Development of a Tellurium Speciation Study Using IC-ICP-MS on Soil Samples Taken from an Area Associated with the Storage, Processing, and Recovery of Electrowaste
Abstract
1. Introduction
2. Results and Discussion
2.1. Optimization of Tellurium Speciation
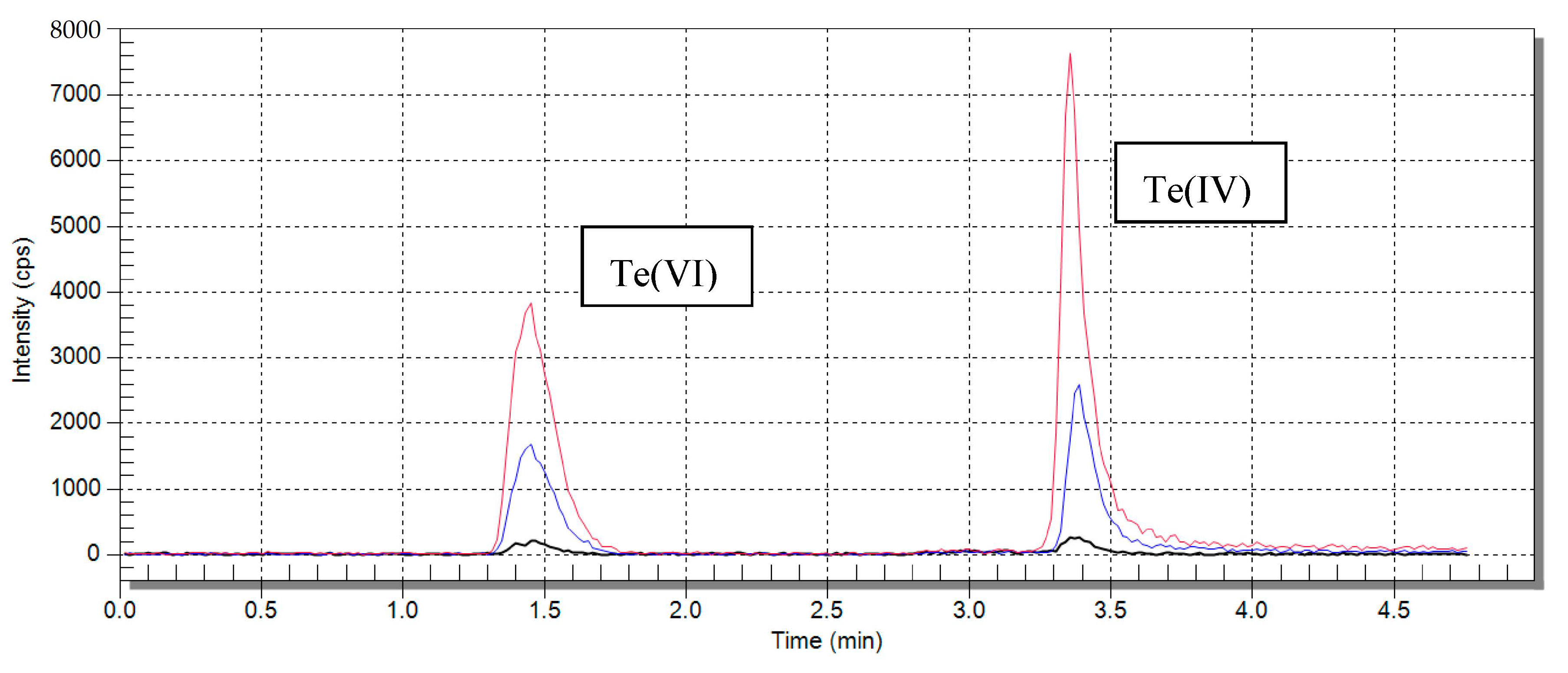
2.1.1. Elution Optimization
2.1.2. The Influence of the Complexing Reagents on Tellurium Species Separation
2.1.3. Optimization of the Chromatographic Separation
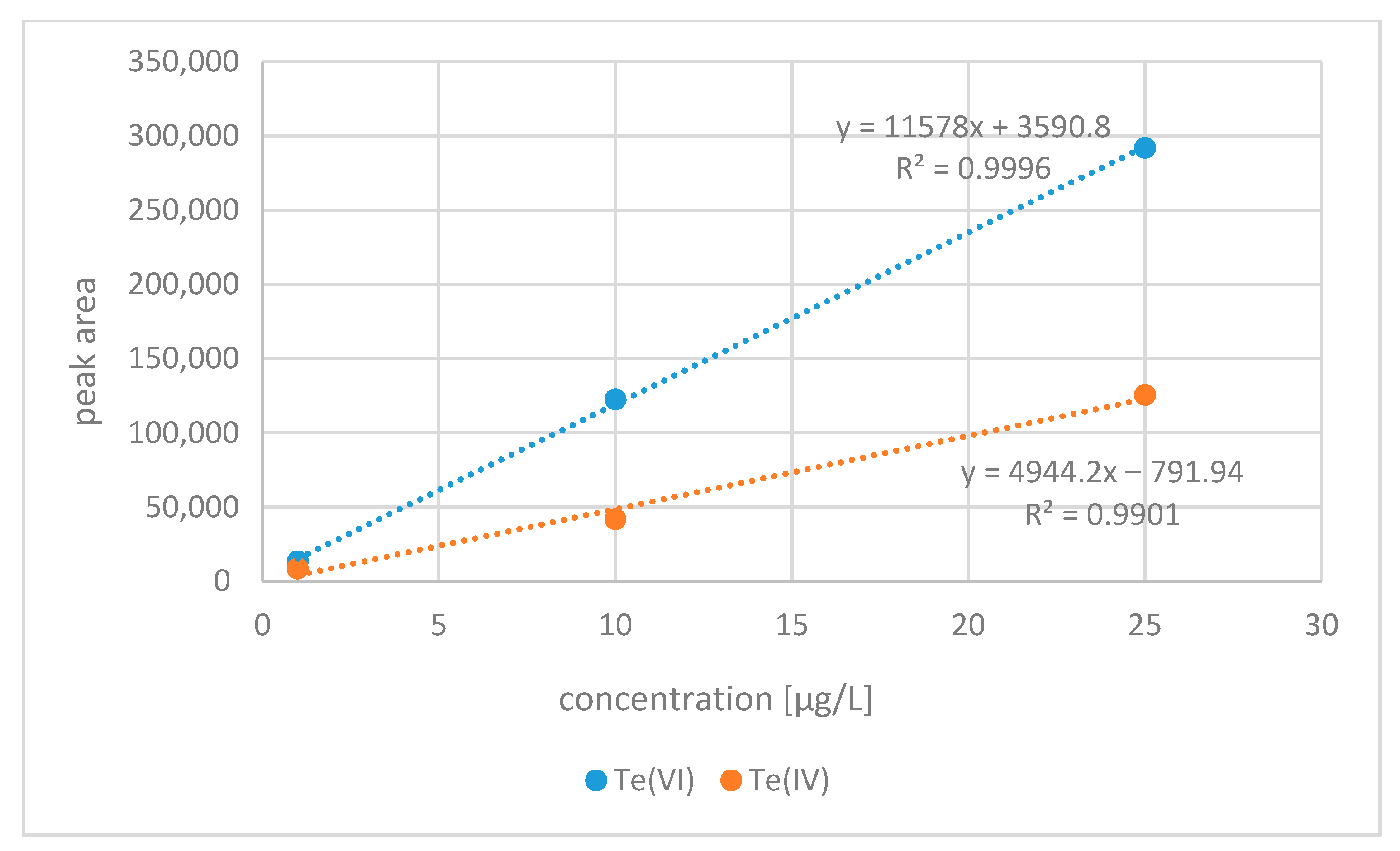
2.1.4. Preparation of Standards
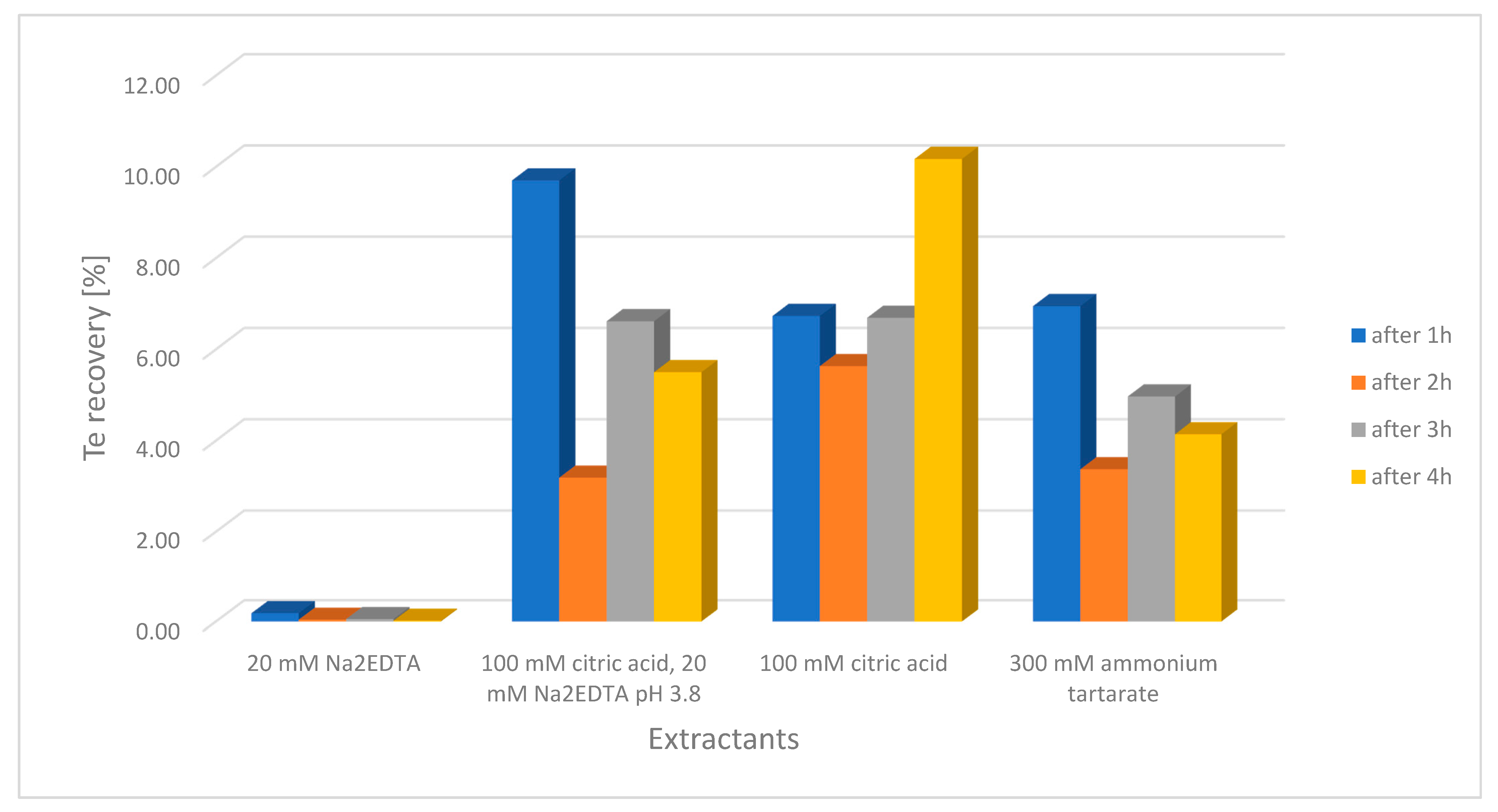
2.1.5. Optimization of Soil Extraction for the IC-ICP-MS Analysis
2.1.6. Sequential Chemical Extraction
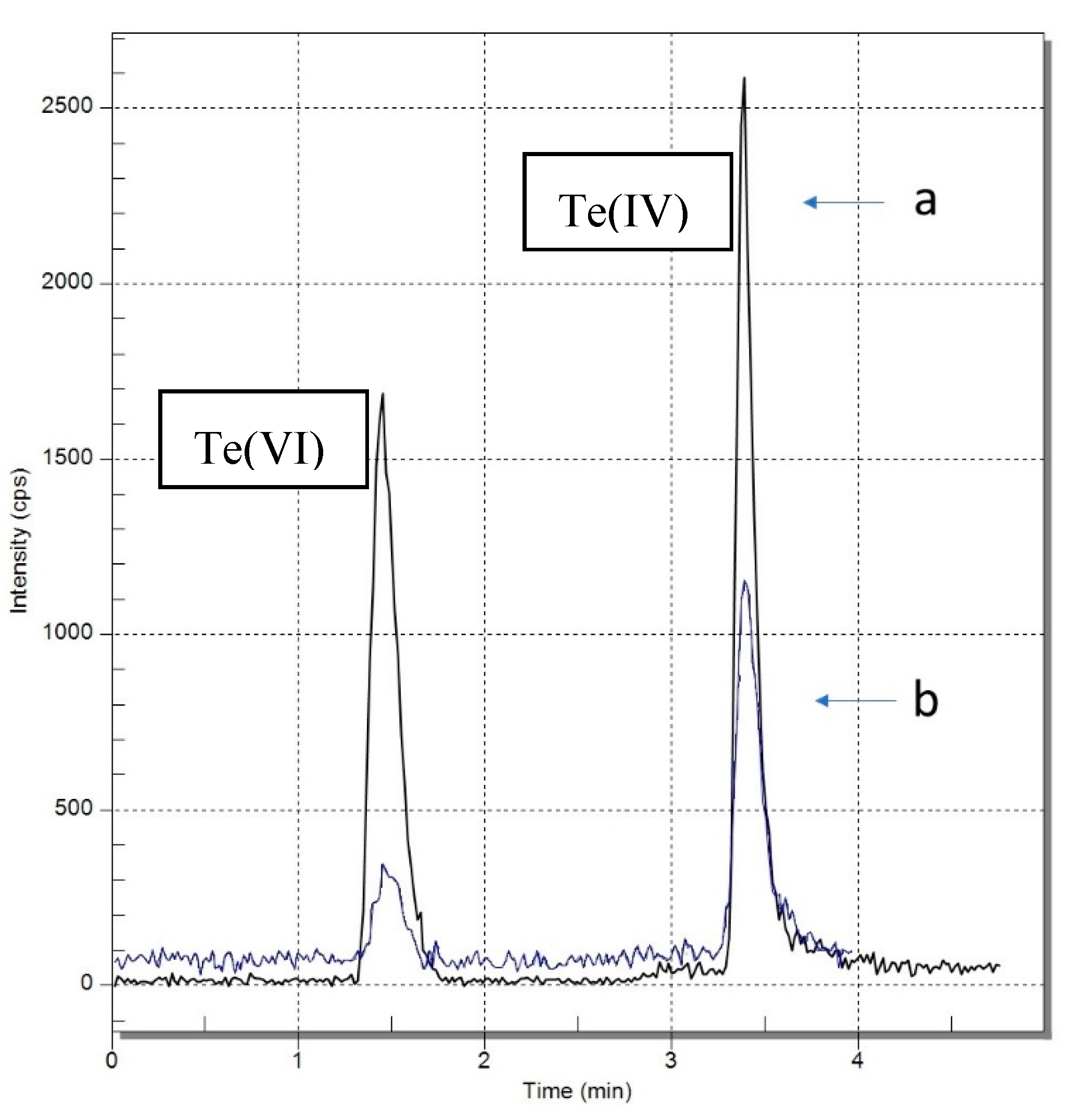
2.1.7. The Matrix Interferences
2.1.8. Quality Control of the Speciation Analysis
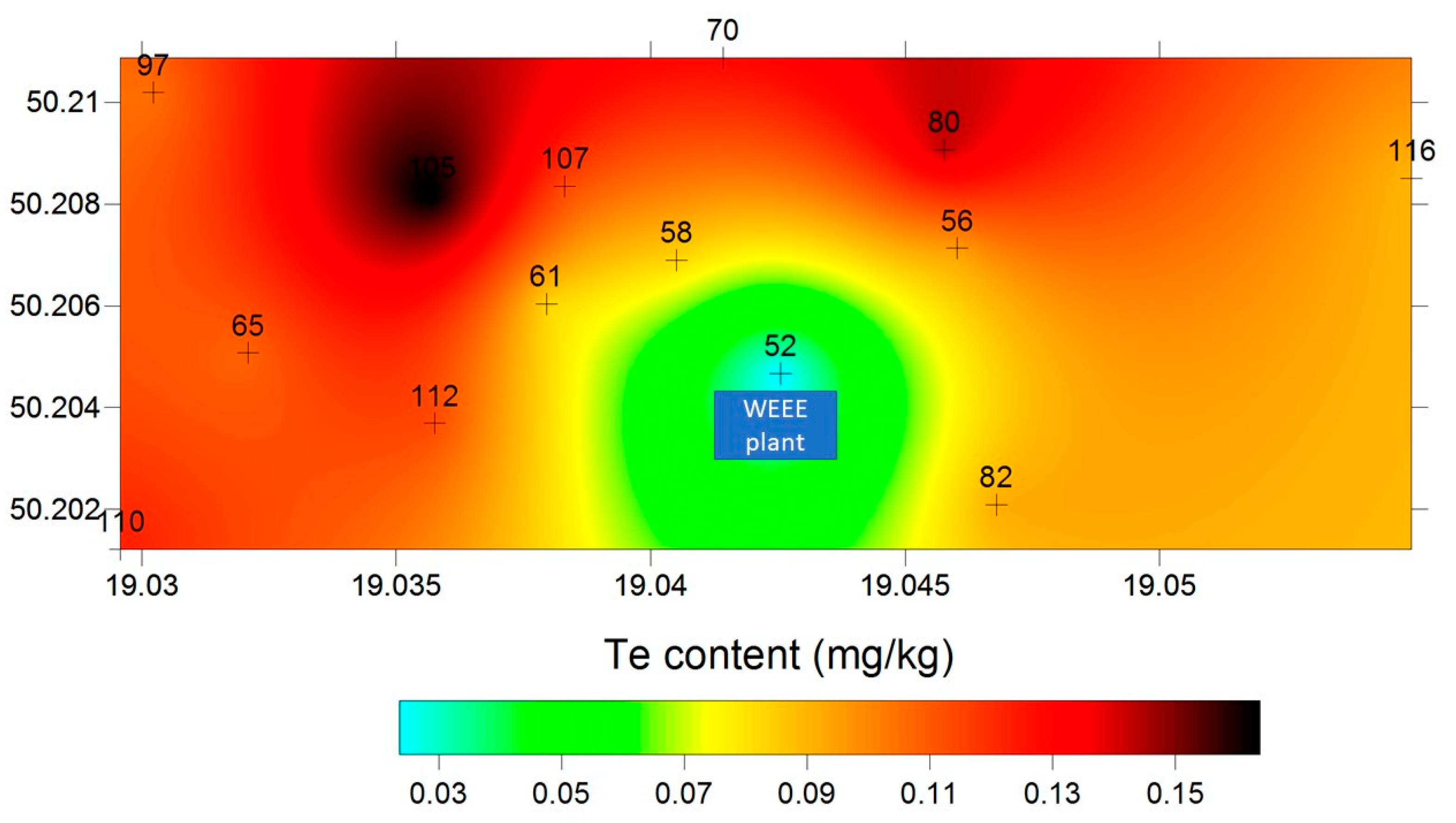
2.2. Total Tellurium Concentration
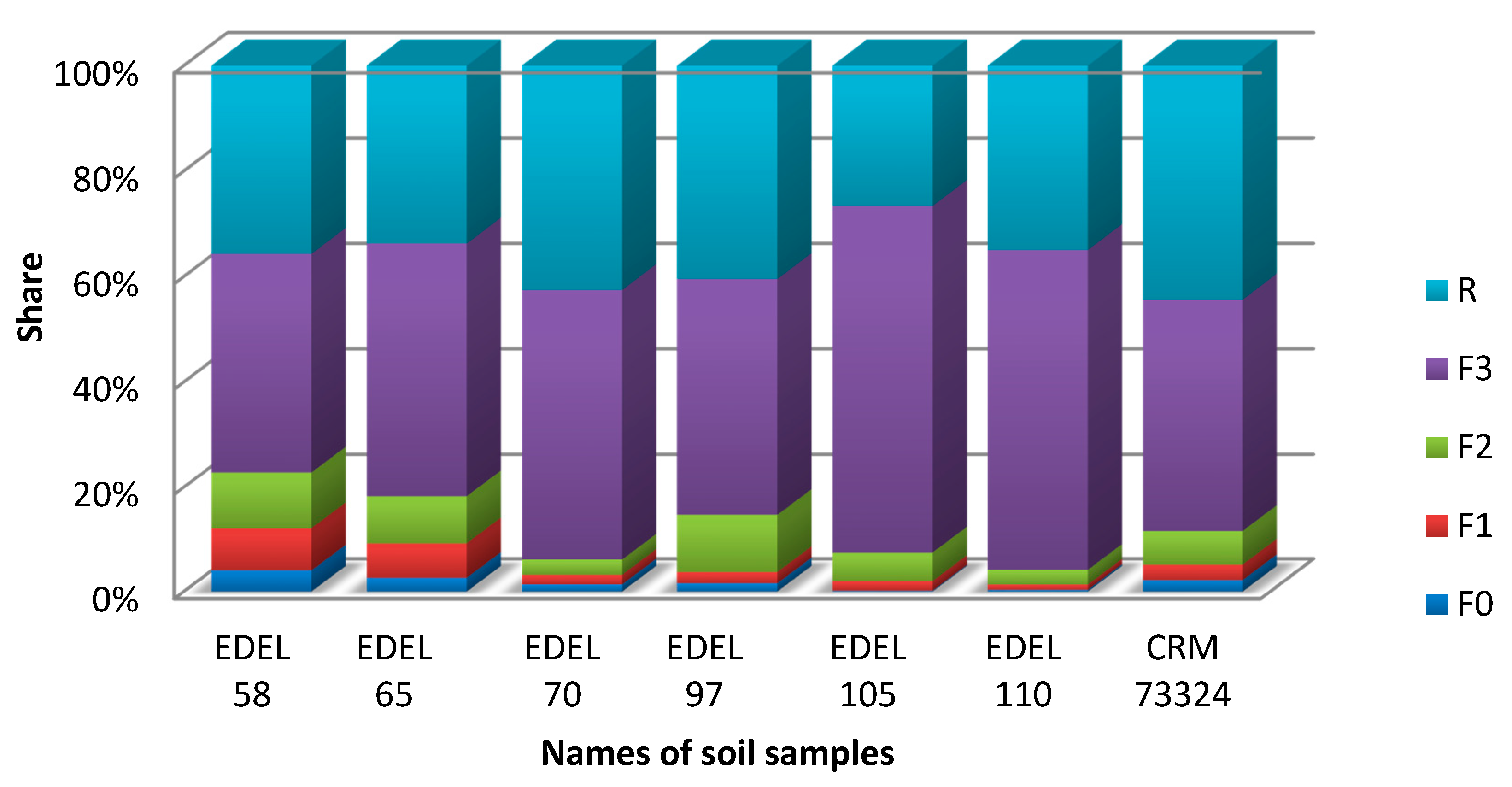
2.3. Tellurium Speciation
3. Material and Methods
3.1. Sampling Area and Soil Preparation
3.2. Apparatus
3.3. Reagents
3.4. Sequential Chemical Extraction of Soil
3.5. Determination of the Total Tellurium and Tellurium Species Content
3.5.1. Quality Control of Total Tellurium Concentration
3.5.2. Soil Extraction for IC-ICP-MS Analysis
4. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Nuss, P.; Blengini, G.A. Towards better monitoring of technology critical elements in Europe: Coupling of natural and anthropogenic cycles. Sci. Total Environ. 2018, 613–614, 569–578. [Google Scholar] [CrossRef]
- Işıldar, A.; Rene, E.R.; van Hullebusch, E.D.; Lens, P.N.L. Electronic waste as a secondary source of critical metals: Management and recovery technologies. Resour. Conserv. Recycl. 2018, 135, 296–312. [Google Scholar] [CrossRef]
- Filella, M.; Reimann, C.; Biver, M.; Rodushkin, I.; Rodushkina, K. Tellurium in the environment: Current knowledge and identification of gaps. Environ. Chem. 2019, 16, 215–228. [Google Scholar] [CrossRef]
