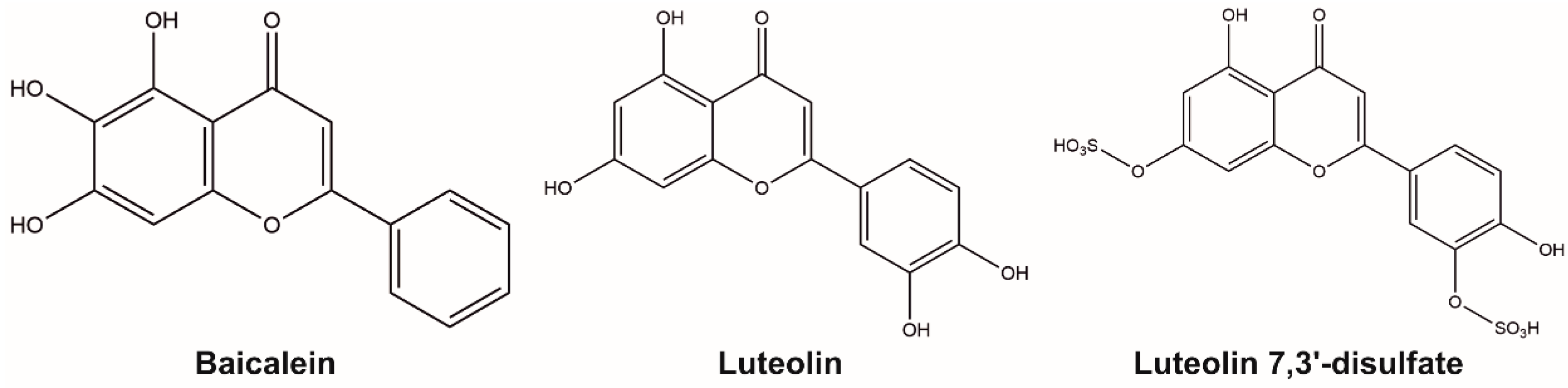
Human Lanosterol 14-Alpha Demethylase (CYP51A1) Is a Putative Target for Natural Flavonoid Luteolin 7,3′-Disulfate
Abstract
1. Introduction
2. Results
2.1. Surface Plasmon Resonance
2.2. Spectral Titration Analysis
2.3. Enzyme Activity Assay
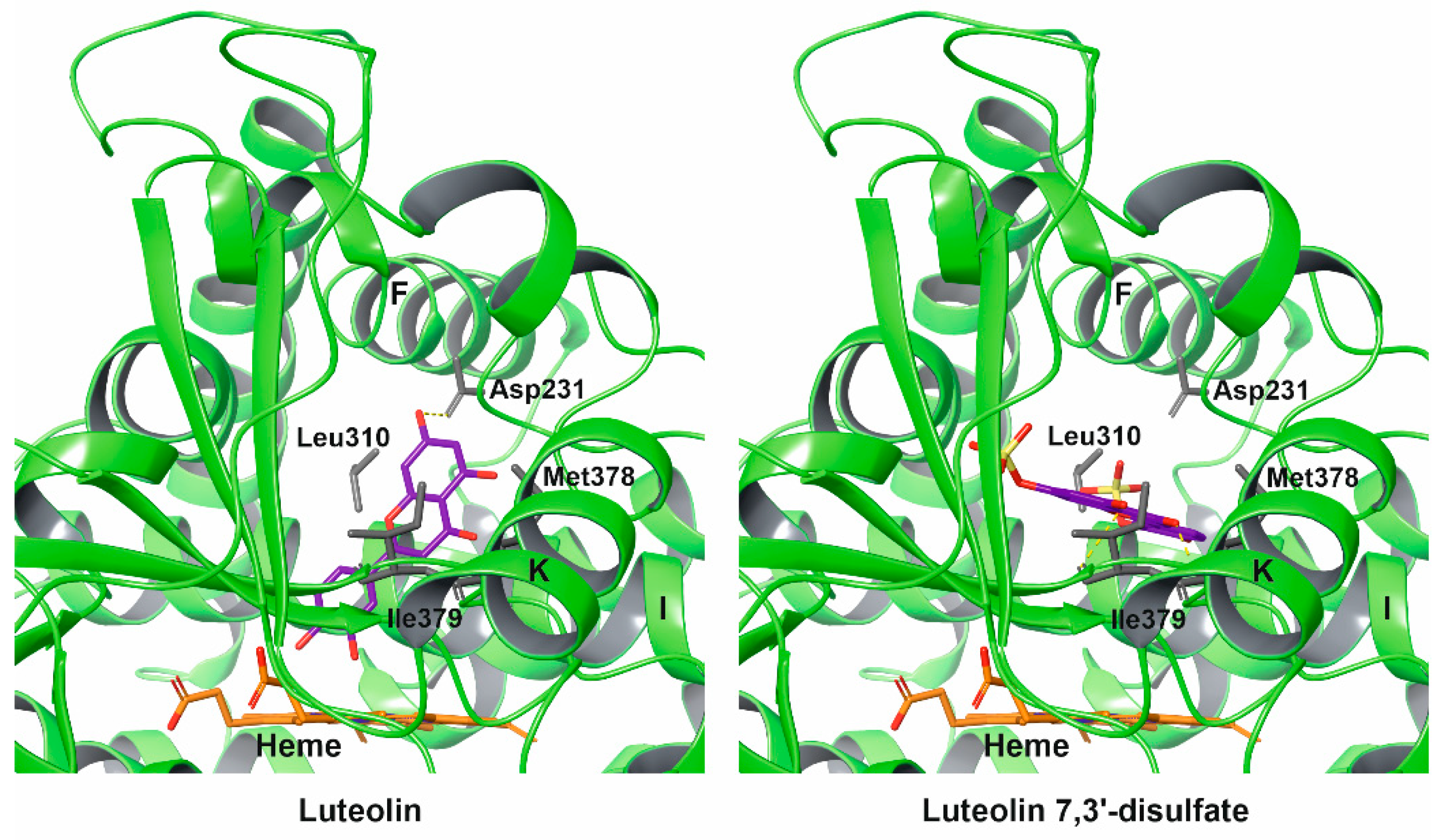
2.4. Molecular Docking
3. Discussion
4. Materials and Methods
4.1. Samples
4.2. Surface Plasmon Resonance
- (1)
- 1:1 (Langmuir) binding [57]: , where Kd—equilibrium dissociation constant, koff—dissociation rate constant, kon—association rate constant.
- (2)
- Two-state (conformational change) binding [58]: , where Kd—equilibrium dissociation constant, koff1—dissociation rate constant, kon1—association rate constant, kon2—forward rate constant for CP ↔ CP* transition, koff2—backward rate constant for CP ↔ CP* transition.
4.3. Spectral Titration Analysis
4.4. Enzyme Assay
4.5. Molecular Docking
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Porter, J.A.; Young, K.E.; Beachy, P.A. Cholesterol Modification of Hedgehog Signaling Proteins in Animal Development. Science 1996, 274, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Tang, J.-J.; Peng, C.; Wang, Y.; Fu, L.; Qiu, Z.-P.; Xiong, Y.; Yang, L.-F.; Cui, H.-W.; He, X.-L.; et al. Cholesterol Modification of Smoothened Is Required for Hedgehog Signaling. Mol. Cell 2017, 66, 154–162.e10. [Google Scholar] [CrossRef] [PubMed]
- McNeill, A.M.; Rosamond, W.D.; Girman, C.J.; Golden, S.H.; Schmidt, M.I.; East, H.E.; Ballantyne, C.M.; Heiss, G. The Metabolic Syndrome and 11-Year Risk of Incident Cardiovascular Disease in the Atherosclerosis Risk in Communities Study. Diabetes Care 2005, 28, 385–390. [Google Scholar] [CrossRef]
- Kumarakulasingham, M.; Rooney, P.H.; Dundas, S.R.; Telfer, C.; Melvin, W.T.; Curran, S.; Murray, G.I. Cytochrome P450 Profile of Colorectal Cancer: Identification of Markers of Prognosis. Clin. Cancer Res. 2005, 11, 3758–3765. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, Y. Recent Progress in the CYP51 Research Focusing on Its Unique Evolutionary and Functional Characteristics as a Diversozyme P450. Front. Biosci. 2005, 10, 1546–1557. [Google Scholar] [CrossRef] [PubMed]
- Murray, G.I.; Patimalla, S.; Stewart, K.N.; Miller, I.D.; Heys, S.D. Profiling the Expression of Cytochrome P450 in Breast Cancer. Histopathology 2010, 57, 202–211. [Google Scholar] [CrossRef]
