; Chen, H.F.; Gao, H.; Chen, J.B.; Yao, X.S.; Zhang, X.K. Xanthone and Benzophenone Glycosides from the Stems of Cratoxylum formosum ssp. Pruniflorum. Chem. Pharm. Bull. 2011, 59, 231–234. [Google Scholar] [CrossRef]
- Sakunpak, A.; Matsunami, K.; Otsuka, H.; Panichayupakaranant, P. Isolation of Chamuangone, a Cytotoxic Compound against Leishmania Major and Cancer Cells from Garcinia cowa Leaves and its HPLC Quantitative Determination Method. J. Can. Res. Updates 2017, 6, 38–45. [Google Scholar] [CrossRef]
- Sakunpak, A.; Panichayupakaranant, P. Antibacterial Activity of Thai Edible Plants against Gastrointestinal Pathogenic Bacteria and Isolation of a New Broad Spectrum Antibacterial Polyisoprenylated Benzophenone, Chamuangone. Food Chem. 2012, 130, 826–831. [Google Scholar] [CrossRef]
- Sae-Lim, P.; Yuenyongsawad, S.; Panichayupakaranant, P. Chamuangone-Enriched Garcinia cowa Leaf Extract with Rice Bran Oil: Extraction and Cytotoxic Activity against Cancer Cells. Pharmacogn. Mag. 2019, 15, 183. [Google Scholar] [CrossRef]
- Shan, T.; Ma, Q.; Guo, K.; Liu, J.; Li, W.; Wang, F.; Wu, E. Xanthones from Mangosteen Extracts as Natural Chemopreventive Agents: Potential Anticancer Drugs. Curr. Mol. Med. 2011, 11, 666–677. [Google Scholar] [CrossRef] [PubMed]
- Watanapokasin, R.; Jarinthanan, F.; Jerusalmi, A.; Suksamrarn, S.; Nakamura, Y.; Sukseree, S.; Uthaisang-Tanethpongtamb, W.; Ratananukul, P.; Sano, T. Potential of Xanthones from Tropical Fruit Mangosteen as Anti-Cancer Agents: Caspase-Dependent Apoptosis Induction in Vitro and in Mice. Appl. Biochem. Biotechnol. 2010, 162, 1080–1094. [Google Scholar] [CrossRef]
- Chen, G.; Li, Y.; Wang, W.; Deng, L. Bioactivity and Pharmacological Properties of α-Mangostin from the Mangosteen Fruit: A Review. Expert Opin. Ther. Pat. 2018, 28, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Nie, Y.; Liang, X.; Ji, L.; Hu, S.; You, Q.; Wang, F.; Ye, H.; Wang, J. A Concise and Efficient Total Synthesis of α-Mangostin and β-Mangostin from Garcinia Mangostana. Nat. Prod. Commun. 2013, 8, 1101–1103. [Google Scholar] [CrossRef] [PubMed]
- Pathak, V.P.; Khanna, R.N. Synthesis of Naturally Occurring Prenylated Benzophenones. Vismiaphenone A, Vismiaphenone B, and Isovismiaphenone B. Bull. Chem. Soc. Jpn. 1982, 55, 2264–2266. [Google Scholar] [CrossRef]
- Trantas, E.A.; Koffas, M.A.G.; Xu, P.; Ververidis, F. When Plants Produce not Enough or at all: Metabolic Engineering of Flavonoids in Microbial Hosts. Front. Plant Sci. 2015, 6, 7. [Google Scholar] [CrossRef]
- Resende, D.I.S.P.; Durães, F.; Maia, M.; Sousa, E.; Pinto, M.M.M. Recent Advances in the Synthesis of Xanthones and Azaxanthones. Org. Chem. Front. 2020, 7, 3027–3066. [Google Scholar] [CrossRef]
- Perkins, C.; Siddiqui, S.; Puri, M.; Demain, A.L. Biotechnological Applications of Microbial Bioconversions. Crit. Rev. Biotechnol. 2016, 36, 1050–1065. [Google Scholar] [CrossRef] [PubMed]
- Pham, J.V.; Yilma, M.A.; Feliz, A.; Majid, M.T.; Maffetone, N.; Walker, J.R.; Kim, E.; Cho, H.J.; Reynolds, J.M.; Song, M.C.; et al. A Review of the Microbial Production of Bioactive Natural Products and Biologics. Front. Microbiol. 2019, 10, 1404. [Google Scholar] [CrossRef] [PubMed]
- Chubukov, V.; Mukhopadhyay, A.; Petzold, C.J.; Keasling, J.D.; Martín, H.G. Synthetic and Systems Biology for Microbial Production of Commodity Chemicals. NPJ Syst. Biol. Appl. 2018, 2, 16009. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, B.A.; Khosla, C. Biosynthesis of Polyketides in Heterologous Hosts. Microbiol. Mol. Biol. Rev. 2001, 65, 106–118. [Google Scholar] [CrossRef]