- Qin, H.B.; Takeichi, Y.; Nitani, H.; Terada, Y.; Takahashi, Y. Tellurium Distribution and Speciation in Contaminated Soils from Abandoned Mine Tailings: Comparison with Selenium. Environ. Sci. Technol. 2017, 51, 6027–6035. [Google Scholar] [CrossRef] [PubMed]
- Belzile, N.; Chen, Y.W. Tellurium in the environment: A critical review focused on natural waters, soils, sediments and airborne particles. Appl. Geochem. 2015, 63, 83–92. [Google Scholar] [CrossRef]
- Govindaraju, K. 1994 compilation of working values and sample description for 383 geostandards. Geostand. Geoanal. Res. 1994, 18, 1–158. [Google Scholar] [CrossRef]
- Ferri, T.; Rossi, S.; Sangiorgio, P. Simultaneous determination of the speciation of selenium and tellurium in geological matrices by use of an iron(III)-modified chelating resin and cathodic stripping voltammetry. Anal. Chim. Acta 1998, 361, 113–123. [Google Scholar] [CrossRef]
- Harada, T.; Takahashi, Y. Origin of the difference in the distribution behavior of tellurium and selenium in a soil-water system. Geochim. Cosmochim. Acta 2009, 72, 1281–1294. [Google Scholar] [CrossRef]
- Kabata-Pendias, A.; Pendias, H. Biogeochemistry of Trace Elements [Biogeochemia pierwiastków śladowych]; PWN: Warszawa, Poland, 1999. (In Polish) [Google Scholar]
- Julander, A.; Lundgren, L.; Skare, L.T.; Grander, M.; Palm, B. Formal recycling of e-waste leads to increased exposure to toxic metals: An occupational exposure study from Sweden. Environ. Int. 2014, 73, 243–251. [Google Scholar] [CrossRef]
- Zeng, X.; Wang, F.; Li, J.; Gong, R. A simplified method to evaluate the recycling potential of e-waste. J. Clean. Prod. 2017, 168, 1518–1524. [Google Scholar] [CrossRef]
- Kumar, A.; Holuszko, M.; Espinosa, D.C.R. E-Waste: An overview on generation, collection, legislation and recycling practices. Resour. Conserv. Recycl. 2017, 122, 32–42. [Google Scholar] [CrossRef]