- Sun, M.; Qiu, J.; Zhai, H.; Wang, Y.; Ma, P.; Li, M.; Chen, B. Prognostic Implications of Novel Gene Signatures in Gastric Cancer Microenvironment. Med. Sci. Monit. 2020, 26, e924604-1–e924604-17. [Google Scholar] [CrossRef] [PubMed]
- Gabitova, L.; Restifo, D.; Gorin, A.; Manocha, K.; Handorf, E.; Yang, D.-H.; Cai, K.Q.; Klein-Szanto, A.J.; Cunningham, D.; Kratz, L.E.; et al. Endogenous Sterol Metabolites Regulate Growth of EGFR/KRAS-Dependent Tumors via LXR. Cell Rep. 2015, 12, 1927–1938. [Google Scholar] [CrossRef]
- Salmina, K.; Huna, A.; Kalejs, M.; Pjanova, D.; Scherthan, H.; Cragg, M.S.; Erenpreisa, J. The Cancer Aneuploidy Paradox: In the Light of Evolution. Genes 2019, 10, 83. [Google Scholar] [CrossRef]
- Keber, R.; Motaln, H.; Wagner, K.D.; Debeljak, N.; Rassoulzadegan, M.; Ačimovič, J.; Rozman, D.; Horvat, S. Mouse Knockout of the Cholesterogenic Cytochrome P450 Lanosterol 14alpha-Demethylase (Cyp51) Resembles Antley-Bixler Syndrome. J. Biol. Chem. 2011, 286, 29086–29097. [Google Scholar] [CrossRef]
- Martínez-Botas, J.; Ferruelo, A.J.; Suárez, Y.; Gómez-Coronado, D.; Lasunción, M.A. Induction of Apoptosis in P53-Null HL-60 Cells by Inhibition of Lanosterol 14-Alpha Demethylase. Biochimie 1998, 80, 887–894. [Google Scholar] [CrossRef]
- Noh, K.; Nepal, M.R.; Jeong, K.S.; Kim, S.-A.; Um, Y.J.; Seo, C.S.; Kang, M.J.; Park, P.-H.; Kang, W.; Jeong, H.G.; et al. Effects of Baicalin on Oral Pharmacokinetics of Caffeine in Rats. Biomol. Ther. 2015, 23, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Kwara, A.; Greenblatt, D.J. Metabolic Interactions between Acetaminophen (Paracetamol) and Two Flavonoids, Luteolin and Quercetin, through in-Vitro Inhibition Studies. J. Pharm. Pharmacol. 2017, 69, 1762–1772. [Google Scholar] [CrossRef] [PubMed]
- Fliszár-Nyúl, E.; Mohos, V.; Csepregi, R.; Mladěnka, P.; Poór, M. Inhibitory Effects of Polyphenols and Their Colonic Metabolites on CYP2D6 Enzyme Using Two Different Substrates. Biomed. Pharmacother. 2020, 131, 110732. [Google Scholar] [CrossRef] [PubMed]
