Head-Group Acylation of Chloroplast Membrane Lipids
Abstract
1. Introduction
2. Discovery of Head Group-Acylated Plastidic Lipids
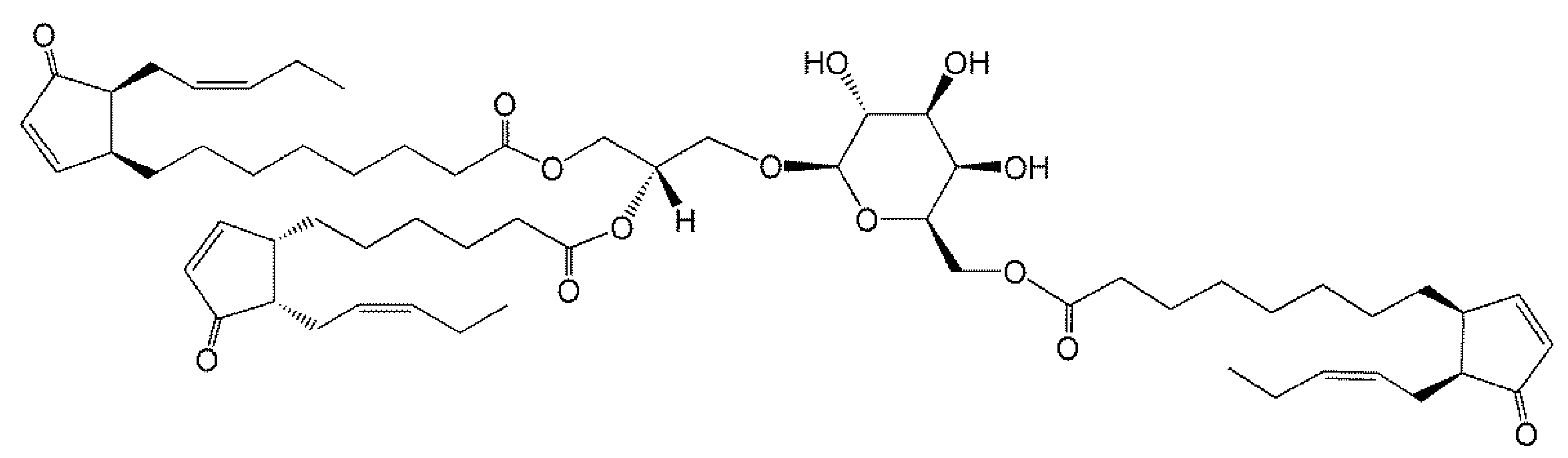
3. Acylated Monogalactosyldiacylglycerol (AcMGDG) Is Ubiquitous in Photosynthetic Tissue
4. Lipidomics Is Increasing Our Knowledge of Head Group-Acylated Lipid Molecular Species
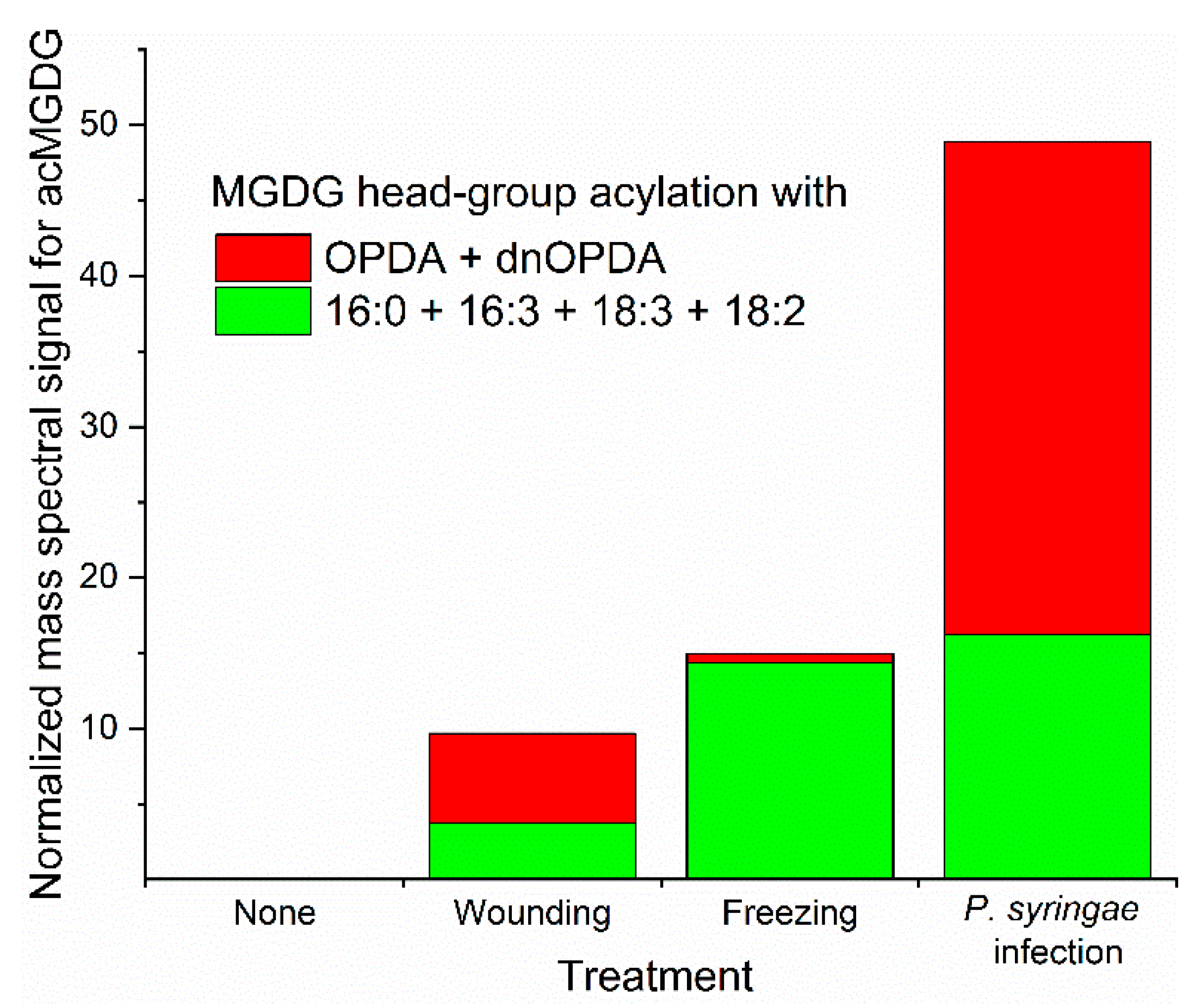
5. Varied Patterns of Acylated Chloroplast Lipids Are Induced by Environmental Stresses
6. AcMGDG Is Produced by Transacylation
7. The Enzymatic Activity Catalyzing Head-Group Acylation of Chloroplast Lipids
8. Sub-Cellular Localization of Acylated Galactolipid-Associated Phospholipase 1 (AGAP1) and Head Group-Acylated Chloroplast Lipids
9. Little Is Known about the Physical Properties of Head Group-Acylated Chloroplasts Lipids
10. The Biological Functions of AGAP1 and Chloroplast Lipid Head-Group Acylation Remain Elusive
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Uemura, M.; Steponkus, P.L. Effect of cold acclimation on the lipid composition of the inner and outer membrane of the chloroplast envelope isolated from rye leaves. Plant Physiol. 1997, 114, 1493–1500. [Google Scholar] [CrossRef] [PubMed]
- Hölzl, G.; Dörmann, P. Chloroplast lipids and their biosynthesis. Annu. Rev. Plant Biol. 2019, 70, 51–81. [Google Scholar] [CrossRef]
- Moellering, E.R.; Muthan, B.; Benning, C. Freezing tolerance in plants requires lipid remodeling at the outer chloroplast membrane. Science 2010, 330, 226–228. [Google Scholar] [CrossRef] [PubMed]
- Moellering, E.R.; Benning, C. Galactoglycerolipid metabolism under stress: A time for remodeling. Trends Plant Sci. 2010, 16, 98–107. [Google Scholar] [CrossRef]
- Vu, H.S.; Roston, R.; Shiva, S.; Hur, M.; Wurtele, E.S.; Wang, X.; Shah, J.; Welti, R. Modifications of membrane lipids in response to wounding of Arabidopsis thaliana leaves. Plant Signal. Behav. 2015, 10, e1056422. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, Y.; Otsuki, H.; Narisawa, T.; Kobayashi, M.; Sawai, S.; Kamide, Y.; Kusano, M.; Aoki, T.; Hirai, M.Y.; Saito, K. A new class of plant lipid is essential for protection against phosphorus depletion. Nat. Commun. 2013, 4, 1510. [Google Scholar] [CrossRef]
- Andersson, M.X.; Hamberg, M.; Kourtchenko, O.; Brunnström, Å.; McPhail, K.L.; Gerwick, W.H.; Göbel, C.; Feussner, I.; Ellerström, M. Oxylipin profiling of the hypersensitive response in Arabidopsis thaliana: Formation of a novel oxo-phytodienoic acid-containing galactolipid, Arabidopside E. J. Biol. Chem. 2006, 281, 31528–31537. [Google Scholar]