- Templeton, D.M.; Fujishiro, H. Terminology of elemental speciation—An IUPAC perspective. Coord. Chem. Rev. 2017, 352, 424–431. [Google Scholar] [CrossRef]
- Jabłońska-Czapla, M.; Szopa, S.; Grygoyć, K.; Łyko, A.; Michalski, R. Development and validation of HPLC–ICP-MS method for the determination inorganic Cr, As and Sb speciation forms and its application for Pławniowice reservoir (Poland) water and bottom sediments variability study. Talanta 2014, 120, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Ou, X.; Wang, C.; He, M.; Chen, B.; Hu, B. Online simultaneous speciation of ultra-trace inorganic antimony and tellurium in environmental water by polymer monolithic capillary microextraction combined with inductively coupled plasma mass spectrometry. Spectrochim. Acta B 2020, 168, 105854. [Google Scholar] [CrossRef]
- Yu, C.; Cai, Q.; Guo, Z.X.; Yang, Z.; Khoo, S.B. Simultaneous speciation of inorganic selenium and tellurium by inductively coupled plasma mass spectrometry following selective solid-phase extraction separation. J. Anal. At. Spectrom. 2004, 19, 410–413. [Google Scholar] [CrossRef]
- Yu, C.; Cai, Q.; Guo, Z.X.; Yang, Z.; Khoo, S.B. Speciation analysis of tellurium by solid-phase extraction in the presence of ammonium pyrrolidine dithiocarbamate and inductively coupled plasma mass spectrometry. Anal. Bioanal. Chem. 2003, 376, 236–242. [Google Scholar] [CrossRef]
- Wojcieszek, J.; Szpunar, J.; Lobinski, R. Speciation of technologically critical elements in the environment using chromatography with element and molecule specific detection. Trends Anal. Chem. 2018, 104, 42–53. [Google Scholar] [CrossRef]
- Garcia-Figueroa, A.; Lavilla, I.; Bendichio, C. Speciation of CdTe quantum dots and Te(IV) following oxidative degradation induced by iodide and headspace single-drop microextraction combined with graphite furnace atomic absorption spectrometry. Spectrochim. Acta B 2019, 158, 105631–105640. [Google Scholar] [CrossRef]