- Da, X.; Nishiyama, Y.; Tie, D.; Hein, K.Z.; Yamamoto, O.; Morita, E. Antifungal Activity and Mechanism of Action of Ou-Gon (Scutellaria Root Extract) Components against Pathogenic Fungi. Sci. Rep. 2019, 9, 1683. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Cao, Y.Y.; Dai, B.D.; Sun, X.R.; Zhu, Z.Y.; Cao, Y.B.; Wang, Y.; Gao, P.H.; Jiang, Y.Y. In Vitro Synergism of Fluconazole and Baicalein against Clinical Isolates of Candida Albicans Resistant to Fluconazole. Biol. Pharm. Bull. 2008, 31, 2234–2236. [Google Scholar] [CrossRef]
- Joung, D.-K.; Lee, Y.-S.; Han, S.-H.; Lee, S.-W.; Cha, S.-W.; Mun, S.-H.; Kong, R.; Kang, O.-H.; Song, H.-J.; Shin, D.-W.; et al. Potentiating Activity of Luteolin on Membrane Permeabilizing Agent and ATPase Inhibitor against Methicillin-Resistant Staphylococcus Aureus. Asian Pac. J. Trop. Med. 2016, 9, 19–22. [Google Scholar] [CrossRef]
- Popov, A.M.; Osipov, A.N.; Korepanova, E.A.; Krivoshapko, O.N.; Artyukov, A.A.; Klimovich, A.A. A Study of the Antioxidant and Membranotropic Activities of Luteolin Using Different Model Systems. BIOPHYSICS 2016, 61, 843–850. [Google Scholar] [CrossRef]
- Ravishankar, D.; Watson, K.A.; Boateng, S.Y.; Green, R.J.; Greco, F.; Osborn, H.M.I. Exploring Quercetin and Luteolin Derivatives as Antiangiogenic Agents. Eur. J. Med. Chem. 2015, 97, 259–274. [Google Scholar] [CrossRef]
- Gao, Y.; Snyder, S.A.; Smith, J.N.; Chen, Y.C. Anticancer Properties of Baicalein: A Review. Med. Chem. Res. 2016, 25, 1515–1523. [Google Scholar] [CrossRef]
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeem, M.; Shariati, M.A.; Khan, I.A.; Imran, A.; Orhan, I.E.; Rizwan, M.; Atif, M.; et al. Luteolin, a Flavonoid, as an Anticancer Agent: A Review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef] [PubMed]
- Tuli, H.S.; Aggarwal, V.; Kaur, J.; Aggarwal, D.; Parashar, G.; Parashar, N.C.; Tuorkey, M.; Kaur, G.; Savla, R.; Sak, K.; et al. Baicalein: A Metabolite with Promising Antineoplastic Activity. Life Sci. 2020, 259, 118183. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Li, P.; Liu, S.; Liu, Q.; Li, Y.; Sun, Y.; He, C.; Xiao, P. Traditional Uses, Ten-Years Research Progress on Phytochemistry and Pharmacology, and Clinical Studies of the Genus Scutellaria. J. Ethnopharmacol. 2021, 265, 113198. [Google Scholar] [CrossRef] [PubMed]
- Nik Salleh, N.N.H.; Othman, F.A.; Kamarudin, N.A.; Tan, S.C. The Biological Activities and Therapeutic Potentials of Baicalein Extracted from Oroxylum Indicum: A Systematic Review. Molecules 2020, 25, 5677. [Google Scholar] [CrossRef]