- Lussier, F.X.; Colatriano, D.; Wiltshire, Z.; Page, J.E.; Martin, V.J.J. Engineering Microbes for Plant Polyketide Biosynthesis. Comput. Struct. Biotechnol. J. 2012, 3, e201210020. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.P.; Parajuli, P.; Koffas, M.A.G.; Sohng, J.K. Microbial Production of Natural and Non-Natural Flavonoids: Pathway Engineering, Directed Evolution and Systems/Synthetic Biology. Biotechnol. Adv. 2016, 34, 634–662. [Google Scholar] [CrossRef]
- Pickens, L.B.; Tang, Y.; Chooi, Y.H. Metabolic Engineering for the Production of Natural Products. Annu. Rev. Chem. Biomol. Eng. 2011, 2, 211–236. [Google Scholar] [CrossRef]
- Nualkaew, N.; Morita, H.; Shimokawa, Y.; Kinjo, K.; Kushiro, T.; De-Eknamkul, W.; Ebizuka, Y.; Abe, I. Benzophenone Synthase from Garcinia mangostana L. Pericarps. Phytochemistry 2012, 77, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Wang, H.; Ye, H.; Du, Z.; Zhang, Y.; Beerhues, L.; Liu, B. Differential Expression of Benzophenone Synthase and Chalcone Synthase in Hypericum Sampsonii. Nat. Prod. Commun. 2012, 7, 1615–1618. [Google Scholar] [CrossRef]
- Liu, B.; Falkenstein-Paul, H.; Schmidt, W.; Beerhues, L. Benzophenone Synthase and Chalcone Synthase from Hypericum androsaemum Cell Cultures: cDNA Cloning, Functional Expression, and Site-Directed Mutagenesis of Two Polyketide Synthases. Plant J. 2003, 34, 847–855. [Google Scholar] [CrossRef]
- Belkheir, A.K.; Gaid, M.; Liu, B.; Hänsch, R.; Beerhues, L. Benzophenone Synthase and Chalcone Synthase Accumulate in the Mesophyll of Hypericum perforatum Leaves at Different Developmental Stages. Front. Plant Sci. 2016, 7, 921. [Google Scholar] [CrossRef]
- Ahluwalia, V.K.; Bhagat, P.; Aggarwal, R.; Chandra, R. Intermediates for Organic Synthesis, 1st ed.; I.K. International Pvt. Ltd.: New Delhi, India, 2005. [Google Scholar]
- Chakraborty, D. Production of Benzophenone Derivatives in Engineered Yeast. Master’s Thesis, Indian Institute of Technology Kharagpur, Kharagpur, India, 2015. [Google Scholar]
- Geissler, J.F.; Harwood, C.S.; Gibson, J. Purification and Properties of Benzoate-Coenzyme A Ligase, a Rhodopseudomonas palustris Enzyme Involved in the Anaerobic Degradation of Benzoate. J. Bacteriol. 1988, 170, 1709–1714. [Google Scholar] [CrossRef]
- Egland, P.G.; Gibson, J.; Harwood, C.S. Benzoate-Coenzyme A Ligase, Encoded by badA, is one of Three Ligases Able to Catalyze Benzoyl-Coenzyme A Formation During Anaerobic Growth of Rhodopseudomonas palustris on Benzoate. J. Bacteriol. 1995, 177, 6545–6551. [Google Scholar] [CrossRef]
- Zirkle, R.; Ligon, J.M.; Molnár, I. Heterologous Production of the Antifugal Polyketide Antibiotic Soraphen A of Sorangium Cellulosum So ce26 in Streptomyces lividans. Microbiology 2004, 150, 2761–2774. [Google Scholar] [CrossRef] [PubMed]
- Tolia, N.H.; Joshua-Tor, L. Strategies for Protein Coexpression in Escherichia coli. Nat. Methods 2006, 3, 55–64. [Google Scholar] [CrossRef]
- Dührkop, K.; Fleischauer, M.; Ludwig, M.; Aksenov, A.A.; Melnik, A.V.; Meusel, M.; Dorrestein, P.C.; Rousu, J.; Böcker, S. SIRIUS 4: A Rapid Tool for Turning Tandem Mass Spectra Into Metabolite Structure Information. Nat. Methods 2019, 16, 299–302. [Google Scholar] [CrossRef] [PubMed]