- Kourtchenko, O.; Andersson, M.X.; Hamberg, M.; Brunnström, Å.; Göbel, C.; McPhail, K.L.; Gerwick, W.H.; Feussner, I.; Ellerström, M. Oxo-phytodienoic acid-containing galactolipids in Arabidopsis: Jasmonate signaling dependence. Plant Physiol. 2007, 145, 1658–1669. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.K.; Johansson, O.N.; Fahlberg, P.; Steinhart, F.; Gustavsson, M.B.; Ellerström, M.; Andersson, M.X. Formation of Oxidized phosphatidylinositol and 12-oxo-phytodienoic acid containing acylated phosphatidylglycerol during the hypersensitive response in Arabidopsis. Phytochemistry 2014, 101, 65–75. [Google Scholar] [CrossRef]
- Vu, H.S.; Roth, M.R.; Tamura, P.; Samarakoon, T.; Shiva, S.; Honey, S.; Lowe, K.; Schmelz, E.A.; Williams, T.D.; Welti, R. Head-group acylation of monogalactosyldiacylglycerol is a common stress response, and the acyl-galactose acyl composition varies with the plant species and applied stress. Physiol. Plant. 2014, 150, 517–528. [Google Scholar] [CrossRef]
- Heinz, E. Acylgalactosyldiglycerides from leaf homogenates. Biochim. Biophys. Acta 1967, 144, 321–332. [Google Scholar] [CrossRef]
- Myhre, D.V. Glycolipids of Soft Wheat Flour. I. Isolation and characterization of 1-O-(6-O-acyl-β-D-galactopyranosyl)-2,3-di-O-acyl-D-glyceritols and phytosteryl 6-O-acyl-Β-D-glucopyranosides. Can. J. Chem. 1968, 46, 3071–3077. [Google Scholar] [CrossRef]
- Heinz, E.; Tulloch, A.P. Reinvestigation of the structure of acyl galactose diglyceride from spinach leaves. Hoppe Seylers Z. Physiol. Chem. 1969, 350, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Heinz, E.; Rullkötter, J.; Budzikiewicz, H. Acyl digalactosyl diglyceride from leaf homogenates. Hoppe Seylers Z. Physiol. Chem. 1974, 355, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Holmbäck, J.; Karlsson, A.A.; Arnoldsson, K.C. Characterization of N-acylphosphatidylethanolamine and acylphosphatidylglycerol in oats. Lipids 2001, 36, 153–165. [Google Scholar] [CrossRef]
- Nilsson, A.K.; Johansson, O.N.; Fahlberg, P.; Kommuri, M.; Topel, M.; Bodin, L.J.; Sikora, P.; Modarres, M.; Ekengren, S.; Nguyen, C.T.; et al. Acylated monogalactosyl diacylglycerol: Prevalence in the plant kingdom and identification of an enzyme catalyzing galactolipid head group acylation in Arabidopsis thaliana. Plant J. 2015, 84, 1152–1166. [Google Scholar] [CrossRef] [PubMed]
- Murakami, N.; Shirahashi, H.; Nagatsu, A.; Sakakibara, J. Studies on glycolipids. VI. New acyl-distributed glyceroglycolipids from the nitrogen-fixing cyanobacterium Anabaena flos-aquae f. flos-aquae. Chem. Pharm. Bull. 1993, 41, 1177–1179. [Google Scholar] [CrossRef][Green Version]