- Farooqi, A.A.; Butt, G.; El-Zahaby, S.A.; Attar, R.; Sabitaliyevich, U.Y.; Jovic, J.J.; Tang, K.-F.; Naureen, H.; Xu, B. Luteolin Mediated Targeting of Protein Network and MicroRNAs in Different Cancers: Focus on JAK-STAT, NOTCH, MTOR and TRAIL-Mediated Signaling Pathways. Pharmacol. Res. 2020, 160, 105188. [Google Scholar] [CrossRef] [PubMed]
- Tuorkey, M.J. Molecular Targets of Luteolin in Cancer. Eur. J. Cancer Prev. 2016, 25, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Barreca, D.; Mandalari, G.; Calderaro, A.; Smeriglio, A.; Trombetta, D.; Felice, M.R.; Gattuso, G. Citrus Flavones: An Update on Sources, Biological Functions, and Health Promoting Properties. Plants 2020, 9, 288. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.-P.; Li, S.; Chen, Y.-M.; Li, H.-B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients 2016, 8. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Occurrence of Sulphated Flavones and Caffeic Acid Esters in Members of the Fluviales. Biochem. Syst. Ecol. 1976, 4, 37–41. [Google Scholar] [CrossRef]
- Popov, A.M.; Krivoshapko, O.N.; Artyukov, A.A. Comparative estimation of pharmacological activities of luteolin and luteolin 7,3′-disulphate at modelling of different pathologies. Russ. J. Biopharm. 2011, 3, 27–33. [Google Scholar]
- Klimovich, A.A.; Popov, A.M.; Styshova, O.N.; Artyukov, A.A.; Tsybulsky, A.V. A Comparative Evaluation of the Actions of Different Secondary Metabolites of Marine Hydrobionts on the Redox Status of Tumor and Immune Cells. Biophysics 2018, 63, 763–768. [Google Scholar] [CrossRef]
- Kwak, J.Y.; Seok, J.K.; Suh, H.-J.; Choi, Y.-H.; Hong, S.S.; Kim, D.S.; Boo, Y.C. Antimelanogenic Effects of Luteolin 7-Sulfate Isolated from Phyllospadix Iwatensis Makino. Br. J. Dermatol. 2016, 175, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Popov, A.M.; Krivoshapko, O.N. Protective Effects of Polar Lipids and Redox-Active Compounds from Marine Organisms at Modeling of Hyperlipidemia and Diabetes. J. Biomed. Sci. Eng. 2013, 6, 543–550. [Google Scholar] [CrossRef]
- Popov, A.M.; Krivoshapko, O.N.; Klimovich, A.A.; Artyukov, A.A. Biological activity and mechanisms of therapeutic action of rosmarinic acid, luteolin and its sulphated derivatives. Biomed. Khim. 2016, 62, 22–30. [Google Scholar] [CrossRef]