- Böcker, S.; Letzel, M.C.; Lipták, Z.; Pervukhin, A. SIRIUS: Decomposing Isotope Patterns for Metabolite Identification. Bioinformatics 2009, 25, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Dührkop, K.; Shen, H.; Meusel, M.; Rousu, J.; Böcker, S. Searching Molecular Structure Databases with Tandem Mass Spectra using CSI: FingerID. Proc. Natl. Acad. Sci. USA 2015, 112, 12580–12585. [Google Scholar] [CrossRef]
- Böcker, S.; Dührkop, K. Fragmentation Trees Reloaded. J. Cheminform. 2016, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Dührkop, K.; Nothias, L.F.; Fleischauer, M.; Reher, R.; Ludwig, M.; Hoffmann, M.A.; Petras, D.; Gerwick, W.H.; Rousu, J.; Dorrestein, P.C.; et al. Systematic Classification of Unknown Metabolites using High-Resolution Fragmentation Mass Spectra. Nat. Biotechnol. 2020, 39, 462–471. [Google Scholar] [CrossRef] [PubMed]
- Braslavsky, S.E. Glossary of Terms used in Photochemistry 3rd edition (IUPAC Recommendations 2006). Pure Appl. Chem 2007, 79, 293–465. [Google Scholar] [CrossRef]
- Yang, X.W.; Grossman, R.B.; Xu, G. Research Progress of Polycyclic Polyprenylated Acylphloroglucinols. Chem. Rev. 2018, 118, 3508–3558. [Google Scholar] [CrossRef] [PubMed]
- Watts, K.T.; Lee, P.C.; Schmidt-Dannert, C. Biosynthesis of Plant-Specific Stilbene Polyketides in Metabolically Engineered Escherichia coli. BMC Biotechnol. 2006, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Beekwilder, J.; Wolswinkel, R.; Jonker, H.; Hall, R.; De Rie Vos, C.H.; Bovy, A. Production of Resveratrol in Recombinant Microorganisms. Appl. Environ. Microbiol. 2006, 72, 5670–5672. [Google Scholar] [CrossRef]
- Leonard, E.; Yan, Y.; Fowler, Z.L.; Li, Z.; Lim, C.G.; Lim, K.H.; Koffas, M.A.G. Strain Improvement of Recombinant Escherichia coli for Efficient Production of Plant Flavonoids. Mol. Pharm. 2008, 5, 257–265. [Google Scholar] [CrossRef]
- Horinouchi, S. Combinatorial Biosynthesis of Non-Bacterial and Unnatural Flavonoids, Stilbenoids and Curcuminoids by Microorganisms. J. Antibiot. 2008, 61, 709–728. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, K.; Xu, S.; Wu, J.; Liu, P.; Du, G.; Li, J.; Chen, J. Identification of Membrane Proteins Associated with Phenylpropanoid Tolerance and Transport in Escherichia coli BL21. J. Proteom. 2015, 113, 15–28. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, B.H.; Liu, Z.N.; Qiao, J.; Zhao, G.R. Combinatorial Optimization of Resveratrol Production in Engineered E. coli. J. Agric. Food Chem. 2018, 66, 13444–13453. [Google Scholar] [CrossRef] [PubMed]
- Leduc, D.; Battesti, A.; Bouveret, E. The hotdog thioesterase EntH (YbdB) plays a Role in Vivo in Optimal Enterobactin Biosynthesis by Interacting with the ArCP Domain of EntB. J. Bacteriol. 2007, 189, 7112–7126. [Google Scholar] [CrossRef]
- Wu, R.; Latham, J.A.; Chen, D.; Farelli, J.; Zhao, H.; Matthews, K.; Allen, K.N.; Dunaway-Mariano, D. Structure and Catalysis in the Escherichia coli Hotdog-Fold Thioesterase Paralogs YdiI and YbdB. Biochemistry 2014, 53, 4788–4805. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Mahajani, M.; Wang, J.; Yang, Y.; Yuan, Q.; Yan, Y.; Lin, Y. Elevating 4-Hydroxycoumarin Production through Alleviating Thioesterase-Mediated Salicoyl-CoA Degradation. Metab. Eng. 2017, 42, 59–65. [Google Scholar] [CrossRef]
- Wilks, J.C.; Slonczewski, J.L. pH of the Cytoplasm and Periplasm of Escherichia coli: Rapid Measurement by Green Fluorescent Protein Fluorimetry. J. Bacteriol. 2007, 189, 5601–5607. [Google Scholar] [CrossRef]