- Najafi, N.M.; Tavakoli, H.; Alizadeh, R.; Seidi, S. Speciation and determination of ultra trace amounts of inorganic tellurium in environmental water samples by dispersive liquid-liquid microextraction and electrothermal atomic absorption spectrometry. Anal. Chim. Acta 2010, 670, 18–23. [Google Scholar] [CrossRef]
- Viñas, P.; López, I.; Merino-Meroño, B.; Hernández-Córdoba, M. Ion chromatography-hydride generation-atomic fluorescence spectrometry speciation of tellurium. Appl. Organomet. Chem. 2005, 19, 930–934. [Google Scholar] [CrossRef]
- Chen, Y.W.; Alzahrani, A.; Deng, T.L.; Belzile, N. Valence properties of tellurium in different chemical systems and its determination in refractory environmental samples using hydride generation-atomic fluorescence spectroscopy. Anal. Chim. Acta 2016, 905, 42–50. [Google Scholar] [CrossRef]
- Chen, M.; Wu, L.; Yi, X.; Yang, K.; Xie, H. Tellurium speciation in a bioleaching solution by hydride generation atomic fluorescence spectrometry. Anal. Methods 2017, 9, 3061–3066. [Google Scholar] [CrossRef]
- Matusiewicz, H.; Krawczyk, M. Determination of tellurium by hydride generation with in situ trapping flame atomic absorption spectrometry. Spectrochim. Acta B 2007, 62, 309–316. [Google Scholar] [CrossRef]
- Pedro, J.; Stripekis, J.; Bonivardi, A.L.; Tudino, M. Surface studies on graphite furnace platforms covered with Pd, Rh and Ir as modifiers in graphite furnace atomic ab- sorption spectrometry of tellurium. Spectrochim. Acta B 2015, 107, 152–158. [Google Scholar] [CrossRef]
- Tan, Q.; Pan, Y.; Liu, L.; Shu, S.; Liu, Y. Determination of ultratrace tellurium in water by hydride generation atomic absorption spectrometry using online separation and pre-concentration with nano-TiO2 microcolumn. Microchem. J. 2019, 144, 495–499. [Google Scholar] [CrossRef]