- Kaluzhskiy, L.A.; Gnedenko, O.V.; Gilep, A.A.; Strushkevich, N.V.; Shkel, T.V.; Chernovetsky, M.A.; Ivanov, A.S.; Lisitsa, A.V.; Usanov, A.S.; Stonik, V.A.; et al. Screening of Human Cytochrome P450(51) (CYP51A1) Inhibitors: Structural Lanosterol Analogues of Plant and Animal Origin. Biochem. (Moscow) Suppl. B Biomed. Chem. 2014, 8, 349–360. [Google Scholar] [CrossRef]
- Shimada, T.; Tanaka, K.; Takenaka, S.; Foroozesh, M.K.; Murayama, N.; Yamazaki, H.; Guengerich, F.P.; Komori, M. Reverse Type I Binding Spectra of Human Cytochrome P450 1B1 Induced by Flavonoid, Stilbene, Pyrene, Naphthalene, Phenanthrene, and Biphenyl Derivatives That Inhibit Catalytic Activity: A Structure-Function Relationship Study. Chem. Res. Toxicol. 2009, 22, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
- Nitahara, Y.; Kishimoto, K.; Yabusaki, Y.; Gotoh, O.; Yoshida, Y.; Horiuchi, T.; Aoyama, Y. The Amino Acid Residues Affecting the Activity and Azole Susceptibility of Rat CYP51 (Sterol 14-Demethylase P450). J. Biochem. 2001, 129, 761–768. [Google Scholar] [CrossRef]
- Strushkevich, N.; Usanov, S.A.; Park, H.-W. Structural Basis of Human CYP51 Inhibition by Antifungal Azoles. J. Mol. Biol. 2010, 397, 1067–1078. [Google Scholar] [CrossRef]
- Yousef, M.I.; Omar, S.A.M.; El-Guendi, M.I.; Abdelmegid, L.A. Potential Protective Effects of Quercetin and Curcumin on Paracetamol-Induced Histological Changes, Oxidative Stress, Impaired Liver and Kidney Functions and Haematotoxicity in Rat. Food Chem. Toxicol. 2010, 48, 3246–3261. [Google Scholar] [CrossRef]
- Cho, Y.-A.; Choi, J.-S.; Burm, J.-P. Effects of the Antioxidant Baicalein on the Pharmacokinetics of Nimodipine in Rats: A Possible Role of P-Glycoprotein and CYP3A4 Inhibition by Baicalein. Pharmacol. Rep. 2011, 63, 1066–1073. [Google Scholar] [CrossRef]
- Si, D.; Wang, Y.; Zhou, Y.-H.; Guo, Y.; Wang, J.; Zhou, H.; Li, Z.-S.; Fawcett, J.P. Mechanism of CYP2C9 Inhibition by Flavones and Flavonols. Drug Metab. Dispos. 2009, 37, 629–634. [Google Scholar] [CrossRef]
- Quintieri, L.; Palatini, P.; Nassi, A.; Ruzza, P.; Floreani, M. Flavonoids Diosmetin and Luteolin Inhibit Midazolam Metabolism by Human Liver Microsomes and Recombinant CYP 3A4 and CYP3A5 Enzymes. Biochem. Pharmacol. 2008, 75, 1426–1437. [Google Scholar] [CrossRef] [PubMed]