- Kim, Y.H.; Choi, J.S.; Hong, J.; Yoo, J.S.; Kim, M.S. Identification of acylated glycoglycerolipids from a cyanobacterium, Synechocystis sp., by tandem mass spectrometry. Lipids 1999, 34, 847–853. [Google Scholar] [CrossRef]
- Al-Fadhli, A.; Wahidulla, S.; D’Souza, L. Glycolipids from the red alga Chondria armata (Kutz.) Okamura. Glycobiology 2006, 16, 902–915. [Google Scholar] [CrossRef]
- Vu, H.S.; Shiva, S.; Roth, M.R.; Tamura, P.; Zheng, L.; Li, M.; Sarowar, S.; Honey, S.; McEllhiney, D.; Hinkes, P.; et al. Lipid changes after leaf wounding in Arabidopsis thaliana: Expanded lipidomic data form the basis for lipid co-occurrence analysis. Plant J. 2014, 80, 728–743. [Google Scholar] [CrossRef] [PubMed]
- Liedvogel, B.; Kleinig, H. Lipid metabolism in chromoplast membranes from the daffodil: Glycosylation and acylation. Planta 1977, 133, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Vu, H.S.; Tamura, P.; Galeva, N.A.; Chaturvedi, R.; Roth, M.R.; Williams, T.D.; Wang, X.; Shah, J.; Welti, R. Direct Infusion Mass spectrometry of oxylipin-containing Arabidopsis membrane lipids reveals varied patterns in different stress responses. Plant Physiol. 2012, 158, 324–339. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Schütz, A.-L.; Galano, J.-M.; Herrfurth, C.; Feussner, K.; Durand, T.; Brodhun, F.; Feussner, I. The alphabet of galactolipids in Arabidopsis thaliana. Front. Plant Sci. 2011, 2, 95. [Google Scholar] [CrossRef]
- Genva, M.; Andersson, M.X.; Fauconnier, M.-L. Simple liquid chromatography-electrospray ionization ion trap mass spectrometry method for the quantification of galacto-oxylipin Arabidopsides in plant samples. Sci. Rep. 2020, 10, 11957. [Google Scholar] [CrossRef]
- Hisamatsu, Y.; Goto, N.; Hasegawa, K.; Shigemori, H. Arabidopsides A and B, Two new oxylipins from Arabidopsis thaliana. Tetrahedron Lett. 2003, 44, 5553–5556. [Google Scholar] [CrossRef]
- Hisamatsu, Y.; Goto, N.; Sekiguchi, M.; Hasegawa, K.; Shigemori, H. Oxylipins Arabidopsides C and D from Arabidopsis thaliana. J. Nat. Prod. 2005, 68, 600–633. [Google Scholar] [CrossRef] [PubMed]
- Buseman, C.M.; Tamura, P.; Sparks, A.A.; Baughman, E.J.; Maatta, S.; Zhao, J.; Roth, M.R.; Esch, S.W.; Shah, J.; Williams, T.D.; et al. Wounding stimulates the accumulation of glycerolipids containing oxophytodienoic acid and dinor-oxophytodienoic acid in Arabidopsis leaves. Plant Physiol. 2006, 142, 28–39. [Google Scholar] [CrossRef]
- Shiva, S.; Samarakoon, T.; Lowe, K.A.; Roach, C.; Vu, H.S.; Colter, M.; Porras, H.; Hwang, C.; Roth, M.R.; Tamura, P.; et al. Leaf lipid alterations in response to heat stress of Arabidopsis thaliana. Plants 2020, 9, 845. [Google Scholar] [CrossRef]