- Cornelis, R.; Caruso, J.; Crews, H.; Heumann, K. Handbook of Elemental Speciation: Techniques and methodology; John Wiley&Sons Ltd.: Chichester, UK, 2003. [Google Scholar]
- Kuo, C.Y.; Jiang, S.J. Determination of selenium and tellurium compounds in biological samples by ion chromatography dynamic reaction cell inductively coupled plasma mass spectrometry. J. Chromatogr. A. 2008, 1181, 60–66. [Google Scholar] [CrossRef]
- Anan, Y.; Yoshida, M.; Hasegawa, S.; Katai, R.; Tokumoto, M.; Ouerdane, L.; Łobiński, R.; Ogra, Y. Speciation and identification of tellurium-containing metabolites in garlic, Allium sativum. Metallomics 2013, 9, 1215–1224. [Google Scholar] [CrossRef]
- Bullock, L.A.; Perez, M.; Armstrong, J.G.; Parnell, J.; Still, J.; Feldmann, J. Selenium and tellurium resources in Kisgruva Proterozoic volcanogenic massive sulphide deposit (Norway). Ore Geol. Rev. 2018, 99, 411–424. [Google Scholar] [CrossRef]
- Liu, Y.; He, M.; Chen, B.; Hu, G. Simultaneous speciation of inorganic arsenic, selenium and tellurium in environmental water samples by dispersive liquid liquid microextraction combined with electrothermal vaporization inductively coupled plasma mass spectrometry. Talanta 2015, 142, 213–220. [Google Scholar] [CrossRef]
- Meng, P.; Xiong, T.; Wu, Y.; Hu, Y.; Wang, H.; Pang, Y.; Jiang, S.; Han, S.; Huang, P.A. Novel strategy to evaluate the degradation of quantum dots: Identification and quantification of CdTe quantum dots and corresponding ionic species by CZE-ICP- MS. Chem. Comm. 2018, 54, 5342–5345. [Google Scholar] [CrossRef]
- Su, C.K.; Cheng, T.Y.; Sun, Y.C. Selective chemical vaporization of exogenous tellurium for characterizing the time-dependent biodistribution and dissolution of quantum dots in living rats. J. Anal. At. Spectrom. 2015, 30, 426–434. [Google Scholar] [CrossRef]
- Jabłońska-Czapla, M.; Grygoyć, K. Speciation and Fractionation of Less-Studied Technology-Critical Elements (Nb, Ta, Ga, In, Ge, Tl, Te): A Review. Pol. J. Environ. Stud. 2021, 30, 1477–1486. [Google Scholar] [CrossRef]