- Ohno, S.; Shinoda, S.; Toyoshima, S.; Nakazawa, H.; Makino, T.; Nakajin, S. Effects of Flavonoid Phytochemicals on Cortisol Production and on Activities of Steroidogenic Enzymes in Human Adrenocortical H295R Cells. J. Steroid Biochem. Mol. Biol. 2002, 80, 355–363. [Google Scholar] [CrossRef]
- Hodek, P.; Trefil, P.; Stiborová, M. Flavonoids-Potent and Versatile Biologically Active Compounds Interacting with Cytochromes P450. Chem.-Biol. Interact. 2002, 139, 1–21. [Google Scholar] [CrossRef]
- Howes, L.G.; James, M.J.; Florin, T.; Walker, C. Nv-52: A Novel Thromboxane Synthase Inhibitor for the Treatment of Inflammatory Bowel Disease. Expert Opin. Investig. Drugs 2007, 16, 1255–1266. [Google Scholar] [CrossRef] [PubMed]
- Swami, S.; Krishnan, A.V.; Peehl, D.M.; Feldman, D. Genistein Potentiates the Growth Inhibitory Effects of 1,25-Dihydroxyvitamin D3 in DU145 Human Prostate Cancer Cells: Role of the Direct Inhibition of CYP24 Enzyme Activity. Mol. Cell. Endocrinol. 2005, 241, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Thilakarathna, S.H.; Rupasinghe, H.P.V. Flavonoid Bioavailability and Attempts for Bioavailability Enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef]
- Williamson, G.; Manach, C. Bioavailability and Bioefficacy of Polyphenols in Humans. II. Review of 93 Intervention Studies. Am. J. Clin. Nutr. 2005, 81, 243S–255S. [Google Scholar] [CrossRef]
- Mohos, V.; Fliszár-Nyúl, E.; Ungvári, O.; Kuffa, K.; Needs, P.W.; Kroon, P.A.; Telbisz, Á.; Özvegy-Laczka, C.; Poór, M. Inhibitory Effects of Quercetin and Its Main Methyl, Sulfate, and Glucuronic Acid Conjugates on Cytochrome P450 Enzymes, and on OATP, BCRP and MRP2 Transporters. Nutrients 2020, 12, 2306. [Google Scholar] [CrossRef]
- Mohos, V.; Fliszár-Nyúl, E.; Ungvári, O.; Bakos, É.; Kuffa, K.; Bencsik, T.; Zsidó, B.Z.; Hetényi, C.; Telbisz, Á.; Özvegy-Laczka, C.; et al. Effects of Chrysin and Its Major Conjugated Metabolites Chrysin-7-Sulfate and Chrysin-7-Glucuronide on Cytochrome P450 Enzymes and on OATP, P-Gp, BCRP, and MRP2 Transporters. Drug Metab. Dispos. 2020, 48, 1064–1073. [Google Scholar] [CrossRef]