- Mongrand, S.; Bessoule, J.-J.; Cabantous, F.; Cassagne, C. The C16:3\C18:3 fatty acid balance in photosynthetic tissues from 468 plant species. Phytochemistry 1998, 49, 1049–1064. [Google Scholar] [CrossRef]
- Hartley, S.E.; Eschen, R.; Horwood, J.M.; Gange, A.C.; Hill, E.M. Infection by a foliar endophyte elicits novel arabidopside-based plant defence reactions in its host, Cirsium arvense. New Phytol. 2015, 205, 816–827. [Google Scholar] [CrossRef]
- Ohashi, T.; Ito, Y.; Okada, M.; Sakagami, Y. Isolation and stomatal opening activity of two oxylipins from Ipomoea tricolor. Bioorganic Med. Chem. Lett. 2005, 15, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Zábranská, M.; Vrkoslav, V.; Sobotníková, J.; Cvačka, J. Analysis of plant galactolipids by reversed-phase high-performance liquid chromatography/mass spectrometry with accurate mass measurement. Chem. Phys. Lipids 2012, 165, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.P.; Unger, M.; Guender, L.; Fekete, A.; Mueller, M.J. Phospholipid:diacylglycerol acyltransferase-mediated triacylglyerol synthesis augments basal thermotolerance. Plant Physiol. 2017, 175, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Heinz, E. On the enzymatic formation of acylgalactosyl diglyceride. Biochim. Biophys. Acta 1967, 144, 333–343. [Google Scholar] [CrossRef]
- Heinz, E.; Dieler, H.P.; Rullkötter, J. Enzymatic acylation of steryl glycoside. Z. Pflanzenphysiol. 1975, 75, 78–87. [Google Scholar] [CrossRef]
- Heinz, E. Some properties of the acyl galactose diglyceride-forming enzyme from leaves. Z. Pflanzenphysiol. 1973, 69, 359–376. [Google Scholar] [CrossRef]
- Heinz, E.; Betrams, M.; Joyard, J.; Douce, R. Demonstration of an acyltransferase activity in chloroplast envelopes. Z. Pflanzenphysiol. 1978, 87, 325–331. [Google Scholar] [CrossRef]
- Heemskerk, J.W.M.; Wintermans, J.F.G.M.; Joyard, J.; Block, M.A.; Dorne, A.; Douce, R. Localization of galactolipid: Galactolipid galactosyltransferase and acyltransferase in outer envelope membrane of spinach chloroplasts. Biochim. Biophys. Acta 1986, 877, 281–289. [Google Scholar] [CrossRef]
- Nilsson, A.K.; Fahlberg, P.; Ellerström, M.; Andersson, M.X. Oxo-phytodienoic acid (OPDA) is formed on fatty acids esterified to galactolipids after tissue disruption in Arabidopsis thaliana. FEBS Lett. 2012, 586, 2483–2487. [Google Scholar] [CrossRef]
- Froehlich, J.E.; Itoh, A.; Howe, G.A. Tomato allene oxide synthase and fatty acid hydroperoxide lyase, two cytochrome P450s involved in oxylipin metabolism, are targeted to different membranes of chloroplast envelope. Plant Physiol. 2001, 125, 306–317. [Google Scholar] [CrossRef]