- Zheng, J.; Iijima, A.; Furuta, N. Complexation effect of antimony compounds with citric acid and its application to the speciation of antimony(III) and antimony(V) using HPLC-ICP-MS. J. Anal. At. Spectrom. 2001, 16, 812–818. [Google Scholar] [CrossRef]
- Salvia, M.V.; Cren-Olivé, C.; Vulliet, E. Statistical evaluation of the influence of soil properties on recoveries and matrix effects during the analysis of pharmaceutical compounds and steroids by quick, easy, cheap, effective, rugged and safe extraction followed by liquid chromatography–tandem mass spectrometry. J. Chromatogr. A. 2013, 1315, 53–60. [Google Scholar] [PubMed]
- Hu, Z.; Gao, S.; Günther, D.; Hu, S.; Liu, X.; Yuan, Y. Direct Determination of Tellurium in Geological Samples by Inductively Coupled Plasma Mass Spectrometry Using Ethanol as a Matrix Modifier. Appl. Spectrosc. 2006, 60, 781–785. [Google Scholar] [CrossRef]
- Casiot, C.; Alonso, M.C.B.; Boisson, J.; Donard, O.F.X.; Potin-Gautier, M. Simultaneous speciation of arsenic, selenium, antimony and tellurium species in waters and soil extracts by capillary electrophoresis and UV detection. Analyst 2008, 123, 2887–2893. [Google Scholar] [CrossRef]
- Geilfus, C.M. Chloride in soil: From nutrient to soil pollutant. Environ. Exp. Bot. 2019, 157, 299–309. [Google Scholar] [CrossRef]
- Missen, O.P.; Mills, S.J.; Etschmann, B.; Reith, F.; Shuster, J.; Smith, D.J.; Brugger, J. Love is in the Earth: A review of tellurium (bio)geochemistry in surface environments. Earth-Sci. Rev. 2020, 204, 103150. [Google Scholar] [CrossRef]
- Yu, M.Z.; Chen, X.G.; Garbe-Schonberg, D.; Ye, Y.; Chen, C.T.A. Volatile chalcophile elements in native sulfur from a submarine hydrothermal; system at Kueishantao, Offshore NE Taiwan. Minerals 2019, 9, 245. [Google Scholar] [CrossRef]