- Monostory, K.; Pascussi, J.-M.; Szabó, P.; Temesvári, M.; Köhalmy, K.; Acimovic, J.; Kocjan, D.; Kuzman, D.; Wilzewski, B.; Bernhardt, R.; et al. Drug Interaction Potential of 2-((3,4-Dichlorophenethyl)(Propyl)Amino)-1-(Pyridin-3-Yl)Ethanol (LK-935), the Novel Nonstatin-Type Cholesterol-Lowering Agent. Drug Metab. Dispos. 2009, 37, 375–385. [Google Scholar] [CrossRef]
- Masamrekh, R.; Kuzikov, A.; Veselovsky, A.; Toropygin, I.; Shkel, T.; Strushkevich, N.; Gilep, A.; Usanov, S.; Archakov, A.; Shumyantseva, V. Interaction of 17α-Hydroxylase, 17(20)-Lyase (CYP17A1) Inhibitors—Abiraterone and Galeterone—with Human Sterol 14α-Demethylase (CYP51A1). J. Inorg. Biochem. 2018, 186, 24–33. [Google Scholar] [CrossRef]
- Korošec, T.; Ačimovič, J.; Seliškar, M.; Kocjan, D.; Tacer, K.F.; Rozman, D.; Urleb, U. Novel Cholesterol Biosynthesis Inhibitors Targeting Human Lanosterol 14α-Demethylase (CYP51). Bioorg. Med. Chem. 2008, 16, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Artjukov, A.A.; Kozlovskaja, E.P.; Krivoshapko, O.N.; Krylova, N.V.; Leonova, G.N.; Popov, A.M. Medication, Possessing Antioxidant, Cardioprotective, Antidiabetic, Anti-Inflammatory, Hepatoprotective, Antitumoral and Antiviral Action 2011. Russian Federation Patent № RU2432959C1, 10 November 2011. [Google Scholar]
- Kim, J.H.; Cho, Y.H.; Park, S.M.; Lee, K.E.; Lee, J.J.; Lee, B.C.; Pyo, H.B.; Song, K.S.; Park, H.D.; Yun, Y.P. Antioxidants and Inhibitor of Matrix Metalloproteinase-1 Expression from Leaves of Zostera Marina L. Arch. Pharm. Res. 2004, 27, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Artjukov, A.A.; Glazunov, V.P.; Kochergina, T.J.; Kozlovskaja, E.P.; Kupera, E.V.; Makhan’kov, V.V.; Novikov, V.L.; Rutskova, T.A. Method of Obtaining Luteolyn 7,3′-Disulphate 2011. Russian Federation Patent № RU2432960C1, 10 November 2011. [Google Scholar]
- Müller, K.M.; Arndt, K.M.; Plückthun, A. Model and Simulation of Multivalent Binding to Fixed Ligands. Anal. Biochem 1998, 261, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Lipschultz, C.A.; Li, Y.; Smith-Gill, S. Experimental Design for Analysis of Complex Kinetics Using Surface Plasmon Resonance. Methods 2000, 20, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Cheeseright, T.; Mackey, M.; Rose, S.; Vinter, A. Molecular Field Extrema as Descriptors of Biological Activity: Definition and Validation. J. Chem. Inf. Model. 2006, 46, 665–676, . [Google Scholar] [CrossRef]
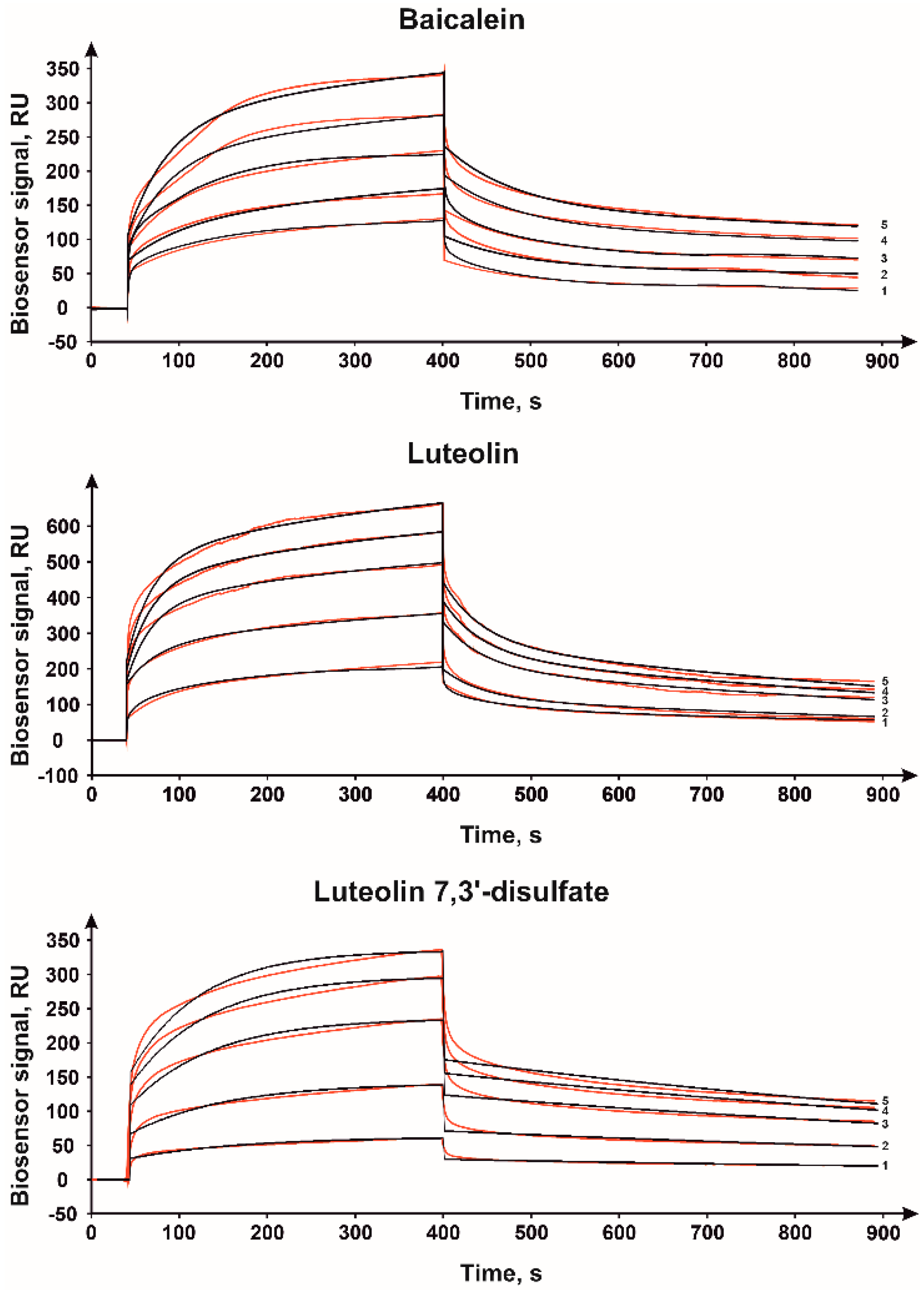