- Martinez, K.A.; Kitko, R.D.; Mershon, J.P.; Adcox, H.E.; Malek, K.A.; Berkmen, M.B.; Slonczewski, J.L. Cytoplasmic pH response to Acid Stress in Individual Cells of Escherichia coli and Bacillus subtilis Observed by Fluorescence Ratio Imaging Microscopy. Appl. Environ. Microbiol. 2012, 78, 3706–3714. [Google Scholar] [CrossRef]
- Eudes, A.; Benites, V.T.; Wang, G.; Baidoo, E.E.K.; Lee, T.S.; Keasling, J.D.; Loqué, D. Precursor-Directed Combinatorial Biosynthesis of Cinnamoyl, Dihydrocinnamoyl, and Benzoyl Anthranilates in Saccharomyces cerevisiae. PLoS ONE 2015, 10, e0138972. [Google Scholar] [CrossRef]
- Austin, M.B.; Bowman, M.E.; Ferrer, J.L.; Schröder, J.; Noel, J.P. An Aldol Switch Discovered in Stilbene Synthases Mediates Cyclization Specificity of Type III Polyketide Synthases. Chem. Biol. 2004, 11, 1179–1194. [Google Scholar] [CrossRef]
- Lim, Y.P.; Go, M.K.; Yew, W.S. Exploiting the Biosynthetic Potential of Type III Polyketide Synthases. Molecules 2016, 21, 806. [Google Scholar] [CrossRef]
- Katsuyama, Y.; Funa, N.; Miyahisa, I.; Horinouchi, S. Synthesis of Unnatural Flavonoids and Stilbenes by Exploiting the Plant Biosynthetic Pathway in Escherichia coli. Chem. Biol. 2007, 14, 613–621. [Google Scholar] [CrossRef]
- Emwas, A.H.M. The Strengths and Weaknesses of NMR Spectroscopy and Mass Spectrometry with Particular Focus on Metabolomics Research. Methods Mol. Biol. 2015, 1277, 161–193. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.F.; Zhou, B.; Ressom, H.W. Metabolite Identification and Quantitation in LC-MS/MS-based Metabolomics. TrAC Trends Anal. Chem. 2012, 32, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Xiao, J.F.; Tuli, L.; Ressom, H.W. LC-MS-Based Metabolomics. Mol. Biosyst. 2012, 8, 470–481. [Google Scholar] [CrossRef]
- Blaženović, I.; Kind, T.; Ji, J.; Fiehn, O. Software Tools and Approaches for Compound Identification of LC-MS/MS Data in Metabolomics. Metabolites 2018, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Shahi, S.; Ang, C.S.; Mathivanan, S. A High-Resolution Mass Spectrometry-Based Quantitative Metabolomic Workflow Highlights Defects in 5-Fluorouracil Metabolism in Cancer Cells with Acquired Chemoresistance. Biology 2020, 9, 96. [Google Scholar] [CrossRef]
- Mzozoyana, V.; van Heerden, F.R. Synthesis of 3-geranyl- and 3-prenyl-2,4,6-trihydroxybenzophenone. Synth. Commun. 2017, 47, 599–603. [Google Scholar] [CrossRef]
- Chen, R.; Gao, B.; Liu, X.; Ruan, F.; Zhang, Y.; Lou, J.; Feng, K.; Wunsch, C.; Li, S.M.; Dai, J.; et al. Molecular Insights into the Enzyme Promiscuity of an Aromatic Prenyltransferase. Nat. Chem. Biol. 2017, 13, 226–234. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klamrak, A.; Nabnueangsap, J.; Nualkaew, N. Biotransformation of Benzoate to 2,4,6-Trihydroxybenzophenone by Engineered Escherichia coli. Molecules 2021, 26, 2779. https://doi.org/10.3390/molecules26092779
Klamrak A, Nabnueangsap J, Nualkaew N. Biotransformation of Benzoate to 2,4,6-Trihydroxybenzophenone by Engineered Escherichia coli. Molecules. 2021; 26(9):2779. https://doi.org/10.3390/molecules26092779
Chicago/Turabian StyleKlamrak, Anuwatchakij, Jaran Nabnueangsap, and Natsajee Nualkaew. 2021. "Biotransformation of Benzoate to 2,4,6-Trihydroxybenzophenone by Engineered Escherichia coli" Molecules 26, no. 9: 2779. https://doi.org/10.3390/molecules26092779
APA StyleKlamrak, A., Nabnueangsap, J., & Nualkaew, N. (2021). Biotransformation of Benzoate to 2,4,6-Trihydroxybenzophenone by Engineered Escherichia coli. Molecules, 26(9), 2779. https://doi.org/10.3390/molecules26092779