- Beceva, K.; Stafilov, T.; Sajn, R.; Tanaselia, C.; Makreski, P. Distribution of chemical elements in soil and stream sediments in the area abandoned Sb-As-Tl Allchar mine, Republic of Macedonia. Environ. Res. 2014, 133, 77–89. [Google Scholar] [CrossRef]
- Perkins, W.T. Extreme selenium and tellurium contamination in soils—An eighty year-old industrial legacy surrounding a Ni refinery in the Swansea Valley. Sci. Total Environ. 2011, 412–413, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Zheng, J.; Tagami, K.; Uchida, S. Rapid and sensitive determination of tellurium in soil and plant samples by sector-field inductively coupled plasma mass spectrometry. Talanta 2013, 116, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Liu, Y.; He, M.; Bai, M.; Xu, W.; Zhao, C. Ore prospecting model and targets for the Dashuigou tellurium deposit, Sichuan Province, China. Acta Geochim. 2018, 37, 578–591. [Google Scholar] [CrossRef]
- Stępniewska, Z.; Ostrowski, J.; Stępniewski, W.; Gliński, J. Classification of redox resistance of Polish arable soils and their spatial characteristics. [Klasyfikacja odporności oksydoredukcyjnej gleb ornych polski i ich przestrzenna charakterystyka]. Water-Environ-Rural. Area 2004, 12, 125–133. (In Polish) [Google Scholar]
- Gliński, J.; Duliban, J. Redox potential in soils [Potencjał oksydoredukcyjny w glebach]. Agrophys. Prob. 1972, 3, 1–43. (In Polish) [Google Scholar]
- Stępniewska, Z. Oxidation reduction properties of Polish mineral soils [Właściwości oksydoredukcyjne gleb mineralnych Polski]. Agrophys. Prob. 1988, 56, 103. (In Polish) [Google Scholar]
- Hayes, S.M.; Ramos, N.A. Surficial geochemistry and bioaccessibility of tellurium in semiarid mine tailigs. Environ. Chem. 2019, 16, 251–265. [Google Scholar] [CrossRef]
- ISO 10390: 2005 Soil Quality-Determination of pH. International Organization for Standardization. Publication Date 2005-02. Available online: https://www.iso.org/standard/40879.html (accessed on 20 April 2021).