- Farmaki, T.; Sanmartín, M.; Jiménez, P.; Paneque, M.; Sanz, C.; Vancanneyt, G.; León, J.; Sánchez-Serrano, J.J. Differential distribution of the lipoxygenase pathway enzymes within potato chloroplasts. J. Exp. Bot. 2007, 58, 555–568. [Google Scholar] [CrossRef]
- Bräutigam, A.; Hoffmann-Benning, S.; Weber, A.P.M. Comparative proteomics of chloroplast envelopes from C3 and C4 plants reveals specific adaptations of the plastid envelope to C4 photosynthesis and candidate proteins required for maintaining C4 metabolite fluxes. Plant Physiol. 2008, 148, 568–579. [Google Scholar] [CrossRef]
- Schaller, F.; Zerbe, P.; Reinbothe, S.; Reinbothe, C.; Hofmann, E.; Pollmann, S. The allene oxide cyclase family of Arabidopsis thaliana-Localization and cyclization. FEBS J. 2008, 275, 2428–2441. [Google Scholar] [CrossRef] [PubMed]
- Peltier, J.-B.; Ytterberg, A.J.; Sun, Q.; van Wijk, K.J. New functions of the thylakoid membrane proteome of Arabidopsis thaliana revealed by a simple, fast, and versatile fractionation strategy. J. Biol. Chem. 2004, 279, 49367–49383. [Google Scholar] [CrossRef]
- Vidi, P.-A.; Kanwischer, M.; Baginsky, S.; Austin, J.R.; Csucs, G.; Dörmann, P.; Kessler, F.; Bréhélin, C. Tocopherol cyclase (VTE1) localization and vitamin E accumulation in chloroplast plastoglobule lipoprotein particles. J. Biol. Chem. 2006, 281, 11225–11234. [Google Scholar] [CrossRef] [PubMed]
- Swamy, M.J.; Ramakrishnan, M.; Angerstein, B.; Marsh, D. Spin-label electron spin resonance studies on the mode of anchoring and vertical location of the N-acyl chain in N-acylphosphatidylethanolamines. Biochemistry 2000, 39, 12476–12484. [Google Scholar] [CrossRef]
- Swamy, M.J.; Tarafdar, P.K.; Kamlekar, R.K. Structure, phase behaviour and membrane interactions of N-acylethanolamines and N-acylphosphatidylethanolamines. Chem. Phys. Lipids 2010, 163, 266–279. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, Y.; Zoong Lwe, Z.S.; Wickramasinghe, P.A.D.B.V.; Welti, R. Head-Group Acylation of Chloroplast Membrane Lipids. Molecules 2021, 26, 1273. https://doi.org/10.3390/molecules26051273
Song Y, Zoong Lwe ZS, Wickramasinghe PADBV, Welti R. Head-Group Acylation of Chloroplast Membrane Lipids. Molecules. 2021; 26(5):1273. https://doi.org/10.3390/molecules26051273
Chicago/Turabian StyleSong, Yu, Zolian S. Zoong Lwe, Pallikonda Arachchige Dona Bashanee Vinusha Wickramasinghe, and Ruth Welti. 2021. "Head-Group Acylation of Chloroplast Membrane Lipids" Molecules 26, no. 5: 1273. https://doi.org/10.3390/molecules26051273
APA StyleSong, Y., Zoong Lwe, Z. S., Wickramasinghe, P. A. D. B. V., & Welti, R. (2021). Head-Group Acylation of Chloroplast Membrane Lipids. Molecules, 26(5), 1273. https://doi.org/10.3390/molecules26051273