| Compound | kon | koff | Kd, μM | Evaluation Model |
|---|---|---|---|---|
| lanosterol | kon (1/Ms) = 41.4 ± 5.0 | koff (1/s × 10−4) = 1.0 ± 0.2 | 2.4 | Langmuir 1:1 |
| baicalein | kon1 (1/Ms) = 146 ± 20 | koff1 (1/s × 10−4) = 100 ± 20 | 12.5 | Two state reaction |
| kon2 (1/s × 10−4) = 27 ± 3 | koff2 (1/s × 10−4) = 6 ± 1 | |||
| luteolin | kon1 (1/Ms) = 282 ± 40 | koff1 (1/s × 10−4) = 190 ± 30 | 20.0 | Two state reaction |
| kon2 (1/s × 10−4) = 33 ± 4 | koff2 (1/s × 10−4) = 14 ± 2 | |||
| luteolin 7,3′-disulfate | kon (1/Ms) = 294.0 ± 32.3 | koff (1/s × 10−4) = 8.4 ± 2.0 | 2.9 | Langmuir 1:1 |
| Compound | Relative Activity, % |
|---|---|
| No compound | 100.0 |
| Baicalein (25 μM) | 89.4 |
| Luteolin (25 μM) | 92.6 |
| Luteolin 7,3′-disulfate (25 μM) | 49.9 |
| Ketoconazole (5 μM) | 5.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaluzhskiy, L.; Ershov, P.; Yablokov, E.; Shkel, T.; Grabovec, I.; Mezentsev, Y.; Gnedenko, O.; Usanov, S.; Shabunya, P.; Fatykhava, S.; et al. Human Lanosterol 14-Alpha Demethylase (CYP51A1) Is a Putative Target for Natural Flavonoid Luteolin 7,3′-Disulfate. Molecules 2021, 26, 2237. https://doi.org/10.3390/molecules26082237
Kaluzhskiy L, Ershov P, Yablokov E, Shkel T, Grabovec I, Mezentsev Y, Gnedenko O, Usanov S, Shabunya P, Fatykhava S, et al. Human Lanosterol 14-Alpha Demethylase (CYP51A1) Is a Putative Target for Natural Flavonoid Luteolin 7,3′-Disulfate. Molecules. 2021; 26(8):2237. https://doi.org/10.3390/molecules26082237
Chicago/Turabian StyleKaluzhskiy, Leonid, Pavel Ershov, Evgeniy Yablokov, Tatsiana Shkel, Irina Grabovec, Yuri Mezentsev, Oksana Gnedenko, Sergey Usanov, Polina Shabunya, Sviatlana Fatykhava, and et al. 2021. "Human Lanosterol 14-Alpha Demethylase (CYP51A1) Is a Putative Target for Natural Flavonoid Luteolin 7,3′-Disulfate" Molecules 26, no. 8: 2237. https://doi.org/10.3390/molecules26082237
APA StyleKaluzhskiy, L., Ershov, P., Yablokov, E., Shkel, T., Grabovec, I., Mezentsev, Y., Gnedenko, O., Usanov, S., Shabunya, P., Fatykhava, S., Popov, A., Artyukov, A., Styshova, O., Gilep, A., Strushkevich, N., & Ivanov, A. (2021). Human Lanosterol 14-Alpha Demethylase (CYP51A1) Is a Putative Target for Natural Flavonoid Luteolin 7,3′-Disulfate. Molecules, 26(8), 2237. https://doi.org/10.3390/molecules26082237