- Jeske, A.; Gworek, B. Review of methods for determining the bioavailability and mobility of heavy metals in soils [Przegląd metod oznaczania biodostępności i mobilności metali ciężkich w glebach]. Environ. Protect. Nat. Res. 2011, 49, 209–218. (In Polish) [Google Scholar]
- Filella, M.; Rodushkin, I. A concise guide for the determination of less-studied technology-critical elements (Nb, Ta, Ga, In, Ge, Te) by inductively coupled plasma mass spectrometry in environmental samples. Spectrochim. Acta Part B 2018, 141, 80–84. [Google Scholar] [CrossRef]
| Parameter | Value |
|---|---|
| Tellurium | |
| Separation column | Hamilton PRP-X100 4.6 mm × 150 mm, 5 µm |
| Temperature | 30 |
| Mobile phase | 10 mM Na2EDTA, 6 mM KHP pH = 4.29–4.32 |
| Elution program | 4 min |
| Retention time of Te species [min] | Te(VI)-1.46 Te(IV)-3.18 |
| Flow rate during analysis [mL/min] | 1.2 |
| Flow rate during the rinsing [mL/min] | 1.2 |
| Volume of sample [μL] | 200 |
| Sample | Total Tellurium Concentration after Digestion [mg/kg] | Total Tellurium Concentration after Extraction [mg/kg] | Extraction Efficiency [%] | Te(VI) Concentration [mg/kg] | Te(IV) Concentration [mg/kg] | Sum Te(IV) and Te(VI) | pH Value in H2O | pH Value in KCl | Eh [mV] |
|---|---|---|---|---|---|---|---|---|---|
| CRM | 0.42 | 0.043 | 10 | 0.004 | 0.036 | 0.040 | 3.46 | 3.12 | 362.2 |
| 52 Edel | 0.021 | 0.010 | 47 | 0.005 | 0.009 | 0.013 | 3.54 | 3.22 | 387.8 |
| 56 Edel | 0.095 | 0.022 | 24 | 0.010 | 0.006 | 0.016 | 3.82 | 3.54 | 395.2 |
| 58 Edel | 0.083 | 0.025 | 30 | 0.013 | 0.009 | 0.022 | 3.83 | 3.49 | 349.3 |
| 61 Edel | 0.083 | 0.028 | 33 | 0.011 | 0.011 | 0.022 | 3.46 | 3.12 | 412.8 |
| 65 Edel | 0.108 | 0.029 | 27 | 0.020 | 0.007 | 0.027 | 3.85 | 3.59 | 384.5 |
| 70 Edel | 0.129 | 0.016 | 12 | 0.003 | 0.009 | 0.013 | 3.43 | 3.14 | 425.5 |
| 80 Edel | 0.143 | 0.016 | 11 | 0.003 | 0.005 | 0.008 | 3.92 | 3.62 | 384.7 |
| 82 Edel | 0.092 | 0.014 | 16 | 0.009 | 0.008 | 0.017 | 4.09 | 3.58 | 365.8 |
| 97 Edel | 0.105 | 0.022 | 21 | 0.017 | 0.005 | 0.022 | 4.33 | 3.68 | 376.2 |
| 105 Edel | 0.166 | 0.017 | 10 | 0.004 | 0.013 | 0.017 | 3.62 | 3.46 | 375.8 |
| 107 Edel | 0.114 | 0.013 | 11 | 0.002 | 0.005 | 0.007 | 3.45 | 3.15 | 382.4 |
| 110 Edel | 0.124 | 0.011 | 9 | 0.005 | 0.011 | 0.015 | 3.35 | 3.11 | 398.5 |
| 112 Edel | 0.114 | 0.014 | 12 | 0.002 | 0.009 | 0.011 | 3.69 | 3.39 | 396.5 |
| 116 Edel | 0.090 | 0.019 | 21 | < LOD | 0.017 | 0.017 | 3.42 | 3.14 | 411.1 |
| Sample No. | N Latitude | E Longitude |
|---|---|---|
| 52 Edel | 50,204670 | 19,042550 |
| 56 Edel | 50,207130 | 19,046020 |
| 58 Edel | 50,206900 | 19,040510 |
| 61 Edel | 50,206040 | 19,037950 |
| 65 Edel | 50,205070 | 19,032090 |
| 70 Edel | 50,210880 | 19,041430 |
| 80 Edel | 50,209070 | 19,045770 |
| 82 Edel | 50,202090 | 19,046790 |
| 97 Edel | 50,210190 | 19,030230 |
| 105 Edel | 50,208170 | 19,035690 |
| 107 Edel | 50,208350 | 19,038310 |
| 110 Edel | 50,201210 | 19,029580 |
| 112 Edel | 50,203690 | 19,035750 |
| 116 Edel | 50,208510 | 19,054950 |
| Extraction Rate | Form | Extracting Reagent |
|---|---|---|
| 0 | Dissolved in pore water | distilled water |
| F1 | Ion exchange and carbonate | 20 mL 0.11 M CH3COOH 16 h, continuous mixing |
| F2 | Oxide | 20 mL 0.1 M NH2OH.HCl (pH 2, supplied HNO3) 16 h, continuous mixing |
| F3 | Organic | (A) 10 mL H2O2 30% pH 2 2 h water bath 85 ± 2 °C 10 mL H2O2 8.8 M pH = 2 2 h water bath 85 ± 2 °C (B) 25 mL 1 M NH4OAc pH 2 16 h continuous mixing (pH 2, supplied HNO3) |
| R | Residual | 6 mL HCl, 2 mL HNO3, 3 mL HF microwave digestion Anton Paar Microwave 3000, power 1400 W, time 45 min. |
| Parameter | Value |
|---|---|
| ICP-MS | |
| RF power [W] | 1125 |
| Plasma gas flow [L/min] | 15 |
| Nebulizer gas flow [L/min] | 0.76–0.82 |
| Auxiliary gas flow [L/min] | 1.15–1.16 |
| Nebulizer type | Cross flow |
| Plasma torch | Quartz |
| Scanning mode | Peak hopping |
| Dwell time [ms] | 100 |
| Sweeps/reading | 20 |
| Number of replicates | 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grygoyć, K.; Jabłońska-Czapla, M. Development of a Tellurium Speciation Study Using IC-ICP-MS on Soil Samples Taken from an Area Associated with the Storage, Processing, and Recovery of Electrowaste. Molecules 2021, 26, 2651. https://doi.org/10.3390/molecules26092651
Grygoyć K, Jabłońska-Czapla M. Development of a Tellurium Speciation Study Using IC-ICP-MS on Soil Samples Taken from an Area Associated with the Storage, Processing, and Recovery of Electrowaste. Molecules. 2021; 26(9):2651. https://doi.org/10.3390/molecules26092651
Chicago/Turabian StyleGrygoyć, Katarzyna, and Magdalena Jabłońska-Czapla. 2021. "Development of a Tellurium Speciation Study Using IC-ICP-MS on Soil Samples Taken from an Area Associated with the Storage, Processing, and Recovery of Electrowaste" Molecules 26, no. 9: 2651. https://doi.org/10.3390/molecules26092651
APA StyleGrygoyć, K., & Jabłońska-Czapla, M. (2021). Development of a Tellurium Speciation Study Using IC-ICP-MS on Soil Samples Taken from an Area Associated with the Storage, Processing, and Recovery of Electrowaste. Molecules, 26(9), 2651. https://doi.org/10.3390/molecules26092651








